Battery Lifespan of an Implantable Middle Ear Device
Abstract
:1. Introduction
2. Materials and Methods
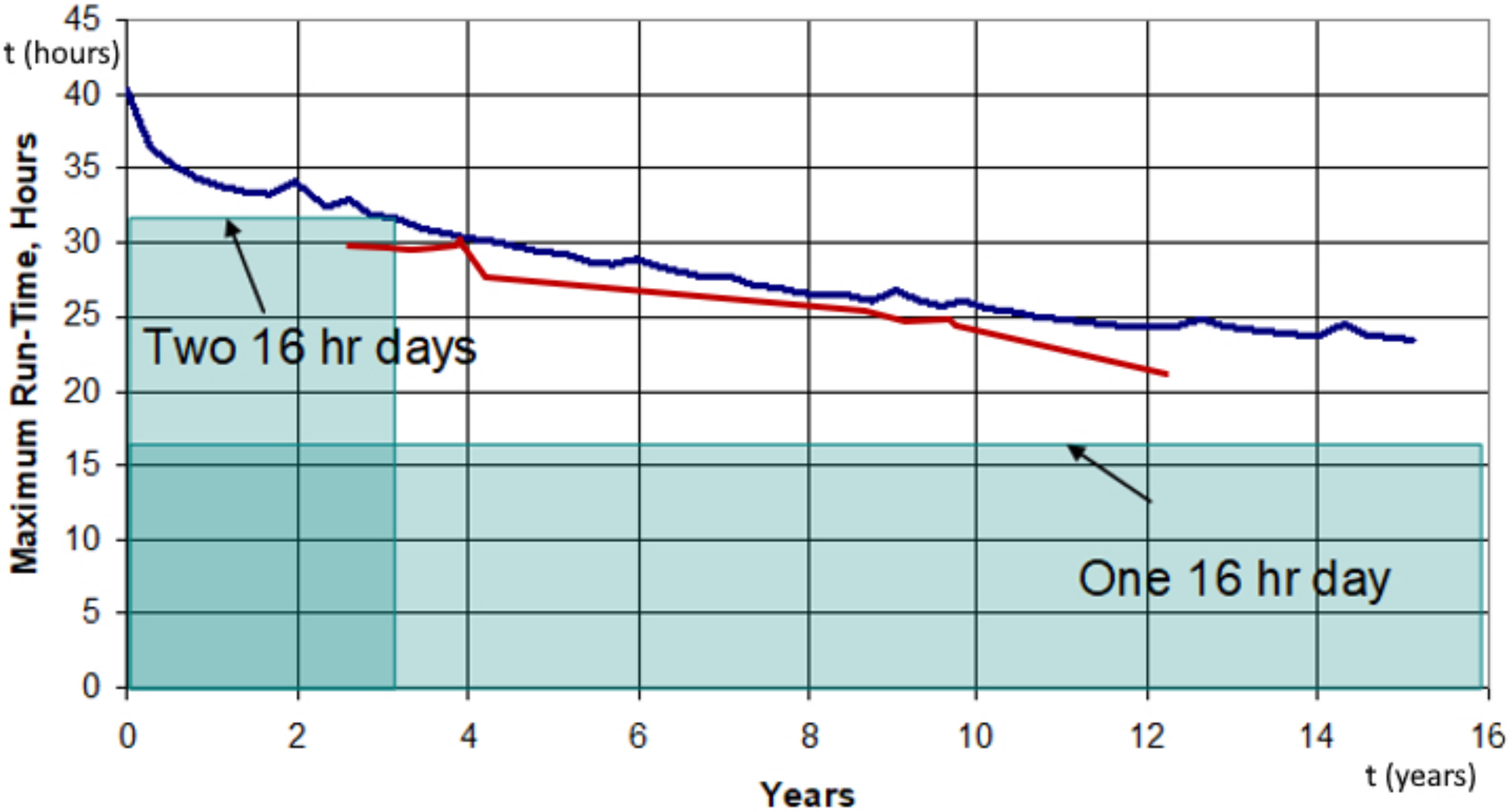
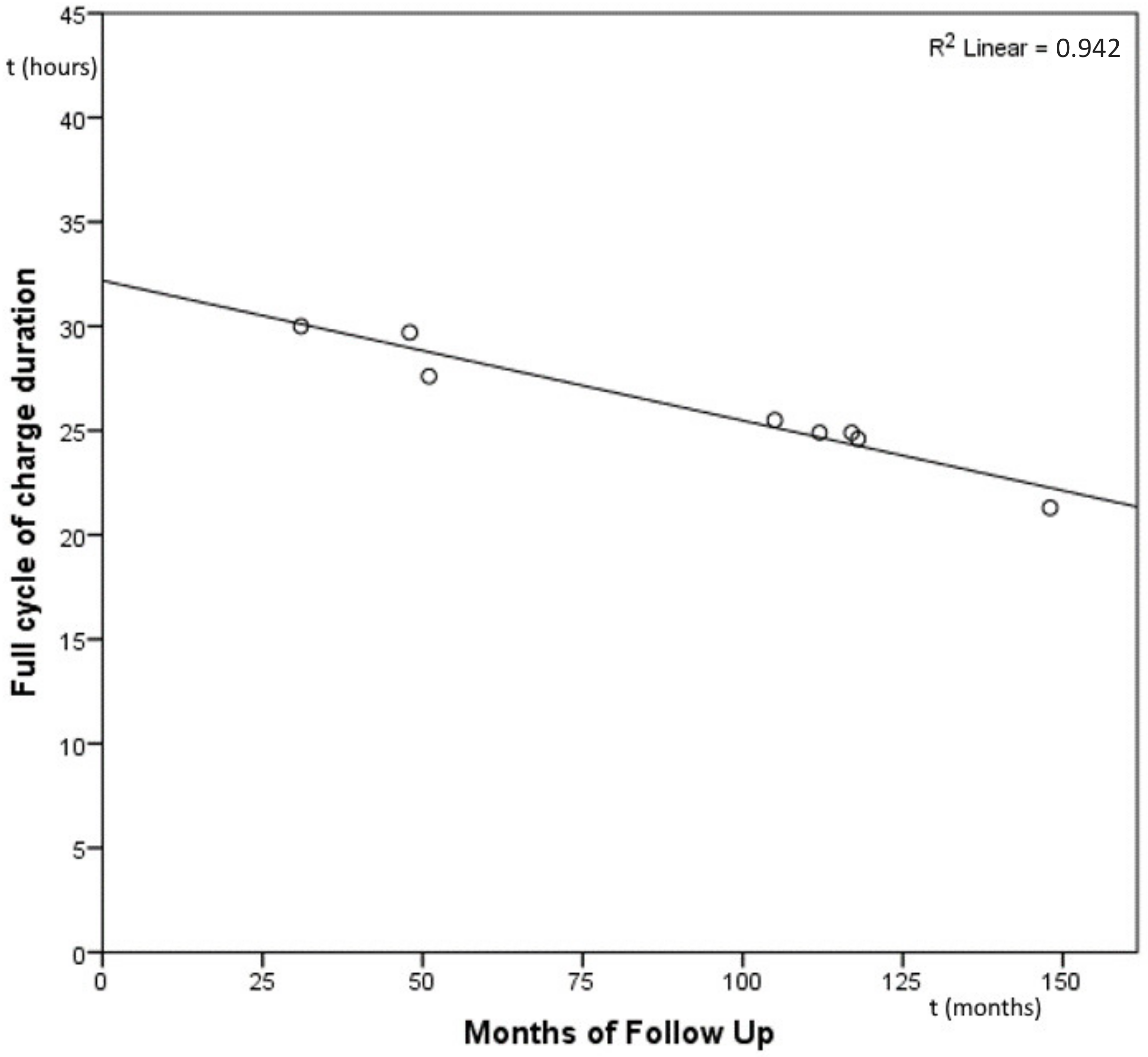
3. Results
- (a)
- Explantation for extrusion after prosthesis infection in two cases;
- (b)
- Explantation for patient’s wish in one case;
- (c)
- A stopping of prosthesis use, due to hearing loss progression over time and subsequent lack of benefit from the amplification through the implanted device in three cases;
- (d)
- Moving to other centres for follow up in two cases (one case with bilateral implant);
- (e)
- Death of the recipient in one case;
- (f)
- Unknown reasons in two cases.
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CE | Conformité Européene |
| GBI | Glasgow Benefit Inventory |
| MET | Middle ear transducer |
| MHL | Mixed hearing loss |
| SNHL | Sensorineural hearing loss |
| TLA | Transducer loading assistant |
References
- Butler, C.L.; Thavaneswaran, P.; Lee, I.H. Efficacy of the Active Middle-Ear Implant in Patients with Sensorineural Hearing Loss. J. Laryngol. Otol. 2013, 127 (Suppl S2), S8–S16. [Google Scholar] [CrossRef] [PubMed]
- Bruschini, L.; Berrettini, S.; Forli, F.; Murri, A.; Cuda, D. The Carina© Middle Ear Implant: Surgical and Functional Outcomes. Eur. Arch. Otorhinolaryngol. 2016, 273, 3631–3640. [Google Scholar] [CrossRef]
- Klein, K.; Nardelli, A.; Stafinski, T. A Systematic Review of the Safety and Effectiveness of Fully Implantable Middle Ear Hearing Devices: The Carina and Esteem Systems. Otol. Neurotol. 2012, 33, 916–921. [Google Scholar] [CrossRef]
- Siegert, R.; Mattheis, S.; Kasic, J. Fully Implantable Hearing Aids in Patients with Congenital Auricular Atresia. Laryngoscope 2007, 117, 336–340. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, H.A.; Atkins, J.S.; Horlbeck, D.; Hoffer, M.E.; Balough, B.; Arigo, J.V.; Alexiades, G.; Garvis, W.U.S. Phase I Preliminary Results of Use of the Otologics MET Fully-Implantable Ossicular Stimulator. Otolaryngol. Head Neck Surg. 2007, 137, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, H.A.; Niparko, J.K.; Slattery, W.H.; Neely, J.G.; Fredrickson, J.M. Otologics Middle Ear Transducer Ossicular Stimulator: Performance Results with Varying Degrees of Sensorineural Hearing Loss. Acta Otolaryngol. 2004, 124, 391–394. [Google Scholar] [CrossRef]
- Kasic, J.F.; Fredrickson, J.M. The Otologics MET Ossicular Stimulator. Otolaryngol. Clin. N. Am. 2001, 34, 501–513. [Google Scholar] [CrossRef]
- Wagner, F.; Todt, I.; Wagner, J.; Ernst, A. Indications and Candidacy for Active Middle Ear Implants. Adv. Otorhinolaryngol. 2010, 69, 20–26. [Google Scholar] [CrossRef]
- Tringali, S.; Pergola, N.; Ferber-Viart, C.; Truy, E.; Berger, P.; Dubreuil, C. Fully Implantable Hearing Device as a New Treatment of Conductive Hearing Loss in Franceschetti Syndrome. Int. J. Pediatr. Otorhinolaryngol. 2008, 72, 513–517. [Google Scholar] [CrossRef]
- Didczuneit-Sandhop, B.; Langer, J. Use of the Carina active middle ear implant in otosclerosis patients. HNO 2021, 69, 828–834. [Google Scholar] [CrossRef]
- Bruschini, L.; Forli, F.; Giannarelli, M.; Bruschini, P.; Berrettini, S. Exclusive Transcanal Surgical Approach for Vibrant Soundbridge Implantation: Surgical and Functional Results. Otol. Neurotol. 2009, 30, 950–955. [Google Scholar] [CrossRef]
- Bruschini, L.; Forli, F.; Passetti, S.; Bruschini, P.; Berrettini, S. Fully Implantable Otologics MET Carina(TM) Device for the Treatment of Sensorineural and Mixed Hearing Loss: Audio-Otological Results. Acta Otolaryngol. 2010, 130, 1147–1153. [Google Scholar] [CrossRef] [PubMed]
- Carina® Middle Ear Implants. Available online: https://www.cochlear.com/mdr/en/home/products-and-accessories/carina-middle-ear-implants (accessed on 6 April 2022).
- Devèze, A. Implantable Hearing Aids. In Recent Advances in Otolaryngology Head and Neck Surgery; Jaypee Brothers Medical Publishers: New Delhi, India, 2014; Volume 3, ISBN 978-93-5152-145-7. [Google Scholar]
- Hendry, J.; Chin, A.; Swan, I.R.C.; Akeroyd, M.A.; Browning, G.G. The Glasgow Benefit Inventory: A Systematic Review of the Use and Value of an Otorhinolaryngological Generic Patient-Recorded Outcome Measure. Clin. Otolaryngol. 2016, 41, 259–275. [Google Scholar] [CrossRef]
- Forli, F.; Lazzerini, F.; Bruschini, L.; Danti, S.; Berrettini, S. Recent and Future Developments in Cochlear Implant Technology: Review of the Literature. Otorhinolaryngology 2021, 71, 196–207. [Google Scholar] [CrossRef]
- Roche, J.P.; Hansen, M.R. On the Horizon: Cochlear Implant Technology. Otolaryngol. Clin. N. Am. 2015, 48, 1097–1116. [Google Scholar] [CrossRef]
- Debeaupte, M.; Decullier, E.; Tringali, S.; Devèze, A.; Mom, T.; Darrouzet, V.; Truy, E. Evolution of the Reliability of the Fully Implantable Middle Ear Transducer over Successive Generations. Otol. Neurotol. 2015, 36, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.; Deveze, A.; Richard, C.; Lefebvre, P.P.; Decat, M.; Ibañez, L.G.; Truy, E.; Mom, T.; Lavieille, J.-P.; Magnan, J.; et al. European Results with Totally Implantable Carina Placed on the Round Window: 2-Year Follow-Up. Otol. Neurotol. 2009, 30, 1196–1203. [Google Scholar] [CrossRef]
- Jones, S.E.M.; Roplekar-Bance, R.; Green, R.; Rae, C.; Ferguson, A.; Spielmann, P.M. Patient-Reported Outcomes in Middle Ear and Active Transcutaneous Bone Conduction Hearing Implants. J. Int. Adv. Otol. 2021, 17, 405–411. [Google Scholar] [CrossRef]
- Barbara, M.; Covelli, E.; Filippi, C.; Volpini, L.; Monini, S. Revision of Active Middle Ear Implants (AMEI): Causes, Surgical Issues and Rehabilitative Transition at a Single Implanting Center. Eur. Arch. Otorhinolaryngol. 2021, 278, 4289–4294. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, P.P.; Gisbert, J.; Cuda, D.; Tringali, S.; Deveze, A. A Retrospective Multicentre Cohort Review of Patient Characteristics and Surgical Aspects versus the Long-Term Outcomes for Recipients of a Fully Implantable Active Middle Ear Implant. Audiol. Neurootol. 2016, 21, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Cebulla, M.; Hagen, R.; Rak, K.; Geiger, U. Intraoperative Determination of Coupling Efficiency of Carina® Middle Ear Implant by Means of Auditory Evoked Potentials. Int. J. Audiol. 2020, 59, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Carlson, M.L.; Driscoll, C.L.W.; Gifford, R.H.; McMenomey, S.O. Cochlear Implantation: Current and Future Device Options. Otolaryngol. Clin. N. Am. 2012, 45, 221–248. [Google Scholar] [CrossRef]
- Cohen, N. The Totally Implantable Cochlear Implant. Ear Hear. 2007, 28, 100S–101S. [Google Scholar] [CrossRef] [PubMed]
- Briggs, R.J.S.; Eder, H.C.; Seligman, P.M.; Cowan, R.S.C.; Plant, K.L.; Dalton, J.; Money, D.K.; Patrick, J.F. Initial Clinical Experience with a Totally Implantable Cochlear Implant Research Device. Otol. Neurotol. 2008, 29, 114–119. [Google Scholar] [CrossRef]
- Hendricks, C.; Williard, N.; Mathew, S.; Pecht, M. A Failure Modes, Mechanisms, and Effects Analysis (FMMEA) of Lithium-Ion Batteries. J. Power Sources 2015, 297, 113–120. [Google Scholar] [CrossRef]
- Waldmann, T.; Wilka, M.; Kasper, M.; Fleischhammer, M.; Wohlfahrt-Mehrens, M. Temperature Dependent Ageing Mechanisms in Lithium-Ion Batteries—A Post-Mortem Study. J. Power Sources 2014, 262, 129–135. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruschini, L.; Forli, F.; Fiacchini, G.; Canelli, R.; Berrettini, S.; Lazzerini, F. Battery Lifespan of an Implantable Middle Ear Device. Audiol. Res. 2022, 12, 485-492. https://doi.org/10.3390/audiolres12050049
Bruschini L, Forli F, Fiacchini G, Canelli R, Berrettini S, Lazzerini F. Battery Lifespan of an Implantable Middle Ear Device. Audiology Research. 2022; 12(5):485-492. https://doi.org/10.3390/audiolres12050049
Chicago/Turabian StyleBruschini, Luca, Francesca Forli, Giacomo Fiacchini, Rachele Canelli, Stefano Berrettini, and Francesco Lazzerini. 2022. "Battery Lifespan of an Implantable Middle Ear Device" Audiology Research 12, no. 5: 485-492. https://doi.org/10.3390/audiolres12050049
APA StyleBruschini, L., Forli, F., Fiacchini, G., Canelli, R., Berrettini, S., & Lazzerini, F. (2022). Battery Lifespan of an Implantable Middle Ear Device. Audiology Research, 12(5), 485-492. https://doi.org/10.3390/audiolres12050049





