Abstract
The suitability of replacing mineral aggregate with carbon-negative ones mainly depends on the properties of the aggregates produced from waste recycling, reducing CO2 emissions. This study aimed to investigate the predictive approaches adapted to concrete mixtures where mineral aggregates are replaced by carbonated aggregates (at different substitution rates from 25 to 100% with aggregates of various origins). A large experimental campaign of aggregates and carbonated aggregate concretes highlighted their physical, mechanical, thermal and hygric properties and the influence of density and porosity of aggregates on these properties. Thanks to these results, predictive approaches were formulated to establish the main engineering properties: mechanical compressive strength, elasticity modulus, thermal conductivity, thermal mass capacity and hygric diffusivity. These empirical and analytical models were based on the density of aggregates. Maximum deviations of around 15% were obtained with the experimental data, highlighting the influence of grain density on carbonated aggregate concretes. These models could then be used to optimize the formulation of concrete mixtures with carbonated aggregates, replacing international standards adapted to mineral aggregates.
1. Introduction
To meet environmental challenges, one of the current avenues is the valorization of industrial by-products from construction, which is the largest consumer of mineral aggregates. The volume of industrial waste generated and then sent to storage and landfill sites is limited [1]. Indeed, waste from industry can currently be recovered in waste incineration plants in order to use its low entropy to satisfy our energy requirements. Unfortunately, this solution generates waste, bottom ash and fly ash, which are buried in class 3 centers (inert waste). A large volume of waste generates leachates which contain toxic substances (heavy metals and organic compounds …) [2]. Landfilling accelerates soil pollution and covers large areas. Thus, the reuse of these industrial by-products would reduce waste production [3,4,5,6,7,8], while ensuring sufficient sanitary conditions for the construction industry. Another benefit would be the reduction in the consumption of natural resources [9]. The construction sector is the most important consumer of aggregates, the extraction of which is one of the main challenges in the management of non-renewable resources [10]. These non-renewable resources could be, in part, replaced by recycled or bio-based aggregates, to meet the requirements of sustainability [11]. A last benefit would be to improve the energy performance of building envelopes. For that purpose, building materials based on these aggregates must have hygrothermal characteristics that enhance the energy performance of walls [12] and contribute to the comfort of the occupants [13].
Several approaches are proposed in order to predict the main mechanical performances of concretes regarding the properties of the components of cementitious mixture [14,15,16,17,18,19,20]. These approaches were developed using the experimental results of usual and lightweight concrete, which makes them unsuitable for other special concretes. Using carbonated aggregates as a replacement for normal ones improves concrete’s thermal properties (conductivity, diffusivity and specific heat capacity) [21]. In order to predict the thermal conductivity for porous materials, there are different theoretical approaches. The most widely used, in the case of building materials, is the model by the auto-coherent homogenization ACH [12,22,23,24,25]. The notion of occupant well-being cannot be limited to thermal properties. For these purposes, the identification of the main hygric characteristics of carbonated aggregate concretes is conducted. A simplified theoretical approach of hygric properties (hygric effusivity and diffusivity) is defined [26] to predict the ability of the materials to take up and store moisture.
Thus, in order to encourage designers and engineers to recommend carbonated aggregates, simplified models have to be proposed to predict the main mechanical, thermal and hygric performances of concretes. This study focuses on the influence of carbonated aggregated density on the most significant properties of concretes. To achieve this goal, after a short presentation of carbonated aggregates, a large experimental campaign was presented to identify the properties of aggregates and concretes. These results are then used to formulate some predictive approaches and models based on the current properties of present phases and on the composition of the mixture.
2. Materials and Methods
2.1. Carbonation Aggregates and Environmental Impact
New treatment methods make it possible to transform industrial waste into co-products in order to reduce landfill volumes and reduce the use of non-renewable raw materials. Despite increased recycling, large quantities of waste are not effectively reused, including incineration residues which are often classified as hazardous due to their alkalinity and heavy metal content. One solution is the use of accelerated carbonation technology (ACT) to permanently bind CO2 into solid carbon by using materials that are naturally reactive, such as waste from thermal processes, due to their high calcium and magnesium content [27]. The accelerated carbonation technology consists of several steps: after being transported and stored at the production facility, the incineration by-products are pre-treated by carbonation and then mixed with cement, sand and water in a carbon dioxide-enriched atmosphere to form a binding paste. The materials are then carbonated in a rotary reactor, forming lightweight aggregates by aggregation around the sand grains. The aggregates produced are then stored and further enriched with CO2.
Carbonation is obtained by waste exposition to a high concentration of carbon dioxide gas in a controlled environment. The exothermic reaction occurs in the presence of water and gas hydrates of CO2 to form carbonic acid. CO2 is first diffused into the solid and then solvated into the pores. In lime-containing materials, such as some wastes, lime hydrates to form calcium hydroxides, thus increasing their reactivity. Then, there is the nucleation of the hydrated calcium silicates CSH and precipitation of the calcium carbonates CaCO3 which act as a binder between the particles. The carbonation in the cementitious matrix presence produces C3S andC2S calcium silicates, which interact with carbon dioxide and water due to their hydrophilic character. The reaction will produce CaCO3 which crystallizes into calcite, a stable polymorph under normal conditions, and CSH, a non-crystalline compound [28]. A second carbonation therefore occurs through the decalcification of the CSH to form hydrates and CaCO3:
C3S + yH2O + (3−x)CO2 → CxSHy + (3−x)CaCO3
+zCO2 → C(x−z)SHy + zCaCo3
+zCO2 → C(x−z)SHy + zCaCo3
These necessary carbonation phases are usually found in many thermal residues and can combine with large amounts of gaseous CO2 during a short period of time [27]. The entanglement of the CxSHy gel governs the aggregates’ strength and participates in inerting the aggregates, thus ensuring the chemical stability of the aggregates. Currently, more than 150 different waste streams were subjected to the accelerated carbonation technology and three different ash supplies have been selected: residues of air-pollution control residues (APCr) ash (C8agg), paper ash (PA) and biomass ash (BAk). Previously dried in an oven, the ashes are then moistened with 10% water and carbonated in a rotating drum under a CO2-enriched atmosphere and at a constant pressure of 2 bars during 24 h. The process is repeated until a constant mass is obtained, ensuring a saturation in carbonate solution.
A life cycle assessment (LCA) of carbonated aggregates has an excellent environmental performance level due to their ability to store CO2 during the production process. The environmental impact of carbonated aggregates is assessed according to nine indicators defined by EN 15804. As shown in Table 1, carbonated aggregate production leads to a strong reduction in the eutrophication indicator (−99%) and a 50% decrease in the indicators of soil and water acidification, photochemical ozone formation, resource depletion (elements and fossil fuels) and water pollution. These aggregates also show a negative carbon balance (close to −50 kgCO2/t).

Table 1.
Environmental impact indicators of mineral and carbonated aggregates.
2.2. Multi-Scale Characterization Methodology
2.2.1. Aggregate Scale
Carbonated aggregates processed from APCr ash (C8agg), paper ash (PA) and biomass ash (BAk) are characterized by standardized methods:
- The identification of granulometry by the sieve method, under dry conditions, according to EN 933-1. The expressions of the differential distributions and cumulative distributions are given by Equation (1), from the amount of material retained in each sieve’s i:The granular class of the aggregates is deduced from the interpretation of the granulometric curves, based on the following acceptability criteria:The granular range can be quantified using the uniformity coefficient and the curve coefficient , whose expressions are:where the percentiles, expressed in mm, are deduced from the granulometric curve. The particle size dispersion is translated by the Trask sorting index and the Folk and Ward skewness index , for which the quartiles are the of the inverse of the percentiles :The symmetry of the granulometric curves is approximated by the Krumbein asymmetry index defined as follows:
- Measurement of bulk, true and absolute densities by the pycnometric method using toluene in order to limit the risk of mineral dissolution (toluene is a non-polar solvent);
- Calculation of total, interparticle and intraparticle porosities using the following equation system:
- Identification of the crush-resistance through a Shimadzu electro-mechanical press with a nominal capacity of 250 kN, equipped with a hollow cylindrical piston (100 mm height and 200 mm2 cross-section), according to EN 13055-1.
2.2.2. Concrete Scale
Concrete specimens are made with a cementitious binder CEM II/B M(L-S) 32.5 R CE NF by varying the substitution rates of mineral aggregates by carbonated aggregates from 0 to 100% with a 25% step. Special attention is paid to the quantity of water introduced during mixing regarding the high capacity of carbonated aggregates to absorb water, which could influence the quality of the mixture’s hydration [29]. An efficient water-to-cement ratio of 0.7 was considered. A 0/5 sand with fineness modulus of 2.5 was used. This sand has a specific density of 2.5. Mineral aggregates are crushed gravel 5/25 with a bulk density of 1150 kg/m3 and a specific density of 2650 kg/m3.
Before mixing the various components for 15 min, aggregates were pre-wetted using the amount of water necessary for their absorption capacity. This amount is equivalent to the water absorbed after 30 min of immersion. The aggregates therefore achieve a saturated surface-dry (SSD) condition. Then, natural sand and cement were added. Finally, the mixing water was slowly added and mixed for 4 min. The prepared testing specimens were demolded after 24 h and stored in a chamber with relative humidity ranging from 60 to 80% and a constant temperature of 23 °C [29].
Characterization tests allow the identification of the main mechanical properties: uniaxial compression tests were carried out using a Shimadzu electro-mechanical press with a 250 kN nominal capacity. Each batch test, consisting of three samples of size cm3 was solicited in simple compression between two parallel plates, hinged at the top. Three potentiometers were used to determine the sample deformation. Tests were performed at 7 days, 28 days, 90 days and one year for each mix of concrete.
A thermal characterization campaign allows the evaluation of the thermal performance of concretes: guarded hot plate (GHP) apparatus is used in order to determine the thermal conductivity. A data logger records the temperature of the cold and hot plates in the central measuring area ( cm2). A thermopile that measures temperatures in the central and guard zones ensures that the heat flow generated is unidirectional. Samples are pre-dried in a controlled oven at 70 °C, until constant mass is reached. The tests are carried out at average temperatures of 10 °C and a temperature gradient between the hot and cold plates of 10 °C.
A non-steady state method can lead to the determination of several thermal characteristics: for this fluxmetric method, one side of the sample in isothermal condition is subjected to a gradient of temperature. The sample is stabilized at a constant temperature of 15 °C using two thermostatic baths connected to two exchange plates. A temperature gradient of 10 °C is then applied to the upper surface (hot plate). Lateral heat losses are limited by an insulation around the periphery of the sample.
The ability of material to absorb ambient water vapor was identified using sorption isotherms. The adsorption and desorption tests were obtained by using desiccators placed in a thermo-regulated room. Five different levels of relative humidity are generated thanks to specific salts: magnesium chloride, magnesium nitrate, sodium chloride, alumina sulphate and potassium sulphate, which control relative humidities of 33%, 55%, 75%, 81% and 97%, respectively, at 23 °C. The samples used are ( cm3) specimens. The wet mass of each sample is regularly measured until it stabilizes, which is a sign of moisture equilibrium.
Finally, a final parameter symbolizing the capacity of a material to regulate the relative humidity can be retained: the moisture buffer value (). The test protocol consists of monitoring the evolution of the wet mass of a sample subjected to moisture solicitation at constant temperature. The cycles of humidity variations follow the protocol defined by the NORDTEST Moisture Buffering Project [30,31,32], i.e., 8 h at 75% relative humidity and 16 h at 33% relative humidity, with a constant temperature of 23 °C. Samples cm3 of each mix are placed in a temperature and relative humidity controlled chamber (Bia Climatic) and regular weighing (ADAM Nimbus analytical balances) allows the material kinetics to be identified.
3. Experimental Identification of Multi-Physic Properties
3.1. Physico-Mechanical Properties
The sieve size analysis of the three carbonated aggregates is presented in Figure 1). The analysis of the physico-morphological properties of aggregates, summarized in Table 2, proves the possibility of replacing all or part of the mineral aggregates when making concrete. Indeed, carbonated aggregates show a similar size distribution to mineral ones. The grading indices show a proportionate () and fairly well-sorted () granular distribution, with a high symmetry between the proportions of fine and coarser elements (). The uniformity and curvature coefficients show that the grain size is well graded and very tight. The granular class, the shape of the curve and sorting indices are very close to those of mineral aggregates. Thus, the morphology of carbonated aggregates does not imply significant differences on concrete behavior. Due to the manufacturing process, all grains have a similar shape (round, governed by the mixing in a rotary reactor) and the grain size analysis tends to demonstrate a similar grain arrangement in concrete regardless of the aggregates.
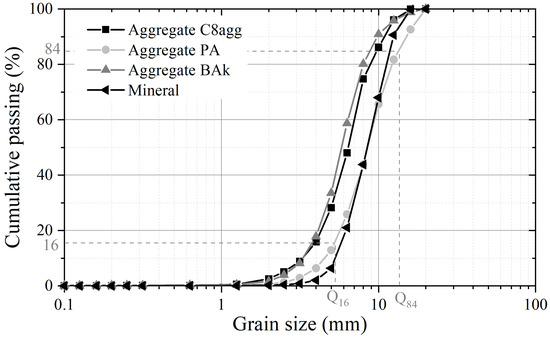
Figure 1.
Gradation curves for carbonated aggregates.

Table 2.
Aggregates physico-morphological characteristics.
The aggregate bulk density is significantly lower than that of a mineral aggregate (close to kg/m3) and is governed by a high total porosity. The intraparticle porosity is much higher than that of a mineral aggregate (+200% to +400%). Crush-resistance tests show a linear relationship between the true density of the grain and the crush-resistance coefficient , highlighting the influence of aggregate density on mechanical performance, regardless of the carbonated aggregate:
Figure 2 shows the evolution of the dry density of concretes regarding the amount of carbonated aggregates used in the mix design. Concrete density varies linearly with the substitution rate due to the gradual introduction of air trapped in the internal pores of the carbonated aggregates. The curves representing the density of C8agg and PA aggregate concretes show identical slopes because of the similar intraparticle porosities (0.18 vs. 0.17) when BAk aggregate concrete traps a smaller amount of air (), represented by a lower slope.
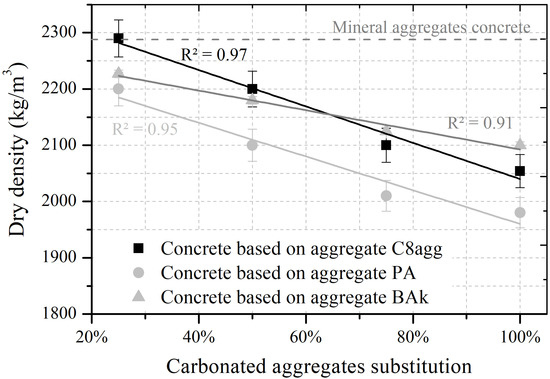
Figure 2.
Evolution of the concrete’s density regarding the substitution rate with carbonated aggregates.
The average strength of carbonated aggregate concretes is about 16 MPa at 28 days (see Table 3), which seems sufficient for load-bearing structures. As expected, the strength of concrete decreases as the substitution rate of aggregate increases. A significant decrease in the compressive strength of concrete is obtained from a low substitution rate (25%). The introduction of carbonated aggregates generates points of weakness in the granular skeleton, favoring the appearance of the first cracks near these aggregates. The initiation of these cracks then governs an anticipated propagation of cracking leading to the failure of the samples. The theory of the weakest link could explain the asymptotic behavior of the compressive strength of concrete at 1 year at a low substitution rate.

Table 3.
Mechanical characterization of the concrete-based carbonated aggregates.
Moreover, C8agg concrete shows the best mechanical strength at one year, followed by BAk concrete and then PA concrete. In the same way as before, the behavior of concrete seems to be dependent of the density of the carbonated aggregates, where C8gg shows a higher density, followed by BAk then PA (Table 2). At 7 days, the differences between concretes are very different. The concrete of PA aggregates shows better mechanical performances than the other two formulations. The crystallization kinematics are impacted by the origin of carbonated aggregates. Aggregates with a high CaO rate (such as that of PA) show the fast formation of Portlandite—and thus the appearance of a crystalline entanglement—which is partly responsible for young age behavior of concrete.
In the same way, the elasticity modulus of concrete is strongly influenced by the introduction of carbonated aggregates in the mix. The carbonated aggregates are porous and have low crushing resistance governing a low modulus of elasticity. Cracks appear within aggregates during compression test representing a good cement matrix-aggregate bonding. Cement paste seems to penetrate into open pores of aggregates, confirming by SEM analysis [29]. The density of aggregates seems to govern the evolution of elasticity modulus which achieves an asymmetrical behavior regarding the substitution rate.
3.2. Thermal Performances
The guarded hot plate test allows for the identification of the experimental thermal conductivity of bulk aggregates. Due to their intraparticle porosity, the thermal conductivity of carbonated aggregates is lower than that of mineral aggregate (from −40% to −20%). Then, a methodology based on Hashin–Shtrikman limits is used to establish the thermal conductivity of solid phase of aggregates. The performance of a two-phase material is defined as follows [29,33]:
The upper and lower limits of Hashin–Shtrikman, respectively, and , can be expressed by the following expressions [24]:
where and are the thermal conductivity of the solid phase and air, respectively. This approach leads to the thermal conductivity of the aggregate and of the solid phase of the grain from the total porosity and the interparticle porosity (Table 2). The results presented in Table 4 show the good thermal performance of paper ash-based aggregate (PA) with a bulk thermal conductivity of 0.15 W/(m·K) at 10 °C. The thermal conductivity of a carbonated aggregate remains interesting compared to that of a mineral aggregate (measured close to 0.25 W/(m·K) at 10 °C, with the same experimental protocol) notably because of their high intraparticle porosity.

Table 4.
Thermal conductivity of the carbonated aggregates.
The thermal conductivity of the solid part of the grain is approximately the same for all aggregates, near 0.9 W/(m·K). The intraparticle porosity seems to govern the evolution of the thermal conductivity of the grain and a linear relation can then be proposed through Equation (15) ():
The characterization of the thermal performances of concretes, using the guarded hot plate and fluxmetric methods, is presented in Table 5. The two thermal conductivity measurement protocols lead to a maximum deviation of approximately 10%. Concretes made of carbonated aggregates show an interesting capacity in the storage of energy (through ) and a slower propagation of the heat flow by conduction under dynamic conditions (a). The thermal performance seems advantageous, compared to mineral concrete. The identification of thermal conductivity and specific heat capacity lead to the establishment of thermal diffusivity and effusivity.

Table 5.
Thermal properties of carbonated aggregate-based concretes.
The use of carbonated aggregates in low proportions (less than or equal to 50%) leads to a small reduction in thermal conductivity compared to standard concrete (greater than 1 W/(m·K)). On the other hand, a higher proportion of carbonated aggregates will have a significant impact with a decrease in the thermal conductivity, lower than 1 W/(m·K). C8 aggregate-based concrete allows to obtain a thermal conductivity lower than 0.8 W/(m·K). These results are explained by the distribution of capillary pores (open and closed) in the concrete. Indeed, mineral aggregate-based concretes present a high thermal conductivity because of the low fraction of air trapped in the pores of the material (matrix and aggregate). For high percentages of carbonated aggregates, the concrete presents a consequent pore network, especially within the aggregates which compose its granular skeleton.
The substitution rate has a significant impact on thermal performance, particularly through the amount of air trapped within the carbonated aggregates. The true density seems to be an interesting property governing the thermal performance of concrete, both for thermal conductivity and thermal mass capacity. On the other hand, the origin of the aggregates does not seem to impact the thermal properties of the concretes, the thermal conductivity of the solid part being appreciably homogeneous among carbonated aggregates (Table 4).
3.3. Hygric Characterization
Figure 3 presents the sorption isotherms at 23 °C of the three carbonated aggregates concretes (100% substitution rate). The large opening of the isotherms shows the capacity of materials to store and release moisture, thus actively participating in the regulation of the indoor environment relative humidity. The experimental data were fitted by the GAB model. GAB model expresses the evolution of water content as a function of relative humidity, using three experimentally determined parameters, representing the molecular water content , the molar heat of adsorption C and the latent molar heat of vaporization K:
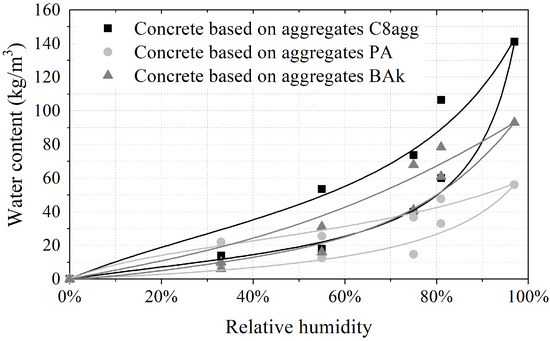
Figure 3.
Sorption isotherms of carbonated aggregate-based concretes.
Then, Table 6 summarizes the results of the measurement. In contrast to mineral concrete (which has an close to 1 (g/(m%HR)), corresponding to a moderate capacity to regulate the relative humidity of the environment), carbonated aggregate-based concretes have a good to excellent hygric performance, depending on the substitution rate. As expected with regard to the internal porosity of the grains, the best performances are identified for the highest substitution rates.

Table 6.
Moisture buffer value of concretes based on carbonated aggregates.
The results show that carbonated aggregates favor water transfer, in liquid or vapor form, because of their microporal structure (quantity and diameter of capillaries). These aggregates then represent a moisture reserve within the concrete itself, governing an ability to regulate the surrounding moisture levels as highlighted by the MBV results.
4. Mechanical, Thermal and Hygric Predictive Approaches
4.1. Empirical Mechanical Model
Conventional approaches of the mechanical properties of concrete tend to over- or underestimate the mechanical characteristics of carbonated aggregates concrete [34]. These approaches lead to the 28-day compressive strength as well as the associated elasticity modulus. Thanks to the experimental campaign, an empirical predictive approach is proposed to assess the main mechanical properties in compression, regarding the density of aggregates. Because of the crystallization times of concretes, depending on the ash origin (Table 3), the approach is focused on the 90-day compressive strength. In a first step, the apparent dry density of the grains is determined, based on the substitution rate of carbonated aggregates :
where can be assessed from the crush-resistance (Equation (11)). Then, the dry density of the formulated concrete can be expressed:
The mechanical resistance in compression and the associated initial tangential elasticity modulus are deduced:
Figure 4 shows the comparison between the experimental data and the results from the empirical predictive model. A deviation close to 12% was reached for the compressive strength at 90 days and approximately 10% for the elasticity modulus, in the same order as the uncertainty of mechanical tests (Table 3). This simplified approach, based on the substitution rate and aggregate density, allows an accurate prediction of 90-day strength and tangential modulus of elasticity.
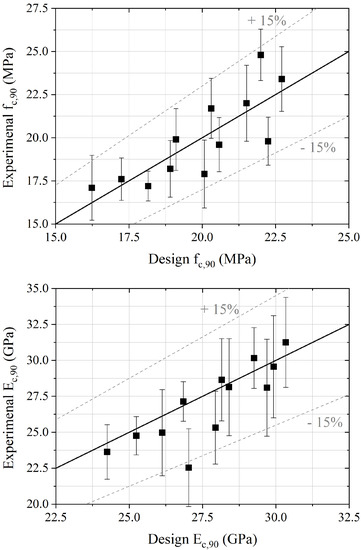
Figure 4.
Design mechanical properties compared to experimental ones.
4.2. Prediction of the Thermal Characteristics
As aforementioned, the thermal conductivity of concrete seems to be governed by the true density of aggregates and the substitution rate. In order to confirm that assumption, a thermal model of auto-coherent homogenization (ACH) with tri-composite inclusion is investigated [23]. This model was used to determine the equivalent thermal conductivity of a spherical inclusion comprising three phases. The three phases represent the included air, the solid part of the grain and the binding matrix. In this approach, the material is considered perfectly dry. The thermal conductivity of concrete can be calculated using the input assumptions of the model and the volume concentration coefficients [25]. The concentration coefficients of the ACH model represent the ratio between the different phases, including the impact of grain porosity. The application of the tri-composite ACH model leads to a satisfactory estimation of the thermal conductivity of carbonated aggregate concrete with a maximum deviation between analytical and experimental results of about 20%, as presented in [34]. The concentration coefficients (dependent on the density ratio) mainly govern the model results, regarding the small differences of the thermal conductivity of the solid phase of the carbonated aggregates . Therefore, a predictive approach is proposed, based on the true homogenized density of the grains (Equation (17)). Depending on the density of the concrete (see Equation (18)), the coefficient is defined as follows:
This approach allows intraparticle and interparticle porosities to be indirectly taken into account through . Then, the thermal conductivity of carbonated aggregate-based concrete was expressed by the following expression:
Similarly, the thermal mass capacity can be approached as follows:
Modeling results are compared with experimental properties in Figure 5 where a maximum deviation of 10% can be obtained for the thermal conductivity and 11% for the thermal mass capacity, whatever the nature of aggregates and the substitution rate. Thus, the model tends to prove that thermal properties seem to be mainly dependent on grain density, as the nature of ashes to produce carbonated aggregates does not seem to be significant.
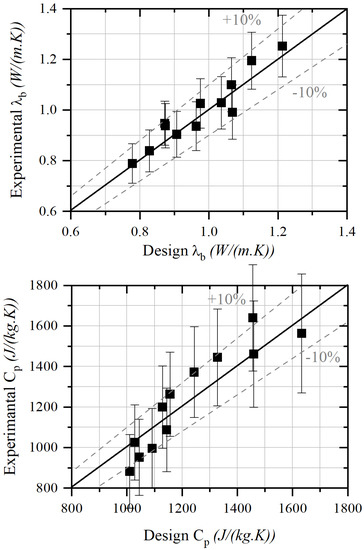
Figure 5.
Design thermal properties compared to experimental ones.
4.3. Simplified Theoretical Approach of the Hygric Properties
The NORDTEST Moisture Buffering project protocol defined two theoretical approaches describing the ability of the material to take up and store moisture: hygric effusivity and ideal moisture buffer value. Hygric effusivity is expressed by the following expression [26]:
The ideal moisture buffer value corresponds to the for an infinite mass convection coefficient [35]. is thus considered as an intrinsic property of a material and is used to establish the hygric performance of materials [36]:
The ratio between and is defined as a function of the Biot mass number [37] which reflects the water vapor diffusion resistance of a material:
When the Biot mass number tends towards 100, is identical to . For a very low Biot number, tends towards 0. In construction fields, the Biot mass number is generally between 10 and 100 [35], leading to slight variation of the to ratio between 0.9 and 1. Assuming an intermediate value of 0.95, a simplified approach leads to the thermal effusivity using the following expression:
Then, by inverting Equation (24), the coefficient of water vapor permeability can be obtained. This coefficient is necessary to assess the hygric diffusivity , from a semi-infinite homogeneous model [38] where the water capacity is the slope of the sorption isotherm (). The water capacity is deduced from Figure 3. Then, the hygric diffusivity is expressed by
Table 7 presents the results obtained from this theoretical method, thanks to the hygric characterization campaign. The water capacity corresponds to the current relative humidity range of the building area, between 40 and 80% (where the evolution of water content regarding relative humidity is linear). The vapor diffusion resistance factor is the ratio between the vapor permeability of air to the vapor permeability of concrete (). This theoretical model leads to a vapor diffusion resistance factor of mineral aggregate concrete near 40 and an hygric diffusion of 5.3 m/s, which is in agreement with the literature (see, e.g., the results displayed in [39] which propose a synthesis of the water vapor diffusion coefficient of hardened concretes).

Table 7.
Hygric properties of carbonated aggregate-based concretes established from the theoretical approach.
The evolution of the vapor diffusion resistance factor regarding the dry density of hardened concrete is plotted in Figure 6, where a linear relation can be highlighted. Thanks to the grain density expressed by Equation (17), the dry density of hardened concrete can be designed (Equation (18)) in order to establish the vapor diffusion resistance factor (R = 0.94):
Modeling results leads to a maximum deviation of 15%, whatever the origin of carbonated aggregates or substitution rates of mineral aggregates.
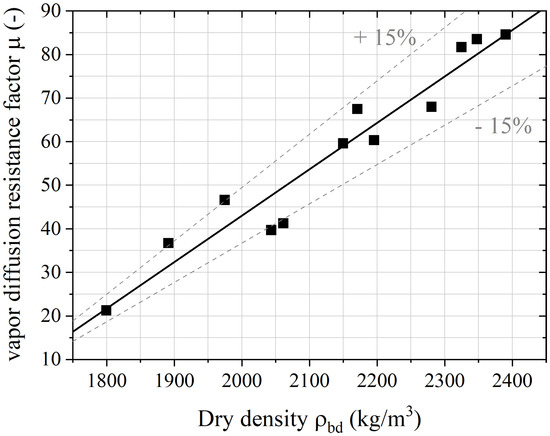
Figure 6.
Evolution of the vapor diffusion resistance factor vs. the dry density of concrete based on carbonated aggregates.
5. Conclusions
The use of carbonated aggregates can be focused on limiting the consumption of mineral resources and the quantity of landfill waste. These aggregates are obtained by the carbonation of waste incinerator ashes, and lead to a negative carbon balance. The inclusion of these lightweight aggregates in a cementitious mixture will increase the porosity of the concrete and affects the strength of the structure and the thermal and hygric performances of the building envelope.
Different wastes were used to produce aggregates: APCr ash (C8agg), paper ash (PA) and biomass ash (BAk), chosen from more than 150 waste streams regarding their ability through carbonation. Firstly, concrete samples were produced with an efficient water to cement ratio of 0.7 and using 25%, 50%, 75% and 100% substitution rates of mineral aggregates. Samples were used to identify the main mechanical, thermal and hygric properties of concretes. Experimental results show an improvement of the thermal and hygric performances of concrete, which could lead, in the building fields, to an increase in the thermal insulation (thermal conductivity can be reduced up to 35%) and of the hygroscopic regulation (good to excellent MBV). The mechanical resistance appears sufficient for load-bearing building envelopes (mostly greater than 16 MPa at 28 days).
Moreover, the experimental campaign highlights the influence of the porosity and density of carbonated aggregates on the properties of concrete, for different mix design (substitution rates from 25 to 100%), regardless the water to cement ratio or kind of binder. The origin of carbonated aggregates does not seem to have significant influence on properties of concrete. To confirm that, some predictive laws are proposed to assess the mechanical strength and elasticity modulus of carbonated aggregate concretes, based on the dry density and the porosity of aggregates. This approach leads to a deviation of around 15% compared to experimental results at 90 days. A second model is expressed to predict the thermal conductivity and the specific heat capacity. This model is still based on the density of aggregates. A deviation of around 10% from the experimental data is highlighted. Finally, a simplified theoretical approach was proposed to establish the main hygric properties of concretes, and especially the hygric effusivity and diffusivity in the relative humidity range from 40 to 80%.
Additional experiments could strengthen these models which are developed for carbonated aggregate-based concrete. More tests could be carried out to validate these approaches, with different mixture designs such as the water-to-cement ratio. The valorization of incinerator ashes for building aggregates is still a subject of numerous studies in order to ensure the durability and sustainability of these building materials, as well as the evolution of thermal and hygric performances over time. In addition, the improvement of tensile performance is critical for making structural concrete. The compatibility with a steel-type reinforcement, in the form of bars or fibers, also appears as an interesting investigative lead.
Author Contributions
Conceptualization, G.P.; methodology, O.D.; validation, I.R.; formal analysis, G.P. and O.D.; investigation, G.P., O.D. and I.R.; writing—original draft preparation, G.P., O.D.; writing—review and editing, I.R. and F.R.; supervision, G.P.; project administration, G.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors would like to thank partners of the SAPICO project which has been selected in the context of the INTERREG European cross-border cooperation program, which is co-financed by the ERDF.
Conflicts of Interest
The authors certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript. The granting bodies had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| porosity | |
| total | |
| interparticle | |
| intraparticle | |
| absolute | |
| true | |
| density, kg/m | |
| a | thermal diffusivity, m/s |
| E | thermal effusivity, J/(mK·s) |
| specific heat capacity, J/(kg·K) | |
| compressive strength, MPa | |
| modulus of elasticity, MPa | |
| carbonated | |
| mineral | |
| substitution rate, % | |
| experimental | |
| aggregate | |
| s | solid |
| hygric diffusivity, m/s | |
| moisture buffer value, kg/(m%RH) | |
| hygric effusivity, kg/(mPa·s) | |
| vapor saturation pressure, Pa | |
| vapor permeability, kg/(m·s·Pa) |
References
- González-Corrochano, B.; Alonso-Azcárate, J.; Rodas, M. Characterization of lightweight aggregates manufactured from washing aggregate sludge and fly ash. Resour. Conserv. Recycl. 2009, 53, 571–581. [Google Scholar] [CrossRef]
- Behera, M.; Bhattacharyya, S.; Minocha, A.; Deoliya, R.; Maiti, S. Recycled aggregate from C&D waste & its use in concrete—A breakthrough towards sustainability in construction sector: A review. Constr. Build. Mater. 2014, 68, 501–516. [Google Scholar]
- Evangelista, L.; de Brito, J. Mechanical behaviour of concrete made with fine recycled concrete aggregates. Cem. Concr. Compos. 2007, 29, 397–401. [Google Scholar] [CrossRef]
- Poon, C.; Kou, S.; Lam, L. Use of recycled aggregates in molded concrete bricks and blocks. Constr. Build. Mater. 2002, 16, 281–289. [Google Scholar] [CrossRef]
- Rao, A.; Jha, K.N.; Misra, S. Use of aggregates from recycled construction and demolition waste in concrete. Resour. Conserv. Recycl. 2007, 50, 71–81. [Google Scholar] [CrossRef]
- Kayali, O. Fly ash lightweight aggregates in high performance concrete. Constr. Build. Mater. 2008, 22, 2393–2399. [Google Scholar] [CrossRef]
- Radonjanin, V.; Malešev, M.; Marinković, S.; Al Malty, A. Green recycled aggregate concrete. Constr. Build. Mater. 2013, 47, 1503–1511. [Google Scholar] [CrossRef]
- Šavija, B.; Luković, M. Carbonation of cement paste: Understanding, challenges, and opportunities. Constr. Build. Mater. 2016, 117, 285–301. [Google Scholar] [CrossRef] [Green Version]
- Motz, H.; Geiseler, J. Products of steel slags an opportunity to save natural resources. Waste Manag. 2001, 21, 285–293. [Google Scholar] [CrossRef]
- INSEE. Indicateurs de Développement Durable en Champagne-Ardenne: La Production des Granulats; Insee dossier n°34; INSEE: Malakoff, France, 2011; pp. 21–22. [Google Scholar]
- Chen, T.; Bai, M.; Gao, X. Carbonation curing of cement mortars incorporating carbonated fly ash for performance improvement and CO2 sequestration. J. CO2 Util. 2021, 51, 101633. [Google Scholar] [CrossRef]
- Cerezo, V. Propriétés Mécanique, Thermique et Acoustique d’un Matériau à Base de Particules Végétales: Approche Expérimentale et Modélisation Théorique. Ph.D. Thesis, Université de Lyon, Lyon, France, 2005. [Google Scholar]
- Latif, E.; Lawrence, M.; Shea, A.; Walker, P. Moisture buffer potential of experimental wall assemblies incorporating formulated hemp-lime. Build. Environ. 2015, 93, 199–209. [Google Scholar] [CrossRef] [Green Version]
- BPEL-91. Régles Techniques de Conception et de Calcul des Ouvrages et Constructions en Béton; Eyrolles: Paris, France, 1993. [Google Scholar]
- Arnould, M.; Virlogeux, M. Granulats et Bétons Légers; Presse de l’ENPC: Toulon, France, 1986. [Google Scholar]
- ACI Committee 318. Building Code Requirements for Structural Concrete and Commentary (ACI 318M-11); ACI Committee: Farmington Hills, MI, USA, 2011; p. 520. [Google Scholar] [CrossRef]
- EN 1992-1-1. EuroCode 2: Design of Concrete Structures: General Rules and Rules for Buildings. 2007. Available online: https://www.phd.eng.br/wp-content/uploads/2015/12/en.1992.1.1.2004.pdf (accessed on 21 July 2021).
- Zhang, M.; Gjorv, O. Mechanical properties of high-strength lightweight concrete. ACI Mater. J. 1991, 88, 240–247. [Google Scholar] [CrossRef]
- Slate, F.; Nilson, A.; Martinez, S. Mechanical properties of high-strength lightweight concrete. J. Am. Concr. Inst. 1986, 83, 606–613. [Google Scholar] [CrossRef]
- Ke, Y. Caractérisation du Comportement Mécanique des Bétons de Granulats Légers: Expérience et Modélisation. Ph.D. Thesis, Université de Cergy Pontoise, Cergy, France, 2008. [Google Scholar]
- Ren, M.; Wen, X.; Gao, X.; Liu, Y. Thermal and mechanical properties of ultra-high performance concrete incorporated with microencapsulated phase change material. Constr. Build. Mater. 2021, 273, 121714. [Google Scholar] [CrossRef]
- Collet, F.; Pretot, S. Thermal conductivity of hemp concretes: Variation with formulation, density and water content. Constr. Build. Mater. 2014, 65, 612–619. [Google Scholar] [CrossRef] [Green Version]
- Rahim, M. Analyse et Caractérisation du Comportement Hygrothermique de Parois Agro-Sourcées à L’échelle 1: Expérimentation et Modélisation. Ph.D. Thesis, Universite de Picardie Jules Verne, Amiens, France, 2015. [Google Scholar]
- Hashin, Z.; Shtrikman, S. A variational approach to the theory of the elastic behaviour of multiphase materials. J. Mech. Phys. Solids 1963, 11, 127–140. [Google Scholar] [CrossRef]
- Bederina, M.; Marmoret, L.; Mezreb, K.; Khenfer, M.; Bali, A.; Quéneudec, M. Effect of the addition of wood shavings on thermal conductivity of sand concretes: Experimental study and modelling. Constr. Build. Mater. 2007, 21, 662–668. [Google Scholar] [CrossRef]
- Rode, P. Moisture Buffering of Building Materials; Report BYG DTU R-126; Deparment of Civil Engineering Technical University of Denmark: Lyngby, Denmark, 2005. [Google Scholar]
- Gunning, P.; Hills, C.; Carey, P. Production of lightweight aggregate from industrial waste and carbon dioxide. Waste Manag. 2009, 29, 2722–2728. [Google Scholar] [CrossRef] [PubMed]
- Fernández Bertos, M.; Simons, S.; Hills, C.; Carey, P. A review of accelerated carbonation technology in the treatment of cement-based materials and sequestration of CO2. J. Hazard. Mater. 2004, 112, 193–205. [Google Scholar] [CrossRef]
- Rahmouni, I.; Promis, G.; R’mili, A.; Beji, H.; Limam, O. Effect of carbonated aggregates on the mechanical properties and thermal conductivity of eco-concrete. Constr. Build. Mater. 2019, 197, 241–250. [Google Scholar] [CrossRef]
- Rode, C.; Clorius, C. Modeling of moisture transport in wood with hysteresis and temperature dependent sorption characteristics. In Performance of Exterior Envelopes of Whole Buildings; International Conference Clearwater: Clearwater, FL, USA, 2004; Volume 80. [Google Scholar]
- Rode, C.; Peuhkuri, R.; Time, B.; Svennberg, K.; Ojanen, T. Moisture buffer value of building materials. J. ASTM Int. 2007, 4, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Rode, C.; Holm, A.; Padfield, T. A Review of Humidity Buffering in the Interior Spaces. J. Therm. Envel. Build. Sci. 2004, 27, 221–226. [Google Scholar] [CrossRef]
- Bourdot, A.; Promis, G.; Tran Le, A.; Douzane, O.; Benazzouk, A.; Rosquoët, F.; Langlet, T. Hygrothermal properties of blocks based on eco-aggregates: Experimental and numerical study. Constr. Build. Mater. 2016, 125, 279–289. [Google Scholar] [CrossRef]
- Rahmouni, I. Optimisation de la Formulation de Bétons a Base D’éco-Granulats Obtenus par Carbonatation Accélérée de Déchets D’incinérateur. Ph.D. Thesis, Université de PIcardie Jules Verne, Amiens, France, 2019. [Google Scholar]
- Tran Le, A.D. Etude des Transferts Hygrothermiques Dans le Béton de Chanvre et leur Application au Bâtiment: Simulation Numérique et Approche Expérimentale. Ph.D. Thesis, Université de Reims Champagne-Ardenne, Reims, France, 2010. [Google Scholar]
- Peuhkuri, R.; Rode, C.; Hansen, K.K. Non-isothermal moisture transport through insulation materials. Build. Environ. 2008, 43, 811–822. [Google Scholar] [CrossRef]
- Abadie, M.; Mendonça, K. Moisture performance of building materials: From material characterization to building simulation using the Moisture Buffer Value concept. Build. Environ. 2009, 44, 388–401. [Google Scholar] [CrossRef]
- Arfvidsson, J. Moisture penetration for periodically varing relative humidity at boundary. Acta Phys. Aedificiorum 1999, 2. [Google Scholar]
- Baroghel-Bouny, V. Water vapour sorption experiments on hardened cementitious materials. Part II: Essential tool for assessment of transport properties and for durability prediction. Cem. Concr. Res. 2007, 37, 438–454. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).