Early Alterations of QTc in Patients with COVID-19 Treated with Hydroxychloroquine or Chloroquine in Libreville, Gabon
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. General Data
3.1.1. Sociodemographic and Clinical Data
3.1.2. Biological Data
3.1.3. Electrocardiogram
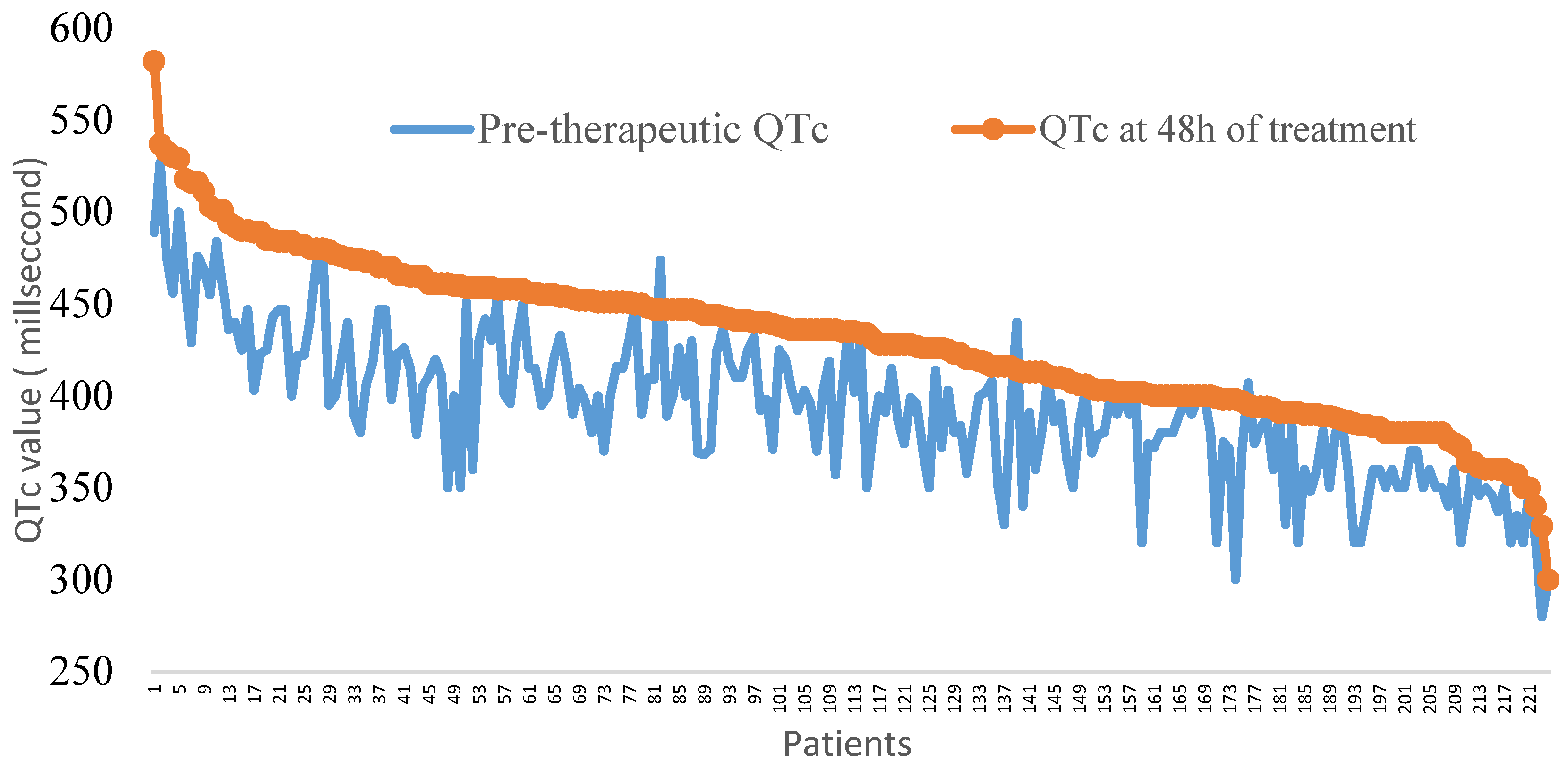
3.2. Electrocardiographic Changes after 48 h of Treatment
3.3. Arrythmia
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Coronavirus Disease 2019 (COVID-19). Weekly Epidemiological Update on COVID-19—25 May 2021. 2020. Available online: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19-25-may-2021 (accessed on 20 July 2021).
- Lawal, Y. Africa’s low COVID-19 mortality rate: A paradox? Int. J. Infect. Dis. 2021, 102, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Jean, S.S.; Lee, P.I.; Hsueh, P.R. Treatment options for COVID-19: The reality and challenges. J. Microbiol. Immunol. Infect. 2020, 53, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Bessière, F.; Roccia, H.; Delinière, A.; Charrière, R.; Chevalier, P.; Argaud, L.; Cour, M. Assessment of QT Intervals in a Case Series of Patients With Coronavirus Disease 2019 (COVID-19) Infection Treated With Hydroxychloroquine Alone or in Combination With Azithromycin in an Intensive Care Unit [published online ahead of print, 1 May 2020]. JAMA Cardiol. 2020, 5, 1067–1069. [Google Scholar] [CrossRef] [PubMed]
- Chorin, E.; Wadhwani, L.; Magnani, S.; Dai, M.; Shulman, E.; Nadeau-Routhier, C.; Knotts, R.; Bar-Cohen, R.; Kogan, E.; Barbhaiya, C.; et al. QT interval prolongation and torsade de pointes in patients with COVID-19 treated with hydroxychloroquine/azithromycin [published online ahead of print, 12 May 2020]. Heart Rhythm. 2020, 17, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Eftekhar, S.P.; Kazemi, S.; Barary, M.; Javanian, M.; Ebrahimpour, S.; Ziaei, N. Effect of Hydroxychloroquine and Azithromycin on QT Interval Prolongation and Other Cardiac Arrhythmias in COVID-19 Confirmed Patients. Cardiovasc. Ther. 2021, 2021, 6683098. [Google Scholar] [CrossRef] [PubMed]
- Saleh, M.; Gabriels, J.; Chang, D.; Kim, B.S.; Mansoor, A.; Mahmood, E.; Makker, P.; Ismail, H.; Goldner, B.; Willner, J.; et al. Effect of Chloroquine, Hydroxychloroquine, and Azithromycin on the Corrected QT Interval in Patients With SARS-CoV-2 Infection. Circ. Arrhythmia Electrophysiol. 2020, 13, e008662. [Google Scholar] [CrossRef]
- Yao, X.; Ye, F.; Zhang, M.; Cui, C.; Huang, B.; Niu, P.; Liu, D. In Vitro Antiviral Activity and Projection of Optimized 345 Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory 346 Syndrome Coronavirus 2 (SARS-CoV-2). Clin. Infect. Dis. 2020, 71, 732–739. [Google Scholar] [CrossRef] [Green Version]
- Mahévas, M.; Tran, V.T.; Roumier, M.; Chabrol, A.; Paule, R.; Guillaud, C.; Costedoat-Chalumeau, N. No evidence of clinical efficacy of hydroxychloroquine in patients hospitalized for COVID-19 infection with oxygen requirement: Results of a study using routinely collected data to emulate a target trial. medRxiv 2020. medRxiv:2020.04.10.20060699. [Google Scholar]
- Gautret, P.; Lagier, J.C.; Parola, P.; Hoang, V.T.; Meddeb, L.; Mailhe, M.; Doudier, B.; Courjon, J.; Giordanengo, V.; Vieira, V.E.; et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial [published online ahead of print, 2020 Mar 20]. Int. J. Antimicrob. Agents 2020, 56, 105949. [Google Scholar] [CrossRef]
- National Directives for the Medical Management of Coronavirus Disease 2019 (COVID-19); Scientific Committee COVID-19: Libreville, Gabon, 2020.
- Thomas, S.H.; Behr, E.R. Pharmacological treatment of acquired QT prolongation and torsades de pointes. Br. J. Clin. Pharmacol. 2016, 81, 420–427. [Google Scholar] [CrossRef]
- Lester, R.M.; Paglialunga, S.; Johnson, I.A. QT Assessment in Early Drug Development: The Long and the Short of It. Int. J. Mol. Sci. 2019, 20, 1324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Obesity: Preventing and Managing the Global Épidémic; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Haeusler, I.L.; Chan, X.H.S.; Guerin, P.J.; White, N.J. The arrhythmogenic cardiotoxicity of the quinoline and structurally related antimalarial drugs: A systematic review. BMC Med. 2018, 16, 200. [Google Scholar] [CrossRef] [PubMed]
- Abena, P.M.; Decloedt, E.H.; Bottieau, E.; Suleman, F.; Adejumo, P.; Sam-Agudu, N.A.; Muyembe TamFum, J.-J.; Seydi, M.; Eholie, S.P.; Mills, E.J.; et al. Chloroquine and Hydroxychloroquine for the Prevention or Treatment of COVID-19 in Africa: Caution for Inappropriate Off-label Use in Healthcare Settings. Am. J. Trop. Med. Hyg. 2020, 102, 1184–1188. [Google Scholar] [CrossRef] [PubMed]
- Sapp, J.L.; Alqarawi, W.; MacIntyre, C.J.; Tadros, R.; Steinberg, C.; Roberts, J.D.; Laksman, Z.; Healey, J.S.; Krahn, A.D. Guidance on Minimizing Risk of Drug-Induced Ventricular Arrhythmia During Treatment of COVID-19: A Statement from the Canadian Heart Rhythm Society. Can. J. Cardiol. 2020, 36, 948–951. [Google Scholar] [CrossRef] [PubMed]
- Yancy, C.W. COVID-19 and African Americans [published online ahead of print, 15 April 2020]. JAMA 2020, 323, 1891. [Google Scholar] [CrossRef] [Green Version]
- Giudicessi, J.R.; Roden, D.M.; Wilde, A.A.M.; Ackerman, M.J. Genetic susceptibility for COVID-19-associated sudden cardiac death in African Americans [published online ahead of print, 5 May 2020]. Heart Rhythm. 2020, 17, 1487–1492. [Google Scholar] [CrossRef]
- Ganatra, S.; Dani, S.S.; Shah, S.; Asnani, A.; Neilan, T.G.; Lenihan, D.; Ky, B.; Barac, A.; Hayek, S.S.; Leja, M.; et al. Management of Cardiovascular Disease During Coronavirus Disease (COVID-19) Pandemic [published online ahead of print, 28 May 2020]. Trends Cardiovasc. Med. 2020, 30, 315–325. [Google Scholar] [CrossRef]
- Mercuro, N.J.; Yen, C.F.; Shim, D.J.; Maher, T.R.; McCoy, C.M.; Zimetbaum, P.J.; Gold, H.S. Risk of QT Interval Prolongation Associated With Use of Hydroxychloroquine With or Without Concomitant Azithromycin Among Hospitalized Patients Testing Positive for Coronavirus Disease 2019 (COVID-19) [published online ahead of print, 1 May 2020]. JAMA Cardiol. 2020, 5, 1036–1041. [Google Scholar] [CrossRef]
- Hsia, B.; Greige, N.; Quiroz, J.A.; Khokhar, A.S.; Daily, J.; Di Biase, L.; Ferrick, K.J.; Fisher, J.D.; Krumerman, A. QT prolongation in a diverse, urban population of COVID-19 patients treated with hydroxychloroquine, chloroquine, or azithromycin [published online ahead of print, 11 July 2020]. J. Interv. Card. Electrophysiol. 2020, 59, 337–345. [Google Scholar] [CrossRef]
- Duckheim, M.; Schreieck, J. COVID-19 and Cardiac Arrhythmias. Hamostaseologie 2021, 41, 372–378. [Google Scholar] [CrossRef]
- Gumilang, R.A.; Siswanto Anggraeni, V.Y.; Trisnawati, I.; Budiono, E.; Hartopo, A.B. QT interval and repolarization dispersion changes during the administration of hydroxychloroquine/chloroquine with/without azithromycin in early COVID 19 pandemic: A prospective observational study from two academic hospitals in Indonesia. J Arrhythm. 2021, 37, 1184–1195. [Google Scholar] [CrossRef] [PubMed]
- Farmakis, I.T.; Minopoulou, I.; Giannakoulas, G.; Boutou, A. Cardiotoxicity of azithromycin in COVID-19: An overall proportion meta-analysis. Adv. Respir. Med. 2022; Online ahead of print. [Google Scholar] [CrossRef]
- Tikkanen, J.T.; Kentta, T.; Porthan, K.; Anttonen, O.; Eranti, A.; Aro, A.L.; Kerola, T.; Rissanen, H.A.; Knekt, P.; Heliövaara, M.; et al. The Risk of Sudden Cardiac Death Associated with QRS, QTc and JTc intervals in the General Population Revision #4. Heart Rhythm. 2022; Online ahead of print. [Google Scholar] [CrossRef]
- Diaz-Arocutipa, C.; Brañez-Condorena, A.; Hernandez, A.V. QTc prolongation in COVID-19 patients treated with hydroxychloroquine, chloroquine, azithromycin, or lopinavir/ritonavir: A systematic review and meta-analysis. Pharmacoepidemiol. Drug Saf. 2021, 30, 694–706. [Google Scholar] [CrossRef] [PubMed]
- Tisdale, J.E.; Jaynes, H.A.; Kingery, J.R.; Mourad, N.A.; Trujillo, T.N.; Overholser, B.R.; Kovacs, R.J. Development and validation of a risk score to predict QT interval prolongation in hospitalized patients. Circ. Cardiovasc. Qual. Outcomes 2013, 6, 479–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hulot, J.S. COVID-19 in patients with cardiovascular diseases. Arch. Cardiovasc. Dis. 2020, 113, 225–226. [Google Scholar] [CrossRef]
- Chen, D., Jr.; Li, X.; Song, Q.; Hu, C., Jr.; Su, F.; Dai, J. Hypokalemia and clinical implications in patients with coronavirus disease 2019 (COVID-19). MedRxiv 2020. medRxiv:2020.02.27.20028530. [Google Scholar]
- Malviya, A. Ventricular arrhythmia risk due to chloroquine/hydroxychloroquine treatment for COVID-19: Should it be given. Indian Heart J. 2020, 72, 131–132. [Google Scholar] [CrossRef]
| Total n (224) | Patients Treated with HCQ n (102) | Patients Treated with CQ (n 122) | p | |
|---|---|---|---|---|
| Age (median, quartile) in years | 48 (38–56) | 49 (39–57) | 47 (37–55) | 0.25 |
| Sex (n, %) | ||||
| Men | 141 (62.9) | 63 (61.8) | 78 (63.9) | 0.73 |
| Women | 83 (37.0) | 39 (38.2) | 44 (36.0) | 0.73 |
| FC beats/minute (median, interquartile) | 80.0 (69.5–90.0) | 79.5 (69.0–90.0) | 80.0 (70.0–90.0) | 0.43 |
| Risk factors (n, %) | ||||
| High blood pressure | 74 (33.0) | 35 (34.3) | 39 (32.0) | 0.71 |
| Diabetes | 43 (19.2) | 22 (21.6) | 21 (17.2) | 0.41 |
| Obesity | 81 (36.2) | 31 (30.3) | 50 (41.0) | 0.10 |
| Tobacco consumption | 8 (3.6) | 3 (2.9) | 5 (4.1) | 0.64 |
| Comorbidities (n, %) | ||||
| Heart failure | 6 (2.7) | 1(1.0) | 5 (4.1) | 0.15 |
| Clinical forms (n, %) | ||||
| Asymptomatic | 41 (18.3) | 18 (17.6) | 23 (18.8) | 0.81 |
| Moderate | 141 (62.9) | 69 (67.6) | 72 (59.0) | 0.12 |
| Severe | 18 (8.0) | 6 (5.8) | 12 (9.8) | 0.27 |
| Critical | 24 (10.7) | 9 (8.8) | 15 (12.3) | 0.40 |
| Baseline ECG abnormalities (n, %) | ||||
| AF | 4(1.8) | 4(3.9) | 0 (0.0) | 0.02 |
| Right bundle branch block | 3(1.3) | 1 (1.0) | 2 (1.6%) | 0.64 |
| Left bundle branch block | 3 (1.3) | 1 (1.0) | 2 (1.6%) | 0.64 |
| Repolarization disorders | 24 (10.7) | 11 (10.8) | 13 (10.6) | 0.97 |
| First degree atrioventricular block | 10 (4.5) | 3 (2.9) | 7 (5.7) | 0.31 |
| Left ventricular hypertrophy | 53 (23.7) | 28 (27.4) | 25 (20.5) | 0.22 |
| Left atrial hypertrophy | 54 (24.1) | |||
| Necrosis sequel | 6 (2.7) | 4 (3.9) | 2 (1.6) | 0.23 |
| Long QTc | 9 (4.0) | 6 (5.9) | 3 (2.4) | 0.40 |
| Kalaemia (median, quartile) mEq/L | 3.8 (3.4–4.2) | 3.8 (3.4–4.0) | 3.8 (3.5–4.2) | 0.49 |
| Treatment | ||||
| Amiodaron | 4 (1.8) | 4 (3.9) | 0 (0.0) | 0.02 |
| Digoxin | 0 | - | - | - |
| Total n (224) | Patients Treated with Hydroxychloroquine n (102) | Patients Treated with Chloroquine; n (122) | p | |
|---|---|---|---|---|
| Arrhythmias | ||||
| Isolated ventricular extrasystoles (n, %) | 4 (1.8) | 0 (0.0) | 4 (3.3) | 0.06 |
| Torsade de pointe (n, %) | 0 (0.0) | 0 (0.0) | 0 (0.0) | - |
| Atrial fibrillation. (n, %) | 1 (0.0) | 0 (0.0) | 0 (0.0) | - |
| QTc alterations | ||||
| QTc (ms) (median, interquartile) | 435 (400–458) | 424 (391–454) | 438 (402–460) | 0.02 |
| QTc interval prolongation at 48 h (ms) (median, interquartile) | 33.0 (18.5–55.0) | 30.0 (19.0–53.0) | 38.5 (17.0–56) | 0.54 |
| QT prolongation > 60 ms at 48 h (n, %) | 41 (18.3) | 16 (15.7) | 25 (20.5) | 0.35 |
| QTc exceeding 500 ms at 48 h (n, %) | 12 (5.3) | 1 (1.0) | 11 (9.0) | <0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ayo Bivigou, E.; Manomba Boulingui, C.; Bouraima, A.; Allognon, C.; Akagha Konde, C.; Atsame, G.; Kinga, A.; Boguikouma, J.B.; Kouna Ndouongo, P.; Bouyou Akotet, M.K. Early Alterations of QTc in Patients with COVID-19 Treated with Hydroxychloroquine or Chloroquine in Libreville, Gabon. Clin. Pract. 2022, 12, 482-490. https://doi.org/10.3390/clinpract12040052
Ayo Bivigou E, Manomba Boulingui C, Bouraima A, Allognon C, Akagha Konde C, Atsame G, Kinga A, Boguikouma JB, Kouna Ndouongo P, Bouyou Akotet MK. Early Alterations of QTc in Patients with COVID-19 Treated with Hydroxychloroquine or Chloroquine in Libreville, Gabon. Clinics and Practice. 2022; 12(4):482-490. https://doi.org/10.3390/clinpract12040052
Chicago/Turabian StyleAyo Bivigou, Elsa, Charlene Manomba Boulingui, Aridath Bouraima, Christian Allognon, Christelle Akagha Konde, Gabrielle Atsame, Armel Kinga, Jean B. Boguikouma, Philomène Kouna Ndouongo, and Marielle K. Bouyou Akotet. 2022. "Early Alterations of QTc in Patients with COVID-19 Treated with Hydroxychloroquine or Chloroquine in Libreville, Gabon" Clinics and Practice 12, no. 4: 482-490. https://doi.org/10.3390/clinpract12040052
APA StyleAyo Bivigou, E., Manomba Boulingui, C., Bouraima, A., Allognon, C., Akagha Konde, C., Atsame, G., Kinga, A., Boguikouma, J. B., Kouna Ndouongo, P., & Bouyou Akotet, M. K. (2022). Early Alterations of QTc in Patients with COVID-19 Treated with Hydroxychloroquine or Chloroquine in Libreville, Gabon. Clinics and Practice, 12(4), 482-490. https://doi.org/10.3390/clinpract12040052






