A Global Synthesis Reveals Gaps in Coastal Habitat Restoration Research
Abstract
1. Introduction
2. Materials and Methods
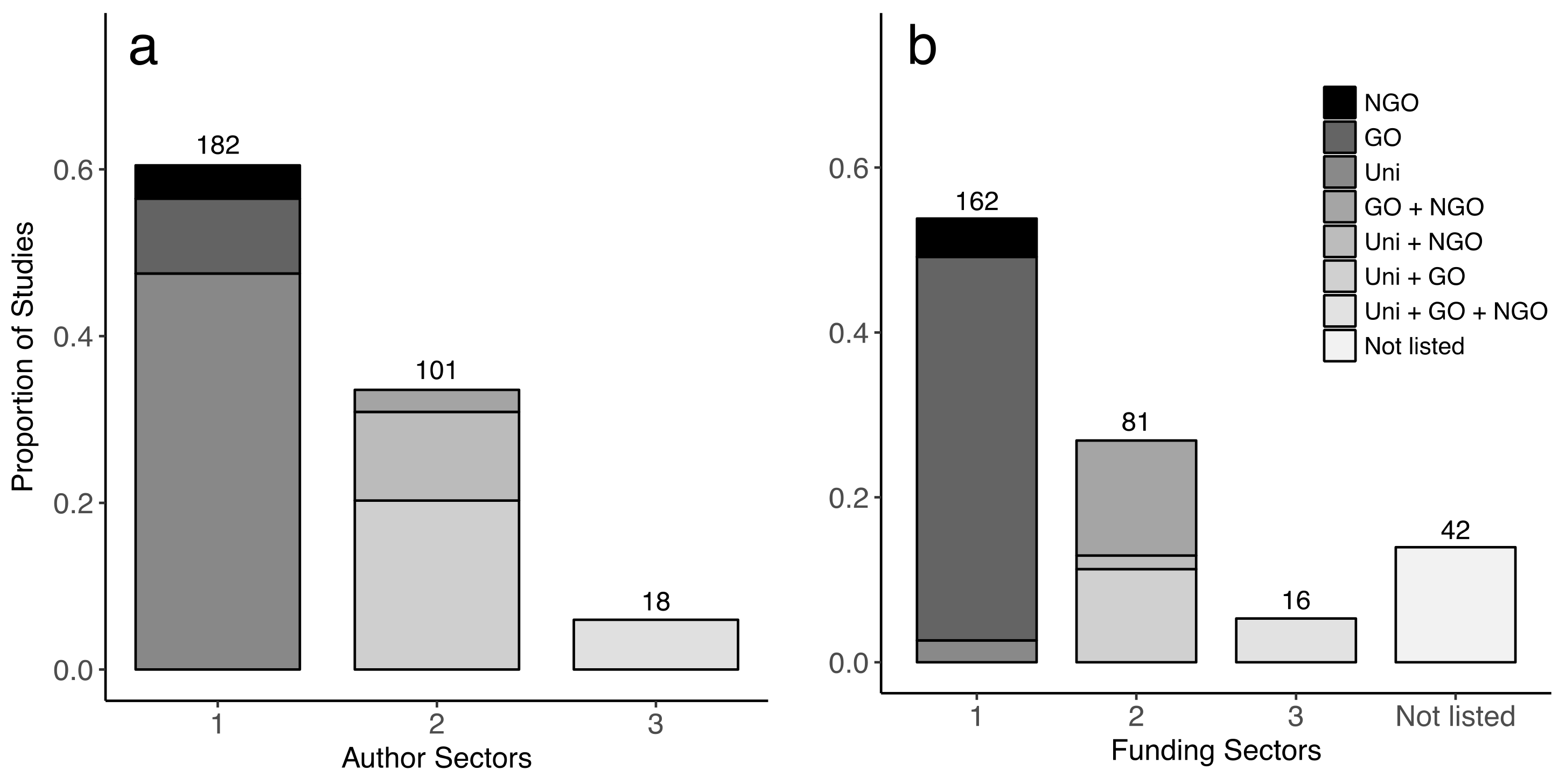
2.1. Cross-Sector Collaborations
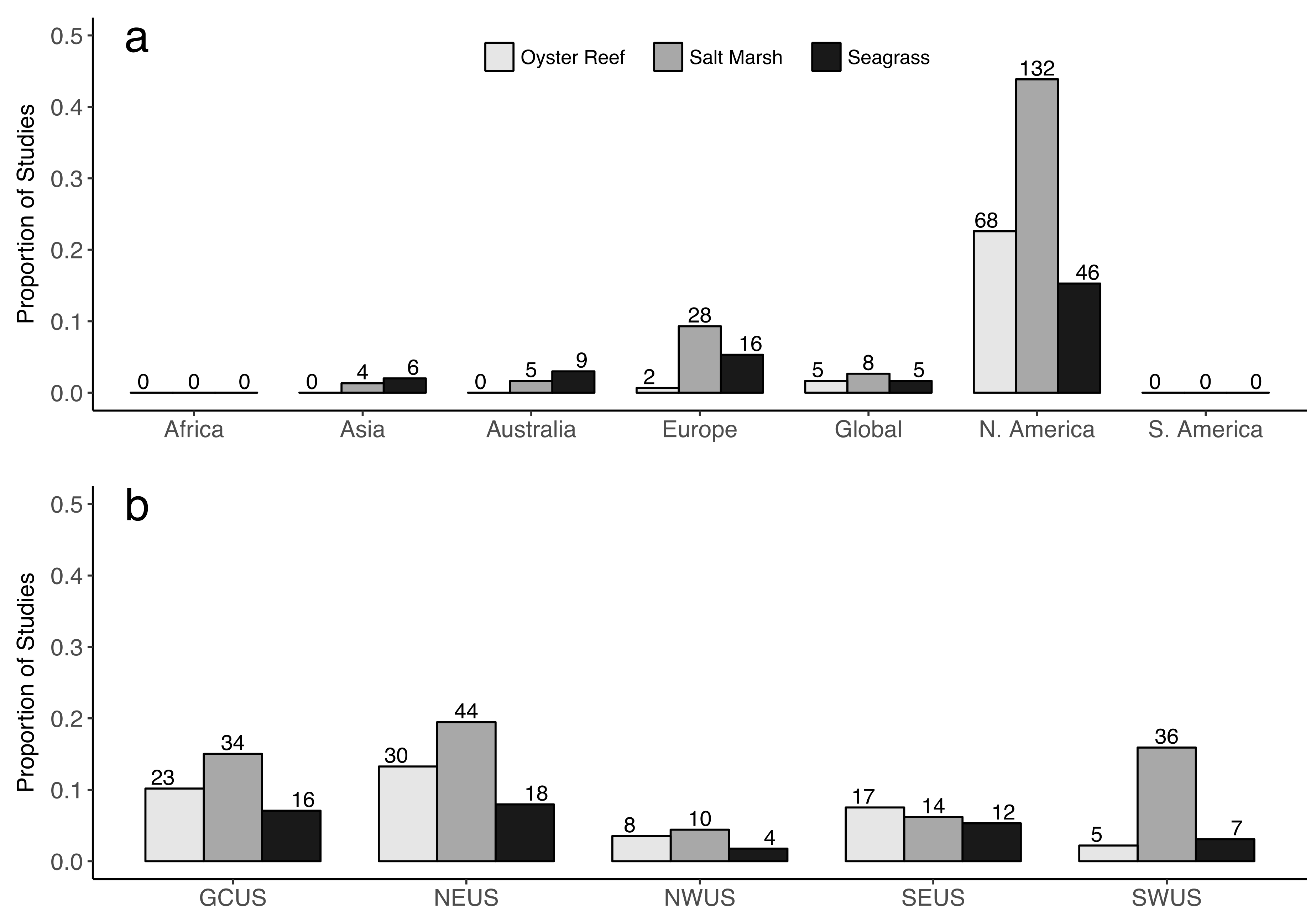
2.2. Restoration Geography
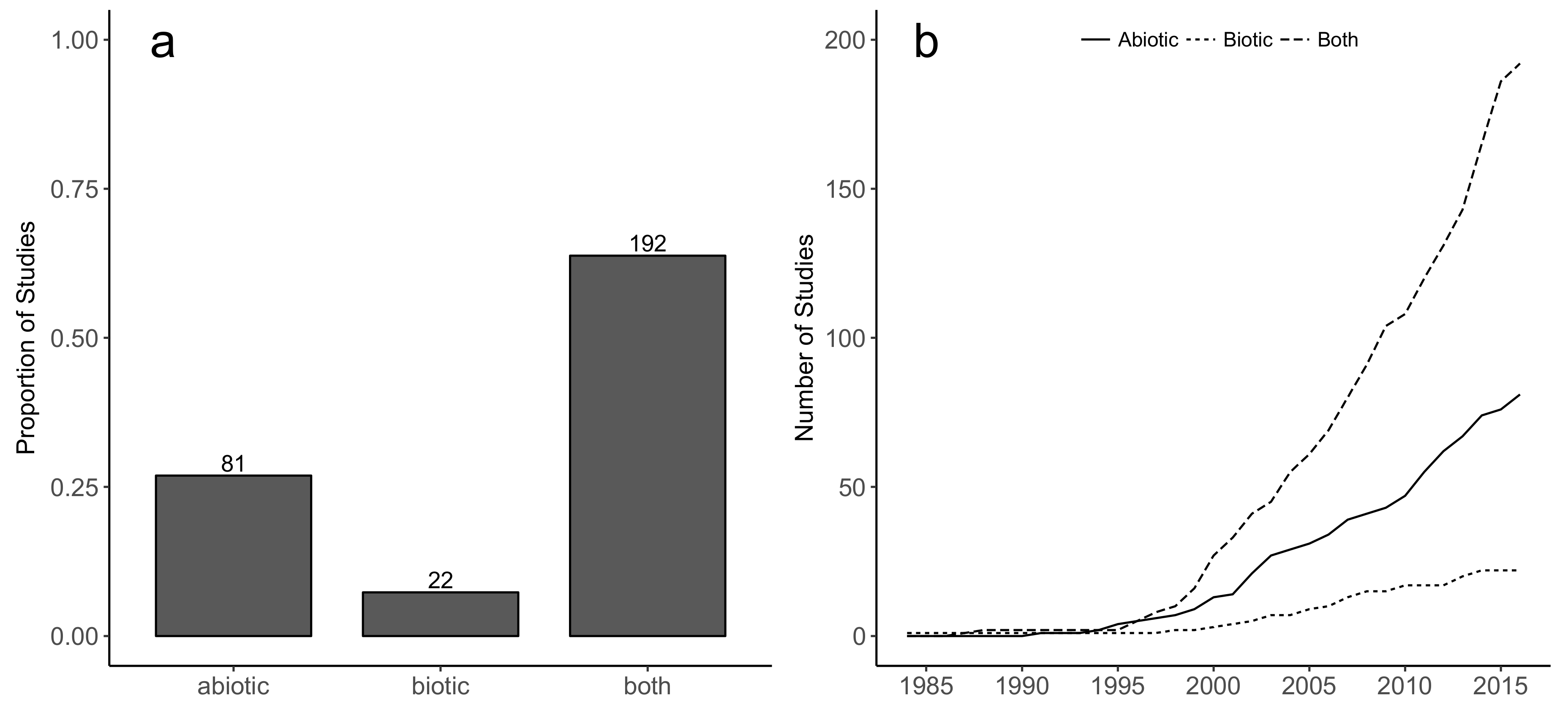
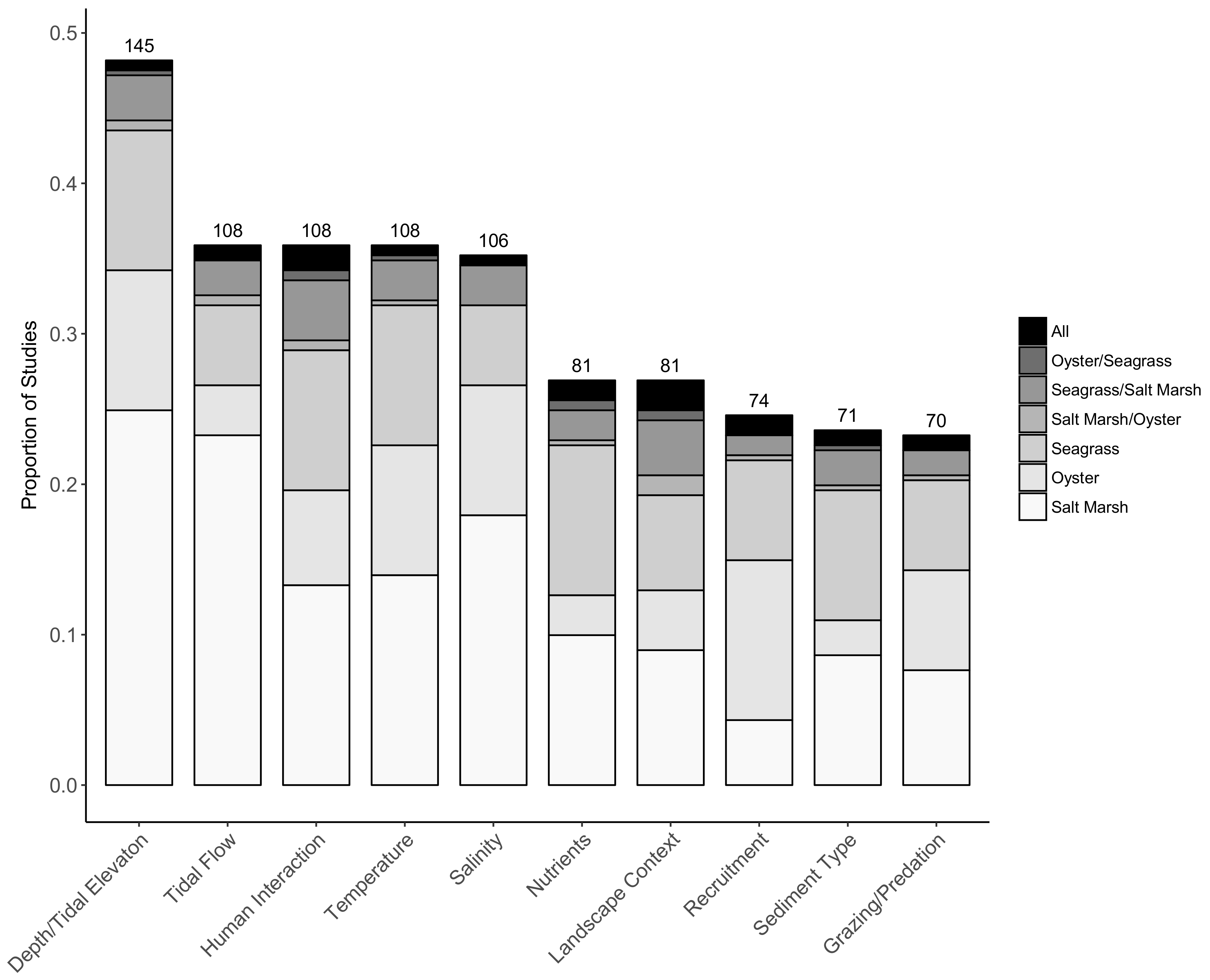
2.3. Factors Considered in Restoration Methodologies
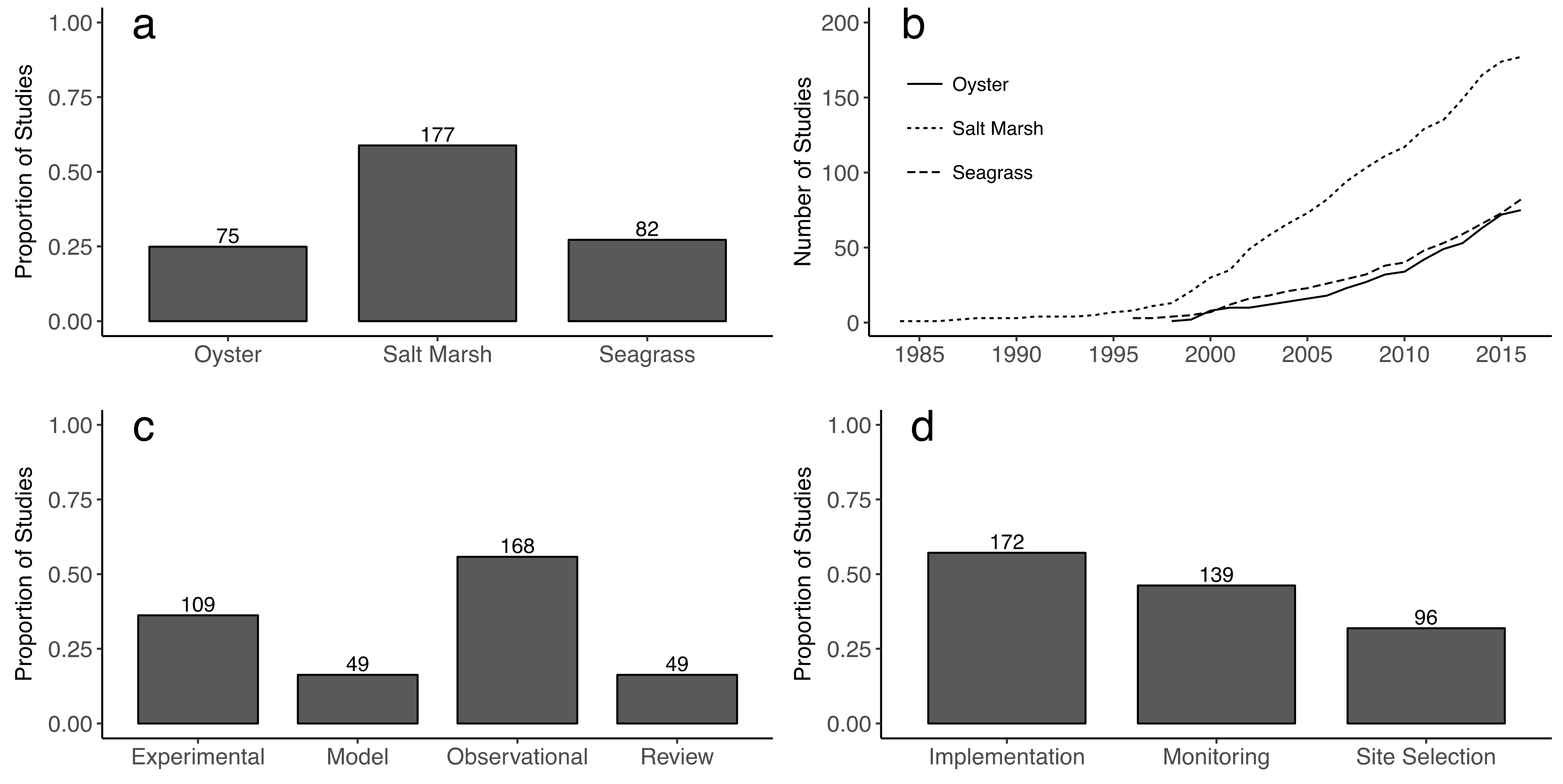
3. Results
4. Discussion
4.1. Cross-Sector Collaborations
4.2. Restoration Geography
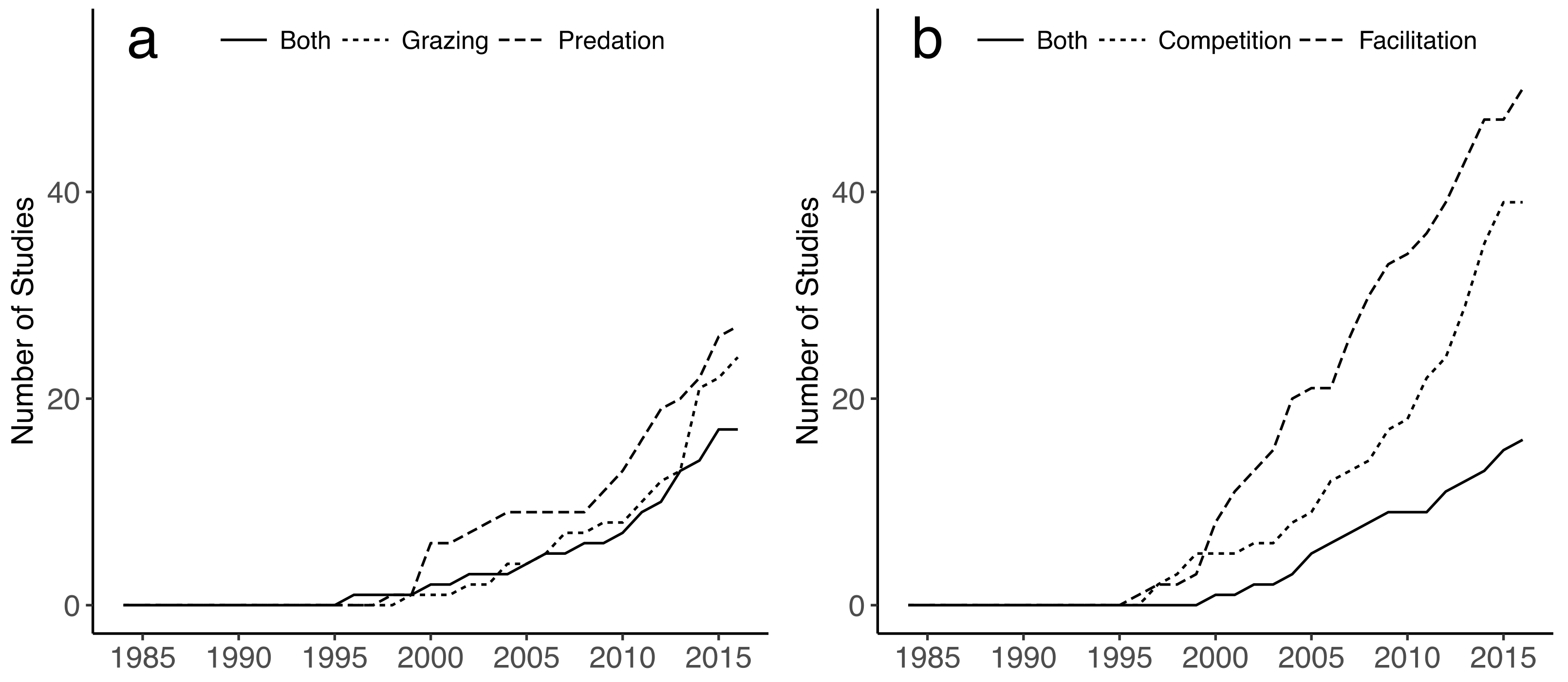
4.3. Factors Considered in Restoration Methodologies
4.4. Recommendations for Coastal Habitat Restoration
- A broad discussion of the extent to which cross-sector collaborations with significant intellectual contributions from all participants occur as well as their effects on knowledge transference and adaptive management of restoration projects.
- A greater emphasis on- and communication of restoration research that occurs outside of the western hemisphere.
- The inclusion of biotic interactions, in addition to the physical template (specifically, top-down effects and facilitation) as a potential means to further enhance restoration yields.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jackson, J.B.C.; Kirby, M.X.; Berger, W.H.; Bjorndal, K.A.; Botsford, L.W.; Bourque, B.J.; Bradbury, R.H.; Cooke, R.; Erlandson, J.; Estes, J.A.; et al. Historical overfishing and the recent collapse of coastal ecosystems. Science 2001, 293, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Leemans, R.; De Groot, R.S. Millennium Ecosystem Assessment: Ecosystems and Human Well-Being: A Framework for Assessment; Island Press: Washington, DC, USA, 2003. [Google Scholar]
- Lotze, H.K.; Lenihan, H.S.; Bourque, B.J.; Bradbury, R.H.; Cooke, R.G.; Kay, M.C.; Kidwell, S.M.; Kirby, M.X.; Peterson, C.H.; Jackson, J.B.C. Depletion, degradation, and recovery potential of estuaries and coastal seas. Science 2006, 312, 1806–1809. [Google Scholar] [CrossRef] [PubMed]
- Beck, M.W.; Brumbaugh, R.D.; Airoldi, L.; Carranza, A.; Coen, L.D.; Crawford, C.; Defeo, O.; Edgar, G.J.; Hancock, B.; Kay, M.C.; et al. Oyster Reefs at Risk and Recommendations for Conservation, Restoration, and Management. BioScience 2011, 61, 107–116. [Google Scholar] [CrossRef]
- Waycott, M.; Duarte, C.M.; Carruthers, T.J.B.; Orth, R.J.; Dennison, W.C.; Olyarnik, S.; Calladine, A.; Fourqurean, J.W.; Heck, K.L.; Hughes, A.R.; et al. Accelerating loss of seagrasses across the globe threatens coastal ecosystems. Proc. Natl. Acad. Sci. USA 2009, 106, 12377–12381. [Google Scholar] [CrossRef] [PubMed]
- Young, T.P. Restoration ecology and conservation biology. Biol. Conserv. 2000, 92, 73–83. [Google Scholar] [CrossRef]
- Young, T.P.; Petersen, D.A.; Clary, J.J. The ecology of restoration: Historical links, emerging issues and unexplored realms. Ecol. Lett. 2005, 8, 662–673. [Google Scholar] [CrossRef]
- Jordan, W.R.; Gilpin, M.E.; Aber, J.D. Restoration Ecology: A Synthetic Approach to Ecological Research; Cambridge University Press: Cambridge, UK, 1990. [Google Scholar]
- Dobson, A.P.; Bradshaw, A.D.; Baker, A.J.M. Hopes for the future: Restoration ecology and conservation biology. Science 1997, 277, 515–522. [Google Scholar] [CrossRef]
- Suding, K.N. Toward an era of restoration in ecology: Successes, failures, and opportunities ahead. Annu. Rev. Ecol. Evol. Syst. 2011, 42, 465–487. [Google Scholar] [CrossRef]
- Tallis, H.; Lubchenco, J. A call for inclusive conservation. Nature 2014, 515, 27. [Google Scholar] [CrossRef] [PubMed]
- Fisher, R.; Radford, B.T.; Knowlton, N.; Brainard, R.E.; Michaelis, F.B.; Caley, M.J. Global mismatch between research effort and conservation needs of tropical coral reefs. Conserv. Lett. 2011, 4, 64–72. [Google Scholar] [CrossRef]
- Jenkins, C.N.; Van Houtan, K.S.; Pimm, S.L.; Sexton, J.O. US protected lands mismatch biodiversity priorities. Proc. Natl. Acad. Sci. USA 2015, 112, 5081–5086. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.K.; Szaro, R.C.; Shapiro, C.D. Adaptive Management: The US Department of the Interior Technical Guide; US Department of the Interior, Adaptive Management Working Group: Washington, DC, USA, 2007.
- Hardy, C.; Phillips, N.; Lawrence, T.B. Resources, knowledge and influence: The organizational effects of interorganizational collaboration. J. Manag. Stud. 2003, 40, 321–347. [Google Scholar] [CrossRef]
- Dietz, T.; Ostrom, E.; Stern, P.C. The Struggle to Govern the Commons. Science 2003, 302, 1907–1912. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Improving Risk Communication; National Academies: Washington, DC, USA, 1989. [Google Scholar]
- Sunderland, T.; Sunderland-Groves, J.; Shanley, P.; Campbell, B. Bridging the Gap: How Can Information Access and Exchange Between Conservation Biologists and Field Practitioners be Improved for Better Conservation Outcomes? Biotropica 2009, 41, 549–554. [Google Scholar] [CrossRef]
- Bennett, N.J.; Roth, R.; Klain, S.C.; Chan, K.; Clark, D.A.; Cullman, G.; Epstein, G.; Nelson, M.P.; Stedman, R.; Teel, T.L. Mainstreaming the social sciences in conservation. Conserv. Biol. 2017, 31, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Nature Publishing Group. The great divide. Nature 2007, 450, 135–136. [Google Scholar]
- Du Toit, J.T.; Walker, B.H.; Campbell, B.M. Conserving tropical nature: Current challenges for ecologists. Trends Ecol. Evol. 2004, 19, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Stinchcombe, J.; Moyle, L.; Hudgens, B.; Bloch, P.; Chinnadurai, S.; Morris, W. The influence of the academic conservation biology literature on endangered species recovery planning. Conserv. Ecol. 2002, 6, 15. [Google Scholar] [CrossRef]
- Juffe-Bignoli, D.; Brooks, T.M.; Butchart, S.H.M.; Jenkins, R.B.; Boe, K.; Hoffmann, M.; Angulo, A.; Bachman, S.; Böhm, M.; Brummitt, N.; et al. Assessing the Cost of Global Biodiversity and Conservation Knowledge. PLoS ONE 2016, 11, e0160640. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, E.S.; Palmer, M.A.; Allan, J.; Alexander, G.; Barnas, K.; Brooks, S.; Carr, J.; Clayton, S.; Dahm, C.; Follstad-Shah, J. Synthesizing US river restoration efforts. Science 2005, 308, 636–637. [Google Scholar] [CrossRef] [PubMed]
- Kareiva, P.; Marvier, M.; West, S.; Hornisher, J. Slow-moving journals hinder conservation efforts. Nature 2002, 420, 15. [Google Scholar] [CrossRef] [PubMed]
- Knight, A.T.; Cowling, R.M.; Rouget, M.; Balmford, A.; Lombard, A.T.; Campbell, B.M. Knowing But Not Doing: Selecting Priority Conservation Areas and the Research—Implementation Gap. Conserv. Biol. 2008, 22, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Kareiva, P.; Groves, C.; Marvier, M. REVIEW: The evolving linkage between conservation science and practice at The Nature Conservancy. J. Appl. Ecol. 2014, 51, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Imperial, M.T. Institutional analysis and ecosystem-based management: The institutional analysis and development framework. Environ. Manag. 1999, 24, 449–465. [Google Scholar] [CrossRef]
- Wilcove, D.S.; Rothstein, D.; Dubow, J.; Phillips, A.; Losos, E. Quantifying threats to imperiled species in the United States. Bioscience 1998, 48, 607–615. [Google Scholar] [CrossRef]
- Kappel, C.V. Losing pieces of the puzzle: Threats to marine, estuarine, and diadromous species. Front. Ecol. Environ. 2005, 3, 275–282. [Google Scholar] [CrossRef]
- Lawler, J.J.; Aukema, J.E.; Grant, J.B.; Halpern, B.S.; Kareiva, P.; Nelson, C.R.; Ohleth, K.; Olden, J.D.; Schlaepfer, M.A.; Silliman, B.R. Conservation science: A 20-year report card. Front. Ecol. Environ. 2006, 4, 473–480. [Google Scholar] [CrossRef]
- Mouillot, D.; Parravicini, V.; Bellwood, D.R.; Leprieur, F.; Huang, D.; Cowman, P.; Albouy, C.; Hughes, T.P.; Thuiller, W.; Guilhaumon, F. Global marine protected areas do not secure the evolutionary history of tropical corals and fishes. Nature Commun. 2016, 7, 10359. [Google Scholar] [CrossRef] [PubMed]
- Brito, D.; Oprea, M. Mismatch of Research Effort and Threat in Avian Conservation Biology. Trop. Conserv. Sci. 2009, 2, 353–362. [Google Scholar] [CrossRef]
- Odum, E.P.; Odum, H.T.; Andrews, J. Fundamentals of Ecology; Saunders: Philadelphia, PA, USA, 1971; Volume 3. [Google Scholar]
- Teal, J.M. Energy flow in the salt marsh ecosystem of Georgia. Ecology 1962, 43, 614–624. [Google Scholar] [CrossRef]
- Fazey, I.; Fischer, J.; Lindenmayer, D.B. What do conservation biologists publish? Biol. Conserv. 2005, 124, 63–73. [Google Scholar] [CrossRef]
- Bertness, M.D.; Grosholz, E. Population dynamics of the ribbed mussel, Geukensia demissa: The costs and benefits of an aggregated distribution. Oecologia 1985, 67, 192–204. [Google Scholar] [CrossRef] [PubMed]
- Bertness, M.D.; Callaway, R. Positive interactions in communities. Trends Ecol. Evol. 1994, 9, 191–193. [Google Scholar] [CrossRef]
- Bertness, M.D.; Hacker, S.D. Physical stress and positive associations among marsh plants. Am. Nat. 1994, 144, 363–372. [Google Scholar] [CrossRef]
- He, Q.; Bertness, M.D.; Altieri, A.H. Global shifts towards positive species interactions with increasing environmental stress. Ecol. Lett. 2013, 16, 695–706. [Google Scholar] [CrossRef] [PubMed]
- Silliman, B.R.; Schrack, E.; He, Q.; Cope, R.; Santoni, A.; Van Der Heide, T.; Jacobi, R.; Jacobi, M.; Van De Koppel, J. Facilitation shifts paradigms and can amplify coastal restoration efforts. Proc. Natl. Acad. Sci. USA 2015, 112, 14295–14300. [Google Scholar] [CrossRef] [PubMed]
- Angelini, C.; Griffin, J.N.; van de Koppel, J.; Lamers, L.P.; Smolders, A.J.; Derksen-Hooijberg, M.; van der Heide, T.; Silliman, B.R. A keystone mutualism underpins resilience of a coastal ecosystem to drought. Nat. Commun. 2016, 7, 12473. [Google Scholar] [CrossRef] [PubMed]
- Halpern, B.S.; Silliman, B.R.; Olden, J.D.; Bruno, J.P.; Bertness, M.D. Incorporating positive interactions in aquatic restoration and conservation. Front. Ecol. Environ. 2007, 5, 153–160. [Google Scholar] [CrossRef]
- Dayton, P.K. Toward an understanding of community resilience and the potential effects of enrichments to the benthos at McMurdo Sound, Antarctica. In Proceedings of the Colloquium on Conservation Problems in Antarctica; Allen Press: Lawrence, KS, USA, 1972; pp. 81–96. [Google Scholar]
- Pigott, T. Advances in Meta-Analysis; Springer: New York, NY, USA, 2012. [Google Scholar]
- Jones, C.G.; Lawton, J.H.; Shachak, M. Positive and negative effects of organisms as physical ecosystem engineers. Ecology 1997, 78, 1946–1957. [Google Scholar] [CrossRef]
- Robins, G.; Bates, L.; Pattison, P. Network governance and environmental management: Conflict and cooperation. Public Adm. 2011, 89, 1293–1313. [Google Scholar] [CrossRef]
- Guerrero, A.M.; Mcallister, R.R.; Wilson, K.A. Achieving cross-scale collaboration for large scale conservation initiatives. Conserv. Lett. 2015, 8, 107–117. [Google Scholar] [CrossRef]
- Lubell, M. Collaborative environmental institutions: All talk and no action? J. Policy Anal. Manag. 2004, 23, 549–573. [Google Scholar] [CrossRef]
- Hobbs, R.J. Setting effective and realistic restoration goals: Key directions for research. Restor. Ecol. 2007, 15, 354–357. [Google Scholar] [CrossRef]
- Cook, C.N.; Mascia, M.B.; Schwartz, M.W.; Possingham, H.P.; Fuller, R.A. Achieving conservation science that bridges the knowledge–action boundary. Conserv. Biol. 2013, 27, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Chapron, G.; Arlettaz, R. Conservation: Academics should ‘conserve or perish’. Nature 2008, 451, 127. [Google Scholar] [CrossRef] [PubMed]
- Toomey, A.H.; Knight, A.T.; Barlow, J. Navigating the space between research and implementation in conservation. Conserv. Lett. 2017, 10, 619–625. [Google Scholar] [CrossRef]
- Holl, K.D.; Howarth, R.B. Paying for restoration. Restor. Ecol. 2000, 8, 260–267. [Google Scholar] [CrossRef]
- Prach, K.; Hobbs, R.J. Spontaneous succession versus technical reclamation in the restoration of disturbed sites. Restor. Ecol. 2008, 16, 363–366. [Google Scholar] [CrossRef]
- Holmgren, M.; Scheffer, M. Strong facilitation in mild environments: The stress gradient hypothesis revisited. J. Ecol. 2010, 98, 1269–1275. [Google Scholar] [CrossRef]
- Zedler, J.B. Success: An unclear, subjective descriptor of restoration outcomes. Ecol. Restor. 2007, 25, 162–168. [Google Scholar] [CrossRef]
- Maron, M.; Hobbs, R.J.; Moilanen, A.; Matthews, J.W.; Christie, K.; Gardner, T.A.; Keith, D.A.; Lindenmayer, D.B.; McAlpine, C.A. Faustian bargains? Restoration realities in the context of biodiversity offset policies. Biol. Conserv. 2012, 155, 141–148. [Google Scholar] [CrossRef]
- Grabowski, J.H.; Peterson, C.H. Restoring oyster reefs to recover ecosystem services. Theor. Ecol. Ser. 2007, 4, 281–298. [Google Scholar]
- Palmer, M.A.; Filoso, S. Restoration of ecosystem services for environmental markets. Science 2009, 325, 575–576. [Google Scholar] [CrossRef] [PubMed]
- An, S.; Li, H.; Guan, B.; Zhou, C.; Wang, Z.; Deng, Z.; Zhi, Y.; Liu, Y.; Xu, C.; Fang, S. China’s natural wetlands: Past problems, current status, and future challenges. Ambio 2007, 36, 335–342. [Google Scholar] [CrossRef]
- Silliman, B.R.; Grosholz, E.; Bertness, M.D. Human Impacts on Salt Marshes: A Global Perspective; University of California Press: Orlando, FL, USA, 2009. [Google Scholar]
- Hairston, N.G.; Smith, F.E.; Slobodkin, L.B. Community structure, population control, and competition. Am. Nat. 1960, 94, 421–425. [Google Scholar] [CrossRef]
- Paine, R.T. Food web complexity and species diversity. Am. Nat. 1966, 100, 65–75. [Google Scholar] [CrossRef]
- McNaughton, S. Ecology of a grazing ecosystem: The Serengeti. Ecol. Monographs 1985, 55, 259–294. [Google Scholar] [CrossRef]
- Dayton, P.K. Competition, disturbance, and community organization: The provision and subsequent utilization of space in a rocky intertidal community. Ecological Monogr. 1971, 41, 351–389. [Google Scholar] [CrossRef]
- Platt, W.J. The Colonization and Formation of Equilibrium Plant Species Associations on Badger Disturbances in a Tall-Grass Prairie. Ecol. Monogr. 1975, 45, 285–305. [Google Scholar] [CrossRef]
- Silliman, B.R.; McCoy, M.W.; Angelini, C.; Holt, R.D.; Griffin, J.N.; van de Koppel, J. Consumer fronts, global change, and runaway collapse in ecosystems. Annu. Rev. Ecol. Evol. Syst. 2013, 44, 503–538. [Google Scholar] [CrossRef]
- Kauffman, M.J.; Brodie, J.F.; Jules, E.S. Are wolves saving Yellowstone’s aspen? A landscape-level test of a behaviorally mediated trophic cascade. Ecology 2010, 91, 2742–2755. [Google Scholar] [CrossRef] [PubMed]
- Douvere, F. The importance of marine spatial planning in advancing ecosystem-based sea use management. Mar. Policy 2008, 32, 762–771. [Google Scholar] [CrossRef]
- Scyphers, S.B. Restoring Oyster Reefs along Eroding Coastlines: An Ecological and Socioeconomic Assessment. Ph.D. Thesis, University of South Alabama, Mobile, AL, USA, May 2012. [Google Scholar]
- Gittman, R.K.; Peterson, C.H.; Currin, C.A.; Fodrie, F.J.; Piehler, M.F.; Bruno, J.F. Living shorelines can enhance the nursery role of threatened estuarine habitats. Ecol. Appl. 2016, 26, 249–263. [Google Scholar] [CrossRef] [PubMed]
- Peterson, B.J.; Heck, K.L., Jr. Positive interactions between suspension-feeding bivalves and seagrass—A facultative mutualism. Mar. Ecol. Prog. Ser. 2001, 213, 143–155. [Google Scholar] [CrossRef]
- Miller, J.R.; Hobbs, R.J. Habitat Restoration—Do We Know What We’re Doing? Restor. Ecol. 2007, 15, 382–390. [Google Scholar] [CrossRef]
- Ruiz-Jaen, M.C.; Mitchell Aide, T. Restoration success: How is it being measured? Restor. Ecol. 2005, 13, 569–577. [Google Scholar] [CrossRef]
- Wortley, L.; Hero, J.-M.; Howes, M. Evaluating Ecological Restoration Success: A Review of the Literature. Restor. Ecol. 2013, 21, 537–543. [Google Scholar] [CrossRef]
- Holl, K.D. Restoring tropical forests from the bottom up. Science 2017, 355, 455–456. [Google Scholar] [CrossRef] [PubMed]
- Holl, K.D.; Crone, E.E.; Schultz, C.B. Landscape restoration: Moving from generalities to methodologies. BioScience 2003, 53, 491–502. [Google Scholar] [CrossRef]
- Bodin, Ö.; Crona, B.I. The role of social networks in natural resource governance: What relational patterns make a difference? Glob. Environ. Chang. 2009, 19, 366–374. [Google Scholar] [CrossRef]
- Aronson, J.; Blignaut, J.N.; Milton, S.J.; Le Maitre, D.; Esler, K.J.; Limouzin, A.; Fontaine, C.; De Wit, M.P.; Prinsloo, P.; Van Der Elst, L. Are Socioeconomic Benefits of Restoration Adequately Quantified? A Meta-analysis of Recent Papers (2000–2008) in Restoration Ecology and 12 Other Scientific Journals. Restor. Ecol. 2010, 18, 143–154. [Google Scholar] [CrossRef]
- Fodrie, F.J.; Rodriguez, A.B.; Baillie, C.J.; Brodeur, M.C.; Coleman, S.E.; Gittman, R.K.; Keller, D.A.; Kenworthy, M.D.; Poray, A.K.; Ridge, J.T. Classic paradigms in a novel environment: Inserting food web and productivity lessons from rocky shores and saltmarshes into biogenic reef restoration. J. Appl. Ecol. 2014, 51, 1314–1325. [Google Scholar] [CrossRef]
- Gittman, R.K.; Fodrie, F.J.; Baillie, C.J.; Brodeur, M.C.; Currin, C.A.; Keller, D.A.; Kenworthy, M.D.; Morton, J.P.; Ridge, J.T.; Zhang, Y.S. Living on the Edge: Increasing Patch Size Enhances the Resilience and Community Development of a Restored Salt Marsh. Estuaries Coasts 2017, 1–12. [Google Scholar] [CrossRef]
- Bell, S.S.; Fonseca, M.S.; Motten, L.B. Linking restoration and landscape ecology. Restor. Ecol. 1997, 5, 318–323. [Google Scholar] [CrossRef]
- Bilkovic, D.M.; Mitchell, M.M.; Isdell, R.E.; Schliep, M.; Smyth, A.R. Mutualism between ribbed mussels and cordgrass enhances salt marsh nitrogen removal. Ecosphere 2017, 8, e01795. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.S.; Cioffi, W.R.; Cope, R.; Daleo, P.; Heywood, E.; Hoyt, C.; Smith, C.S.; Silliman, B.R. A Global Synthesis Reveals Gaps in Coastal Habitat Restoration Research. Sustainability 2018, 10, 1040. https://doi.org/10.3390/su10041040
Zhang YS, Cioffi WR, Cope R, Daleo P, Heywood E, Hoyt C, Smith CS, Silliman BR. A Global Synthesis Reveals Gaps in Coastal Habitat Restoration Research. Sustainability. 2018; 10(4):1040. https://doi.org/10.3390/su10041040
Chicago/Turabian StyleZhang, Y. Stacy, William R. Cioffi, Rebecca Cope, Pedro Daleo, Eleanor Heywood, Carmen Hoyt, Carter S. Smith, and Brian. R. Silliman. 2018. "A Global Synthesis Reveals Gaps in Coastal Habitat Restoration Research" Sustainability 10, no. 4: 1040. https://doi.org/10.3390/su10041040
APA StyleZhang, Y. S., Cioffi, W. R., Cope, R., Daleo, P., Heywood, E., Hoyt, C., Smith, C. S., & Silliman, B. R. (2018). A Global Synthesis Reveals Gaps in Coastal Habitat Restoration Research. Sustainability, 10(4), 1040. https://doi.org/10.3390/su10041040




