Single Cell Protein: A Potential Substitute in Human and Animal Nutrition
Abstract
:1. Introduction
1.1. Historical Background of SCP
1.2. Applications of SCP
- In animal feed and nutrition, for the stuffing and fattening of poultry, laying hens, calves and pigs.
- As food additives (vitamin and aroma carriers and emulsifying agents), to enhance nutritional value (of baked food items, ready-made meals, soups, etc.) and as starter cultures (baker’s, brewer’s and wine yeast).
- In industrial processes, as a foam-stabilizing agent and in paper and leather processing.
1.3. Mechanism of Production
- High-energy resources: gas oil, natural gas, ethanol, methanol, n-alkanes and acetic acid
- Renewable plant resources: starch, sugar and cellulose
- Various wastes: sulfite waste liquor, molasses, whey, milk and fruit waste
- Carbon dioxide
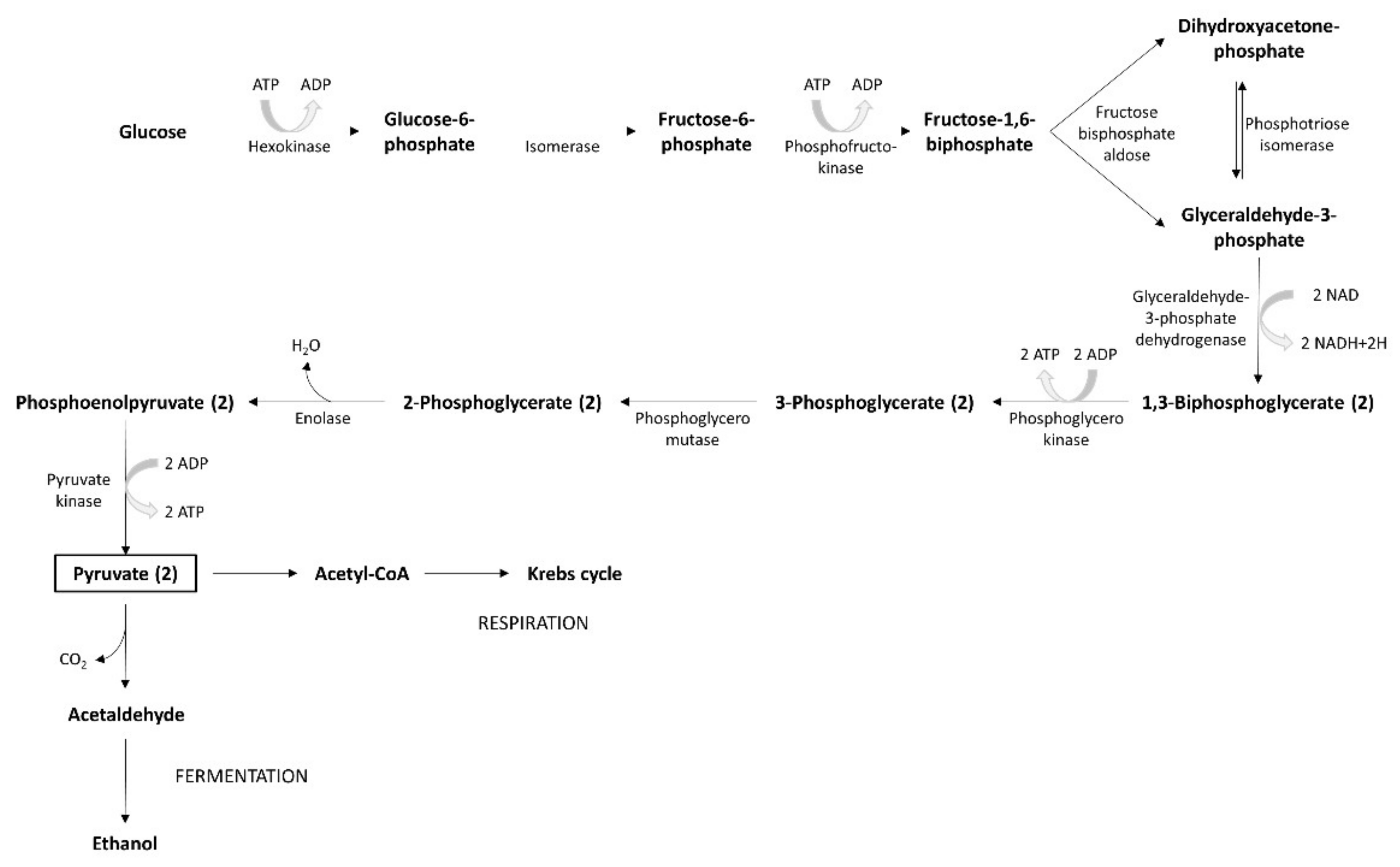
2. Bacterial Metabolism
Bacterial Sources
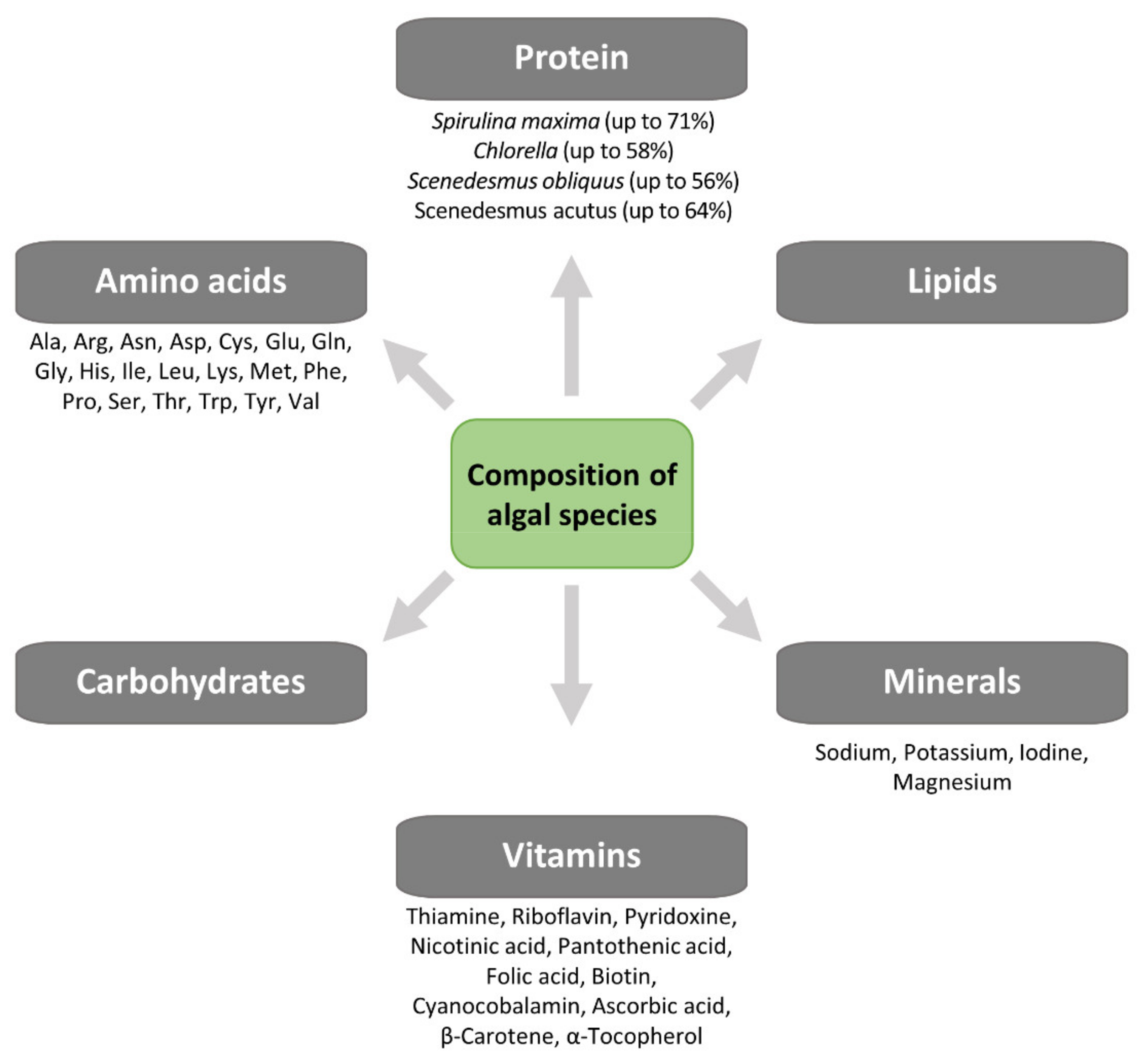
3. Algal Metabolism
Algae Sources
| Algae | Substrate | SCP (%) | Reference |
|---|---|---|---|
| Chaetomorpha antennina | Soda ash effluent | 14.0–18.2% | [74] |
| Ulva fasciata | Soda ash effluent | 13.7–18.6% | [74] |
| Chlorella sp. | Tofu waste | 52.32% ± 3.31 | [75] |
| Tempeh waste | 52.00% ± 1.80 | [75] | |
| Cheese waste | 15.43% ± 2.55 | [75] | |
| Chlorella salina | Saline sewage effluents | 51% | [76] |
| Gracilaria domingensis | Natural habit | 6.2% | [77] |
| Gracilaria birdiae | Natural habit | 7.1% | [77] |
| Laurencia filiformis | Natural habit | 18.3% | [77] |
| Laurencia intricate | Natural habit | 4.6% | [77] |
| Palmaria palmate | Natural habit | 8–35% | [78] |
| Natural habit | 8.0–35.0% | [79] | |
| Natural habit | 11.9–21.9% | [80] | |
| Natural habit | 13.5% | [77] | |
| Natural habit | 12–21% | [81] | |
| Chondrus crispus | Natural habit | 20.1% | [82] |
| Porphyra umbilicalis | Natural habit | 15–37% | [81] |
| Gracilaria verrucosa | Natural habit | 7.0–23.0% | [79] |
| Chlorella sorokiniana | Wastewater | 45% | [35] |
| Scenedesmus obliquus | Wastewater | 52% | [83] |
| Spirulina | Salinated water | 48.59% | [84] |
| Desalinated wastewater | 56.17% | [84] |
4. Fungal Metabolism
Fungal Sources
5. Yeast Metabolism
Yeast Sources
| Yeast | Substrate | SCP (%) | Reference |
|---|---|---|---|
| Saccharomyces cerevisiae | Orange pulp, molasses, brewer’s spent grain | 39 | [112] |
| Candida krusei | Cheese whey | 48 | [113] |
| Candida tropicalis | Molasses | 56 | [114] |
| Candida tropicalis | Bagasse | 31 | [115] |
| Candida utilis | Waste capsicum powder | 29 | [116] |
| Candida utilis | Poultry litter | 48 | [117] |
| Candida utilis | Potato starch industry waste | 46 | [30] |
| Candida utilis | Potato wastewater | 49 | [118] |
| Hanseniaspora uvarum | Spoiled date palm fruit | 49 | [119] |
| Kefir sp. | Cheese whey | 54 | [120] |
| Kefir sp. | Orange pulp, molasses, brewer’s spent grain, whey, potato pulp, malt spent | 23 | [112] |
| Debaryomyces hansenii | Brewery’s spent grains (hemicellulosic hydrolysate) | 32 | [121] |
| Kluyveromyces marxianus | Cheese whey | 43 | [113] |
| Kluyveromyces marxianus | Orange pulp, molasses, brewer’s spent grain, whey, potato pulp | 34 | [112] |
| Zygosaccharomyces rouxi | Spoiled date palm fruit | 49 | [119] |
| Yarrowia lipolytica | Inulin, crude oil, glycerol waste hydrocarbons | 48–54 | [122] |
6. Nutritional Benefits of SCP
7. Economic Analysis
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pihlajaniemi, V.; Ellilä, S.; Poikkimäki, S.; Nappa, M.; Rinne, M.; Lantto, R.; Siika-aho, M. Comparison of pretreatments and cost-optimization of enzymatic hydrolysis for production of single cell protein from grass silage fibre. Bioresour. Technol. Rep. 2020, 9, 100357. [Google Scholar] [CrossRef]
- Verstraete, W.; Clauwaert, P.; Vlaeminck, S.E. Used water and nutrients: Recovery perspectives in a ‘panta rhei’ context. Bioresour. Technol. 2016, 215, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Ritala, A.; Häkkinen, S.T.; Toivari, M.; Wiebe, M.G. Single Cell Protein—State-of-the-Art, Industrial Landscape and Patents 2001–2016. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The State of Food Security and Nutrition in the World 2020: Transforming Food Systems for Affordable Healthy Diets; Food and Agriculture Organization: Rome, Italy, 2020. [Google Scholar]
- Food and Agricultural Organization of the United Nations. How to Feed the World in 2050. Available online: http://www.fao.org/fileadmin/templates/wsfs/docs/expert_paper/How_to_Feed_the_World_in_2050.pdf (accessed on 18 June 2021).
- Junaid, F.; Khawaja, L.A.; Ali, S. Single cell proteins as a potential meat substitute: A critical review. World J. Pharm. Res. 2020, 9, 141–161. [Google Scholar] [CrossRef]
- Srividya, A.R.; Vishnuvarthan, V.J.; Murugappan, M.; Dahake, P.G. Single Cell Protein- A Review. Int. J. Pharm. Res. Sch. 2013, 2, 472–485. [Google Scholar]
- Suman, G.; Nupur, M.; Anuradha, S.; Pradeep, B. Single Cell Protein Production: A Review. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 251–262. [Google Scholar]
- Zhou, Y.-M.; Chen, Y.-P.; Guo, J.-S.; Shen, Y.; Yan, P.; Yang, J.-X. Recycling of orange waste for single cell protein production and the synergistic and antagonistic effects on production quality. J. Clean. Prod. 2019, 213, 384–392. [Google Scholar] [CrossRef]
- Nagare, B.; Bhambere, S.; Kumar, S.; Kakad, K.; Nagare, N. In Situ Gelling System: Smart Carriers for Ophthalmic Drug Delivery. Int. J. Pharm. Res. Sch. 2015, 4, 10–23. [Google Scholar]
- Yunus, F.-u.-N.; Nadeem, M.; Rashid, F. Single-cell protein production through microbial conversion of lignocellulosic residue (wheat bran) for animal feed. J. Inst. Brew. 2015, 121, 553–557. [Google Scholar] [CrossRef] [Green Version]
- Matassa, S.; Boon, N.; Pikaar, I.; Verstraete, W. Microbial protein: Future sustainable food supply route with low environmental footprint. Microb. Biotechnol. 2016, 9, 568–575. [Google Scholar] [CrossRef]
- Ali, S.; Mushtaq, J.; Nazir, F.; Sarfraz, H. Production and Processing of Single Cell Protein (SCP)—A review. Eur. J. Pharm. Med. Res. 2017, 4, 86–94. [Google Scholar]
- Raziq, A.; Lateef, M.; Ullah, A.; Ullah, H.; Khan, M.W. Single cell protein (SCP) production and potential substrates: A comprehensive review. Philipp. Accredit. Bur. 2020, 9. [Google Scholar] [CrossRef]
- Queiroz, M.I.; Lopes, E.J.; Zepka, L.Q.; Bastos, R.G.; Goldbeck, R. The kinetics of the removal of nitrogen and organic matter from parboiled rice effluent by cyanobacteria in a stirred batch reactor. Bioresour. Technol. 2007, 98, 2163–2169. [Google Scholar] [CrossRef]
- Kadim, I.T.; Mahgoub, O.; Baqir, S.; Faye, B.; Purchas, R. Cultured meat from muscle stem cells: A review of challenges and prospects. J. Integr. Agric. 2015, 14, 222–233. [Google Scholar] [CrossRef] [Green Version]
- Zepka, L.Q.; Jacob-Lopes, E.; Goldbeck, R.; Queiroz, M.I. Production and biochemical profile of the microalgae Aphanothece microscopica Nägeli submitted to different drying conditions. Chem. Eng. Process. 2008, 47, 1305–1310. [Google Scholar] [CrossRef]
- John, R.P.; Anisha, G.S.; Nampoothiri, K.M.; Pandey, A. Micro and macroalgal biomass: A renewable source for bioethanol. Bioresour. Technol. 2011, 102, 186–193. [Google Scholar] [CrossRef]
- Zha, X.; Tsapekos, P.; Zhu, X.; Khoshnevisan, B.; Lu, X.; Angelidaki, I. Bioconversion of wastewater to single cell protein by methanotrophic bacteria. Bioresour. Technol. 2021, 320, 124351. [Google Scholar] [CrossRef]
- Jurtshuk, P.J. Bacterial Metabolism. In Medical Microbiology, 4th ed.; Baron, S., Ed.; University of Texas Medical Branch at Galveston: Galveston, TX, USA, 1996; Chapter 4. [Google Scholar]
- Vasdekis, A.E.; Stephanopoulos, G. Review of methods to probe single cell metabolism and bioenergetics. Metab. Eng. 2015, 27, 115–135. [Google Scholar] [CrossRef] [Green Version]
- Müller, W.E.G.; Schröder, H.C.; Wang, X. Inorganic Polyphosphates As Storage for and Generator of Metabolic Energy in the Extracellular Matrix. Chem. Rev. 2019, 119, 12337–12374. [Google Scholar] [CrossRef] [PubMed]
- Garimella, S.; Kugle, K.; Kssoju, A.; Merugu, R. Current status on single cell protein (SCP) production from photosynthetic purple non sulfur bacteria. J. Chem. Pharma. Sci. 2017, 10, 915–922. [Google Scholar]
- Bamberg, J. British Petroleum and Global Oil 1950–1975. The Challenge of Nationalism; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Øverland, M.; Tauson, A.-H.; Shearer, K.; Skrede, A. Evaluation of methane-utilising bacteria products as feed ingredients for monogastric animals. Arch. Anim. Nutr. 2010, 64, 171–189. [Google Scholar] [CrossRef]
- Carrillo, W. Biotechnology and Food Production; ED-Tech Press: Essex, UK, 2020. [Google Scholar]
- Chumpol, S.; Kantachote, D.; Nitoda, T.; Kanzaki, H. Administration of purple nonsulfur bacteria as single cell protein by mixing with shrimp feed to enhance growth, immune response and survival in white shrimp (Litopenaeus vannamei) cultivation. Aquaculture 2018, 489, 85–95. [Google Scholar] [CrossRef]
- Kurbanoglu, E.B.; Algur, O.F. Single-cell protein production from ram horn hydrolysate by bacteria. Bioresour. Technol. 2002, 85, 125–129. [Google Scholar] [CrossRef]
- Liu, B.; Li, Y.; Song, J.; Zhang, L.; Dong, J.; Yang, Q. Production of single-cell protein with two-step fermentation for treatment of potato starch processing waste. Cellulose 2014, 21, 3637–3645. [Google Scholar] [CrossRef]
- Liu, B.; Song, J.; Li, Y.; Niu, J.; Wang, Z.; Yang, Q. Towards industrially feasible treatment of potato starch processing waste by mixed cultures. Appl. Biochem. Biotechnol. 2013, 171, 1001–1010. [Google Scholar] [CrossRef]
- Wongputtisin, P.; Khanongnuch, C.; Kongbuntad, W.; Niamsup, P.; Lumyong, S.; Sarkar, P.K. Use of Bacillus subtilis isolates from Tua-nao towards nutritional improvement of soya bean hull for monogastric feed application. Lett. Appl. Microbiol. 2014, 59, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.P.; Kim, J.D.; Kim, J.E.; Kim, I.H. Amino acid digestibility of single cell protein from Corynebacterium ammoniagenes in growing pigs. Anim. Feed Sci. Technol. 2013, 180, 111–114. [Google Scholar] [CrossRef]
- Kunasundari, B.; Murugaiyah, V.; Kaur, G.; Maurer, F.H.J.; Sudesh, K. Revisiting the Single Cell Protein Application of Cupriavidus necator H16 and Recovering Bioplastic Granules Simultaneously. PLoS ONE 2013, 8, e78528. [Google Scholar] [CrossRef] [Green Version]
- Taran, M.; Asadi, N. A Novel Approach for Environmentally Friendly Production of Single Cell Protein From Petrochemical Wastewater Using a Halophilic Microorganism in Different Conditions. Petrol. Sci Technol. 2014, 32, 625–630. [Google Scholar] [CrossRef]
- Rasouli, Z.; Valverde-Pérez, B.; D’Este, M.; De Francisci, D.; Angelidaki, I. Nutrient recovery from industrial wastewater as single cell protein by a co-culture of green microalgae and methanotrophs. Biochem. Eng. 2018, 134, 129–135. [Google Scholar] [CrossRef]
- Xu, M.; Zhou, H.; Yang, X.; Angelidaki, I.; Zhang, Y. Sulfide restrains the growth of Methylocapsa acidiphila converting renewable biogas to single cell protein. Water Res. 2020, 184, 116138. [Google Scholar] [CrossRef]
- Yazdian, F.; Hajizadeh, S.; Shojaosadati, S.A.; Khalilzadeh, R.; Jahanshahi, M.; Nosrati, M. Production of single cell protein from natural gas: Parameter optimization and RNA evaluation. Iran. J. Biotechnol. 2005, 3, 235–242. [Google Scholar]
- Lee, J.Z.; Logan, A.; Terry, S.; Spear, J.R. Microbial response to single-cell protein production and brewery wastewater treatment. Microb. Biotechnol. 2015, 8, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Kornochalert, N.; Kantachote, D.; Chaiprapat, S.; Techkarnjanaruk, S. Use of Rhodopseudomonas palustris P1 stimulated growth by fermented pineapple extract to treat latex rubber sheet wastewater to obtain single cell protein. Ann. Microbiol. 2014, 64, 1021–1032. [Google Scholar] [CrossRef]
- Getha, K.; Vikineswary, S.; Chong, V.C. Isolation and growth of the phototrophic bacterium Rhodopseudomonas palustris strain B1 in sago-starch-processing wastewater. World J. Microbiol. Biotechnol. 1998, 14, 505–511. [Google Scholar] [CrossRef]
- Ponsano, E.H.G.; Lacava, P.M.; Pinto, M.F. Chemical composition of Rhodocyclus gelatinosus biomass produced in poultry slaughterhouse wastewater. Braz. Arch. Biol. Technol. 2003, 46, 143–147. [Google Scholar] [CrossRef] [Green Version]
- Noparatnaraporn, N.; Nagai, S. Selection of Rhodobacter sphaeroides P47 as a useful source of single cell protein. J. Gen. Appl. Microbiol. 1986, 32, 351–359. [Google Scholar] [CrossRef]
- He, J.; Zhang, G.; Lu, H. Treatment of soybean wastewater by a wild strain Rhodobacter sphaeroides and to produce protein under natural conditions. Front. Environ. Sci. Eng. 2010, 4, 334–339. [Google Scholar] [CrossRef]
- Suwansaard, M. Production of Hydrogen and 5-Aminolevulinic Acid by Photosynthetic Bacteria from Palm Oil Mill Effluent. Prince of Songkla University. 2010. Available online: https://kb.psu.ac.th/psukb/handle/2016/10300 (accessed on 22 June 2021).
- Balloni, W.; Carlozzi, P.; Ventura, S.; Sacchi, A. Microbial biomass for fertilizer use from the photo-anaerobic treatment of pig-wastes. In Proceedings of the International Symposium on Compost Recycling of Wastes, Udine, Italy, 17–19 April 1986. [Google Scholar]
- Balloni, W.; Carlozzi, P.; Ventura, S.; De Phillipis, R.; Bosco, M. A three years experiment on the production of Rhodopseudomonas and Rhodospirillum biomass by outdoor culture on different wastes. In Proceedings of the 4th International Conference on Biomass for Energy and Industry, Orleans, France, 11–15 May 1987. [Google Scholar]
- Sasaki, K.; Noparatnaraporn, N.; Hayashi, M.; Nishizawa, Y.; Nagai, S. Single cell protein production by treatment of soybean wastes with Rhodopseudomonas gelatinos. J. Ferment. Technol. 1981, 59, 471–477. [Google Scholar]
- Prasertsan, P.; Choorit, W.; Suwanno, S. Isolation, identification and growth conditions of photosynthetic bacteria found in seafood processing wastewater. World J. Microbiol. Biotechnol. 1993, 9, 590–592. [Google Scholar] [CrossRef]
- Kar Soon, T.; Al-Azad, S.; Ransangan, J. Isolation and characterization of purple non-sulfur bacteria, Afifella marina, producing large amount of carotenoids from mangrove microhabitats. J. Microbiol. Biotechnol. 2014, 24, 1034–1043. [Google Scholar] [CrossRef] [Green Version]
- Whangchenchom, W.; Chiemchaisri, W.; Tapaneeyaworawong, P.; Powtongsook, S. Wastewater from Instant Noodle Factory as the Whole Nutrients Source for the Microalga Scenedesmus sp. Cultivation. Environ. Eng. Res. 2014, 19, 283–287. [Google Scholar] [CrossRef]
- Saejung, C.; Thammaratana, T. Biomass recovery during municipal wastewater treatment using photosynthetic bacteria and prospect of production of single cell protein for feedstuff. Environ. Technol. 2016, 37, 3055–3061. [Google Scholar] [CrossRef]
- Prasertsan, P.; Prasertsan, S.; H-Kittikun, A. Recycling of agro-industrial wastes through cleaner technology. Biotechnol. Encycl. Life Support Syst. 1997, 10, 1–11. [Google Scholar]
- Poulain, A.J.; Newman, D.K. Rhodobacter capsulatus catalyzes light-dependent Fe(II) oxidation under anaerobic conditions as a potential detoxification mechanism. Appl. Environ. Microbiol. 2009, 75, 6639–6646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfennig, N. Rhodopseudomonas acidophila, sp. n., a new species of the budding purple nonsulfur bacteria. J. Bacteriol. 1969, 99, 597–602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noparatnaraporn, N.; Trakulnaleumsai, S.; Silveira, R.G.; Nishizawa, Y.; Nagai, S. SCP production by mixed culture of Rhodocyclus gelatinosus and Rhodobacter sphaeroides from Cassava Waste. J. Ferment. Technol. 1987, 65, 11–16. [Google Scholar] [CrossRef]
- Honda, R.; Fukushi, K.; Yamamoto, K. Optimization of wastewater feeding for single-cell protein production in an anaerobic wastewater treatment process utilizing purple non-sulfur bacteria in mixed culture condition. J. Biotechnol. 2006, 125, 565–573. [Google Scholar] [CrossRef]
- Kim, J.K.; Lee, B.-K.; Kim, S.-H.; Moon, J.-H. Characterization of denitrifying photosynthetic bacteria isolated from photosynthetic sludge. Aquac. Eng. 1999, 19, 179–193. [Google Scholar] [CrossRef]
- Kelechi, M.; Ukaegbu-Obi, K.M. Single Cell Protein: A Resort to Global Protein Challenge and Waste Management. J. Microbiol. Microb. Technol. 2016, 1, 5. [Google Scholar]
- Johnson, E.A. Biotechnology of non-Saccharomyces yeasts--the ascomycetes. Appl. Microbiol. Biotechnol. 2013, 97, 503–517. [Google Scholar] [CrossRef] [PubMed]
- Ding, C. Experiments on the biodegradation of phenol wastewater by immobilized photosynthetic bacteria. Water Resour. Prot. 2008, 24, 93–95. [Google Scholar]
- Madukasi, E.I.; Dai, X.; He, C.; Zhou, J. Potentials of phototrophic bacteria in treating pharmaceutical wastewater. Int. J. Environ. Sci. Technol. 2010, 7, 165–174. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.C.; Shih, Y.C.; Chiou, P.; Yu, B. Evaluating Nutritional Quality of Single Stage- and Two Stage-fermented Soybean Meal. Asian Australas. J. Anim. Sci. 2010, 23. [Google Scholar] [CrossRef]
- Kantachote, D.; Torpee, S.; Umsakul, K. The potential use of anoxygenic phototrophic bacteria for treating latex rubber sheet wastewater. Electron. J. Biotechnol. 2005, 8, 314–323. [Google Scholar] [CrossRef] [Green Version]
- Kalyuzhnaya, M.G.; Puri, A.W.; Lidstrom, M.E. Metabolic engineering in methanotrophic bacteria. Metabol. Eng. 2015, 29, 142–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beardall, J.; Raven, J.A. Algal Metabolism. Encycl. Life Sci. 2012. [Google Scholar] [CrossRef]
- Falkowski, P.G.; Raven, J.A. Aquatic Photosynthesis, 2nd ed.; STU-Student Edition, Ed.; Princeton University Press: Princeton, NJ, USA, 2007. [Google Scholar]
- Radmer, R.J.; Parker, B.C. Commercial applications of algae: Opportunities and constraints. J. Appl. Phycol. 1994, 6, 93–98. [Google Scholar] [CrossRef]
- Darcy Vrillon, B. Nutritional aspects of the developing use of marine macroalgae for the human food industry. Int. J. Food Sci. Nutr. 1993, 44, S23–S35. [Google Scholar]
- Wood, A.; Toerien, D.F.; Robinson, R.K. Developments in Food Proteins-7; Hudson, B.J.F., Ed.; Elsevier Applied Science: London, UK, 1991. [Google Scholar]
- Heimann, K.; Huerlimann, R. Microalgal Classification: Major Classes and Genera of Commercial Microalgal Species. In Handbook of Marine Microalgae; Kim, S.-K., Ed.; Academic Press: Boston, MA, USA, 2015; Chapter 3; pp. 25–41. [Google Scholar]
- Nasseri, A.T.; Rasoul-Amini, S.; Morowvat, M.H.; Younes, G. Single Cell Protein: Production and Process. Am. J. Food Technol. 2011, 6. [Google Scholar] [CrossRef]
- Chronakis, I.S. Biosolar proteins from aquatic algae. In Developments in Food Science; Doxastakis, G., Kiosseoglou, V., Eds.; Elsevier: Amsterdam, The Netherlands, 2000; Volume 41, pp. 39–75. [Google Scholar]
- García-Garibay, M.; Gómez-Ruiz, L.; Cruz-Guerrero, A.E.; Bárzana, E. Single cell protein|The Algae. In Encyclopedia of Food Microbiology, 2nd ed.; Batt, C.A., Tortorello, M.L., Eds.; Academic Press: Oxford, UK, 2014; pp. 425–430. [Google Scholar]
- Jadeja, R.N.; Tewari, A. Effect of soda ash industry effluent on protein content of two green seaweeds. J. Hazard. Mater. 2008, 151, 559–561. [Google Scholar] [CrossRef]
- Putri, D.; Ulhidayati, A.; Musthofa, I.A.; Wardani, A.K. Single cell protein production of Chlorella sp. using food processing waste as a cultivation medium. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2018; Volume 131, p. 012052. [Google Scholar] [CrossRef]
- Spalvins, K.; Zihare, L.; Blumberga, D. Single cell protein production from waste biomass: Comparison of various industrial by-products. Energy Procedia 2018, 147, 409–418. [Google Scholar] [CrossRef]
- Gressler, V.; Yokoya, N.S.; Fujii, M.T.; Colepicolo, P.; Filho, J.M.; Torres, R.P.; Pinto, E. Lipid, fatty acid, protein, amino acid and ash contents in four Brazilian red algae species. Food Chem. 2010, 120, 585–590. [Google Scholar] [CrossRef]
- Fleurence, J. Seaweed proteins: Biochemical, nutritional aspects and potential uses. Trends Food Sci. Technol. 1999, 10, 25–28. [Google Scholar] [CrossRef]
- Fleurence, J.; Morançais, M.; Dumay, J.; Decottignies, P.; Turpin, V.; Munier, M.; Garcia-Bueno, N.; Jaouen, P. What are the prospects for using seaweed in human nutrition and for marine animals raised through aquaculture? Trends Food Sci. Technol. 2012, 27, 57–61. [Google Scholar] [CrossRef]
- Galland-Irmouli, A.V.; Fleurence, J.; Lamghari, R.; Luçon, M.; Rouxel, C.; Barbaroux, O.; Bronowicki, J.P.; Villaume, C.; Guéant, J.L. Nutritional value of proteins from edible seaweed Palmaria palmata (dulse). J. Nutr. Biochem. 1999, 10, 353–359. [Google Scholar] [CrossRef]
- Morrissey, J.; Kraan, S.; Guiry, M.D. A Guide to Commercially Important Seaweeds on the Irish Coast; Irish Bord Iascaigh Mhara/Irish Sea Fisheries Board: Dublin, Ireland, 2001. [Google Scholar]
- Rupérez, P.; Saura-Calixto, F. Dietary fibre and physicochemical properties of edible Spanish seaweeds. Eur. Food Res. Technol. 2001, 212, 349–354. [Google Scholar] [CrossRef]
- Nuñez, V.c.J.; Voltolina, D.; Nieves, M.; Piña, P.; Medina, A.; Guerrero, M.n. Nitrogen budget in Scenedesmus obliquus cultures with artificial wastewater. Bioresour. Technol. 2001, 78, 161–164. [Google Scholar] [CrossRef]
- Volkmann, H.; Imianovsky, U.; Oliveira, J.; Sant’Anna, E. Cultivation of Arthrospira (spirulina) platensis in desalinator wastewater and salinated synthetic medium: Protein content and amino-acid profile Cultivo de Arthrospira (Spirulina) platensis em rejeito de dessalinizador e meio sintético salinizado: Teor protéico e perfil de aminoácidos. Braz. J. Microbiol. 2008, 39. [Google Scholar] [CrossRef] [Green Version]
- Rouxel, C.; Daniel, A.; Jérôme, M.; Etienne, M.; Fleurence, J. Species identification by SDS-PAGE of red algae used as seafood or a food ingredient. Food Chem. 2001, 74, 349–353. [Google Scholar] [CrossRef]
- Sharif, M.; Zafar, M.H.; Aqib, A.I.; Saeed, M.; Farag, M.R.; Alagawany, M. Single cell protein: Sources, mechanism of production, nutritional value and its uses in aquaculture nutrition. Aquaculture 2021, 531, 735885. [Google Scholar] [CrossRef]
- Takahashi, J.A.; Barbosa, B.V.R.; Martins, B.d.A.; Guirlanda, C.P.; Moura, M.A.F. Use of the Versatility of Fungal Metabolism to Meet Modern Demands for Healthy Aging, Functional Foods, and Sustainability. J. Fungi 2020, 6, 223. [Google Scholar] [CrossRef]
- Demain, A.L.; Fang, A. The Natural Functions of Secondary Metabolites. In History of Modern Biotechnology I; Fiechter, A., Ed.; Springer: Heidelberg, Germany, 2000; pp. 1–39. [Google Scholar] [CrossRef]
- Keller, N.P.; Turner, G.; Bennett, J.W. Fungal secondary metabolism—From biochemistry to genomics. Nat. Rev. Microbiol. 2005, 3, 937–947. [Google Scholar] [CrossRef]
- Dufossé, L.; Fouillaud, M.; Caro, Y.; Mapari, S.A.; Sutthiwong, N. Filamentous fungi are large-scale producers of pigments and colorants for the food industry. Curr. Opin. Biotechnol. 2014, 26, 56–61. [Google Scholar] [CrossRef]
- Hoffmeister, D.; Keller, N.P. Natural products of filamentous fungi: Enzymes, genes, and their regulation. Nat. Prod. Rep. 2007, 24, 393–416. [Google Scholar] [CrossRef] [PubMed]
- Kohlhaw, G.B. Leucine biosynthesis in fungi: Entering metabolism through the back door. Microbiol. Mol. Biol. Rev. 2003, 67, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schardl, C.L.; Young, C.A.; Hesse, U.; Amyotte, S.G.; Andreeva, K.; Calie, P.J.; Fleetwood, D.J.; Haws, D.C.; Moore, N.; Oeser, B.; et al. Plant-Symbiotic Fungi as Chemical Engineers: Multi-Genome Analysis of the Clavicipitaceae Reveals Dynamics of Alkaloid Loci. PLoS Genet. 2013, 9, e1003323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brakhage, A.A. Regulation of fungal secondary metabolism. Nat. Rev. Microbiol. 2013, 11, 21–32. [Google Scholar] [CrossRef]
- Ravinder, R.; Linga, V.; Ravindra, P. Studies on Aspergillus oryzae Mutants for the Production of Single Cell Proteins from Deoiled Rice Bran. Food Technol. Biotechnol. 2003, 41, 243–246. [Google Scholar]
- Turnbull, W.H.; Leeds, A.R.; Edwards, D.G. Mycoprotein reduces blood lipids in free-living subjects. Am. J. Clin. Nutr. 1992, 55, 415–419. [Google Scholar] [CrossRef]
- Nangul, A.; Bhatia, R. Microorganisms: A marvelous source of single cell proteins. J. Microbiol. Biotechnol. Food Sci. 2013, 3, 15–18. [Google Scholar]
- Valentino, M.J.; Ganado, L.; Undan, J.R. Single cell protein potential of endophytic fungi associated with bamboo using rice bran as substrate. Adv. Appl. Sci. Res. 2016, 7, 68–72. [Google Scholar]
- Ahmadi, A.R.; Ghoorchian, H.; Hajihosaini, R.; Khanifar, J. Determination of the amount of protein and amino acids extracted from the microbial protein (SCP) of lignocellulosic wastes. Pak. J. Biol. Sci. 2010, 13, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, J.; Ferraz, A.; Nogueira, R.F.; Ferrer, I.; Esposito, E.; Durán, N. Lignin biodegradation by the ascomycete Chrysonilia sitophila. Appl. Biochem. Biotechnol. 1997, 62, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, T.C.; Joshi, M. Protein enrichment of apple pomace by co-culture of cellulolytic moulds and yeasts. World J. Microbiol. Biotechnol. 1994, 10, 116–117. [Google Scholar] [CrossRef]
- Baldensperger, J.; Le Mer, J.; Hannibal, L.; Quinto, P.J. Solid state fermentation of banana wastes. Biotechnol. Lett. 1985, 7, 743–748. [Google Scholar] [CrossRef]
- De Gregorio, A.; Mandalari, G.; Arena, N.; Nucita, F.; Tripodo, M.M.; Lo Curto, R.B. SCP and crude pectinase production by slurry-state fermentation of lemon pulps. Bioresour. Technol. 2002, 83, 89–94. [Google Scholar] [CrossRef]
- Chiou, P.W.S.; Chiu, S.W.; Chen, C.R. Value of Aspergillus niger fermentation product as a dietary ingredient for broiler chickens. Anim. Feed Sci. Technol. 2001, 91, 171–182. [Google Scholar] [CrossRef]
- Wiebe, M.G. Myco-protein from Fusarium venenatum: A well-established product for human consumption. Appl. Microbiol. Biotechnol. 2002, 58, 421–427. [Google Scholar] [CrossRef]
- Şişman, T.; Gür, Ö.; Doğan, N.; Özdal, M.; Algur Ö, F.; Ergon, T. Single-cell protein as an alternative food for zebrafish, Danio rerio: A toxicological assessment. Toxicol. Ind. Health 2013, 29, 792–799. [Google Scholar] [CrossRef]
- Klug, L.; Daum, G. Yeast lipid metabolism at a glance. FEMS Yeast Res. 2014, 14, 369–388. [Google Scholar] [CrossRef] [Green Version]
- Sitepu, I.; Selby, T.; Lin, T.; Zhu, S.; Boundy-Mills, K. Carbon source utilization and inhibitor tolerance of 45 oleaginous yeast species. J. Ind. Microbiol. Biotechnol. 2014, 41, 1061–1070. [Google Scholar] [CrossRef] [Green Version]
- Stincone, A.; Prigione, A.; Cramer, T.; Wamelink, M.M.; Campbell, K.; Cheung, E.; Olin-Sandoval, V.; Grüning, N.M.; Krüger, A.; Tauqeer Alam, M.; et al. The return of metabolism: Biochemistry and physiology of the pentose phosphate pathway. Biol. Rev. Camb. Philos. Soc. 2015, 90, 927–963. [Google Scholar] [CrossRef] [Green Version]
- Kieliszek, M.; Kot, A.; Bzducha-Wróbel, A.; Błażejak, S.; Gientka, I.; Kurcz, A. Biotechnological use of Candida yeasts in the food industry: A review. Fungal Biol. Rev. 2017, 31, 185–198. [Google Scholar] [CrossRef]
- Bennett, J.W.; Keller, N.P. Mycotoxins and their prevention. In Fungal Biotechnology; Chapman & Hall: Weinheim, Germany, 1997; pp. 265–271. [Google Scholar]
- Aggelopoulos, T.; Katsieris, K.; Bekatorou, A.; Pandey, A.; Banat, I.M.; Koutinas, A.A. Solid state fermentation of food waste mixtures for single cell protein, aroma volatiles and fat production. Food Chem. 2014, 145, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Yadav, J.S.; Bezawada, J.; Ajila, C.M.; Yan, S.; Tyagi, R.D.; Surampalli, R.Y. Mixed culture of Kluyveromyces marxianus and Candida krusei for single-cell protein production and organic load removal from whey. Bioresour. Technol. 2014, 164, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Li, D.; Liu, Y. Production of single cell protein from soy molasses using Candida tropicalis. Ann. Microbiol. 2012, 62, 1165–1172. [Google Scholar] [CrossRef]
- Pessoa, A.; Mancilha, I.M.; Sato, S. Cultivation of Candida tropicalis in sugar cane hemicellulosic hydrolyzate for microbial protein production. J. Biotechnol. 1996, 51, 83–88. [Google Scholar] [CrossRef]
- Zhao, G.; Zhang, W.; Zhang, G. Production of single cell protein using waste capsicum powder produced during capsanthin extraction. Lett. Appl. Microbiol. 2010, 50, 187–191. [Google Scholar] [CrossRef]
- Jalasutram, V.; Kataram, S.; Gandu, B.; Anupoju, G.R. Single cell protein production from digested and undigested poultry litter by Candida utilis: Optimization of process parameters using response surface methodology. Clean Technol. Environ. Policy 2013, 15, 265–273. [Google Scholar] [CrossRef]
- Kurcz, A.; Błażejak, S.; Kot, A.M.; Bzducha-Wróbel, A.; Kieliszek, M. Application of Industrial Wastes for the Production of Microbial Single-Cell Protein by Fodder Yeast Candida utilis. Waste Biomass Valorization 2018, 9, 57–64. [Google Scholar] [CrossRef] [Green Version]
- Hashem, M.; Hesham, A.E.-L.; Alamri, S.A.; Alrumman, S.A. Production of single-cell protein from wasted date fruits by Hanseniaspora uvarum KKUY-0084 and Zygosaccharomyces rouxii KKUY-0157. Ann. Microbiol. 2014, 64, 1505–1511. [Google Scholar] [CrossRef]
- Paraskevopoulou, A.; Athanasiadis, I.; Kanellaki, M.; Bekatorou, A.; Blekas, G.; Kiosseoglou, V. Functional properties of single cell protein produced by kefir microflora. Int. Food Res. J. 2003, 36, 431–438. [Google Scholar] [CrossRef]
- Duarte, L.C.; Carvalheiro, F.; Lopes, S.; Neves, I.; Gírio, F.M. Yeast Biomass Production in Brewery’s Spent Grains Hemicellulosic Hydrolyzate. Appl. Biochem. Biotechnol. 2008, 148, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Wang, Q.; Zhang, F.; Zhang, S.-C.; Chi, Z.-M.; Madzak, C. Direct conversion of inulin into single cell protein by the engineered Yarrowia lipolytica carrying inulinase gene. Process. Biochem. 2011, 46, 1442–1448. [Google Scholar] [CrossRef]
- Wu, G.; Fanzo, J.; Miller, D.D.; Pingali, P.; Post, M.; Steiner, J.L.; Thalacker-Mercer, A.E. Production and supply of high-quality food protein for human consumption: Sustainability, challenges, and innovations. Ann. N. Y. Acad. Sci. 2014, 1321, 1–19. [Google Scholar] [CrossRef]
- Boland, M.J.; Rae, A.N.; Vereijken, J.M.; Meuwissen, M.P.M.; Fischer, A.R.H.; van Boekel, M.A.J.S.; Rutherfurd, S.M.; Gruppen, H.; Moughan, P.J.; Hendriks, W.H. The future supply of animal-derived protein for human consumption. Trends Food Sci. Technol. 2013, 29, 62–73. [Google Scholar] [CrossRef]
- Linder, T. Making the case for edible microorganisms as an integral part of a more sustainable and resilient food production system. Food Secur. 2019, 11, 265–278. [Google Scholar] [CrossRef] [Green Version]
- Finnigan, T.; Needham, L.; Abbott, C. Mycoprotein: A Healthy New Protein With a Low Environmental Impact. In Sustainable Protein Sources; Nadathur, S.R., Wanasundara, J.P.D., Scanlin, L., Eds.; Academic Press: San Diego, CA, USA, 2017; Chapter 19; pp. 305–325. [Google Scholar] [CrossRef]
- Bogdahn, I. Agriculture-independent, sustainable, fail-safe and efficient food production by autotrophic single-cell protein. PeerJ PrePrints 2015, 3, e1279v1273. [Google Scholar] [CrossRef]
- Ferreira, I.M.P.L.V.O.; Pinho, O.; Vieira, E.; Tavarela, J.G. Brewer’s Saccharomyces yeast biomass: Characteristics and potential applications. Trends Food Sci. Technol. 2010, 21, 77–84. [Google Scholar] [CrossRef]
- Yousufi, M.K. To determine protein content of single cell protein produced by using various combination of fruit wastes and two standard food fungi. Int. J. Adv. Biotechnol. Res. 2012, 3, 533–536. [Google Scholar]
- Anupama; Ravindra, P. Value-added food:: Single cell protein. Biotechnol. Adv. 2000, 18, 459–479. [Google Scholar] [CrossRef]
- Zhou, Y.-M.; Chen, Y.-P.; Shen, Y. Single cell protein-feed: Taking orange waste as raw material for fermentation. In Advanced Materials and Energy Sustainability; World Scientific: Singapore, 2017; pp. 323–335. [Google Scholar] [CrossRef]
- Aruna, T.E.; Aworh, O.C.; Raji, A.O.; Olagunju, A.I. Protein enrichment of yam peels by fermentation with Saccharomyces cerevisiae (BY4743). Ann. Agric. Sci. 2017, 62, 33–37. [Google Scholar] [CrossRef]
- Adedayo, M.R.; Ajiboye, E.A.; Akintunde, J.K.; Odaibo, A. Single Cell Proteins: As Nutritional Enhancer. Adv. Appl. Sci. Res. 2011, 2, 396–409. [Google Scholar]
- Bhalla, T.; Sharma, N.N.; Sharma, M. Production of Metabolites, Industrial enzymes, Amino acid, Organic acids, Antibiotics, Vitamins and Single Cell Proteins. J. Environ. 2007, 6, 34–78. [Google Scholar]
- Jamal, P.; Alam, M.; Salleh, N. Medai optimization for bioproteins productions from cheaper carbon source. J. Eng. Sci. Technol. 2008, 3, 124–130. [Google Scholar]
- Mahajan, A.; Dua, S. A perspective on biotechnological potential. J. Food Sci. Technol. 1995, 32, 162–165. [Google Scholar]
- McEvoy, L.A.; Navarro, J.C.; Hontoria, F.; Amat, F.; Sargent, J.R. Two novel Anemia enrichment diets containing polar lipid. Aquaculture 1996, 144, 339–352. [Google Scholar] [CrossRef]
- Olvera-Novoa, M.A.; MartÍNez-Palacios, C.A.; Olivera-Castillo, L. Utilization of torula yeast (Candida utilis) as a protein source in diets for tilapia (Oreochromis mossambicus Peters) fry. Aquac. Nutr. 2002, 8, 257–264. [Google Scholar] [CrossRef]
- Hardy, R.W.; Patro, B.; Pujol-Baxley, C.; Marx, C.J.; Feinberg, L. Partial replacement of soybean meal with Methylobacterium extorquens single-cell protein in feeds for rainbow trout (Oncorhynchus mykiss Walbaum). Aquac. Res. 2018, 49, 2218–2224. [Google Scholar] [CrossRef]
- Oliva-Teles, A.; Gonçalves, P. Partial replacement of fishmeal by brewers yeast (Saccaromyces cerevisae) in diets for sea bass (Dicentrarchus labrax) juveniles. Aquaculture 2001, 202, 269–278. [Google Scholar] [CrossRef]
- Tovar, D.; Zambonino, J.; Cahu, C.; Gatesoupe, F.J.; Vázquez-Juárez, R.; Lésel, R. Effect of live yeast incorporation in compound diet on digestive enzyme activity in sea bass (Dicentrarchus labrax) larvae. Aquaculture 2002, 204, 113–123. [Google Scholar] [CrossRef]
- Tesfaw, A.; Assefa, F. Co-culture: A great promising method in single cell protein production. Biotechnol. Mol. Biol. Rev. 2014, 9, 12–20. [Google Scholar] [CrossRef]
- Protein Extracts from Single Cell Protein and Other Conventional Sources Market Analysis By Sources (Plant, Bacteria, Yeast, Algae, Fungi), By Applications (Pharma, Animal Feed {Isolate, Concentrate, Hydrolysate}, Agriculture), And Segment Forecasts, 2018–2025. Available online: https://www.grandviewresearch.com/industry-analysis/protein-extracts-from-single-cell-protein-other-conventional-sources-market (accessed on 25 June 2021).
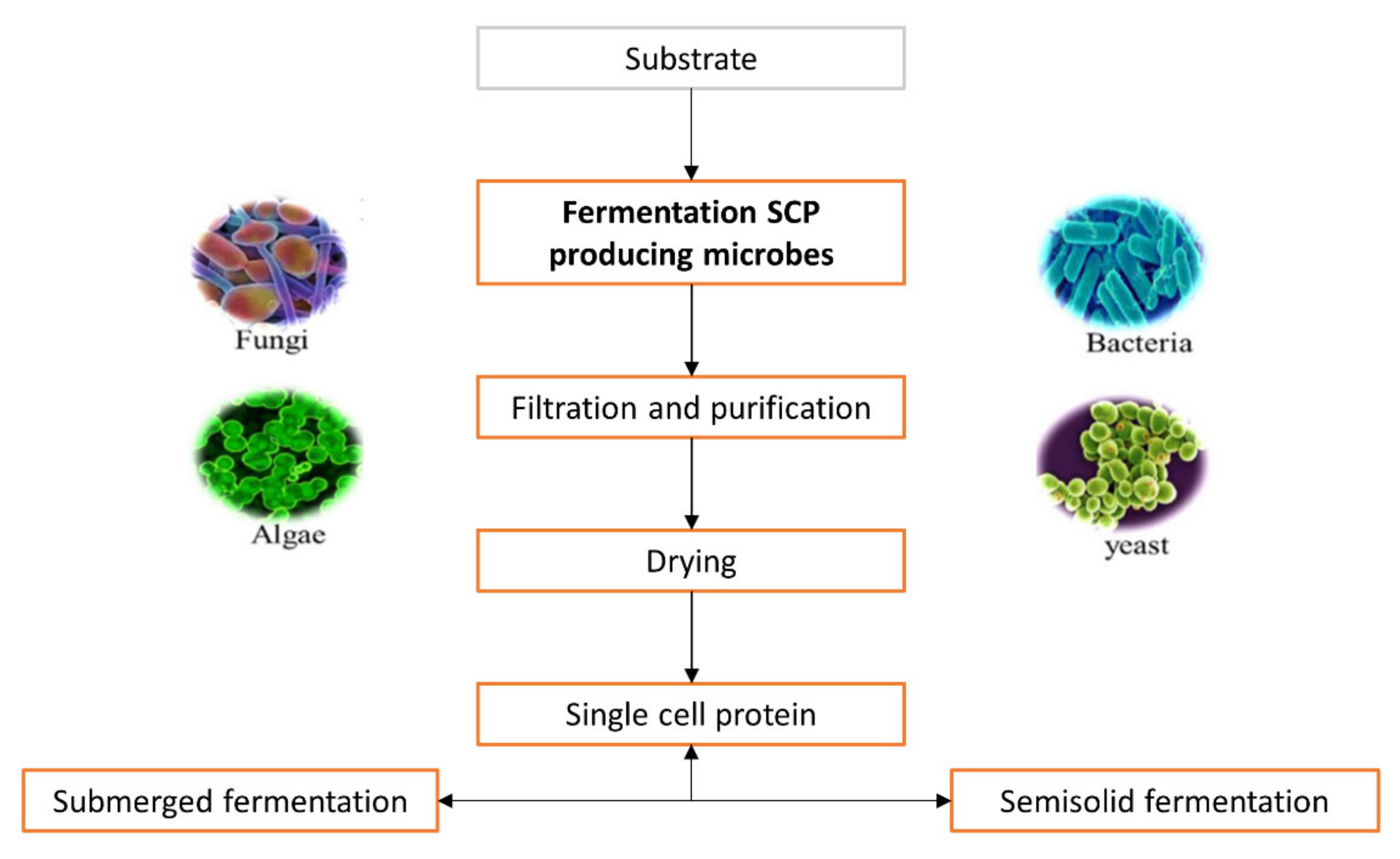
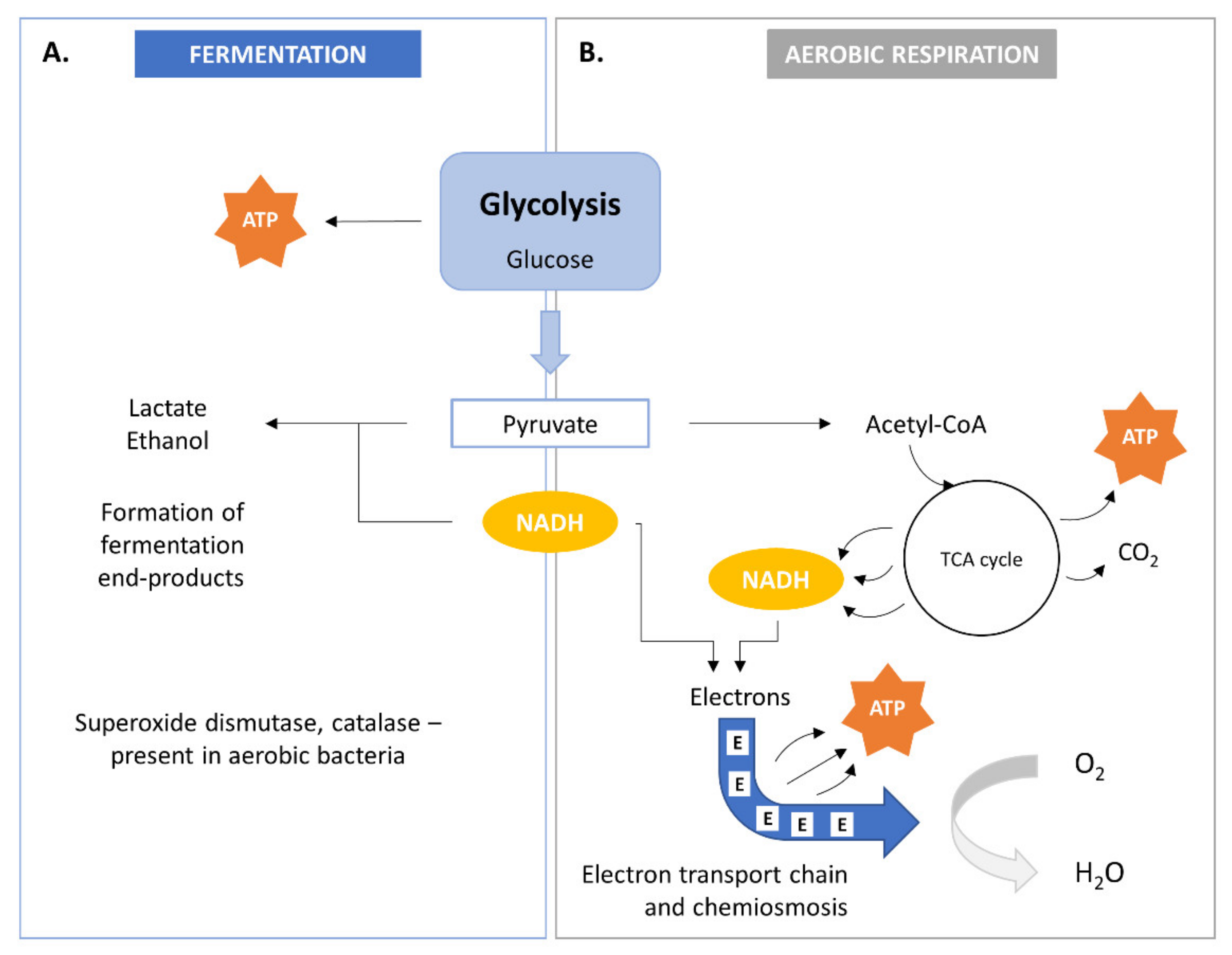
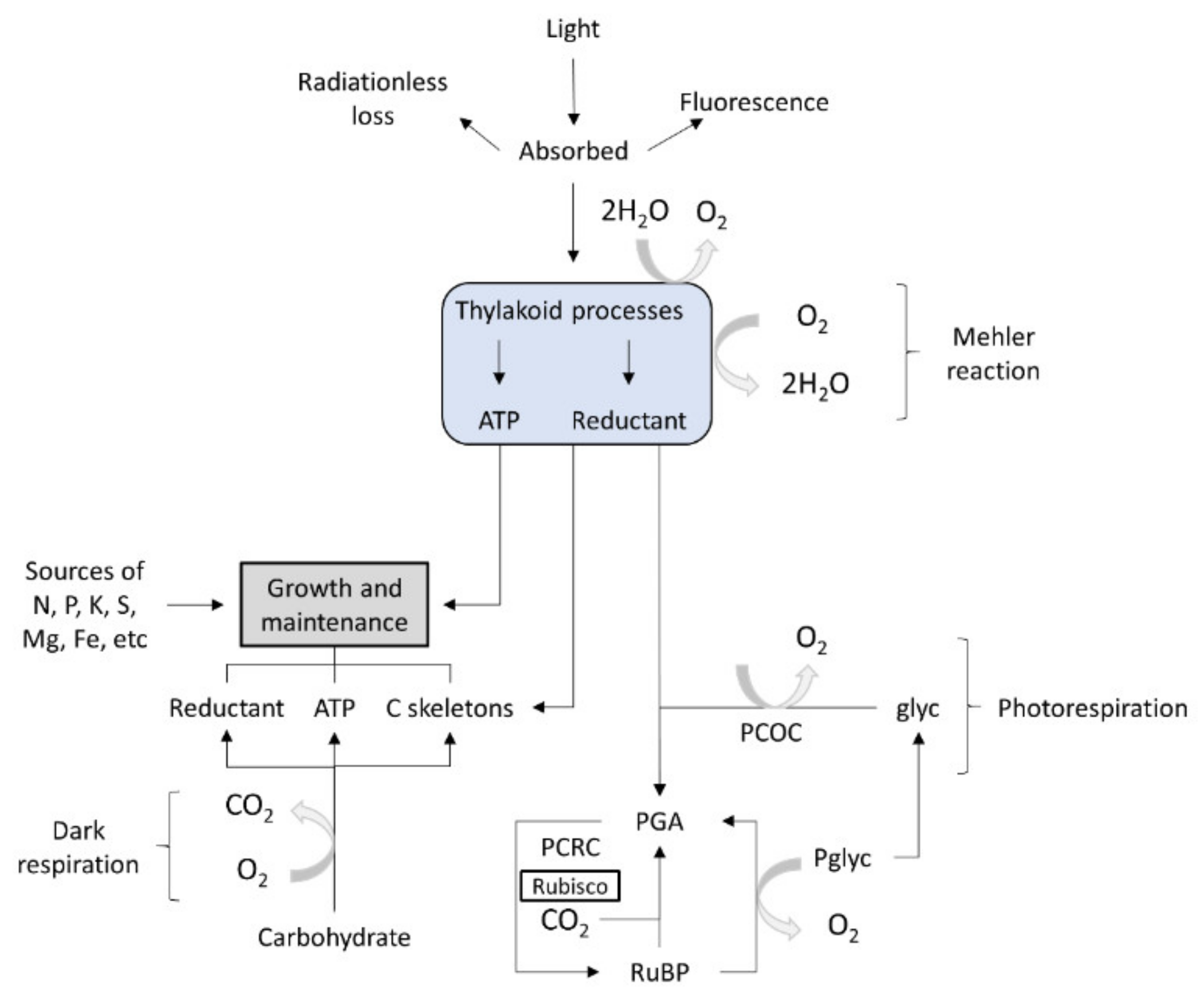
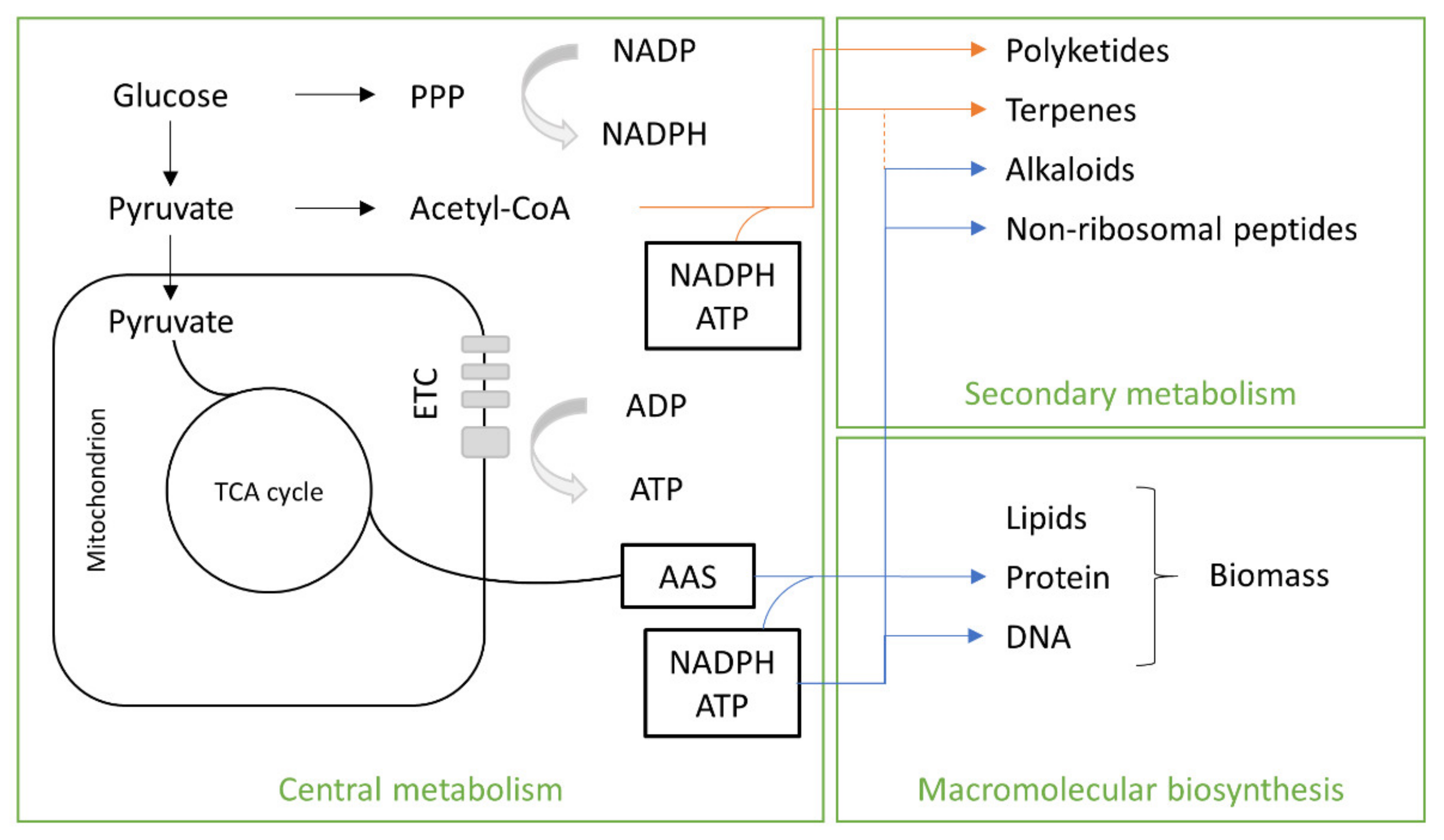
| Bacteria | Substrate | SCP (%) | Reference |
|---|---|---|---|
| Afifella marina STW181 | Commercial shrimp feed | >46 | [27] |
| Bacillus cereus | Ram horn | 68 | [28] |
| Bacillus licheniformis | Potato starch processing waste | 38 | [29] |
| Bacillus pumilis | Potato starch processing waste | 46 | [30] |
| Bacillus subtilis | Ram horn | 71 | [28] |
| Bacillus subtilis sp | Soybean hull | 26 | [31] |
| Corynebacterium ammoniagenes | Glucose + fructose | 61 | [32] |
| Cupriavidus necator | Synthetic growth medium | 40–46 | [33] |
| Escherichia coli | Ram horn | 66 | [28] |
| Haloarcula sp. IRU1 | Petrochemical wastewater | 76 | [34] |
| Methylococcus capsulatus, Ralstonia sp., Brevibacillus agri, Aneurinibacillus sp. | Methane (natural gas) | 67–73 | [25] |
| Methylomonas and Methylophilus spp. Metilococ capsulatus | Gas and liquid products of sewage | <41 | [19] |
| Methylophilus methylotrophus | Methanol | 81 | [25] |
| Methylococcus capsulatus (bath) | Methane | 53 | [35] |
| Methylophilus methylotrophus | Methanol | <70 | [3] |
| Methylophilus sp | Supernatant and biogas | 24 | [19] |
| Methylocapsa acidiphila | Methane | 59 | [36] |
| Methilomonas.sp | Natural gas | 69.3 | [37] |
| Methilomonas.sp | Biogas and supernatant of sewage sludge | 56 | [19] |
| Rhizospheric diazotrophs (whole microbial community | Brewery wastewater | >55 | [38] |
| Rhodopseudomonas blastica | Wastewater from a latex rubber | 66.7 | [39] |
| Rhodopseudomonas palustris | Sludge and sago starch processing | 72–74 | [40] |
| Rhodocyclus gelatinosus | Poultry slaughterhouse wastewater | 67.6 | [41] |
| Rhodobacter sphaeroides P47 | Pineapple waste | 66.6 | [42] |
| Rhodopseudomon as palustris P1 | Fermented pineapple extract | 65 | [39] |
| Rhodobacter sphaeroides Z08 | Soyabean wastewater | 52 | [43] |
| Rhodovulvum sulphidophilum | Glutamate malate medium | 15,6 | [44] |
| Rhodocyclus gelatinosus | Pig farm waste | 50.6 | [45] |
| Rhodopseudomonas and R. fulvum | Sugar refinery wastewater | 58 | [46] |
| Rhodocyclus gelatinosus | Miso-like effluent medium | 63 | [47] |
| Rhodobacter sphaeroides P47 | Dehydrated medium from pineapple peel waste | 66.6 | [42] |
| Rhodocyclus gelatinosus | Seafood processing wastewater | 50 | [48] |
| Rhodopseudomonas sp. | Synthetic medium | 11 | [49] |
| Rhodopseudomonas palustris Rhodopseudomonas sp. | Wastewater from noodle production | 50 | [50] |
| Rhodopseudomonas sp. CSK01 | Municipal wastewater | 60.1 | [51] |
| Rhodocyclusg elatinosus R7 | Tuna condensate | 56 | [52] |
| Rhodobacter capsulatus | Synthetic medium | 45 | [53] |
| Rhodopseudomonas acidophila | Synthetic medium | 23 | [54] |
| Rhodocyclus gelatinosus | Cassava waste | 56 | [55] |
| Rhodopseudomonas palustris | Simulated wastewater | 45 | [56] |
| Rhodopseudomonas palustris | Photosynthetic sludge | 74 | [57] |
| Fungi | Substrate | SCP (%) | Reference |
|---|---|---|---|
| Cladosporium cladosporioides | Rice bran | 10% | [98] |
| Penicillium citrinum | Rice bran | 10% | [98] |
| Pleurotus florida | Wheat straw | 63% | [99] |
| Chrysonilia sitophilia | Lignin | 39% | [100] |
| Aspergillus flavus | Rice bran | 10% | [98] |
| Aspergillus niger | Apple pomace | 17–20% | [101] |
| Aspergillus niger | Banana waste | 18% | [102] |
| Aspergillus niger | Rice bran | 11% | [98] |
| Aspergillus niger | Citrus pulp | 25.6% | [103] |
| Aspergillus niger | Potato starch processing waste | 38% | [29,30] |
| Aspergillus niger | Waste liquor | 50% | [104] |
| Aspergillus ochraceus | Rice bran | 10% | [98] |
| Aspergillus oryzae | Rice bran (deoiled) | 24% | [95] |
| Fusarium semitectum and sp1 and sp2 | Rice bran | 10% | [98] |
| Fusarium venenatum | Glucose (Product: QuornTM) | 44% | [105] |
| Monascus ruber | Rice bran | 9% | [98] |
| Trichoderma harzianum | Cheese whey filtrate | 34% | [106] |
| Trichoderma virideae | Citrus pulp | 32 | [103] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bratosin, B.C.; Darjan, S.; Vodnar, D.C. Single Cell Protein: A Potential Substitute in Human and Animal Nutrition. Sustainability 2021, 13, 9284. https://doi.org/10.3390/su13169284
Bratosin BC, Darjan S, Vodnar DC. Single Cell Protein: A Potential Substitute in Human and Animal Nutrition. Sustainability. 2021; 13(16):9284. https://doi.org/10.3390/su13169284
Chicago/Turabian StyleBratosin, Bogdan Constantin, Sorina Darjan, and Dan Cristian Vodnar. 2021. "Single Cell Protein: A Potential Substitute in Human and Animal Nutrition" Sustainability 13, no. 16: 9284. https://doi.org/10.3390/su13169284
APA StyleBratosin, B. C., Darjan, S., & Vodnar, D. C. (2021). Single Cell Protein: A Potential Substitute in Human and Animal Nutrition. Sustainability, 13(16), 9284. https://doi.org/10.3390/su13169284







