Acetyleugenol from Acacia nilotica (L.) Exhibits a Strong Antibacterial Activity and Its Phenyl and Indole Analogues Show a Promising Anti-TB Potential Targeting PknE/B Protein Kinases
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Collection and Authentication
2.2. Preparation of Ethyl Acetate Extracts
2.3. Isolation and Purification of the Bioactive Compounds
2.4. Thin Layer Chromatography
2.5. Column Chromatography
2.6. Gas Chromatography/Mass Spectrometry Analysis
2.7. Nuclear Magnetic Resonance Spectroscopy
2.8. Bioactivity Antibacterial Sensitivity Assays
2.8.1. Microorganisms, Culture Media and Inoculum Preparations
2.8.2. Antibacterial Sensitivity Assay of the Extracts
2.8.3. Minimum Inhibitory Concentration and Minimum Bactericidal Concentration
2.9. Molecular Docking Experiment
2.9.1. Ligands and Protein Preparations
2.9.2. Docking Procedure
3. Results and Discussion
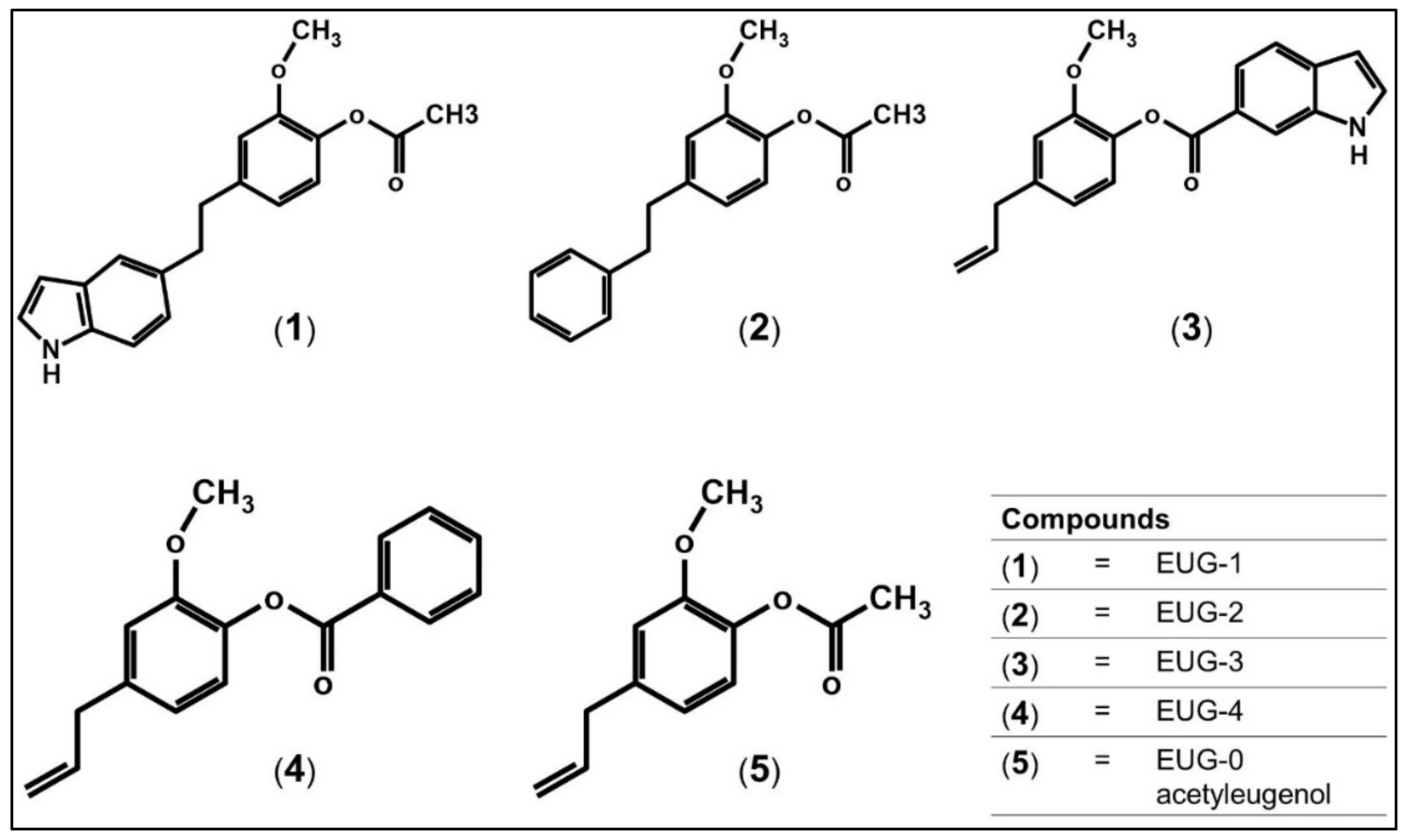
3.1. Isolation and Structural Elucidation of Eugenol and Its Antibacterial Activity
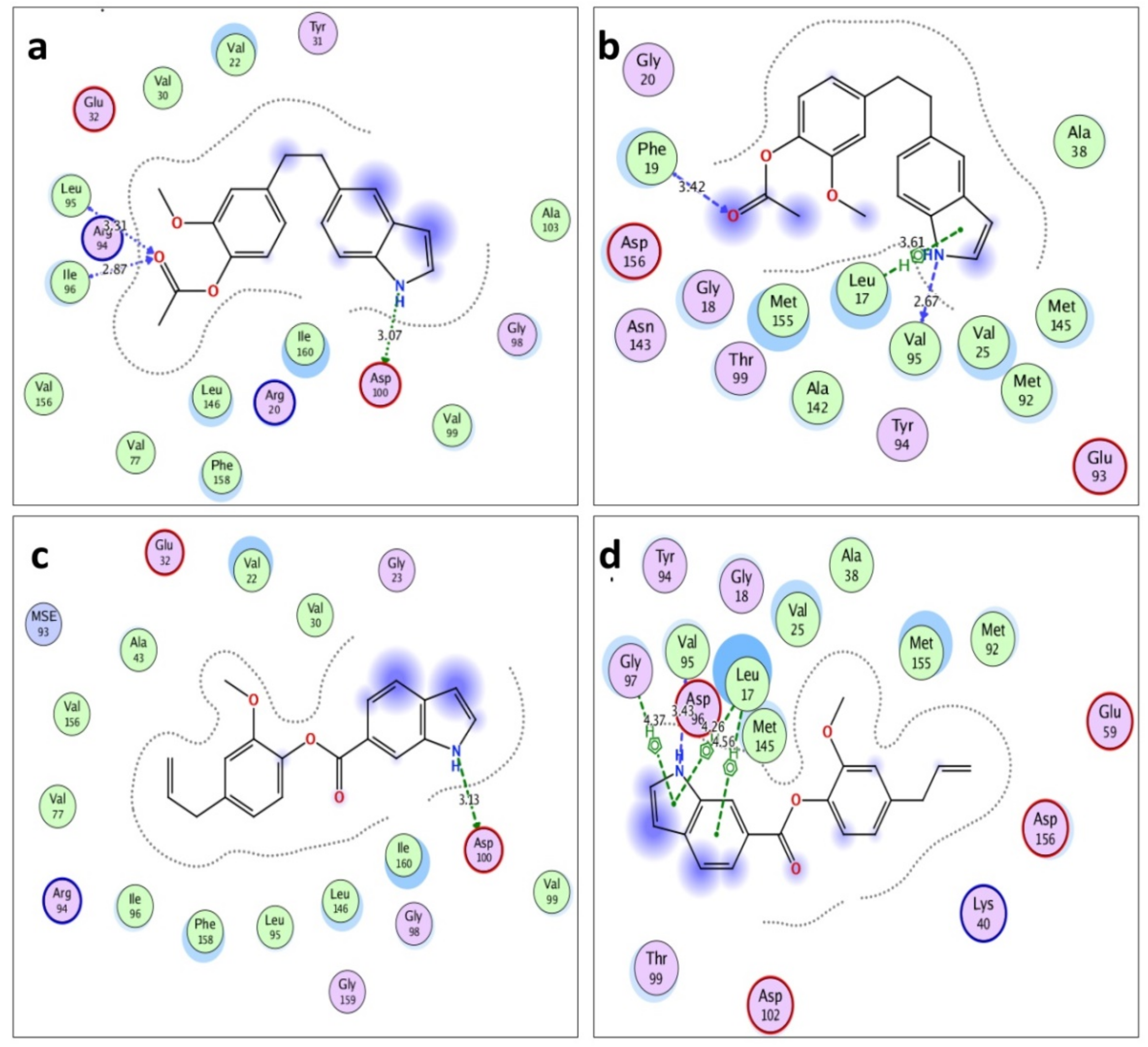
3.2. Molecular Docking of Acetyleugenol and Its Analogues Against MtPknB and MtPknE
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- MacNeil, A.; Glaziou, P.; Sismanidis, C.; Maloney, S.; Floyd, K. Global epidemiology of tuberculosis and progress toward achieving global targets—2017. MMWR Morb. Mortal. Wkly Rep. 2019, 68, 263–266. [Google Scholar] [CrossRef] [Green Version]
- Seung, K.J.; Keshavjee, S.; Rich, M.L. Multidrug-resistant tuberculosis and extensively drug-resistant tuberculosis. Cold Spring Harb. Perspect. Med. 2015, 5, a017863. [Google Scholar] [CrossRef] [Green Version]
- Sakamoto, H.; Lee, S.; Ishizuka, A.; Hinoshita, E.; Hori, H.; Ishibashi, N.; Komada, K.; Norizuki, M.; Katsuma, Y.; Akashi, H.; et al. Challenges and opportunities for eliminating tuberculosis—Leveraging political momentum of the UN high-level meeting on tuberculosis. BMC Public Health 2019, 19, 76. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Available online: https://www.who.int/news-room/fact-sheets/detail/tuberculosis (accessed on 24 March 2020).
- Liang, L.; Wu, Q.; Gao, L.; Hao, Y.; Liu, C.; Xie, Y.; Sun, H.; Yan, X.; Li, F.; Li, H.; et al. Factors contributing to the high prevalence of multidrug-resistant tuberculosis: A study from China. Thorax 2012, 67, 632–638. [Google Scholar] [CrossRef] [Green Version]
- Gandhi, N.R.; Moll, A.; Sturm, A.W.; Pawinski, R.; Govender, T.; Lalloo, U.; Zeller, K.; Andrews, J.; Friedland, G. Extensively drug-resistant tuberculosis as a cause of death in patients co-infected with tuberculosis and HIV in a rural area of South Africa. Lancet 2006, 368, 1575–1580. [Google Scholar] [CrossRef]
- Glaziou, P.; Sismanidis, C.; Floyd, K.; Raviglione, M. Global epidemiology of tuberculosis. Cold Spring Harb. Perspect. Med. 2015, 5, a017798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Heym, B.; Allen, B.; Young, D.; Cole, S. The catalase-peroxidase gene and isoniazid resistance of Mycobacterium tuberculosis. Nature 1992, 358, 591–593. [Google Scholar] [CrossRef] [PubMed]
- Rawat, R.; Whitty, A.; Tonge, P.J. The isoniazid-NAD adduct is a slow, tight-binding inhibitor of InhA, the Mycobacterium tuberculosis enoyl reductase: Adduct affinity and drug resistance. Proc. Natl. Acad. Sci. USA 2003, 100, 13881–13886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palomino, J.C.; Martin, A. Drug resistance mechanism in Mycobacterium tuberculosis. Antibiotics 2014, 3, 317–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maruri, F.; Sterling, T.R.; Kaiga, A.W.; Blackman, A.; van der Heijden, Y.F.; Mayer, C.; Cambau, E.; Aubry, A. A systematic review of gyrase mutations associated with fluoroquinolone-resistant Mycobacterium tuberculosis and a proposed gyrase numbering system. J. Antimicrob. Chemother. 2012, 67, 819–831. [Google Scholar] [CrossRef] [Green Version]
- Reeves, A.Z.; Campbell, P.J.; Sultana, R.; Malik, S.; Murray, M.; Plikaytis, B.B.; Shinnick, T.M.; Posey, J.E. Aminoglycoside Cross-Resistance in Mycobacterium tuberculosis due to mutations in the 5′ untranslated region of whiB7. Antimicrob. Agents Chemother. 2013, 57, 1857–1865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asadbeigi, M.; Mohammedi, T.; Rafieian-Kopaei, M.; Saki, K.; Bahmani, M.; Delfan, M. Traditional effects of medicinal plants in the treatment of respiratory diseases and disorders: An ethnobotanical study in the Urmia. Asian Pac. J. Trop. Med. 2014, 7, S364–S368. [Google Scholar] [CrossRef] [Green Version]
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta 2013, 1830, 3670–3695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [Green Version]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharm. 2014, 4, 177. [Google Scholar] [CrossRef] [Green Version]
- Van Wyk, B.-E. The potential of South African plants in the development of new medicinal products. S. Afr. J. Bot. 2011, 77, 812–829. [Google Scholar] [CrossRef] [Green Version]
- Semenya, S.S.; Maroyi, A. Data on medicinal plants used to treat respiratory infections and related symptoms in South Africa. Data Brief 2018, 21, 419–423. [Google Scholar] [CrossRef]
- Karar, M.G.E.; Kuhnert, N. Herbal drugs from Sudan: Traditional uses and phytoconstituents. Pharm. Rev. 2017, 11, 83–103. [Google Scholar] [CrossRef] [Green Version]
- Hussein, G.; Miyashiro, H.; Nakamura, N.; Hattori, M.; Kakiuchi, N.; Shimotohno, K. Inhibitory effects of Sudanese medicinal plant extracts on hepatitis C virus (HCV) protease. Phytother. Res. 2000, 14, 510–516. [Google Scholar] [CrossRef]
- Alli, L.A.; Adesokan, A.A.; Salawu, A.O. Antimalarial activity of fractions of aqueous extract of Acacia nilotica root. J. Intercult. Ethnopharmacol. 2018, 5, 180–185. [Google Scholar] [CrossRef]
- Singh, R.; Singh, B.; Singh, S.; Kumar, N.; Kumar, S.; Arora, S. Anti-free radical activities of kaempferol isolated from Acacia nilotica (L.) Willd. Ex. Del. Toxicol. In Vitro 2008, 22, 1965–1970. [Google Scholar] [CrossRef] [PubMed]
- Kaur, K.; Michael, H.; Arora, S.; Härkönen, P.; Kumar, S. In vitro bioactivity-guided fractionation and characterization of polyphenolic inhibitory fractions from Acacia nilotica (L.). Willd. ex Del. J. Ethnopharmacol. 2005, 99, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Saini, M.L.; Saini, R.; Roy, S.; Kumar, A. Comparative pharmacognostical and antimicrobial studies of Acacia species (Mimosaceae). J. Med. Plants Res. 2008, 2, 378–386. [Google Scholar]
- Musa, M.S.; Abdelrasool, F.E.; Elsheikh, E.A.; Ahmed, L.A.; Mahmoud, A.L.; Yagi, S.M. Ethnobotanical study of medicinal plants in the Blue Nile State, South-eastern Sudan. J. Med. Plants Res. 2011, 5, 4287–4297. [Google Scholar]
- Grush, J.; Noakes, D.L.; Moccia, R.D. The efficacy of clove oil as an anesthetic for the zebrafish, Danio rerio (Hamilton). Zebrafish 2004, 1, 46–53. [Google Scholar] [CrossRef] [Green Version]
- Mallavarapu, G.R.; Ramesh, S.; Chandrasekhara, R.S.; Rajeswara Rao, B.R.; Kaul, P.N.; Bhattacharya, A.K. Investigation of the essential oil of cinnamon leaf grown at Bangalore and Hyderabad. Flavour Fragr. J. 1995, 10, 239–242. [Google Scholar] [CrossRef]
- Clarke, S. Families of compounds that occur in essential oils. In Essential Chemistry for Aromatheraphy, 2nd ed.; Churchill Livingstone: London, UK, 2008; pp. 41–77. [Google Scholar] [CrossRef]
- Bai, S.; Seasotiya, L.; Malik, A.; Bharti, P.; Dalal, S. GC-MS analysis of chloroform extract of Acacia nilotica L. leaves. J. Pharm. Phytochem. 2014, 2, 79–82. [Google Scholar]
- Batiha, G.E.S.; Alkazmi, L.M.; Wasef, L.G.; Beshbishy, A.M.; Nadwa, E.H.; Rashwan, E.K. Syzygium aromaticum L. (Myrtaceae): Traditional uses, bioactive chemical constituents, pharmacological and toxicological activities. Biomolecules 2020, 10, 202. [Google Scholar] [CrossRef] [Green Version]
- Barboza, J.N.; da Silva Maia Bezerra Filho, C.; Silva, R.O.; Medeiros, J.V.R.; de Sousa, D.P. An overview on the anti-inflammatory potential and antioxidant profile of eugenol. Oxid. Med. Cell. Longev. 2018, 2018, 3957262. [Google Scholar] [CrossRef]
- De Almeida, A.L.; Caleffi-Ferracioli, R.; de L Scodro, R.B.; Baldin, V.P.; Montaholi, D.C.; Spricigo, L.F.; Nakamura-Vasconcelos, S.S.; Hegeto, L.A.; Sampiron, E.G.; Costacurta, G.F.; et al. Eugenol and derivatives activity against Mycobacterium tuberculosis, nontuberculous mycobacteria and other bacteria. Future Microbiol. 2019, 14, 331–344. [Google Scholar] [CrossRef]
- Rohane, S.H.; Chauhan, A.J.; Fuloria, N.K.; Fuloria, S. Synthesis and in vitro antimycobacterial potential of novel hydrazones of eugenol. Arab. J. Chem. 2020, 13, 4495–4504. [Google Scholar] [CrossRef]
- Khan, M.Z.; Kaur, P.; Nandicoori, V.K. Targeting the messengers: Serine/threonine protein kinases as potential targets for antimycobacterial drug development. IUBMB Life 2018, 70, 889–904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mori, M.; Sammartino, J.C.; Costantino, L.; Gelain, A.; Meneghetti, F.; Villa, S.; Chiarelli, L.R. An overview on the potential antimycobacterial agents targeting serine/threonine protein kinases from Mycobacterium tuberculosis. Curr. Top. Med. Chem. 2019, 19, 646–661. [Google Scholar] [CrossRef]
- Wehenkel, A.; Bellinzoni, M.; Grana, M.; Duran, R.; Villarino, A.; Fernandez, P.; Andre-Leroux, G.; England, P.; Takiff, H.; Cervenansky, C.; et al. Mycobacterial Ser/Thr protein kinases and phosphatases: Physiological roles and therapeutic potential. Biochim. Biophys. Acta 2008, 1784, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.M.; Abbott, D.W.; Park, S.T.; Dascher, C.C.; Cantley, L.C.; Husson, R.N. The Mycobacterium tuberculosis serine/threonine kinases PknA and PknB: Substrate identification and regulation of cell shape. Genes Dev. 2005, 19, 1692–1704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hatzios, S.K.; Baer, C.E.; Rustad, T.R.; Siegrist, M.S.; Pang, J.M.; Ortega, C.; Alber, T.; Grundner, C.; Sherman, D.R.; Bertozzi, C.R. Osmosensory signaling in Mycobacterium tuberculosis mediated by a eukaryotic-like Ser/Thr protein kinase. Proc. Natl. Acad. Sci. USA 2013, 110, E5069–E5077. [Google Scholar] [CrossRef] [Green Version]
- Jayakumar, D.; Jacobs, W.R.J.; Narayanan, S. Protein kinase E of Mycobacterium tuberculosis has a role in the nitric oxide stress response and apoptosis in a human macrophage model of infection. Cell. Microbiol. 2008, 10, 365–374. [Google Scholar] [CrossRef] [Green Version]
- Kumar, D.; Narayanan, S. PknE, a serine/threonine kinase of Mycobacterium tuberculosis modulates multiple apoptotic paradigms. Infect. Genet. Evol. 2012, 12, 737–747. [Google Scholar] [CrossRef]
- Gupta, V.K.; Kumar, M.M.; Bisht, D.; Kaushik, A. Plants in our combating strategies against Mycobacterium tuberculosis: Progress made and obstacles met. Pharm. Biol. 2017, 55, 1536–1544. [Google Scholar] [CrossRef] [Green Version]
- Ali, M.T.; Blicharska, N.; Shilpi, J.A.; Seidel, V. Investigation of the anti-TB potential of selected propolis constituents using a molecular docking approach. Sci. Rep. 2018, 8, 12238. [Google Scholar] [CrossRef]
- Young, T.A.; Delagoutte, B.; Endrizzi, J.A.; Falick, A.M.; Alber, T. Structure of Mycobacterium tuberculosis PknB supports a universal activation mechanism for Ser/Thr protein kinases. Nat. Struct. Biol. 2003, 10, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Gay, L.M.; Ng, H.L.; Alber, T. A conserved dimer and global conformational changes in the structure of apo-PknE Ser/Thr protein kinase from Mycobacterium tuberculosis. J. Mol. Biol. 2006, 360, 409–420. [Google Scholar] [CrossRef]
- Greenstein, A.E.; Echols, N.; Lombana, T.N.; King, D.S.; Alber, T. Allosteric activation by dimerization of the PknD receptor Ser/Thr protein kinase from Mycobacterium tuberculosis. J. Biol. Chem. 2007, 282, 11427–11435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cannell, R.J.P. Natural Products Isolation, Methods in Biotechnology; Humana Press: Totowa, NJ, USA, 1998. [Google Scholar]
- Patil, P.; Shettigar, R. An advancement of analytical techniques in herbal research. J. Adv. Sci. Res. 2010, 1, 8–14. [Google Scholar]
- Baker, P.J.; Slots, J.; Genco, R.J.; Evans, R.T. Minimal inhibitory concentrations of various antimicrobial agents for human oral anaerobic bacteria. Antimicrob. Agents Chemother. 1983, 24, 420–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dahiya, P.; Purkayastha, S. Phytochemical screening and antimicrobial activity of some medicinal plants against multi-drug resistant bacteria from clinical isolates. Indian J. Pharm. Sci. 2012, 74, 443–450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koua, F.H.M.; Babiker, H.A.; Halfawi, A.; Ibrahim, R.O.; Abbas, F.M.; Elgaail, E.I.; Khalafallah, M.M. Phytochemical and biolofical study of Striga hermonthica (Del.) Benth callus and intact plant. Res. Pharm. Biotechnol. 2011, 3, 85–92. [Google Scholar]
- Vollekova, A.D.; Kostalova, R.; Sochorova, R. Isoquinoline alkaloids from Mahonia aquifolium stem bark is active against Malassezia sp. Folia. Microbiology 2001, 46, 107–111. [Google Scholar] [CrossRef]
- Taylor, P.C.; Schoenknecht, F.D.; Sherris, J.C.; Linner, E.C. Determination of minimum bactericidal concentrations of oxacillin for Staphylococcus aureus: Influence and significance of technical factors. Antimicrob. Agents Chemother. 1983, 23, 142–150. [Google Scholar] [CrossRef] [Green Version]
- Lazarevic, J.; Kolarevic, A.; Stojanovic, G.; Smelcerovic, A.; Ciuffred, P.; Santaniello, E. Synthesis, antimicrobial activity and in silico studies on eugenol eters. Acta Chim. Slov. 2018, 65, 801–810. [Google Scholar] [CrossRef]
- Banfi, D.; Patiny, L. Resurrecting and processing NMR spectra on-line. Chimia 2008, 62, 280–281. [Google Scholar] [CrossRef]
- Carrasco, A.H.; Espinoza, C.L.; Cardile, V.; Gallardo, C.; Cardona, W.; Lombardo, L.; Catalán, M.K.; Cuellar, F.M.; Russo, A. Eugenol and its synthetic analogues inhibit cell growth of human cancer cells (Part I). J. Braz. Chem. Soc. 2008, 19, 543–548. [Google Scholar] [CrossRef] [Green Version]
- Dhara, L.; Tripathi, A. Antimicrobial activity of eugenol and cinnamaldehyde against extended spectrum beta lactamase producing enterobacteriaceae by in vitro and molecular docking analysis. Eur. J. Integr. Med. 2013, 5, 527–536. [Google Scholar] [CrossRef]
- Thosar, N.; Basak, S.; Bahadure, R.N.; Rajurkar, M. Antimicrobial efficacy of five essential oils against oral pathogens: An in vitro study. Eur. J. Dent. 2013, 7, S071–S077. [Google Scholar] [CrossRef] [Green Version]
- Hipólito, T.M.M.; Bastos, G.T.L.; Barbosa, T.W.L.; de Souza, T.B.; Coelho, L.F.L.; Dias, A.L.T.; Rodriguez, I.C.; Dos Santos, M.H.; Dias, D.F.; Franco, L.L.; et al. Synthesis, activity, and docking studies of eugenol- based glucosides as new agents against Candida sp. Chem. Biol. Drug Des. 2018, 92, 1514–1524. [Google Scholar] [CrossRef]
- Xu, J.; Wang, J.; Zhou, J.; Xu, C.-L.; Huang, B.; Xing, Y.; Wang, B.; Luo, R.; Wang, Y.-C.; You, X.-F.; et al. A novel protein kinase inhibitor IMB-YH-8 with anti-tuberculosis activity. Sci. Rep. 2017, 7, 5093. [Google Scholar] [CrossRef] [Green Version]

| Bacterial Isolates | Zone of Inhibition (mm) | ||||
|---|---|---|---|---|---|
| Fraction-I | Fraction-II | Fraction-III | Methanol 5% | Amox. | |
| E. coli | 22.0 ± 3.00 | 5.33 ± 1.52 | 4.33 ± 2.52 | n.d | 28.7 ± 1.15 |
| K. pneumoniae | 15.0 ± 4.58 | 5.67 ± 2.31 | 5.33 ± 1.53 | n.d | 26.7 ± 0.58 |
| Proteus sp. | 29.0 ± 2.00 | 4.67 ± 2.08 | 3.33 ± 1.53 | n.d | 30.7 ± 1.15 |
| P. aeruginosa | 24.7 ± 3.51 | 7.67 ± 3.06 | 4.33 ± 1.15 | n.d | 25.4 ± 1.53 |
| S. aureus | 24.3 ± 4.16 | 6.00 ± 1.73 | 3.61 ± 2.08 | n.d | 23.0 ± 1.00 |
| S. typhi | 28.0 ± 5.57 | 2.00 ± 1.00 | 3.00 ± 2.00 | n.d | 28.0 ± 2.00 |
| S. pneumonia | 26.7 ± 1.53 | 5.33 ± 1.53 | 3.33 ± 1.52 | n.d | 21.7 ± 1.53 |
| Bacterial Isolates | MIC (mg/mL) | MBC (mg/mL) |
|---|---|---|
| E. coli | 0.31 | 5.00 |
| K. pneumoniae | 1.25 | 10.0 |
| Proteus sp. | 0.63 | 5.00 |
| P. aeruginosa | 1.25 | 5.00 |
| S. aureus | 0.63 | 10.0 |
| S. typhi | 1.25 | n.d |
| S. pneumonia | 2.50 | n.d |
| Peak# | Name | Formula | Molecular Weight (g/mol) | Retention Time (min) | Retention Index | Area (%) |
|---|---|---|---|---|---|---|
| 1 | Phenol, 2-methoxy-4-(2-propenyl)-, acetate (acetyleugenol) | C12H14O3 | 206 | 8.242 | 1552 | 25.66 |
| 2 | Dodecanoic acid | C12H24O2 | 200 | 11.425 | 1570 | 7.11 |
| 3 | Tetradecanoic acid | C14H28O2 | 228 | 13.642 | 1769 | 3.01 |
| 4 | Hexadecanoic acid, methyl ester | C17H34O2 | 270 | 15.625 | 1878 | 3.35 |
| 5 | n-Hexadecanoic acid | C16H32O2 | 256 | 16.817 | 1968 | 22.99 |
| 6 | 3,4,5-Trihydroxybenzaldehyde | C7H6O4 | 154 | 18.325 | 1644 | 1.47 |
| 7 | 11-Octadecanoic acid, methyl ester | C19H36O2 | 296 | 18.900 | 2085 | 3.19 |
| 8 | Heneicosanoic acid, methyl ester | C22H44O2 | 340 | 19.267 | 2375 | 1.09 |
| 9 | Erucic acid | C22H42O2 | 338 | 19.958 | 2572 | 19.87 |
| 10 | Octadecanoic acid | C18H36O2 | 284 | 20.150 | 2167 | 9.54 |
| 11 | 1,E-11,Z-13-Octadecatriene | C18H32 | 248 | 21.617 | 1817 | 1.52 |
| 12 | 13-Oxabicyclo[9.3.1]pentadecane | C14H26O | 210 | 23.492 | 1690 | 0.38 |
| 13 | Methyl hexacosanoate | C27H54O2 | 410 | 23.858 | 2872 | 0.45 |
| 14 | 1-Gala-1-ido-octose | C8H16O8 | 240 | 25.567 | 2221 | 0.39 |
| Compound | Binding Energy (kcal/mol) | Reference | |
|---|---|---|---|
| MtPknE | MtPknB | ||
| EUG-1 | −11.08 | −10.46 | This study |
| EUG-2 | −10.29 | −10.19 | This study |
| EUG-3 | −10.05 | −9.64 | This study |
| EUG-4 | −9.25 | −9.35 | This study |
| Acetyleugenol | −8.53 | −7.60 | This study |
| Mitoxantrone | n.a * | −10.8 | [42] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdulhamid, A.; Awad, T.A.; Ahmed, A.E.; Koua, F.H.M.; Ismail, A.M. Acetyleugenol from Acacia nilotica (L.) Exhibits a Strong Antibacterial Activity and Its Phenyl and Indole Analogues Show a Promising Anti-TB Potential Targeting PknE/B Protein Kinases. Microbiol. Res. 2021, 12, 1-15. https://doi.org/10.3390/microbiolres12010001
Abdulhamid A, Awad TA, Ahmed AE, Koua FHM, Ismail AM. Acetyleugenol from Acacia nilotica (L.) Exhibits a Strong Antibacterial Activity and Its Phenyl and Indole Analogues Show a Promising Anti-TB Potential Targeting PknE/B Protein Kinases. Microbiology Research. 2021; 12(1):1-15. https://doi.org/10.3390/microbiolres12010001
Chicago/Turabian StyleAbdulhamid, Abubakar, Talal Ahmed Awad, Abdalla E. Ahmed, Faisal Hammad Mekky Koua, and Amar Mohamed Ismail. 2021. "Acetyleugenol from Acacia nilotica (L.) Exhibits a Strong Antibacterial Activity and Its Phenyl and Indole Analogues Show a Promising Anti-TB Potential Targeting PknE/B Protein Kinases" Microbiology Research 12, no. 1: 1-15. https://doi.org/10.3390/microbiolres12010001
APA StyleAbdulhamid, A., Awad, T. A., Ahmed, A. E., Koua, F. H. M., & Ismail, A. M. (2021). Acetyleugenol from Acacia nilotica (L.) Exhibits a Strong Antibacterial Activity and Its Phenyl and Indole Analogues Show a Promising Anti-TB Potential Targeting PknE/B Protein Kinases. Microbiology Research, 12(1), 1-15. https://doi.org/10.3390/microbiolres12010001






