Abstract
Every year the oil refining industry consumes thousand tons of fluid catalytic cracking zeolite from the E-cat generated in the fluid catalytic cracking (FCC) unit. In the present paper, a new process for recycling of fluid catalytic cracking catalysts (FCCCs) is presented. The process, previously tested at laboratory scale, was simulated by SuperPro Designer catalysts (FCCCs, also known as equilibrium catalysts, E-cat), which are mainly landfilled. Their intrinsic value is quite low and the content of rare earth elements (REEs), as lanthanum and cerium oxides, is around 3%wt. Moreover, their reuse in other industrial processes as raw material is very scarce. For each metric ton of spent FCCC treated for recovery of REEs, nearly the same amount of waste is generated from the process, the majority of which is represented by the solid residue resulting from the leaching stage. The manuscript presents a technological study and an economic analysis for the recovery of REEs, as well as the production of synthetic © software package. The plant was designed for a capacity of 4000 metric tons per year. The discounted cash flow (DCF) method was applied and Net Present Value (NPV) equal to about two-million € and Discounted Payback Time (DPBT) equal to two years defined the profitability of the process for recycling of FCCCs. This result depends on the selling price of zeolite. Consequently, a break-even point (BEP) analysis was conducted on this critical variable and the condition of economic feasibility was verified with a price of 1070 €/ton. This study tried to implement recycling strategies towards circular economy models.
1. Introduction
In the last half century, the use of catalysts for refinery processes, such as catalytic reforming (CR), fluid catalytic cracking (FCC), hydrodesulphurization (HDS), alkylation, isomerization, and others increased enormously [1]. Once exhaust, some of them can be regenerated by thermal treatments, but after a certain number of regenerations they need to be replaced as their catalytic activity becomes too low. Other catalysts, such as FCCCs, are poisoned by heavy metals and they cannot be regenerated; periodically, they are replaced with fresh catalyst [2]. Rare earths (REs) are used in the structure of FCC catalysts in order to improve the activity and obtain hydrothermally stable products with enhanced yield [3]. Rare earth oxides (REOs) are able to enhance the catalytic activity and prevent loss of acid sites. As the need for increased amounts of gasoline grew over time, refiners tended to increase the level of rare earths in their catalyst formulation to meet their profitability targets [4]. REOs concentration gradually increased over the years, and their average is nowadays between 1 and 3%wt, depending on the producer and the feedstock to treat. REs extraction and refining from primary ores are concentrated in few countries; the majority of mines are located in China [5,6], hence the recovery of such elements from industrial waste will be crucial for countries, like the European ones, which do not own primary mines [4]. It is difficult to quantify the total amount of FCCCs produced every year in the world, as well as their price. The main manufacturers (Grace Davison, Columbia (MD), USA; Johnson Matthey, London, UK; Albemarle, Charlotte (NC), USA; Basf, Ludwigshafen am Rhein, Germany; Sinopec, Beijing, China) do not release this data freely and some available reports are very expensive [7]. Moreover, the amount of spent FCCCs disposed of annually can be confidential or even unknown. Nevertheless, some reliable estimations were done in recent years and a production of 840,000 ton per year was estimated [8,9].
There are several groups that focused their researches on direct reuse of spent E-cat, for instance, as additive for partial replacement of sand in cement or mortar powder mixtures [10,11,12,13]. Nevertheless, the amount of FCCC powder than can be used does not exceed 20%wt, in order not to negatively affect the chemical and mechanical properties of cements and mortars. Thus, nowadays, the fraction of spent E-cat reused is still low if compared to the huge amount disposed of annually. Nevertheless, no updated global official data currently exists about the recycling rate of E-cat as cement and mortar additive, refractory material, production of mineral wool, and ladle covering powder in steelmaking industry.
Regarding regeneration and rejuvenation of FCCC, additional research is required to find reliable methods, since no effective techniques are currently available in the industrial scale. Direct reuse as catalyst in other industrial processes has very limited applications, and in lab- or pilot-scale only; for instance, spent E-cat was used as catalysts for plastic and biomass pyrolysis/gasification or for the production of synthetic fuels [14,15]. Nonetheless, such processes were not widely developed in industrial scale. Disposal in landfill or the use for concrete blend are still the main choices for handling of spent catalysts, despite the content of rare earths elements and sometimes other minor metals in terms of concentration, like vanadium and nickel, which could be recovered as well [7].
There are several works that studied the extraction of the two rare earth elements (REEs) contained in FCCCs or waste slag from their production, i.e., lanthanum (La) and cerium (Ce), although no industrial processes were developed in industrial scale. This is caused by the unprofitability of the recovery of La and Ce. Some authors recovered La and Ce from FCC waste slag by leaching with HCl and selective precipitation of the REs as NaRE(SO4)2·xH2O [16,17]. Furthermore, aluminum is converted into soluble NaAlO2 that can be used as secondary raw material. Besides selective precipitation [18], the most used technique for extraction and the further refining of REs from leach liquors is solvent extraction [18,19,20]. Ion-exchange is another hydrometallurgical technique that was used to extract REEs from leach liquors [21,22,23,24]. Bioleaching was also investigated to obtain an environmentally friendly process [25].
Some works were focused on synthesis of zeolites from spent FCCC and the recovery of La and Ce was not considered [26,27,28]. Thermal treatment can be carried out by fusion with NaOH or NaHCO3. For instance, some authors tested alkaline fusion with several FCCC to Na2CO3 ratios at 800 °C for 2 h, followed by hydrothermal crystallization with 4 mol/L NaOH solution at 80 °C and dissolved NaAlO2 [27].
REEs are included in the list of 27 critical materials released by the European Union (EU) in 2017 and their recycling and reuse is a fundamental step for the sustainable development in the next years [29]. As a last option, since E-cat is sometimes classified as hazardous waste, a thermal treatment at high temperature with a plasma arc furnace is able to convert spent E-cat into an inert waste. The vitrification that occurs in the temperature range of 1400–1600 °C avoids any possible release of metals caused by rainfall water leaching when such waste is put in landfill. Nevertheless, this high temperature process determines an increase of the disposal cost. Such treatment was tested, for instance, by Tetronics (Swindon, UK).
The present paper is focused on the profitability analysis for a new recycling process developed at the laboratories of Chemical Engineering of the University of L’Aquila. This process extracts Ce and La and reuses the leaching residue for the production of a synthetic Na-A zeolite (also known as LTA, Linde Type A). Such zeolite is widely used in several industrial and environmental processes, like the adsorption of metals and organics from wastewater and spent solutions, ion-exchange, catalysis, formulation of phosphorus free detergents as water softener and for alkane/alkene separation [30,31]. According to the Ellen MacArthur Foundation, a circular economy is an economic system where products and services are traded in closed loops, which is regenerative by design, with the aim to retain as much value as possible of products and materials. Hence, the aim is to create a system that allows for the long life, optimal reuse, refurbishment, remanufacturing, and recycling of products and materials. This is the aim of the process presented in this paper, that was applied to a plant with a realistic capacity of 4000 metric tons per year; the total amount of E-cat annually stockpiled or disposed of in Italy is indeed in the range 10,000–12,000 tons.
The methodology used is based on DCF and multiple indexes, such as NPV, DPBT, and Internal Rate of Return (IRR) are proposed. At the same time, other indexes, such as Gross Margin Ratio (GMR) and Return on Investment (ROI), are useful to describe the economic performance of the project according to the metric used in the balance sheet. The analysis is conducted in a baseline scenario and varying the critical variables (selling price of zeolite, cost of sodium hydroxide, cost of sulfuric acid, treatment/disposal cost for solid waste, treatment/disposal of aqueous liquid, and cost opportunity of capital) alternative scenarios are investigated.
2. Materials and Methods
The first sub-section presents the entire recycling process as simulated by the software. The following Section 2.2 lists the different techniques that are used for the characterization of solid samples and liquid solutions. Section 2.3 and Section 2.4 explain how the leaching tests were carried out and how the two REEs were recovered from the leach liquor. The detailed experimental procedure for the synthesis of zeolite is shown in Section 2.5. Finally, Section 2.6 presents the economic model developed, and all input data and assumptions are described in Section 2.7.
2.1. The Description of the Process
Spent E-cat does not require any mechanical treatment like grinding or even roasting, as it is already composed of fine oxidized particles (D50 = 80 µm). Hence, the process is characterized by a direct leaching of the catalyst in sulfuric acid solution. After that, the unreacted material is filtered by a plate and frame (P&F) filter. The solid residue undergoes synthesis of zeolite, whereas the pregnant solution is heated and stirred for 1 h, while 50%wt NaOH solution is slowly added to increase the pH at around 2 and cause the precipitation of RE double sulphate salts. Such salt is thus filtered by a P&F filter or a two-phase centrifuge, washed, and dried for final storage in bags. The spent solution, still containing an interesting amount of sulfuric acid, undergoes nanofiltration (NF) in a membrane module in order to recover around 85% of the solution in the permeate with approximately the same molarity of the feed solution; salts and other rejected compounds are recovered in the retentate stream, a concentrated solution that is routed to the wastewater treatment. This recovery stage was already tested by our research group for spent sulfuric acid solutions coming from the production of optical quartz: in this way, the operating costs are strongly reduced as a small make-up of fresh acid is required only.
The synthesis of the zeolite is carried out according to the procedure that is explained in Section 2.4. Sodium carbonate is thoroughly mixed with the solid residue that acts as base silicoaluminate. The mixture is put into a rotary kiln for roasting, and after cooling the hydrothermal treatment with NaOH solution takes place. Afterwards, the solution is filtered by a two-phase decanter and the solution, together with the water used for zeolite washing, is routed to the wastewater treatment.
The zeolite is dried in a rotary or belt kiln, thus cooled down, mildly milled, and stored in a dried environment prior to final packaging. The flow-sheet of the process is shown in Figure 1. Nearly 85% of the total water consumed is recycled back to the process after its treatment. This is very important, since the process is water-intensive, as several washings are required (leaching cake and zeolite), thus the recycling of water is necessary in the perspective of a circular economy approach. After the first treatment, part of the water has to be discharged into the industrial sewage network to avoid the overconcentration of salts and any other compound in the recycled water.
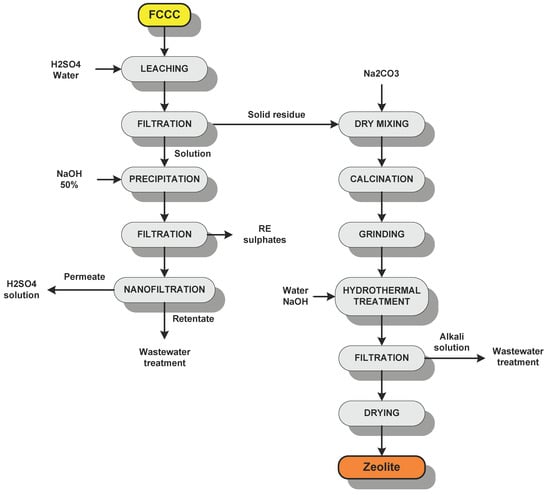
Figure 1.
Flow-sheet of the recycling process.
2.2. Characterization of the Samples
The characterization of the spent FCCC sample, products, and solutions was carried out at the laboratories of Chemical Engineering at the University of L’Aquila, Italy. The spent E-cat catalyst sample was provided by Orim SpA, Macerata, Italy. The catalysts and the zeolites synthesized during the experimental campaign were characterized by X-ray fluorescence (XRF) (Spectro, Xepos, Kleve, Germany) and Inductively Coupled Plasma-Optical Emission Spectrometry (ICP-OES, 5110 Agilent Technologies, Santa Clara (CA), USA), X-ray diffraction (XRD, PANalytical X’Pert PRO, Malvern PANalytical, Malvern, UK), particle size distribution (PSD, Mastersizer 2000, Malvern PANalytical, Malvern, UK), Brunauer-Emmet-Teller (BET) specific surface area (SSA, NOVA 1200e, Quantachrome porosimeter, Boynton Beach (FL), USA), and scanning electron microscopy (SEM, 20 kV Philips XL30 CP microscope, Amsterdam, The Netherlands).
2.3. Leaching Stage
Several leaching tests were carried out with different acids [32]. Sulfuric acid was selected as the most promising leachant. The leaching stage was carried out in a 2 L glass jacketed stirred reactor equipped with a mechanical stirrer, with 1 L of 1.5 mol/L H2SO4 solution, and solid to liquid ratio (S/L) equal to 200 g/L at 80 °C. The reaction time was set at 3 h. These optimized conditions are based on the results that were obtained in a previous research [18]. After the reaction time, the remaining E-cat was filtered by a 142 mm air pressure filter (Millipore, cut off 0.45 µm, Burlington (MA), USA) in order to separate the solid from the leach liquor. The solid was washed by distilled water and dried at 105 °C for 24 h. The leaching trials were repeated twice, in the same conditions, to have an estimation of the reproducibility of the results. The concentration of Ce, La, Al, and Si in the leach liquors was measured by ICP-OES. Al and Si were determined as are the most concentrated elements that can affect the final grade of the REs precipitate. The extraction yield was calculated, as indicated in [33].
2.4. Recovery of REEs
La and Ce were recovered from the leach liquor by a direct precipitation with 50%wt NaOH solution. A soon as the pH reached 1.5, a white precipitate appeared in the flask. The precipitation was carried out in a beaker on a heated plate, under mild magnetic stirring at 50 °C. Further NaOH solution was added to achieve a pH equal to 2, thus the reaction was stopped after 1 h. The solution was filtered by the pressure filter and the precipitate washed with distilled water and then dried in oven at 105 °C for 24 h. The RE precipitate was characterized by ICP and XRD analyses.
2.5. Synthesis and Characterization of Zeolite
The solid residue from the leaching stage was used as raw silicoaluminate for the production of synthetic zeolite. Unlike the hydrothermal synthesis that is described in [32], the roasting stage was carried out with Na2CO3, which is a cheaper reagent. The E-cat residue from the leaching stage was mixed with Na2CO3 in a 50%wt ratio and roasted at 600 °C for 2 h in a muffle oven (MF 100, Galli). Hence, once cooled down, the material was ground by a ball mill (PM 100, Retsch). The hydrothermal treatment was conducted in a 500 mL flask at 80 °C under stirring in a water bath (Dubnoff, ISCO) and S/L ratio of 15% wt/vol for 24 h. A concentration of NaOH in the range 1–1.5 mol/L was used.
After that, the material was filtered by pressure filter and washed with distilled water until the wash water reached a pH value between 7 and 8. The synthetic zeolite, hence, was dried at 105 °C for 24 h, ground manually, and stored in a closed flask in order to avoid moistening. The characterization of physico-chemical properties was done by BET, XRD, and XRF analyses.
2.6. The Description of Economic Model
DCF is a well-known economic assessment method estimating the attractiveness of a project. It tries to work out the value of an asset today, based on projections of all the cash flows that it could make available to investors in the future. Several economic indexes can be chosen to assess the profitability [34]:
- NPV is defined as the sum of present values of individual cash flows.
- DPBT represents the number of years needed to balance cumulative discounted cash flows and initial investment.
- IRR identifies the discount rate at which the present value of all future cash flows will balance the initial investment.in which It = discounted cash inflows; Ot = discounted cash outflows; r = cost opportunity of capital, t = time period; and, n = lifetime of investment.
Other useful indicators are typically used in the balance sheet. For this purpose, their use characterizes all firms. A combination with previous indexes can be useful, according to the existing literature [35,36]:
- GMR is calculated by dividing the gross profit by sales.
- ROI measures the amount of return on a particular investment, relevant to the investment’s cost.
2.7. Input Data
The simulation of the entire process was carried out by the software package SuperPro Designer©, Intelligen Inc. (Cambridge (MA), USA). The process is divided into two main sections: the first hydrometallurgical section in which La and Ce are extracted and recovered and the second one in which the synthesis of the zeolite is performed. There is an additional section in which all spent solutions and wastewaters are treated.
Spent FCC catalyst is usually classified as non-hazardous waste, but some samples are classified as hazardous, because of the content and the particular speciation of metals, like nickel and vanadium; for instance, V5+ (V2O5) is considered to be very toxic to living organisms [7]. Nevertheless, the concentration of such two metals is usually quite low (0.5–1.5% wt as oxides), depending on quality of the feedstock and the operating time before the unloading of the column’s bed. After the leaching stage, the solid residue is very stable as all the metals are mostly leached by a strong acid solution, so the toxicity is eliminated. The toxicity characteristic leaching procedure (TCLP) test demonstrated that such material is inert (results are not reported here). The testing methodology is used to determine whether a waste has to be classified as hazardous, i.e. belongs to list according to the United States (U.S.) Environmental Protection Agency (EPA).
Spent FCCC is usually collected by specialized companies that dispose it of responsibly in appropriate landfills, and for such service refineries pay a fee that in Europe varies from 200 €/ton to 300 €/ton. This amount represents additional revenue for the recycling process. The plant works in a continuous operating mode for a total of 7920 h/year (330 working days), considering the normal maintenance periods. The total capacity of the plant is 4000 tons/year, with an hourly flow-rate of around 505 kg/h. This capacity was chosen as case study for a country like Italy, where the annual amount of spent E-cat is estimated to be between 10,000 and 12,000 tons. The project lifetime is set at 15 years, with a depreciation period of 10 years.
The raw materials that were used in the process are concentrated sulfuric acid 96%wt solution, water, sodium hydroxide, and sodium carbonate. Utilities include electrical energy and natural gas, burnt in a boiler to heat the jacketed stirred reactors. Natural gas is also used to heat the kiln up in order to roast the mixture FCCC-Na2CO3, as well as to dry the final zeolite before packaging and storage.
The unit revenues of the plant and the cost of raw materials and utilities that are used in the simulation are indicated in Table 1. Table 2 presents the list of the main equipment required to develop the plant. The total purchase cost is 2450 k€; there are additional devices and machinery not included in the list, such as conveyor belts, hoppers, pumps, compressor, weighting system, and other minor auxiliary facilities, but whose purchasing cost was taken into account. The total plant direct cost amounts to 3370 k€: the latter includes the equipment purchase cost, installation, piping, instrumentation and control system, insulation, electrical, yard improvement, and buildings. Engineering and construction costs plus contingency give the final direct fixed capital cost that amounts to 3710 k€.

Table 1.
Economic unit input.

Table 2.
List of the main equipment.
Operative costs regard several items as raw materials (Ca hydroxide, Na2CO3, NaOH, Sodium Hydroxide, Sulfuric Acid, Water), labor-dependent (nine workers distributed on three shifts), facility-dependent (the staff is completed by an office worker and the technical director of the plant), laboratory, consumables, waste treatment/disposal (solid waste, aqueous liquid), utilities (natural gas, electric energy and compressed air), transportation, miscellaneous, running royalties, and failed product disposal.
Concentrated sulfuric acid is one of the greatest costs among raw materials; for this reason, the spent acid from the NF membrane module is recycled back and, taking into account such saved amount, the equivalent cost of the fresh acid would be 100 €/ton instead of the market price of 120 €/ton. The cost of sodium hydroxide is equal to 300 €/ton; in this process, such chemical is used as aqueous solution in the REEs precipitation stage and in the hydrothermal treatment of the leaching residue. The solutions are freshly prepared with water in two stirred tanks. Consequently, the price per ton remains fixed but the amount of NaOH that is requested by the processes halved. According to this assumption, it is possible to declare that the cost of NaOH 50% sol. is equal to 150 €/ton.
Finally, three items characterize the revenues: (i) E-cat collection (disposal fee), (ii) selling of RE oxides, and (iii) selling of zeolite. The first item is derived from the treatment of a product that is potentially dangerous and it is paid by oil industry. The other two items are linked to the materials recovered by the process.
3. Results
In this section, a quick overview on the main results achieved during the experimental campaign is given. After the results from the characterization of the spent FCCC (Section 3.1), the extraction yields for La, Ce, Al, and Si are reported in Section 3.2, whereas in Section 3.3, the zeolite obtained from the synthesis procedure is fully characterized. Several economic indexes (NPV, DPBT, IRR, GMR, and ROI) are proposed in Section 3.4 regarding the baseline scenario. In addition, Section 3.5 investigates the variation of these indexes in function of critical variables.
3.1. Characterization of Spent FCCC
The concentration of the main elements is shown in Table 3. It underlines that the total concentration of REs is rather low.

Table 3.
X-ray fluorescence (XRF) and inductively coupled plasma (ICP) analyses of the spent E-cat.
The XRD analysis showed that the most probable compounds are dealuminated zeolite Na-Y, that is the main crystalline phase, followed by other minor phases, like zeolite ZSM-5 and alumina. Regarding the BET analysis, the SSA was around 112 m2/g, a bit lower than the area of a fresh FCCC, which is usually in the range 120–180 m2/g. The PSD was a Gaussian curve centered at 80.4 µm, with a Sauter’s diameter D [2,3] equal to 73.6 µm.
3.2. Extraction and Recovery of REEs
The extraction yields of the main elements as a function of reaction time are listed in Table 4; such yields are reported as medium values and standard deviation.

Table 4.
Extraction yield for the main elements (ICP-OES).
Hence, the best extraction yields for La and Ce were obtained after 3 h. The extraction of Si was lower than 2%, but such element is present as colloidal silica that, once the solution cools down, makes the filtration difficult. For this reason, the following filtration and precipitation shall be carried out with warm solution. The extraction of Al was low as well, but the concentration in leach liquor was not negligible. The initial pH of solution was 0.45 and NaOH was added to increase the pH to 2. The precipitation occurred slowly as soon as the NaOH solution was added. The precipitation of insoluble NaRE(SO4)3·H2O is expressed, as follows [37]:
La2(SO4)3 + 2 Na+ + SO42- + 2 H2O → 2 NaLa(SO4)2·H2O
Ce2(SO4)3 + 2 Na+ + SO42- +2 H2O → 2 NaCe(SO4)2·H2O
This precipitation mechanism as double sulfate salt is also caused by the addition of Na2SO4 instead of NaOH [17]. The solubility of NaRE(SO4)2·H2O in water (in particular, for Ce, La, Pr, Nd, Pm, Sm, Eu, Sc) is very low and it decreases with the increase of temperature [18]. After cooling down, the white salt was filtered, washed with distilled water and dried at 105 °C for 24 h, and thus used for the analysis. The results of ICP-OES showed the following element concentration: La 36.54%, Ce 2.87%, Na 2.88%, Al 6.14%, and Si 0.7% by weight. The rest is mainly water, oxygen, and sulphur.
The XRD analysis indicated that the RE precipitate was mainly composed of NaLa(SO4)2·xH2O and NaCe(SO4)2·xH2O, but Al2SO4 and small amount of Al(OH)3 cannot be excluded. The XRD spectrum of the RE salt was indeed not crystalline and characterized by large and not well-defined peaks. This RE concentrate shall be leached again and La be separated from Ce by solvent extraction. Hence, the precipitated salt can be sold to companies with expertise in separation and refining of REEs like Rhodia (France), Umicore (Belgium) or Solvay (Belgium).
3.3. Characterization of the Synthesized Zeolite
After the synthesis procedure that is described in Section 2.5, the zeolite from the acid leaching was fully characterized. In particular, the SSA was 83.2 m2/g, whereas the BJH cumulative average pore volume was 0.062 cm3/g with an average pore size of 3 nm. Spent E-cat and synthetic zeolite obtained from the hydrothermal procedure are shown in Figure 2. The XRD spectrum of the latter is shown in Figure 3, whereas Figure 4 proposes a SEM picture of the same zeolite. The zeolite is light brown in color, whereas the spent E-cat is grey.

Figure 2.
Spent fluid catalytic cracking (FCC) catalyst (left) and synthetic zeolite (right).
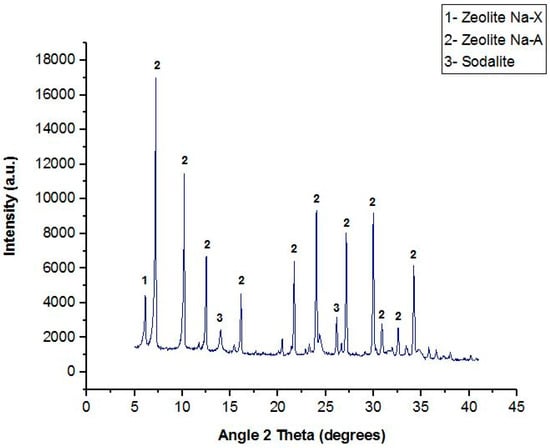
Figure 3.
X-ray diffraction (XRD) spectrum of the synthetic zeolite.

Figure 4.
Scanning electron microscopy (SEM) picture of the synthetic zeolite.
The main phase detected in the zeolite was Na-A, with formula Na12Al12Si12O48·27H2O. Small percentages of dehydrated zeolite Na-X (Na92Al92Si100O384) and Cl-free sodalite (Na8Al6Si6O24), another aluminosilicate mineral, were also present: this means that the conversion to phase-A was not complete. The zeolite Na-A presents a cubic crystalline system with the lattice parameters a = b = c = 24.61 Å and α = β = γ = 90°. The presence of high crystallinity was demonstrated by the high intensity of the peaks. Na-A is the major constituent phase (around 91%wt) and spheroidal aggregates of remaining sodalite and Na-X phase. Table 5 shows the composition of the main elements of the zeolite that was obtained from the residue of the sulfuric acid leaching.

Table 5.
Element concentration of zeolite from H2SO4 leaching measured by XRF.
Although this analysis is not as accurate as ICP, especially for light elements, like Na and Al, it gives an indication about the Si/Al ratio: hence, from the XRF analysis, it can be inferred that this is a low silica synthetic zeolite.
3.4. Economic Evaluation Report—Baseline Scenario
The description of the technical aspects was already elaborated and in this phase of the work, the economic analysis is developed. There is a clear relationship between two perspectives. The simulation software quantifies the several amounts of resources that were employed in the process and some economic results (GMR, ROI) are obtained directly by this software, while other results (NPV, DPBT, IRR) are elaborated through Excel programme—Table 6.

Table 6.
Economic executive summary (baseline scenario).
The project is profitable in the baseline scenario and the results contribute to developing models of circular economy. In fact, the evaluation of this typology of investment firstly considers the environmental advantages derived from the recovery of these wastes and at the same time the achievement of environmental goals set by the European Union (COM (2017) 490 final). The following step is represented by the economic study. NPV quantifies the amount of potential profits when considering the lifetime of the project. The value obtained is extremely relevant: it is equal to 2170 k€. NPV is a realistic metric for decision makers because the DCF method is based on two relevant aspects: (i) the time value of the money and (ii) the lifetime of the project.
The distribution of cash flows permits to understand this result. From the revenues side, the recovery of zeolite plays a key role. Its weight is equal to about three quarters (77%). The price of RE oxides is greater than that of zeolite, but the amount of materials recovered in this waste is extremely lower (about 5%). It is opportune to underline as the REO concentrate represents the lowest value among the three revenues of the plant. This confirms that the recovery of REEs from spent E-cat is unprofitable, even if they would be separated and recovered with 99.9% grade. The use of raw materials (29%) and the phases linked to the waste treatment and/or disposal (26%) are the main items of costs. Instead, the share of investment costs is not relevant (8%). The main item of the materials cost is due to NaOH, used in solution for precipitation of La and Ce, as well as for hydrothermal treatment, with nearly 837 k€/year. The use of Na2CO3 (around 277 k€/year) in the roasting stage results in significant savings with respect to the use of sodium hydroxide. Despite the recycling of sulfuric acid solution, the cost for such reagent is equal to 506 k€/year (cost of fresh acid only). Industrial water is another raw material, but around 85% is reused after the wastewater treatment. The disposal cost of solid and liquid waste is almost high, but the process produces a great amount of sludge and waste solution containing metals. This cost is equal to 644 k€ and 851 k€, respectively—Figure 5.
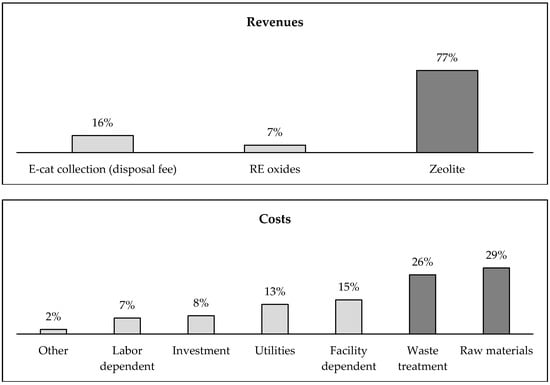
Figure 5.
The distribution of discounted cash flows (baseline scenario).
DPBT is coherent with the result of NPV. A cut-off period is not fixed in this work but certainly the result is positive, since those cash flows allow the re-entering from the investment already at the end of the second year. This depends on three elements: (i) the positive value of gross profit, (ii) the application of third-party funds that distribute the investment cost over the years of loan, and (iii) the share of investment costs is lower than the operative ones.
Finally, also IRR defines the profitability of this project. A possible concern for users of the IRR method is when the discount rate of a project is unknown, but in this case, it is significantly greater than the cost opportunity of capital. The analysis of cash flows underlines the presence of a single change of sign (it is negative during the year zero and it is positive in the following years due to the gross profit). For this reason, the possible issue of multiple values of IRR is not verified.
NPV, DPBT, and IRR measure the profitability of a project, while other two indexes (GMR, ROI) are referred only to a single year of activity. It is linear with the role of balance sheet that characterizes the world of firms. Useful indications are provided, but these indexes are more appropriate for other scopes as: (i) a comparison with the value of benchmarking of the sector; (ii) a comparison with other firms employed in the same sector; (iii) a monitoring to measure the performance of a company over time; and, (iv) for the purposes of the regulatory rules.
Gross profit is the difference between the annual revenues and the annual operating costs. Gross margin is the direct percentage of profit in the sale price. Its positive value is determined by the revenues that are greater than the operative costs (6305 k€ against 5306 €). When this value becomes negative, not only it does not produce wealth, but it even generates economic losses.
Net Profit also considers the profit tax and the depreciation in comparison to the gross profit. ROI determines how a purchase is profitable. Its positive value is determined by the positive value of net profit. ROI and GMR are two metrics that give different information. For example, if money is invested in a project, ROI measures the result of the added investment. Instead, GMR indicates whether revenues are going up faster than costs.
3.5. Economic Analysis—Alternative Scenarios
The aim of this sub-section is to give solidity to economic results obtained in the baseline scenario. In fact, they are based on the assumption of a set of input parameters. Hence, a variance of the expected results could occur. This issue can be overcome by implementing a sensitivity analysis on the critical variables [38].
The choice of critical variables is linked to the results proposed in Figure 5 regarding the main items that characterize the cash flows. A pessimistic and an optimistic scenario are evaluated for each critical variable and a variation of ± 20% is applied.
- The zeolite has a weight equal to four-fifths of discounted cash inflows. Its price of reference is equal to 1150 €/ton (see Table 1). The potential values of 920 €/ton and 1380 €/ton are hypothesized in alternative scenarios.
- Raw materials have a weight equal to one-third of discounted cash outflows. In particular, the net cost of NaOH (50% w/w) covers 35.7% of this cost, followed by the net cost of sulfuric acid with the 30%. NaOH 50% sol. has a baseline value of 150 €/ton and alternative values of 120 €/ton and 180 €/ton are proposed. Instead, 96%wt H2SO4 when considering its recycling has a value of reference of 100 €/ton and potential alternative values are 80 €/ton and 120 €/ton.
- Waste treatment/disposal have a weight slightly lower than raw materials (−3.5%). In particular, liquid waste disposal covers 56.9% of this cost and the remaining share is linked to the solid one. The difference is not significant and both variables are analysed. Solid waste has a baseline value of 130 €/ton and alternative values of 104 €/ton and 156 €/ton are hypothesized. Regarding liquid waste, instead, 120 €/ton is the baseline value and 96 €/ton and 144 €/ton are the alternative values.
- A macro-variable of all cash flows is represented by the cost opportunity of capital. This variable measures the return coming from an alternative project, which has the same risk level. It is hypothesized to equal 5%. In this case, four scenarios are assessed with values varying from 3% up to 7%.
The presentation of six critical variables has determined the elaboration of fourteen alternative case studies. Several indexes change in function of both variables regarding revenues and costs—Table 7—while cost opportunity of capital does not modify the value of balance sheet (GMR and ROI) and at the same time the value of IRR is constant (baseline value equal to 56.7% is always greater than values examined in this work)—Figure 6.

Table 7.
Economic results (alternative scenarios).
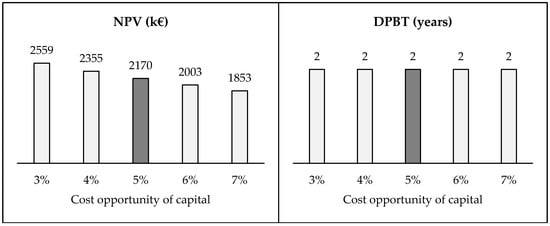
Figure 6.
Sensitivity analysis—Cost opportunity of capital.
Results that were obtained in these alternative case studies confirm the profitability of the project. Economic losses are obtained in only one scenario (price of zeolite equal to 920 €/ton). In this situation, it is opportune to elaborate a BEP analysis. It defines the value of the price of zeolite from which a null value of NPV is obtained—Figure 7.
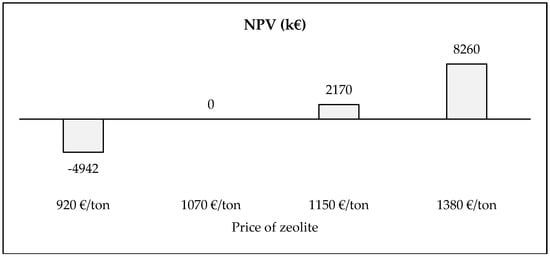
Figure 7.
Break even point analysis—Price of zeolite in presence of disposal fee.
BEP underlines that the profitability of this project is strictly linked to the price of zeolite. In the baseline scenario, a consistent profit is obtained (equal to 2170 k€), but in a pessimistic scenario, a relevant economic loss is reached (equal to −4942 k€). From a mathematical point of view, the price of zeolite that characterizes NPV = 0 is contained in the range from 920 €/ton to 1150 €/ton. The high level of accuracy of this analysis defines the value of 1070 €/ton that is close to the baseline value and it is lower than 7%.
The other critical variables, instead, do not modify the profitability of the project. In fact, NPV presents its lower value (equal to 1100 k€) when the cost of the liquid waste is 144 €/ton. In this same scenario, DPBT is equal to four years that is, however, a reasonable value for investors. It remains fixed equal to two years when the cost opportunity of capital is modified. NPV increased by 18% if the cost opportunity of capital is equal to 3%, while it decreased by 15% when this variable is equal to 7%.
DPBT in other scenarios varies from two years to three years. There is a particular situation, only when the price of zeolite is modified. The investment is recovered already after one year if the price of zeolite is fixed to 1380 €/ton, while the value >15 indicates that during the lifetime of the project, the value of discounted cash inflows never will be match one of discounted cash outflows.
In this scenario, IRR cannot be calculated, since all cash flows are always negative (n.d. acronym stands for “not definable”). In other scenarios, the minimum value is equal to 34.1%, which is significantly greater than the cost opportunity of capital used in this analysis.
GMR proposes a comparison between revenues and operating costs. Such an index is positive, even in the worst scenario (equal to 0.4%). In fact, the value of annual revenues is greater than the operating costs, but, when also considering investment costs, the profitability is not accomplished. This demonstrates as this index has a great limit. The value is significantly low, but it does not give any information on relevant data of a project (as cost opportunity of capital and lifetime of the project). The same is verified also for ROI.
The concept of circular economy requires not only to preserve the environmental value through initiatives of recovery/recycling as those defined in this work, but also at the same time economic opportunities must be developed. The sole recovery of REEs from spent E-cat is absolutely unprofitable, so the production of zeolite plays a key-role. However, also the presence of a disposal fee is required to make recycling of spent E-cat profitable. In this direction, another alternative scenario can be evaluated considering the absence of a disposal fee. A BEP analysis is conducted on the price of zeolite—Figure 8.
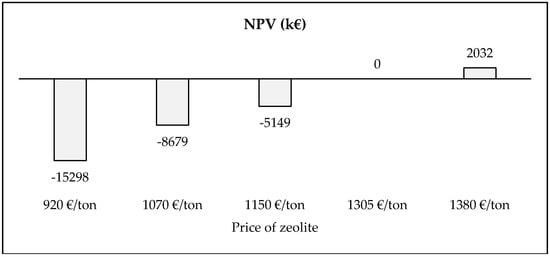
Figure 8.
Break even point analysis—Price of zeolite in absence of disposal fee.
Results highlight that the profitability of baseline scenario with a price of zeolite equal to 1150 €/ton is no longer verified. In fact, NPV is negative and is equal to −5149 k€ (a difference of about 7319 k€—see Figure 7). At the same time, in the optimistic scenario in which the price of zeolite is increased by 20%, NPV is positive (equal to 2032 k€). This economic result is lower than that from the baseline scenario (2170 k€), despite that the price of zeolite is increased of 230 €/ton. Consequently, the zeolite has a key-role in the economic analysis, but in absence of disposal fee for E-cat collection, the profitability is significantly lower. The profitability is achieved with a price of the zeolite equal to 1305 €/ton without the disposal fee. Majority of zeolites are manufactured using alkaline and alkaline earth metals. The most common natural zeolites include analcime, chabazite, clinoptilolite, erionite, ferrierite, heulandite, laumontite, mordenite, and phillipsite. Natural and synthetic zeolites are most commonly used in various end-use industries, owing to various properties, including unique adsorption, molecular sieve, superior catalytic, and ion-exchange properties. Natural zeolites were the largest product segment, accounting for over 60% of the global market volume in 2014. Synthetic zeolites market demand has been tremendously increasing over the past few years. Most common synthetic zeolites include types A, X, Y, and ZMS-5. The segment accounted for nearly 40% in 2014 and share is expected to increase by the end of 2022. Growing utilization of these zeolites in the petroleum industry, owing to its ability to purify gas streams, is expected to be the primary factor for the demand growth over the forecast period. The global zeolite market was valued at around USD 3.50 billion in 2014 and it is expected to reach USD 4.50 billion in 2020 and over 2000 ktons of zeolites produced worldwide, both natural and synthetic [39].
4. Conclusions
In this paper, a novel recycling process was developed for the full recycling of spent E-cat, also known as FCC catalyst. Lanthanum and cerium were recovered by a leaching stage with sulfuric acid solution, thus precipitated as RE sulphates with NaOH. The solid residue from the leaching stage is used for the synthesis of zeolite. The analysis demonstrated that zeolite Na-A is the most concentrated phase, and minor amounts of Na-X and sodalite are also present. Nevertheless, changing the synthesis conditions (amount of Na2CO3, roasting, and hydrothermal stage temperature and time) it could be possible to maximize the Na-A phase. This is the aim of the next experimental campaign. Spent sulfuric acid solution is regenerated by nanofiltration, and also most of the water is recycled back after treatment.
The economic analysis demonstrated that the process is profitable if the solid residue from the leaching stage is used for production of zeolite; moreover, another important revenue of the plant is the disposal fee for the E-cat. The current metal market prices do not make the recovery of La and Ce profitable, as their concentration in spent E-cat is rather low. The baseline scenario is profitable and several alternative case studies present a positive sign of NPV. However, the profitability cannot be verified in two case studies:
- The first concerns the price of zeolite, when such value is lower than 1070 €/ton in presence of the disposal fee for E-cat collection.
- The second regards the price of zeolite when this value is lower than 1305 €/ton in the absence of the disposal fee for E-cat collection.
The process and the relevant plant, thus, demonstrated a reliable and useful management of spent E-cat; although part of such catalyst is already used for the partial replacement of cement and mortar powders, powder in refractory materials and mineral woods, the majority of it is still landfilled. Hence, it is crucial to find the market for such synthetic zeolites, which have many industrial uses and could certainly reduce the consumption of natural zeolites that are mined worldwide and the amount of spent E-cat landfilled every year.
Author Contributions
Conceptualization, F.F.; methodology, S.L. and I.D.M.; software, S.L.; validation, F.V.; investigation, F.F. and V.I.; data curation, V.I.; writing—original draft preparation, F.F. and I.D.; writing—review and editing, I.D.; visualization, I.D.M.; supervision, F.V.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sector.
Acknowledgments
The authors would like to warmly thank Orim S.p.A., whose President Alfredo Mancini, MEng., and R&D Manager Fabio Maggiore, MEng, have supplied the FCCC sample and other precious background information. Moreover, our research group at the University of L’Aquila wants to dedicate such work to Orim’s former CEO Andrea Mancini, who prematurely passed away in 2017. We all remember his efforts and diligence in the recovery and recycling of industrial waste.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Costa, C.; Marques, J. Feasibility of eco-friendly binary and ternary blended binders made of fly-ash and oil-refinery spent catalyst in ready-mixed concrete production. Sustainability 2018, 10, 3136. [Google Scholar] [CrossRef]
- Molnár, Á.; Papp, A. Catalyst recycling—A survey of recent progress and current status. Coord. Chem. Rev. 2017, 349, 1–65. [Google Scholar] [CrossRef]
- Akah, A. Application of rare earths in fluid catalytic cracking: A review. J. Rare Earths 2017, 35, 941–956. [Google Scholar] [CrossRef]
- BASF. Fluid Catalytic Cracking (FCC) Catalyst Optimization to Cope with High Rare Earth Oxide Price Environment. Technical Note. Available online: http://www.catalysts.basf.com/p02/USWeb-Internet/catalysts/en/function/conversions:/publish/content/microsites/catalysts/prods-inds/process-catalysts/BF-9626_US_REAL_Technical_Note.pdf (accessed on 8 November 2018).
- Wang, X.; Yao, M.; Li, J.; Zhang, K.; Zhu, H.; Zheng, M. China’s rare earths production forecasting and sustainable development policy implications. Sustainability 2017, 9, 1003. [Google Scholar] [CrossRef]
- Liang, X.; Ye, M.; Yang, L.; Fu, W.; Li, Z. Evaluation and Policy Research on the Sustainable Development of China’s Rare Earth Resources. Sustainability 2018, 10, 3792. [Google Scholar] [CrossRef]
- Ferella, F.; Innocenzi, V.; Maggiore, F. Oil refining spent catalysts: A review of possible recycling technologies. Resour. Conserv. Recycl. 2016, 108, 10–20. [Google Scholar] [CrossRef]
- Letzsch, W. Global Demand for Catalytic Technology Increases. Hart’s Fuel 2010, 1, 10–14. [Google Scholar]
- Vogt, E.; Weckhuysen, B. Fluid catalytic cracking: Recent: Recent developments on the grand old lady of zeolite catalysis. Chem. Soc. Rev. 2015, 44, 7342–7370. [Google Scholar] [CrossRef]
- Pacewska, B.; Nowacka, M.; Wilińska, I.; Kubissa, W.; Antonovich, V. Studies on the influence of spent FCC catalyst on hydration of calcium aluminate cements at ambient temperature. J. Therm. Anal. Calorim. 2011, 105, 129–140. [Google Scholar] [CrossRef]
- Zornoza, E.; Payá, J.; Monzó, J.; Borrachero, M.V.; Garcés, P. The carbonation of OPC mortars partially substituted with spent fluid catalytic catalyst (FC3R) and its influence on their mechanical properties. Constr. Build. Mater. 2009, 23, 1323–1328. [Google Scholar] [CrossRef]
- Wu, J.-H.; Wu, W.-L.; Hsu, K.-C. The effect of waste oil-cracking catalyst on the compressive strength of cement pastes and mortars. Cem. Concr. Res. 2003, 33, 245–253. [Google Scholar] [CrossRef]
- Chen, H.-L.; Tseng, Y.-S.; Hsu, K.-C. Spent FCC catalyst as a pozzolanic material for high-performance mortars. Cem. Concr. Compos. 2004, 26, 657–664. [Google Scholar] [CrossRef]
- Zhang, H.; Xiao, R.; Wang, D.; Zhong, Z.; Song, M.; Pan, Q.; He, G. Catalytic Fast Pyrolysis of Biomass in a Fluidized Bed with Fresh and Spent Fluidized Catalytic Cracking (FCC) Catalysts. Energy Fuels 2009, 23, 6199–6206. [Google Scholar] [CrossRef]
- Zhang, H.; Xiao, R.; Jin, B.; Shen, D.; Chen, R.; Xiao, G. Catalytic fast pyrolysis of straw biomass in an internally interconnected fluidized bed to produce aromatics and olefins: Effect of different catalysts. Bioresour. Technol. 2013, 137, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xu, Y.; Wang, L.; Zhao, L.; Wang, Q.; Cui, D.; Long, Z.; Huang, X. Recovery of rare earths and aluminum from FCC catalysts manufacturing slag by stepwise leaching and selective precipitation. J. Environ. Chem. Eng. 2017, 5, 3711–3718. [Google Scholar] [CrossRef]
- Wang, J.; Huang, X.; Cui, D.; Wang, L.; Feng, Z.; Hu, B.; Long, Z.; Zhao, N. Recovery of rare earths and aluminum from FCC waste slag by acid leaching and selective precipitation. J. Rare Earths 2017, 35, 1141–1148. [Google Scholar] [CrossRef]
- Innocenzi, V.; Ferella, F.; De Michelis, I.; Vegliò, F. Treatment of fluid catalytic cracking spent catalysts to recover lanthanum and cerium: Comparison between selective precipitation and solvent extraction. J. Ind. Eng. Chem. 2015, 24, 92–97. [Google Scholar] [CrossRef]
- Zhao, Z.; Qiu, Z.; Yang, J.; Lu, S.; Cao, L.; Zhang, W.; Xu, Y. Recovery of rare earth elements from spent fluid catalytic cracking catalysts using leaching and solvent extraction techniques. Hydrometallurgy 2017, 167, 183–188. [Google Scholar] [CrossRef]
- Ye, S.; Jing, Y.; Wang, Y.; Fei, W. Recovery of rare earths from spent FCC catalysts by solvent extraction using saponified 2-ethylhexyl phosphoric acid-2-ethylhexyl ester (EHEHPA). J. Rare Earths 2017, 35, 716–722. [Google Scholar] [CrossRef]
- Page, M.J.; Soldenhoff, K.; Ogden, M.D. Comparative study of the application of chelating resins for rare earth recovery. Hydrometallurgy 2017, 169, 275–281. [Google Scholar] [CrossRef]
- Hubicka, H.; Kołodyńska, D. Studies on application of polyacrylate anion-exchangers in sorption and separation of iminodiacetate rare earth element(III) complexes. Hydrometallurgy 2001, 62, 107–113. [Google Scholar] [CrossRef]
- Dave, S.R.; Kaur, H.; Menon, S.K. Selective solid-phase extraction of rare earth elements by the chemically modified Amberlite XAD-4 resin with azacrown ether. React. Funct. Polym. 2010, 70, 692–698. [Google Scholar] [CrossRef]
- Jha, M.K.; Kumari, A.; Panda, R.; Rajesh Kumar, J.; Yoo, K.; Lee, J.Y. Review on hydrometallurgical recovery of rare earth metals. Hydrometallurgy 2016, 165, 2–26. [Google Scholar] [CrossRef]
- Reed, D.W.; Fujita, Y.; Daubaras, D.L.; Jiao, Y.; Thompson, V.S. Bioleaching of rare earth elements from waste phosphors and cracking catalysts. Hydrometallurgy 2016, 166, 34–40. [Google Scholar] [CrossRef]
- Basaldella, E.I.; Paladino, J.C.; Solari, M.; Valle, G.M. Exhausted fluid catalytic cracking catalysts as raw materials for zeolite synthesis. Appl. Catal. B Environ. 2006, 66, 186–191. [Google Scholar] [CrossRef]
- Basaldella, E.I.; Torres Sánchez, R.M.; Conconi, M.S. Conversion of exhausted fluid cracking catalysts into zeolites by alkaline fusion. Appl. Clay Sci. 2009, 42, 611–614. [Google Scholar] [CrossRef]
- Liu, H.; Ma, J.; Gao, X. Synthesis, characterization and evaluation of a novel resid FCC catalyst based on in situ synthesis on kaolin microspheres. Catal. Lett. 2006, 110, 229–234. [Google Scholar] [CrossRef]
- European Commision. COM (2017) 490 Final. 2017. Available online: http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=COM:2017:0490:FIN (accessed on 5 November 2018).
- Tounsi, H.; Mseddi, S.; Djemel, S. Preparation and characterization of Na-LTA zeolite from Tunisian sand and aluminum scrap. Phys. Procedia 2009, 2, 1065–1074. [Google Scholar] [CrossRef]
- Sallam, A.E.-A.; Al-Zahrani, M.S.; Al-Wabel, M.I.; Al-Farraj, A.S.; Usman, A.R. Removal of Cr (VI) and Toxic Ions from Aqueous Solutions and Tannery Wastewater Using Polymer-Clay Composites. Sustainability 2017, 9, 1993. [Google Scholar] [CrossRef]
- Leone, S.; Ferella, F.; Innocenzi, V.; De Michelis, I.; Veglio, F. Synthesis and Characterization of Zeolites from Spent FCC Catalysts. Chem. Eng. Trans. 2018, 67, 601–606. [Google Scholar]
- Ferella, F.; Belardi, G.; Marsilii, A.; De Michelis, I.; Vegliò, F. Separation and recovery of glass, plastic and indium from spent LCD panels. Waste Manag. 2017, 60, 569–581. [Google Scholar] [CrossRef] [PubMed]
- D’Adamo, I.; Rosa, P.; Terzi, S. Challenges in Waste Electrical and Electronic Equipment Management: A Profitability Assessment in Three European Countries. Sustainability 2016, 8, 633. [Google Scholar] [CrossRef]
- Charoensiddhi, S.; Lorbeer, A.J.; Franco, C.M.M.; Su, P.; Conlon, M.A.; Zhang, W. Process and economic feasibility for the production of functional food from the brown alga Ecklonia radiata. Algal Res. 2018, 29, 80–91. [Google Scholar] [CrossRef]
- Kwan, T.H.; Pleissner, D.; Lau, K.Y.; Venus, J.; Pommeret, A.; Lin, C.S.K. Techno-economic analysis of a food waste valorization process via microalgae cultivation and co-production of plasticizer, lactic acid and animal feed from algal biomass and food waste. Bioresour. Technol. 2015, 198, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Porvali, A.; Wilson, B.P.; Lundström, M. Lanthanide-alkali double sulfate precipitation from strong sulfuric acid NiMH battery waste leachate. Waste Manag. 2018, 71, 381–389. [Google Scholar] [CrossRef]
- Banos-Gonzalez, I.; Martínez-Fernández, J.; Esteve-Selma, M.-Á.; Esteve-Guirao, P. Sensitivity analysis in socio-ecological models as a tool in environmental policy for sustainability. Sustainability 2018, 10, 2928. [Google Scholar] [CrossRef]
- Grand View Research Inc. Zeolite Market. Size, Share & Trends Analysis Report by Product, by Application (Catalysts, Detergent Builders), by Region. (North. America, Europe, Asia Pacific, CSA, MEA), and Segment Forecasts, 2012–2022. 2018. Available online: https://www.grandviewresearch.com/industry-analysis/zeolites-market (accessed on 17 December 2018).
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).