Mechanisms by Which Soil Microbial Communities Regulate Ecosystem Multifunctionality in Tea Gardens of Longnan City, China
Abstract
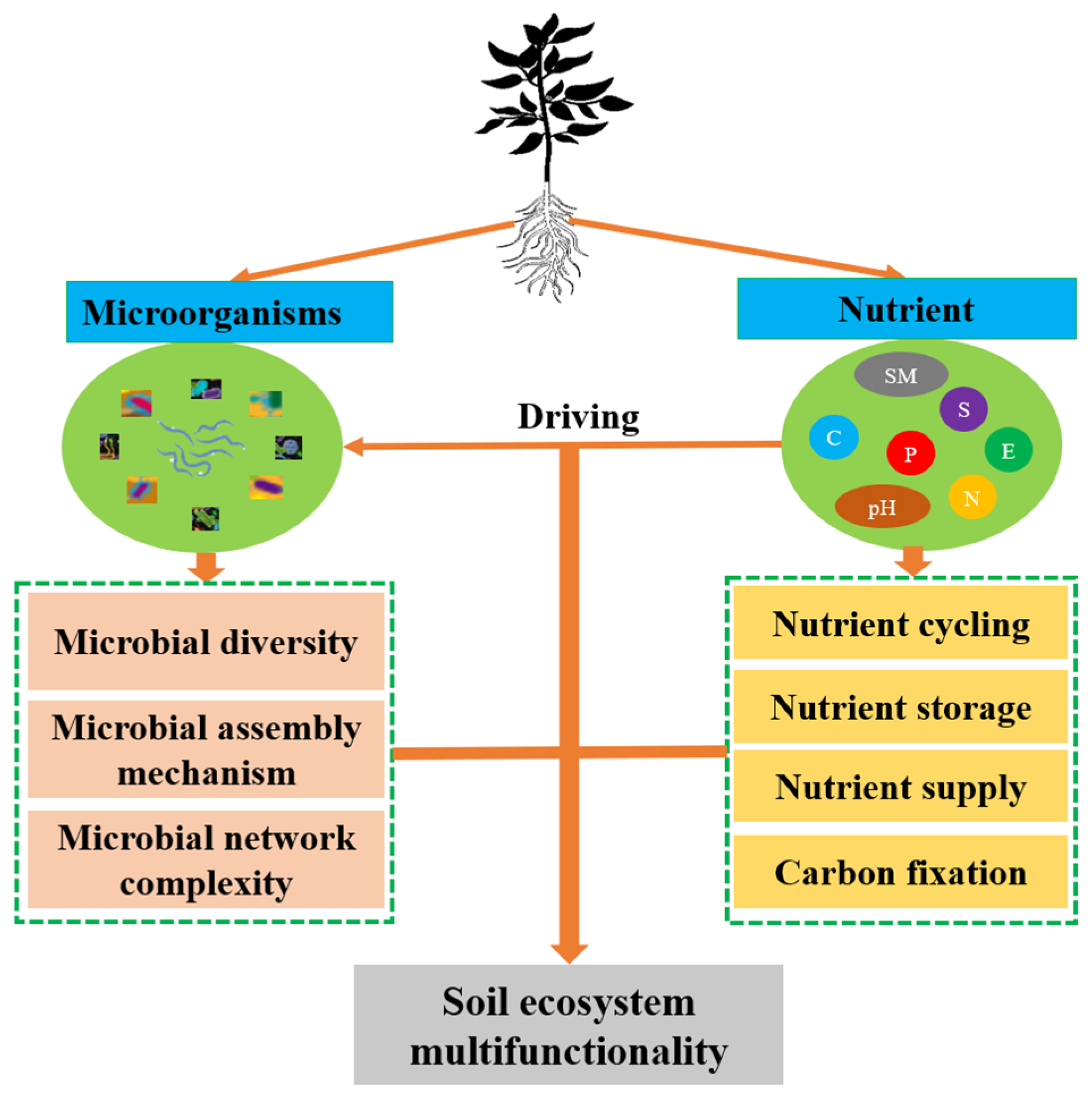
1. Introduction
2. Materials and Methods
2.1. Study Area Overview
2.2. Soil Sample Collection
2.3. Methods for Determining Soil Physicochemical Properties
2.4. Soil Microbial High-Throughput Sequencing
2.5. Determination of Ecosystem Multifunctionality
2.6. Calculation of Soil Element and Enzyme Stoichiometry
2.7. Data Analysis
3. Results and Analyses
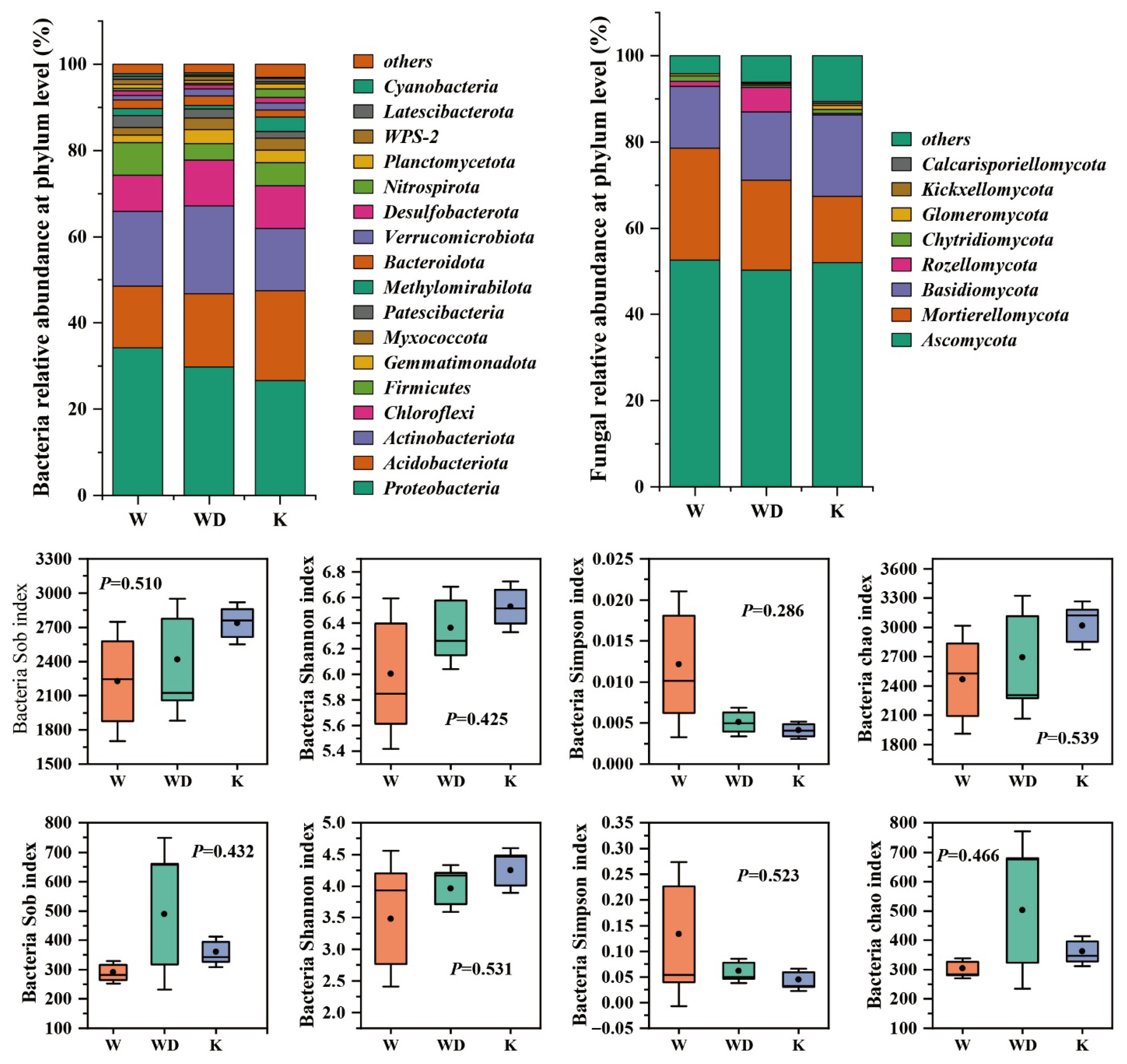
3.1. Soil Microbial Community Structure
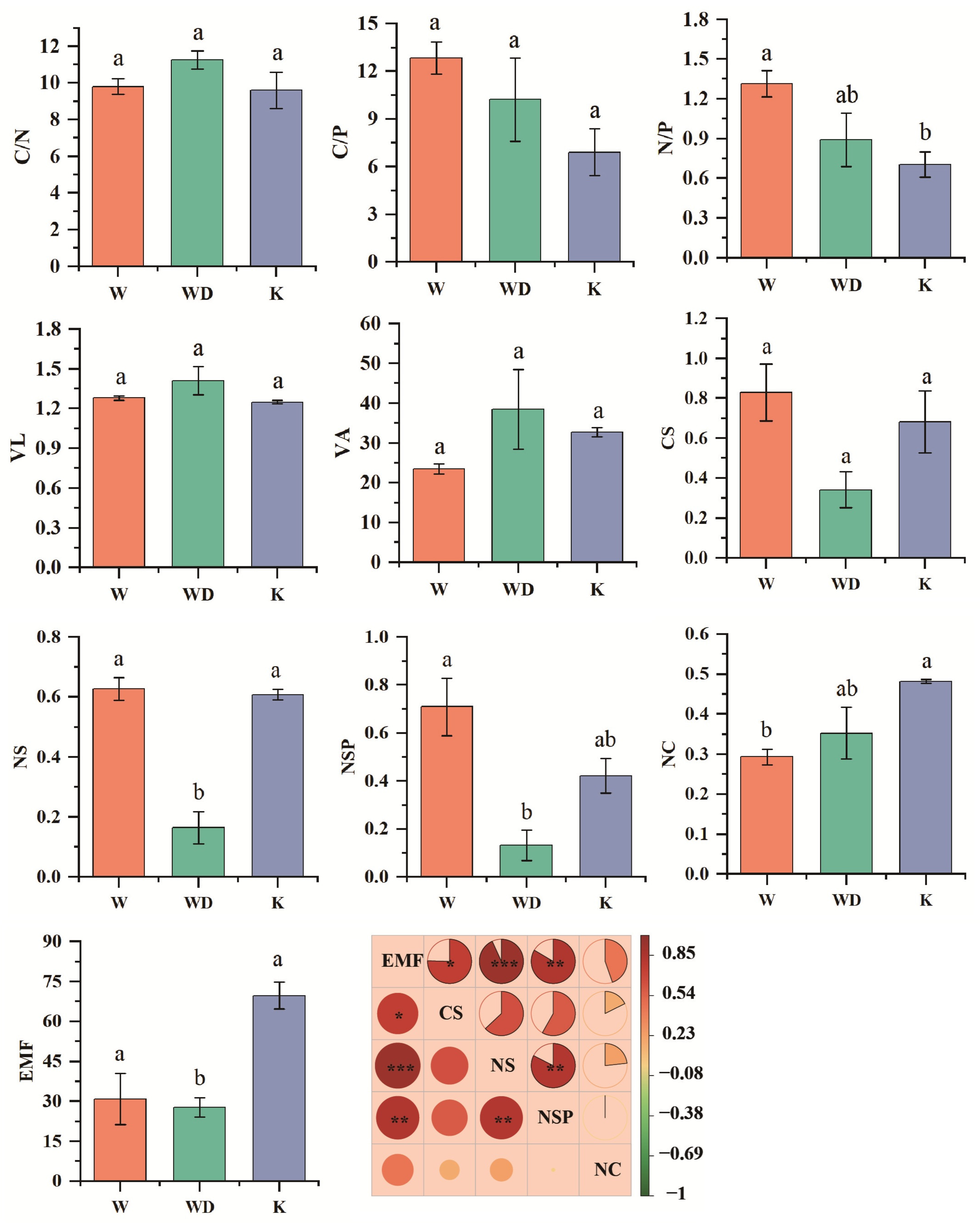
3.2. Soil Ecosystem Multifunctionality
3.3. Matching Analysis of Spatial Interpolation Results of Soil Heavy Metals with Irrigation Water Sampling Points
4. Discussion
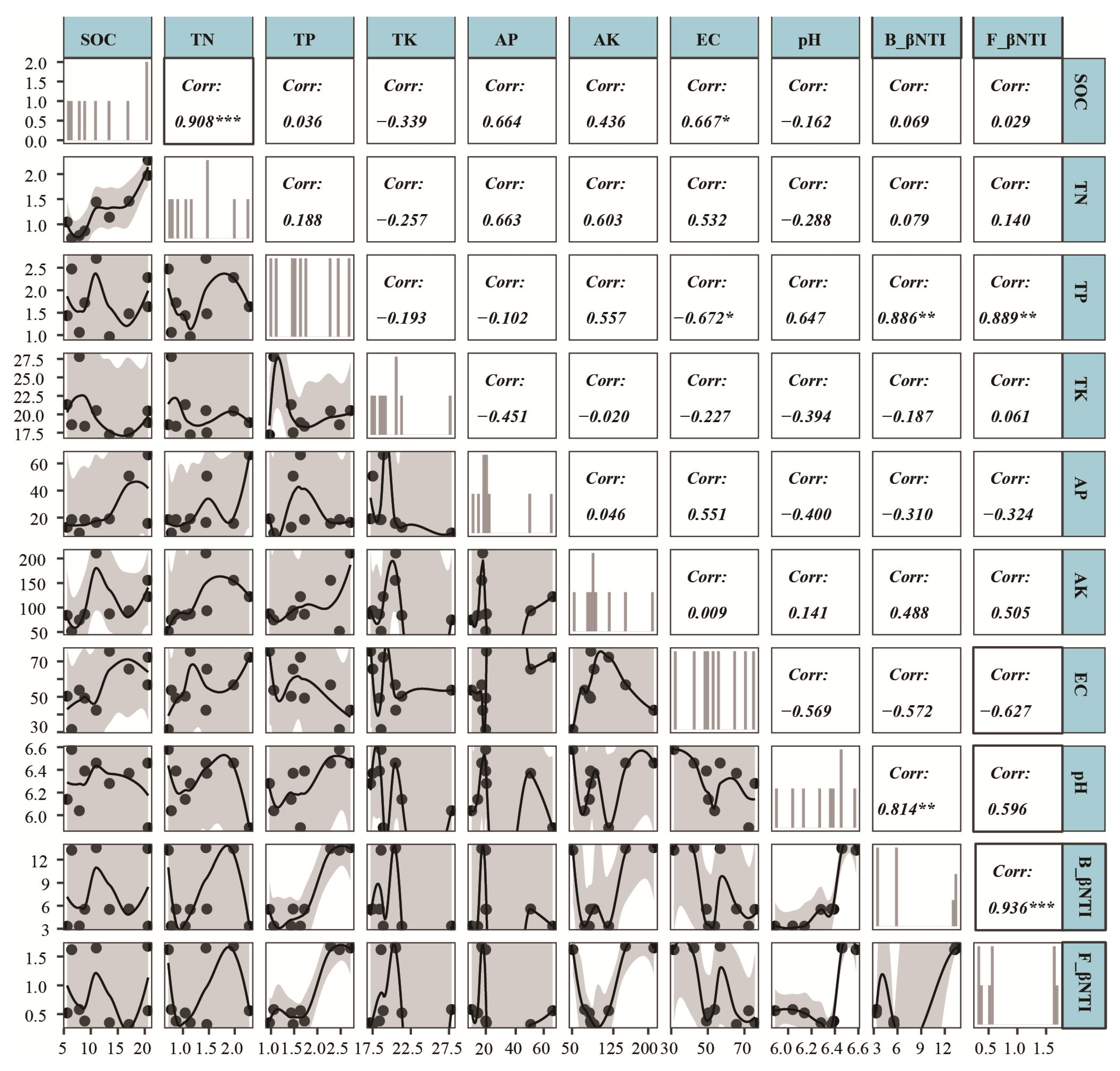
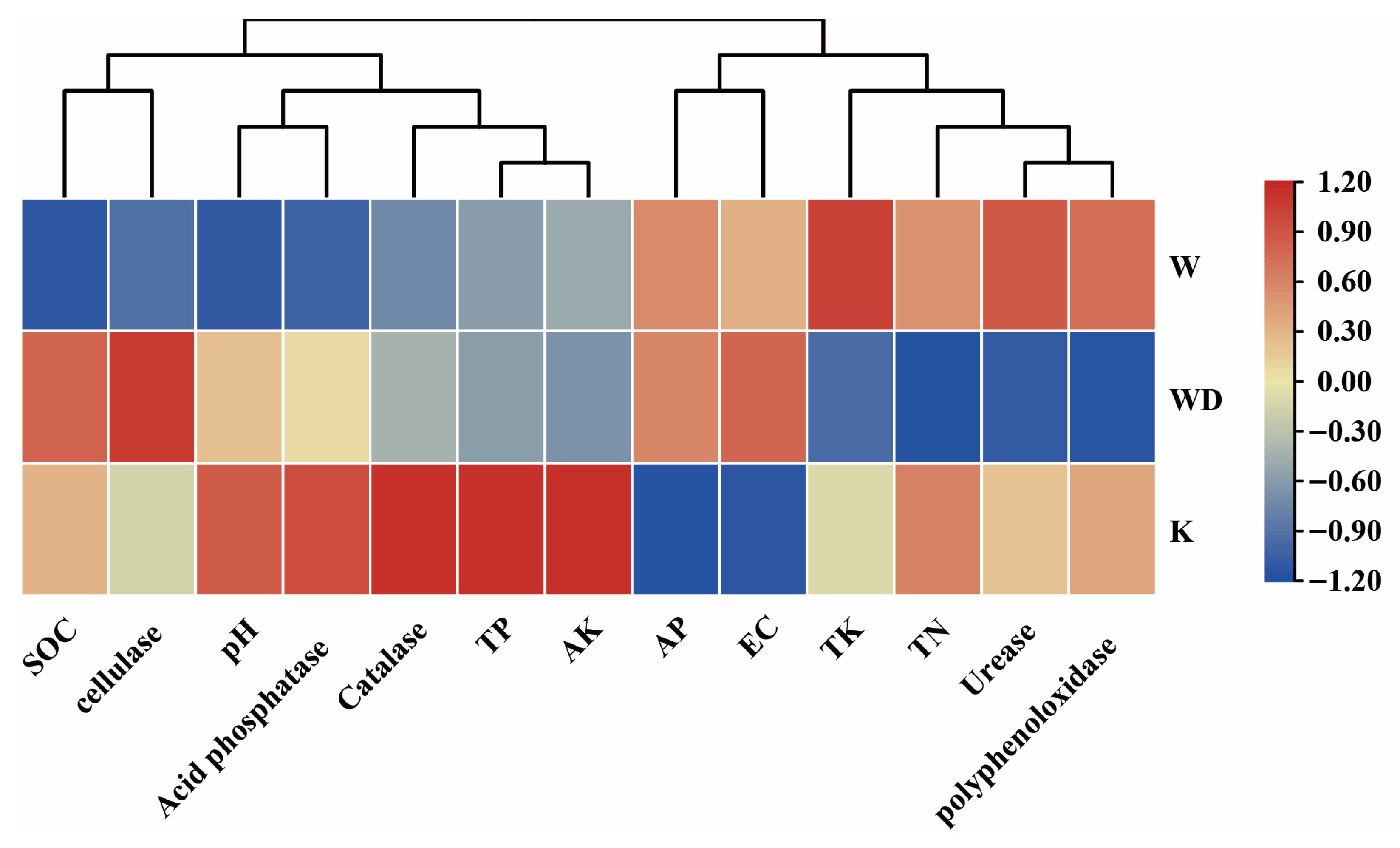
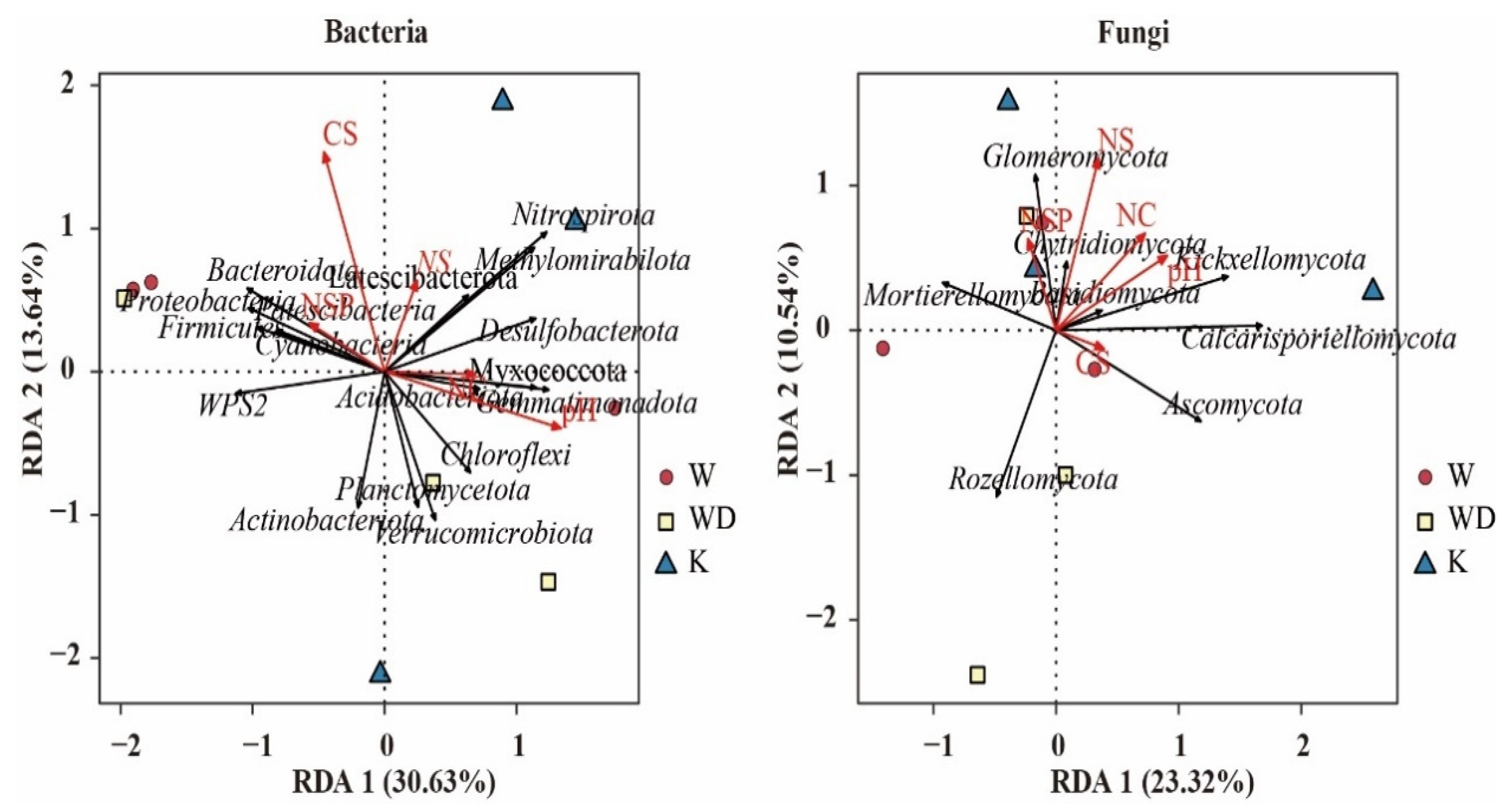
4.1. Drivers of Soil Microbial Community Structure in Tea Gardens
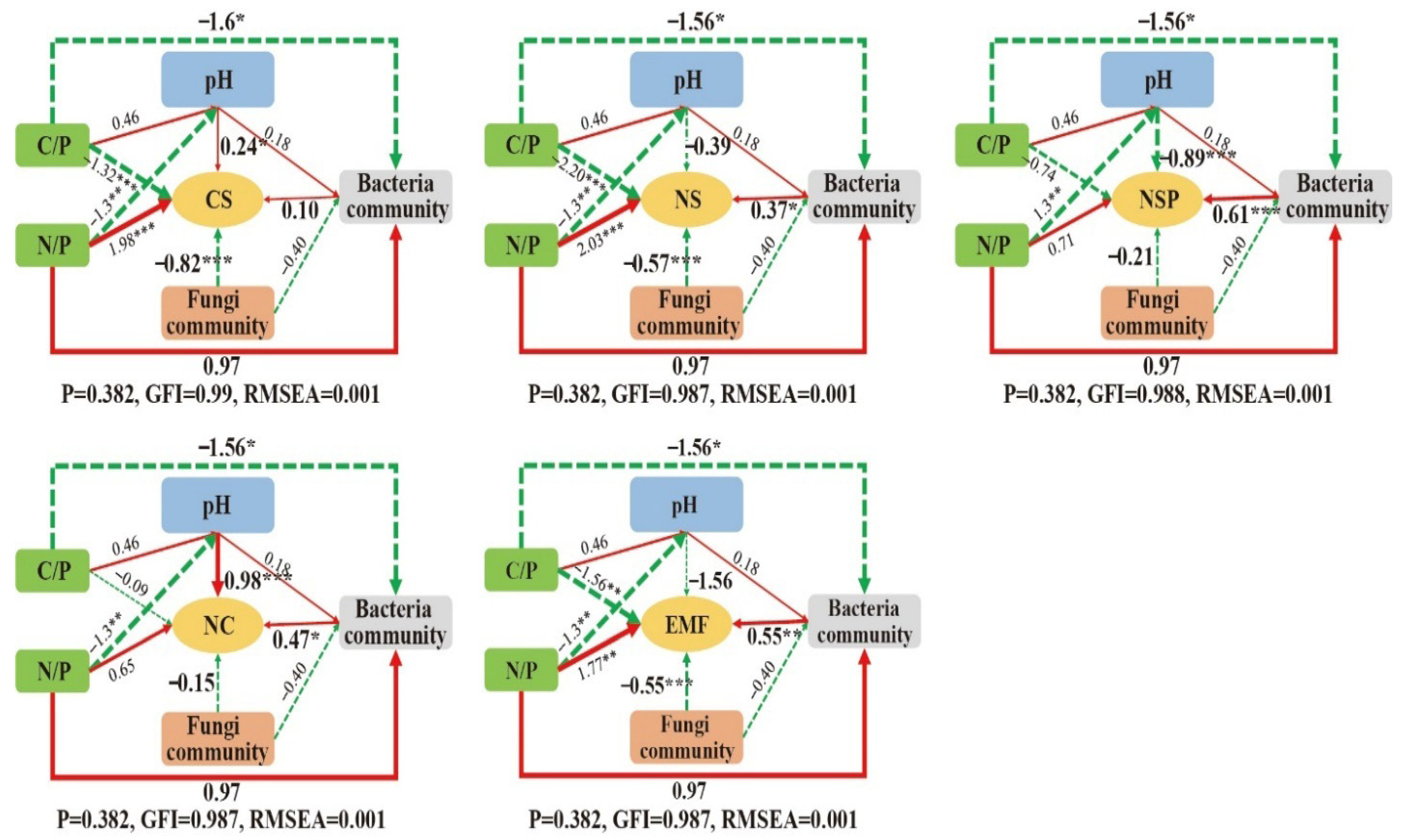
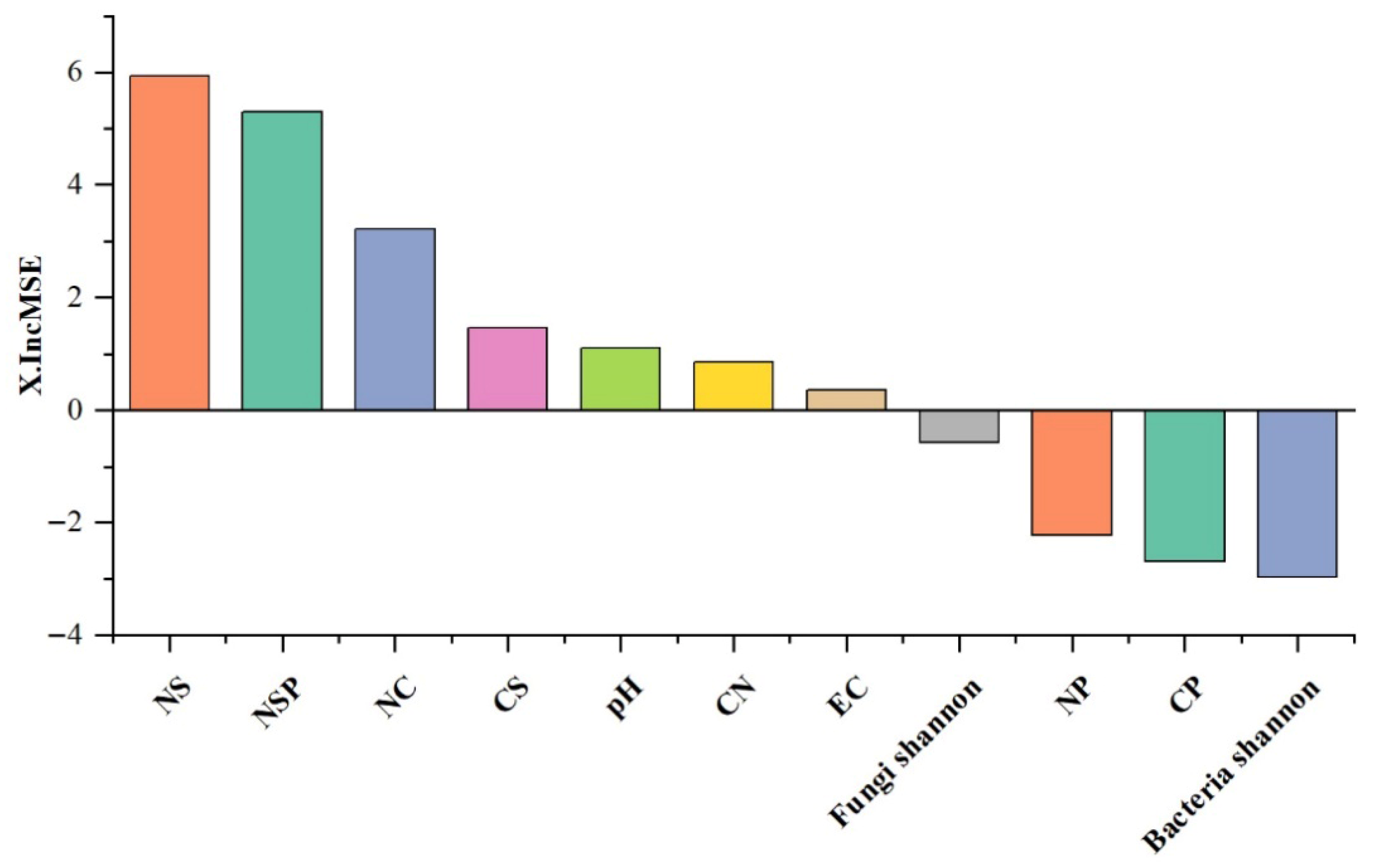
4.2. Soil Ecosystem Multifunctionality in Tea Gardens
4.3. Limitations and Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pan, S.-Y.; Nie, Q.; Tai, H.-C.; Song, X.-L.; Tong, Y.-F.; Zhang, L.-J.-F.; Wu, X.-W.; Lin, Z.-H.; Zhang, Y.-Y.; Ye, D.-Y. Tea and tea drinking: China’s outstanding contributions to the mankind. Chin. Med. 2022, 17, 27. [Google Scholar] [CrossRef]
- Jiang, T.; Wang, M.; Zhang, W.; Zhu, C.; Wang, F. A comprehensive analysis of agricultural non-point source pollution in China: Current status, risk assessment and management strategies. Sustainability 2024, 16, 2515. [Google Scholar] [CrossRef]
- Ding, K.; Chen, L.; Zhang, Y.; Ge, S.; Zhang, Y.; Lu, M.; Shen, Z.; Tong, Z.; Zhang, J. Long-term cover crops boost multi-nutrient cycling and subsurface soil carbon sequestration by alleviating microbial carbon limitation in a subtropical forest. Catena 2024, 244, 108252. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, F.; Wu, Z.; Jiang, F.; Yu, W.; Yang, J.; Chen, J.; Jian, G.; You, Z.; Zeng, L. Effects of long-term nitrogen fertilization on the formation of metabolites related to tea quality in subtropical China. Metabolites 2021, 11, 146. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ye, C.; Su, Y.; Peng, W.; Lu, R.; Liu, Y.; Huang, H.; He, X.; Yang, M.; Zhu, S. Soil Acidification caused by excessive application of nitrogen fertilizer aggravates soil-borne diseases: Evidence from literature review and field trials. Agric. Ecosyst. Environ. 2022, 340, 108176. [Google Scholar] [CrossRef]
- Liu, S.; Yang, R.; Peng, X.; Hou, C.; Ma, J.; Guo, J. Contributions of plant litter decomposition to soil nutrients in ecological tea gardens. Agriculture 2022, 12, 957. [Google Scholar] [CrossRef]
- Wang, H.; Sheng, M.; Yang, X.; Zhao, Q.; Jiao, J.; Pan, H.; Ye, X.; Chen, X. Overview and trends of international research on soil acidification in tea plantations. Chin. J. Eco-Agric. 2024, 32, 2107–2116. [Google Scholar] [CrossRef]
- Naz, M.; Dai, Z.; Hussain, S.; Tariq, M.; Danish, S.; Khan, I.U.; Qi, S.; Du, D. The soil pH and heavy metals revealed their impact on soil microbial community. J. Environ. Manag. 2022, 321, 115770. [Google Scholar] [CrossRef]
- Singh, J.S.; Gupta, V.K. Soil microbial biomass: A key soil driver in management of ecosystem functioning. Sci. Total Environ. 2018, 634, 497–500. [Google Scholar] [CrossRef]
- Tarafdar, J.C. Role of soil biology on soil health for sustainable agricultural production. In Structure and Functions of Pedospher; Springer: Berlin/Heidelberg, Germany, 2022; pp. 67–81. [Google Scholar] [CrossRef]
- Nadarajah, K.; Abdul Rahman, N.S.N. The microbial connection to sustainable agriculture. Plants 2023, 12, 2307. [Google Scholar] [CrossRef]
- Coban, O.; De Deyn, G.B.; van der Ploeg, M. Soil microbiota as game-changers in restoration of degraded lands. Science 2022, 375, abe0725. [Google Scholar] [CrossRef]
- Schimel, J.P. Life in dry soils: Effects of drought on soil microbial communities and processes. Annu. Rev. Ecol. Evol. Syst. 2018, 49, 409–432. [Google Scholar] [CrossRef]
- Harris, J. Soil microbial communities and restoration ecology: Facilitators or followers? Science 2009, 325, 573–574. [Google Scholar] [CrossRef]
- Hall, E.K.; Bernhardt, E.S.; Bier, R.L.; Bradford, M.A.; Boot, C.M.; Cotner, J.B.; Del Giorgio, P.A.; Evans, S.E.; Graham, E.B.; Jones, S.E. Understanding how microbiomes influence the systems they inhabit. Nat. Microbiol. 2018, 3, 977–982. [Google Scholar] [CrossRef]
- Saha, I.; Datta, S.; Biswas, D. Exploring the role of bacterial extracellular polymeric substances for sustainable development in agriculture. Curr. Microbiol. 2020, 77, 3224–3239. [Google Scholar] [CrossRef] [PubMed]
- Jibola-Shittu, M.Y.; Heng, Z.; Keyhani, N.O.; Dang, Y.; Chen, R.; Liu, S.; Lin, Y.; Lai, P.; Chen, J.; Yang, C. Understanding and exploring the diversity of soil microorganisms in tea (Camellia sinensis) gardens: Toward sustainable tea production. Front. Microbiol. 2024, 15, 1379879. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Zhang, L.; Wang, Y.; Huang, Z. Impact of ecological restoration on the physicochemical properties and bacterial communities in alpine mining area soils. Microorganisms 2023, 12, 41. [Google Scholar] [CrossRef] [PubMed]
- Rillig, M.C.; Ryo, M.; Lehmann, A.; Aguilar-Trigueros, C.A.; Buchert, S.; Wulf, A.; Iwasaki, A.; Roy, J.; Yang, G. The role of multiple global change factors in driving soil functions and microbial biodiversity. Science 2019, 366, 886–890. [Google Scholar] [CrossRef]
- Manning, P.; Van Der Plas, F.; Soliveres, S.; Allan, E.; Maestre, F.T.; Mace, G.; Whittingham, M.J.; Fischer, M. Redefining ecosystem multifunctionality. Nat. Ecol. Evol. 2018, 2, 427–436. [Google Scholar] [CrossRef]
- Zhang, L.; Du, H.; Song, T.; Yang, Z.; Peng, W.; Gong, J.; Huang, G.; Li, Y. Conversion of farmland to forest or grassland improves soil carbon, nitrogen, and ecosystem multi-functionality in a subtropical karst region of southwest China. Sci. Rep. 2024, 14, 17745. [Google Scholar] [CrossRef]
- Craven, D.; Eisenhauer, N.; Pearse, W.D.; Hautier, Y.; Isbell, F.; Roscher, C.; Bahn, M.; Beierkuhnlein, C.; Bönisch, G.; Buchmann, N. Multiple facets of biodiversity drive the diversity–stability relationship. Nat. Ecol. Evol. 2018, 2, 1579–1587. [Google Scholar] [CrossRef]
- Wagg, C.; Bender, S.F.; Widmer, F.; Van Der Heijden, M.G. Soil biodiversity and soil community composition determine ecosystem multifunctionality. Proc. Natl. Acad. Sci. USA 2014, 111, 5266–5270. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Baquerizo, M.; Maestre, F.T.; Reich, P.B.; Jeffries, T.C.; Gaitan, J.J.; Encinar, D.; Berdugo, M.; Campbell, C.D.; Singh, B.K. Microbial diversity drives multifunctionality in terrestrial ecosystems. Nat. Commun. 2016, 7, 10541. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, X.; Liu, G.; Zhang, C.; Wang, G. Bacterial richness is negatively related to potential soil multifunctionality in a degraded alpine meadow. Ecol. Indic. 2021, 121, 106996. [Google Scholar] [CrossRef]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis: Part 3 Chemical Methods; American Society of Agronomy; Soil Science Society of America: Madison, WI, USA, 1996; Volume 5, pp. 961–1010. [Google Scholar] [CrossRef]
- Bremner, J. Total nitrogen. Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties; American Society of Agronomy; Soil Science Society of America: Madison, WI, USA, 1965; Volume 9, pp. 1149–1178. [Google Scholar]
- Olsen, S.R. Estimation of Available Phosphorus in Soils by Extraction with Sodium Bicarbonate; US Department of Agriculture: Washington, DC, USA, 1954.
- Kandeler, E.; Gerber, H. Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol. Fertil. Soils 1988, 6, 68–72. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol. Biochem. 1969, 1, 301–307. [Google Scholar] [CrossRef]
- Saxena, M.; Saxena, J.; Nema, R.; Singh, D.; Gupta, A. Phytochemistry of medicinal plants. J. Pharmacogn. Phytochem. 2013, 1, 168–182. [Google Scholar]
- Peter-Katalinić, J. Methods in enzymology: O-glycosylation of proteins. Methods Enzymol. 2005, 405, 139–171. [Google Scholar] [CrossRef]
- Stohner, J.; Ganesan, A. Pure and Applied Chemistry; Blackwell Scientific Publications: Oxford, UK, 1970; Volume 24. [Google Scholar]
- Wang, G.C.; Wang, Y. The frequency of chimeric molecules as a consequence of PCR co-amplification of 16S rRNA genes from different bacterial species. Microbiology 1996, 142, 1107–1114. [Google Scholar] [CrossRef]
- Yang, Y.; Dou, Y.; Huang, Y.; An, S. Links between soil fungal diversity and plant and soil properties on the Loess Plateau. Front. Microbiol. 2017, 8, 2198. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Wang, J.; Chen, X.; Meng, Z.; Xu, R.; Duoji, D.; Zhang, J.; He, J.; Wang, Z.; Chen, J. Soil microbial network complexity predicts ecosystem function along elevation gradients on the Tibetan Plateau. Soil Biol. Biochem. 2022, 172, 108766. [Google Scholar] [CrossRef]
- Jiao, S.; Lu, Y.; Wei, G. Soil multitrophic network complexity enhances the link between biodiversity and multifunctionality in agricultural systems. Glob. Change Biol. 2022, 28, 140–153. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Wang, Y.; Ye, S.; Liu, S.; Stirling, E.; Gilbert, J.A.; Faust, K.; Knight, R.; Jansson, J.K.; Cardona, C. Earth microbial co-occurrence network reveals interconnection pattern across microbiomes. Microbiome 2020, 8, 82. [Google Scholar] [CrossRef] [PubMed]
- Maestre, F.T.; Quero, J.L.; Gotelli, N.J.; Escudero, A.; Ochoa, V.; Delgado-Baquerizo, M.; García-Gómez, M.; Bowker, M.A.; Soliveres, S.; Escolar, C. Plant species richness and ecosystem multifunctionality in global drylands. Science 2012, 335, 214–218. [Google Scholar] [CrossRef]
- Moorhead, D.L.; Sinsabaugh, R.L.; Hill, B.H.; Weintraub, M.N. Vector analysis of ecoenzyme activities reveal constraints on coupled C, N and P dynamics. Soil Biol. Biochem. 2016, 93, 1–7. [Google Scholar] [CrossRef]
- Zhong, Z.; Li, W.; Lu, X.; Gu, Y.; Wu, S.; Shen, Z.; Han, X.; Yang, G.; Ren, C. Adaptive pathways of soil microorganisms to stoichiometric imbalances regulate microbial respiration following afforestation in the Loess Plateau, China. Soil Biol. Biochem. 2020, 151, 108048. [Google Scholar] [CrossRef]
- Hartmann, M.; Six, J. Soil structure and microbiome functions in agroecosystems. Nat. Rev. Earth Environ. 2023, 4, 4–18. [Google Scholar] [CrossRef]
- Xiong, W.; Jousset, A.; Guo, S.; Karlsson, I.; Zhao, Q.; Wu, H.; Kowalchuk, G.A.; Shen, Q.; Li, R.; Geisen, S. Soil protist communities form a dynamic hub in the soil microbiome. ISME J. 2018, 12, 634–638. [Google Scholar] [CrossRef]
- Xiong, F.; Yao, C.; Zhai, D.; Liu, H.; Dong, W.; Chen, Y. Heavy metal contamination and eutrophication effects on bacterial communities in the Han River basin. Ecol. Indic. 2025, 170, 113108. [Google Scholar] [CrossRef]
- Zhang, Z.; Lu, J.; Zhang, S.; Tian, Z.; Feng, C.; Liu, Y. Analysis of bacterial community structure, functional variation, and assembly mechanisms in multi-media habitats of lakes during the frozen period. Ecotoxicol. Environ. Saf. 2024, 284, 116903. [Google Scholar] [CrossRef]
- Li, X.; Su, L.; Jing, M.; Wang, K.; Song, C.; Song, Y. Nitrogen addition restricts key soil ecological enzymes and nutrients by reducing microbial abundance and diversity. Sci. Rep. 2025, 15, 5560. [Google Scholar] [CrossRef]
- Zhu, J.; Niu, W.; Zhang, Z.; Siddique, K.H.; Sun, D.; Yang, R. Distinct roles for soil bacterial and fungal communities associated with the availability of carbon and phosphorus under aerated drip irrigation. Agric. Water Manag. 2022, 274, 107925. [Google Scholar] [CrossRef]
- Schä, H.; Bernard, L.; Courties, C.; Lebaron, P.; Servais, P.; Pukall, R.; Stackebrandt, E.; Troussellier, M.; Teresa, G.; Vives-Rego, J. Microbial community dynamics in Mediterranean nutrient-enriched seawater mesocosms: Changes in the genetic diversity of bacterial populations. FEMS Microbiol. Ecol. 2001, 34, 243–253. [Google Scholar] [CrossRef]
- Li, N.; Zhang, Y.; Qu, Z.; Liu, B.; Huang, L.; Ming, A.; Sun, H. Mixed and continuous cropping eucalyptus plantation facilitated soil carbon cycling and fungal community diversity after a 14-year field trail. Ind. Crops Prod. 2024, 210, 118157. [Google Scholar] [CrossRef]
- Yan, P.; Shen, C.; Zou, Z.; Fan, L.; Li, X.; Zhang, L.; Zhang, L.; Dong, C.; Fu, J.; Han, W. Increased soil fertility in tea gardens leads to declines in fungal diversity and complexity in subsoils. Agronomy 2022, 12, 1751. [Google Scholar] [CrossRef]
- Carr, A.; Diener, C.; Baliga, N.S.; Gibbons, S.M. Use and abuse of correlation analyses in microbial ecology. ISME J. 2019, 13, 2647–2655. [Google Scholar] [CrossRef]
- Jiao, H.; Xu, M.; Ji, J.; Wang, Y.; Zhang, Y.; Su, B.; Zhang, H.; Gao, C. Bacterial and fungal diversity, community composition, functional groups, and co-occurrence network succession in dryland and paddy soils along a 3000-year chronosequence. Catena 2025, 248, 108582. [Google Scholar] [CrossRef]
- Li, X.; Xie, J.; Zhang, Q.; Lyu, M.; Xiong, X.; Liu, X.; Lin, T.; Yang, Y. Substrate availability and soil microbes drive temperature sensitivity of soil organic carbon mineralization to warming along an elevation gradient in subtropical Asia. Geoderma 2020, 364, 114198. [Google Scholar] [CrossRef]
- Lv, H.; Yang, M.; Cheng, Y.; Li, K.; Ji, G.; Huang, T.; Wen, G. Disentangling the assembly patterns and drivers of microbial communities during thermal stratification and mixed periods in a deep-water reservoir. Sci. Total Environ. 2024, 946, 174398. [Google Scholar] [CrossRef]
- Adamo, I.; Ortiz-Malavasi, E.; Chazdon, R.; Chaverri, P.; Ter Steege, H.; Geml, J. Soil fungal community composition correlates with site-specific abiotic factors, tree community structure, and forest age in regenerating tropical rainforests. Biology 2021, 10, 1120. [Google Scholar] [CrossRef]
- Creamer, R.E.; Hagens, M.; Baartman, J.; Helming, K. Editorial for special issue on “understanding soil functions–from ped to planet”. Eur. J. Soil Sci. 2021, 72, 1493–1496. [Google Scholar] [CrossRef]
- Kome, G.K.; Enang, R.K.; Tabi, F.O.; Yerima, B.P.K. Influence of clay minerals on some soil fertility attributes: A review. Open J. Soil Sci. 2019, 9, 155–188. [Google Scholar] [CrossRef]
- Adnan, M.; Islam, W.; Gang, L.; Chen, H.Y. Advanced research tools for fungal diversity and its impact on forest ecosystem. Environ. Sci. Pollut. Res. 2022, 29, 45044–45062. [Google Scholar] [CrossRef]
- Rodrigues, C.I.D.; Brito, L.M.; Nunes, L.J. Soil carbon sequestration in the context of climate change mitigation: A review. Soil Syst. 2023, 7, 64. [Google Scholar] [CrossRef]
- Das, P.P.; Singh, K.R.; Nagpure, G.; Mansoori, A.; Singh, R.P.; Ghazi, I.A.; Kumar, A.; Singh, J. Plant-soil-microbes: A tripartite interaction for nutrient acquisition and better plant growth for sustainable agricultural practices. Environ. Res. 2022, 214, 113821. [Google Scholar] [CrossRef]
- Bargaz, A.; Lyamlouli, K.; Chtouki, M.; Zeroual, Y.; Dhiba, D. Soil microbial resources for improving fertilizers efficiency in an integrated plant nutrient management system. Front. Microbiol. 2018, 9, 1606. [Google Scholar] [CrossRef]
- Naresh, R.; Timsina, J.; Bhaskar, S.; Gupta, R.; Singh, A.; Dhaliwal, S.; Rathore, R.; Kumar, V.; Singh, P.; Singh, S. Effects of tillage, residue and nutrient management on soil organic carbon dynamics and its fractions, soil aggregate stability and soil carbon sequestration: A review. EC Nutr. 2017, 12, 53–80. [Google Scholar]
- Raza, T.; Qadir, M.F.; Khan, K.S.; Eash, N.S.; Yousuf, M.; Chatterjee, S.; Manzoor, R.; ur Rehman, S.; Oetting, J.N. Unraveling the potential of microbes in decomposition of organic matter and release of carbon in the ecosystem. J. Environ. Manag. 2023, 344, 118529. [Google Scholar] [CrossRef]
- Li, S.; Cui, Y.; Xia, Z.; Zhang, X.; Zhou, C.; An, S.; Zhu, M.; Gao, Y.; Yu, W.; Ma, Q. Microbial nutrient limitations limit carbon sequestration but promote nitrogen and phosphorus cycling: A case study in an agroecosystem with long-term straw return. Sci. Total Environ. 2023, 870, 161865. [Google Scholar] [CrossRef]
- Thepbandit, W.; Athinuwat, D. Rhizosphere microorganisms supply availability of soil nutrients and induce plant defense. Microorganisms 2024, 12, 558. [Google Scholar] [CrossRef]
- Xiao, D.; Huang, Y.; Feng, S.; Ge, Y.; Zhang, W.; He, X.; Wang, K. Soil organic carbon mineralization with fresh organic substrate and inorganic carbon additions in a red soil is controlled by fungal diversity along a pH gradient. Geoderma 2018, 321, 79–89. [Google Scholar] [CrossRef]
- Rahman, M.A.; Lee, S.-H.; Ji, H.C.; Kabir, A.H.; Jones, C.S.; Lee, K.-W. Importance of mineral nutrition for mitigating aluminum toxicity in plants on acidic soils: Current status and opportunities. Int. J. Mol. Sci. 2018, 19, 3073. [Google Scholar] [CrossRef] [PubMed]
- Baldrian, P.; in der Wiesche, C.; Gabriel, J.; Nerud, F.; Zadražil, F. Influence of cadmium and mercury on activities of ligninolytic enzymes and degradation of polycyclic aromatic hydrocarbons by Pleurotus ostreatus in soil. Appl. Environ. Microbiol. 2000, 66, 2471–2478. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Kumar, G.; Chhabra, S.; Prasad, R. Role of soil microbes in biogeochemical cycle for enhancing soil fertility. In New and Future Developments in Microbial Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2021; pp. 149–157. [Google Scholar] [CrossRef]
- Biswas, T.; Kole, S.C. Soil organic matter and microbial role in plant productivity and soil fertility. In Advances in Soil Microbiology: Recent Trends and Future Prospects; Volume 2: Soil-Microbe-Plant Interaction; Springer: Berlin/Heidelberg, Germany, 2018; pp. 219–238. [Google Scholar] [CrossRef]
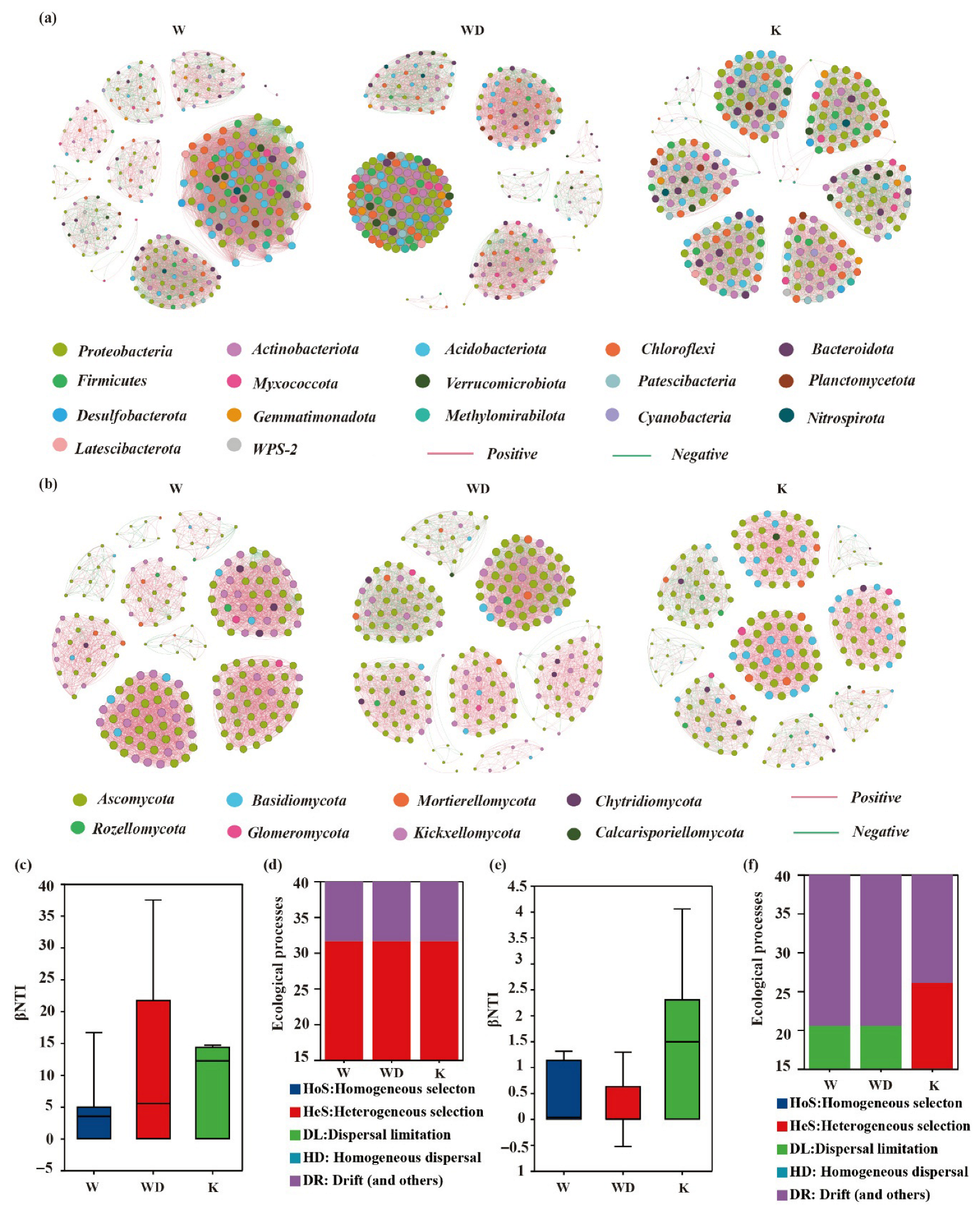
| Topological | Bacteria | Fungi | ||||
|---|---|---|---|---|---|---|
| Parameters | W | WD | K | W | WD | K |
| Nodes | 288 | 284 | 281 | 211 | 232 | 224 |
| Links | 8623 | 9259 | 5624 | 3328 | 3913 | 3205 |
| Positive links % | 68.76% | 60.16% | 57.52% | 87.80% | 77.31% | 85.87% |
| Negative links % | 31.24% | 39.84% | 42.48% | 12.20% | 22.69% | 14.13% |
| Average degree | 59.882 | 65.204 | 40.028 | 31.545 | 33.733 | 28.616 |
| Grpah density | 0.209 | 0.23 | 0.143 | 0.15 | 0.146 | 0.128 |
| Modularity | 0.474 | 0.515 | 0.834 | 0.768 | 0.778 | 0.825 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nian, L.; Li, J.; Tang, Y.; Haider, F.U.; Wang, Z.; Dong, L.; Zhang, J.; Long, Q.; Wang, W.; Zhao, X. Mechanisms by Which Soil Microbial Communities Regulate Ecosystem Multifunctionality in Tea Gardens of Longnan City, China. Microbiol. Res. 2025, 16, 192. https://doi.org/10.3390/microbiolres16090192
Nian L, Li J, Tang Y, Haider FU, Wang Z, Dong L, Zhang J, Long Q, Wang W, Zhao X. Mechanisms by Which Soil Microbial Communities Regulate Ecosystem Multifunctionality in Tea Gardens of Longnan City, China. Microbiology Research. 2025; 16(9):192. https://doi.org/10.3390/microbiolres16090192
Chicago/Turabian StyleNian, Lili, Juan Li, Ying Tang, Fasih Ullah Haider, Zining Wang, Liuwen Dong, Jie Zhang, Qian Long, Wenli Wang, and Xu Zhao. 2025. "Mechanisms by Which Soil Microbial Communities Regulate Ecosystem Multifunctionality in Tea Gardens of Longnan City, China" Microbiology Research 16, no. 9: 192. https://doi.org/10.3390/microbiolres16090192
APA StyleNian, L., Li, J., Tang, Y., Haider, F. U., Wang, Z., Dong, L., Zhang, J., Long, Q., Wang, W., & Zhao, X. (2025). Mechanisms by Which Soil Microbial Communities Regulate Ecosystem Multifunctionality in Tea Gardens of Longnan City, China. Microbiology Research, 16(9), 192. https://doi.org/10.3390/microbiolres16090192






