Oil Pollution Affects the Central Metabolism of Keystone Vachellia (Acacia) Trees
Abstract
1. Introduction
2. Materials and Methods
2.1. Evrona Nature Reserve
2.2. Leaf Collection, Adult Trees
2.3. One-Year-Old Seedlings
2.4. Targeted Metabolic Analysis Using Gas Chromatography-Mass Spectrometry (GC-MS)
2.5. Statistical Analysis
3. Results
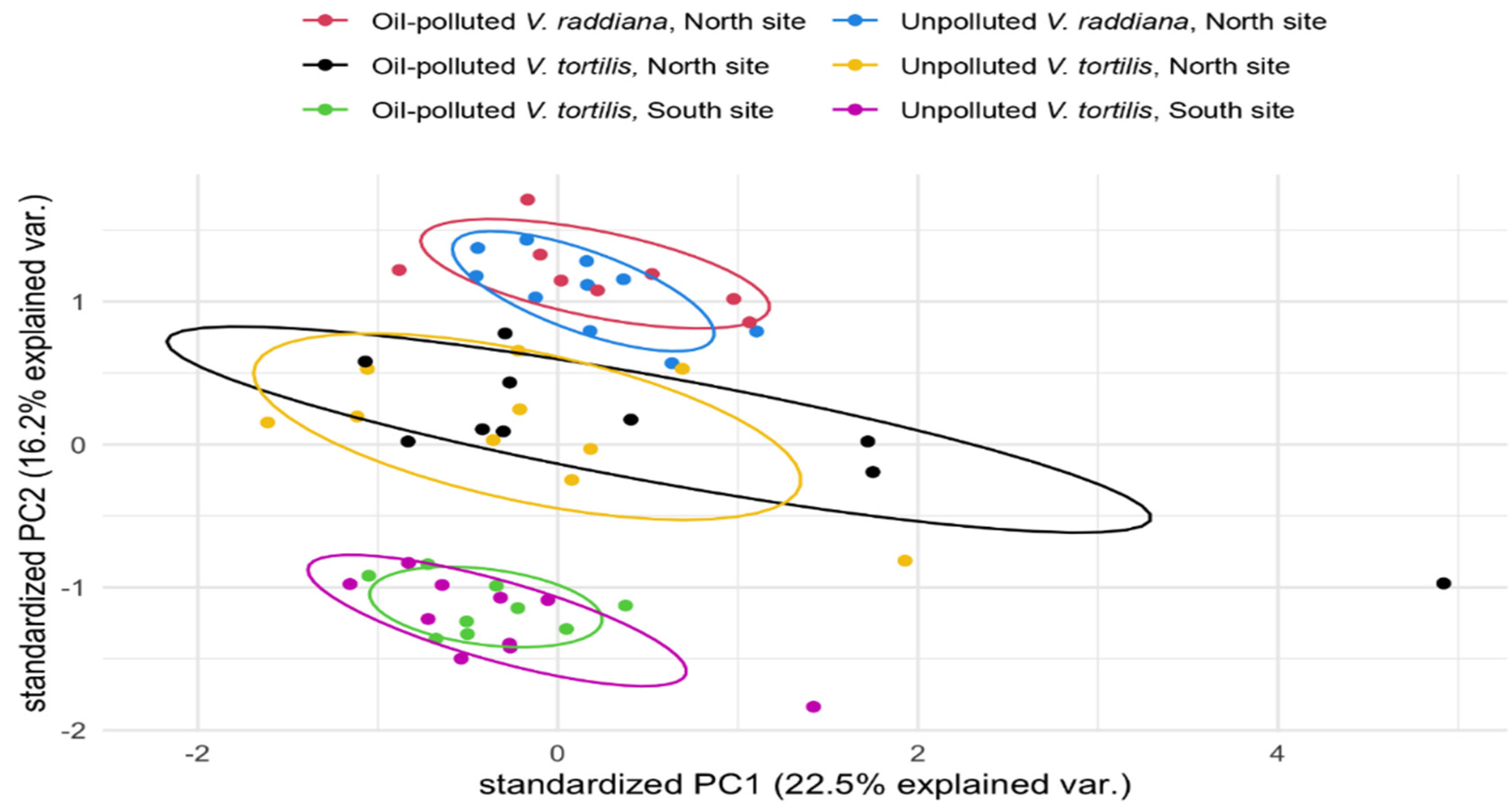
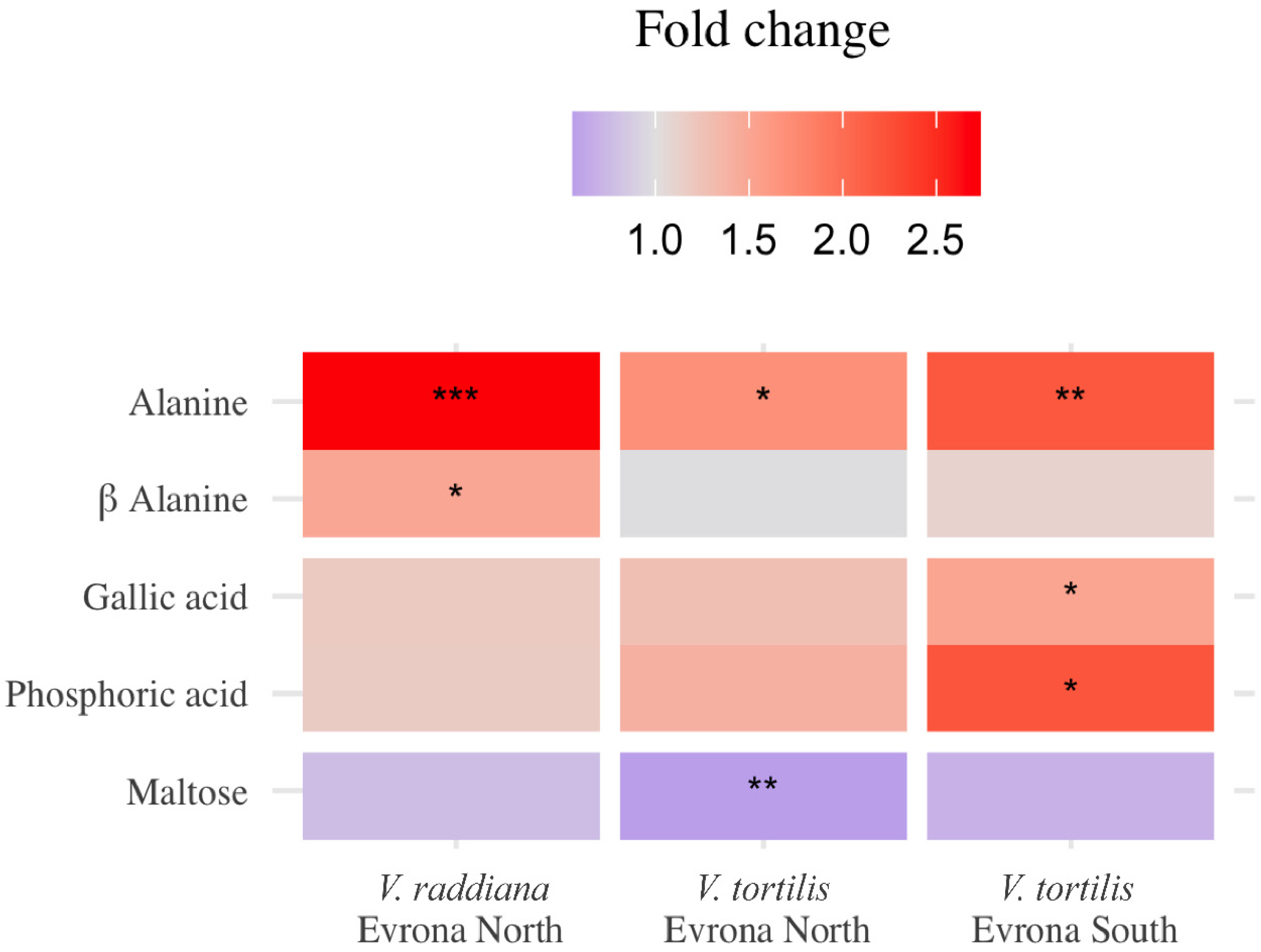
3.1. Metabolic Profile of the Adult Trees
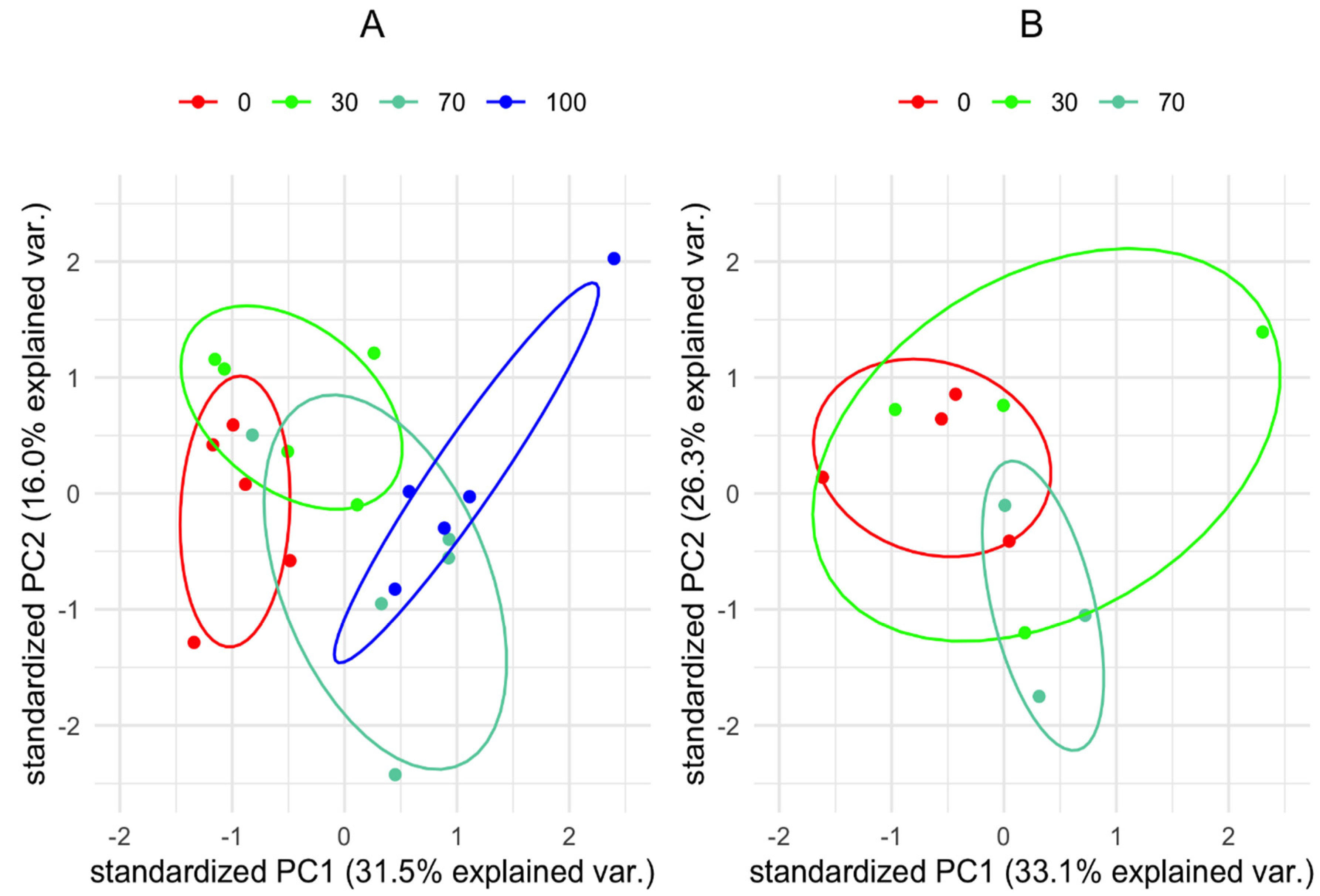
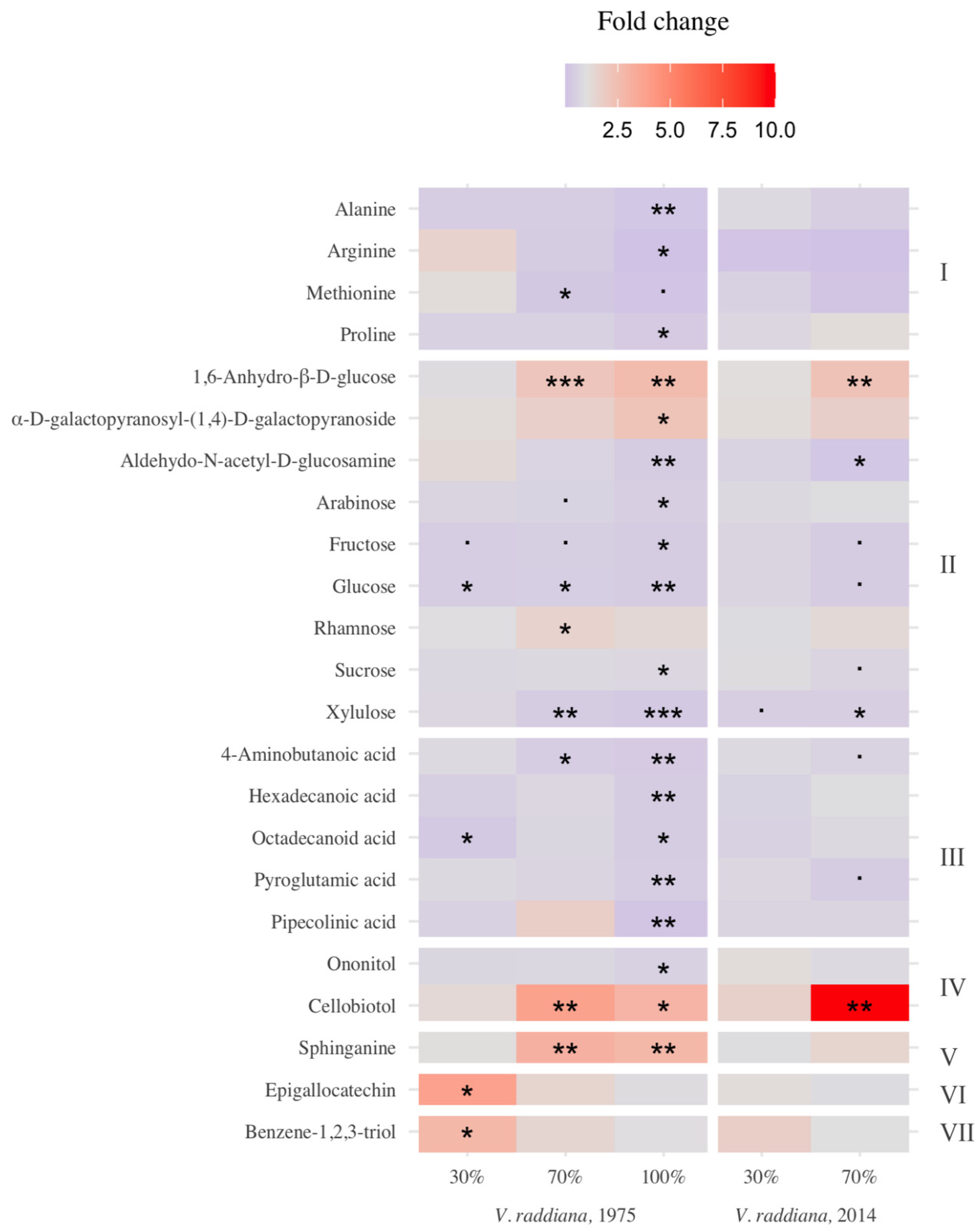
3.2. Metabolic Profile of Vachellia Raddiana Seedlings
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moriarty, P.; Honnery, D. Feasibility of a 100% Global Renewable Energy System. Energies 2020, 13, 5543. [Google Scholar] [CrossRef]
- Martins, F.; Felgueiras, C.; Smitkova, M.; Caetano, N. Analysis of Fossil Fuel Energy Consumption and Environmental Impacts in European Countries. Energies 2019, 12, 964. [Google Scholar] [CrossRef]
- Duke, N.C. Oil Spill Impacts on Mangroves: Recommendations for Operational Planning and Action Based on a Global Review. Mar. Pollut. Bull. 2016, 109, 700–715. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhang, W.; Wan, Z.; Li, S.; Huang, T.; Fei, Y. Oil Spills from Global Tankers: Status Review and Future Governance. J. Clean. Prod. 2019, 227, 20–32. [Google Scholar] [CrossRef]
- Thyng, K.M. Deepwater Horizon Oil Could Have Naturally Reached Texas Beaches. Mar. Pollut. Bull. 2019, 149, 110527. [Google Scholar] [CrossRef] [PubMed]
- Kingston, P.F. Long-Term Environmental Impact of Oil Spills. Spill Sci. Technol. B 2002, 7, 53–61. [Google Scholar] [CrossRef]
- Kinako, P.D.S. Short-Term Effects of Oil Pollution on Species Numbers and Productivity of a Simple Terrestrial Ecosystem. Environ. Pollut. Ser. A 1981, 26, 87–91. [Google Scholar] [CrossRef]
- Hegazy, A.K. Plant Succession and Its Optimization on Tar-Polluted Coasts in the Arabian Gulf Region. Environ. Conserv. 1997, 24, 149–158. [Google Scholar] [CrossRef]
- Nicolotti, G.; Egli, S. Soil Contamination by Crude Oil: Impact on the Mycorrhizosphere and on the Revegetation Potential of Forest Trees. Environ. Pollut. 1998, 99, 37–43. [Google Scholar] [CrossRef]
- Ward, D. The Biology of Deserts, 2nd ed.; Oxford University Press: Oxford, UK, 2016. [Google Scholar]
- Noy-Meir, I. Desert Ecosystems: Environment and Producers. Annu. Rev. Ecol. Evol. Syst. 1973, 4, 25–51. [Google Scholar] [CrossRef]
- Lovich, J.E.; Bainbridge, D. Anthropogenic Degradation of the Southern California Desert Ecosystem and Prospects for Natural Recovery and Restoration. Environ. Manag. 1999, 24, 309–326. [Google Scholar] [CrossRef]
- Hester, M.W.; Willis, J.M.; Rouhani, S.; Steinhoff, M.A.; Baker, M.C. Impacts of the Deepwater Horizon Oil Spill on the Salt Marsh Vegetation of Louisiana. Environ. Pollut. 2016, 216, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Arellano, P.; Tansey, K.; Balzter, H.; Tellkamp, M. Plant Family-Specific Impacts of Petroleum Pollution on Biodiversity and Leaf Chlorophyll Content in the Amazon Rainforest of Ecuador. PLoS ONE 2017, 12, e0169867. [Google Scholar] [CrossRef]
- Odukoya, J.; Lambert, R.; Sakrabani, R. Understanding the Impacts of Crude Oil and Its Induced Abiotic Stresses on Agrifood Production: A Review. Horticulturae 2019, 5, 47. [Google Scholar] [CrossRef]
- Baker, J.M. The Effects of Oils on Plants. Environ. Pollut. 1970, 1, 27–44. [Google Scholar] [CrossRef]
- Noori, A.S.; Maivan, H.Z.; Alaie, E. Changes in Total Phenol and Flavonoid Contents in Chrysanthemum leucanthemum under Crude Oil Contamination. Adv. Environ. Biol. 2012, 6, 3057–3065. [Google Scholar]
- Malallah, G.; Afzal, M.; Kurian, M.; Gulshan, S.; Dhami, M.S.I. Impact of Oil Pollution on Some Desert Plants. Environ. Int. 1998, 24, 919–924. [Google Scholar] [CrossRef]
- Johnson, C.M.; Hoskins, W.M. The Relation of Acids and Peroxides in Spray Oils to the Respiration of Sprayed Bean Leaves and the Development of Injury. Plant Physiol. 1952, 27, 507–525. [Google Scholar] [CrossRef] [PubMed]
- Knight, H.; Chamberlin, J.C.; Samuels, C.D. On Some Limiting Factors in the Use of Saturated Petroleum Oils as Insecticides. Plant Physiol. 1929, 4, 299–321. [Google Scholar] [CrossRef]
- Achuba, F.I. The Effect of Sublethal Concentrations of Crude Oil on the Growth and Metabolism of Cowpea (Vigna unguiculata) Seedlings. Environmentalist 2006, 26, 17–20. [Google Scholar] [CrossRef]
- Tran, T.H.; Mayzlish Gati, E.; Eshel, A.; Winters, G. Germination, Physiological and Biochemical Responses of Acacia Seedlings (Acacia raddiana and Acacia tortilis) to Petroleum Contaminated Soils. Environ. Pollut. 2018, 234, 642–655. [Google Scholar] [CrossRef]
- Walters, M.B.; Reich, P.B. Are Shade Tolerance, Survival, and Growth Linked? Low Light and Nitrogen Effects on Hardwood Seedlings. Ecology 1996, 77, 841–853. [Google Scholar] [CrossRef]
- Simberloff, D. Flagships, Umbrellas, and Keystones: Is Single-Species Management Passé in the Landscape Era? Biol. Conserv. 1998, 83, 247–257. [Google Scholar] [CrossRef]
- Paine, R.T. A Note on Trophic Complexity and Community Stability. Am. Nat. 1969, 103, 91–93. [Google Scholar] [CrossRef]
- Smith, D.W.; Peterson, R.O.; Houston, D.B. Yellowstone after Wolves. BioScience 2003, 53, 330–340. [Google Scholar] [CrossRef]
- Tanner, J.E.; Hughes, T.P.; Connell, J.H. Species Coexistence, Keystone Species, and Succession: A Sensitivity Analysis. Ecology 1994, 75, 2204–2219. [Google Scholar] [CrossRef]
- Power, M.E.; Tilman, D.; Estes, J.A.; Menge, B.A.; Bond, W.J.; Mills, L.S.; Daily, G.; Castilla, J.C.; Lubchenco, J.; Paine, R.T. Challenges in the Quest for Keystones: Identifying Keystone Species Is Difficult—but Essential to Understanding How Loss of Species Will Affect Ecosystems. BioScience 1996, 46, 609–620. [Google Scholar] [CrossRef]
- Gordon, G.; Stavi, I.; Shavit, U.; Rosenzweig, R. Oil Spill Effects on Soil Hydrophobicity and Related Properties in a Hyper-Arid Region. Geoderma 2018, 312, 114–120. [Google Scholar] [CrossRef]
- Munzbergova, Z.; Ward, D. Acacia Trees as Keystone Species in Negev Desert Ecosystems. J. Veg. Sci. 2002, 13, 227–236. [Google Scholar] [CrossRef]
- Hackett, T.D.; Korine, C.; Holderied, M.W. The Importance of Acacia Trees for Insectivorous Bats and Arthropods in the Arava Desert. PLoS ONE 2013, 8, e52999. [Google Scholar] [CrossRef] [PubMed]
- Rodger, Y.S.; Greenbaum, G.; Silver, M.; Bar-David, S.; Winters, G. Detecting Hierarchical Levels of Connectivity in a Population of Acacia tortilis at the Northern Edge of the Species’ Global Distribution: Combining Classical Population Genetics and Network Analyses. PLoS ONE 2018, 13, e0194901. [Google Scholar] [CrossRef]
- Zohary, M. Flora Palaestina; Israel Academy of Sciences and Humanities: Jerusalem, Israel, 1972; ISBN 978-965-208-000-4. [Google Scholar]
- Ferrante, M.; Möller, D.M.; Möller, G.M.; Lubin, Y.; Segoli, M. Seed Predation on Oil-Polluted and Unpolluted Vachellia (Acacia) Trees in a Hyper-Arid Desert Ecosystem. Insects 2020, 11, 665. [Google Scholar] [CrossRef]
- Stavi, I.; Silver, M.; Avni, Y. Latitude, Basin Size, and Microhabitat Effects on the Viability of Acacia Trees in the Negev and Arava, Israel. Catena 2014, 114, 149–156. [Google Scholar] [CrossRef]
- Ward, D.; Rohner, C. Anthropogenic Causes of High Mortality and Low Recruitment in Three Acacia Tree Taxa in the Negev Desert, Israel. Biodivers. Conserv. 1997, 6, 877–893. [Google Scholar] [CrossRef]
- Sher, A.A.; Wiegand, K.; Ward, D. Do Acacia and Tamarix Trees Compete for Water in the Negev Desert? J. Arid Environ. 2010, 74, 338–343. [Google Scholar] [CrossRef]
- Bruins, H.J.; Sherzer, Z.; Ginat, H.; Batarseh, S. Degradation of Springs in the Arava Valley: Anthropogenic and Climatic Factors. Land Degrad. Dev. 2012, 23, 365–383. [Google Scholar] [CrossRef]
- Stavi, I.; Rosenzweig, R. Tillage Effect on Hydrophobicity and Hydrological Properties of Oil-Contaminated Sediments in a Hyper-Arid Region. Arid Land Res. Manag. 2020, 34, 26–35. [Google Scholar] [CrossRef]
- Nothers, M.; Segev, N.; Kreyling, J.; Hjazin, A.; Groner, E. Desert Vegetation Forty Years after an Oil Spill. J. Environ. Qual. 2017, 46, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Möller, D.M.; Ferrante, M.; Möller, G.M.; Rozenberg, T.; Segoli, M. The Impact of Terrestrial Oil Pollution on Parasitoid Wasps Associated with Vachellia (Fabales: Fabaceae) Trees in a Desert Ecosystem, Israel. Environ. Entomol. 2020. [Google Scholar] [CrossRef]
- Batyrshina, Z.S.; Yaakov, B.; Shavit, R.; Singh, A.; Tzin, V. Comparative Transcriptomic and Metabolic Analysis of Wild and Domesticated Wheat Genotypes Reveals Differences in Chemical and Physical Defense Responses against Aphids. BMC Plant Biol. 2020, 20, 1–20. [Google Scholar] [CrossRef]
- Hochberg, U.; Degu, A.; Toubiana, D.; Gendler, T.; Nikoloski, Z.; Rachmilevitch, S.; Fait, A. Metabolite Profiling and Network Analysis Reveal Coordinated Changes in Grapevine Water Stress Response. BMC Plant Biol. 2013, 13, 184. [Google Scholar] [CrossRef]
- Lisec, J.; Schauer, N.; Kopka, J.; Willmitzer, L.; Fernie, A.R. Gas Chromatography Mass Spectrometry–Based Metabolite Profiling in Plants. Nat. Protoc. 2006, 1, 387–396. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; Version 4.0.2; Vienna, Austria, 2020; Available online: https://www.r-project.org/ (accessed on 11 June 2021).
- R Studio Team. RStudio: Integrated Development Environment for R; Boston, MA, USA, 2018; Available online: https://www.rstudio.com/ (accessed on 11 June 2021).
- Vu, V.Q. Ggbiplot: A Ggplot2 Based Biplot. R Package; Version 0.55; 2011. [Google Scholar]
- Lenth, R.V. Least-Squares Means: The R Package Lsmeans. J. Stat. Softw. 2016, 69, 1–33. [Google Scholar] [CrossRef]
- Wang, L.; Meeus, I.; Rombouts, C.; Van Meulebroek, L.; Vanhaecke, L.; Smagghe, G. Metabolomics-Based Biomarker Discovery for Bee Health Monitoring: A Proof of Concept Study Concerning Nutritional Stress in Bombus terrestris. Sci. Rep. 2019, 9, 11423. [Google Scholar] [CrossRef]
- Miller, I.J.; Peters, S.R.; Overmyer, K.A.; Paulson, B.R.; Westphall, M.S.; Coon, J.J. Real-Time Health Monitoring through Urine Metabolomics. NPJ Digit. Med. 2019, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Castro-Moretti, F.R.; Gentzel, I.N.; Mackey, D.; Alonso, A.P. Metabolomics as an Emerging Tool for the Study of Plant–Pathogen Interactions. Metabolites 2020, 10, 52. [Google Scholar] [CrossRef]
- Nassan, F.L.; Kelly, R.S.; Kosheleva, A.; Koutrakis, P.; Vokonas, P.S.; Lasky-Su, J.A.; Schwartz, J.D. Metabolomic Signatures of the Long-Term Exposure to Air Pollution and Temperature. Environ. Health 2021, 20, 3. [Google Scholar] [CrossRef]
- Gago-Tinoco, A.; González-Domínguez, R.; García-Barrera, T.; Blasco-Moreno, J.; Bebianno, M.J.; Gómez-Ariza, J.-L. Metabolic Signatures Associated with Environmental Pollution by Metals in Doñana National Park Using P. clarkii as Bioindicator. Environ. Sci. Pollut. Res. 2014, 21, 13315–13323. [Google Scholar] [CrossRef] [PubMed]
- Stien, D.; Suzuki, M.; Rodrigues, A.M.S.; Yvin, M.; Clergeaud, F.; Thorel, E.; Lebaron, P. A Unique Approach to Monitor Stress in Coral Exposed to Emerging Pollutants. Sci. Rep. 2020, 10, 9601. [Google Scholar] [CrossRef] [PubMed]
- Richter, C.M.; Kranig, S.; Wild, A. Contents of Free Amino Acids in Needles of Norway Spruce Trees in Relation to Novel Forest Decline. Studies on Trees from a Site in the Northern Black Forest. Environ. Pollut. 1995, 87, 303–312. [Google Scholar] [CrossRef]
- Honour, S.L.; Bell, J.N.B.; Ashenden, T.W.; Cape, J.N.; Power, S.A. Responses of Herbaceous Plants to Urban Air Pollution: Effects on Growth, Phenology and Leaf Surface Characteristics. Environ. Pollut. 2009, 157, 1279–1286. [Google Scholar] [CrossRef]
- Skrypnik, L.; Maslennikov, P.; Novikova, A.; Kozhikin, M. Effect of Crude Oil on Growth, Oxidative Stress and Response of Antioxidative System of Two Rye (Secale cereale L.) Varieties. Plants 2021, 10, 157. [Google Scholar] [CrossRef]
- Al-Hawas, G.H.S.; Shukry, W.M.; Azzoz, M.M.; Al-Moaik, R.M.S. The Effect of Sublethal Concentrations of Crude Oil on the Metabolism of Jojoba (Simmodsia chinensis) Seedlings. Int. Res. J. Plant Sci. 2012, 3, 54–62. [Google Scholar]
- López-Bucio, J.; Nieto-Jacobo, M.F.; Ramírez-Rodríguez, V.; Herrera-Estrella, L. Organic Acid Metabolism in Plants: From Adaptive Physiology to Transgenic Varieties for Cultivation in Extreme Soils. Plant Sci. 2000, 160, 1–13. [Google Scholar] [CrossRef]
- Timpa, J.D.; Burke, J.J.; Quisenberry, J.E.; Wendt, C.W. Effects of Water Stress on the Organic Acid and Carbohydrate Compositions of Cotton Plants. Plant Physiol. 1986, 82, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shi, D.-C. Photosynthesis, Chlorophyll Fluorescence, Inorganic Ion and Organic Acid Accumulations of Sunflower in Responses to Salt and Salt-Alkaline Mixed Stress. Photosynthetica 2010, 48, 127–134. [Google Scholar] [CrossRef]
- Lu, Y.; Sharkey, T.D. The Importance of Maltose in Transitory Starch Breakdown. Plant Cell Environ. 2006, 29, 353–366. [Google Scholar] [CrossRef]
- Kaplan, F.; Guy, C.L. RNA Interference of Arabidopsis Beta-Amylase8 Prevents Maltose Accumulation upon Cold Shock and Increases Sensitivity of PSII Photochemical Efficiency to Freezing Stress. Plant J. 2005, 44, 730–743. [Google Scholar] [CrossRef]
- Parthasarathy, A.; Savka, M.A.; Hudson, A.O. The Synthesis and Role of β-Alanine in Plants. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Vandermeulen, J.H.; Harper, J.; Humphrey, B. Environmental Factors Influencing Oil Penetration and Persistence in Fine Sediment Tidal Flats. Oil Chem. Pollut. 1988, 4, 155–177. [Google Scholar] [CrossRef]
- de Jong, E. The Effect of a Crude Oil Spill on Cereals. Environ. Pollut. Ser. A Ecol. Biol. 1980, 22, 187–196. [Google Scholar] [CrossRef]
- Aharoni, A.; Galili, G. Metabolic Engineering of the Plant Primary–Secondary Metabolism Interface. Curr. Opin. Biotechnol. 2011, 22, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Wöll, S.; Kim, S.H.; Greten, H.J.; Efferth, T. Animal Plant Warfare and Secondary Metabolite Evolution. Nat. Prod. Bioprospect. 2013, 3, 1–7. [Google Scholar] [CrossRef]
- Or, K.; Ward, D. The Effects of Seed Quality and Pipecolic and Djenkolic Acids on Bruchid Beetle Infestation in Water Deficit-Stressed Acacia Trees. J. Chem. Ecol. 2004, 30, 2297–2307. [Google Scholar] [CrossRef] [PubMed]
- Meiri, R.; Winters, G. The Evrona 5-Year Monitoring Program: Monitoring Acacia Trees in the Evrona Nature Reserve Following the December 2014 Oil Spill; Dead Sea Arava Science Center: Hatzeva, Israel, 2019; p. 46. [Google Scholar]
- Winters, G.; Otieno, D.; Cohen, S.; Bogner, C.; Ragowloski, G.; Paudel, I.; Klein, T. Tree Growth and Water-Use in Hyper-Arid Acacia Occurs during the Hottest and Driest Season. Oecologia 2018, 188, 695–705. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrante, M.; Dangol, A.; Didi-Cohen, S.; Winters, G.; Tzin, V.; Segoli, M. Oil Pollution Affects the Central Metabolism of Keystone Vachellia (Acacia) Trees. Sustainability 2021, 13, 6660. https://doi.org/10.3390/su13126660
Ferrante M, Dangol A, Didi-Cohen S, Winters G, Tzin V, Segoli M. Oil Pollution Affects the Central Metabolism of Keystone Vachellia (Acacia) Trees. Sustainability. 2021; 13(12):6660. https://doi.org/10.3390/su13126660
Chicago/Turabian StyleFerrante, Marco, Anuma Dangol, Shoshana Didi-Cohen, Gidon Winters, Vered Tzin, and Michal Segoli. 2021. "Oil Pollution Affects the Central Metabolism of Keystone Vachellia (Acacia) Trees" Sustainability 13, no. 12: 6660. https://doi.org/10.3390/su13126660
APA StyleFerrante, M., Dangol, A., Didi-Cohen, S., Winters, G., Tzin, V., & Segoli, M. (2021). Oil Pollution Affects the Central Metabolism of Keystone Vachellia (Acacia) Trees. Sustainability, 13(12), 6660. https://doi.org/10.3390/su13126660










