Acute Hepatitis in a Yemeni Immigrant Associated with Khat: A “Biological Amphetamine” Carried in Cultures
Abstract
1. Introduction
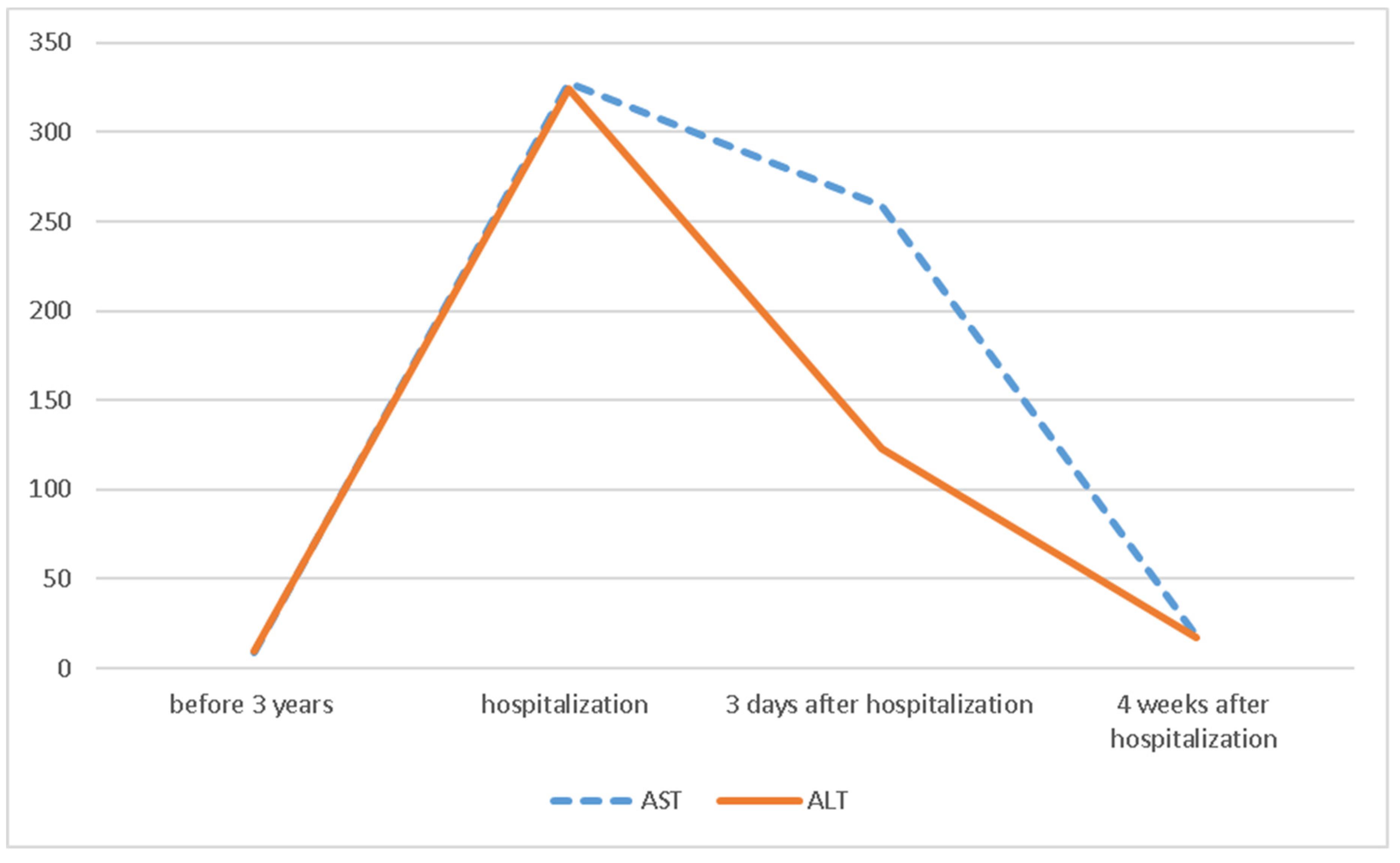
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Ryder, S.D.; Beckingham, I.J. Acute hepatitis. BMJ 2001, 322, 151–153. [Google Scholar] [CrossRef] [PubMed]
- Bunchorntavakul, C.; Reddy, K.R. Review article: Herbal and dietary supplement hepatotoxicity. Aliment. Pharmacol. Ther. 2013, 37, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Stirnimann, G.; Kessebohm, K.; Lauterburg, B. Liver injury caused by drugs: An update. Swiss Med. Wkly. 2010, 140. [Google Scholar] [CrossRef]
- Andrade, R.J.; Medina-Caliz, I.; Gonzalez-Jimenez, A.; Garcia-Cortes, M.; Lucena, M.I. Hepatic Damage by Natural Remedies. In Seminars in Liver Disease; Thieme Medical Publishers: New York, NY, USA, 2018; Volume 38, pp. 21–40. [Google Scholar] [CrossRef]
- Teni, F.S.; Surur, A.S.; Hailemariam, A.; Aye, A.; Mitiku, G.; Gurmu, A.E.; Tessema, B. Prevalence, Reasons, and Perceived Effects of Khat Chewing Among Students of a College in Gondar Town, Northwestern Ethiopia: A Cross-Sectional Study. Ann. Med. Health Sci. Res. 2015, 5, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.L. Attitudes to khat use within the Somali community in England. Drugs Educ. Prev. Policy 2008, 15, 37–53. [Google Scholar] [CrossRef]
- Thomas, S.; Williams, T. Khat (Catha edulis): A systematic review of evidence and literature pertaining to its harms to UK users and society. Drug Sci. Policy Law 2013, 1. [Google Scholar] [CrossRef]
- Chapman, M.H.; Kajihara, M.; Borges, G.; O’Beirne, J.; Patch, D.; Dhillon, A.P.; Crozier, A.; Morgan, M.Y. Severe, Acute Liver Injury and Khat Leaves. N. Engl. J. Med. 2010, 362, 1642–1644. [Google Scholar] [CrossRef] [PubMed]
- Getahun, W.; Gedif, T.; Tesfaye, F. Regular Khat (Catha edulis) chewing is associated with elevated diastolic blood pressure among adults in Butajira, Ethiopia: A comparative study. BMC Public Health 2010, 10, 390. [Google Scholar] [CrossRef] [PubMed]
- Abid, M.D.; Chen, J.; Xiang, M.; Zhou, J.; Chen, X.; Gong, F. Khat (Catha edulis) generates reactive oxygen species and promotes hepatic cell apoptosis via MAPK activation. Int. J. Mol. Med. 2013, 32, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Al-Mamary, M.; Al-Habori, M.; Al-Aghbari, A.M.; Baker, M.M. Investigation into the toxicological effects of Catha edulis leaves: A short term study in animals. Phytother. Res. 2002, 16, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Al-Habori, M.; Al-Aghbari, A.; Al-Mamary, M.; Baker, M. Toxicological evaluation of Catha edulis leaves: A long term feeding experiment in animals. J. Ethnopharmacol. 2002, 83, 209–217. [Google Scholar] [CrossRef]
- Riyaz, S.; Imran, M.; Gleeson, D.; Karajeh, M.A. Khat (Catha Edulis) as a possible cause of autoimmune hepatitis. World J. Hepatol. 2014, 6, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Stuyt, R.J.; Willems, S.M.; Wagtmans, M.J.; van Hoek, B. Chewing khat and chronic liver disease. Liver Int. Off. J. Int. Assoc. Study Liver 2011, 31, 434–436. [Google Scholar] [CrossRef] [PubMed]
- Daba, D.; Hymete, A.; Bekhit, A.A.; Mohamed, A.M.; Bekhit Ael, D. Multi residue analysis of pesticides in wheat and khat collected from different regions of Ethiopia. Bull. Environ. Contam. Toxicol. 2011, 86, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Ben Rhouma, K.; Tebourbi, O.; Sakly, M. Acute hepatotoxicity of DDT: Effect on glucocorticoid receptors and serum transcortin. Indian J. Exp. Biol. 2000, 38, 452–456. [Google Scholar]
- Douglas, H.; Boyle, M.; Lintzeris, N. The health impacts of khat: A qualitative study among Somali-Australians. Med. J. Aust. 2011, 195, 666–669. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food & Drug Administration. Import Alert 66-23. Available online: https://www.accessdata.fda.gov/cms_ia/importalert_180.html (accessed on 18 March 2011).
- Griffiths, P.; Lopez, D.; Sedefov, R.; Gallegos, A.; Hughes, B.; Noor, A.; Royuela, L. Khat use and monitoring drug use in Europe: The current situation and issues for the future. J. Ethnopharmacol. 2010, 132, 578–583. [Google Scholar] [CrossRef][Green Version]
- Orlien, S.M.S.; Berhe, N.B.; Morgan, M.Y.; Johannessen, A. Khat-related liver disease in sub-Saharan Africa: Neglected, yet important. Lancet Glob. Health 2019, 7, e310. [Google Scholar] [CrossRef]


| At Admission | Follow-Up 3 Weeks Later | |
|---|---|---|
| HepA IgG | Positive | |
| Hep B surface Ab | Positive | |
| HCV Ab | Positive | |
| HCV RNA | Undetectable | |
| Hep E IgG | Undetectable | |
| Hep E IgM | Undetectable | |
| Alpha-1 Antitrypsin | Normal | |
| Anti-nuclear antibody | Negative | |
| Antimitochondrial antibody | Negative | |
| Celiac panel | Negative | |
| Ceruloplasmin mg/dL | Normal | |
| Acetaminophen | Not detectable | |
| Smooth muscle antibody | Negative | |
| Transferrin saturation | 40% | |
| Ferritin ng/mL | 195 | |
| Liver kidney microsomal | Negative | |
| Immunoglobulin G level | 946 mg/dL | |
| Eosinophils (percent, count) | 8.7%, 0.4 K/uL | |
| International Normalized Rato (INR) | 1.0 | |
| Albumin g/dL | 4.9 | 4.7 |
| ALT units/L | 328 | 18 |
| AST units/L | 324 | 17 |
| Alkaline phosphatase units/L | 78 | 47 |
| Total Bilirubin md/dL | 2.0 | 0.7 |
| Direct Bilirubin md/dL | 0.7 | 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patel, H.; Kumar, K.; Essrani, R.K.; Niazi, M.; Makker, J.; Nayudu, S.K. Acute Hepatitis in a Yemeni Immigrant Associated with Khat: A “Biological Amphetamine” Carried in Cultures. Clin. Pract. 2021, 11, 167-173. https://doi.org/10.3390/clinpract11010023
Patel H, Kumar K, Essrani RK, Niazi M, Makker J, Nayudu SK. Acute Hepatitis in a Yemeni Immigrant Associated with Khat: A “Biological Amphetamine” Carried in Cultures. Clinics and Practice. 2021; 11(1):167-173. https://doi.org/10.3390/clinpract11010023
Chicago/Turabian StylePatel, Harish, Kishore Kumar, Rajesh Kumar Essrani, Masooma Niazi, Jasbir Makker, and Suresh Kumar Nayudu. 2021. "Acute Hepatitis in a Yemeni Immigrant Associated with Khat: A “Biological Amphetamine” Carried in Cultures" Clinics and Practice 11, no. 1: 167-173. https://doi.org/10.3390/clinpract11010023
APA StylePatel, H., Kumar, K., Essrani, R. K., Niazi, M., Makker, J., & Nayudu, S. K. (2021). Acute Hepatitis in a Yemeni Immigrant Associated with Khat: A “Biological Amphetamine” Carried in Cultures. Clinics and Practice, 11(1), 167-173. https://doi.org/10.3390/clinpract11010023




