Recent Advances in Carbon Dioxide Conversion: A Circular Bioeconomy Perspective
Abstract
:1. Introduction
2. CO2 Conversion Processes
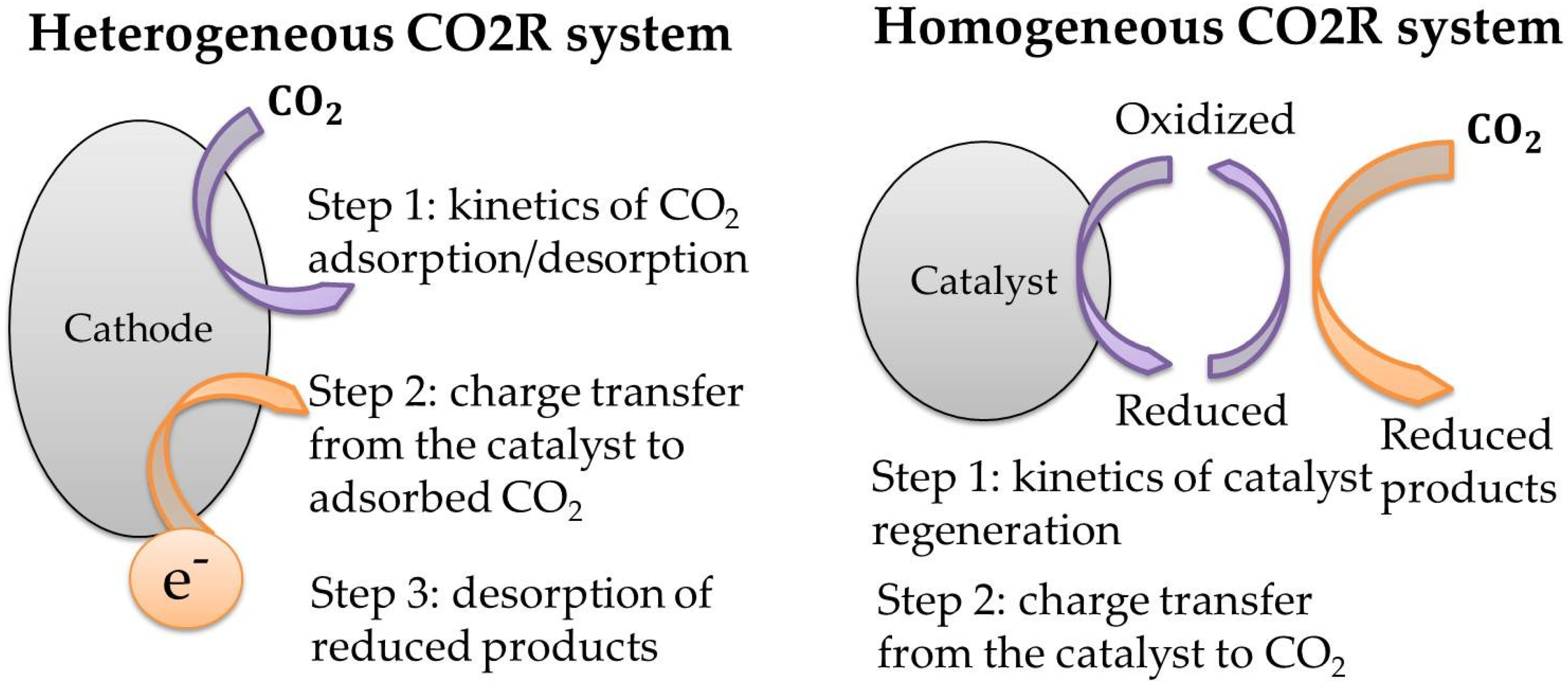
2.1. Homogeneous Catalysts for CO2R
2.2. Heterogeneous Catalysts for CO2R
3. The Chemistry of Abiotic CO2R
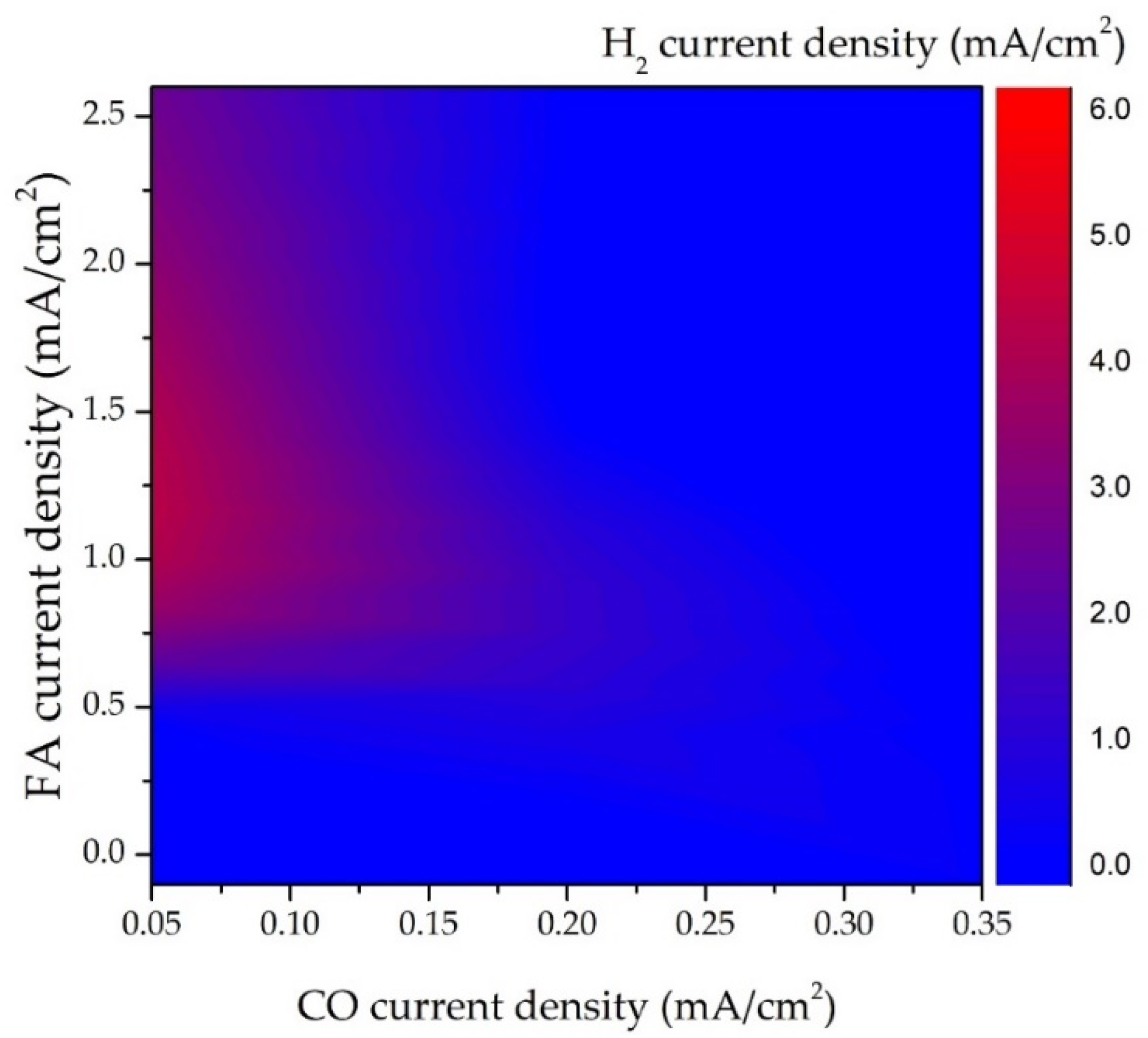
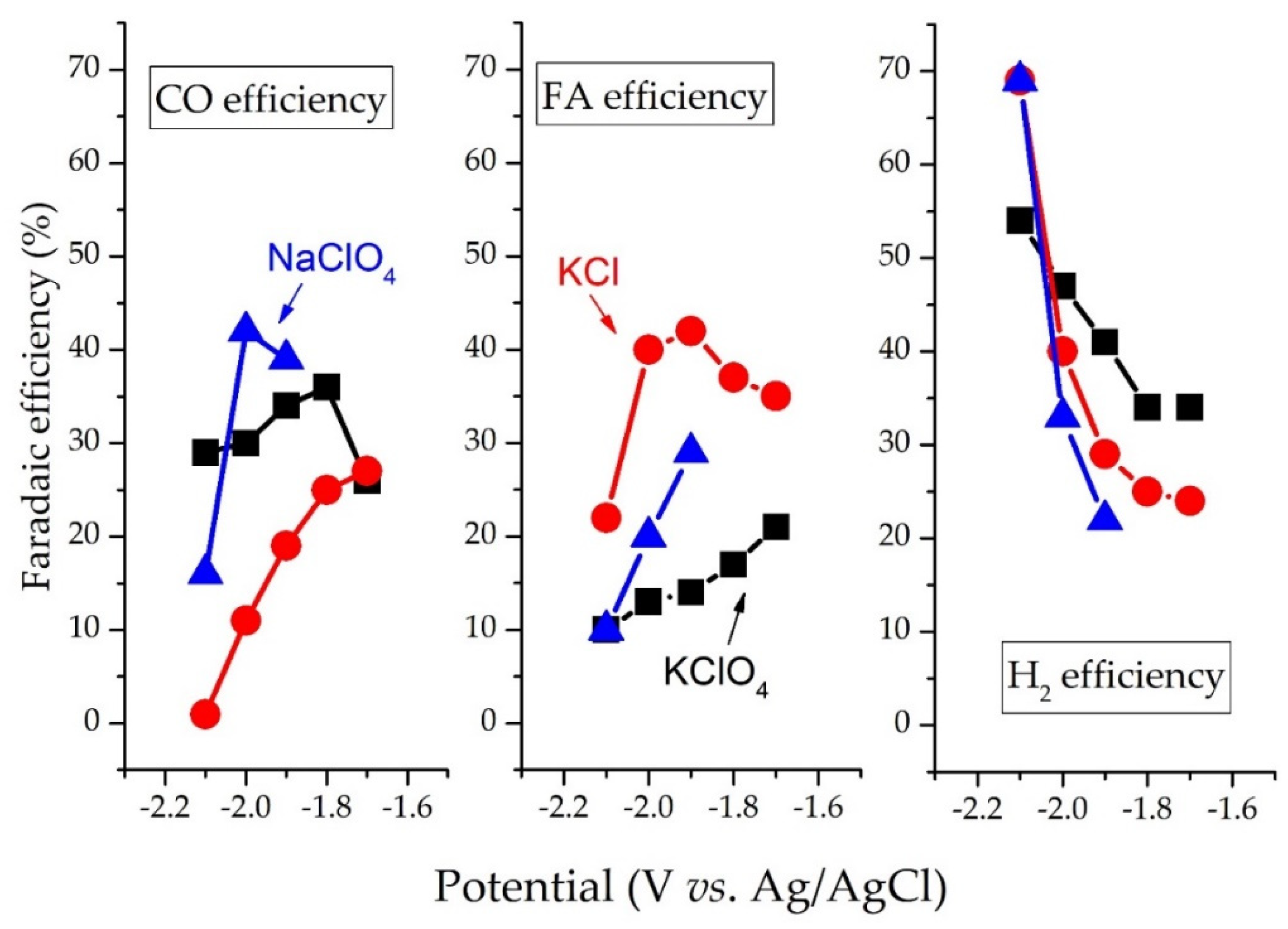
3.1. Abiotic CO2R in Water Phase
3.1.1. Effect of Cu Surface Chemistry on Abiotic CO2R
3.1.2. Effect of Surface Chemistry of Non Cu-Family Catalysts on Abiotic CO2R
3.2. The Chemistry of Gaseous Phase Abiotic CO2R
3.2.1. Hydrogenation of CO2 to CO and CH4
3.2.2. Hydrogenation of CO2 to Methanol
3.2.3. Hydrogenation of CO2 to Low and Long Chain Chemicals
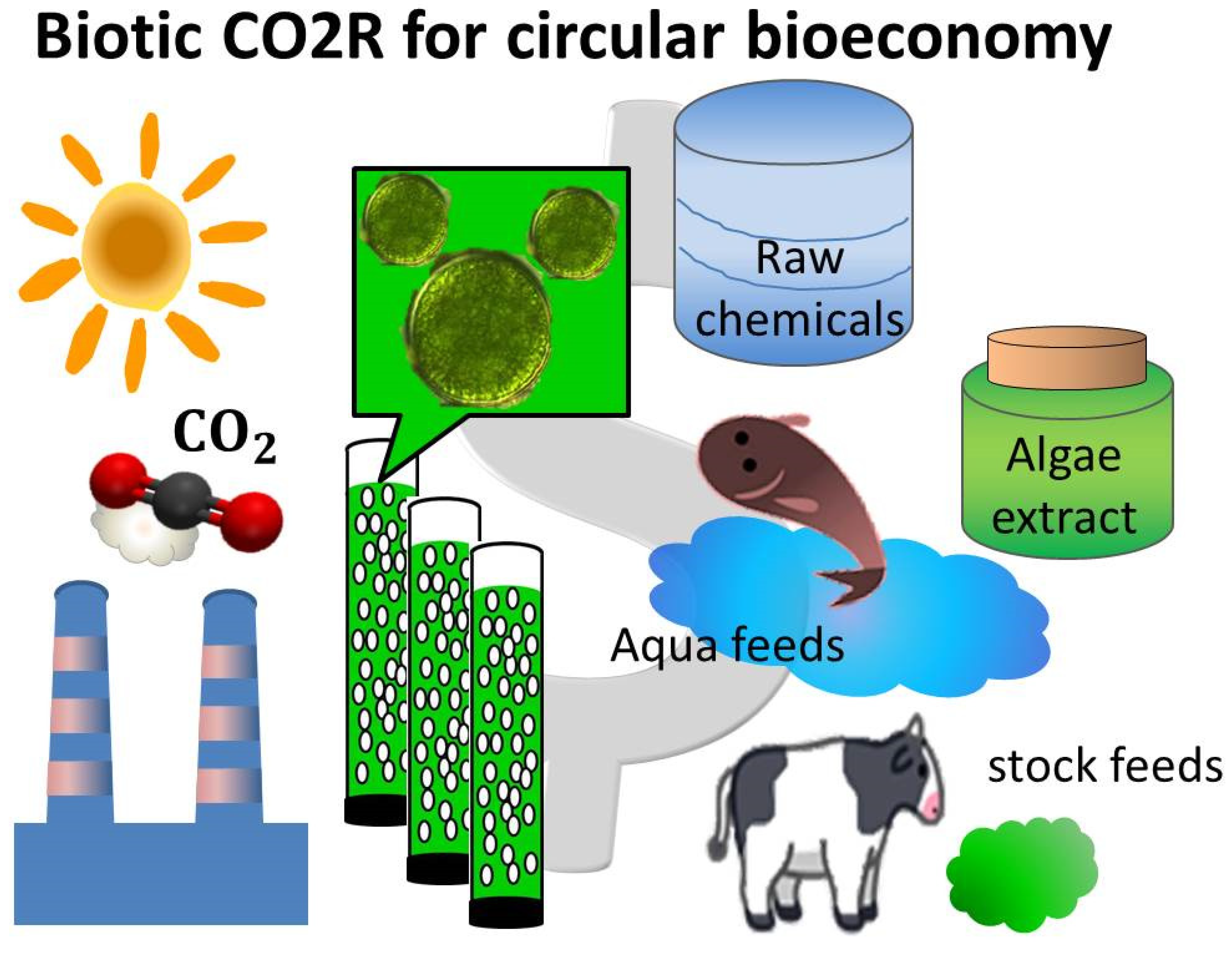
4. Biotic CO2R
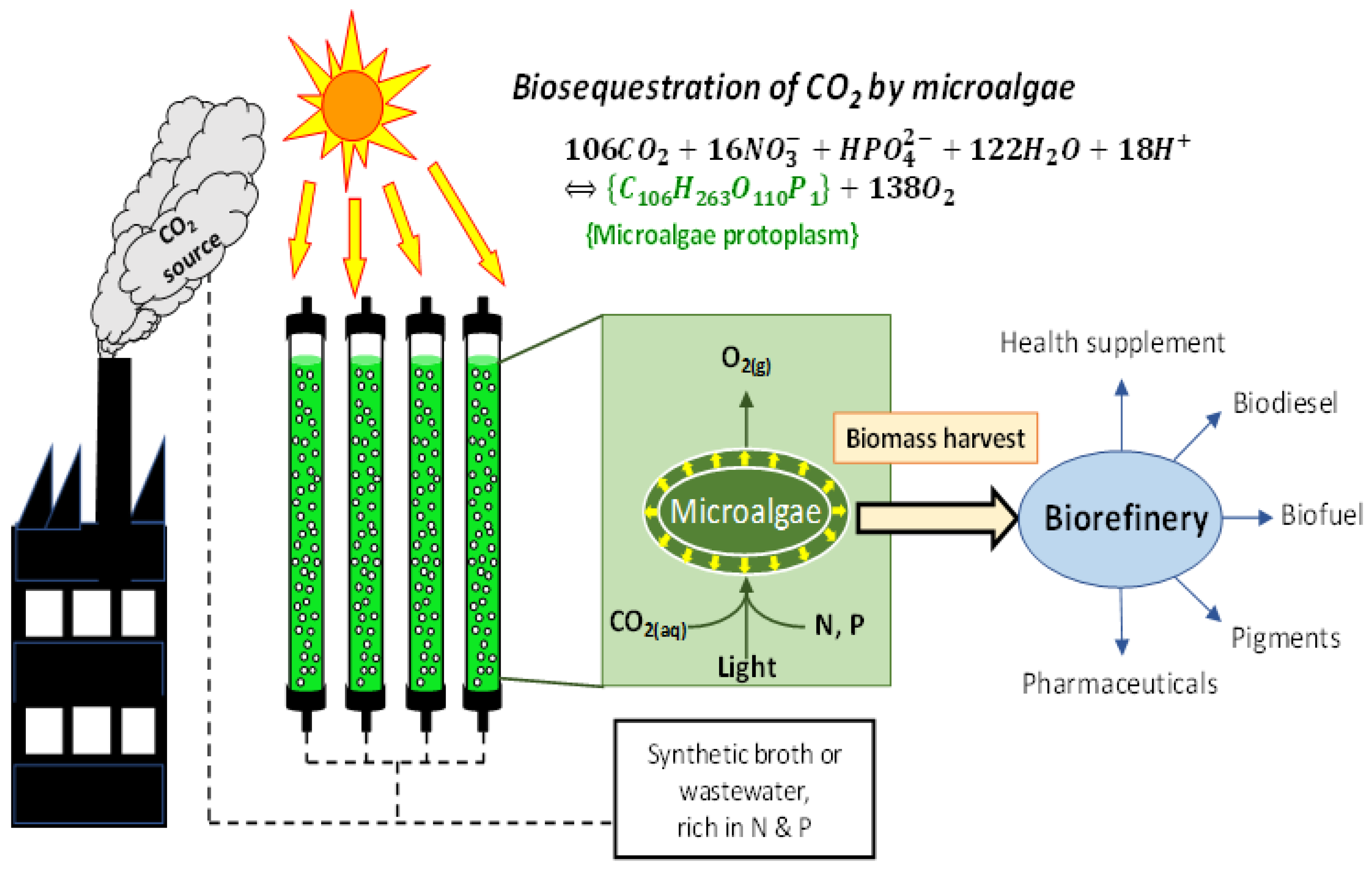
4.1. Biotic CO2R by Microalgae Farming
4.2. The Unit Processes for Biotic CO2R
4.3. Additional Ecomomic Benefit of Biotic CO2R
4.4. The Utilization of Microalgae Biomass
4.5. The Circular Bioeconomy
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Redfield, A.C.; Ketchum, B.K.; Richards, F.A. The influence of organisms on the composition of sea-water. In The Sea; Wiley: New York, NY, USA, 1963; Volume 2, pp. 26–77. [Google Scholar]
- Francke, R.; Schille, B.; Roemelt, M. Homogeneously catalyzed electroreduction of carbon dioxide—Methods, mechanisms, and catalysts. Chem. Rev. 2018, 118, 4631–4701. [Google Scholar] [CrossRef] [PubMed]
- Qiao, J.; Liu, Y.; Hong, F.; Zhang, J. A review of catalysts for the electroreduction of carbon dioxide to produce low-carbon fuels. Chem. Soc. Rev. 2014, 43, 631–675. [Google Scholar] [CrossRef] [PubMed]
- Rakowski Dubois, M.; Dubois, D.L. Development of molecular electrocatalysts for CO2 reduction and H2 production/oxidation. Acc. Chem. Res. 2009, 42, 1974–1982. [Google Scholar] [CrossRef] [PubMed]
- Mellmann, D.; Sponholz, P.; Junge, H.; Beller, M. Formic acid as a hydrogen storage material—Development of homogeneous catalysts for selective hydrogen release. Chem. Soc. Rev. 2016, 45, 3954–3988. [Google Scholar] [CrossRef]
- Sordakis, K.; Tang, C.; Vogt, L.K.; Junge, H.; Dyson, P.J.; Beller, M.; Laurenczy, G. Homogeneous catalysis for sustainable hydrogen storage in formic acid and alcohols. Chem. Rev. 2018, 118, 372–433. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.Z.; Wang, L.S. Bifunctional wood for electrocatalytic CO2 reduction to formate and electroanalytical detection of myricetin and cadmium (II). Electrochim. Acta 2019, 319, 569–576. [Google Scholar] [CrossRef]
- Kim, Y.E.; Lee, W.; Youn, M.H.; Jeong, S.K.; Kim, H.J.; Park, J.C.; Park, K.T. Leaching-resistant SnO2/gamma-Al2O3 nanocatalyst for stable electrochemical CO2 reduction into formate. J. Indust. Eng. Chem. 2019, 78, 73–78. [Google Scholar] [CrossRef]
- Asset, T.; Garcia, S.T.; Herrera, S.; Andersen, N.; Chen, Y.C.; Peterson, E.J.; Matanovic, I.; Artyushkova, K.; Lee, J.; Minteer, S.D.; et al. Investigating the nature of the active sites for the CO2 reduction reaction on carbon-based electrocatalysts. ACS Catal. 2019, 9, 7668–7678. [Google Scholar] [CrossRef]
- Zhang, N.; Gao, C.; Xiong, Y.J. Defect engineering: A versatile tool for tuning the activation of key molecules in photocatalytic reactions. J. Energy Chem. 2019, 37, 43–57. [Google Scholar] [CrossRef] [Green Version]
- Zhou, L.Q.; Ling, C.; Zhou, H.; Wang, X.; Liao, J.; Reddy, G.K.; Deng, L.Z.; Peck, T.C.; Zhang, R.G.; Whittingham, M.S.; et al. A high-performance oxygen evolution catalyst in neutral-pH for sunlight-driven CO2 reduction. Nat. Comm. 2019, 10, 4081. [Google Scholar] [CrossRef] [Green Version]
- Yang, D.R.; Yu, H.D.; He, T.; Zuo, S.W.; Liu, X.Z.; Yang, H.Z.; Ni, B.; Li, H.Y.; Gu, L.; Wang, D. Visible-light-switched electron transfer over single porphyrin-metal atom center for highly selective electroreduction of carbon dioxide. Nat. Comm. 2019, 10, 3844. [Google Scholar] [CrossRef] [Green Version]
- Chang, X.X.; Wang, T.; Zhang, P.; Wei, Y.J.; Zhao, J.B.; Gong, J.L. Stable aqueous photoelectrochemical CO2 reduction by a Cu2O dark cathode with improved selectivity for carbonaceous products. Angew. Chim. Int. Ed. 2016, 55, 8840–8845. [Google Scholar] [CrossRef]
- Cheng, J.; Zhang, M.; Liu, J.Z.; Zhou, J.H.; Cen, K.F. A Cu foam cathode used as a Pt-RGO catalyst matrix to improve CO2 reduction in a photoelectrocatalytic cell with a TiO2 photoanode. J. Mater. Chem. A 2015, 3, 12947–12957. [Google Scholar] [CrossRef]
- Pletcher, D. The cathodic reduction of carbon dioxide—What can it realistically achieve? A mini review. Electrochem. Comm. 2015, 61, 97–101. [Google Scholar] [CrossRef] [Green Version]
- Honoré, M.N.; Belmonte-Ureña, L.J.; Navarro-Velasco, A.; Camacho-Ferre, F. Profit analysis of papaya crops under greenhouses as an alternative to traditional intensive horticulture in southeast Spain. Int. J. Environ. Res. Public Health 2019, 16, 2908. [Google Scholar] [CrossRef] [Green Version]
- Hamnett, A. Mechanism and electrocatalysis in the direct methanol fuel cell. Catal. Today 1997, 38, 445–457. [Google Scholar] [CrossRef]
- Liu, H.; Song, C.; Zhang, L.; Zhang, J.; Wang, H.; Wilkinson, D.P. A review of anode catalysis in the direct methanol fuel cell. J. Power Sources 2006, 155, 95–110. [Google Scholar] [CrossRef]
- Jiang, J.; Wieckowski, A. Prospective direct formate fuel cell. Electrochem. Comm. 2012, 18, 41–43. [Google Scholar] [CrossRef]
- Bartrom, A.M.; Haan, J.L. The direct formate fuel cell with an alkaline anion exchange membrane. J. Power Sources 2012, 214, 68–74. [Google Scholar] [CrossRef]
- An, L.; Chen, R. Direct formate fuel cells: A review. J. Power Sources 2016, 320, 127–139. [Google Scholar] [CrossRef]
- Aresta, M. Carbon Dioxide as Chemical Feedstock; Wiley-VCH: Weinheim, Germany, 2010. [Google Scholar]
- Brennführer, A.; Neumann, H.; Beller, M. Palladium-catalyzed carbonylation reactions of aryl halides and related compounds. Angew. Chem. Int. Ed. 2009, 48, 4114–4133. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhang, H.; Lei, A. Oxidative carbonylation reactions: Organometallic compounds (R-M) or hydrocarbons (R-H) as nucleophiles. Angew. Chem. Int. Ed. 2011, 50, 10788–10799. [Google Scholar] [CrossRef]
- Pegis, M.L.; Roberts, J.A.S.; Wasylenko, D.J.; Mader, E.A.; Appel, A.M.; Mayer, J.M. Standard reduction potentials for oxygen and carbon dioxide couples in acetonitrile and N,N-dimethylformamide. Inorg. Chem. 2015, 54, 11883–11888. [Google Scholar] [CrossRef] [PubMed]
- Gai, S.L.; Yu, J.L.; Yu, H.; Eagle, J.; Zhao, H.; Lucas, J.; Doroodchi, E.; Moghtaderi, B. Process simulation of a near-zero-carbon-emission power plant using CO2 as the renewable energy storage medium. Int. J. Greenh. Gas Control 2016, 447, 240–249. [Google Scholar] [CrossRef]
- Sun, Z.; Zeng, L.; Russell, C.K.; Assabumrungrat, S.; Chen, S.Y.; Duan, L.B.; Xiang, W.G.; Gong, J.L. Solar–wind–bio ecosystem for biomass cascade utilization with multigeneration of formic acid, hydrogen, and graphene. ACS Sustain. Chem. Eng. 2019, 7, 22558–22568. [Google Scholar] [CrossRef]
- Zouaoui, N.; Ossonon, B.D.; Fan, M.Y.; Mayilukila, D.; Garbarino, S.; de Silveira, G.; Botton, G.A.; Guay, D.; Tavares, A.C. Electroreduction of CO2 to formate on amine modified Pb electrodes. J. Mater. Chem. A 2019, 7, 11272–11281. [Google Scholar] [CrossRef]
- Benson, E.E.; Kubiak, C.P.; Sathrum, A.J.; Smieja, J.M. Electrocatalytic and homogeneous approaches to conversion of CO2 to liquid fuels. Chem. Soc. Rev. 2009, 38, 89–99. [Google Scholar] [CrossRef]
- Xu, S.Z.; Carter, E.A. Theoretical insights into heterogeneous (photo)electrochemical CO2 reduction. Chem. Rev. 2019, 119, 6631–6669. [Google Scholar] [CrossRef]
- Nitopi, S.; Bertheussen, E.; Scott, S.B.; Liu, X.Y.; Engstfeld, A.K.; Horch, S.; Seger, B.; Stephens, I.E.L.; Chan, K.; Hahn, C.; et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem. Rev. 2019, 119, 7610–7672. [Google Scholar] [CrossRef] [Green Version]
- Ma, M.; Trzesniewski, B.J.; Xie, J.; Smith, W.A. Selective and efficient reduction of carbon dioxide to carbon monoxide on oxide-derived nanostructured silver electrocatalysts. Angew. Chem. Int. Ed. 2016, 55, 9748–9752. [Google Scholar] [CrossRef]
- Taheri, A.; Thompson, E.J.; Fettinger, J.C.; Berben, L.A. An iron electrocatalyst for selective reduction of CO2 to formate in water: Including thermochemical insights. ACS Catal. 2015, 5, 7140–7151. [Google Scholar] [CrossRef]
- Rosas-Hernández, A.; Junge, H.; Beller, M.; Roemelt, M.; Francke, R. Cyclopentadienone iron complexes as efficient and selective catalysts for the electroreduction of CO2 to CO. Catal. Sci. Technol. 2017, 7, 459–465. [Google Scholar] [CrossRef]
- Buckley, A.K.; Lee, M.; Cheng, T.; Kazantsev, R.V.; Larson, D.M.; Goddard, W.A., III; Toste, F.D.; Toma, F.M. Electrocatalysis at organic-metal interfaces: Identification of structure-reactivity relationships for CO2 reduction at modified Cu surfaces. J. Am. Chem. Soc. 2019, 141, 7355–7364. [Google Scholar] [CrossRef] [Green Version]
- Lobaccaro, P.; Singh, M.R.; Clark, E.L.; Kwon, Y.; Bell, A.T.; Ager, J.W. Effects of temperature and gas-liquid mass transfer on the operation of small electrochemical cells for the quantitative evaluation of CO2 reduction electrocatalysts. Phys. Chem. Chem. Phys. 2016, 18, 26777–26785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomisaki, M.; Kasahara, S.; Natsui, K.; Ikemiya, N.; Einaga, Y. Switchable product selectivity in the electrochemical reduction of carbon dioxide using boron-doped diamond electrodes. J. Am. Chem. Soc. 2019, 141, 7414–7420. [Google Scholar] [CrossRef] [PubMed]
- Hori, Y. Modern Aspects of Electrochemistry; Vayenas, C.G., White, R.E., Gamboa-Aldeco, M.E., Eds.; Springer: New York, NY, USA, 2008. [Google Scholar]
- Taheri, A.; Berben, L.A. Tailoring electrocatalysts for selective CO2 or H+ reduction: Iron carbonyl clusters as a case study. Inorg. Chem. 2016, 55, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Waldie, K.M.; Ostericher, A.L.; Reineke, M.H.; Sasayama, A.F.; Kubiak, C.P. Hydricity of transition-metal hydrides: Thermodynamic considerations for CO2 reduction. ACS Catal. 2018, 8, 1313–1324. [Google Scholar] [CrossRef] [Green Version]
- Kortlever, R.; Shen, J.; Schouten, K.J.P.; Calle-Vallejo, F.; Koper, M.T.M. Catalysts and reaction pathways for the electrochemical reduction of carbon dioxide. J. Phys. Chem. Lett. 2015, 6, 4073–4082. [Google Scholar] [CrossRef]
- Yu, X.; Pickup, P.G. Recent advances in direct formic acid fuel cells (DFAFC). J. Power Sources 2008, 182, 124–132. [Google Scholar] [CrossRef]
- Feaster, J.T.; Shi, C.; Cave, E.R.; Hatsukade, T.; Abram, D.N.; Kuhl, K.P.; Hahn, C.; Nørskov, J.K.; Jaramillo, T.F. Understanding selectivity for the electrochemical reduction of carbon dioxide to formic acid and carbon monoxide on metal electrodes. ACS Catal. 2017, 7, 4822–4827. [Google Scholar] [CrossRef]
- Shinagawa, T.; Larrazábal, G.O.; Martín, A.J.; Krumeich, F.; Pérez-Ramírez, J. Sulfur-modified copper catalysts for the electrochemical reduction of carbon dioxide to formate. ACS Catal. 2018, 8, 837–844. [Google Scholar] [CrossRef] [Green Version]
- Huan, T.N.; Simon, P.; Rousse, G.; Génois, I.; Artero, V.; Fontecave, M. Porous dendritic copper: An electrocatalyst for highly selective CO2 reduction to formate in water/ionic liquid electrolyte. Chem. Sci. 2017, 8, 742–747. [Google Scholar] [CrossRef] [Green Version]
- Li, C.W.; Ciston, J.; Kanan, M.W. Electroreduction of carbon monoxide to liquid fuel on oxide-derived nanocrystalline copper. Nature 2014, 508, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Kuhl, K.P.; Cave, E.R.; Abram, D.N.; Jaramillo, T.F. New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces. Energy Environ. Sci. 2012, 5, 7050–7059. [Google Scholar] [CrossRef]
- Hori, Y.; Murata, A.; Takahashi, R. Formation of hydrocarbons in the electrochemical reduction of carbon dioxide at a copper electrode in aqueous solution. J. Chem. Soc. Faraday Trans. 1989, 1, 2309–2326. [Google Scholar] [CrossRef]
- Hori, Y.; Takahashi, R.; Yoshinami, Y.; Murata, A. Electrochemical reduction of CO at a copper electrode. J. Phys. Chem. B 1997, 101, 7075–7081. [Google Scholar] [CrossRef]
- Hori, Y.; Murata, A.; Takahashi, R.; Suzuki, S. Electroreduction of CO to CH4 and C2H4 at a copper electrode in aqueous solutions at ambient temperature and pressure. J. Am. Chem. Soc. 1987, 109, 5022–5023. [Google Scholar] [CrossRef]
- Li, C.W.; Kanan, M.W. CO2 reduction at low overpotential on Cu electrodes resulting from the reduction of thick Cu2O films. J. Am. Chem. Soc. 2012, 134, 7231–7234. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Cao, Z.; Liu, W.; Nichols, E.M.; Smith, P.T.; Derrick, J.S.; Liu, Y.S.; Liu, J.; Wen, X.; Chang, C.J. Supramolecular porphyrin cages assembled at molecular–materials interfaces for electrocatalytic co reduction. ACS Cent. Sci. 2017, 3, 1032–1040. [Google Scholar] [CrossRef] [Green Version]
- Bertheussen, E.; Verdaguer-Casadevall, A.; Ravasio, D.; Montoya, J.H.; Trimarco, D.B.; Roy, C.; Meier, S.; Wendland, J.; Nørskov, J.K.; Stephens, I.E.L.; et al. Acetaldehyde as an intermediate in the electroreduction of carbon monoxide to ethanol on oxide-derived copper. Angew. Chem. Int. Ed. 2016, 55, 1450–1454. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Z.; Zhang, Y.; Li, Y.; Zhao, H.; Quan, X. Electrochemical reduction of carbondioxide to formate with Fe-C electrodes in anaerobic sludge digestion process. Water Res. 2016, 106, 339–343. [Google Scholar] [CrossRef]
- Lu, M.; Li, Q.; Liu, J.; Zhang, F.M.; Zhang, L.; Wang, J.L.; Kang, Z.H.; Lan, Y.Q. Installing earth-abundant metal active centers to covalent organic frameworks for efficient heterogeneous photocatalytic CO2 reduction. Appl. Catal. B Environ. 2019, 254, 624–633. [Google Scholar] [CrossRef]
- Swierk, J.R.; Mallouk, T.E. Design and development of photoanodes for water-splitting dye-sensitized photoelectrochemical cells. Chem. Soc. Rev. 2013, 42, 2357–2387. [Google Scholar] [CrossRef] [PubMed]
- Todorova, T.K.; Huan, T.N.; Wang, X.; Agarwala, H.; Fontecave, M. Controlling hydrogen evolution during photoreduction of CO2 to formic acid using [Rh(R-bpy)(Cp*)Cl](+) catalysts: A structure-activity study. Inorg. Chem. 2019, 58, 6893–6903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chardon-Noblat, S.; Deronzier, A.; Ziessel, R.; Zsoldos, D. Electroreduction of CO2 catalyzed by polymeric [Ru(bpy)(CO)2]n films in aqueous media: Parameters influencing the reaction selectivity. J. Electroanal. Chem. 1998, 444, 253–260. [Google Scholar] [CrossRef]
- Chen, J.; Yin, J.; Zheng, X.P.; Ahsaine, H.A.; Zhou, Y.; Dong, C.W.; Mohammed, O.F.; Takanabe, K.; Bakr, O.M. Compositionally screened eutectic catalytic coatings on halide perovskite photocathodes for photoassisted selective CO2 reduction. ACS Energy Lett. 2019, 4, 1279–1286. [Google Scholar] [CrossRef] [Green Version]
- Ajmal, S.; Yang, Y.; Li, K.J.; Tahir, M.A.; Liu, Y.Y.; Wang, T.; Bacha, A.U.R.; Feng, Y.Q.; Deng, Y.; Zhang, L.W. Zinc-modified copper catalyst for efficient (photo-) electrochemical CO2 reduction with high selectivity of HCOOH production. J. Phys. Chem. C 2019, 123, 11555–11563. [Google Scholar] [CrossRef]
- An, X.W.; Li, S.S.; Yoshida, A.; Wang, Z.D.; Hao, X.G.; Abudula, A.; Guan, G.Q. Electrodeposition of tin-based electrocatalysts with different surface tin species distributions for electrochemical reduction of CO2 to HCOOH. ACS Sustain. Chem. Eng. 2019, 7, 9360–9368. [Google Scholar] [CrossRef]
- Chatterjee, S.; Griego, C.; Hart, J.L.; Li, Y.W.; Taheri, M.L.; Keith, J.; Snyder, J.D. Free standing nanoporous palladium alloys as CO poisoning tolerant electrocatalysts for the electrochemical reduction of CO2 to formate. ACS Catal. 2019, 6, 5290–5301. [Google Scholar] [CrossRef] [Green Version]
- Raju, R.K.; Rodriguez, P.; Johnston, R.L. Can a single valence electron alter the electrocatalytic activity and selectivity for CO2 reduction on the subnanometer scale? J. Phys. Chem. C 2019, 123, 14591–14609. [Google Scholar] [CrossRef]
- Qin, C.L.; Fan, A.X.; Zhang, X.; Dai, X.P.; Sun, H.; Ren, D.H.; Dong, Z.; Wang, Y.; Luan, C.L.; Ye, J.Y.; et al. The in situ etching assisted synthesis of Pt-Fe-Mn ternary alloys with high-index facets as efficient catalysts for electro-oxidation reactions. Nanoscale 2019, 11, 9061–9075. [Google Scholar] [CrossRef]
- Pan, F.P.; Li, B.Y.; Deng, W.; Du, Z.C.; Gang, Y.; Wang, G.F.; Li, Y. Promoting electrocatalytic CO2 reduction on nitrogen-doped carbon with sulfur addition. Appl. Catal. B Environ. 2019, 252, 240–249. [Google Scholar] [CrossRef]
- Yin, Y.; Gao, D.F.; Yao, S.Y.; Zhao, B.; Cai, F.; Lin, L.L.; Tang, P.; Zhai, P.; Wang, G.X.; Ma, D.; et al. Highly selective palladium-copper bimetallic electrocatalysts for the electrochemical reduction of CO2 to CO. Nano Energy 2016, 27, 35–43. [Google Scholar] [CrossRef]
- Gong, Q.F.; Ding, P.; Xu, M.Q.; Zhu, X.R.; Wang, M.Y.; Deng, J.; Ma, Q.; Han, N.; Zhu, Y.; Lu, J.; et al. Structural defects on converted bismuth oxide nanotubes enable highly active electrocatalysis of carbon dioxide reduction. Nat. Comm. 2019, 10, 2807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, D.; Zhou, H.; Wang, J.; Miao, S.; Yang, F.; Wang, G.; Wang, J.; Bao, X. Size-dependent electrocatalytic reduction of CO2 over Pd nanoparticles. J. Am. Chem. Soc. 2015, 137, 4288–4291. [Google Scholar] [CrossRef]
- Jiang, B.; Zhang, X.G.; Jiang, K.; Wu, D.Y.; Cai, W.B. Boosting formate production in electrocatalytic CO2 reduction over wide potential window on Pd surfaces. J. Am. Chem. Soc. 2018, 140, 2880–2889. [Google Scholar] [CrossRef] [PubMed]
- Eder, G.M.; Pyles, D.A.; Wolfson, E.R.; McGrier, P.L. A ruthenium porphyrin-based porous organic polymer for the hydrosilylative reduction of CO2 to formate. Chem. Comm. 2019, 55, 7195–7198. [Google Scholar] [CrossRef]
- Weetman, C.; Bag, P.; Szilvasi, T.; Jandl, C.; Inoue, S. CO2 Fixation and catalytic reduction by a neutral aluminum double bond. Angew. Chem. Int. Ed. 2019, 58, 10961–10965. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Wang, T.H.; Wang, Y.B.; Hao, R.L.; Wang, H.; Han, Y.H. Catalytic reduction of CO2 to HCO2− by nanoscale nickel-based bimetallic alloy under atmospheric pressure. J. Ind. Eng. Chem. 2019, 77, 291–302. [Google Scholar] [CrossRef]
- Porosoff, M.D.; Yan, B.; Chen, J.G. Catalytic reduction of CO2 by H2 for synthesis of CO, methanol and hydrocarbons: Challenges and opportunities. Energy Environ. Sci. 2016, 9, 62–73. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Liu, P.; Stacchiola, D.J.; Senanayake, S.D.; White, M.G.; Chen, J.G. Hydrogenation of CO2 to methanol: Importance of metal-oxide and metal-carbide interfaces in the activation of CO2. ACS Catal. 2015, 5, 6696–6706. [Google Scholar] [CrossRef]
- Gao, P.; Dang, S.; Li, S.; Bu, X.; Liu, Z.; Qiu, M.; Yang, C.; Wang, H.; Zhong, L.; Han, Y.; et al. Direct production of lower olefins from CO2 conversion via bifunctional catalysis. ACS Catal. 2018, 8, 571–578. [Google Scholar] [CrossRef]
- Sun, K.; Fan, Z.; Ye, J.; Yan, J.; Ge, Q.; Li, Y.; He, W.; Yang, W.; Liu, C. Hydrogenation of CO2 to methanol over In2O3 catalyst. J. CO2 Util. 2015, 12, 1–6. [Google Scholar] [CrossRef]
- Gnanamani, M.K.; Jacobs, G.; Hamdeh, H.H.; Shafer, W.D.; Liu, F.; Hopps, S.D.; Thomas, G.A.; Davis, B.H. Hydrogenation of carbon dioxide over Co-Fe bimetallic catalysts. ACS Catal. 2016, 6, 913–927. [Google Scholar] [CrossRef]
- Dreyer, J.A.H.; Li, P.; Zhang, L.; Beh, G.K.; Zhang, R.; Sit, P.H.L.; Teoh, W.Y. Influence of the oxide support reducibility on the CO2 methanation over Ru-based catalysts. Appl. Catal. B Environ. 2017, 219, 715–726. [Google Scholar] [CrossRef]
- Li, W.; Nie, X.; Jiang, X.; Zhang, A.; Ding, F.; Liu, M.; Liu, Z.; Guo, X.; Song, C. ZrO2 support imparts superior activity and stability of Co catalysts for CO2 methanation. Appl. Catal. B Environ. 2018, 220, 397–408. [Google Scholar] [CrossRef]
- Zhan, G.; Zeng, H.C. ZIF-67-derived nanoreactors for controlling product selectivity in CO2 hydrogenation. ACS Catal. 2017, 7, 7509–7519. [Google Scholar] [CrossRef]
- Li, Y.; Cai, X.; Chen, S.; Zhang, H.; Zhang, K.H.L.; Hong, J.; Chen, B.; Kuo, D.H.; Wang, W. Highly dispersed metal carbide on ZIF-derived pyridinic-N-doped carbon for CO2 enrichment and selective hydrogenation. ChemSusChem 2018, 11, 1040–1047. [Google Scholar] [CrossRef] [PubMed]
- Kattel, S.; Yu, W.; Yang, X.; Yan, B.; Huang, Y.; Wan, W.; Liu, P.; Chen, J.G. CO2 hydrogenation over oxide-supported PtCo catalysts: The role of the oxide support in determining the product selectivity. Angew. Chem. Int. Ed. 2016, 55, 7968–7973. [Google Scholar] [CrossRef]
- Ren, J.; Qin, X.; Yang, J.Z.; Qin, Z.F.; Guo, H.L.; Lin, J.Y.; Li, Z. Methanation of carbon dioxide over Ni-M/ZrO2 (M = Fe, Co, Cu) catalysts: Effect of addition of a second metal. Fuel Process. Technol. 2015, 137, 204–211. [Google Scholar] [CrossRef]
- Kirchner, J.; Anolleck, J.K.; Loesch, H.; Kureti, S. Methanation of CO2 on iron based catalysts. Appl. Catal. B Environ. 2018, 223, 47–59. [Google Scholar] [CrossRef]
- Bi, Q.; Huang, X.; Yin, G.; Chen, T.; Du, X.; Cai, J.; Xu, J.; Liu, Z.; Han, Y.; Huang, F. Cooperative catalysis of nickel and nickel oxide for efficient reduction of CO2 to CH4. ChemCatChem 2019, 11, 1295–1302. [Google Scholar] [CrossRef]
- Dietz, L.; Piccinin, S.; Maestri, M. Mechanistic insights into CO2 activation via reverse water-gas shift on metal surfaces. J. Phys. Chem. C 2015, 119, 4959–4966. [Google Scholar] [CrossRef]
- Kattel, S.; Yan, B.; Chen, J.G.; Liu, P. CO2 hydrogenation on Pt, Pt/SiO2 and Pt/TiO2: Importance of synergy between Pt and oxide support. J. Catal. 2016, 343, 115–126. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Su, X.; Chen, X.; Duan, H.; Liang, B.; Liu, Q.; Liu, X.; Ren, Y.; Huang, Y.; Zhang, T. Promotion effects of potassium on the activity and selectivity of Pt/zeolite catalysts for reverse water gas shift reaction. Appl. Catal. B Environ. 2017, 216, 95–105. [Google Scholar] [CrossRef]
- Wang, X.; Shi, H.; Kwak, J.H.; Szanyi, J. Mechanism of CO2 hydrogenation on Pd/Al2O3 catalysts: Kinetics and transient DRIFTS-MS studies. ACS Catal. 2015, 5, 6337–6349. [Google Scholar] [CrossRef]
- Guo, Y.; Mei, S.; Yuan, K.; Wang, D.J.; Liu, H.C.; Yan, C.H.; Zhang, Y.W. Low-temperature CO2 methanation over CeO2-supported Ru single atoms, nanoclusters, and nanoparticles competitively tuned by strong metal-support interactions and H-spillover effect. ACS Catal. 2018, 8, 6203–6215. [Google Scholar] [CrossRef]
- Xu, J.; Su, X.; Duan, H.; Hou, B.; Lin, Q.; Liu, X.; Pan, X.; Pei, G.; Geng, H.; Huang, Y.; et al. Influence of pretreatment temperature on catalytic performance of rutile TiO2-supported ruthenium catalyst in CO2 methanation. J. Catal. 2016, 333, 227–237. [Google Scholar] [CrossRef]
- Wu, H.C.; Chang, Y.C.; Wu, J.H.; Lin, J.H.; Lin, I.K.; Chen, C.S. Methanation of CO2 and reverse water gas shift reactions on Ni/SiO2 catalysts: The influence of particle size on selectivity and reaction pathway. Catal. Sci. Technol. 2015, 5, 4154–4163. [Google Scholar] [CrossRef]
- Ren, H.; Xu, C.H.; Zhao, H.Y.; Wang, Y.X.; Liu, J.; Liu, J.Y. Methanol synthesis from CO2 hydrogenation over Cu/gamma-Al2O3 catalysts modified by ZnO, ZrO2 and MgO. J. Ind. Eng. Chem. 2015, 28, 261–267. [Google Scholar] [CrossRef]
- Hus, M.; Dasireddy, V.D.B.C.; Stefancic, N.S.; Likozar, B. Mechanism, kinetics and thermodynamics of carbon dioxide hydrogenation to methanol on Cu/ZnAl2O4 spinel-type heterogeneous catalysts. Appl. Catal. B Environ. 2017, 207, 267–278. [Google Scholar] [CrossRef]
- Studt, F.; Behrens, M.; Kunkes, E.L.; Thomas, N.; Zander, S.; Tarasov, A.; Schumann, J.; Frei, E.; Varley, J.B.; Abild-Pedersen, F.; et al. The mechanism of CO and CO2 hydrogenation to methanol over Cu-based catalysts. ChemCatChem 2015, 7, 1105–1111. [Google Scholar] [CrossRef] [Green Version]
- Kunkes, E.L.; Studt, F.; Abild-Pedersen, F.; Schloegl, R.; Behrens, M. Hydrogenation of CO2 to methanol and CO on Cu/ZnO/Al2O3: Is there a common intermediate or not? J. Catal. 2015, 328, 43–48. [Google Scholar] [CrossRef] [Green Version]
- Karelovic, A.; Ruiz, P. The role of copper particle size in low pressure methanol synthesis via CO2 hydrogenation over Cu/ZnO catalysts. Catal. Sci. Technol. 2015, 5, 869–881. [Google Scholar] [CrossRef]
- Ro, I.; Liu, Y.; Ball, M.R.; Jackson, D.H.K.; Chada, J.P.; Sener, C.; Kuech, T.F.; Madon, R.J.; Huber, G.W.; Dumesic, J.A. Role of the Cu-ZrO2 interfacial sites for conversion of ethanol to ethyl acetate and synthesis of methanol from CO2 and H2. ACS Catal. 2016, 6, 7040–7050. [Google Scholar] [CrossRef]
- Phongamwong, T.; Chantaprasertporn, U.; Witoon, T.; Numpilai, T.; Poo-Arporn, Y.; Limphirat, W.; Donphai, W.; Dittanet, P.; Chareonpanich, M.; Limtrakul, J. CO2 hydrogenation to methanol over CuO-ZnO-ZrO2-SiO2 catalysts: Effects of SiO2 contents. Chem. Eng. J. 2017, 316, 692–703. [Google Scholar] [CrossRef]
- Jiang, X.; Koizumi, N.; Guo, X.; Song, C. Bimetallic Pd-Cu catalysts for selective CO2 hydrogenation to methanol. Appl. Catal. B Environ. 2015, 170, 173–185. [Google Scholar] [CrossRef]
- Bahruji, H.; Bowker, M.; Hutchings, G.; Dimitratos, N.; Wells, P.; Gibson, E.; Jones, W.; Brookes, C.; Morgan, D.; Lalev, G. Pd/ZnO catalysts for direct CO2 hydrogenation to methanol. J. Catal. 2016, 343, 133–146. [Google Scholar] [CrossRef] [Green Version]
- Nie, X.; Jiang, X.; Wang, H.; Luo, W.; Janik, M.J.; Chen, Y.; Guo, X.; Song, C. Mechanistic understanding of alloy effect and water promotion for Pd-Cu bimetallic catalysts in CO2 hydrogenation to methanol. ACS Catal. 2018, 8, 4873–4892. [Google Scholar] [CrossRef]
- Stangeland, K.; Kalai, D.Y.; Ding, Y.; Yu, Z. Mesoporous manganese-cobalt oxide spinel catalysts for CO2 hydrogenation to methanol. J. CO2 Util. 2019, 32, 146–154. [Google Scholar] [CrossRef]
- Martin, O.; Martín, A.J.; Mondelli, C.; Mitchell, S.; Segawa, T.F.; Hauert, R.; Drouilly, C.; Curulla-Ferré, D.; Pérez-Ramírez, J. Indium oxide as a superior catalyst for methanol synthesis by CO2 Hydrogenation. Angew. Chem. Int. Ed. 2016, 55, 6261–6265. [Google Scholar] [CrossRef]
- Wei, J.; Ge, Q.; Yao, R.; Wen, Z.; Fang, C.; Guo, L.; Xu, H.; Sun, J. Directly converting CO2 into a gasoline fuel. Nat. Comm. 2017, 8, 15174. [Google Scholar] [CrossRef] [Green Version]
- Visconti, C.G.; Martinelli, M.; Falbo, L.; Infantes-Molina, A.; Lietti, L.; Forzatti, P.; Iaquaniello, G.; Palo, E.; Picutti, B.; Brignoli, F. CO2 hydrogenation to lower olefins on a high surface area K-promoted bulk Fe-catalyst. Appl. Catal. B Environ. 2017, 200, 530–542. [Google Scholar] [CrossRef]
- Albrecht, M.; Rodemerck, U.; Schneider, M.; Broering, M.; Baabe, D.; Kondratenko, E.V. Unexpectedly efficient CO2 hydrogenation to higher hydrocarbons over non-doped Fe2O3. Appl. Catal. B Environ. 2017, 204, 119–126. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, A.; Liu, M.; Hu, S.; Ding, F.; Song, C.; Guo, X. Fe-MOF-derived highly active catalysts for carbon dioxide hydrogenation to valuable hydrocarbons. J. CO2 Util. 2017, 21, 100–107. [Google Scholar] [CrossRef]
- Choi, Y.H.; Jang, Y.J.; Park, H.; Kim, W.Y.; Lee, Y.H.; Choi, S.H.; Lee, J.S. Carbon dioxide Fischer-Tropsch synthesis: A new path to carbon-neutral fuels. Appl. Catal. B Environ. 2017, 202, 605–610. [Google Scholar] [CrossRef]
- Wei, J.; Sun, J.; Wen, Z.; Fang, C.; Ge, Q.; Xu, H. New insights into the effect of sodium on Fe3O4-based nanocatalysts for CO2 hydrogenation to light olefins. Catal. Sci. Technol. 2016, 6, 4786–4793. [Google Scholar] [CrossRef]
- Satthawong, R.; Koizumi, N.; Song, C.; Prasassarakich, P. Light olefin synthesis from CO2 hydrogenation over K-promoted Fe-Co bimetallic catalysts. Catal. Today 2015, 251, 34–40. [Google Scholar] [CrossRef]
- Gao, P.; Li, S.; Bu, X.; Dang, S.; Liu, Z.; Wang, H.; Zhong, L.; Qiu, M.; Yang, C.; Cai, J.; et al. Direct conversion of CO2 into liquid fuels with high selectivity over a bifunctional catalyst. Nat. Chem. 2017, 9, 1019–1024. [Google Scholar] [CrossRef]
- Cheah, W.Y.; Show, P.L.; Chang, J.S.; Ling, T.C.; Juan, J.C. Biosequestration of atmospheric CO2 and flue gas-containing CO2 by microalgae. Bioresour. Technol. 2015, 184, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Yadav, G.; Sen, R. Microalgal green refinery concept for biosequestration of carbon-dioxide vis-à-vis wastewater remediation and bioenergy production: Recent technological advances in climate research. J. CO2 Util. 2017, 17, 188–206. [Google Scholar] [CrossRef]
- Kuo, C.M.; Jian, J.F.; Sun, Y.L.; Lin, T.H.; Yang, Y.C.; Zhang, W.X.; Chang, H.F.; Lai, J.T.; Chang, J.S.; Lin, C.S. An efficient photobioreactors/raceway circulating system combined with alkaline-CO2 capturing medium for microalgal cultivation. Bioresour. Technol. 2018, 266, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.; Shi, F. Bioreactor design for algal growth as a sustainable energy source. In Reactor and Process Design in Sustainable Energy Technology; Elsevier: Amsterdam, The Netherlands, 2014; pp. 27–60. [Google Scholar]
- Xie, Y.P.; Ho, S.H.; Chen, C.Y.; Chen, C.N.N.; Liu, C.C.; Ng, I.S.; Jing, K.J.; Yang, S.C.; Chen, C.H.; Chang, J.S.; et al. Simultaneous enhancement of CO2 fixation and lutein production with thermo-tolerant Desmodesmus sp. F51 using a repeated fed-batch cultivation strategy. Biochem. Eng. J. 2014, 86, 33–40. [Google Scholar] [CrossRef]
- Gonçalves, A.L.; Rodrigues, C.M.; Pires, J.C.M.; Simões, M. The effect of increasing CO2 concentrations on its capture, biomass production and wastewater bioremediation by microalgae and cyanobacteria. Algal Res. 2016, 14, 127–136. [Google Scholar] [CrossRef]
- Chen, H.; Wang, J.; Zheng, Y.; Zhan, J.; He, C.; Wang, Q. Algal biofuel production coupled bioremediation of biomass power plant wastes based on Chlorella sp C2 cultivation. Appl. Energy 2018, 211, 296–305. [Google Scholar] [CrossRef]
- Jain, D.; Ghonse, S.S.; Trivedi, T.; Fernandes, G.L.; Menezes, L.D.; Damare, S.R.; Mamatha, S.S.; Kumar, S.; Gupta, V. CO2 fixation and production of biodiesel by Chlorella vulgaris NIOCCV under mixotrophic cultivation. Bioresour. Technol. 2019, 273, 672–676. [Google Scholar] [CrossRef]
- Kumar, K.; Banerjee, D.; Das, D. Carbon dioxide sequestration from industrial flue gas by Chlorella sorokiniana. Bioresour. Technol. 2014, 152, 225–233. [Google Scholar] [CrossRef]
- Tripathi, R.; Singh, J.; Thakur, I.S. Characterization of microalga Scenedesmus sp ISTGA1 for potential CO2 sequestration and biodiesel production. Renew. Energy 2015, 74, 774–781. [Google Scholar] [CrossRef]
- Varshney, P.; Beardall, J.; Bhattacharya, S.; Wangikar, P.P. Isolation and biochemical characterisation of two thermophilic green algal species- Asterarcys quadricellulare and Chlorella sorokiniana, which are tolerant to high levels of carbon dioxide and nitric oxide. Algal Res. 2018, 30, 28–37. [Google Scholar] [CrossRef]
- Yun, H.S.; Ji, M.K.; Park, Y.T.; Salama, E.S.; Choi, J. Microalga, Acutodesmus obliquus KGE 30 as a potential candidate for CO2 mitigation and biodiesel production. Environ. Sci. Pollut. Res. 2016, 23, 17831–17839. [Google Scholar] [CrossRef]
- Bhakta, J.N.; Lahiri, S.; Pittman, J.K.; Jana, B.B. Carbon dioxide sequestration in wastewater by a consortium of elevated carbon dioxide-tolerant microalgae. J. CO2 Util. 2015, 10, 105–112. [Google Scholar] [CrossRef]
- Kuo, C.M.; Lin, T.H.; Yang, Y.C.; Zhang, W.X.; Lai, J.T.; Wu, H.T.; Chang, J.S.; Lin, C.S. Ability of an alkali-tolerant mutant strain of the microalga Chlorella sp. AT1 to capture carbon dioxide for increasing carbon dioxide utilization efficiency. Bioresour. Technol. 2017, 244, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Liu, J.; Ma, X.; Guo, B.; Liu, B.; Wu, T.; Jiang, Y.; Chen, F. Genetic engineering of the Calvin cycle toward enhanced photosynthetic CO2 fixation in microalgae. Biotechnol. Biofuels 2017, 10, 229. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wang, L.; Zhao, Q.; Wei, W.; Sun, Y. Improving high carbon dioxide tolerance and carbon dioxide fixation capability of Chlorella sp by adaptive laboratory evolution. Bioresour. Technol. 2015, 185, 269–275. [Google Scholar] [CrossRef]
- Bilanovic, D.; Holland, M.; Starosvetsky, J.; Armon, R. Co-cultivation of microalgae and nitrifiers for higher biomass production and better carbon capture. Bioresour. Technol. 2016, 220, 282–288. [Google Scholar] [CrossRef]
- Aslam, A.; Thomas-Hall, S.R.; Mughal, T.A.; Schenk, P.M. Selection and adaptation of microalgae to growth in 100% unfiltered coal-fired flue gas. Bioresour. Technol. 2017, 233, 271–283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nayak, M.; Suh, W.I.; Lee, B.; Chang, Y.K. Enhanced carbon utilization efficiency and FAME production of Chlorella sp HS2 through combined supplementation of bicarbonate and carbon dioxide. Energy Convers. Manag. 2018, 156, 45–52. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhang, W.; Wang, J.; Chen, Y.; Shen, S.; Liu, T. Utilization of simulated flue gas for cultivation of Scenedesmus dimorphus. Bioresour. Technol. 2013, 128, 359–364. [Google Scholar] [CrossRef]
- Vo, H.N.P.; Bui, X.T.; Nguyen, T.T.; Nguyen, D.D.; Dao, T.S.; Cao, N.D.T.; Vo, T.K.Q. Effects of nutrient ratios and carbon dioxide bio-sequestration on biomass growth of Chlorella sp in bubble column photobioreactor. J. Environ. Manag. 2018, 219, 1–8. [Google Scholar] [CrossRef]
- Liu, X.; Ying, K.; Chen, G.; Zhou, C.; Zhang, W.; Zhang, X.; Cai, Z.; Holmes, T.; Tao, Y. Growth of Chlorella vulgaris and nutrient removal in the wastewater in response to intermittent carbon dioxide. Chemosphere 2017, 186, 977–985. [Google Scholar] [CrossRef]
- Posadas, E.; Morales, M.d.M.; Gomez, C.; Acién, F.G.; Muñoz, R. Influence of pH and CO2 source on the performance of microalgae-based secondary domestic wastewater treatment in outdoors pilot raceways. Chem. Eng. J. 2015, 265, 239–248. [Google Scholar] [CrossRef] [Green Version]
- Kuo, C.M.; Jian, J.F.; Lin, T.H.; Chang, Y.B.; Wan, X.H.; Lai, J.T.; Chang, J.S.; Lin, C.S. Simultaneous microalgal biomass production and CO2 fixation by cultivating Chlorella sp. GD with aquaculture wastewater and boiler flue gas. Bioresour. Technol. 2016, 221, 241–250. [Google Scholar] [CrossRef]
- Eze, V.C.; Velasquez-Orta, S.B.; Hernandez-Garcia, A.; Monje-Ramirez, I.; Orta-Ledesma, M.T. Kinetic modelling of microalgae cultivation for wastewater treatment and carbon dioxide sequestration. Algal Res. 2018, 32, 131–141. [Google Scholar] [CrossRef]
- Fu, W.; Gudmundsson, S.; Wichuk, K.; Palsson, S.; Palsson, B.O.; Salehi-Ashtiani, K.; Brynjolfsson, S. Sugar-stimulated CO2 sequestration by the green microalga Chlorella vulgaris. Sci. Total Environ. 2019, 654, 275–283. [Google Scholar] [CrossRef]
- Dineshkumar, R.; Subramanian, G.; Dash, S.K.; Sen, R. Development of an optimal light-feeding strategy coupled with semi-continuous reactor operation for simultaneous improvement of microalgal photosynthetic efficiency, lutein production and CO2 sequestration. Biochem. Eng. J. 2016, 113, 47–56. [Google Scholar] [CrossRef]
- Jacob-Lopes, E.; Franco, T.T. From oil refinery to microalgal biorefinery. J. CO2 Util. 2013, 2, 1–7. [Google Scholar] [CrossRef]
- Kassim, M.A.; Meng, T.K. Carbon dioxide (CO2) biofixation by microalgae and its potential for biorefinery and biofuel production. Sci. Total Environ. 2017, 584, 1121–1129. [Google Scholar] [CrossRef]
- Kao, C.Y.; Chen, T.Y.; Chang, Y.B.; Chiu, T.W.; Lin, H.Y.; Chen, C.D.; Chang, J.S.; Lin, C.S. Utilization of carbon dioxide in industrial flue gases for the cultivation of microalga Chlorella sp. Bioresour. Technol. 2014, 166, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Sun, Y.; Liao, Q.; Fu, Q.; Xia, A.; Zhu, X. Improvement on light penetrability and microalgae biomass production by periodically pre-harvesting Chlorella vulgaris cells with culture medium recycling. Bioresour. Technol. 2016, 216, 669–676. [Google Scholar] [CrossRef]
- Moheimani, N.R. Tetraselmis suecica culture for CO2 bioremediation of untreated flue gas from a coal-fired power station. J. Appl. Phycol. 2016, 28, 2139–2146. [Google Scholar] [CrossRef]
- Ou-Yang, C.; Chen, H.W.; Ho, C.H.; Chou, J.C.; Yuan, Y.T.; Ho, C.L.; Hsueh, H.T.; Chen, S.T.; Liao, P.C.; Chao, L.K. Value chain analysis of algal bioenergy and carbon capture integrated with a biotechnology innovation. J. Clean. Prod. 2018, 180, 349–359. [Google Scholar] [CrossRef]
- Zhu, L.D.; Xu, Z.B.; Qin, L.; Wang, Z.M.; Hiltunen, E.; Li, Z.H. Oil production from pilot-scale microalgae cultivation: An economics evaluation. Energy Sources Part B Econ. Plan. Policy 2016, 11, 11–17. [Google Scholar] [CrossRef]
- Rezvani, S.; Moheimani, N.R.; Bahri, P.A. Techno-economic assessment of CO2 bio-fixation using microalgae in connection with three different state-of-the-art power plants. Comput. Chem. Eng. 2016, 84, 290–301. [Google Scholar] [CrossRef] [Green Version]
- Venkata Mohan, S.; Modestra, J.A.; Amulya, K.; Butti, S.K.; Velvizhi, G. A circular bioeconomy with biobased products from CO2 sequestration. Trends Biotechnol. 2016, 34, 506–519. [Google Scholar] [CrossRef] [PubMed]
- Chew, K.W.; Yap, J.Y.; Show, P.L.; Suan, N.H.; Juan, J.C.; Ling, T.C.; Lee, D.J.; Chang, J.S. Microalgae biorefinery: High value products perspectives. Bioresour. Technol. 2017, 229, 53–62. [Google Scholar] [CrossRef]
- Duque-Acevedo, M.; Belmonte-Ureña, L.J.; Plaza-Úbeda, J.A.; Camacho-Ferre, F. The management of agricultural waste biomass in the framework of circular economy and bioeconomy: An opportunity for greenhouse agriculture in southeast Spain. Agronomy 2020, 10, 489. [Google Scholar] [CrossRef] [Green Version]
- Dębowski, M.; Zieliński, M.; Kazimierowicz, J.; Kujawska, N.; Talbierz, S. Microalgae cultivation technologies as an opportunity for bioenergetic system development—Advantages and limitations. Sustainability 2020, 12, 9980. [Google Scholar] [CrossRef]
- Khan, M.I.; Shin, J.H.; Kim, J.D. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb. Cell Factories 2018, 17, 36. [Google Scholar] [CrossRef]
- Alobwede, E.; Leake, J.R.; Pandhal, J. Circular economy fertilization: Testing micro and macro algal species as soil improvers and nutrient sources for crop production in greenhouse and field conditions. Geoderma 2019, 334, 113–123. [Google Scholar] [CrossRef]
- Sprague, M.; Dick, J.R.; Tocher, D.R. Impact of sustainable feeds on omega-3 long-chain fatty acid levels in farmed Atlantic salmon, 2006–2015. Sci. Rep. 2016, 6, 21892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dineshbabu, G.; Goswami, G.; Kumar, R.; Sinha, A.; Das, D. Microalgae—Nutritious, sustainable aqua- and animal feed source. J. Funct. Foods 2019, 62, 103545. [Google Scholar] [CrossRef]
- Shakya, R.; Whelen, J.; Adhikari, S.; Mahadevan, R.; Neupane, S. Effect of temperature and Na2CO3 catalyst on hydrothermal liquefaction of algae. Algal Res. 2015, 12, 80–90. [Google Scholar] [CrossRef] [Green Version]
- Gai, C.; Zhang, Y.; Chen, W.T.; Zhang, P.; Dong, Y. An investigation of reaction pathways of hydrothermal liquefaction using Chlorella pyrenoidosa and Spirulina platensis. Energy Convers. Manag. 2015, 96, 330–339. [Google Scholar] [CrossRef]
- Chen, Y.; Mu, R.; Yang, M.; Fang, L.; Wu, Y.; Wu, K.; Liu, Y.; Gong, J. Catalytic hydrothermal liquefaction for bio-oil production over CNTs supported metal catalysts. Chem. Eng. Sci. 2017, 161, 299–307. [Google Scholar] [CrossRef]
- López Barreiro, D.; Bauer, M.; Hornung, U.; Posten, C.; Kruse, A.; Prins, W. Cultivation of microalgae with recovered nutrients after hydrothermal liquefaction. Algal Res. 2015, 9, 99–106. [Google Scholar] [CrossRef]
- Chen, W.H.; Lin, B.J.; Huang, M.Y.; Chang, J.S. Thermochemical conversion of microalgal biomass into biofuels: A review. Bioresour. Technol. 2015, 184, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Raheem, A.; Dupont, V.; Channa, A.Q.; Zhao, X.; Vuppaladadiyam, A.K.; Taufiq-Yap, Y.H.; Zhao, M.; Harun, R. Parametric characterization of air gasification of Chlorella vulgaris biomass. Energy Fuels 2017, 31, 2959–2969. [Google Scholar] [CrossRef] [Green Version]
- Tiong, L.; Komiyama, M.; Uemura, Y.; Nguyen, T.T. Catalytic supercritical water gasification of microalgae: Comparison of Chlorella vulgaris and Scenedesmus quadricauda. J. Supercrit. Fluids 2016, 107, 408–413. [Google Scholar] [CrossRef]
- Peng, G.; Vogel, F.; Refardt, D.; Ludwig, C. Catalytic supercritical water gasification: Continuous methanization of Chlorella vulgaris. Ind. Eng. Chem. Res. 2017, 56, 6256–6265. [Google Scholar] [CrossRef]
- Hu, Z.; Ma, X.; Jiang, E. The effect of microwave pretreatment on chemical looping gasification of microalgae for syngas production. Energy Convers. Manag. 2017, 143, 513–521. [Google Scholar] [CrossRef]
- Liu, G.; Liao, Y.; Wu, Y.; Ma, X. Synthesis gas production from microalgae gasification in the presence of Fe2O3 oxygen carrier and CaO additive. Appl. Energy 2018, 212, 955–965. [Google Scholar] [CrossRef]
- Azizi, K.; Keshavarz Moraveji, M.; Abedini Najafabadi, H. A review on bio-fuel production from microalgal biomass by using pyrolysis method. Renew. Sustain. Energy Rev. 2018, 82, 3046–3059. [Google Scholar] [CrossRef]
- Wang, X.; Sheng, L.; Yang, X. Pyrolysis characteristics and pathways of protein, lipid and carbohydrate isolated from microalgae Nannochloropsis sp. Bioresour. Technol. 2017, 229, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Guo, H.; Zhou, M.; Cui, M.; Dong, C.; Yang, Y. Selective preparation of monocyclic aromatic hydrocarbons from catalytic cracking of biomass fast pyrolysis vapors over Mo2N/HZSM-5 catalyst. Fuel Process. Technol. 2018, 173, 134–142. [Google Scholar] [CrossRef]
- Zainan, N.H.; Srivatsa, S.C.; Li, F.; Bhattacharya, S. Quality of bio-oil from catalytic pyrolysis of microalgae Chlorella vulgaris. Fuel 2018, 223, 12–19. [Google Scholar] [CrossRef]
- Zabed, H.M.; Qi, X.; Yun, J.; Zhang, H. Anaerobic digestion of microalgae biomass for methane production. In Microalgae Biotechnology for Development of Biofuel and Wastewater Treatment; Alam, M.A., Wang, Z., Eds.; Springer: Singapore, 2019; pp. 397–421. [Google Scholar]
- Wang, Y.; Guo, W.; Cheng, C.L.; Ho, S.H.; Chang, J.S.; Ren, N. Enhancing bio-butanol production from biomass of Chlorella vulgaris JSC-6 with sequential alkali pretreatment and acid hydrolysis. Bioresour. Technol. 2016, 200, 557–564. [Google Scholar] [CrossRef]
- Abomohra, A.E.F.; Elshobary, M. Biodiesel, bioethanol, and biobutanol production from microalgae. In Microalgae Biotechnology for Development of Biofuel and Wastewater Treatment; Alam, M.A., Wang, Z., Eds.; Springer: Singapore, 2019; pp. 293–321. [Google Scholar]
- Passos, F.; Carretero, J.; Ferrer, I. Comparing pretreatment methods for improving microalgae anaerobic digestion: Thermal, hydrothermal, microwave and ultrasound. Chem. Eng. J. 2015, 279, 667–672. [Google Scholar] [CrossRef]
- Bohutskyi, P.; Betenbaugh, M.J.; Bouwer, E.J. The effects of alternative pretreatment strategies on anaerobic digestion and methane production from different algal strains. Bioresour. Technol. 2014, 155, 366–372. [Google Scholar] [CrossRef]
- Passos, F.; Hom-Diaz, A.; Blanquez, P.; Vicent, T.; Ferrer, I. Improving biogas production from microalgae by enzymatic pretreatment. Bioresour. Technol. 2016, 199, 347–351. [Google Scholar] [CrossRef]
- Hernandez, D.; Riano, B.; Coca, M.; Garcia-Gonzalez, M.C. Saccharification of carbohydrates in microalgal biomass by physical, chemical and enzymatic pre-treatments as a previous step for bioethanol production. Chem. Eng. J. 2015, 262, 939–945. [Google Scholar] [CrossRef]
- Shokrkar, H.; Ebrahimi, S.; Zamani, M. Bioethanol production from acidic and enzymatic hydrolysates of mixed microalgae culture. Fuel 2017, 200, 380–386. [Google Scholar] [CrossRef]
- Cheng, L.M.; Lee, K.T.; Chan, D.J.C. Synergistic effect of pretreatment and fermentation process on carbohydrate-rich Scenedesmus dimorphus for bioethanol production. Energy Convers. Manag. 2017, 141, 410–419. [Google Scholar] [CrossRef]
- Dickinson, S.; Mientus, M.; Frey, D.; Amini-Hajibashi, A.; Ozturk, S.; Shaikh, F.; Sengupta, D.; El-Halwagi, M.M. A review of biodiesel production from microalgae. Clean Technol. Environ. Policy 2017, 19, 637–668. [Google Scholar] [CrossRef]
- Jeevan Kumar, S.P.; Vijay Kumar, G.; Dash, A.; Scholz, P.; Banerjee, R. Sustainable green solvents and techniques for lipid extraction from microalgae: A review. Algal Res. 2017, 21, 138–147. [Google Scholar] [CrossRef]
- Lai, Y.S.; De Francesco, F.; Aguinaga, A.; Parameswaran, P.; Rittmann, B.E. Improving lipid recovery from Scenedesmus wet biomass by surfactant-assisted disruption. Green Chem. 2016, 18, 1319–1326. [Google Scholar] [CrossRef] [Green Version]
- Orr, V.C.A.; Plechkova, N.V.; Seddon, K.R.; Rehmann, L. Disruption and wet extraction of the microalgae Chlorella vulgaris using room-temperature ionic liquids. ACS Sustain. Chem. Eng. 2016, 4, 591–600. [Google Scholar] [CrossRef]
- Lee, O.K.; Oh, Y.K.; Lee, E.Y. Bioethanol production from carbohydrate-enriched residual biomass obtained after lipid extraction of Chlorella sp. KR-1. Bioresour. Technol. 2015, 196, 22–27. [Google Scholar] [CrossRef]
- El-Dalatony, M.M.; Salama, E.S.; Kurade, M.B.; Kim, K.Y.; Govindwar, S.P.; Kim, J.R.; Kwon, E.E.; Min, B.; Jang, M.; Oh, S.E.; et al. Whole conversion of microalgal biomass into biofuels through successive high-throughput fermentation. Chem. Eng. J. 2019, 360, 797–805. [Google Scholar] [CrossRef]
- Chen, C.Y.; Liu, C.C. Optimization of lutein production with a two-stage mixotrophic cultivation system with Chlorella sorokiniana MB-1. Bioresour. Technol. 2018, 262, 74–79. [Google Scholar] [CrossRef]
- Xie, Y.; Zhao, X.; Chen, J.; Yang, X.; Ho, S.H.; Wang, B.; Chang, J.S.; Shen, Y. Enhancing cell growth and lutein productivity of Desmodesmus sp F51 by optimal utilization of inorganic carbon sources and ammonium salt. Bioresour. Technol. 2017, 244, 664–671. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. Closing the Loop—An EU Action Plan for the Circular Economy; European Commission: Brussels, Belgium, 2015. [Google Scholar]
- Bertolucci Paes, L.A.; Bezerra, B.S.; Deus, R.M.; Jugend, D.; Gomes Battistelle, R.A. Organic solid waste management in a circular economy perspective—A systematic review and SWOT analysis. J. Clean. Prod. 2019, 239, 118086. [Google Scholar] [CrossRef]
- Teigiserova, D.A.; Hamelin, L.; Thomsen, M. Review of high-value food waste and food residues biorefineries with focus on unavoidable wastes from processing. Resour. Conserv. Recycl. 2019, 149, 413–426. [Google Scholar] [CrossRef]
- Cao, Y.; Chen, S.S.; Zhang, S.C.; Ok, Y.S.; Matsagar, B.M.; Wu, K.C.W.; Tsang, D.C.W. Advances in lignin valorization towards bio-based chemicals and fuels: Lignin biorefinery. Bioresour. Technol. 2019, 291, 121878. [Google Scholar] [CrossRef] [PubMed]
- Mitra, M.; Mishra, S. Multiproduct biorefinery from Arthrospira spp. towards zero waste: Current status and future trends. Bioresour. Technol. 2019, 291, 121928. [Google Scholar] [CrossRef] [PubMed]
- Hemalatha, M.; Sarkar, O.; Mohan, S.V. Self-sustainable azolla-biorefinery platform for valorization of biobased products with circular-cascading design. Chem. Eng. J. 2019, 373, 1042–1053. [Google Scholar] [CrossRef]
- Milledge, J.J.; Nielsen, B.V.; Maneein, S.; Harvey, P.J. A brief review of anaerobic digestion of algae for bioenergy. Energies 2019, 12, 1166. [Google Scholar] [CrossRef] [Green Version]
- Abomohra, A.E.; Zheng, X.; Wang, Q.; Huang, J.; Ebaid, R. Enhancement of biodiesel yield and characteristics through in-situ solvo-thermal co-transesterification of wet microalgae with spent coffee grounds. Bioresour. Technol. 2021, 323, 124640. [Google Scholar] [CrossRef]
- Ma, X.; Zheng, H.; Addy, M.; Anderson, E.; Liu, Y.; Chen, P.; Ruan, R. Cultivation of Chlorella vulgaris in wastewater with waste glycerol: Strategies for improving nutrients removal and enhancing lipid production. Bioresour. Technol. 2016, 207, 252–261. [Google Scholar] [CrossRef]
- Abomohra, A.E.F.; Eladel, H.; El-Esawi, M.; Wang, S.; Wang, Q.; He, Z.X.; Feng, Y.Q.; Shang, H.; Hanelt, D. Effect of lipid-free microalgal biomass and waste glycerol on growth and lipid production of Scenedesmus obliquus: Innovative waste recycling for extraordinary lipid production. Bioresour. Technol. 2018, 249, 992–999. [Google Scholar] [CrossRef]
- Yarnold, J.; Karan, H.; Oey, M.; Hankamer, B. Microalgal Aquafeeds as Part of a Circular Bioeconomy. Trends Plant Sci. 2019, 24, 959–970. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chew, K.W.; Chia, S.R.; Yen, H.W.; Nomanbhay, S.; Ho, Y.C.; Show, P.L. Transformation of biomass waste into sustainable organic fertilizers. Sustainability 2019, 11, 2266. [Google Scholar] [CrossRef] [Green Version]
- Abad-Segura, E.; De La Fuente, A.B.; González-Zamar, M.; Belmonte-Ureña, L.J. Effects of circular economy policies on the environment and sustainable growth: Worldwide research. Sustainability 2020, 12, 5792. [Google Scholar] [CrossRef]

| Base of Catalyst | FE (%) in FA | CO2R Condition | Reference |
|---|---|---|---|
| Cu modified with polymeric | 38–45 | −0.7 VRHE in 0.05 M K2CO3 and 4 mM KCl with 5 sccm CO2 | [35] |
| boron-doped diamond | ~70 | −2.1 V (vs. Ag/AgCl) in KCl aqueous solution | [37] |
| polycrystalline Sn | ~70 | −1.0 VRHE in 0.1 M KHCO3with 20 sccm CO2 | [43] |
| sulfur-modified copper | ~80 | −0.8 VRHE in 0.1 M KHCO3with 20 sccm CO2 | [44] |
| Cu | ~85 | −1.6 V vs. ferrocenium voltage in CO2-saturated [EMIM](BF4)/H2O (92/8 v/v) ionic liquid solution | [45] |
| Cu (1.5 cm 3 cm) | ~20 | −0.8 VRHE in 0.1 M KHCO3 with 20 sccm CO2 | [47] |
| Cu | ~20 | −1.4 VRHE in 0.5 M KCl with 70 sccm CO2 | [48] |
| Cu2O@Cu | ~40 | −0.7 VRHE in 0.1 M KHCO3 with 5 sccm CO2 | [51] |
| Iron-graphite electrode pair | ~18 | −0.6 VAg/AgCl with CO2 saturated 0.5 MNaHCO3 in anaerobic sludge digestion process | [54] |
| Co incovalentorganic frameworks | in CO | MeCN with triethanolamineas sacrificial reducing agent and Ru(bpy)3Cl2·asphotosensitizerunder simulated sunlight | [55] |
| In0.6Bi0.2Sn0.2 alloy on a halide perovskite | ~95 | −1.3 VRHE in 0.5 M KHCO3with 20 sccmCO2under simulated sunlight | [59] |
| Cu0.5Zn0.5 | ~60 | −1.3 VRHE in CO2 saturated 0.1 M KHCO3 under simulated sunlight | [60] |
| SnOx/Sn | ~80 | −1.7 VAg/AgCl in 0.1 M CO2-saturated KHCO3 | [61] |
| Pd15Ni85 | ~50 | −0.5 VRHE in 1.0 M CO2-saturated KHCO3 | [62] |
| Pb modified with 4-aminomethylbenzene | ~80 | −1.3 VRHE in 1.0 M CO2-saturated KHCO3 | [28] |
| N,S-codoped carbon catalysts | ~90 in CO | −0.6 VRHE in 0.1 M KHCO3with 34 sccm CO2 | [65] |
| Pd85Cu15/C | ~86 in CO | −0.9 VRHE in 1.0 M CO2-saturated KHCO3 | [66] |
| defective β-Bi2O3 double-walled nanotubes | ~90 | −0.8 VRHE in 0.5M KHCO3with 20 sccm CO2 | [67] |
| 3.7 nm Pd nanoparticles | ~90 in CO | −0.9 VRHE in 1.0 M CO2-saturated KHCO3 | [68] |
| boron-doped Pd catalyst | ~70 | −0.5 VRHE in 1.0 M CO2-saturated KHCO3 | [69] |
| Catalyst | T (°C) | P (MPa) | H2/CO2/Inert | GHSV (mL g−1 h−1) | XCO2 (%) | SCO (%) | SCH4 (%) | Reference |
|---|---|---|---|---|---|---|---|---|
| Ru/MnOx | 300 | 0.1 | 22/5/73 | 150,000 | 25 | 10 | 90 | [78] |
| Ru/Al2O3 | 32 | 6 | 94 | |||||
| Ru/CeO2 | 83 | 1 | 99 | |||||
| Ru/ZnO | 1 | 94 | 6 | |||||
| PtCo/TiO2 | 300 | 0.1 | 67/33/0 | 36,000 | 8.2 | 99 | 1 | [82] |
| PtCo/CeO2 | 9.1 | 92 | 8 | |||||
| PtCo/ZrO2 | 7.8 | 90 | 11 | |||||
| Co/ZrO2 | 400 | 3.0 | 80/20/0 | 3600 | 92.5 | < 1 | > 99 | [81] |
| Co/SiO2 | 80.1 | 2 | 98 | |||||
| Co/Al2O3 | 77.8 | 3 | 97 | |||||
| Co/TiO2 | 30.9 | 96 | 4 | |||||
| Ni/ZIF-8 a | 420 | 0.1 | 80/20/0 | 15,000 | 43.8 | 97 | 3 | [81] |
| Fe/ZIF-8 a | 43.8 | 97 | 3 | |||||
| Ni/Fe/ZrO2 | 230 | 0.5 | 80/20/0 | 5000 | 82 | 14 | 86 | [83] |
| γ-Fe2O3 | 400 | 0.1 | 20/0.1/80 | 1,500,000 | 45 | 30 | 70 | [84] |
| Ni/MCM b | 400 | 0.1 | 80/20/0 | 90,000 | 73.2 | 8 | 92 | [85] |
| Catalyst | T (°C) | P (MPa) | CO2/H2/Inert | GHSV (mL g−1 h−1) | XCO2 (%) | SCH3OH (%) | Reference |
|---|---|---|---|---|---|---|---|
| Cu/SiO2 | 250 | 4.1 | 72/24/4 | 3600 | 2.8 | 15 | [100] |
| Cu/ZnO | 180 | 0.7 | 90/10/0 | 4000 | 0.9 | 94 | [97] |
| Cu/ZnO/ZrO2/SiO2 | 240 | 2.0 | 30/90/10 | 39,000 | 5.2 | 38 | [99] |
| Cu/ZnO/ZrO2/MgO/Al2O3 | 250 | 2.0 | 75/25/0 | 2000 | 12.1 | 36 | [93] |
| Pd/ZnO | 250 | 2.0 | 25/75/0 | 3600 | 10.7 | 60 | [101] |
| Pd/SiO2 | 250 | 4.1 | 72/24/4 | 3600 | 3.0 | 23 | [100] |
| Pd-Cu/SiO2 | 6.6 | 34 | |||||
| Pd-Cu/SiO2 | 250 | 5.0 | 75/25/0 | 30,000 | 1.6 | 27 | [102] |
| MnOx-Co3O4 | 250 | 1.0 | 60/20/20 | 120,000 | 45.1 | 22 | [103] |
| In2O3 | 270 | 4.0 | 60/20/20 | 15,000 | 1.1 | 55 | [76] |
| In2O3 | 330 | 4.0 | 60/20/20 | 15,000 | 7.1 | 40 | |
| In2O3/ZrO2 | 300 | 5.0 | 80/20/0 | 16,000 | 5.2 | >99 | [104] |
| Catalyst | T (°C) | P (MPa) | H2/CO2/Inert | GHSV (mL g−1 h−1) | XCO2 (%) | SCO (%) | Hydrocarbon Distribution (%) | Reference | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| CH4 | C2=-C4= | C2−-C4− | C5+ | ||||||||
| Fe3O4 | 320 | 3 | 72/24/4 | 2000 | 29 | 17 | 60 | <1 | 36 | 3 | [110] |
| Na-Fe3O4 | 320 | 3 | 72/24/4 | 2000 | 41 | 14 | 16 | 47 | 8 | 30 | |
| Fe2O3 | 350 | 1.5 | 70/23/7 | 1150 | 23 | 21 | 18 | 82 | [107] | ||
| K-Fe3O4/Fe2O3 | 270 | 5 | 73/25/2 | 2700 | 37 | 14 | 24 | 42 | 9 | 29 | [106] |
| CuFeO2 | 300 | 1 | 75/25/0 | 1800 | 18 | 32 | 4 | 31 | 5 | 60 | [109] |
| CuFe2O4 | 300 | 1 | 75/25/0 | 1800 | 16 | 28 | 38 | 1 | 49 | 11 | |
| K-Fe-Co/Al2O3 | 300 | 1.1 | 72/24/4 | 700 | 31 | 18 | 16 | 27 | 6 | 51 | [111] |
| Co/Fe oxide a | 270 | 0.9 | 72/28/0 | 2000 | 27 | 14 | 82 | 15 | <1 | [77] | |
| Na-Co/Fe oxide a | 270 | 0.9 | 72/28/0 | 2000 | 23 | 42 | 60 | 29 | 2 | ||
| Pyrolyzed Fe-MIL-88B b | 400 | 3 | 75/25/0 | 3600 | 46 | 18 | 32 | 23 | 18 | 27 | [108] |
| K-pyrolyzed Fe-MIL-88B b | 400 | 3 | 75/25/0 | 3600 | 43 | 26 | 32 | 33 | 6 | 19 | |
| In2O3/HZSM | 340 | 3 | 73/24/3 | 9000 | 13 | 45 | 1 | 20 | 79 | [112] | |
| Ga2O3/HZSM | 340 | 3 | 73/24/3 | 9000 | 9 | 86 | 5 | 35 | 61 | ||
| Fe2O3/HZSM | 340 | 3 | 73/24/3 | 9000 | 7 | 74 | 2 | 28 | 71 | ||
| Na-Fe3O4/HZSM | 350 | 3 | 72/24/4 | 4000 | 33 | 26 | 8 | 18 | 74 | [105] | |
| In2O3-ZrO2/SAPO | 400 | 3 | 73/24/3 | 9000 | 36 | 85 | 4 | 76 | 17 | 3 | [75] |
| Microalgae Species | CO2 Conc. (v/v%) | CO2 Fixation Rate (g-CO2 L−1 d−1) | Biomass Production Rate (g-DCW L−1 d−1) | Specific Growth Rate (d−1) | Cultivation System | CO2 Injection Rate (vvm), Mode | Light Intensity (umol m−2 s−1), Light/Dark Cycle | Reference |
|---|---|---|---|---|---|---|---|---|
| Acutodesmusobliquus | 14.1 | 0.22 b | 0.12 | 1.09 | Flask | 1.66, intermittent | 120, 24/0 | [124] |
| Aphanothecemicroscopica Nägeli | 15 a | 14.14 (from carbon balance) | 0.47 | 1.4 | Bubble column | 1, intermittent | 150, 24/0 | [140] |
| Asterarcysquadricellulare | 5 a | 0.39 b | 0.21 | 1.2 | Flask | 0.5, continuous | 250, 14/10 | [123] |
| Chlorella minutissima | 3.5 | 1.17 | 0.64 | 1.66 | Airlift | 0.43, continuous | 50~260 f, 24/0 | [139] |
| Chlorella sorokiniana | 5 a | 0.39 b | 0.22 | 2.42 | Flask | 0.5, continuous | 250, 14/10 | [123] |
| Chlorella sorokiniana | 15.6 | 0.31 | 0.17 | 0.46 | Airlift | 0.33, continuous | 120, 24/0 | [121] |
| Chlorella sp. | 1 | 1.00 | 0.53 | 0.615 | Flat-panel | 0.25, continuous | 130, 24/0 | [131] |
| Chlorella sp. | 2 | 0.77 b | 0.42 | 0.62 | Bubble column | 0.2, continuous | 400, 24/0 | [115] |
| Chlorella sp. | 3 | 0.46 | 0.31 | NA | Bubble column | -, continuous | 115, 24/0 | [119] |
| Chlorella sp. | 5 | 1.65 | 0.87 | 0.15 | Bubble column | 0.5, continuous | 56, 24/0 | [133] |
| Chlorella sp. | 5 | 0.10 | 0.06 | 0.25 | Flask | No aeration | 450, 24/0 | [141] |
| Chlorella sp. | 8 a | 2.33 | 0.84 | 1.11 | Bubble column | 0.2, continuous | 300, 24/0 | [136] |
| Chlorella sp. c | 10 | 1.33 | 0.73 | 0.55 | Bubble column | 0.2, intermittent | 300, 24/0 | [126] |
| Chlorella sp. | 12.5 a | 0.97 b | 0.53 | 0.827 | Bubble column | 0.2, continuous | 300, 24/0 | [142] |
| Chlorella sp. d | 30 | 0.61 b | 0.33 | NA | Bubble column | 0.67, continuous | 95, 24/0 | [128] |
| Chlorella vulgaris | 2.5 | 3.71 | 2.06 | NA | Bubble column | 0.36, continuous | 300, 18/6 | [138] |
| Chlorella vulgaris e | 3 | 0.31 | 0.17 | NA | Flask | No aeration | 40, 12/12 | [88] |
| Chlorella vulgaris | 5 | 1.32 b | 0.72 | NA | Bubble column | 0.1, continuous | 160, 24/0 | [143] |
| Chlorella vulgaris | 5 | 0.10 | 0.12 | 1.36 | Flask | 0.5, continuous | 160, 24/0 | [118] |
| Chlorella vulgaris | 10 | 0.43 | 0.26 | 0.46 | Tubular | 0.5, continuous | 100, 12/12 | [120] |
| Chlorella vulgaris | 10 | 0.31 b | 0.17 | 1.34 c | Flask | -, intermittent | 40, 14/10 | [134] |
| Desmodesmus sp. | 2.5 | 1.58 | 0.88 | NA | Tubular | 0.2, continuous | 300~1000 f, 24/0 | [117] |
| Microcystisaeruginosa g | 5 | 0.11 | 0.14 | 1.5 | Flask | 0.5, continuous | 160, 24/0 | [118] |
| Pseudokirchneriellasubcapitata | 5 | 0.09 | 0.10 | 0.89 | Flask | 0.5, continuous | 160, 24/0 | [118] |
| Scenedesmusdimorphus | 15 a | 0.89 | 0.49 | NA | Tubular | 0.25, intermittent | 100, 24/0 | [132] |
| Scenedesmus sp. | 15 | 0.17 b | 0.09 | NA | Flask | No aeration | -, 16/8 | [122] |
| Synechocystissalina h | 5 | 0.11 | 0.14 | 1.65 | Flask | 0.5, continuous | 160, 24/0 | [118] |
| Tetraselmissuecica, | 5 | 0.11 | 0.07 | 0.29 | Flask | No aeration | 450, 24/0 | [141] |
| Chlorella sp., Scenedesmus sp., Sphaerocystis sp., Spirulina sp. | 20 | 0.27 b | 0.15 | NA | Flask | No aeration | 80, 16/8 | [125] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, T.; Dong, C.-D.; Lin, J.-Y.; Chen, C.-W.; Chang, J.-S.; Kim, H.; Huang, C.-P.; Hung, C.-M. Recent Advances in Carbon Dioxide Conversion: A Circular Bioeconomy Perspective. Sustainability 2021, 13, 6962. https://doi.org/10.3390/su13126962
Wang T, Dong C-D, Lin J-Y, Chen C-W, Chang J-S, Kim H, Huang C-P, Hung C-M. Recent Advances in Carbon Dioxide Conversion: A Circular Bioeconomy Perspective. Sustainability. 2021; 13(12):6962. https://doi.org/10.3390/su13126962
Chicago/Turabian StyleWang, TsingHai, Cheng-Di Dong, Jui-Yen Lin, Chiu-Wen Chen, Jo-Shu Chang, Hyunook Kim, Chin-Pao Huang, and Chang-Mao Hung. 2021. "Recent Advances in Carbon Dioxide Conversion: A Circular Bioeconomy Perspective" Sustainability 13, no. 12: 6962. https://doi.org/10.3390/su13126962
APA StyleWang, T., Dong, C.-D., Lin, J.-Y., Chen, C.-W., Chang, J.-S., Kim, H., Huang, C.-P., & Hung, C.-M. (2021). Recent Advances in Carbon Dioxide Conversion: A Circular Bioeconomy Perspective. Sustainability, 13(12), 6962. https://doi.org/10.3390/su13126962









