Investigation on Cycling and Calendar Aging Processes of 3.4 Ah Lithium-Sulfur Pouch Cells
Abstract
:1. Introduction
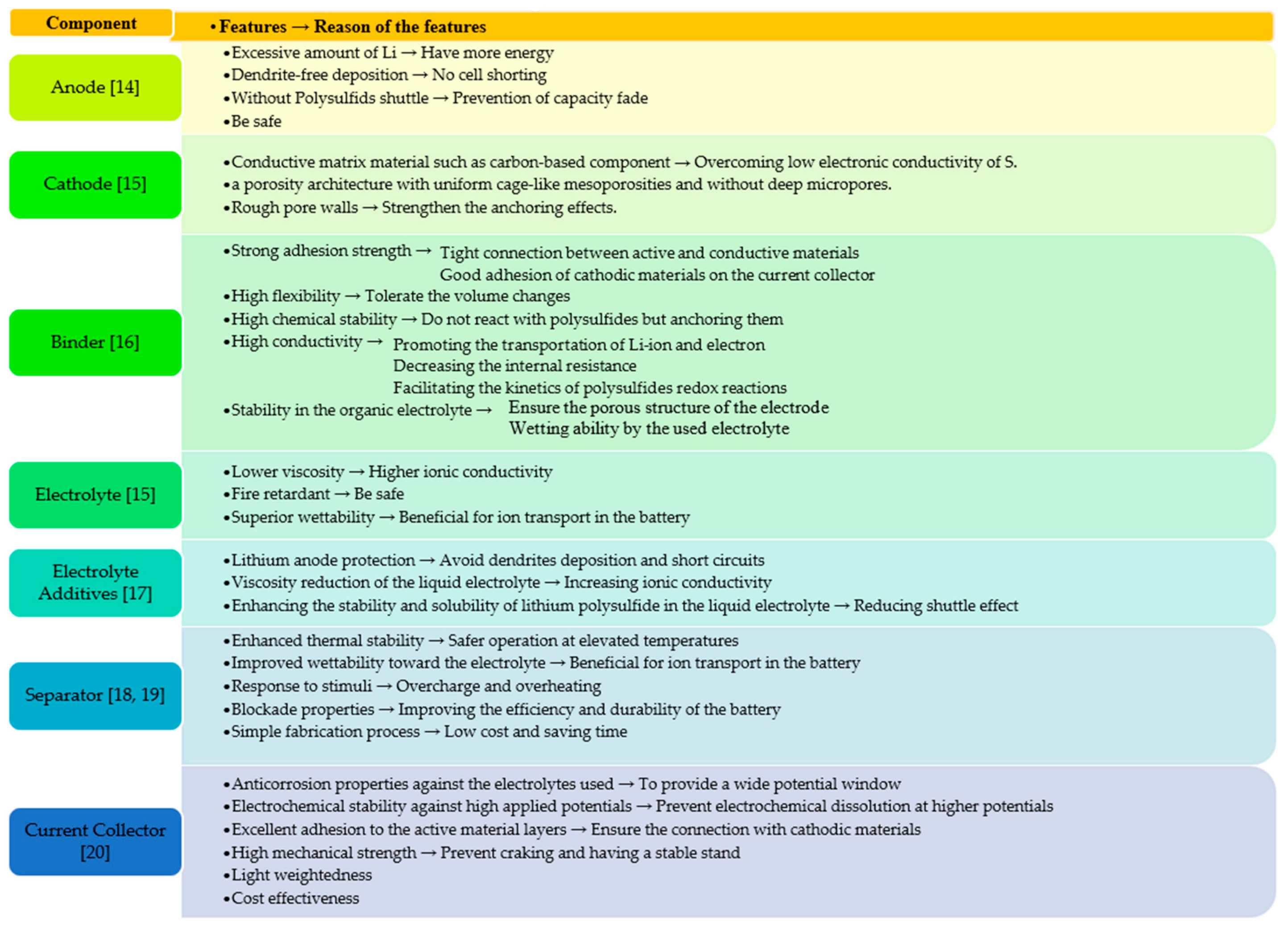
- Expansion of sulfur (∼80% change upon lithiation) → causes pulverization of cathodic materials and loss of electrical connection with the conductive substrate [13];
- 5.
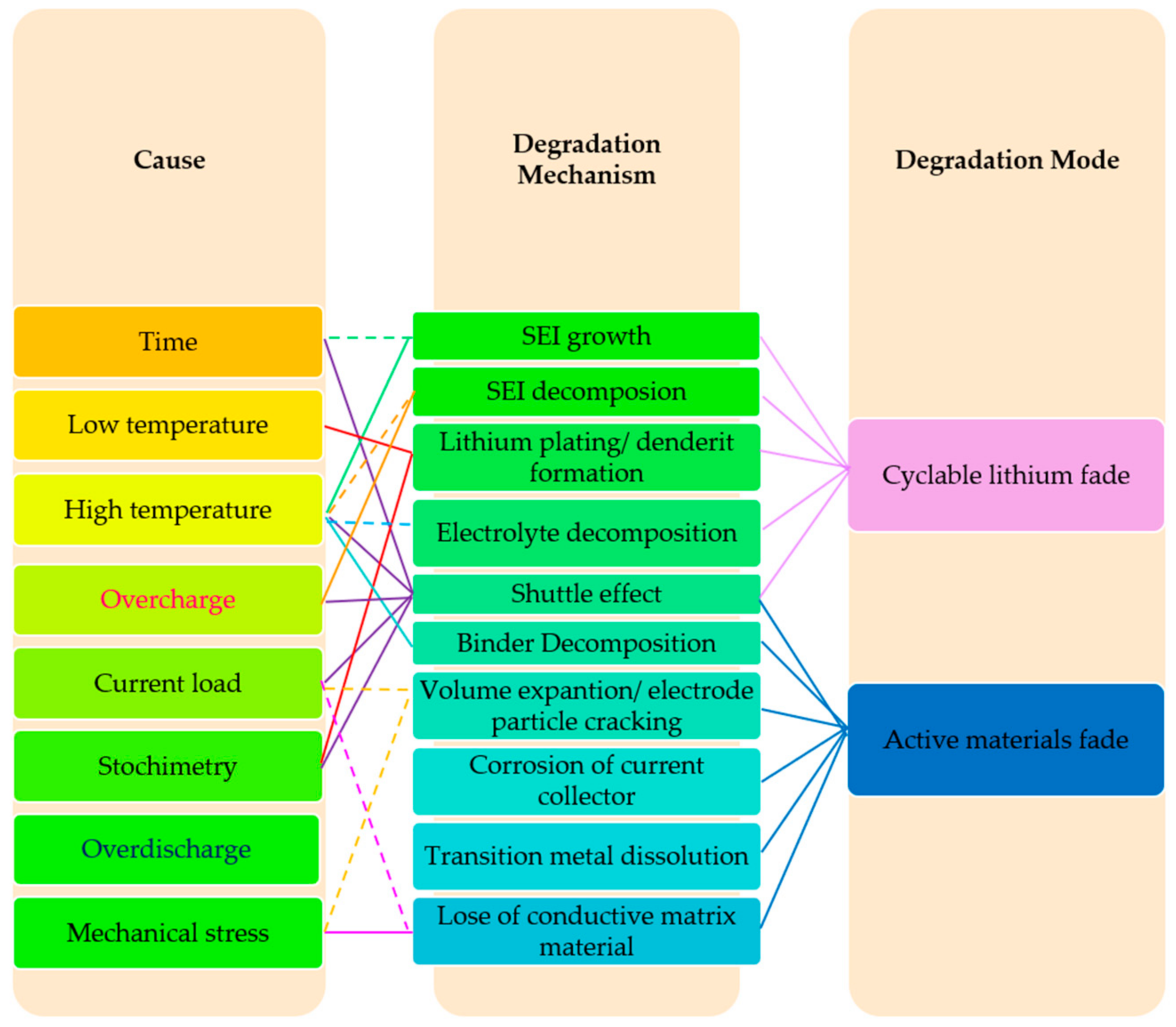
- The capacity loss can be linked to the degradation of the electrolyte components through side reactions with Li+ resulting in less cyclable Li+ in the electrodes by loss of active electrode material. The power losses consist of contact resistance in interphases between the materials, growth of resistive film on the active material, loss of active surface area, and impaired mass transport [21]. Generally, there are two main reasons that cause lithium- based batteries to deteriorate [21,22]:
- 6.
- Cyclable lithium fade: The performance of lithium-ion-based batteries depends on the transportation of lithium ions between the positive and negative electrodes of the battery. If the lithium ions lose their mobility during side reactions, then the battery performance will decrease;
- 7.
- Active materials fade: The active materials of a battery are those that participate in the electrochemical charge/discharge reaction. These include the two electrode materials of a cell and the electrolyte between them. If for any reason (such as material dissolution, particle isolation, electrode delamination, and structure degradation) these materials are subject to change, the battery capacity will decrease.
2. Experimental Results
- A discharge step: obtaining the residual capacity of the cell after storage;
- A pre-condition cycle: for resetting ‘a cumulative history’ of the cell;
- A cycle to measure the cell’s maximum charge and discharge capacity;
- Measuring the cell’s resistance (at different SOC levels with 10% resolution, with 30 s long current pulses with amplitudes of 0.1, 0.2, and 0.5 C for charging and 0.2, 0.5, and 1 C for discharging);
- Measuring the shuttle current;
- Discharging the cell to the considered SOC level for storage.
3. Results and Discussion
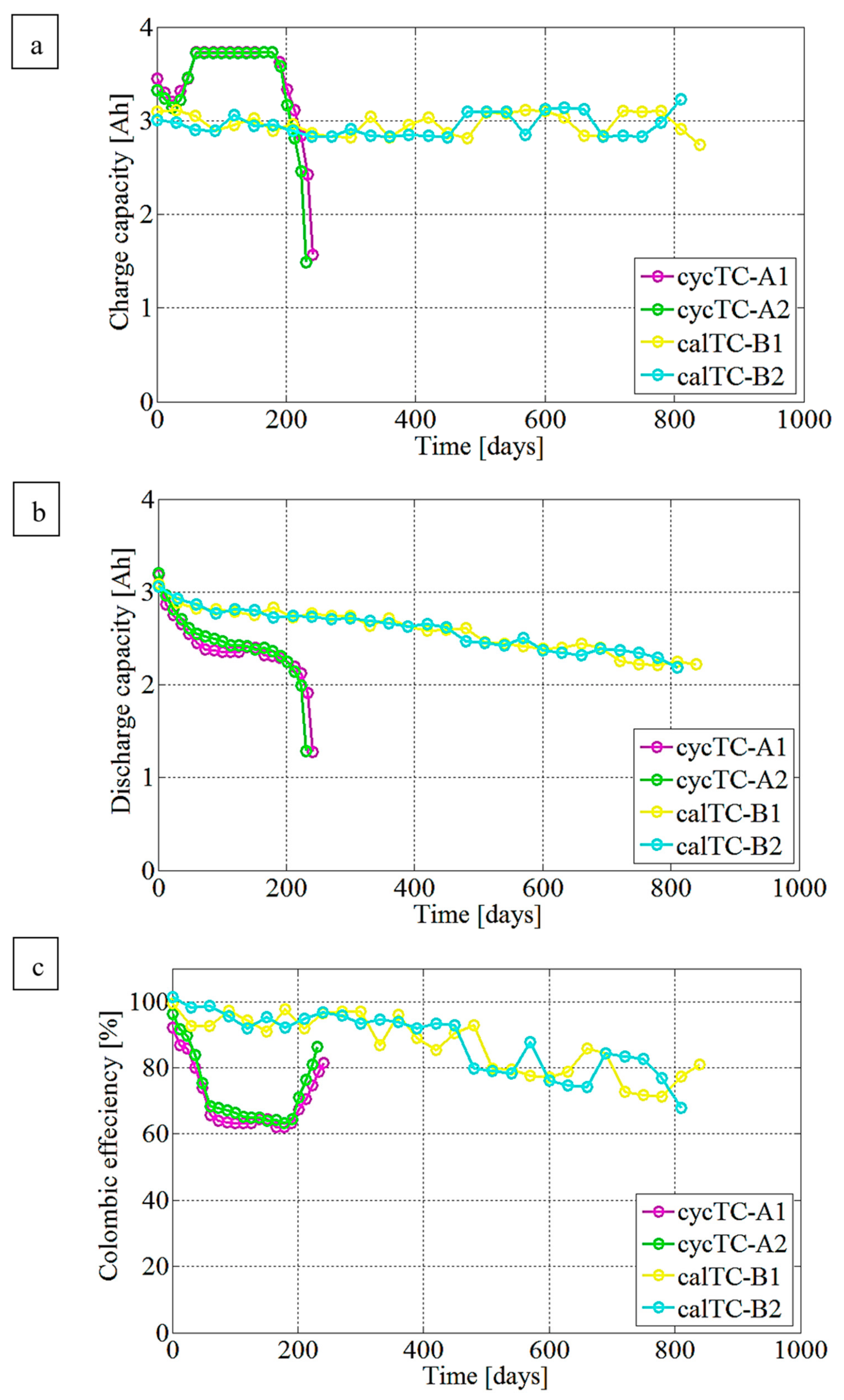
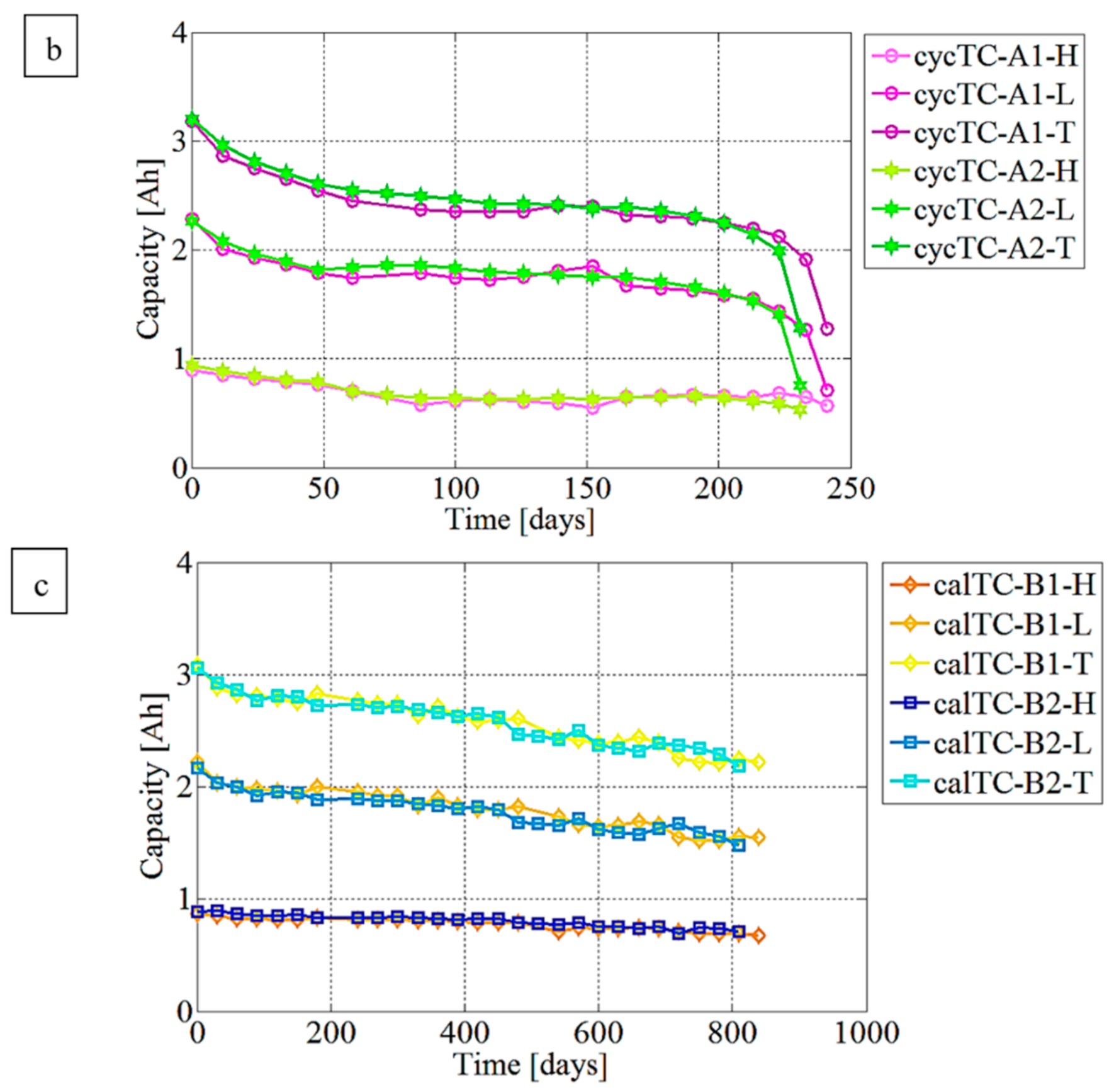
3.1. Capacity Fading Evaluation Trend
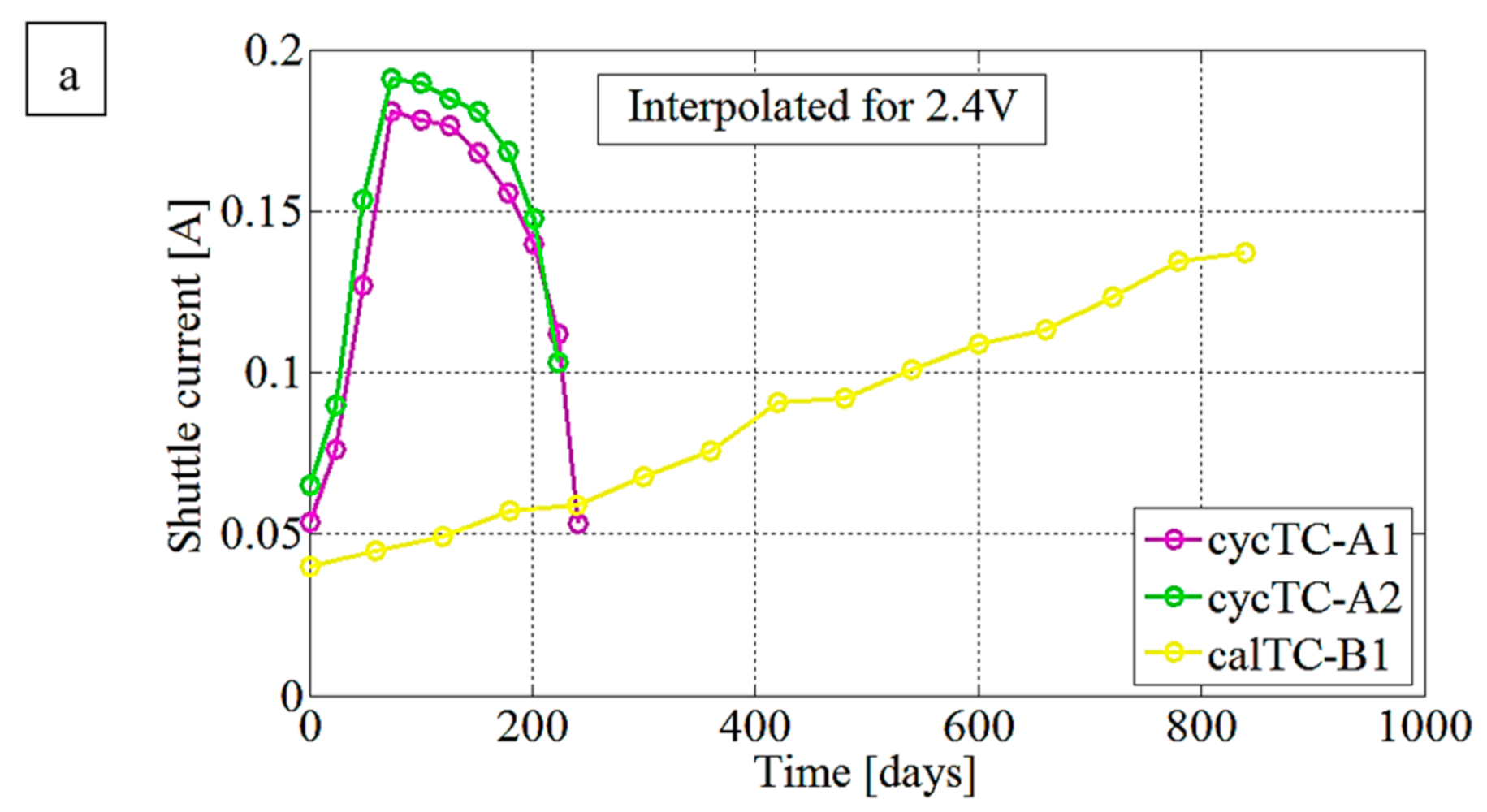
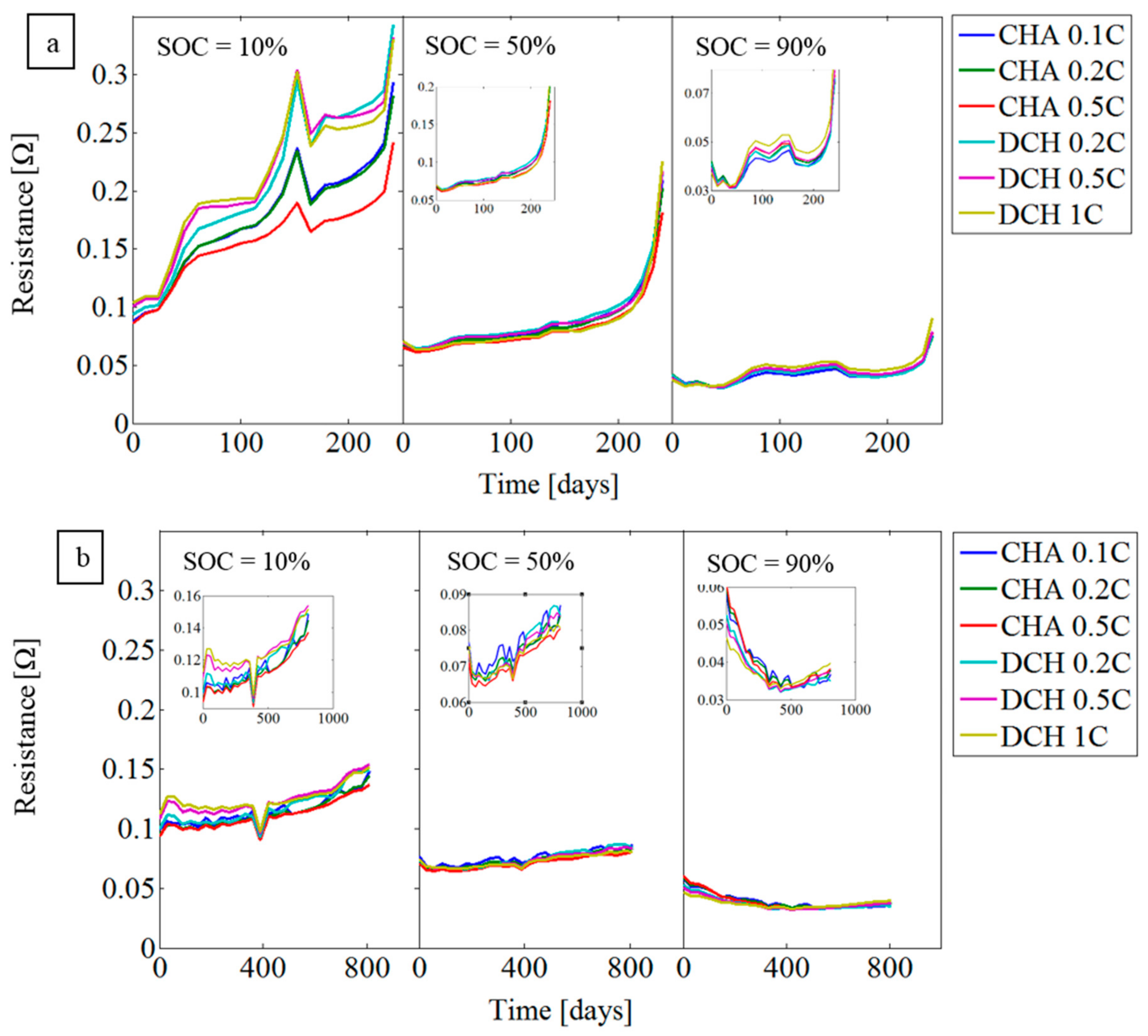
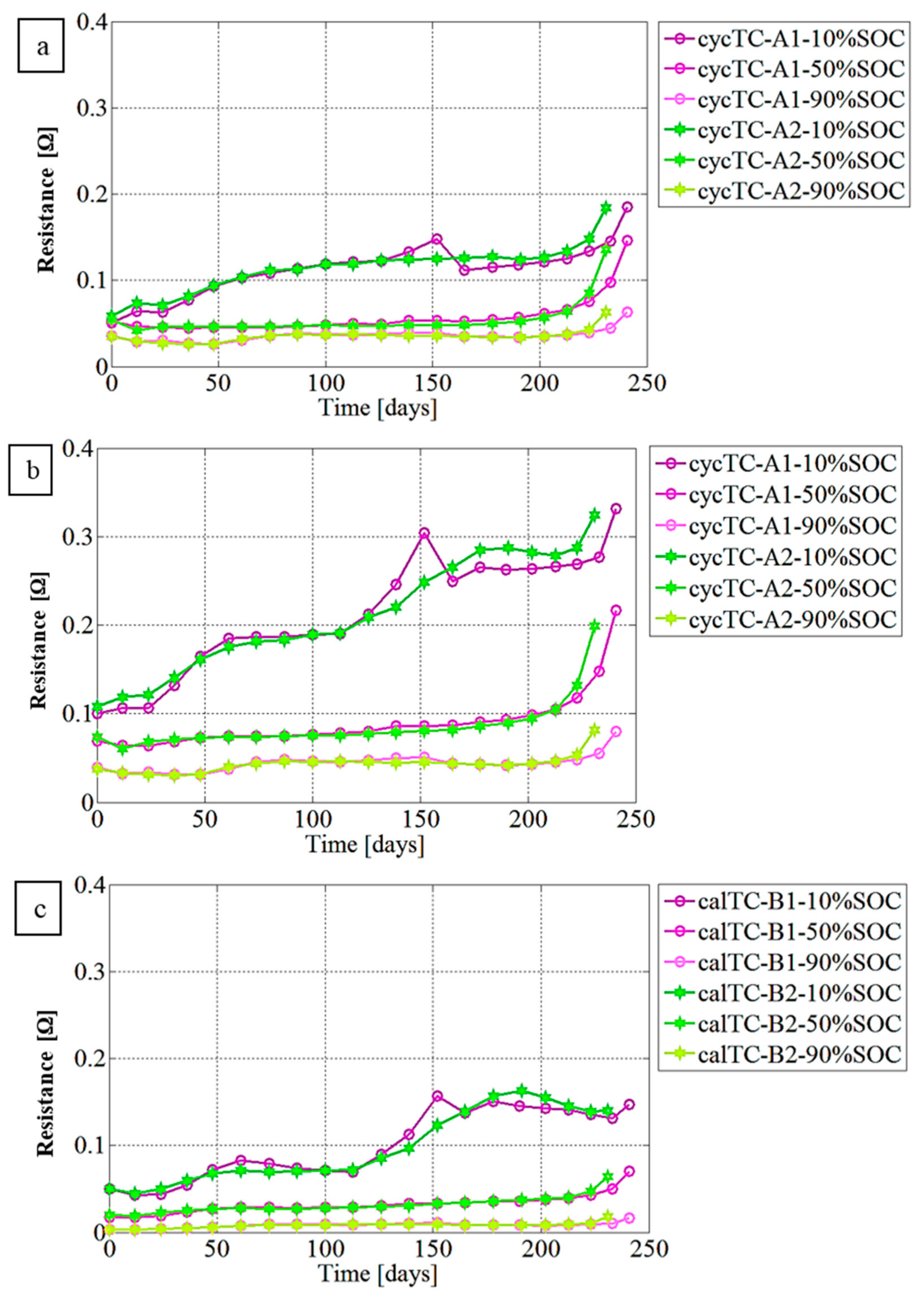
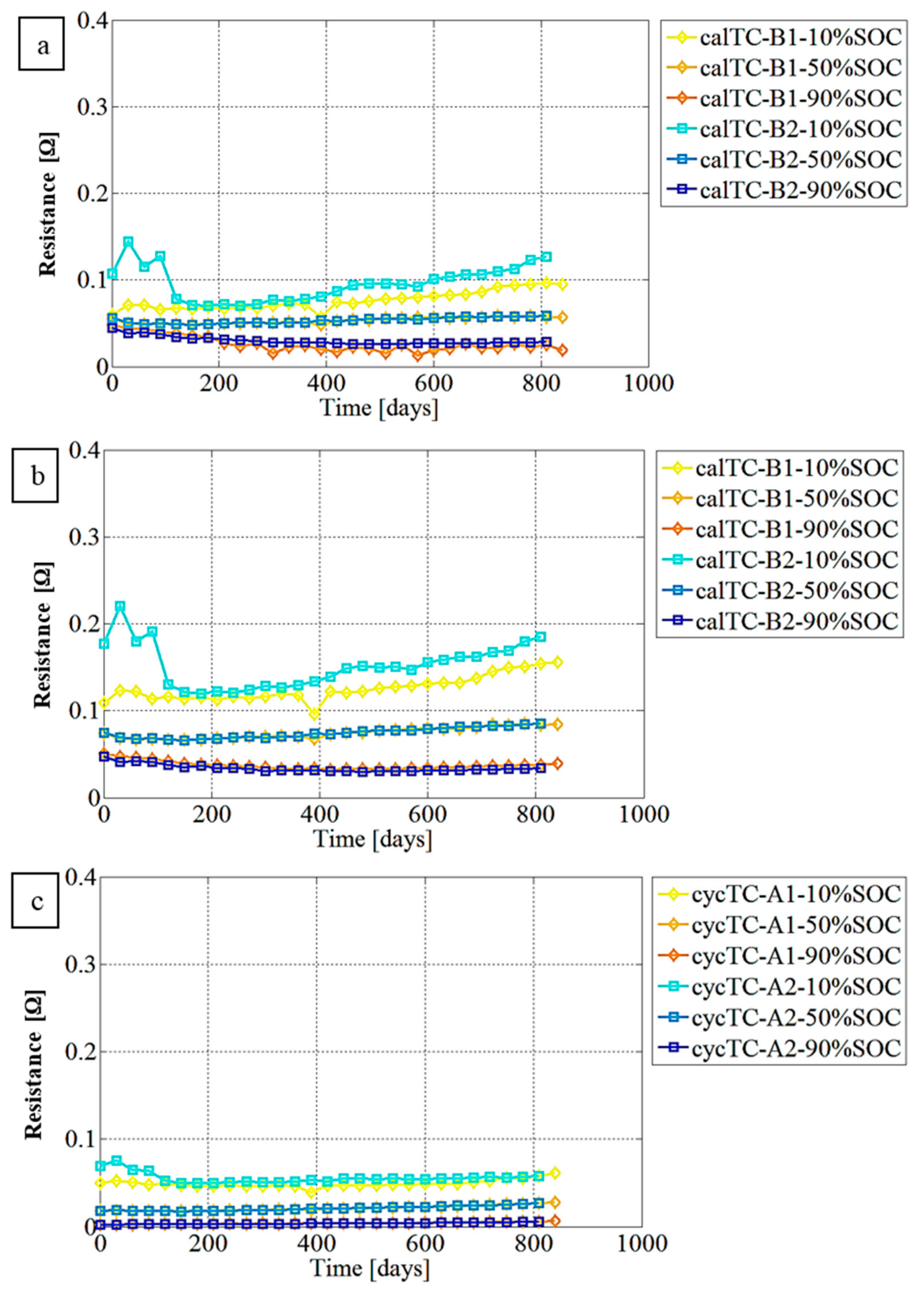
3.2. Resistance Evaluation Trend
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fang, R.; Zhao, S.; Sun, Z.; Wang, D.W.; Cheng, H.M.; Li, F. More reliable lithium-sulfur batteries: Status, solutions and prospects. Adv. Mater. 2017, 29, 1606823. [Google Scholar] [CrossRef]
- Manthiram, A.; Fu, Y.; Su, Y.-S. Challenges and prospects of lithium-sulfur batteries. Acc. Chem. Res. 2013, 46, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Bruce, P.G.; Freunberger, S.A.; Hardwick, L.J.; Tarascon, J.-M. Li-O2 and Li-S batteries with high energy storage. Nat. Mater. 2012, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Nazar, L.F. Advances in Li-S batteries. J. Mater. Chem. 2010, 20, 9821–9826. [Google Scholar] [CrossRef]
- Zheng, D.; Zhang, X.; Wang, J.; Qu, D.; Yang, X.; Qu, D. Reduction mechanism of sulfur in lithium-sulfur battery: From elemental sulfur to polysulfide. J. Power Sources 2016, 301, 312–316. [Google Scholar] [CrossRef]
- Yun, S.; Park, S.H.; Yeon, J.S.; Park, J.; Jana, M.; Suk, J.; Park, H.S. Materials and device constructions for aqueous lithium-sulfur batteries. Adv. Funct. Mater. 2018, 28, 1707593. [Google Scholar] [CrossRef]
- Chen, W.; Lei, T.; Wu, C.; Deng, M.; Gong, C.; Hu, K.; Ma, Y.; Dai, L.; Lv, W.; He, W. Designing safe electrolyte systems for a high-stability lithium-sulfur battery. Adv. Energy Mater. 2018, 8, 1702348. [Google Scholar] [CrossRef]
- Li, K.; Wang, B.; Su, D.; Park, J.; Ahn, H.; Wang, G. Enhance electrochemical performance of lithium sulfur battery through a solution-based processing technique. J. Power Sources 2012, 202, 389–393. [Google Scholar] [CrossRef]
- Wild, M.; O’neill, L.; Zhang, T.; Purkayastha, R.; Minton, G.; Marinescu, M.; Offer, G. Lithium sulfur batteries, a mechanistic review. Energy Environ. Sci. 2015, 8, 3477–3494. [Google Scholar] [CrossRef]
- Yang, X.; Li, X.; Adair, K.; Zhang, H.; Sun, X. Structural design of lithium-sulfur batteries: From fundamental research to practical application. Electrochem. Energy Rev. 2018, 1, 239–293. [Google Scholar] [CrossRef] [Green Version]
- Pang, Q.; Liang, X.; Kwok, C.Y.; Nazar, L.F. Advances in lithium-sulfur batteries based on multifunctional cathodes and electrolytes. Nat. Energy 2016, 1, 1–11. [Google Scholar] [CrossRef]
- Su, Y.S.; Manthiram, A. Sulfur-lithium-insertion compound composite cathodes for Li-S batteries. J. Power Source 2014, 270, 101–105. [Google Scholar] [CrossRef]
- Cheon, S.-E.; Ko, K.-S.; Cho, J.-H.; Kim, S.-W.; Chin, E.-Y.; Kim, H.-T. Rechargeable lithium sulfur battery: I. Structural change of sulfur cathode during discharge and charge. J. Electrochem. Soc. 2003, 150, A796. [Google Scholar] [CrossRef]
- Zhao, H.; Deng, N.; Yan, J.; Kang, W.; Ju, J.; Ruan, Y.; Wang, X.; Zhuang, X.; Li, Q.; Cheng, B. A review on anode for lithium-sulfur batteries: Progress and prospects. Chem. Eng. J. 2018, 347, 343–365. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, G.; Ju, J.; Zhao, Y.; Kang, W.; Deng, N.; Cheng, B. Towards high energy density Li-S batteries with high sulfur loading: From key issues to advanced strategies. Energy Storage Mater. 2020, 32, 320–355. [Google Scholar] [CrossRef]
- Qi, Q.; Lv, X.; Lv, W.; Yang, Q.-H. Multifunctional binder designs for lithium-sulfur batteries. J. Energy Chem. 2019, 39, 88–100. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.S. Liquid electrolyte lithium/sulfur battery: Fundamental chemistry, problems, and solutions. J. Power Sources 2013, 231, 153–162. [Google Scholar] [CrossRef]
- Xiang, Y.; Li, J.; Lei, J.; Liu, D.; Xie, Z.; Qu, D.; Li, K.; Deng, T.; Tang, H. Advanced separators for lithium-ion and lithium-sulfur batteries: A review of recent progress. ChemSusChem 2016, 9, 3023–3039. [Google Scholar] [CrossRef]
- Chung, S.-H.; Manthiram, A. Lithium-Sulfur batteries with the lowest self-discharge and the longest shelf life. ACS Energy Lett. 2017, 2, 1056–1061. [Google Scholar] [CrossRef]
- Yamada, M.; Watanabe, T.; Gunji, T.; Wu, J.; Matsumoto, F. Review of the design of current collectors for improving the battery performance in lithium-ion and post-lithium-ion batteries. Electrochem 2020, 1, 124–159. [Google Scholar] [CrossRef]
- Safari, M.; Delacourt, C. Aging of a commercial graphite/LiFePO4 cell. J. Electrochem. Soc. 2011, 158, A1123. [Google Scholar] [CrossRef]
- Grolleau, S.; Delaille, A.; Gualous, H.; Gyan, P.; Revel, R.; Bernard, J.; Redondo-Iglesias, E.; Peter, J.; SIMCAL Network. Calendar aging of commercial graphite/LiFePO4 cell—Predicting capacity fade under time dependent storage conditions. J. Power Sources 2014, 255, 450–458. [Google Scholar] [CrossRef]
- Li, W.; Yao, H.; Yan, K.; Zheng, G.; Liang, Z.; Chiang, Y.-M.; Cui, Y. The synergetic effect of lithium polysulfide and lithium nitrate to prevent lithium dendrite growth. Nat. Commun. 2015, 6, 7436. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Zhou, Y.-N.; Han, D.; Zhou, J.; Zhou, D.; Tang, W.; Goodenough, J.B. Thermodynamic understanding of Li-dendrite formation. Joule 2020, 4, 864–1879. [Google Scholar] [CrossRef]
- Birkl, C.R.; Roberts, M.R.; McTurk, E.; Bruce, P.G.; Howey, D.A. Degradation diagnostics for lithium ion cells. J. Power Sources 2017, 341, 373–386. [Google Scholar] [CrossRef]
- Yu, X.; Manthiram, A. Electrode-Electrolyte interfaces in lithium-sulfur batteries with liquid or inorganic solid electrolytes. Acc. Chem. Res. 2017, 50, 2653–2660. [Google Scholar] [CrossRef] [PubMed]
- Wolff, D.; Canals Casals, L.; Benveniste, G.; Corchero, C.; Trilla, L. The effects of lithium sulfur battery ageing on second-life possibilities and environmental life cycle assessment studies. Energies 2019, 12, 2440. [Google Scholar] [CrossRef] [Green Version]
- Ren, Y.; Zhao, T.; Liu, M.; Tan, P.; Zeng, Y. Modeling of lithium-sulfur batteries incorporating the effect of Li2S precipitation. J. Power Sources 2016, 336, 115–125. [Google Scholar] [CrossRef]
- Mikhaylik, Y.V.; Akridge, J.R. Polysulfide shuttle study in the Li/S battery system. J. Electrochem. Soc. 2004, 151, A1969. [Google Scholar] [CrossRef]
- Siczek, K.J. Next-Generation Batteries with Sulfur Cathodes; Academic Press: Cambridge, MA, USA, 2019. [Google Scholar]
- Kumar, R.; Liu, J.; Hwang, J.-Y.; Sun, Y.-K. Recent research trends in Li-S batteries. J. Mater. Chem. A 2018, 6, 11582–11605. [Google Scholar] [CrossRef]
- Deng, C.; Wang, Z.; Feng, L.; Wang, S.; Yu, J. Electrocatalysis of sulfur and polysulfides in Li-S batteries. J. Mater. Chem. A 2020, 8, 19704–19728. [Google Scholar] [CrossRef]
- Benveniste, G.; Rallo, H.; Casals, L.C.; Merino, A.; Amante, B. Comparison of the state of lithium-sulphur and lithium-ion batteries applied to electromobility. J. Environ. Manag. 2018, 226, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Redondo-Iglesias, E.; Venet, P.; Pelissier, S. Measuring reversible and irreversible capacity losses on lithium-ion batteries. In Proceedings of the 2016 IEEE Vehicle Power and Propulsion Conference (VPPC), Hangzhou, China, 17–20 October 2016; pp. 1–5. [Google Scholar]
- Redondo-Iglesias, E.; Venet, P.; Pelissier, S. Calendar and cycling ageing combination of batteries in electric vehicles. Microelectron. Reliab. 2018, 88, 1212–1215. [Google Scholar] [CrossRef] [Green Version]
- Su, B.; Ke, X.; Yuan, C. Electrochemical modeling of calendar capacity loss of nickel-manganese-cobalt (NMC)-graphite lithium ion batteries. arXiv 2021, arXiv:2103.02166. [Google Scholar]
- Knap, V.; Stroe, D.-I.; Purkayastha, R.; Walus, S.; Auger, D.J.; Fotouhi, A.; Propp, K. Reference performance test methodology for degradation assessment of lithium-sulfur batteries. J. Electrochem. Soc. 2018, 165, A1601. [Google Scholar] [CrossRef] [Green Version]
- Xiao, J.; Li, Q.; Bi, Y.; Cai, M.; Dunn, B.; Glossmann, T.; Liu, J.; Osaka, T.; Sugiura, R.; Wu, B. Understanding and applying coulombic efficiency in lithium metal batteries. Nat. Energy 2020, 5, 561–568. [Google Scholar] [CrossRef]
- Hofmann, A.F.; Fronczek, D.N.; Bessler, W.G. Mechanistic modeling of polysulfide shuttle and capacity loss in lithium-sulfur batteries. J. Power Sources 2014, 259, 300–310. [Google Scholar] [CrossRef] [Green Version]
- Pastor-Fernández, C.; Bruen, T.; Widanage, W.; Gama-Valdez, M.; Marco, J. A study of cell-to-cell interactions and degradation in parallel strings: Implications for the battery management system. J. Power Sources 2016, 329, 574–585. [Google Scholar] [CrossRef] [Green Version]
- Alber, G. Predicting Battery Performance using Internal Cell Resistance; Albercorp: Boca Raton, FL, USA, 2013; Available online: http://www.alber.com/docs/predictbatt.pdf (accessed on 25 November 2014).
- Ernst, S.; Heins, T.P.; Schlüter, N.; Schröder, U. Capturing the current-overpotential nonlinearity of lithium-ion batteries by nonlinear electrochemical impedance spectroscopy (NLEIS) in charge and discharge direction. Front. Energy Res. 2019, 7, 1–13. [Google Scholar] [CrossRef]
- Fan, F.Y.; Carter, W.C.; Chiang, Y.M. Mechanism and kinetics of Li2S precipitation in lithium-sulfur batteries. Adv. Mater. 2015, 27, 5203–5209. [Google Scholar] [CrossRef] [PubMed]
- Maurer, C.; Commerell, W.; Hintennach, A.; Jossen, A. Continuous shuttle current measurement method for lithium sulfur cells. J. Electrochem. Soc. 2020, 167, 090534. [Google Scholar] [CrossRef]
- Fotouhi, A.; Auger, D.J.; O’Neill, L.; Cleaver, T.; Walus, S. Lithium-Sulfur battery technology readiness and applications—A review. Energies 2017, 10, 1937. [Google Scholar] [CrossRef] [Green Version]
- Moy, D.; Manivannan, A.; Narayanan, S. Direct measurement of polysulfide shuttle current: A window into understanding the performance of lithium-sulfur cells. J. Electrochem. Soc. 2014, 162, A1. [Google Scholar] [CrossRef]
- Fotouhi, A.; Auger, D.J.; Propp, K.; Longo, S. Lithium-Sulfur battery state-of-charge observability analysis and estimation. IEEE Trans. Power Electron. 2017, 33, 5847–5859. [Google Scholar] [CrossRef]
- Chien, Y.-C.; Menon, A.S.; Brant, W.R.; Brandell, D.; Lacey, M.J. Simultaneous monitoring of crystalline active materials and resistance evolution in lithium-sulfur batteries. J. Am. Chem. Soc. 2019, 142, 1449–1456. [Google Scholar] [CrossRef]
- Cheon, S.-E.; Ko, K.-S.; Cho, J.-H.; Kim, S.-W.; Chin, E.-Y.; Kim, H.-T. Rechargeable lithium sulfur battery: II. Rate capability and cycle characteristics. J. Electrochem. Soc. 2003, 150, A800. [Google Scholar] [CrossRef]
- Jiang, J.; Liang, Y.; Ju, Q.; Zhang, L.; Zhang, W.; Zhang, C. An equivalent circuit model for lithium-sulfur batteries. Energy Procedia 2017, 105, 3533–3538. [Google Scholar] [CrossRef]
- Zhao, M.; Li, B.-Q.; Zhang, X.-Q.; Huang, J.-Q.; Zhang, Q. A perspective toward practical lithium-sulfur batteries. ACS Cent. Sci. 2020, 6, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Belharouak, I.; Li, J.C.; Zhang, X.; Bloom, I.; Bareño, J. Role of polysulfides in self-healing lithium-sulfur batteries. Adv. Energy Mater. 2013, 3, 833–838. [Google Scholar] [CrossRef]
- Fan, F.Y.; Pan, M.S.; Lau, K.C.; Assary, R.S.; Woodford, W.H.; Curtiss, L.A.; Carter, W.C.; Chiang, Y.-M. Solvent effects on polysulfide redox kinetics and ionic conductivity in lithium-sulfur batteries. J. Electrochem. Soc. 2016, 163, A3111. [Google Scholar] [CrossRef] [Green Version]
- Fan, F.Y.; Chiang, Y.-M. Electrodeposition kinetics in Li-S batteries: Effects of low electrolyte/sulfur ratios and deposition surface composition. J. Electrochem. Soc. 2017, 164, A917. [Google Scholar] [CrossRef]
- Seyed Ehsan, S. Modeling of Lithium-ion Battery Performance and Thermal Behavior in Electrified Vehicles. Ph.D. Thesis, University of Waterloo, Waterloo, ON, Canada, 2015. [Google Scholar]
| Characteristics | Values |
|---|---|
| Nominal capacity (30 °C) | 3.4 Ah |
| Nominal charging current | 0.34 A (0.1 C-rate) |
| Nominal discharging current | 0.68 A (0.2 C-rate) |
| Nominal voltage | 2.05 V |
| Charge cutoff voltage | 2.45 V |
| Discharge cutoff voltage | 1.5 V |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gohari, S.; Knap, V.; Yaftian, M.R. Investigation on Cycling and Calendar Aging Processes of 3.4 Ah Lithium-Sulfur Pouch Cells. Sustainability 2021, 13, 9473. https://doi.org/10.3390/su13169473
Gohari S, Knap V, Yaftian MR. Investigation on Cycling and Calendar Aging Processes of 3.4 Ah Lithium-Sulfur Pouch Cells. Sustainability. 2021; 13(16):9473. https://doi.org/10.3390/su13169473
Chicago/Turabian StyleGohari, Salimeh, Vaclav Knap, and Mohammad Reza Yaftian. 2021. "Investigation on Cycling and Calendar Aging Processes of 3.4 Ah Lithium-Sulfur Pouch Cells" Sustainability 13, no. 16: 9473. https://doi.org/10.3390/su13169473
APA StyleGohari, S., Knap, V., & Yaftian, M. R. (2021). Investigation on Cycling and Calendar Aging Processes of 3.4 Ah Lithium-Sulfur Pouch Cells. Sustainability, 13(16), 9473. https://doi.org/10.3390/su13169473







