1. Introduction
Currently, biodiversity is considered crucial for agricultural landscapes also having a beneficial impact for cropping systems, especially if it does not limit the achievement of optimal yield [
1,
2]. In recent decades, biodiversity in farmland gradually declined, mainly because of simplified crop rotations, and ever more intensive cultivation practices [
3,
4]. Consequently, current crop production systems should be rescheduled to limit the dependence on external inputs and at the same time to foster an adequate enlargement of sustainability and thus biodiversity and crop yield [
5,
6].
Weed flora is considered, to date, one of the main causes that interfere in a relevant way with the quantity and quality of agricultural production, even if, on the other hand, some authors point out that weed flora is also an important element that characterizes the floristic biodiversity of countryside [
7,
8]. Furthermore, weed flora is fundamental in favoring the biodiversity of a landscape since it offers shelter and nourishment to a wide range of fauna (insects, birds, small mammals) [
9,
10,
11,
12].
Currently, weed control management scheduling is addressed to limit dependence on herbicides by keeping the weed flora at a tolerable threshold of control instead of maintaining the crop totally free of weeds [
13]. The aim is to develop sustainable cropping systems reducing reliance on herbicides by means of lower herbicide doses than labelled ones [
14], covering crops or using mulch [
15,
16,
17], mechanical [
18], and flame weeding [
19]. From the perspective of herbicide underdosage adoption, some components of weed communities are expected to be altered. Other than weed species density and richness, these components also involved the overall composition and association of weeds [
20,
21]. The investigation of the phytostructural effects due to herbicide underdosage is a key step in order to set up weed management systems characterized by a reduced reliance on chemical inputs [
21].
Serious concerns arose about the adoption of lower herbicide doses than labeled ones, due to the potential onset of herbicide-resistant weed species [
22,
23]. However, it is strategical to provide herbicides at the right dose and time in order to manage weeds at a tolerable level and to preserve the crop from the phytotoxicity of the herbicide. Previous studies have demonstrated that labelled herbicide doses are recommended to guarantee a successful degree of weed control in different environments characterized by various pedo-climatic characteristics, and weed flora. Moreover, the same manufacturing industries are aware that there is a broad variety of conditions where herbicide use may be kept at reduced doses, although these conditions are somewhat unpredictable [
24,
25,
26]. Underdosed herbicide use (50–80% reduction with respect to labeled dose) has been reported and applied for maize on over 50% of the maize-cropped area in The Netherlands and over 80% of the maize-cropped in Denmark, German, and France [
27]. Additionally, according to Neve et al. [
28] the onset of herbicide-resistant weeds is a great threat in monoculture systems with respect to cropping systems where rotation is regularly practiced [
29]. Other authors report that crop rotation is one of the determining factors of the soil seedbank and absence of crop rotation may lead to a weed flora assemblage with a lower biodiversity and thus reducing herbicide available options to manage weeds [
30,
31].
The oilseed rape crop, which we studied in the present paper, is considered a strong rooting-break species and an improver of soil structure [
32,
33]. For this last reason, in Mediterranean environments, it is commonly grown in an annual rotation with winter cereals [
32,
34,
35], thus minimizing the risk to cause the selection of herbicide-weed resistance when herbicides are applied at reduced doses.
To our knowledge, studies on the effects of underdosage herbicide in oilseed rape are very limited, and, on the same crop, investigations highlighting typical weed composition were mainly carried out on continental environments [
36,
37,
38,
39,
40].
This study was aimed to analyze the effects of underdosed herbicides and untreated plots on floristic composition and species diversity in a Mediterranean oilseed rape cropping system. Specifically, this analysis was focused on detecting possible changes of the weed flora community in terms of relative weed abundance, richness, and diversity as a result of underdosed herbicide application.
4. Discussion
Sustainable intensification, assuming a decrease in the use of agrochemicals (fertilizer, herbicide etc.), has the positive aspect to promote ecosystem services within which the conservation of weed flora diversity occurs without a reduction of arable land for food production [
52,
53]. The primary goal of our study was to determine whether reduction of herbicide dose used would significantly increase weed diversity and composition. Our study represents an assessment of the weed vegetation of oilseed rape fields in a Mediterranean environment, aiming to highlight how weed composition is affected by an underdose herbicide factor. In our experiment, weeding management based upon the non- and low use of herbicides fostered a significantly greater weed species diversity than did conventional management, namely with the labelled herbicide dose. The magnitude of change in weed community composition observed in this study was slightly smaller than that observed by Fried et al. [
37] in France and by Hanzlik and Gerowitt [
38] in Germany. However, our findings showed a slightly higher diversity than studies carried out in Denmark and United Kingdom in oilseed rape arable fields [
40,
54].
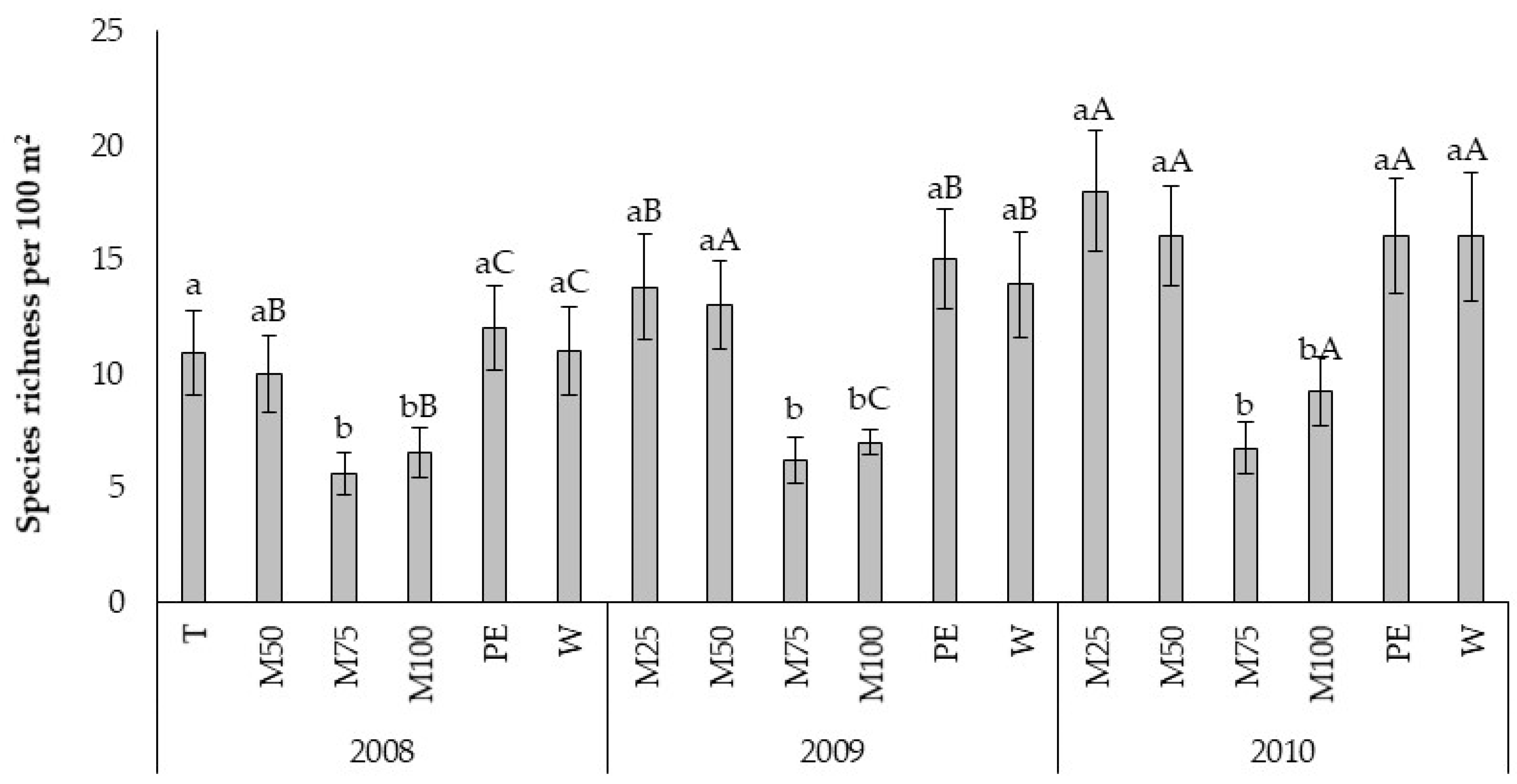
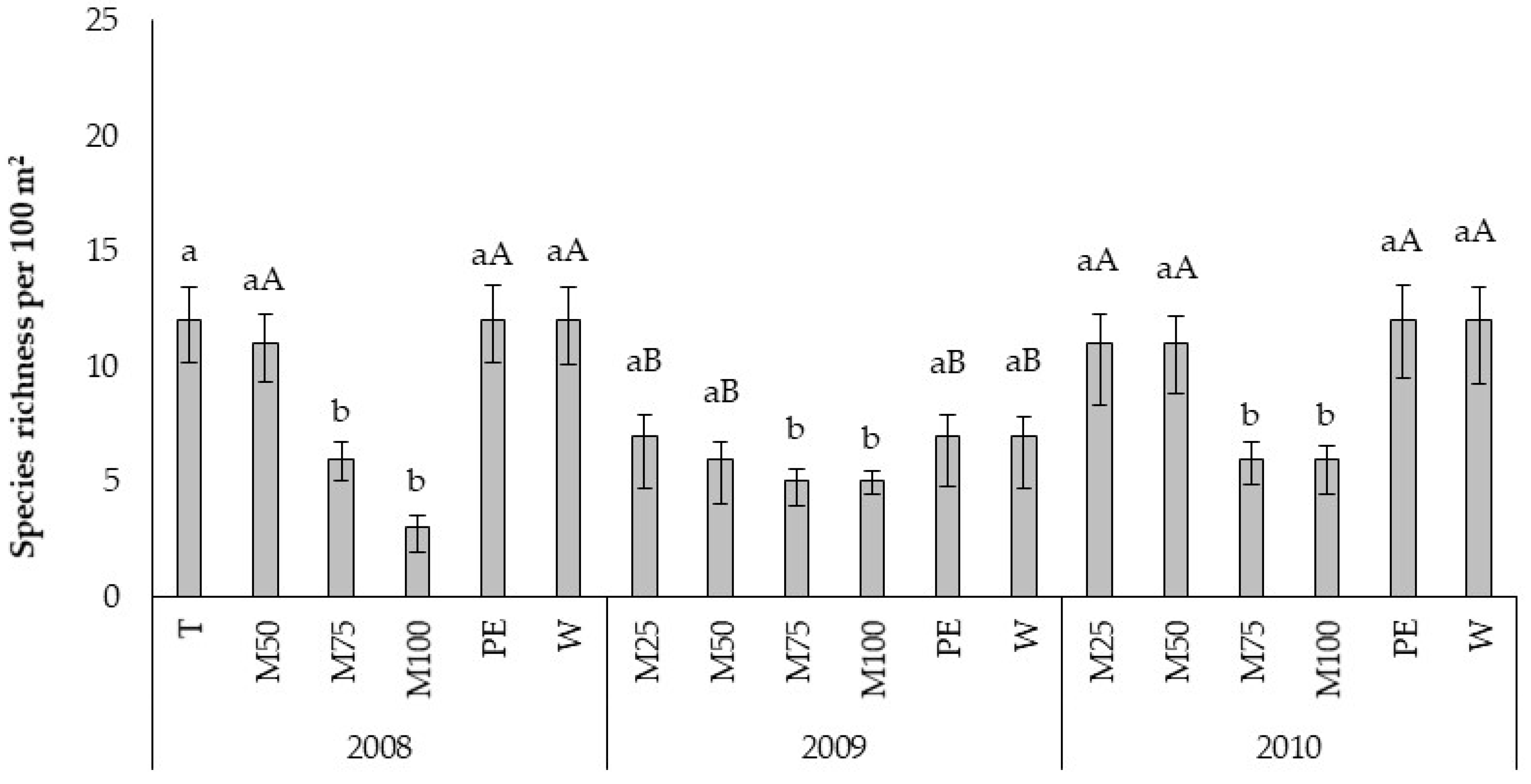
In particular, in the USN experiment, environmental and site effects played a key role in highlighting specific patterns in weed composition, as, being equal treatments, the site of Ussana recorded each year a number of species lower than Ottava. The importance of landscape heterogeneity (e.g., pedo-climatic characteristics, previous land-uses) in the arrangement of a weed community was previously highlighted by other authors [
55], and sometimes, contrasting results were reported, likely as a consequence of weed assembly variation rather than consistent shifts in community composition [
56,
57,
58]. As expected, the weed species richness and diversity increased in the untreated control [
59]. Only six weed species (
Avena fatua,
Fumaria officinalis,
Lolium rigidum,
Papaver rhoeas,
Raphanus raphanistrum, and
Sinapis arvensis), out of all observed species, were dominant being recorded every year in every site. Among these, only two grass weeds were highly abundant (
Avena fatua and
Lolium rigidum), and the relative abundance of these two species was higher in underdosed treatments. This last result is also supported by other studies that found high grass weed count in low-input management systems and high broad-leaf count in conventional managed system [
58]. Specifically, with respect to other studies focused on oilseed rape weed flora (even if in a different environment), some species resulted common for oilseed rape independently from location, underlining that those weeds are oilseed rape crop-specific [
37,
39]. The latter include species such as
Galium aparine;
Convolvolus arvensis;
Papaver rhoeas;
Senecio vulgaris; Stellaria media;
Capsella bursa-pastoris;
Fumaria officinalis;
Raphanus raphanistrum, and species belonging to genus
Chenopodium,
Polygonum, Geranium,
Veronica,
Sonchus, and
Vicia. By contrast, some other species are not typical of oilseed rape crop weed flora community, but in any case some of them (e.g.,
Anagallis arvensis,
Chrisanthemum coronarium,
Medicago sp. pl.,
Ranunculus sp. pl.,
Plantago sp.pl.,
Sylibum marianum,
Trifolium sp. pl.) are included as weed common species of arable fields in the Mediterranean region [
60,
61,
62]. In particular,
Chrysanthemum coronarium and
Sylibum marianum are considered common weeds species of cereal crops of the Mediterranean basin [
63,
64,
65,
66], thus, suggesting the importance of previous crop and rotation in developing the weed flora community of a species [
67]. This assertion is also supported by other studies who stated that crop rotation influences weed species [
68] as well as weed seed banks [
69]. Moreover, Koocheki et al. [
70] pointed out that different rotations that include crops with different life cycles such as winter wheat-maize and winter wheat-sugar beet could lead to additional benefits in reducing the weed seed bank and the incidence of perennial grasses and broadleaf species.
Less common species were
Beta vulgaris,
Calendula officinalis,
Ecballium elaterium, and
Plantago lanceolata. Environmental conditions greatly influenced the occurrence of some weeds. The observed differences in species frequency and diversity of weed flora assemblage among years might be attributed to the impact of environmental factors such as rainfall. Some authors [
71,
72], indeed, reported that in addition to nutrients (N), weeds growth cycle is affected by water availability. In our findings,
Ranunculus repens was observed only during the last growing season (2010) and in both sites. The spring of the last growing season was characterized by a cumulated rainfall higher than the long-term series typical for the region (
Tables S1 and S2), and this fostered the occurrence of
R. repens which is a weed species mostly found during wet spring [
73] and in low-input herbicide management system [
74]. Other authors stated that at least 10 mm of rainfall is required for the emergence of all the species, with significantly higher germination rates at rainfall amounts of >20 mm [
75] and with cumulated rainfall over consecutive days, rather than single rainfall events of the same amount. The length of time that the soil surface remained above the permanent wilting point, allowing the imbibition of water, was also an important factor [
75]. Furthermore, in our study
C. album was observed at the Ottava site in each growing season, whereas, at the Ussana site, it failed to be found in 2009 and in 2010. Grundy et al. [
76] found a relationship between the winter mean temperatures and the relative intensity of
C. album infestation observed across various environments. The latter ascribed the flush of emergence of
C. album to the cumulated time spent below some critical temperature greater than 0 °C since a deeper winter chilling may have a greater dormancy breaking effect than a relatively mild winter chill. This last explanation is consistent with the results achieved in our study and with the climatic conditions of the sites under study. Indeed, Ottava site, during the period under study, was characterized by a minimum temperature at least 2–3 °C lower than Ussana site during winter season (
Tables S1 and S2). The decline in weed flora composition and diversity caused by herbicide treatments is confirmed by different studies. Edesi et al. [
77] reported a downward trend of Shannon’s index due to herbicide use. Other previous studies [
74,
78] supported our findings reporting the highest species diversity in low-input system and at untreated plots level. It is noteworthy to mention that different diversity patterns concerning treatments among sites suggests that different agronomic practices and environmental factors may interact in a complex way with treatments and affect the weed diversity within communities [
79]. According with findings of previous studies [
80,
81,
82], it seems that weed communities differed first among sites, while weed shifts within each site is mainly associated with growing season and herbicide treatments.
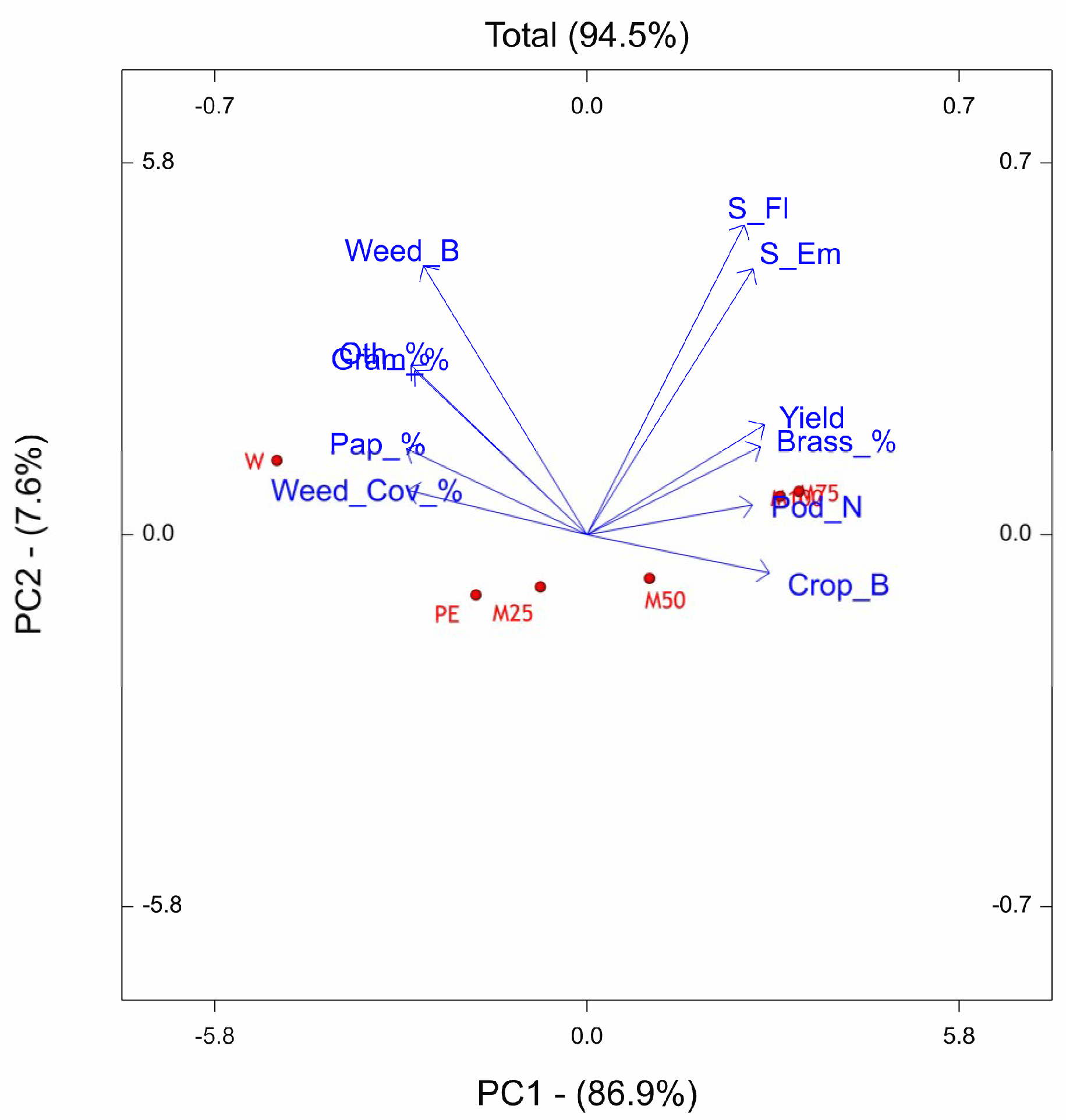
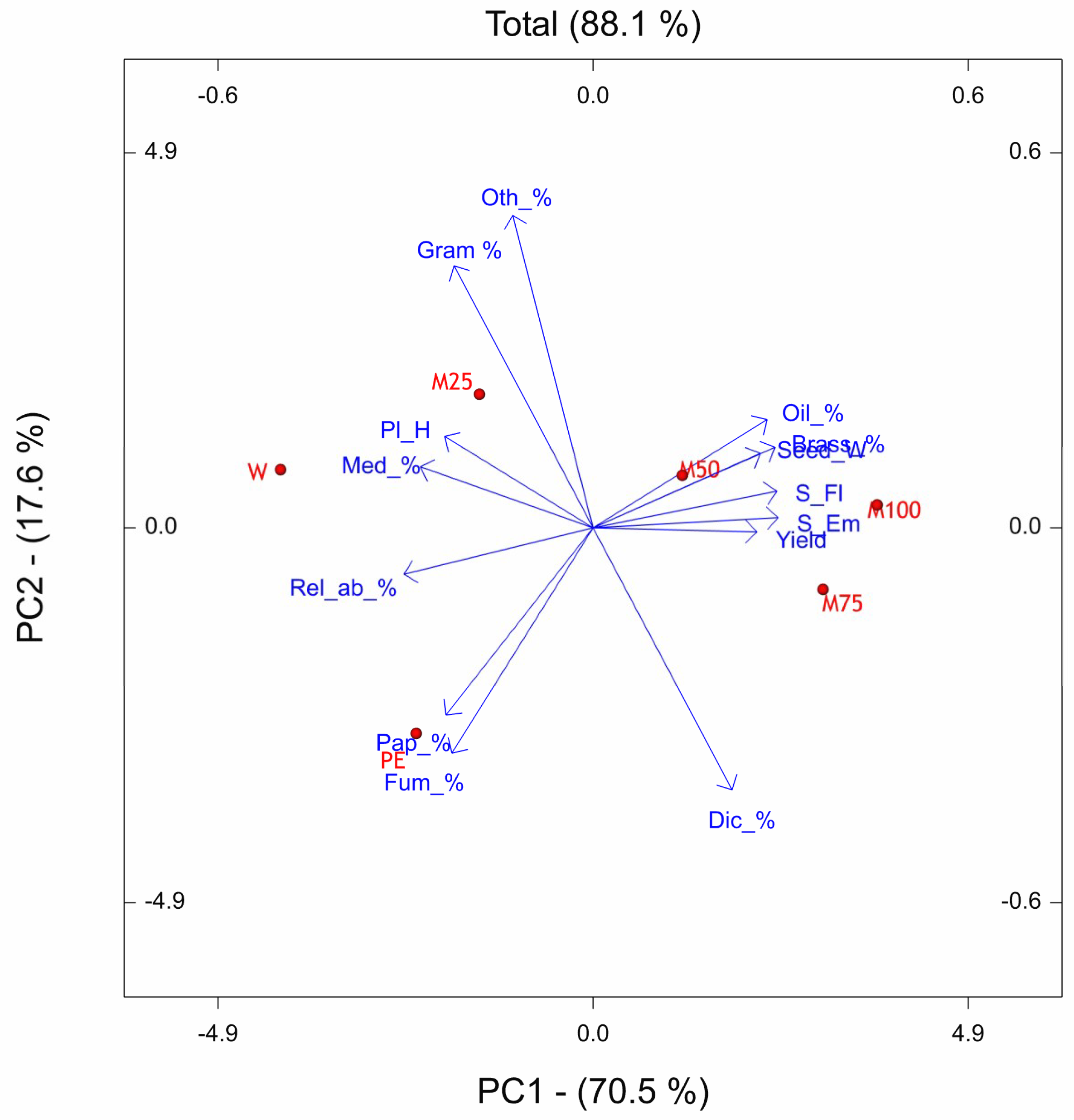
The PCs of OTV site reveals that the underdosed treatments PE, M25, and M50 resulted shifted downward with respect to W, M100, and M75. The weed flora related traits were mainly associated with W treatment, while, the crop related traits were associated to M75 and M100. The period from sowing to emergence and sowing to flowering was most sensitive to M75 and M100, indeed the number of days to emerge and to flower resulted higher with respect to the other treatments. We observed the same finding also in the site of Ussana, where higher doses of pre-emergence treatments (M100, M75, M50) caused phytotoxic effects on oilseed rape by lengthening the phase between sowing and emergence and sowing-flowering and reducing crop growth. The phytoxicity of metazachlor on the oilseed rape crop when applied at labelled dose was also reported by Vercampt et al. [
83,
84].
As a result of application of labelled dose, the metazachlor sensitive species were strongly restrained and thus the community consists of fewer species mainly belonging to Brassicaceae botanical family. If M100, M75, M50 treatments are effective for the general control of weeds, these favor, in proportion, the presence of weeds belonging to the Brassicaceae family (Brassicaceae % vector with smaller angle with M100, M75, M50 vectors). This effect is due to the fact that with metazachlor, which is effective both with monocotyledons and dicotyledons, results are less effective with the physiologically closer weeds such as those of the Brassicaceae family. Some of these findings are consistent with results reported by other authors who claimed that in oilseed rape intensively cropped, a shift in weed vegetation occurred by favoring the Brassicaceae weed species [
39] and suggesting that herbicide pressure may lead to a genetically closer community structure [
85]. This behaviour at community level might arise from the phenomenon known as ‘crop mimicry’ [
86], in which each individual of a population is selected on the basis of its morphological or bio-chemical similarity to the crop enabling them to escape some selection pressures. An increase in the abundance of tolerant and perennial species is not desirable because they are more harmful than annuals. Therefore, weed species diversity should be sought, as the interspecific competition of weeds prohibits one species from becoming a dominant, ‘problem’ weed. Finally, that time of herbicide application (pre- vs. post-emergence application) influenced weed flora assemblage as observed in previous studies, even though they are not considered as the main cause of weed community variation [
87].