Evaluation of the Correlation between the rs4918 Polymorphism of AHSG Gene and Coronary Artery Calcification in Patients with Coronary Artery Disease
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Genotype Analysis
2.3. Statistical Analysis
3. Results
3.1. Patients Characteristics
3.2. Comparison of Coronary Artery Calcium Score between Different Groups
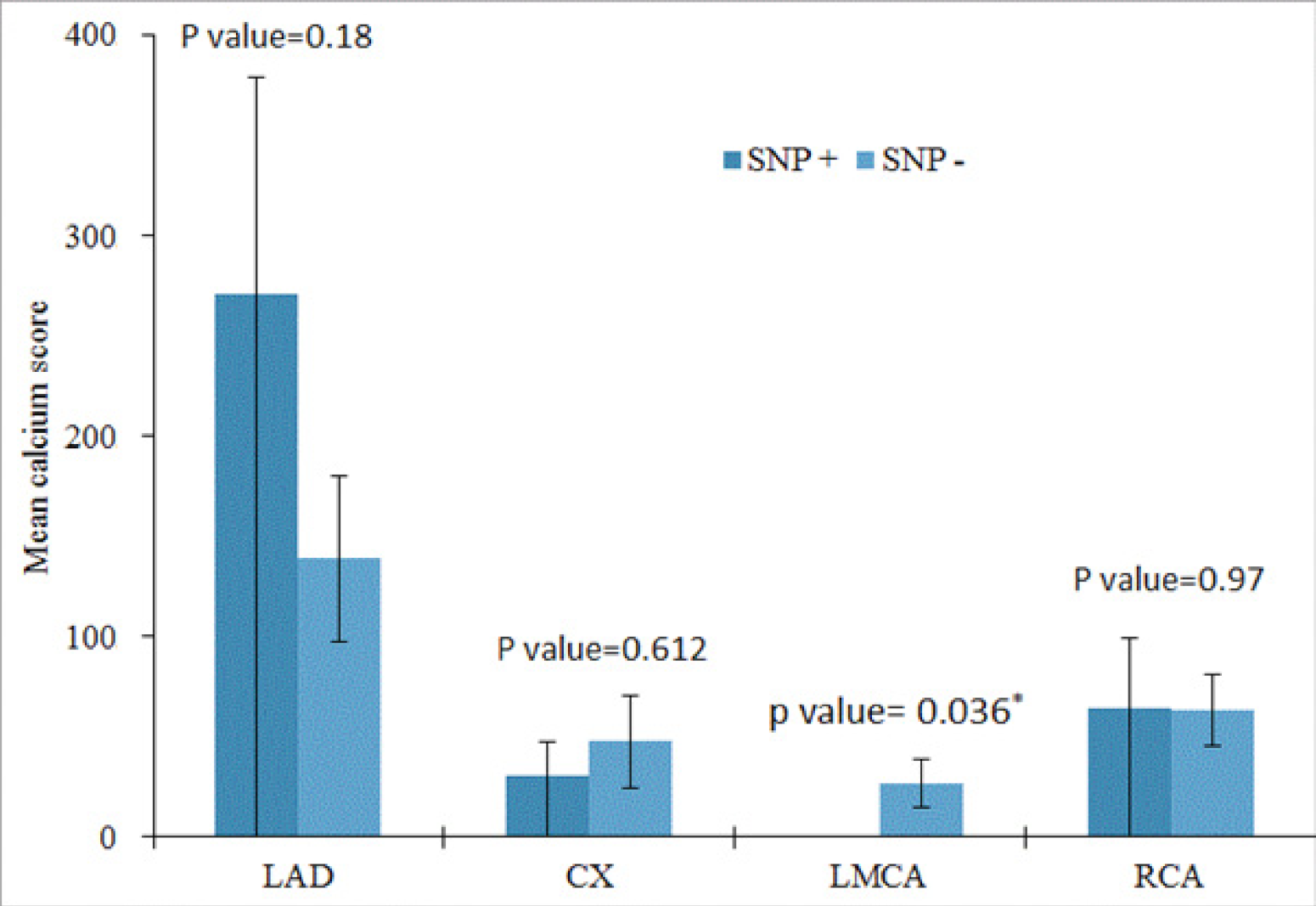
3.3. Comparison between Calcium Scores of the Main Coronary Arteries in the Presence and Absence of rs4918 SNP
3.4. Frequency of rs4918 SNP in Different Groups
4. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lusis, A.J. Atherosclerosis. Nature 2000, 407, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Sage, A.P.; Tintut, Y.; Demer, L.L. Regulatory mechanisms in vascular calcification. Nat. Rev. Cardiol. 2010, 7, 528–536. [Google Scholar] [CrossRef]
- Peyser, P.A.; Bielak, L.F.; Chu, J.S.; Turner, S.T.; Ellsworth, D.L.; Boerwinkle, E.; Sheedy, P.F. Heritability of Coronary Artery Calcium Quantity Measured by Electron Beam Computed Tomography in Asymptomatic Adults. Circulation 2002, 106, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Fisher, E.; Stefan, N.; Saar, K.; Drogan, D.; Boeing, H.; Fritsche, A.; Joost, H.-G.; Häring, H.-U.; Hübner, N.; Boeing, H.; et al. Association of AHSG Gene Polymorphisms With Fetuin-A Plasma Levels and Cardiovascular Diseases in the EPIC-Potsdam Study. Circ. Cardiovasc. Genet. 2009, 2, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Cuspidi, C.; Sala, C. Is fetuin-A a biomarker of preclinical atherosclerosis in essential hypertension? Hypertens. Res. 2012, 36, 104–106. [Google Scholar] [CrossRef]
- Mori, K.; Emoto, M.; Inaba, M. Fetuin-A: A multifunctional protein. Recent Pat. Endocr. Metab. Immune Drug Discov. 2011, 5, 124–146. [Google Scholar] [CrossRef]
- Ombrellino, M.; Wang, H.; Yang, H.; Zhang, M.; Vishnubhakat, J.; Frazier, A.; Scher, L.A.; Friedman, S.G.; Tracey, K.J. Fetuin, a negative acute phase protein, attenuates tnf synthesis and the innate inflammatory response to carrageenan. Shock 2001, 15, 181–185. [Google Scholar] [CrossRef]
- Schafer, C.; Heiss, A.; Schwarz, A.; Westenfeld, R.; Ketteler, M.; Floege, J.; Müller-Esterl, W.; Schinke, T.; Jahnen-Dechent, W. The serum protein alpha 2-Heremans-Schmid glycoprotein/fetuin-A is a systemically acting inhibitor of ectopic calcification. J. Clin. Invest. 2003, 112, 357–366. [Google Scholar] [CrossRef]
- Fiore, C.E.; Celotta, G.; Politi, G.G.; Di Pino, L.; Castelli, Z.; Mangiafico, R.A.; Signorelli, S.S.; Pennisi, P. Association of high alpha2-Heremans–Schmid glycoprotein/fetuin concentration in serum and intima-media thickness in patients with atherosclerotic vascular disease and low bone mass. Atherosclerosis 2007, 195, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Ketteler, M.; Bongartz, P.; Westenfeld, R.; Wildberger, J.E.; Mahnken, A.H.; Böhm, R.; Metzger, T.; Wanner, C.; Jahnen-Dechent, W.; Floege, J. Association of low fetuin-A (AHSG) concentrations in serum with cardiovascular mortality in patients on dialysis: A cross-sectional study. Lancet 2003, 361, 827–833. [Google Scholar] [CrossRef]
- Stenvinkel, P.; Wang, K.; Qureshi, A.R.; Axelsson, J.; Pecoits-Filho, R.; Gao, P.; Barany, P.; Lindholm, B.; Jogestrand, T.; Heimberger, O.; et al. Low fetuin-A levels are associated with cardiovascular death: Impact of variations in the gene encoding fetuin. Kidney Int. 2005, 67, 2383–2392. [Google Scholar] [CrossRef]
- Lehtinen, A.B.; Burdon, K.P.; Lewis, J.P.; Langefeld, C.D.; Ziegler, J.T.; Rich, S.S.; Register, T.C.; Carr, J.J.; Freedman, B.I.; Bowden, D.W. Association of α2-Heremans-Schmid Glycoprotein Polymorphisms with Subclinical Atherosclerosis. J. Clin. Endocrinol. Metab. 2007, 92, 345–352. [Google Scholar] [CrossRef][Green Version]
- Roos, M.; Richart, T.; Kouznetsova, T.; Von Eynatten, M.; Lutz, J.; Heemann, U.; Baumann, M.; Staessen, J.A. Fetuin-A and arterial stiffness in patients with normal kidney function. Regul. Pept. 2009, 154, 39–43. [Google Scholar] [CrossRef]
- Cozzolino, M.; Biondi, M.L.; Galassi, A.; Gallieni, M.; D’Eril, G.V.M.; Brancaccio, D. Gene Polymorphisms and Serum Alpha-2-Heremans-Schmid Levels in Italian Haemodialysis Patients. Am. J. Nephrol. 2007, 27, 639–642. [Google Scholar] [CrossRef]
- Bellia, C.; Agnello, L.; Sasso, B.L.; Milano, S.; Bivona, G.; Scazzone, C.; Pivetti, A.; Novo, G.; Palermo, C.; Bonomo, V.; et al. Fetuin-A is Associated to Serum Calcium and AHSG T256S Genotype but Not to Coronary Artery Calcification. Biochem. Genet. 2016, 54, 222–231. [Google Scholar] [CrossRef]
- Verduijn, M.; Prein, R.A.; Stenvinkel, P.; Carrero, J.J.; Le Cessie, S.; Witasp, A.; Nordfors, L.; Krediet, R.T.; Boeschoten, E.W.; Dekker, F. Is fetuin-A a mortality risk factor in dialysis patients or a mere risk marker? A Mendelian randomization approach. Nephrol. Dial. Transplant. 2010, 26, 239–245. [Google Scholar] [CrossRef]
- Roos, M.; Lutz, J.; Salmhofer, H.; Luppa, P.; Knauß, A.; Braun, S.; Martinof, S.; Schömig, A.; Heemann, U.; Kastrati, A.; et al. Relation between plasma fibroblast growth factor-23, serum fetuin-A levels and coronary artery calcification evaluated by multislice computed tomography in patients with normal kidney function. Clin. Endocrinol. 2008, 68, 660–665. [Google Scholar] [CrossRef]
- Osawa, M.; Umetsu, K.; Ohki, T.; Nagasawa, T.; Suzuki, T.; Takeichi, S. Molecular evidence for human alpha 2-HS glycoprotein (AHSG) polymorphism. Hum. Genet. 1997, 99, 18–21. [Google Scholar] [CrossRef]
- Falquerho, L.; Paquereau, L.; Vilarem, M.J.; Galas, S.; Patey, G.; Le Cam, A. Functional characterization of the promoter of pp63, a gene encoding a natural inhibitor of the insulin receptor tyrosine kinase. Nucleic Acids Res. 1992, 20, 1983–1990. [Google Scholar] [CrossRef][Green Version]
- Gangneux, C.; Daveau, M.; Hiron, M.; Derambure, C.; Papaconstantinou, J.; Salier, J.P. The inflammation-induced down-regulation of plasma Fetuin-A (α2HS-Glycoprotein) in liver results from the loss of interaction between long C/EBP isoforms at two neighbouring binding sites. Nucleic Acids Res. 2003, 31, 5957–5970. [Google Scholar] [CrossRef]
- Daveau, M.; Davrinche, C.; Djelassi, N.; Lemetayer, J.; Julen, N.; Hiron, M.; Arnaud, P.; Lebreton, J. Partial hepatectomy and mediators of inflammation decrease the expression of liver α2 -HS glycoprotein gene in rats. FEBS Lett. 1990, 273, 79–81. [Google Scholar] [CrossRef]
- Daveau, M.; Davrinche, C.-; Julen, N.; Hiron, M.; Amaud, P.; Lebreton, J.-P. The synthesis of human α-2-HS glycoprotein is down-regulated by cytokines in hepatoma HepG2 cells. FEBS Lett. 1988, 241, 191–194. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Y.; Chen, Q.; Li, Q.; Li, Y.; Ling, W. Lower Plasma Fetuin-A Levels Are Associated with a Higher Mortality Risk in Patients with Coronary Artery Disease. Arter. Thromb. Vasc. Biol. 2017, 37, 2213–2219. [Google Scholar] [CrossRef]
- Jensen, M.K.; Bartz, T.M.; Mukamal, K.J.; Djoussé, L.; Kizer, J.R.; Tracy, R.P.; Zieman, S.J.; Rimm, E.B.; Siscovick, D.S.; Shlipak, M.; et al. Fetuin-A, Type 2 Diabetes, and Risk of Cardiovascular Disease in Older Adults. Cardiovasc. Health Study 2013, 36, 1222–1228. [Google Scholar] [CrossRef]
- Laughlin, G.A.; Cummins, K.M.; Wassel, C.L.; Daniels, L.B.; Ix, J.H. The Association of Fetuin-A with Cardiovascular Disease Mortality in Older Community-Dwelling Adults: The Rancho Bernardo study. J. Am. Coll. Cardiol. 2012, 59, 1688–1696. [Google Scholar] [CrossRef]
- Blake, G.J.; Ridker, P.M. C-reactive protein and other inflammatory risk markers in acute coronary syndromes. J. Am. Coll. Cardiol. 2003, 41, S37–S42. [Google Scholar] [CrossRef]
| Characteristics | Patients (n = 81) | Control (n = 42) |
|---|---|---|
| Male, % | 72.84 | 35.71 |
| Age (Year) | 57.08 ± 10.54 | 34.47 ± 10.46 |
| Hypertension, % | 48.14 | 0 |
| Dyslipidemia, % | 62.96 | 0 |
| Diabetes mellitus type II, % | 18.51 | 0 |
| Smoking status, % | 30.86 | 0 |
| Family history, % | 45.67 | 0 |
| rs4918 SNP, % frequency | 38.27 | 35.71 |
| Calcium score (mean ± STD) | 348.29 ± 588.7 | 56.3 ± 26.62 |
| Mean Calcium Score (SNP +) ± Std. Error | Mean Calcium Score (SNP −) ± Std. Error | p Value | |
|---|---|---|---|
| LAD | 2.7111E+02 ± 108.12 | 1.3879E+02 ± 41.76 | 0.180 |
| CX | 30.38 ± 17.07 | 47.51 ± 22.66 | 0.612 |
| LMCA | 0.14 ± 0.14 | 27.04 ± 12.18 | 0.036 |
| RCA | 64.58 ± 35.03 | 63.24 ± 18.06 | 0.970 |
| Calcium Group | CACS | Frequency of the rs4918 SNP | p Value |
|---|---|---|---|
| Group 1 | 0–100 | 37.66 | 0.9 |
| Group 2 | 101–400 | 36.36 | |
| Group 3 | >400 | 34.78 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmadihosseini, Z.; Moeinian, M.; Nazemi, S.; Elyasi, S.; Mohammadpour, A.H. Evaluation of the Correlation between the rs4918 Polymorphism of AHSG Gene and Coronary Artery Calcification in Patients with Coronary Artery Disease. Cardiogenetics 2020, 10, 33-41. https://doi.org/10.3390/cardiogenetics10020007
Ahmadihosseini Z, Moeinian M, Nazemi S, Elyasi S, Mohammadpour AH. Evaluation of the Correlation between the rs4918 Polymorphism of AHSG Gene and Coronary Artery Calcification in Patients with Coronary Artery Disease. Cardiogenetics. 2020; 10(2):33-41. https://doi.org/10.3390/cardiogenetics10020007
Chicago/Turabian StyleAhmadihosseini, Zeynab, Morteza Moeinian, Saeed Nazemi, Sepideh Elyasi, and Amir Hooshang Mohammadpour. 2020. "Evaluation of the Correlation between the rs4918 Polymorphism of AHSG Gene and Coronary Artery Calcification in Patients with Coronary Artery Disease" Cardiogenetics 10, no. 2: 33-41. https://doi.org/10.3390/cardiogenetics10020007
APA StyleAhmadihosseini, Z., Moeinian, M., Nazemi, S., Elyasi, S., & Mohammadpour, A. H. (2020). Evaluation of the Correlation between the rs4918 Polymorphism of AHSG Gene and Coronary Artery Calcification in Patients with Coronary Artery Disease. Cardiogenetics, 10(2), 33-41. https://doi.org/10.3390/cardiogenetics10020007





