1. Introduction and Outline
Price fluctuations of phosphorus fertilizers since about 2005 indicate that the market for P has become volatile. During 2007/8, prices increased by 300%. Some reasons were the closing of two phosphate rock mines in the USA and the export stop for P in the USA and China. At the same time, P markets in India and Southeast Asia increased. Soils in sub-saharan Africa are still poor in plant available P; thus, new markets will also develop in future. Although prices had dropped again after this tremendous increase, they never reached their old level and a steady increase can be expected as already outlined in the CRU-report ‘Phosphate rock: ten year outlook (2011)’ (refer to scope newsletter No. 81, November 2011,
https://phosphorusplatform.eu/images/download/ScopeNewsletter81.pdf).
Of course, dynamics of the P market depend also on the sources of phosphate rock (PR). Phosphorus is the eleventh most abundant element worldwide. According to [
1] global P can be divided into three groups due to its accessibility. The largest amount of P most probably belongs to the global potential; however, these natural deposits are not known or mining is economically or technically not feasible. For instance, P in submarine sediments belongs to this group. Phosphorus resources are known deposits, but mining would be not economic, for example, deposits in Siberia far away from any infrastructure. Potential deposits deduced from geological formations but still unexplored also belong to the resources. Reserves are deposits undergoing mining or known deposits where mining would be technically and economically feasible. The development of phosphate rock reserves in recent years is shown in
Table 1 [
2]. The quotient between reserves and worldwide production results in the static lifetime of phosphate rock. In the early 2000s static lifetime was about one hundred years, pointing to the scarcity of phosphate rock. In 2010, Morocco announced another 50 billion tons of PR reserves, extending static lifetime to nearly 400 years. In recent years, production in China has increased dramatically, reducing static lifetime down to 250–300 years. Hence, as seen from the figures, PR reserves are limited but there will be no scarcity over the next centuries. Furthermore, world resources of PR are more than 300 billion tons.
However, PR reserves are not evenly distributed worldwide (
Table 2). In Europe, only Finland runs a small mine; hence, Europe has to import PR. Worldwide, the largest reserves are located in countries south and east of the Mediterranean Sea ranging from Morocco to Jordan and Syria, in China, and in the USA. The largest worldwide producers are China, USA, and Morocco, but China and the USA have stopped exporting PR. Importing from countries around the Mediterranean Sea contains uncertainties for different reasons. (I) Quite a few of these countries are politically unstable, for example, Syria. The largest producer in this region, Morocco, shows a comparatively good political stability. However, the PR mines are mainly located in Western Sahara, which is only annexed by Morocco and this annexation is not recognised by the UN; (II) Moroccan PR contains also high concentrations of cadmium and uranium. The removal of these pollutants makes the fertilizer production more expensive; (III) Production of PR is very water demanding, in these arid regions there is strong competition for water between the human population, agriculture, and PR production. Europe should reduce its dependency on PR imports.
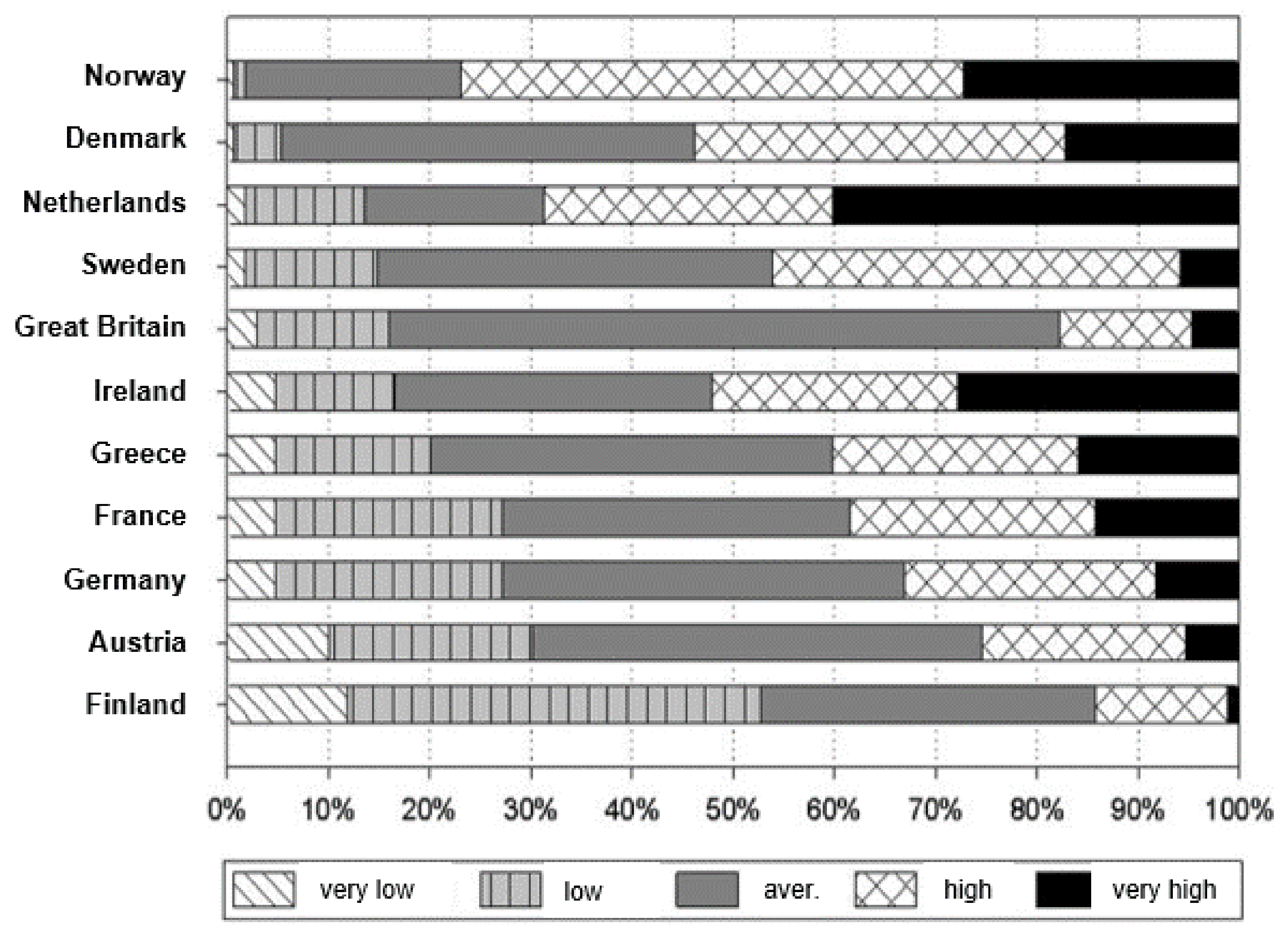
Figure 1 shows the soil P status in some European countries based on the particular national soil P extraction methods and advisory services, as such, data are not directly comparable [
3]. However, it is obvious that most soils are well or even highly supplied with P; hence, there is no reason for alarmism when PR imports drop. In Germany, only about 3% of soils are in a very low supply level (class A,
Table 3), whereas more than 40% of the soils are in a high or very high supply level (class D, E) [
4]. Of course, there are regional differences. In areas with intense animal production like north western Germany, soils are extremely over-supplied with P, meaning there is no need for P fertilization for the next 200 years (refer to [
5]). Even in 85 cm soil depth, P concentrations can be as high as 90 mg P/kg soil, which would be equivalent to the well supply level in the plow layer (class C). In areas with mainly plant production, the P supply status of soils is less but is usually still sufficient.
The increasing prices for P fertilizer and the necessity to become independent from PR imports draw attention to a reduction of P fertilization. In Germany, soils are classified according to their P supply level (class A–E) and recommended fertilization is double the removal (class A), removal (class C), or nil (class E). However, the threshold values of these classes, as set by the Association of German Agricultural Analytic and Research Institutes (VDLUFA) in 1997 [
6], are relatively high. This resulted in high soil P concentrations and concomitantly eutrophication of water bodies. Therefore, recent discussions stress the necessity to reduce these threshold values (e.g., the 121. VDLUFA congress 2009 in Karlsruhe [
7]). Another attempt to reduce PR imports would be the recycling of phosphorus from organic wastes. This will be the topic of this manuscript.
The potential of P recycling in Germany is given in
Table 4. The yearly production of sewage sludge is about 2.4 million tons of dry matter. Assuming an average P concentration of 2% results in about 48,000 t of P. Another 20,000 t are in meat-and-bone meal. The consumption of mineral P fertilizer ranged between 100 and 130 thousand tons in recent years. Hence, about 50–70% of mineral fertilizer P could theoretically be replaced by organic wastes. The direct use of sewage sludge and meat-and-bone meal as fertilizer is restricted or forbidden. The new German sewage sludge ordinance (2017) stipulates the recycling of P for larger wastewater treatment plants, however, with a transition period of 12–15 years. Hence, the necessity for developing recycling techniques is given.
In 2004, the German government represented by three ministries (research, environment, and agriculture) started an initiative to support 17 different research and industrial projects for developing techniques of P recycling and assessing their practicability [
10]. These projects covered a wide range of technical approaches like precipitation of P, adsorption, crystallization, nano-filtration, electro-dialyses, elution, wet oxidation, pyrolysis, ion exchange, or bioleaching. This resulted in 32 different recycling products which were tested by five institutes for their agronomic efficiency, that is, plant availability. This has been done mainly in pot experiments on different soils. Measured P uptake was evaluated in comparison to water soluble P fertilizer like single super phosphate (SSP) or triple super phosphate (TSP). The objective of these projects was to identify promising techniques and products for future developments. Similar projects are supported by the EU (e.g., “SUSAN” [
11] or ‘P-REX’ [
12]) or by other institutions like Fachagentur für Nachwachsende Rohstoffe (FNR, [
13]).
In this manuscript, we will summarize and compare the results of these projects. We start with a short introduction to some recycling techniques. This might be of help to understand the differences in the agronomic efficiency. Furthermore, it gives valuable information for farmers because some management systems restrict the use of certain fertilizers, for instance, water soluble P fertilizers are not allowed in organic farming. More emphasis should be on published results about the fertilizer effect of different recycling products.
4. Discussion
Plant growth depends, besides the uptake of nutrients, on many other soil and environmental factors. In contrast, P uptake depends mainly on P availability in soil and accordingly on solubility of P fertilizers and their impact with soil chemistry. Therefore, the cited experiments were mainly evaluated by measured P uptake. Biomass production would also be influenced by the growth parameter mentioned above and the internal utilization efficiency of different crops or varieties. Hence, the amount of P taken up is a good measure in pot experiments to assess the availability of fertilizers and P recycling products. In previous decades, mainly water soluble P fertilizers like ammoniumphosphate, single superphosphate (SSP), or triple superphosphate (TSP) have been used in conventional farming, for example, in Germany 94 percent of P fertilizers used were water soluble. Therefore, SSP or TSP was used as control in the described experiments; in both fertilizers Ca(H2PO4)2·H2O is the dominating P form.
Table 21 tries to summarize all results by comparing the uptake from P recycling products relative to SSP or TSP in four ‘efficiency groups’, these are 0–25, 26–50, 51–75, and 76–100 percent. This would mean the products are categorized as completely unsuitable as fertilizer, widely unsuitable, limitedly suitable, or suitable, respectively.
Most results (from three institutions) are available for thermo-chemically digested products, that is, sewage sludge ashes reheated to about 800–1000 °C together with chloride to reduce heavy metal concentration. In 23 experiments only one product reached the group of 51–75 percent (54%) uptake on sand. For this product, the digestion was with MgCl
2 (
Table 6). The second positive result on loam is not reliable, because uptake of fertilized plants was only 5 to 7 mg higher than in the unfertilized control (101 mg P) and this was statistically not different.
Altogether, in 10 experiments out of 23 (43 percent), plant uptake (mainly by maize) was less than 25 percent of uptake from TSP, and in another 11 experiments (48 percent) it was less than 50 percent. Hence, in over 90 percent of all experiments availability was not or hardly given and the products have to be categorized as completely or widely unsuitable, independently of soil texture or soil pH. The reason is most probably the formation of chloride apatites during the second heating when CaCl
2 has been used as chloride donator; reference [
33] identified chloride apatite minerals in such products. People should also be warned to use these products for NPK fertilizers as long as their chemical structure is not altered by producing these NPKs. Farmers will purchase a fertilizer with a good N and K availability but without any P availability leading to an unlevelled plant nutrition.
Meat-and-bone meal ashes failed on sandy and loamy soils, too. This is most probably also due to apatites as the main binding form of P in ashes. However, sintering of meat-and-bone meal together with soda and quartz sand results in a product (Ulo-Phos) which reached a relative availability of 87 percent on sand and 65–74 percent on loam in comparison to TSP, that is, the product is suitable as fertilizer independently of the soil. A similar alkaline digestion, but based on sewage sludge ashes, was used to produce Ash-Dec. It also showed a good availability of 74–100% of TSP even at high soil pH values. Both products are similar to Rhenania phosphate or alkaline sinter phosphates from decades ago. However, using meat-and-bone meal instead of phosphate rock as substrate results in a product low in heavy metals. Furthermore, the production process doesn’t need harmful chemicals and is less accident-sensitive than a chemical wet digestion.
The results of the experiments with plant ashes cannot be discussed because SSP or TSP controls were lacking as well as a balancing fertilization with potassium and magnesium. However, by using plant ashes it has to be considered that their pH value is about 11 to 13, that is, similar to hydrated lime [
13,
33]. Consequently, soil pH will rise by using plant ashes as K or P fertilizer. This pH rise was from 6.9 to 7.5, that is, 0.6 pH units, on a highly pH buffered clay soil [
33]. On less buffered sandy soils, a much higher rise of soil pH can be expected with the risk of micronutrient fixation, while on acid sandy soils the ash application could have an additional liming effect.
The cupola slag (Mephrec) failed on the acid sandy soil and performed very well on the neutral loamy soil. This is in contrast to most of the other P fertilizers and leads to the question about the P binding form in this product, which is still not known and needs further investigation. However, the slag also had a relatively high Al concentration. Therefore, phosphate might have been bound to Al, which would explain the relation between soil pH and solubility.
For the tested Ca-phosphates the results are heterogeneous, regardless if precipitated with Ca(OH)
2 or crystallized on calcium-silicate-hydrate surfaces (
Table 21). P-Roc 09, Fix-P, SESAL and a further Ca-phosphate failed completely on the loamy soil; they showed a relative availability of 0–25 percent of TSP. The Pasch products reached 26–50 percent on sand and once 51–75 percent each on sand and loam. The experiment with rye seedlings (
Table 18) indicated that the solubility of Ca phosphates steadily decreased with an increasing Ca content in the binding forms up to apatites. All tested products might have a mixture of primary, secondary, and tertiary phosphates and apatites. The composition defines the fertilizer efficiency. With the technologies used it seems impossible to influence the precipitation or sorption conditions in a way that mainly primary and secondary phosphates occur which would be desirable. An exception might be the PASCH process, where P is first leached out of the sewage sludge ash and afterwards precipitated by increasing the pH value of the leachate. This allows a better control of the precipitation process compared to the other procedures and might be the reason for the better availability on loamy soils. In general, Ca-phosphates are better soluble at low pH values. This is confirmed in the described pot experiments, because availability on the strongly acid sands was higher than on the slightly acid loamy soils. It is rather astonishing that even at pH values as low as 4.4–4.7 availability remains less than 75% of TSP. Therefore, these products are unsuitable for most soils. However, the products can be used as substrate for digestion with sulfuric or phosphoric acids, as is done for production of SSP or TSP. Another possibility would be the production of elemental P under reductive conditions with coke [
34]. Elemental P can be used for further industrial processes.
The good fertilizer effect of the tested Mg-phosphates produced by the Seaborn procedure is remarkable. They performed as well as the pure Mg
3(PO
4)·4H
2O. All Mg-phosphates had a better availability than the recycled Ca-phosphates, which is also given for the Mg-treated ashes (
Table 6 and
Table 13). Magnesium-ammonium-phosphates (MgAP), that is, struvite, showed the best availability of all tested substances and can be recommended as P fertilizer. The MgAP from Stuttgart (MgAP St) was tested on four soils by two institutes and led to a relative P uptake of 81–103 percent, MgAP Seaborne (MgAP Sb) reached 64–129 percent, and only uptake from MgAP Gifhorn (MgAP Gf) was a bit lower with 45–92 percent (
Table 19). The good availability of MgAPs was also shown by [
29] in an experiment with rye seedlings. The comparably low availability of the MgAP Gf is probably due to a relative high Fe concentration. It was, with 5.5 percent, much higher than for the other two tested MgAPs, with 0.4–1 percent [
35]. The availability of Fe-phosphates is lower than for Ca- or Mg-phosphates (
Table 18). It is possible that during the production of the MgAP product in the sewage works part of the phosphate is already precipitated as Fe-phosphate, or that this happens in the soil after solving of the MgAP in close vicinity to Fe. In a first attempt, the good availability of MgAPs is surprising, because water solubility is regarded as very low. With the method of VDLUFA [
36] only 0.5–2 percent of MgAPs is water soluble [
29,
35], whereas water solubility of SSP or TSP is 84 and 93 percent, respectively. However, the VDLUFA method measures water solubility with a narrow relation of fertilizer to water (10g in 500 mL). At this relation, the equilibrium concentration of MgAP is easily reached and no further MgAP can be solved. In [
37], it was shown that the equilibrium concentration of MgAP is higher than an average soil solution concentration. In soils, the P buffer capacity keeps the soil solution concentration relatively stable and below the equilibrium concentration of MgAP, resulting in a steady state equilibrium by which all MgAP can be solved.
Therefore, and also according to [
35] the parameter ‘water solubility’ as well as ‘citrate solubility’ are not good measures to describe plant availability of P fertilizer. A much better correlation to plant uptake was given for soil solution concentration (C
L) or the isotopically exchangeable phosphate (IEP). Both parameters had been measured in soil samples without plants and three weeks after fertilization. For MgAPs the values of C
L and IEP were in the same order than for TSP. For cupola slag, IEP was much higher in the neutral loam compared to the acid sand which is in accordance to plant availability. The same relation between IEP and availability was found for Ca-phosphates, however with a different relation to soil pH, higher IEP values on the acid sands and lower ones for neutral loam. The conclusion of the authors was that isotopically exchangeable phosphate would be a good method for assessing the plant availability of phosphate in P recycling products.
Most of the described experiments had been performed as pot experiments; thus, for a final conclusion about the suitability of the recycling products as fertilizers, field experiments are necessary, at least with the most promising products like MgAP or Sinter-P. However, a number of field experiments are still relatively scarce and a P fertilizer effect is often not visible due to the high P supply level of most soils. In parallel to the pot experiments, [
17] also tested some of the products in field experiments on three different sites in a 3 year crop rotation with winter barley, rapeseed, and winter wheat. Despite the fact that all sites were in a ‘very poor’ P supply level according to VDLUFA [
6] (CAL-P: 18–21 mg/kg) there was no fertilizer effect. All fertilized treatments achieved the same biomass production, grain yield, and P uptake as the unfertilized control.
5. Conclusions
The fertilizer effect of the tested P recycling products can clearly be differentiated: TSP = MgAP > Mg-P = sinter-P > Ca-P, cupola-slag > thermally treated sewage sludge ashes > meat-and-bone meal ash = Fe-P.
A phosphate availability comparable to TSP or SSP was only given for struvite, that is, magnesium-ammonium-phosphate, if it was poor in Fe impurities. An unacceptable low availability was found for Fe-phosphates, meat-and-bone meal ashes, and sewage sludge ashes, even though they were thermally treated together with CaCl2. All other products, precipitates or slags, are grouped in between these two extremes. Magnesium phosphates, either precipitates or treated ashes, had a higher availability than Ca-phosphates. The tested Ca-phosphates are most probably tertiary phosphates or apatites which are unsuitable as fertilizers. However, they can be used as substrate for further chemical digestions to produce fertilizers like SSP or TSP. Also, a reduction, together with coke, to elemental P is possible. An acceptable availability was achieved with alkaline sintered meat-and-bone meal (Ulo-Phos) and at least on neutral soil for cupola slag. Further developments in P recycling processes should take these findings into account.
Most of the described results were obtained in pot experiments, and need a confirmation in field experiments. Field experiments need a longer time scale, because soil processes for P are much slower compared to, for instance, nitrogen. Relatively quick and probably good information about expected availability can be achieved by the isotopically exchangeable P (IEP) method. Hence, this method can be used to select promising products for field experiments.