Factors Controlling the Spatial Distribution and Temporal Trend of Nationwide Groundwater Quality in Korea
Abstract
1. Introduction
2. Methods and Materials
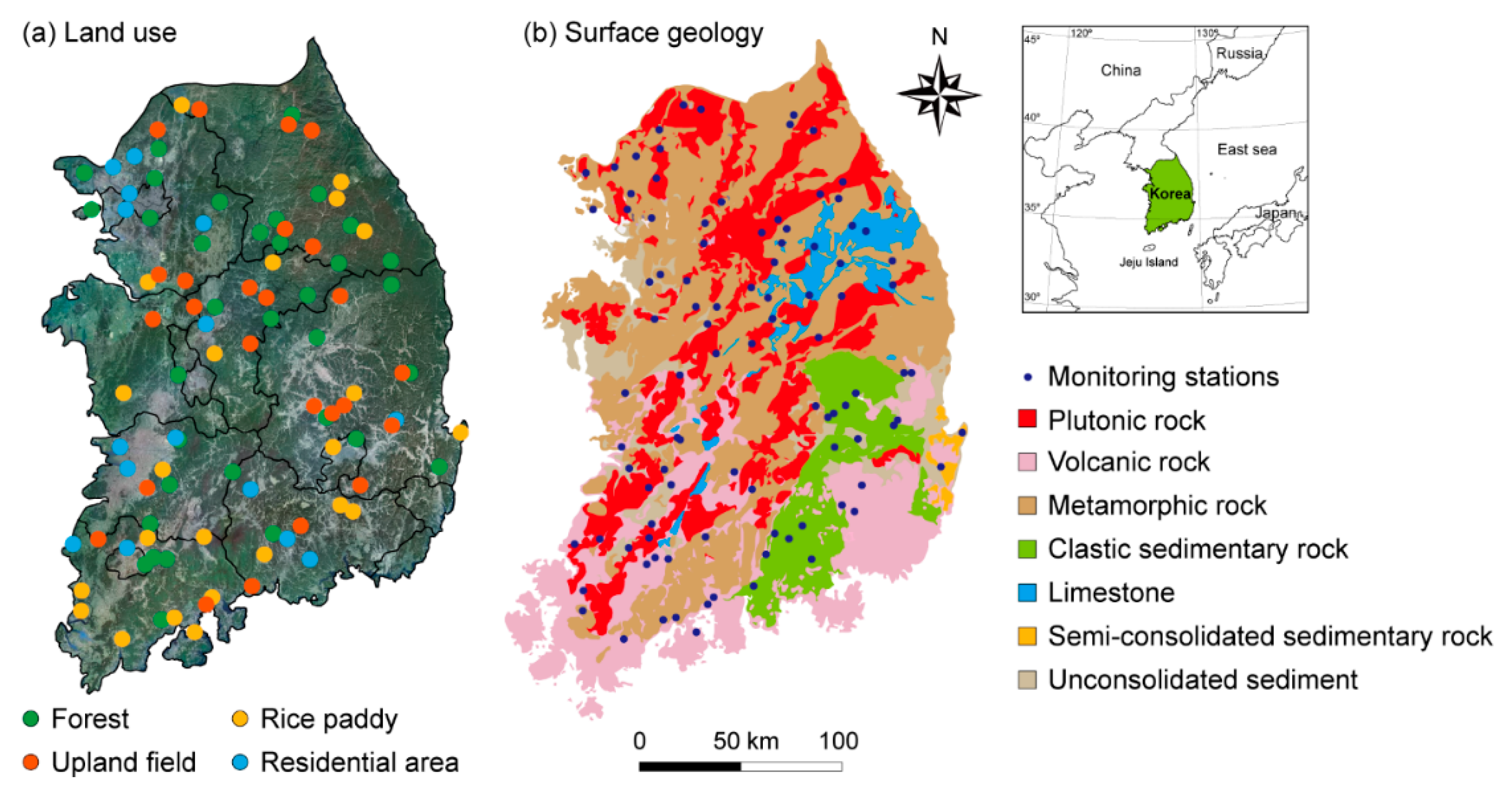
2.1. Study Area, Hydrometeorology, and Land Use
2.2. Collection of Groundwater Quality Data
2.3. Statistical Data Analysis
3. Results and Discussion
3.1. General Chemistry of Groundwater
3.2. Spatial Distribution of Groundwater Quality
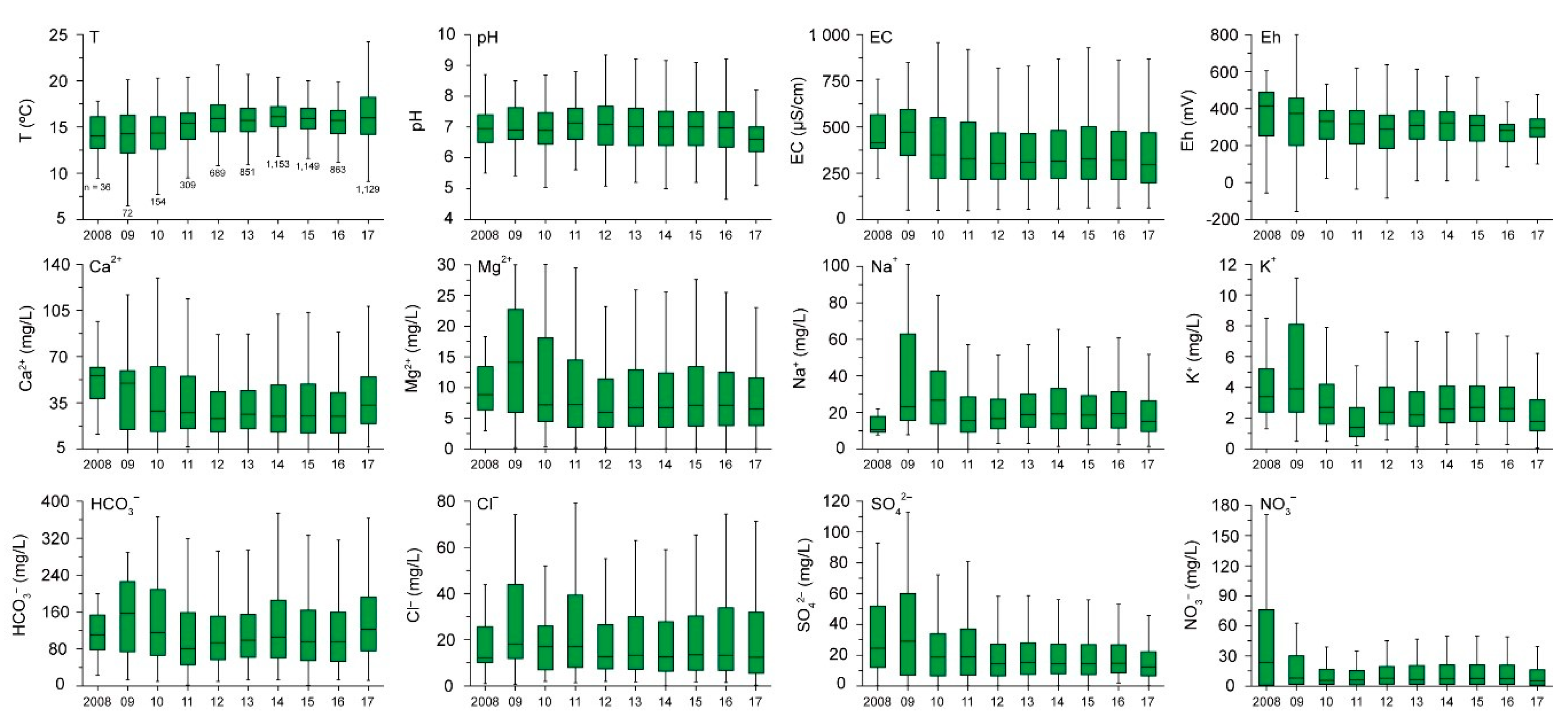
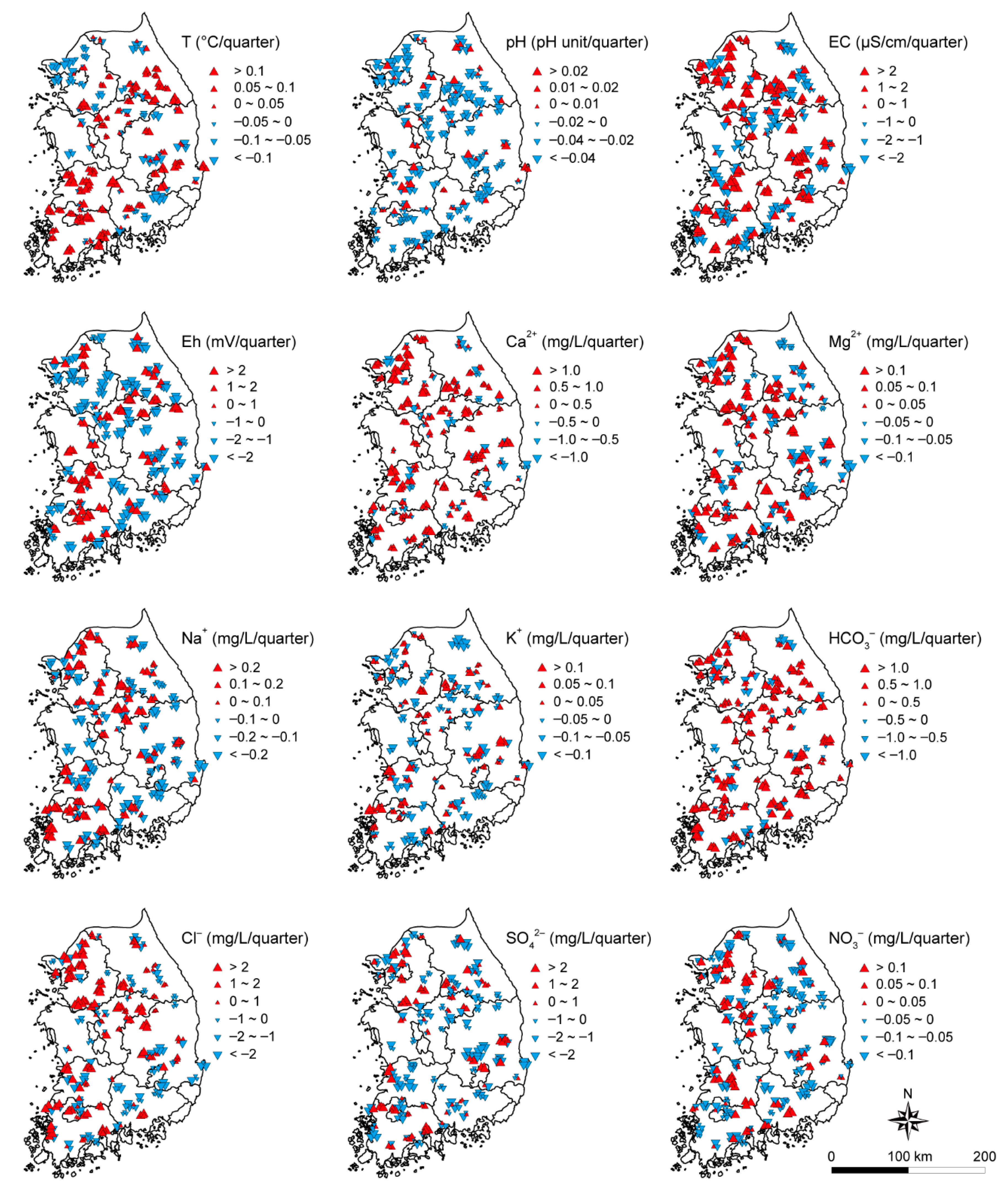
3.3. Temporal Trend of Groundwater Quality
3.4. Major Factors Controlling Spatiotemporal Distribution of Groundwater Quality
3.4.1. Fertilizers Application
3.4.2. Sewage Leakage
3.4.3. Seawater Intrusion
3.5. Mechanism for Controlling the Hydrochemistry of Groundwater
3.6. Statistical Verification of Ions Co-Occurrence and Sources
4. Conclusions
- Six parameters; T, EC, Ca2+, Mg2+, HCO3−, and Cl− had dominant increasing trends in groundwater during the studied period, revealing the effect of natural and anthropogenic factors. Other parameters, pH, Eh, Na+, K+, SO42−, and NO3− showed dominant decreasing trends over time.
- Among land uses, upland fields had the highest mean value for groundwater NO3− (22.2 mg/L), confirming that many fertilizers were applied to upland fields. Thus, agricultural activities were the main controlling factor of groundwater NO3− contamination in agricultural areas.
- In residential areas, Cl− and Na+ had high mean values in groundwater of 705.3 mg/L and 298.4 mg/L, respectively. Thus, domestic activities were the controlling factor of groundwater Cl− and Na+ contamination in residential areas.
- Some monitoring wells exceeded the Korean drinking water standards for NO3− and Cl−. The contamination derived from human activities can be sufficiently reduced by effective active measures. Therefore, the government has to strengthen corresponding regulations in order to minimize deterioration of groundwater quality.
- Mean Cl− (457.4 mg/L) of 15 monitoring stations in coastal areas was much higher than that of the monitoring stations in inland. Seawater intrusion naturally or as a result of groundwater over-pumping was the controlling factor of groundwater Cl− in coastal areas.
- Groundwater chemistry was predominantly controlled by the mechanism of geogenic rock dominance. The results of Pearson correlation and PCA collectively verified mineral dissolution or carbonate rock weathering as the major source of ions in groundwater, along with concurrence of evaporite dissolution.
- Expansion of monitoring stations to areas where the GQMN is not currently installed will provide a better evaluation of groundwater quality in the future.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Diamantini, E.; Lutz, S.R.; Mallucci, S.; Majone, B.; Merz, R.; Bellin, A. Driver detection of water quality trends in three large European river basins. Sci. Total Environ. 2018, 612, 49–62. [Google Scholar] [CrossRef]
- Xu, B.; Wang, G. Surface water and groundwater contaminations and the resultant hydrochemical evolution in the Yongxiu area, west of Poyang Lake, China. Environ. Earth Sci. 2016, 75, 184. [Google Scholar] [CrossRef]
- Li, P.; He, S.; Yang, N.; Xiang, G. Groundwater quality assessment for domestic and agricultural purposes in Yan’an City, northwest China: Implications to sustainable groundwater quality management on the Loess Plateau. Environ. Earth Sci. 2018, 77, 775. [Google Scholar] [CrossRef]
- Lee, J.Y.; Raza, M.; Kwon, K.D. Land use and land cover changes in the Haean Basin of Korea: Impacts on soil erosion. Episodes 2019, 42, 17–32. [Google Scholar] [CrossRef]
- Giri, S.; Qiu, Z. Understanding the relationship of land uses and water quality in Twenty First Century: A review. J. Environ. Manag. 2016, 173, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Jahnke, C.; Wannous, M.; Troeger, U.; Falk, M.; Struck, U. Impact of seawater intrusion and disposal of desalinization brines on groundwater quality in El Gouna, Egypt, Red Sea Area. Process analyses by means of chemical and isotopic signatures. Appl. Geochem. 2019, 100, 64–76. [Google Scholar] [CrossRef]
- Yadav, S.; Babel, M.S.; Shrestha, S.; Deb, P. Land use impact on the water quality of large tropical river: Mun River Basin, Thailand. Environ. Monit. Assess. 2019, 191, 614. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Wu, Y.; Groves, C.; Yuan, D.; Kambesis, P. Natural and anthropogenic factors affecting the groundwater quality in the Nandong karst underground river system in Yunan, China. J. Contam. Hydrol. 2009, 109, 49–61. [Google Scholar] [CrossRef]
- Kim, S.G.; Kim, G.B. Are groundwater monitoring networks economical? Cost-benefit analysis on the long-term groundwater supply project of South Korea. Water 2019, 11, 753. [Google Scholar] [CrossRef]
- Ministry of Environment and Korea Environment Institute. Management Method of Groundwater Monitoring System and Intensification with New Technology; Ministry of Environment: Sejong, Korea, 2018; pp. 31–48.
- Duan, W.; He, B.; Nover, D.; Yang, G.; Chen, W.; Meng, H.; Zou, S.; Liu, C. Water quality assessment and pollution source identification of the eastern Poyang Lake Basin using multivariate statistical methods. Sustainability 2016, 8, 133. [Google Scholar] [CrossRef]
- Raza, M.; Farooqi, A.; Niazi, N.K.; Ahmad, A. Geochemical control on spatial variability of fluoride concentrations in groundwater from rural areas of Gujrat in Punjab, Pakistan. Environ. Earth Sci. 2016, 75, 1364. [Google Scholar] [CrossRef]
- Belkhiri, L.; Mouni, L.; Tiri, A. Water-rock interaction and geochemistry of groundwater from the Ain Azel aquifer, Algeria. Environ. Geochem. Health 2012, 34, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Stuart, M.; Chilton, P.; Kinniburgh, D.; Cooper, D. Screening for long-term trends in groundwater nitrate monitoring data. Q. J. Eng. Geol. Hydrogeol. 2007, 40, 361–376. [Google Scholar] [CrossRef]
- Kim, J.; Ryoo, R.; Lee, J.; Song, D.; Lee, Y.J.; Jun, H.B. Study on major mineral distribution characteristics in groundwater in South Korea. J. Korean Soc. Environ. Eng. 2016, 38, 566–573. [Google Scholar] [CrossRef]
- Lee, L.; Ahn, K.; Min, B.; Yang, M.; Choi, I.; Chung, H.; Park, J. The study on the ion water characteristics of raw water in the domestic natural mineral water. J. Korean Soc. Water Environ. 2016, 32, 442–449. [Google Scholar] [CrossRef][Green Version]
- Lee, J.Y.; Kwon, K.D.; Park, Y.C.; Jeon, W.H. Unexpected nationwide nitrate declines in groundwater of Korea. Hydrol. Process. 2017, 31, 4693–4704. [Google Scholar] [CrossRef]
- Verma, S.; Mukherjee, A.; Mahanta, C.; Choudhury, R.; Mitra, K. Influence of geology on groundwater–sediment interactions in arsenic enriched tectono-morphic aquifers of the Himalayan Brahmaputra river basin. J. Hydrol. 2016, 540, 176–195. [Google Scholar] [CrossRef]
- Jeon, C.; Raza, M.; Lee, J.Y.; Kim, H.; Kim, C.S.; Kim, B.; Kim, J.W.; Kim, R.H.; Lee, S.W. Countrywide groundwater quality trend and suitability for use in key sectors of Korea. Water 2020, 12, 1193. [Google Scholar] [CrossRef]
- Bacal, M.C.J.O.; Hwang, S.; Guevarra-Segura, I. Predictive lithologic mapping of South Korea from geochemical data using decision trees. J. Geochem. Explor. 2019, 205, 106326. [Google Scholar] [CrossRef]
- Korea Environment Corporation. Investigation of Groundwater Quality by Groundwater Quality Monitoring Network; Korea Environment Corporation: Incheon, Korea, 2018; pp. 1–12. [Google Scholar]
- Wetzel, H.A.; Mooi, E.; Sarstedt, M.; Mooi-Reci, I. Market Research: The Process, Data, and Methods Using Stata; Springer: Singapore, 2018; pp. 1–416. [Google Scholar]
- A Correspondence Table for Non Parametric and Parametric Tests. Available online: https://blog.minitab.com/blog/applying-statistics-in-quality-projects/a-correspondence-table-for-non-parametric-and-parametric-tests (accessed on 27 August 2013).
- Lee, S.Y.; Roh, Y.; Jeong, J.T. Changes of the oxidation/reduction potential of groundwater by the biogeochemical activity of indigenous bacteria. Econ. Environ. Geol. 2014, 47, 61–69. [Google Scholar] [CrossRef][Green Version]
- Oehler, T.; Eiche, E.; Putra, D.; Adyasari, D.; Hennig, H.; Mallast, U.; Moosdorf, N. Seasonal variability of land-ocean groundwater nutrient fluxes from a tropical karstic region (southern Java, Indonesia). J. Hydrol. 2018, 565, 662–671. [Google Scholar] [CrossRef]
- Moujabber, M.E.; Samra, B.B.; Darwish, T.; Atallah, T. Comparison of different indicators for groundwater contamination by seawater intrusion on the Lebanese Coase. Water Resour. Manag. 2006, 20, 161–180. [Google Scholar] [CrossRef]
- Lee, J.Y.; Hahn, J.S. Characterization of groundwater temperature obtained from the Korean national groundwater monitoring stations: Implications for heat pumps. J. Hydrol. 2006, 329, 514–526. [Google Scholar] [CrossRef]
- Kaown, D.; Hyun, Y.; Bae, G.O.; Oh, C.W.; Lee, K.K. Evaluation of spatio-temporal trends of groundwater quality in different land uses using Kendall test. Geosci. J. 2012, 16, 65–75. [Google Scholar] [CrossRef]
- Watari, T.; Kotcharoen, W.; Ormine, T.; Hatamoto, M.; Araki, N.; Oshiki, M.; Mimura, K.; Nagano, A.; Yamaguchi, T. Formation of denitrifying granules in an upflow sludge blanket reactor with municipal sewage and sodium nitrate feeding. Environ. Technol. Innov. 2020, 19, 100861. [Google Scholar] [CrossRef]
- Rajmohan, N.; Elango, L. Nutrient chemistry of groundwater in an intensively irrigated region of southern India. Environ. Geol. 2005, 47, 820–830. [Google Scholar] [CrossRef]
- Hem, J.D. Study and Interpretation of the Chemical Characteristics of Natural Water, 3rd ed.; U.S. Government Printing Office: Washington, DC, USA, 1985; pp. 1–109.
- Li, J.Y.; Li, T.Y. Seasonal and annual changes in soil/cave air pCO2 and the δ13CDIC of cave drip water in response to changes in temperature and rainfall. Appl. Geochem. 2018, 93, 94–101. [Google Scholar] [CrossRef]
- Saha, S.; Reza, A.S.; Roy, M.K. Hydrochemical evaluation of groundwater quality of the Tista floodplain, Rangpur, Bangladesh. Appl. Water Sci. 2019, 9, 198. [Google Scholar] [CrossRef]
- Woltersdorf, L.; Scheidegger, R.; Liehr, S.; Doll, P. Municipal water reuse for urban agriculture in Namibia: Modeling nutrient and salt flows as impacted by sanitation user behavior. J. Environ. Manag. 2016, 169, 272–284. [Google Scholar] [CrossRef]
- Lee, C.S.; Gil, S.G.; Jeong, J.H.; Kim, J.H.; Choi, G.Y. Research for Effective Management and Reasonable Evaluation Standard of Eco-Friendly Fertilizer; Ministry for Food, Agriculture, Forestry and Fisheries: Sejong, Korea, 2012; pp. 1–149.
- Park, S.C.; Yun, S.T.; Chae, G.T.; Yoo, I.S.; Shin, K.S.; Heo, C.H.; Lee, S.K. Regional hydrochemical study on salinization of coastal aquifers, western coastal aquifers, western coastal area of South Korea. J. Hydrol. 2005, 313, 182–194. [Google Scholar] [CrossRef]
- Lee, H.; Koo, M.H.; Kim, K.; Kim, Y. Spatio-temporal variations in stream-aquifer interactions following construction of weirs in Korea. Groundwater 2015, 54, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.K.; Lee, Y.J.; Kang, S.S.; Yoon, H.B.; Hong, S.Y.; Kim, L.H.; Kim, Y.H.; Kim, M.S.; Seong, J.K.; Lee, J.Y.; et al. Manual of Fertilizer Recommendation Program of Heug-Toram; National Institute of Agricultural Sciences: Wanju, Korea, 2013; pp. 1–97. [Google Scholar]
- Fertilizer Application. Available online: http://kosis.kr/statHtml/statHtml.do?orgId=101&tblId=DT_2KAA415_OECD (accessed on 10 October 2019).
- Kim, H.S.; Um, D.H.; Seo, I.G.; Kim, Y.I.; Kim, H.I.; Kim, H.J.; Kim, J.S.; Oh, K.Y.; Oh, S.Y. Environment Friendly Agricultural Infrastructures Scheme to Reduce Nutrients (N, P) Loading of Agricultural Drainage Water; Korea Rural Community Corporation: Naju, Korea, 2002; pp. 1–112. [Google Scholar]
- Ministry of Environment. The Guidelines for Groundwater Quality Preservation; Ministry of Environment: Sejong, Korea, 2014; pp. 1–416.
- Overbo, A.; Heger, S.; Kyser, S.; Asleson, B.; Gulliver, J. Chloride Contributions from Water Softeners and Other Domestic, Commercial, Industrial, and Agricultural Sources to Minnesota Waters; University of Minnesota: Minneapolis, MN, USA, 2019; pp. 1–34. [Google Scholar]
- Eo, H.J.; Kim, J.S. Analysis of correlations between mineral contents in waters and sensory characteristics of coffee. Culi. Sci. Hos. Res. 2017, 23, 105–115. [Google Scholar]
- Statistics of Sewer Pipe in 2018. Available online: http://me.go.kr/home/web/policy_data/read.do?pagerOffset=0&maxPageItems=10&maxIndexPages=10&searchKey=&searchValue=&menuId=10264&orgCd=&condition.code=A5&condition.deleteYn=N&seq=7440 (accessed on 31 December 2019).
- Zhang, B.; Zhao, D.; Zhou, P.; Qu, S.; Liao, F.; Wang, G. Hydrochemical characteristics of groundwater and dominant water-rock interactions in the Delingha Area, Qaidam Basin, Northwest China. Water 2020, 12, 836. [Google Scholar] [CrossRef]
- Marandi, A.; Shand, P. Groundwater chemistry and the Gibbs diagram. Appl. Geochem. 2018, 97, 209–212. [Google Scholar] [CrossRef]
- Sarwade, D.V.; Nandakumar, M.V.; Kesari, M.P.; Mondal, N.C.; Singh, V.S.; Singh, B. Evaluation of seawater ingress into an Indian atoll. Environ. Geol. 2007, 52, 1475–1483. [Google Scholar] [CrossRef]
- Sidhu, N.; Rishi, M.S.; Herojeet, R.K. Groundwater quality variation with respect to aquifer dispositioning in urbanized watershed of Chandigarh, India. Int. J. Environ. Ecol. Fam. Urban Stud. 2013, 3, 87–98. [Google Scholar]
- Pant, R.R.; Zhang, F.; Rehman, F.U.; Wang, G.; Ye, M.; Zeng, C.; Tang, H. Spatiotemporal variations of hydrogeochemistry and its controlling factors in the Gandaki River Basin, Central Himalaya Nepal. Sci. Total Environ. 2018, 622, 770–782. [Google Scholar] [CrossRef]
- Li, C.; Gao, X.; Wang, Y. Hydrogeochemistry of high-fluoride groundwater at Yuncheng Basin, northern China. Sci. Total Environ. 2015, 508, 155–165. [Google Scholar] [CrossRef]
- Kaown, D.; Koh, D.C.; Yu, H.E.; Kim, H.; Yoon, Y.Y.; Yum, B.W.; Lee, K.K. Combined effects of recharge and hydrogeochemical conditions on nitrate in groundwater of a highland agricultural basin based on multiple environmental tracers. Agric. Water Manag. 2020, 240, 106327. [Google Scholar] [CrossRef]

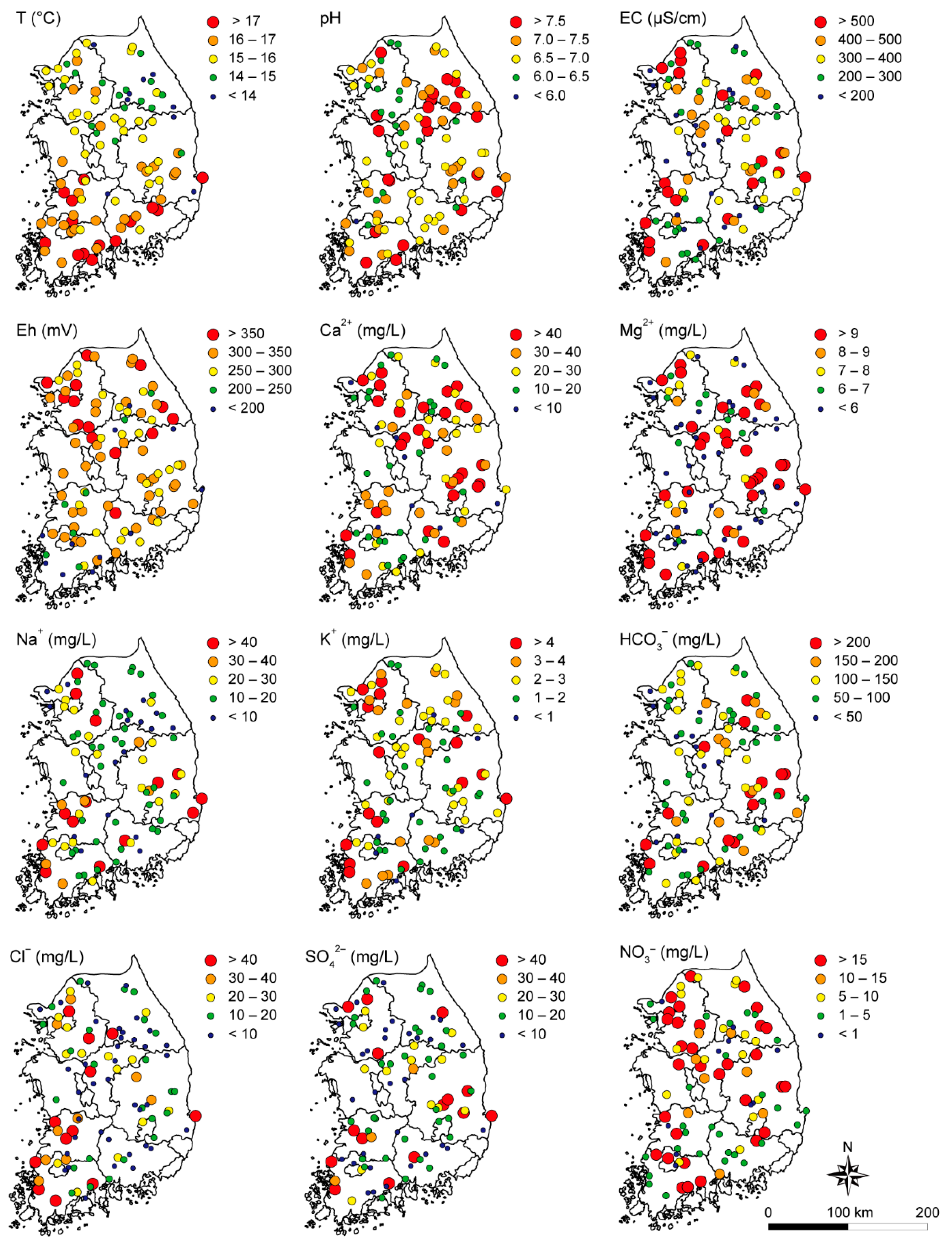

| Parameters | Maximum | Minimum | Mean | Median | SD 1 | CV 2 | ND 3 (%) |
|---|---|---|---|---|---|---|---|
| T (°C) | 27.8 | 4.4 | 15.9 | 15.8 | 2.3 | 0.15 | 0 |
| pH | 10.4 | 4.3 | 7.0 | 6.9 | 0.8 | 0.11 | 0 |
| EC (µS/cm) | 48,100 | 46 | 769 | 317 | 3586 | 4.66 | 0 |
| Eh (mV) | 892 | −159 | 288 | 302 | 123 | 0.43 | 0 |
| Ca2+ (mg/L) | 4000 | <0.05 | 57.1 | 26.5 | 232 | 4.06 | 0.2 |
| Mg2+ (mg/L) | 2357 | 0.03 | 20.8 | 6.8 | 113 | 5.43 | 0.2 |
| Na+ (mg/L) | 8640 | <0.05 | 99.8 | 17.8 | 601 | 6.02 | 0.02 |
| K+ (mg/L) | 3200 | <0.05 | 5.0 | 2.4 | 41.3 | 8.26 | 0.02 |
| HCO3− (mg/L) | 1198 | 1.0 | 127 | 102 | 95.1 | 0.75 | 0 |
| Cl− (mg/L) | 30,479 | <0.05 | 201 | 13.3 | 1550 | 7.71 | 0.02 |
| SO42− (mg/L) | 1983 | <0.05 | 46.6 | 14.5 | 162 | 3.48 | 2.6 |
| NO3− (mg/L) | 368 | <0.05 | 16.8 | 6.9 | 30.4 | 1.81 | 13.1 |
| Parameters | Maximum | Minimum | Mean | Median | Increasing (%) | Decreasing (%) |
|---|---|---|---|---|---|---|
| T | 0.3 | −0.5 | 0.01 | 0.02 | 63 | 37 |
| pH | 0.07 | −0.2 | −0.03 | −0.02 | 19 | 81 |
| EC | 97.3 | −379 | −0.7 | 0.7 | 57 | 43 |
| Eh | 15.2 | −23.1 | −1.4 | −1.3 | 37 | 63 |
| Ca2+ | 16.8 | −86.7 | 0.2 | 0.3 | 72 | 28 |
| Mg2+ | 4.8 | −34.9 | −0.09 | 0.03 | 61 | 39 |
| Na+ | 46.9 | −72.7 | 0.1 | −0.05 | 41 | 59 |
| K+ | 1.5 | −12.9 | −0.06 | −0.01 | 40 | 60 |
| HCO3− | 11.0 | −8.9 | 0.9 | 0.7 | 73 | 27 |
| Cl− | 70.7 | −375 | −0.2 | 0.01 | 52 | 48 |
| SO42− | 6.4 | −31.6 | −0.6 | −0.1 | 34 | 66 |
| NO3− | 2.1 | −8.5 | −0.2 | −0.05 | 33 | 67 |
| Parameters | Land Use | |||
|---|---|---|---|---|
| Upland Field | Rice Paddy | Forest | Residential Area | |
| NO3− (mg/L) | 22.2 | 16.6 | 13.4 | 16.2 |
| Cl− (mg/L) | 19.4 | 297.3 | 27.8 | 705.3 |
| Na+ (mg/L) | 24.2 | 144.1 | 29.6 | 298.4 |
| Parameters | Mean (Coastal Area, n = 15) | Mean (Inland Area, n = 82) |
|---|---|---|
| Cl− (mg/L) | 457.4 | 17.9 |
| Na+ (mg/L) | 280.9 | 20.5 |
| Ca2+ (mg/L) | 40.1 | 33.1 |
| Mg2+ (mg/L) | 26.3 | 8.8 |
| K+ (mg/L) | 9.5 | 2.7 |
| EC (µS/cm) | 1445 | 348 |
| Parameters | T | pH | EC | Eh | Ca2+ | Mg2+ | Na+ | K+ | HCO3− | Cl− | SO42− | NO3− |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T | 1 | |||||||||||
| pH | −0.03 * | 1 | ||||||||||
| EC | 0.06 ** | 0.02 | 1 | |||||||||
| Eh | −0.13 ** | −0.31 ** | −0.20 ** | 1 | ||||||||
| Ca2+ | 0.05 ** | 0.00 | 0.88 ** | −0.16 ** | 1 | |||||||
| Mg2+ | 0.06 ** | 0.01 | 0.90 ** | −0.17 ** | 0.95 ** | 1 | ||||||
| Na+ | 0.08 ** | 0.02 | 0.93 ** | −0.21 ** | 0.87 ** | 0.89 ** | 1 | |||||
| K+ | 0.00 | 0.00 | 0.32 ** | −0.07 ** | 0.34 ** | 0.26 ** | 0.26 ** | 1 | ||||
| HCO3− | 0.09 ** | 0.26 ** | 0.07 ** | −0.25 ** | 0.06 ** | 0.08 ** | 0.10 ** | 0.05 ** | 1 | |||
| Cl− | 0.07 ** | 0.01 | 0.93 ** | −0.18** | 0.94 ** | 0.95 ** | 0.96 ** | 0.30 ** | 0.04 ** | 1 | ||
| SO42− | 0.08 ** | 0.01 | 0.83 ** | −0.20 ** | 0.74 ** | 0.76 ** | 0.85 ** | 0.33 ** | 0.23 ** | 0.81 ** | 1 | |
| NO3− | −0.08 ** | −0.22 ** | −0.03 ** | 0.20 ** | −0.01 | −0.04 ** | −0.06 ** | 0.00 | −0.18 ** | −0.05 ** | −0.03 ** | 1 |
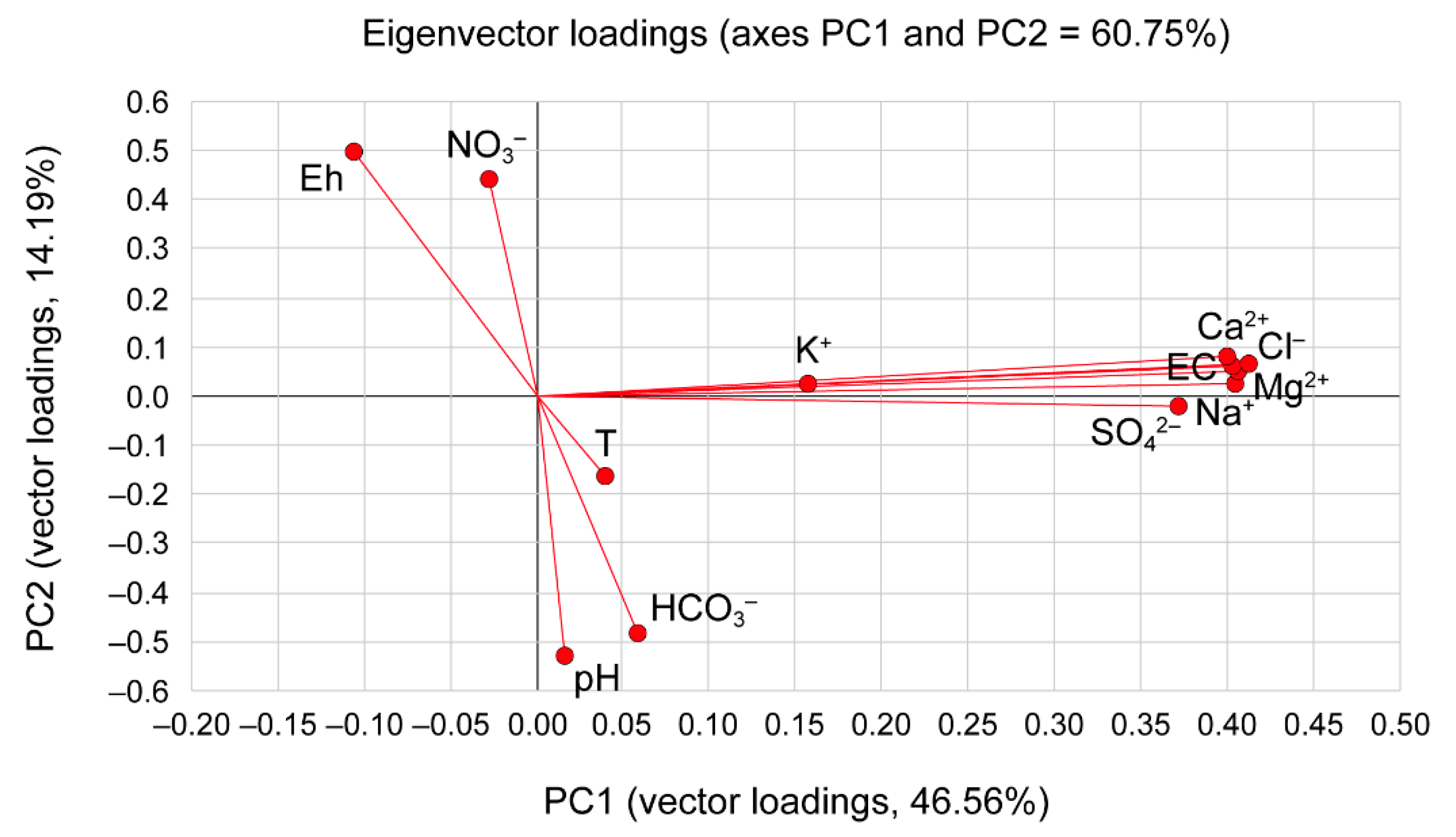
| Parameters | PC1 | PC2 | PC3 |
|---|---|---|---|
| T | 0.09 | −0.21 | 0.92 |
| pH | 0.04 | −0.69 | −0.35 |
| EC | 0.96 | 0.06 | −0.01 |
| Eh | −0.24 | 0.65 | −0.04 |
| Ca2+ | 0.94 | 0.10 | −0.03 |
| Mg2+ | 0.95 | 0.08 | −0.01 |
| Na+ | 0.96 | 0.03 | 0.03 |
| K+ | 0.37 | 0.03 | −0.18 |
| HCO3− | 0.14 | −0.63 | −0.03 |
| Cl− | 0.98 | 0.09 | 0.00 |
| SO42− | 0.88 | −0.03 | 0.01 |
| NO3− | −0.06 | 0.58 | −0.06 |
| Eigenvalues | 5.59 | 1.70 | 1.02 |
| Variance (%) | 46.56 | 14.19 | 8.48 |
| Cumulative (%) | 46.56 | 60.75 | 69.23 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, C.-S.; Raza, M.; Lee, J.-Y.; Kim, H.; Jeon, C.; Kim, B.; Kim, J.-W.; Kim, R.-H. Factors Controlling the Spatial Distribution and Temporal Trend of Nationwide Groundwater Quality in Korea. Sustainability 2020, 12, 9971. https://doi.org/10.3390/su12239971
Kim C-S, Raza M, Lee J-Y, Kim H, Jeon C, Kim B, Kim J-W, Kim R-H. Factors Controlling the Spatial Distribution and Temporal Trend of Nationwide Groundwater Quality in Korea. Sustainability. 2020; 12(23):9971. https://doi.org/10.3390/su12239971
Chicago/Turabian StyleKim, Chang-Seong, Maimoona Raza, Jin-Yong Lee, Heejung Kim, Chanhyeok Jeon, Bora Kim, Jeong-Woo Kim, and Rak-Hyeon Kim. 2020. "Factors Controlling the Spatial Distribution and Temporal Trend of Nationwide Groundwater Quality in Korea" Sustainability 12, no. 23: 9971. https://doi.org/10.3390/su12239971
APA StyleKim, C.-S., Raza, M., Lee, J.-Y., Kim, H., Jeon, C., Kim, B., Kim, J.-W., & Kim, R.-H. (2020). Factors Controlling the Spatial Distribution and Temporal Trend of Nationwide Groundwater Quality in Korea. Sustainability, 12(23), 9971. https://doi.org/10.3390/su12239971









