Niche Analysis and Conservation of Bird Species Using Urban Core Areas
Abstract
1. Introduction
2. Materials and Methods
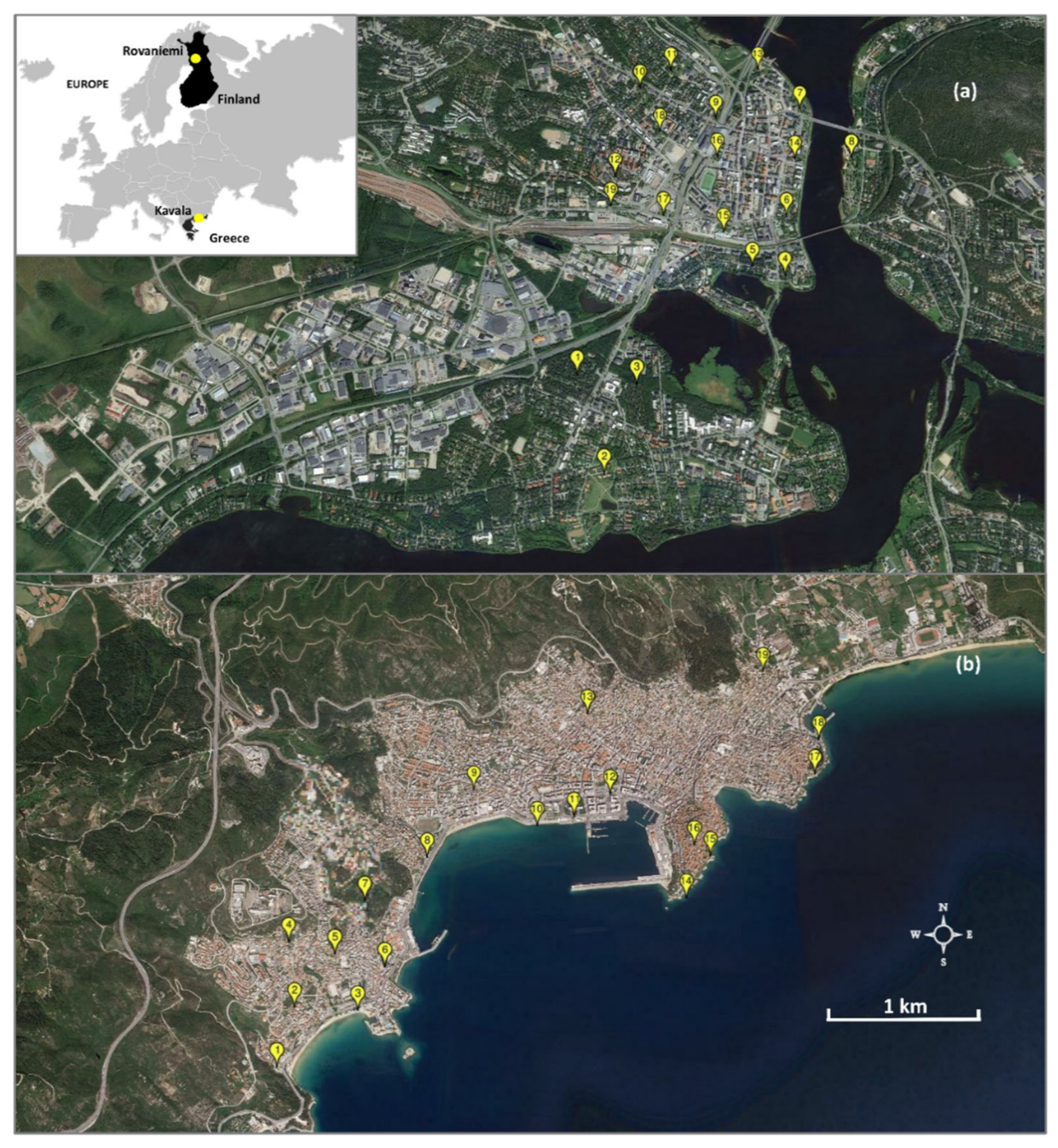
2.1. Study Area
2.2. Bird Surveys
2.3. Environmental Variables
2.4. Data Analysis
3. Results
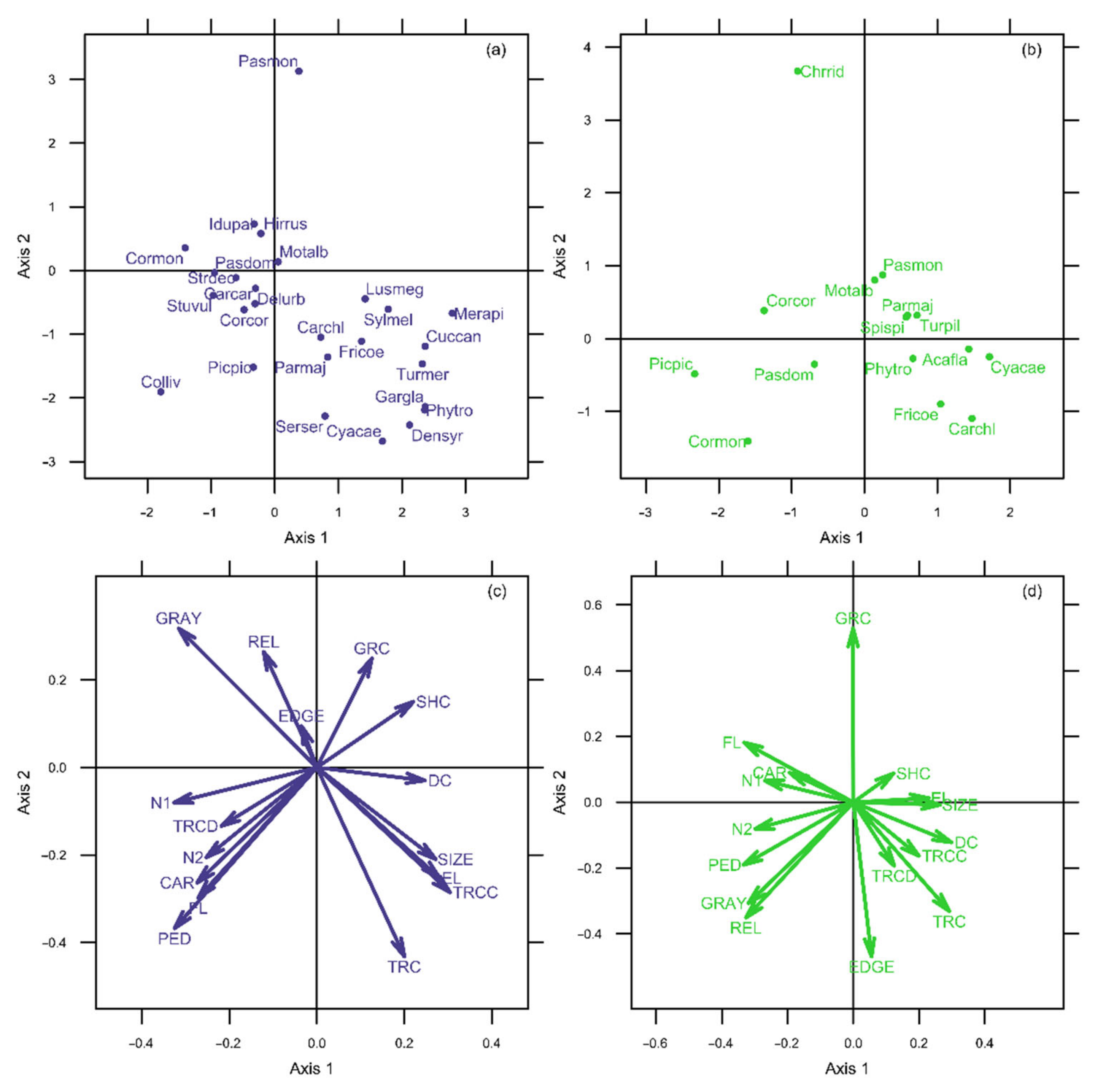
3.1. Niche Analysis for the Breeding Bird Community of Kavala
3.2. Niche Analysis for the Breeding Bird Community of Rovaniemi
3.3. Conservation Status and Priority
4. Discussion
4.1. Niche Specialization and Habitat Conditions
4.2. Conservation Management Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hutchinson, G.E. Concluding remarks. Cold Spring Harb. Symp. Quant. Biol. 1957, 22, 145–159. [Google Scholar] [CrossRef]
- Whittaker, R.H.; Levin, S.A.; Root, R.B. Niche, habitat and ecotope. Am. Nat. 1973, 107, 321–338. [Google Scholar] [CrossRef]
- Lehmann, S. Growing Biodiverse Urban Futures: Renaturalization and Rewilding as Strategies to Strengthen Urban Resilience. Sustainability 2021, 13, 2932. [Google Scholar] [CrossRef]
- Lepczyk, C.A.; Warren, P.S. Urban Bird Ecology and Conservation; University of California Press: Berkeley, CA, USA, 2012. [Google Scholar]
- Xu, X.; Xie, Y.; Qi, K.; Luo, Z.; Wang, X. Detecting the response of bird communities and biodiversity to habitat loss and fragmentation due to urbanization. Sci. Total Environ. 2018, 624, 1561–1576. [Google Scholar] [CrossRef]
- Chace, J.F.; Walsh, J.J. Urban effects on native avifauna: A review. Landsc. Urban Plan. 2006, 7, 46–69. [Google Scholar] [CrossRef]
- Pellissier, V.; Cohen, M.; Boulay, A.; Clergeau, P. Birds are also sensitive to landscape composition and configuration within the city centre. Landsc. Urban Plan. 2012, 104, 181–188. [Google Scholar] [CrossRef]
- Jones, G.A.; Sieving, K.E. Sieving Intercropping sunflower in organic vegetables to augment bird predators of arthropods. Agric. Ecosyst. Environ. 2006, 117, 171–177. [Google Scholar] [CrossRef]
- Klein, A.M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B Biol. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef]
- Whelan, C.J.; Sekercioglu, C.H.; Wenny, D.G. Why birds matter: From economic ornithology to ecosystem services. J. Ornithol. 2015, 156, 227–238. [Google Scholar] [CrossRef]
- Fuller, R.A.; Irvine, K.N.; Devine-Wright, P.; Warren, P.H.; Gaston, K.J. Psychological benefits of greenspace increase with biodiversity. Biol. Lett. 2007, 3, 390–394. [Google Scholar] [CrossRef]
- Cameron, R.W.F.; Brindley, P.; Mears, M.; McEwan, K.; Ferguson, F.; Sheffield, D.; Jorgensen, A.; Riley, J.; Goodrick, J.; Ballard, L.; et al. Where the wild things are! Do urban green spaces with greater avian biodiversity promote more positive emotions in humans? Urban Ecosyst. 2020, 23, 301–317. [Google Scholar] [CrossRef]
- Randler, C.; Tryjanowski, P.; Jokimäki, J.; Kaisanlahti-Jokimäki, M.-L.; Staller, N. SARS-CoV2 (COVID-19) Pandemic lockdown influences nature-based recreational activity: The case of birders. Int. J. Environ. Res. Public Health 2020, 17, 7310. [Google Scholar] [CrossRef]
- Grimm, N.B.; Faeth, S.H.; Golubiewski, N.E.; Redman, C.L.; Wu, J.G.; Bai, X.M.; Briggs, J.M. Global change and the ecology of cities. Science 2008, 319, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Jokimäki, J.; Suhonen, J.; Benedetti, Y.; Diaz, M.; Kaisanlahti-Jokimäki, M.-L.; Morelli, F.; Pérez-Contreras, T.; Rubio, E.; Sprau, P.; Tryjanowski, P.; et al. Land-sharing vs. land-sparing urban development modulate predator-prey interactions in Europe. Ecol. Appl. 2020, 30, 02049. [Google Scholar] [CrossRef] [PubMed]
- Loss, S.R.; Will, T.; Marra, P.P. The impact of domestic cats on wildlife of the United States. Nat. Commun. 2013, 4, 1396. [Google Scholar] [CrossRef] [PubMed]
- Pollack, L.; Ondrasek, N.R.; Calisi, R. Urban health and ecology: The promise of an avian biomonitoring tool. Curr. Zool. 2017, 63, 205–212. [Google Scholar] [CrossRef]
- Jiguet, F.; Sunnen, L.; Prévot, A.-C.; Princé, K. Urban pigeons losing toes due to human activities. Biol. Conserv. 2019, 240, 108241. [Google Scholar] [CrossRef]
- MacGregor-Fors, I.; Ortega-Álvarez, R. Fading from the forest: Bird community shifts related to urban park site-specific and landscape traits. Urban For. Urban Green. 2011, 10, 239–246. [Google Scholar] [CrossRef]
- Schneider, S.C.; Fischer, J.D.; Miller, J.R. Two-sided edge responses of avian communities in an urban landscape. Urban Ecosyst. 2015, 18, 539–551. [Google Scholar] [CrossRef]
- Leveau, L.M.; Leveau, C.M.; Villegas, M.; Cursach, J.A.; Suazo, C.G. Bird communities along urbanization gradients: A comparative analysis among three neo-tropical cities. Ornitol. Neotrop. 2017, 28, 77–87. [Google Scholar]
- Tzortzakaki, O.; Kati, V.; Kassara, C.; Tietze, D.T.; Giokas, S. Seasonal patterns of urban bird diversity in a Mediterranean coastal city: The positive role of open green spaces. Urban Ecosyst. 2018, 21, 27–39. [Google Scholar] [CrossRef]
- Kontsiotis, V.J.; Valsamidis, E.; Liordos, V. Organization and differentiation of breeding bird communities across a forested to urban landscape. Urban For. Urban Green. 2019, 38, 242–250. [Google Scholar] [CrossRef]
- Blair, R.B. Land use and avian species diversity along an urban gradient. Ecol. Appl. 1996, 6, 506–519. [Google Scholar] [CrossRef]
- McKinney, M.L. Urbanization as a major cause of biotic homogenization. Biol. Conserv. 2006, 127, 247–260. [Google Scholar] [CrossRef]
- Fernández-Juricic, E.; Jokimäki, J. A habitat island approach to conserving birds in urban landscapes-case studies from southern and northern Europe. Biodivers. Conserv. 2001, 10, 2023–2043. [Google Scholar] [CrossRef]
- Jokimäki, J.; Suhonen, J. Effects of urbanization on the breeding bird species richness in Finland: A biogeographical comparison. Ornis Fenn. 1993, 70, 71–77. [Google Scholar]
- Ives, C.D.; Lentini, P.E.; Threlfall, C.G.; Ikin, K.; Shanahan, D.F.; Garrard, G.E.; Bekessy, S.A.; Fuller, R.A.; Mumaw, L.; Rayner, L.; et al. The importance of cities for threatened species. Glob. Ecol. Biogeogr. 2016, 25, 117–126. [Google Scholar] [CrossRef]
- Jokimäki, J.; Suhonen, J.; Kaisanlahti-Jokimäki, M.-L. Urban core areas are important for species conservation: A European-level analysis of breeding bird species. Landsc. Urban Plan. 2018, 178, 73–81. [Google Scholar] [CrossRef]
- Miller, J.R.; Hobbs, R.J. Conservation where people live and work. Conserv. Biol. 2002, 16, 330–337. [Google Scholar] [CrossRef]
- Gilbert, O.L. The Ecology of Urban Habitats; Chapman and Hall: London, UK, 1989. [Google Scholar]
- Jokimäki, J. Occurrence of breeding bird species in urban parks: Effects of park structure and broad-scale variables. Urban Ecosyst. 1999, 3, 21–34. [Google Scholar] [CrossRef]
- Carbó-Ramírez, P.; Zuria, I. The value of small urban green spaces for birds in a Mexican city. Landsc. Urban Plan. 2011, 100, 213–222. [Google Scholar] [CrossRef]
- Strohbach, M.W.; Lerman, S.B.; Warren, P.S. Are small greening areas enhancing bird diversity? Insights from community-driven greening projects in Boston. Landsc. Urban Plan. 2013, 114, 69–79. [Google Scholar] [CrossRef]
- Peris, S.; Montelongo, T. Birds and small urban parks: A study in a high plateau city. Turk. J. Zool. 2014, 38, 316–325. [Google Scholar] [CrossRef]
- Jasmani, Z.; Ravn, H.P.; van den Bosch, C.C.K. The influence of small urban parks characteristics on bird diversity: A case study of Petaling Jaya, Malaysia. Urban Ecosyst. 2017, 20, 227–243. [Google Scholar] [CrossRef]
- Amaya-Espinel, J.D.; Hostetler, M.; Henríquez, C.; Bonacic, C. The influence of building density on Neotropical bird communities found in small urban parks. Landsc. Urban Plan. 2019, 190, 103578. [Google Scholar] [CrossRef]
- Clergeau, P.; Jokimäki, J.; Savard, J.P.L. Are urban bird communities influenced by the bird diversity of adjacent landscapes? J. Appl. Ecol. 2001, 38, 1122–1134. [Google Scholar] [CrossRef]
- Jokimäki, J.; Kaisanlahti-Jokimäki, M.-L. Spatial similarity of urban bird communities: A multiscale approach. J. Biogeogr. 2003, 8, 1183–1193. [Google Scholar] [CrossRef]
- Shwartz, A.; Muratet, A.; Simon, L.; Julliard, R. Local and management variables outweigh landscape effects in enhancing the diversity of different taxa in a big metropolis. Biol. Conserv. 2013, 157, 285–292. [Google Scholar] [CrossRef]
- Callaghan, C.T.; Major, R.E.; Lyons, M.B.; Martin, J.M.; Kingsford, R.T. The effects of local and landscape habitat attributes on bird diversity in urban greenspaces. Ecosphere 2018, 9, e02347. [Google Scholar] [CrossRef]
- Muñoz-Pedreros, A.; González-Urrutia, M.; Encina-Montoya, F.; Norambuena, H.V. Effects of vegetation strata and human disturbance on bird diversity in green areas in a city in southern Chile. Avian Res. 2018, 9, 38. [Google Scholar] [CrossRef]
- Titoko, R.; Lowry, J.H.; Osborne, T.; Naikatini, A.; Comely, J.; Riley, R. Relationship of bird richness, abundance and assemblage to the built environment in a small island tropical urban setting: A Suva, Fiji case study. Urban Ecosyst. 2019, 22, 709–719. [Google Scholar] [CrossRef]
- Yang, X.; Tan, X.; Chen, C.; Wang, Y. The influence of urban park characteristics on bird diversity in Nanjing, China. Avian Res. 2020, 11, 45. [Google Scholar] [CrossRef]
- Da Silva, B.F.; Pena, J.C.; Viana-Junior, A.B.; Vergne, M.; Pizo, M.A. Noise and tree species richness modulate the bird community inhabiting small public urban green spaces of a Neotropical city. Urban Ecosyst. 2021, 24, 71–81. [Google Scholar] [CrossRef]
- MacGregor-Fors, I.; Schondube, J.E. Gray vs. green urbanization: Relative importance of urban features for urban bird communities. Basic Appl. Ecol. 2011, 12, 372–381. [Google Scholar] [CrossRef]
- Ortega-Álvarez, R.; MacGregor-Fors, I. Living in the big city: Effects of urban land-use on bird community structure, diversity, and composition. Landsc. Urban Plan. 2009, 90, 189–195. [Google Scholar] [CrossRef]
- Ferenc, M.; Sedláček, O.; Fuchs, R.; Dinetti, M.; Fraissinet, M.; Storch, D. Are cities different? Patterns of species richness and beta diversity of urban bird communities and regional species assemblages in Europe. Glob. Ecol. Biogeogr. 2014, 23, 479–489. [Google Scholar] [CrossRef]
- Jokimäki, J.; Suhonen, J.; Kaisanlahti-Jokimäki, M.-L. Urbanization and species occupancy frequency distribution patterns in core zone areas of European towns. Eur. J. Ecol. 2016, 2, 23–43. [Google Scholar] [CrossRef]
- Ibáñez-Álamo, J.D.; Morelli, F.; Benedetti, Y.; Rubio, E.; Jokimäki, J.; Pérez-Contreras, T.; Sprau, P.; Suhonen, J.; Tryjanowski, P.; Kaisanlahti-Jokimäki, M.-L.; et al. Biodiversity within the city: Effects of land sharing and land sparing urban development on avian diversity. Sci. Total Environ. 2020, 707, 135477. [Google Scholar] [CrossRef] [PubMed]
- Kark, S.; Iwaniuk, A.; Schalimtzek, A.; Banker, E. Living in the city: Can anyone become an ‘Urban Exploiter’? J. Biogeogr. 2007, 34, 638–651. [Google Scholar] [CrossRef]
- Croci, S.; Butet, A.; Clergeau, P. Does urbanization filter birds on the basis of their biological traits. Condor 2008, 110, 223–240. [Google Scholar] [CrossRef]
- Evans, K.L.; Chaberlain, D.E.; Hatchwell, B.J.; Gregory, R.D.; Gaston, K.J. What makes an urban bird? Glob. Chang. Biol. 2011, 17, 32–44. [Google Scholar] [CrossRef]
- Callaghan, C.T.; Major, R.E.; Wilshire, J.H.; Martin, J.M.; Kingsford, R.T.; Cornwell, W.K. Generalists are the most urban-tolerant of birds: A phylogenetically controlled analysis of ecological and life history traits using a novel continuous measure of bird responses to urbanization. Oikos 2019, 128, 845–858. [Google Scholar] [CrossRef]
- Callaghan, C.T.; Benedetti, Y.; Wilshire, J.H.; Morelli, F. Avian trait specialization is negatively associated with urban tolerance. Oikos 2020, 129, 1541–1551. [Google Scholar] [CrossRef]
- Shugart, H.H.; Patten, B.C. Niche Quantification and the Concept of Niche Pattern. In Systems Analysis and Simulation Ecology; Patten, B.C., Ed.; Academic Press: New York, NY, USA, 1972; pp. 283–327. [Google Scholar]
- Gregory, R.D.; Gaston, K.J. Explanations of commonness and rarity in British breeding birds, separating resource use and resource availability. Oikos 2000, 88, 5515–5526. [Google Scholar] [CrossRef]
- Shultz, S.; Bradbury, R.; Evans, K.L.; Gregory, R.; Blackburn, T. Brain size and resource specialization predict long-term population trends in British birds. Proc. R. Soc. B 2005, 272, 2305–2311. [Google Scholar] [CrossRef]
- Hurlbert, A.H.; White, E.P. Ecological correlates of geographical range occupancy in North American birds. Glob. Ecol. Biogeogr. 2007, 16, 764–773. [Google Scholar] [CrossRef]
- Rannap, R.; Lõhmus, A.; Briggs, L. Niche position, but not niche breadth, differs in two coexisting amphibians having contrasting trends in Europe. Divers. Distrib. 2009, 15, 692–700. [Google Scholar] [CrossRef]
- Greek Statistical Authority. Population Census 2011. Available online: http://www.statistics.gr/portal/page/portal/ESYE/PAGE-census2011 (accessed on 5 January 2021).
- Statistics Finland. Rovaniemi Population, January 2021. Available online: https://pxnet2.stat.fi/PXWeb/pxweb/en/StatFin/StatFin__vrm__vamuu/statfin_vamuu_pxt_11lj.px/table/tableViewLayout1/ (accessed on 5 January 2021).
- Bibby, C.; Burgess, N.; Hill, D.; Mustoe, S. Bird Census Techniques, 3rd ed.; Academic Press: London, UK, 2000. [Google Scholar]
- Van Heezik, Y.; Seddon, P.J. Accounting for detectability when estimating avian abundance in an urban area. N. Z. J. Ecol. 2012, 36, 391–397. [Google Scholar]
- Sólymos, P.; Lele, S.R.; Bayne, E. Conditional likelihood approach for analyzing single visit abundance survey data in the presence of zero inflation and detection error. Environmetrics 2012, 23, 197–205. [Google Scholar] [CrossRef]
- Green, D.M.; Baker, M.G. Urbanization impacts on habitat and bird communities in a Sonoran desert ecosystem. Landsc. Urban Plan. 2003, 36, 225–239. [Google Scholar] [CrossRef]
- Donnelly, R.; Marzluff, J.M. Importance of reserve size and landscape context to urban bird conservation. Conserv. Biol. 2004, 18, 733–745. [Google Scholar] [CrossRef]
- Litteral, J.; Wu, J. Urban landscape matrix affects avian diversity in remnant vegetation fragments: Evidence from the Phoenix metropolitan region, USA. Urban Ecosyst. 2012, 15, 939–959. [Google Scholar] [CrossRef]
- Rico-Silva, J.F.; Cruz-Trujillo, E.J.; Colorado, Z.G.J. Influence of environmental factors on bird diversity in greenspaces in an Amazonian city. Urban Ecosyst. 2021, 24, 365–374. [Google Scholar] [CrossRef]
- BirdLife International. European Birds of Conservation Concern: Populations, Trends and National Responsibilities; BirdLife International: Cambridge, UK, 2017. [Google Scholar]
- Hyvärinen, E.; Juslén, A.; Kemppainen, E.; Uddström, A.; Liukko, U.-M. The 2019 Red List of Finnish Species; Ministry of the Environment; Finnish Environment Institute: Helsinki, Finland, 2019.
- Legakis, A.; Maragou, P. The Red Data Book of Threatened Animals of Greece; Hellenic Zoological Society: Athens, Greece, 2009. [Google Scholar]
- Dolédec, S.; Chessel, D.; Gimaret-Carpentier, C. Niche separation in community analysis: A new method. Ecology 2000, 81, 2914–2927. [Google Scholar] [CrossRef]
- Dray, S.; Dufour, A.B. The ade4 Package: Implementing the Duality Diagram for Ecologists. J. Stat. Softw. 2007, 22, 1–20. [Google Scholar] [CrossRef]
- Harrell, F.E., Jr.; Dupont, C. Hmisc: Harrell Miscellaneous—R Package Version 4.4-1. 2020. Available online: https://CRAN.R-project.org/package=Hmisc (accessed on 5 January 2021).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 5 January 2021).
- Keller, V.; Herrando, S.; Voříšek, P.; Franch, M.; Kipson, M.; Milanesi, P.; Martí, D.; Anton, M.; Klvaňová, A.; Kalyakin, M.V.; et al. European Breeding Bird Atlas 2: Distribution, Abundance and Change; European Bird Census Council: Beek, The Netherlands; Lynx Edicions: Barcelona, Spain, 2020. [Google Scholar]
- Ahvenainen, J. Rovaniemen Historia II 1632–1960; Kirjapaino Oy: Kuopio, Finland, 1970. [Google Scholar]
- Jokimäki, J.; Huhta, E. Artificial nest predation and abundance of birds along an urban gradient. Condor 2000, 102, 838–847. [Google Scholar] [CrossRef]
- Murgui, E. Breeding habitat selection in the House martin Delichon urbica in the city of Valencia (Spain). Acta Ornithol. 2002, 37, 75–83. [Google Scholar] [CrossRef]
- Moudra, L.; Zasadil, P.; Moudry, V.; Salek, M. What makes new housing development unsuitable for house sparrows (Passer domesticus)? Landsc. Urban Plan. 2018, 169, 124–130. [Google Scholar] [CrossRef]
- Gaston, K.J.; Warren, P.H.; Thompson, K.; Smith, R.M. Urban domestic gardens (IV): The extent of the resource and its associated features. Biodivers. Conserv. 2005, 14, 3327–3349. [Google Scholar] [CrossRef]
- Balaji, S. Artificial nest box for house sparrow: An apt method to save the dwindling species in an urban environment. Int. J. Biodivers. Conserv. 2014, 6, 194–198. [Google Scholar] [CrossRef][Green Version]
- Ciach, M.; Fröhlich, A. Habitat type, food resources, noise and light pollution explain the species composition, abundance and stability of a winter bird assemblage in an urban environment. Urban Ecosyst. 2017, 20, 547–559. [Google Scholar] [CrossRef]
- Murgui, E. Factors influencing the bird community of urban wooded streets along an annual cycle. Ornis Fenn. 2007, 84, 66–77. [Google Scholar]
- Ferenc, M.; Sedláček, O.; Fuchs, R. How to improve urban greenspace for woodland birds: Site and local-scale determinants of bird species richness. Urban Ecosyst. 2014, 17, 625–640. [Google Scholar] [CrossRef]
- Tryjanowski, P.; Morelli, F.; Mikula, P.; Krištín, A.; Indykiewicz, P.; Grzywaczewski, G.; Kronenberg, J.; Jerzak, L. Bird diversity in urban green space: A large-scale analysis of differences between parks and cemeteries in Central Europe. Urban For. Urban Green. 2017, 27, 264–271. [Google Scholar] [CrossRef]
- Witt, K.; Mitschke, A.; Luniak, M. A comparison of common breeding bird populations in Hamburg, Berlin and Warsaw. Acta Ornithol. 2005, 40, 139–146. [Google Scholar] [CrossRef]
| Species Name | Species Code | Kavala | Rovaniemi | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Inertia | OMI (%) | T (%) | RT (%) | Inertia | OMI (%) | T (%) | RT (%) | ||
| Black-headed Gull | Chrrid | - | - | - | - | 12.1 | 79.0 | 3.9 | 17.1 |
| Feral Pigeon | Colliv | 23.6 | 37.0 * | 36.4 | 26.6 | - | - | - | - |
| Collared Dove | Strdec | 15.1 | 5.3 | 24.8 | 69.9 | - | - | - | - |
| Common Cuckoo | Cuccan | 27.8 | 41.8 * | 31.7 | 26.6 | - | - | - | - |
| European Bee-eater | Merapi | 40.1 | 47.0 * | 21.3 | 31.7 | - | - | - | - |
| Syrian Woodpecker | Densyr | 32.8 | 39.5 * | 29.1 | 31.3 | - | - | - | - |
| Northern House Martin | Delurb | 16.1 | 1.8 | 23.9 | 74.3 | - | - | - | - |
| Barn Swallow | Hirrus | 9.6 | 9.2 | 9.5 | 81.3 | - | - | - | - |
| White Wagtail | Motalb | 10.9 | 42.7 | 0.7 | 56.6 | 16.8 | 6.8 | 10.3 | 82.9 |
| Eurasian Blackbird | Turmer | 28.6 | 39.5 * | 33.0 | 27.5 | - | - | - | - |
| Fieldfare | Turpil | - | - | - | - | 15.5 | 10.8 | 22.4 | 66.8 |
| Sardinian Warbler | Sylmel | 23.1 | 29.4 | 25.4 | 45.2 | - | - | - | - |
| Willow Warbler | Phytro | 32.6 | 45.0 * | 30.4 | 24.7 | 19.7 | 15.3 | 15.8 | 68.9 |
| Olivaceous Warbler | Idupal | 14.9 | 25.1 | 0.0 | 74.9 | - | - | - | - |
| Common Nightingale | Lusmeg | 19.1 | 22.6 | 16.1 | 61.4 | - | - | - | - |
| Great Tit | Parmaj | 24.7 | 11.1 | 34.9 | 54.0 | 17.5 | 6.6 | 25.9 | 67.5 |
| Eurasian Blue Tit | Cyacae | 32.6 | 36.2 * | 20.6 | 43.2 | 23.1 | 38.0 * | 8.0 | 54.1 |
| Eurasian Magpie | Picpic | 13.1 | 58.1 * | 5.0 | 36.9 | 25.9 | 64.7 * | 4.9 | 30.5 |
| Eurasian Jay | Gargla | 34.2 | 41.7 * | 33.5 | 24.8 | - | - | - | - |
| Western Jackdaw | Cormon | 17.4 | 33.1 * | 14.4 | 52.4 | 19.1 | 43.5 * | 21.9 | 34.6 |
| Hooded Crow | Corcor | 15.7 | 6.0 | 26.4 | 67.6 | 17.3 | 34.7 * | 14.6 | 50.7 |
| Common Starling | Stuvul | 13.5 | 21.7 | 17.3 | 61.0 | - | - | - | - |
| House Sparrow | Pasdom | 14.8 | 11.2 | 26.1 | 62.8 | 14.6 | 11.9 | 16.6 | 71.5 |
| Eurasian Tree Sparrow | Pasmon | 13.8 | 87.2 * | 0.3 | 12.5 | 11.9 | 20.9 | 11.4 | 67.7 |
| Common Chaffinch | Fricoe | 22.3 | 20.2 * | 33.8 | 46.0 | 16.2 | 22.4 * | 26.0 | 51.6 |
| European Goldfinch | Carcar | 17.3 | 4.3 | 15.4 | 80.3 | - | - | - | - |
| European Greenfinch | Carchl | 21.2 | 8.9 | 34.2 | 56.9 | 17.0 | 42.2 * | 14.1 | 43.6 |
| Redpoll | Acafla | - | - | - | - | 19.5 | 38.4 | 17.9 | 43.6 |
| European Serin | Serser | 21.7 | 60.7 | 17.1 | 22.2 | - | - | - | - |
| Eurasian Siskin | Spispi | - | - | - | - | 12.4 | 12.8 | 4.3 | 82.9 |
| Environmental Variables | Code | Kavala | Rovaniemi | ||
|---|---|---|---|---|---|
| Axis1 | Axis2 | Axis1 | Axis2 | ||
| Tree cover (%) | TRC | 0.538 * | −0.799 *** | 0.554 * | −0.578 ** |
| Shrub cover (%) | SHC | 0.395 | 0.362 | 0.269 | 0.043 |
| Bare ground cover (%) | GRC | 0.302 | 0.393 | 0.120 | 0.933 *** |
| Gray cover (%) | GRAY | −0.783 *** | 0.591 ** | −0.754 *** | −0.509 * |
| Deciduous tree cover (%) | TRCD | −0.437 | −0.275 | 0.252 | −0.323 |
| Coniferous tree cover (%) | TRCC | 0.726 *** | −0.510 * | 0.349 | −0.356 |
| Green space size (ha) | SIZE | 0.696 *** | −0.551 * | 0.712 ** | 0.069 |
| Green space perimeter (km) | EL | 0.666 ** | −0.590 ** | 0.658 ** | 0.221 |
| Relative edge length (km/km2) | REL | −0.369 | 0.617 ** | −0.745 *** | −0.538 * |
| Distance from city edge (km) | EDGE | 0.045 | 0.062 | 0.035 | −0.766 *** |
| Distance from city center (km) | DC | 0.440 | −0.035 | 0.788 *** | −0.011 |
| Building height (floors/building) | FL | −0.576 ** | −0.366 | −0.734 *** | 0.077 |
| Morning noise level (dBA) | N1 | −0.872 *** | −0.111 | −0.688 ** | 0.029 |
| Midday noise level (dBA) | N2 | −0.699 *** | −0.417 | −0.746 *** | −0.246 |
| Car traffic (cars/min) | CAR | −0.679 *** | −0.492 * | −0.530 * | −0.172 |
| Pedestrian traffic (people/min) | PED | −0.644 ** | −0.593 ** | −0.727 *** | −0.213 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liordos, V.; Jokimäki, J.; Kaisanlahti-Jokimäki, M.-L.; Valsamidis, E.; Kontsiotis, V.J. Niche Analysis and Conservation of Bird Species Using Urban Core Areas. Sustainability 2021, 13, 6327. https://doi.org/10.3390/su13116327
Liordos V, Jokimäki J, Kaisanlahti-Jokimäki M-L, Valsamidis E, Kontsiotis VJ. Niche Analysis and Conservation of Bird Species Using Urban Core Areas. Sustainability. 2021; 13(11):6327. https://doi.org/10.3390/su13116327
Chicago/Turabian StyleLiordos, Vasilios, Jukka Jokimäki, Marja-Liisa Kaisanlahti-Jokimäki, Evangelos Valsamidis, and Vasileios J. Kontsiotis. 2021. "Niche Analysis and Conservation of Bird Species Using Urban Core Areas" Sustainability 13, no. 11: 6327. https://doi.org/10.3390/su13116327
APA StyleLiordos, V., Jokimäki, J., Kaisanlahti-Jokimäki, M.-L., Valsamidis, E., & Kontsiotis, V. J. (2021). Niche Analysis and Conservation of Bird Species Using Urban Core Areas. Sustainability, 13(11), 6327. https://doi.org/10.3390/su13116327







