Leptin, Nesfatin-1, Glucagon-like Peptide 1, and Short-Chain Fatty Acids in Colon Cancer and Inflammatory Bowel Disease
Abstract
1. Introduction
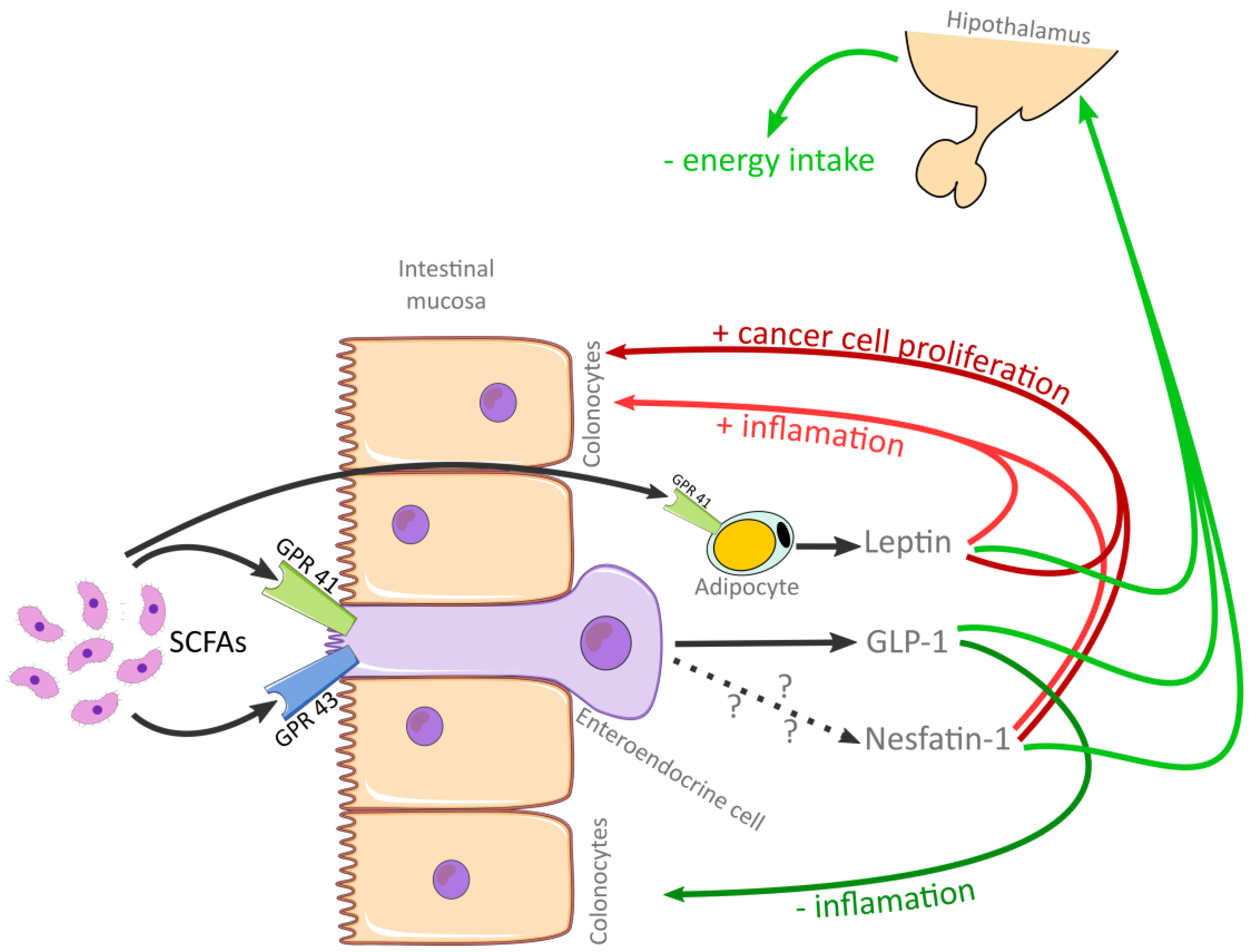
1.1. Short-Chain Fatty Acids, Leptin, Glucagon-like Peptide-1, and Nesfatin-1
1.2. Inflammatory Bowel Disease and Colorectal Cancer
2. Materials and Methods
2.1. Sample Selection and Inclusion
2.2. Laboratory Analysis
2.3. Statistical Analysis
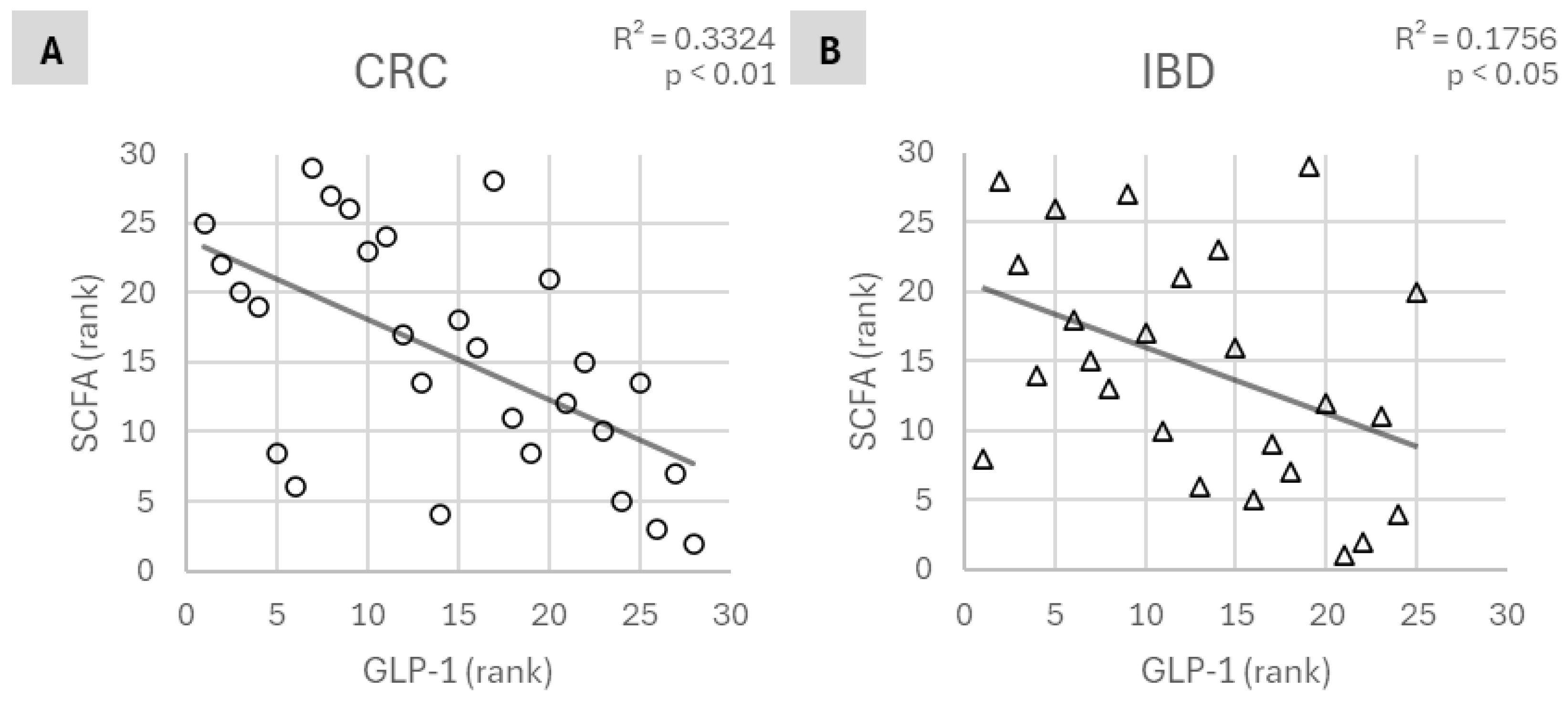
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CRC | Colorectal Cancer |
| IBD | Inflammatory Bowel Disease |
| CD | Crohn’s Disease |
| UC | Ulcerative Colitis |
| SCFAs | Short-Chain Fatty Acids |
| GLP-1 | Glucagon-like Peptide 1 |
| Hb | Hemoglobin |
| Ht | Hematocrit |
| MCV | Mean Corpuscular Volume |
| MCHC | Mean Corpuscular Hemoglobin Concentration |
| PLT | Platelets |
| WBC | White Blood Cells |
| Neu | Neutrophils |
| Eo | Eosinophils |
| Ba | Basophils |
References
- Ilyés, T.; Pop, M.; Surcel, M.; Pop, D.M.; Rusu, R.; Silaghi, C.N.; Zaharie, G.C.; Crăciun, A.M. First Comparative Evaluation of Short-Chain Fatty Acids and Vitamin-K-Dependent Proteins Levels in Mother–Newborn Pairs at Birth. Life 2023, 13, 847. [Google Scholar] [CrossRef]
- Ilyés, T.; Silaghi, C.N.; Crăciun, A.M. Diet-Related Changes of Short-Chain Fatty Acids in Blood and Feces in Obesity and Metabolic Syndrome. Biology 2022, 11, 1556. [Google Scholar] [CrossRef]
- Xiong, Y.; Miyamoto, N.; Shibata, K.; Valasek, M.A.; Motoike, T.; Kedzierski, R.M.; Yanagisawa, M. Short-Chain Fatty Acids Stimulate Leptin Production in Adipocytes through the G Protein-Coupled Receptor GPR41. Proc. Natl. Acad. Sci. USA 2004, 101, 1045–1050. [Google Scholar] [CrossRef]
- Karaki, S.-I.; Tazoe, H.; Hayashi, H.; Kashiwabara, H.; Tooyama, K.; Suzuki, Y.; Kuwahara, A. Expression of the Short-Chain Fatty Acid Receptor, GPR43, in the Human Colon. J. Mol. Histol. 2008, 39, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshini, M.; Lednovich, K.; Xu, K.; Gough, S.; Wicksteed, B.; Layden, B.T. FFAR from the Gut Microbiome Crowd: SCFA Receptors in T1D Pathology. Metabolites 2021, 11, 302. [Google Scholar] [CrossRef] [PubMed]
- Psichas, A.; Sleeth, M.L.; Murphy, K.G.; Brooks, L.; Bewick, G.A.; Hanyaloglu, A.C.; Ghatei, M.A.; Bloom, S.R.; Frost, G. The Short Chain Fatty Acid Propionate Stimulates GLP-1 and PYY Secretion via Free Fatty Acid Receptor 2 in Rodents. Int. J. Obes. 2015, 39, 424–429. [Google Scholar] [CrossRef]
- Shin, Y.; Han, S.; Kwon, J.; Ju, S.; Choi, T.G.; Kang, I.; Kim, S.S. Roles of Short-Chain Fatty Acids in Inflammatory Bowel Disease. Nutrients 2023, 15, 4466. [Google Scholar] [CrossRef]
- Parada Venegas, D.; De la Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277. [Google Scholar] [CrossRef] [PubMed]
- Deleu, S.; Machiels, K.; Raes, J.; Verbeke, K.; Vermeire, S. Short Chain Fatty Acids and Its Producing Organisms: An Overlooked Therapy for IBD? eBioMedicine 2021, 66, 103293. [Google Scholar] [CrossRef]
- Stidham, R.W.; Higgins, P.D.R. Colorectal Cancer in Inflammatory Bowel Disease. Clin. Colon Rectal Surg. 2018, 31, 168–178. [Google Scholar] [CrossRef]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; McKee, L.J.; Bauer, T.L.; et al. Serum Immunoreactive-Leptin Concentrations in Normal-Weight and Obese Humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, F.C.; Fantuzzi, G. The Association of Short-Chain Fatty Acids and Leptin Metabolism: A Systematic Review. Nutr. Res. 2019, 72, 18–35. [Google Scholar] [CrossRef]
- Baggio, L.L.; Drucker, D.J. Biology of Incretins: GLP-1 and GIP. Gastroenterology 2007, 132, 2131–2157. [Google Scholar] [CrossRef]
- Tolhurst, G.; Heffron, H.; Lam, Y.S.; Parker, H.E.; Habib, A.M.; Diakogiannaki, E.; Cameron, J.; Grosse, J.; Reimann, F.; Gribble, F.M. Short-Chain Fatty Acids Stimulate Glucagon-Like Peptide-1 Secretion via the G-Protein–Coupled Receptor FFAR2. Diabetes 2012, 61, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Hills, R.D.; Pontefract, B.A.; Mishcon, H.R.; Black, C.A.; Sutton, S.C.; Theberge, C.R. Gut Microbiome: Profound Implications for Diet and Disease. Nutrients 2019, 11, 1613. [Google Scholar] [CrossRef]
- Kim, H.R. Obesity-Related Colorectal Cancer: The Role of Leptin. Ann. Coloproctol. 2015, 31, 209–210. [Google Scholar] [CrossRef][Green Version]
- Socol, C.T.; Chira, A.; Martinez-Sanchez, M.A.; Nuñez-Sanchez, M.A.; Maerescu, C.M.; Mierlita, D.; Rusu, A.V.; Ruiz-Alcaraz, A.J.; Trif, M.; Ramos-Molina, B. Leptin Signaling in Obesity and Colorectal Cancer. Int. J. Mol. Sci. 2022, 23, 4713. [Google Scholar] [CrossRef]
- Stattin, P.; Palmqvist, R.; Söderberg, S.; Biessy, C.; Ardnor, B.; Hallmans, G.; Kaaks, R.; Olsson, T. Plasma Leptin and Colorectal Cancer Risk: A Prospective Study in Northern Sweden. Oncol. Rep. 2003, 10, 2015–2021. [Google Scholar] [CrossRef]
- Trejo-Vazquez, F.; Garza-Veloz, I.; Villela-Ramirez, G.A.; Ortiz-Castro, Y.; Mauricio-Saucedo, P.; Cardenas-Vargas, E.; Diaz-Baez, M.; Cid-Baez, M.A.; Castañeda-Miranda, R.; Ortiz-Rodriguez, J.M.; et al. Positive Association Between Leptin Serum Levels and Disease Activity on Endoscopy in Inflammatory Bowel Disease: A Case-Control Study. Exp. Ther. Med. 2018, 15, 3336–3344. [Google Scholar] [CrossRef]
- Chouliaras, G.; Panayotou, I.; Margoni, D.; Mantzou, E.; Pervanidou, P.; Manios, Y.; Chrousos, G.P.; Roma, E. Circulating Leptin and Adiponectin and Their Relation to Glucose Metabolism in Children with Crohn’s Disease and Ulcerative Colitis. Pediatr. Res. 2013, 74, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Seino, Y.; Fukushima, M.; Yabe, D. GIP and GLP-1, the Two Incretin Hormones: Similarities and Differences. J. Diabetes Investig. 2010, 1, 8–23. [Google Scholar] [CrossRef]
- Dotania, K.; Tripathy, M.; Rai, U. Nesfatin-1 as a Crucial Mediator of Glucose Homeostasis in the Reptile, Hemidactylus Flaviviridis. Sci. Rep. 2024, 14, 31565. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, W.; Kaelber, D.C.; Xu, R.; Berger, N.A. GLP-1 Receptor Agonists and Colorectal Cancer Risk in Drug-Naive Patients with Type 2 Diabetes, with and without Overweight/Obesity. JAMA Oncol. 2024, 10, 256–258. [Google Scholar] [CrossRef]
- Gorelik, Y.; Ghersin, I.; Lujan, R.; Shlon, D.; Loewenberg Weisband, Y.; Ben-Tov, A.; Matz, E.; Zacay, G.; Dotan, I.; Turner, D.; et al. GLP-1 Analog Use Is Associated with Improved Disease Course in Inflammatory Bowel Disease: A Report from the Epi-IIRN. J. Crohn’s Colitis 2024, 19, jjae160. [Google Scholar] [CrossRef] [PubMed]
- Oh-I, S.; Shimizu, H.; Satoh, T.; Okada, S.; Adachi, S.; Inoue, K.; Eguchi, H.; Yamamoto, M.; Imaki, T.; Hashimoto, K.; et al. Identification of Nesfatin-1 as a Satiety Molecule in the Hypothalamus. Nature 2006, 443, 709–712. [Google Scholar] [CrossRef] [PubMed]
- Schalla, M.A.; Stengel, A. Current Understanding of the Role of Nesfatin-1. J. Endocr. Soc. 2018, 2, 1188–1206. [Google Scholar] [CrossRef]
- Beyaz, Ş.; Akbal, E. Increased Serum Nesfatin-1 Levels in Patients with Inflammatory Bowel Diseases. Postgrad. Med. J. 2022, 98, 446–449. [Google Scholar] [CrossRef]
- Kan, J.-Y.; Yen, M.-C.; Wang, J.-Y.; Wu, D.-C.; Chiu, Y.-J.; Ho, Y.-W.; Kuo, P.-L. Nesfatin-1/Nucleobindin-2 Enhances Cell Migration, Invasion, and Epithelial-Mesenchymal Transition via LKB1/AMPK/TORC1/ZEB1 Pathways in Colon Cancer. Oncotarget 2016, 7, 31336–31349. [Google Scholar] [CrossRef]
- Seyedian, S.S.; Nokhostin, F.; Malamir, M.D. A Review of the Diagnosis, Prevention, and Treatment Methods of Inflammatory Bowel Disease. J. Med. Life 2019, 12, 113–122. [Google Scholar] [CrossRef]
- Torres, J.; Mehandru, S.; Colombel, J.-F.; Peyrin-Biroulet, L. Crohn’s Disease. Lancet 2017, 389, 1741–1755. [Google Scholar] [CrossRef]
- Magro, F.; Gionchetti, P.; Eliakim, R.; Ardizzone, S.; Armuzzi, A.; Barreiro-de Acosta, M.; Burisch, J.; Gecse, K.B.; Hart, A.L.; Hindryckx, P.; et al. Third European Evidence-Based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 1: Definitions, Diagnosis, Extra-Intestinal Manifestations, Pregnancy, Cancer Surveillance, Surgery, and Ileo-Anal Pouch Disorders. J. Crohn’s Colitis 2017, 11, 649–670. [Google Scholar] [CrossRef]
- Feuerstein, J.D.; Moss, A.C.; Farraye, F.A. Ulcerative Colitis. Mayo Clin. Proc. 2019, 94, 1357–1373. [Google Scholar] [CrossRef]
- Ko, I.K.; Kim, B.-G.; Awadallah, A.; Mikulan, J.; Lin, P.; Letterio, J.J.; Dennis, J.E. Targeting Improves MSC Treatment of Inflammatory Bowel Disease. Mol. Ther. 2010, 18, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, J.; Mortensen, P.B. Hydrogen Sulfide and Colonic Epithelial Metabolism: Implications for Ulcerative Colitis. Dig. Dis. Sci. 2001, 46, 1722–1732. [Google Scholar] [CrossRef]
- Levine, J.; Ellis, C.J.; Furne, J.K.; Springfield, J.; Levitt, M.D. Fecal Hydrogen Sulfide Production in Ulcerative Colitis. Am. J. Gastroenterol. 1998, 93, 83–87. [Google Scholar] [CrossRef]
- Duan, B.; Zhao, Y.; Bai, J.; Wang, J.; Duan, X.; Luo, X.; Zhang, R.; Pu, Y.; Kou, M.; Lei, J.; et al. Colorectal Cancer: An Overview. In Gastrointestinal Cancers; Morgado-Diaz, J.A., Ed.; Exon Publications: Brisbane, Australia, 2022; ISBN 978-0-6453320-6-3. [Google Scholar]
- Fleming, M.; Ravula, S.; Tatishchev, S.F.; Wang, H.L. Colorectal Carcinoma: Pathologic Aspects. J. Gastrointest. Oncol. 2012, 3, 3. [Google Scholar] [CrossRef]
- Remo, A.; Fassan, M.; Vanoli, A.; Bonetti, L.R.; Barresi, V.; Tatangelo, F.; Gafà, R.; Giordano, G.; Pancione, M.; Grillo, F.; et al. Morphology and Molecular Features of Rare Colorectal Carcinoma Histotypes. Cancers 2019, 11, 1036. [Google Scholar] [CrossRef]
- Crippa, J.; Grass, F.; Achilli, P.; Behm, K.T.; Mathis, K.L.; Day, C.N.; Harmsen, W.S.; Mari, G.M.; Larson, D.W. Surgical Approach to Transverse Colon Cancer: Analysis of Current Practice and Oncological Outcomes Using the National Cancer Database. Dis. Colon Rectum 2021, 64, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Tsujinaka, S.; Miura, T.; Kitamura, Y.; Suzuki, H.; Shibata, C. Inflammatory Bowel Disease and Colorectal Cancer: Epidemiology, Etiology, Surveillance, and Management. Cancers 2023, 15, 4154. [Google Scholar] [CrossRef] [PubMed]
- Mattar, M.C.; Lough, D.; Pishvaian, M.J.; Charabaty, A. Current Management of Inflammatory Bowel Disease and Colorectal Cancer. Gastrointest. Cancer Res. 2011, 4, 53–61. [Google Scholar]
- Liang, Y.; Ma, Y.; Wang, K.; Xiang, M.; Yi, B. NUCB-2/Nesfatin-1 Promotes the Proliferation of Nasopharyngeal Carcinoma Cells. Cancer Cell Int. 2023, 23, 181. [Google Scholar] [CrossRef]
- Skorupska, A.; Lenda, R.; Ożyhar, A.; Bystranowska, D. The Multifaceted Nature of Nucleobindin-2 in Carcinogenesis. Int. J. Mol. Sci. 2021, 22, 5687. [Google Scholar] [CrossRef]
- Haemmerle, R.J.; Jatoi, A. Loss of Appetite in Patients with Cancer: An Update on Characterization, Mechanisms, and Palliative Therapeutics. Curr. Opin. Support. Palliat. Care 2023, 17, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Kelesidis, T.; Kelesidis, I.; Chou, S.; Mantzoros, C.S. Narrative Review: The Role of Leptin in Human Physiology: Emerging Clinical Applications. Ann. Intern. Med. 2010, 152, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Bakal, U.; Saraç, M.; Ciftci, H.; Tartar, T.; Kazez, A.; Aydin, S. Leptin and NUCB2/Nesfatin-1 in Acute Appendicitis. Int. J. Clin. Med. 2015, 6, 919–927. [Google Scholar] [CrossRef]
- Moran, G.W.; Thapaliya, G. The Gut–Brain Axis and Its Role in Controlling Eating Behavior in Intestinal Inflammation. Nutrients 2021, 13, 981. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, C.; Zhang, H.; Li, L.; Fan, T.; Jin, Z. The Alleviating Effect and Mechanism of GLP-1 on Ulcerative Colitis. Aging 2023, 15, 8044–8060. [Google Scholar] [CrossRef]
- Zatorski, H.; Sałaga, M.; Fichna, J. Role of Glucagon-like Peptides in Inflammatory Bowel Diseases—Current Knowledge and Future Perspectives. Naunyn-Schmiedeb. Arch. Pharmacol. 2019, 392, 1321–1330. [Google Scholar] [CrossRef]
- Singh, N.; Baby, D.; Rajguru, J.P.; Patil, P.B.; Thakkannavar, S.S.; Pujari, V.B. Inflammation and Cancer. Ann. Afr. Med. 2019, 18, 121–126. [Google Scholar] [CrossRef]
- Zeng, Y.; Wu, Y.; Zhang, Q.; Xiao, X. Crosstalk between Glucagon-like Peptide 1 and Gut Microbiota in Metabolic Diseases. mBio 2024, 15, e02032-23. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, H.; Chen, T.; Shi, L.; Wang, D.; Tang, D. Regulatory Role of Short-Chain Fatty Acids in Inflammatory Bowel Disease. Cell Commun. Signal. 2022, 20, 64. [Google Scholar] [CrossRef] [PubMed]

| Parameter | Study Group | p | ||||
|---|---|---|---|---|---|---|
| CRC | IBD | CD | UC | Control | ||
| Age (Years) | 66 (40–84) | 38 (19–78) | 38 (19–78) | 42 (19–75) | 28 (25–76) | <0.01 |
| Disease Activity Score | N/A | N/A | CDAI: 127.6 (10–400) | MAYO: 2.72 ± 2.3 | N/A | N/A |
| Nesfatin-1 (pg/mL) | 6185.6 (338.2–9315.6) | 2219.6 (258–5624.5) | 1506.3 (765–4069.4) | 3043.5 (258–5624.5) | 2729.85 (433.5–6514.2) | <0.05 |
| SCFAs (pg/mL) | 217.4 (104.7–1047.71) | 238.8 (114.77–504.36) | 277.26 (122.79–504.36) | 225.10 (114.77–445.76) | 201.3 (126.49–7103.72) | 0.746 |
| Leptin (pg/mL) | 2376.5 (828–12,493) | 2143 (944–5774) | 2104.5 (1601–5480.5) | 2342 (944–5774) | 2584.75 (944–7130) | 0.376 |
| GLP-1 (pg/mL) | 207.55 (23–611.4) | 132.4 (26–422) | 130.7 (26–373.7) | 167.95 (26.4–422) | 214.4 (30.9–687.1) | 0.323 |
| Parameter | Study Group | p | |||
|---|---|---|---|---|---|
| CRC | IBD | CD | UC | ||
| Hb (g/dL) | 10.9 (±1.69) | 13.46 (±2.02) | 13.8 (±1.97) | 13.26 (±2.08) | <0.01 |
| Ht (%) | 33.54 (±5.24) | 39.83 (±5.33) | 40.67 (±5.13) | 39.32 (±5.53) | <0.01 |
| MCV (fl) | 81.6 (±8.17) | 88.03 (±6.21) | 85.79 (±5.27) | 89.4 (±6.48) | <0.01 |
| MCHC (g/dL) | 32.86 (±1.54) | 29.72 (±2.53) | 29.09 (±2.64) | 30.11 (±2.46) | <0.01 |
| # PLT | 281 (86–718) | 290 (164–651) | 266 (164–373) | 316 (180–651) | 0.591 |
| # WBC | 9050 (4230–30,460) | 7920 (3220–18,590) | 7920 (3220–18,590) | 8045 (5510–13,210) | 0.2 |
| # Neu | 6020 (3100–19,200) | 4920 (1580–15,790) | 4920 (1580–15,790) | 3445 (2880–9280) | 0.125 |
| # Eo | 120 (0–960) | 130 (0–920) | 150 (0–450) | 90 (0–920) | 0.345 |
| # Ba | 30 (0–100) | 40 (10–100) | 30 (10–70) | 45 (10–100) | 0.239 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ilyés, T.; Grama, P.; Gheorghe, S.R.; Anton, V.; Silaghi, C.N.; Crăciun, A.M. Leptin, Nesfatin-1, Glucagon-like Peptide 1, and Short-Chain Fatty Acids in Colon Cancer and Inflammatory Bowel Disease. Gastroenterol. Insights 2025, 16, 32. https://doi.org/10.3390/gastroent16030032
Ilyés T, Grama P, Gheorghe SR, Anton V, Silaghi CN, Crăciun AM. Leptin, Nesfatin-1, Glucagon-like Peptide 1, and Short-Chain Fatty Acids in Colon Cancer and Inflammatory Bowel Disease. Gastroenterology Insights. 2025; 16(3):32. https://doi.org/10.3390/gastroent16030032
Chicago/Turabian StyleIlyés, Tamás, Paul Grama, Simona R. Gheorghe, Vlad Anton, Ciprian N. Silaghi, and Alexandra M. Crăciun. 2025. "Leptin, Nesfatin-1, Glucagon-like Peptide 1, and Short-Chain Fatty Acids in Colon Cancer and Inflammatory Bowel Disease" Gastroenterology Insights 16, no. 3: 32. https://doi.org/10.3390/gastroent16030032
APA StyleIlyés, T., Grama, P., Gheorghe, S. R., Anton, V., Silaghi, C. N., & Crăciun, A. M. (2025). Leptin, Nesfatin-1, Glucagon-like Peptide 1, and Short-Chain Fatty Acids in Colon Cancer and Inflammatory Bowel Disease. Gastroenterology Insights, 16(3), 32. https://doi.org/10.3390/gastroent16030032





