1. Introduction
According to the European Environmental Agency (EEA) greenhouse gas emissions in the EU-28 in 2015 were 4452 million tonnes CO
2 equivalent and the share from the agriculture sector was 11.3%, mainly from ruminants [
1]. In the EU, poultry meat production has increased by 11% in a single decade (2005–2015) reaching over 12 million tonnes of poultry meat in 2015, and it is expected to rise by another 6% by 2025 [
2]. Among the different meat segments, the growth rate of poultry meat is predicted to have the highest development globally in the near future. In order to cope with the increased demand for poultry meat, intensive poultry farming is required. However, intensive livestock production generates significant waste disposal issues and environmental impacts. The estimated accumulation of animal manure in the EU is approximately 1.4 billion tonnes/year and the share of poultry litter is about 7.9% [
3]. Traditionally, poultry litter which includes manure, bedding material, egg shells, feathers, etc., has been applied as a fertiliser and soil conditioner via direct land application [
4]. However, land application alone is no longer a sustainable solution to the growing volume of animal waste and likely to be uneconomic [
5]. Therefore, new waste treatment technologies addressing sustainability challenges at the nexus of food production and energy are needed to tackle the disposal problem of poultry litter.
The EU’s 2030 Framework for Climate and Energy Policies has recently been revised and the share of EU energy to be produced from renewable resources was raised to 27% [
6]. Bioenergy technologies could play a vital role in achieving the EU2030 targets by reducing the greenhouse gas emissions arising from manure management and fertiliser application while increasing the share of renewable bioenergy. Alternatively, poultry litter or animal manure (dried) could be converted into liquid fuel and value-added products using thermochemical conversion technologies [
7,
8]. Small or modular pyrolysis units situated directly on poultry farms could produce sufficient bio-oil to make a farm self-sustainable by off-setting fossil fuel consumption for heating poultry houses resulting in economic sustainability and also providing bio-fertiliser for soil amendment (nutrient rich biochar). A recent review paper analysed the characteristics of poultry litter as fuel and explored different thermochemical conversion processes for energy integration [
9].
Pyrolysis is one of the most attractive alternative options amongst the bioenergy conversion technologies to produce biofuels and soil conditioners from poultry litter. Pyrolysis is a thermal decomposition process that converts biomass and other organic matter into high energy density solids (biochar), liquids (bio-oil) and gases (syngas/biogas) in the absence of an oxidising agent (oxygen-free environment). It is a complex thermochemical process involving both simultaneous and successive reactions of drying and devolatilisation [
10]. The product yields from a pyrolysis process is dependent on various process parameters such as the temperature within the reactor, residence time, heating rate of the feedstock, particle size of the feedstock, pressure and reactor configuration [
11]. Pyrolysis can be categorised into slow, medium and fast pyrolysis. In fast pyrolysis, the decomposition of feedstock occurs rapidly, resulting in the production of vapour and aerosols and smaller amounts of biochar and gas. Following condensation, a dark-brown homogenous liquid is formed, with a calorific value roughly half that of fossil fuel oil [
10]. Fluidised bed reactors are the recommended technology for fast pyrolysis because of their ease of operation, stability, scaling-up potential and high bio-oil yield [
12].
Poultry litter has a relatively high ash content and at an elevated temperature this can give rise to ash fusion and sintering associated with potassium, phosphorous and other alkali materials present in the ash leading to defluidisation or bed agglomeration problem. Since the pyrolysis temperatures are normally kept in the range of 400 to 600 °C, defluidisation or bed agglomeration problems should not occur [
13,
14].
Pyrolysis technology has so far been used predominantly on biomass with very few studies addressing feedstock originating from livestock. In an investigation, five manure-based feedstocks (i.e., swine-separated solids; paved feedlot manure; dairy manure; poultry litter; and turkey litter) were pyrolysed at 300 and 700 °C. Amongst these poultry litters, char exhibited the highest electro conductivity (a parameter measuring nutrients in the substrate) [
8]. In another investigation, biochar produced from chicken manure using a batch-type tubular pyrolysis reactor was used as a green catalyst to produce biodiesel (fatty acid methyl ester). The pseudo-catalytic transesterification reaction performance of chicken manure biochar compared well against commercially available porous materials (SiO
2), resulting in 95.6% fatty acid methyl ester yield at 350 °C. The study also recommended that in order to avoid thermal cracking of inorganic species (Ca) in chicken manure biochar, it should be produced at temperatures lower than 350 °C [
15]. The pattern of product gas evolution during the pyrolysis and gasification of chicken manure was studied at different temperatures and oxygen enrichment in a batch reactor, and the authors concluded that the cumulative energy yields from the gasification process were higher compared to pyrolysis [
16]. Pyrolysis and gasification of chicken manure have been experimentally studied using different gasifying media (i.e., nitrogen, air, carbon dioxide and steam) and temperatures. It was recommended that the gasifying media should be chosen based on the availability of resources and the desired output [
17]. Recently, an experimental analysis of slow pyrolysis of poultry litter was investigated at different temperatures using a laboratory-scale fixed-bed reactor. The pyrolytic gases evolved were estimated to be energetically adequate for the process to be self-sufficient. The energy transfer analysis showed that one-third of the heating value of the poultry litter was transferred to the organic liquid condensate and roughly the same was retained in the char. In this study, the maximum fraction of liquid condensate that could be upgraded to liquid biofuels was reported to be obtained at 550 °C [
18].
The biocrude oil produced from fast pyrolysis of poultry litter is less acidic (pH values between 3.6–6.3) and with a higher calorific value (26–30 MJ/kg) compared to hardwood biocrude oil (23 MJ/kg). The higher calorific value and nitrogen content in the biocrude oils produced from poultry litter were due to the protein and nitrogen contents, respectively, in the raw poultry litter [
19,
20,
21]. Nevertheless, biocrude oils produced from poultry litter are viscous and unstable compared to fossil derived petroleum fuels [
19,
22]. Biochar produced from poultry litter can also be used as a source for the preparation of activated carbon [
23]. Several other batch and fixed-bed pyrolysis experiments were conducted for bio-oil production using a wide variety of feedstocks such as poultry industry waste [
24], swine manure [
25], food waste [
26] and other agricultural and urban organic waste for biochar production [
27].
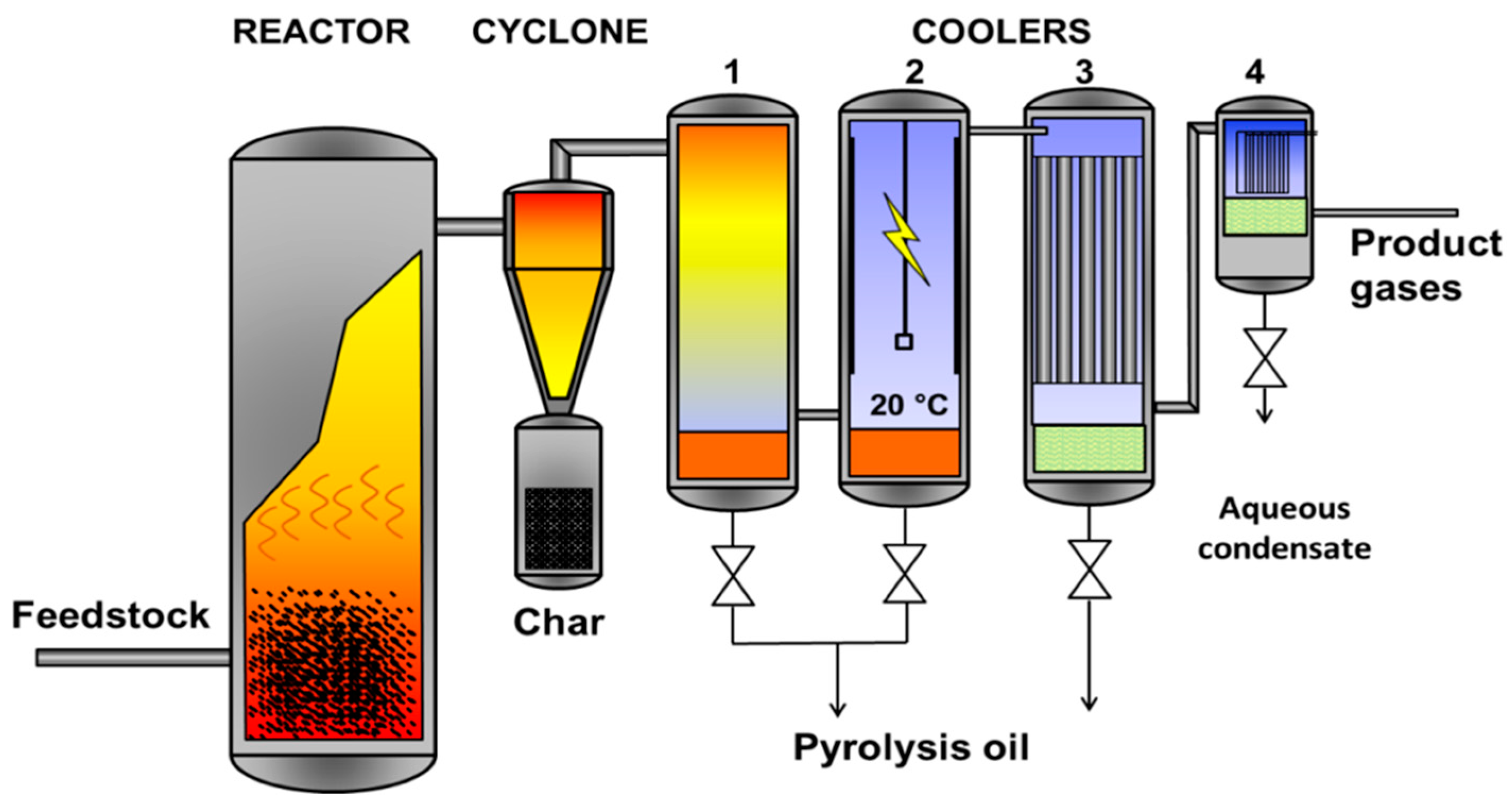
Several fixed-bed and batch pyrolysis studies have been carried out on poultry litter and they validated its potential for sanitation and waste-to-energy valorisation. However, the results obtained from batch pyrolysis units cannot be used for up-scaling and design purposes. In contrast, relatively few studies have focused on fast pyrolysis of poultry litter in a fluidised bed reactor due to the high ash and moisture contents. The present study proposes a small-scale fluidised bed fast pyrolysis unit operating at relatively low temperature as a safe and economical way of disposing of poultry litter. The aim of this study is to explore the suitability of poultry litter as an alternative feedstock in fast pyrolysis for bio-oil production. Furthermore, the outcome of this work can be helpful in the design, scale-up and deployment of poultry litter-based pyrolysis units at farm level for combined heat and power generation and to make the poultry farms self-sufficient in energy. This work investigated the technical feasibility of fast pyrolysis processing of poultry litter as well as assessing the potential utilisation of the evolved gases and biochar (potential source of organic nutrients).
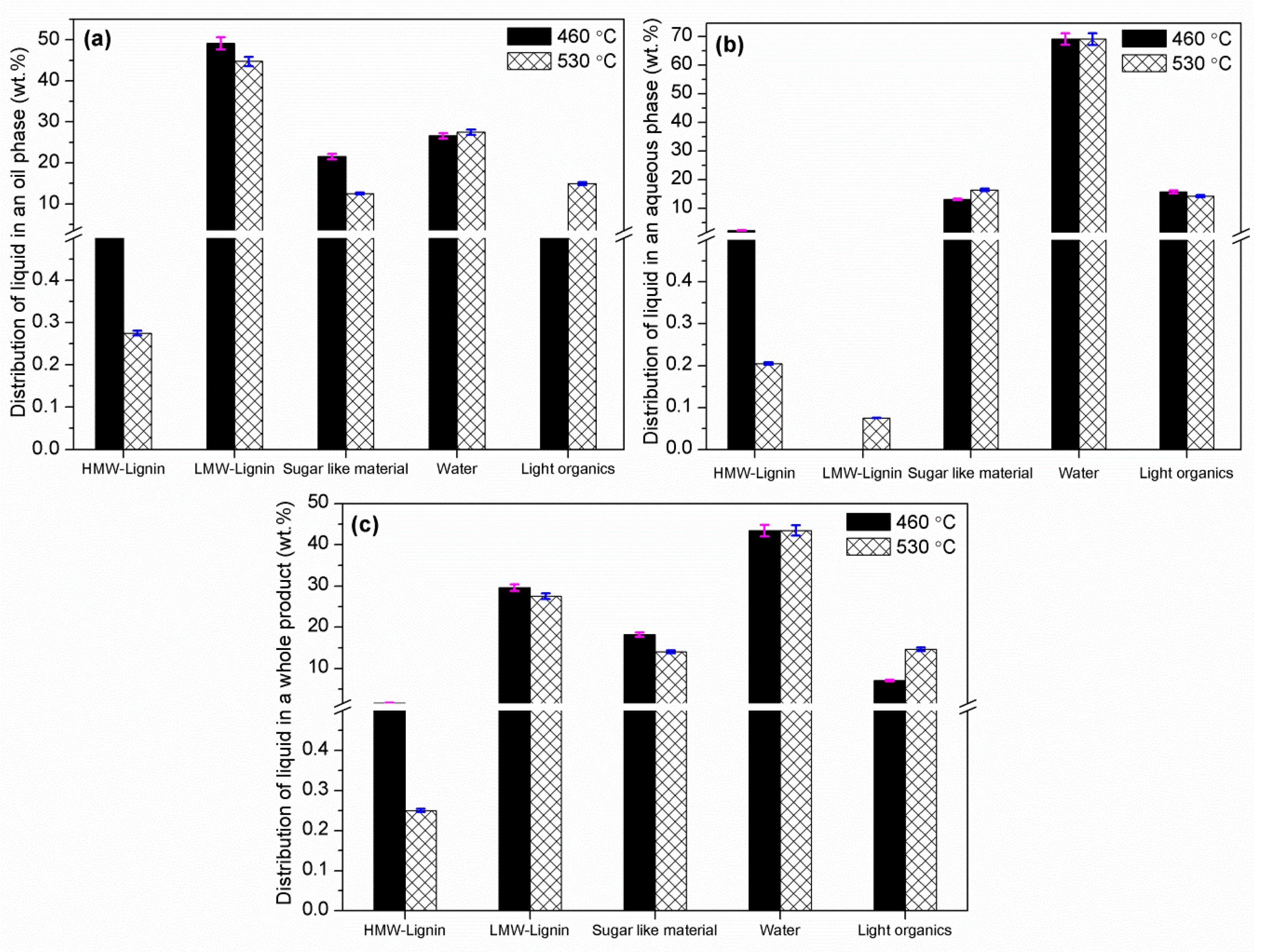
4. Conclusions
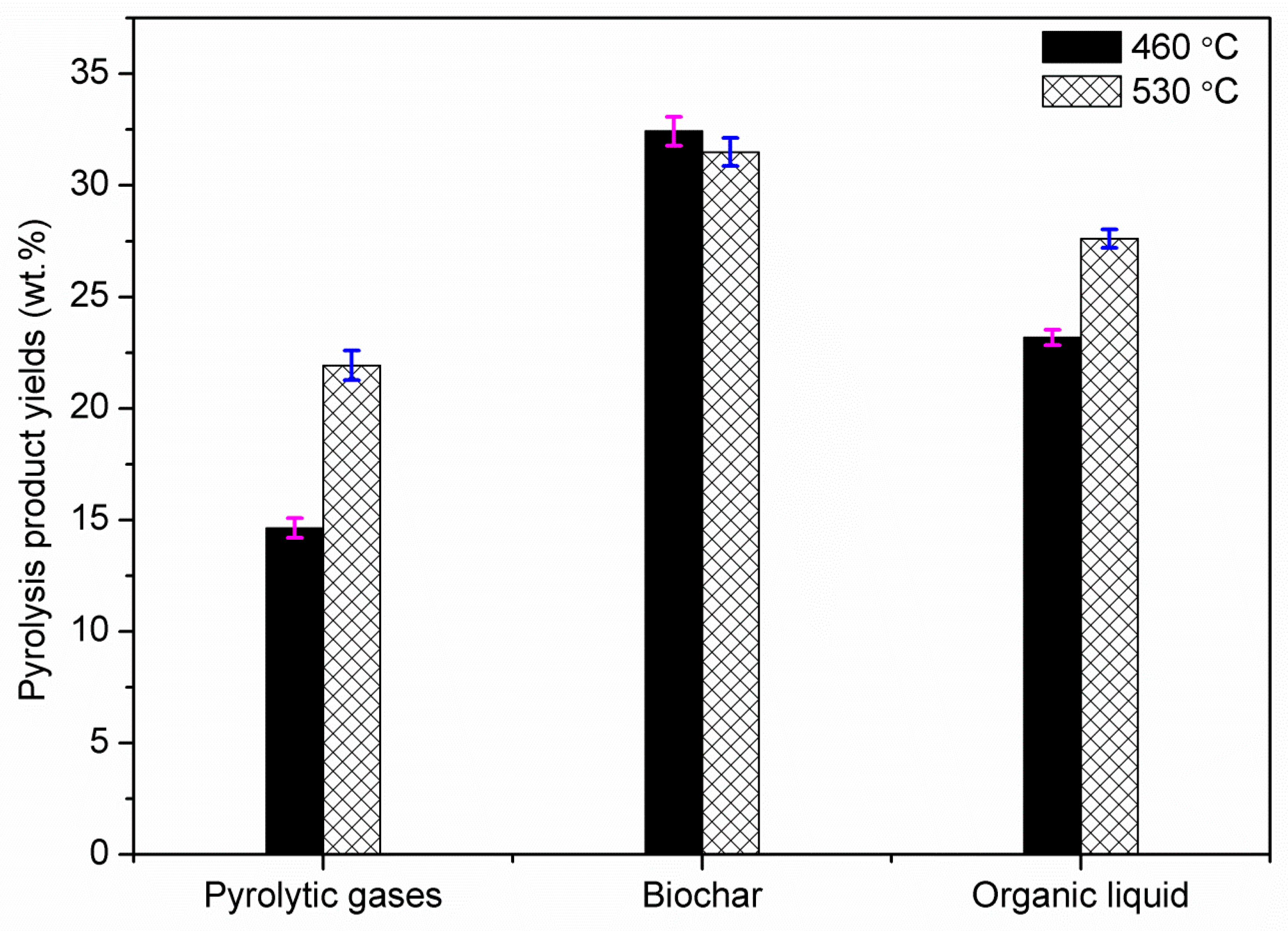
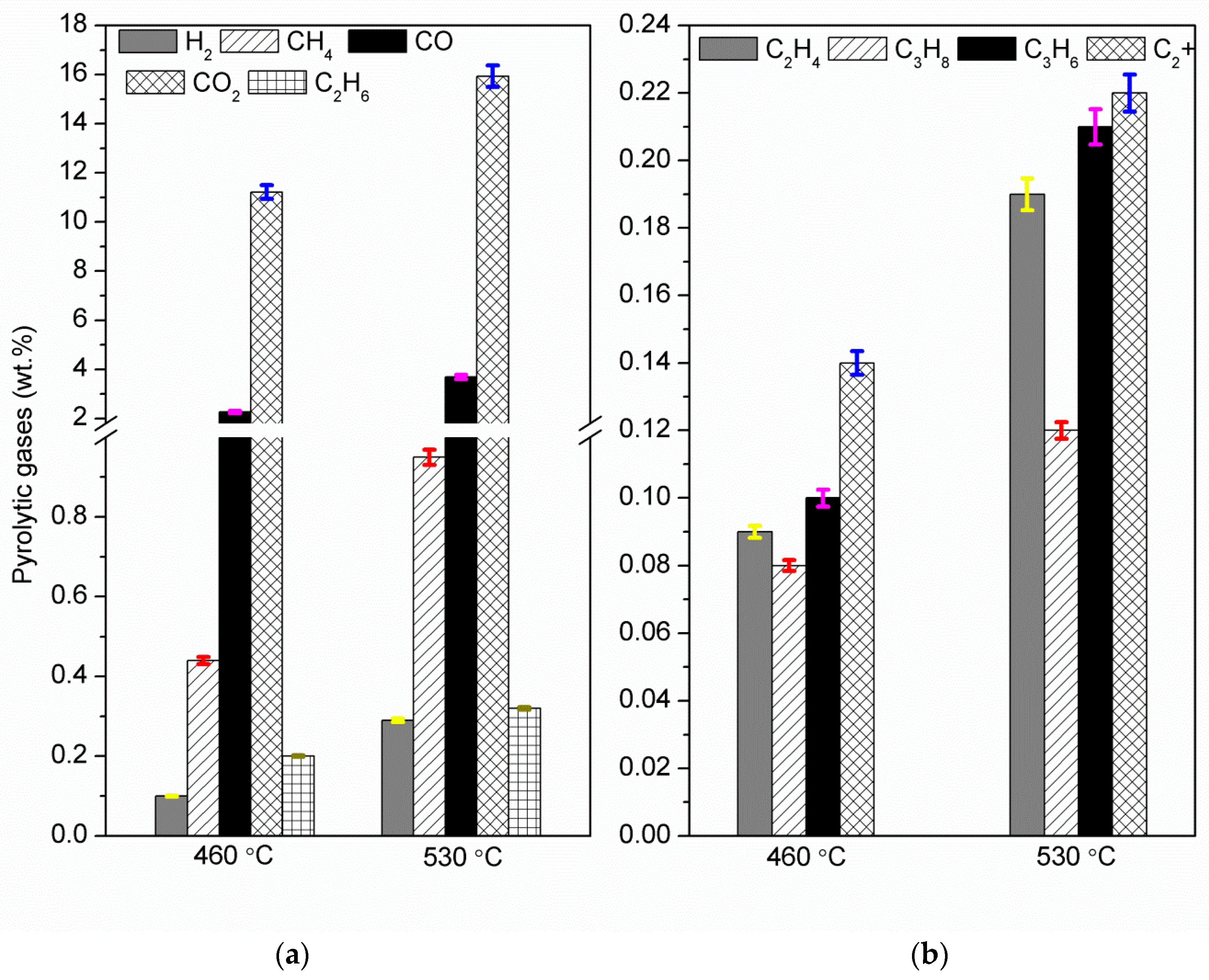
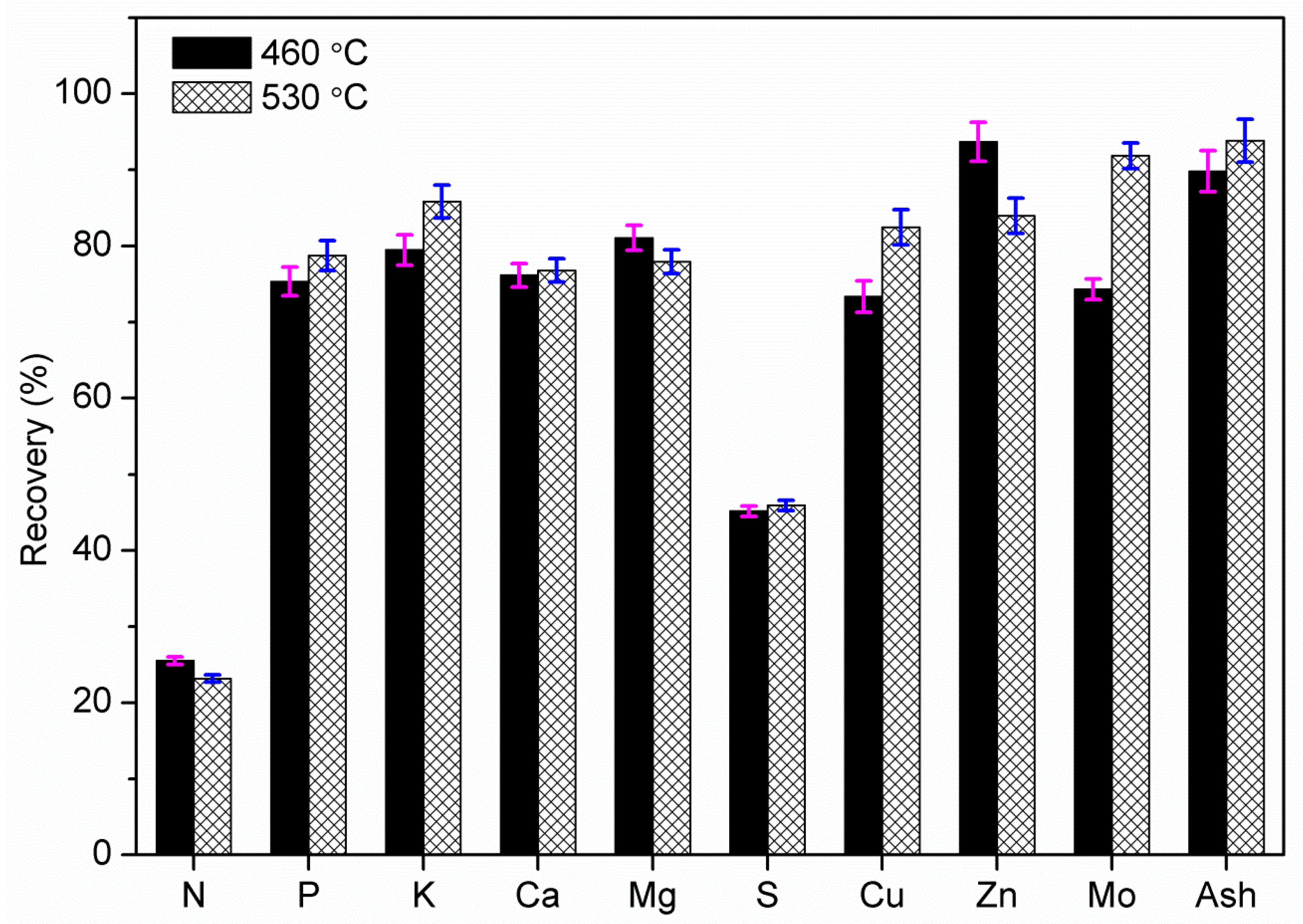
Fast pyrolysis of poultry litter was experimentally studied at two different temperatures (460 and 530 °C) in a bubbling fluidised bed reactor. The effect of reactor temperature on product gas, bio-oil and biochar were studied. The maximum organic bio-oil (27.62 wt.%) and pyrolytic gas yield (~22 wt.%) were obtained at 530 °C with CO2, CO and CH4 as the main gas products, whereas the maximum biochar yield was obtained at 460 °C. Owing to high ash content in poultry litter, biochar yield was comparatively higher than the bio-oil yield. Nevertheless, bio-oil from fast pyrolysis of poultry litter gave a 50% higher HHV compared to the oil produced from wood. The bio-oil produced was further characterised using elemental and ICP analyses. The ICP showed the presence of S, K, P, Si, Mg, and Ca and Fe were also detected in the liquid products. Therefore, further thermal and/or catalytic treatment is required before the bio-oil can be utilised in downstream application for heat and power generation. Most of the heavy metals and trace elements present in poultry litter were retained in the biochar including non-volatile minerals P and K. The retention of the non-volatile minerals (alkali and alkaline earth metals) in the biochar emphasises that the manure-derived biochar could be used in agriculture systems, but the concentration of trace elements may restrict its use. Therefore, before applying these biochars as a soil amendment, an exhaustive investigation is needed to assess their impact on ground water (leaching), soil conditions and on pot-plant growth. The carbon content in the biochar collected from the cyclone was in the range of 47–48.5 wt.% (db) with a lower concentration of hydrogen. The increase in pyrolysis temperature showed an increase of ash content and slight decrease in H, N and O contents in biochar.