Design of an Open Remote Electrocardiogram (ECG) Service
Abstract
1. Introduction
- the next section is a review of on-topic literature that introduces our methods and results;
- Section 3 applies defined methods in the design of a remote ECG service;
- in Section 4, we describe the materials to implement a functional prototype
- in Section 5, we perform a exhaustive test of all components, to assess the results and guide future improvements;
- finally, we discuss the potential of our prototype in some use cases.
2. Related Works and Results Summary
- Open Source: components and protocols are exhaustively documented and freely reproducible;
- Low Cost: the less expensive option is always preferable. As a corollary, if one functionality is already available for free, it is not re-implemented;
- Commercial Devices: devices must be available on retail (aka Commercial Off-the-shelf (COTS)).
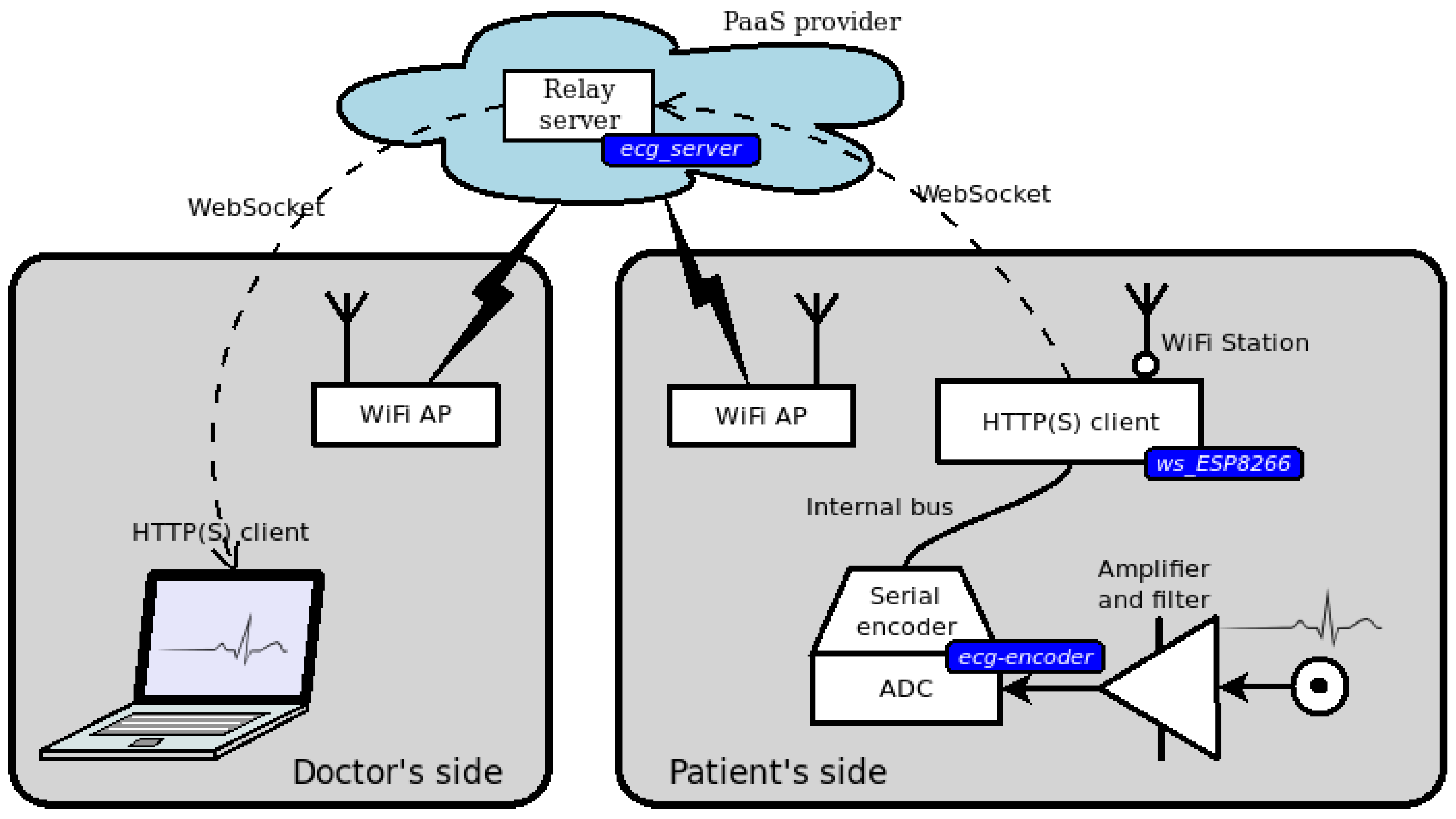
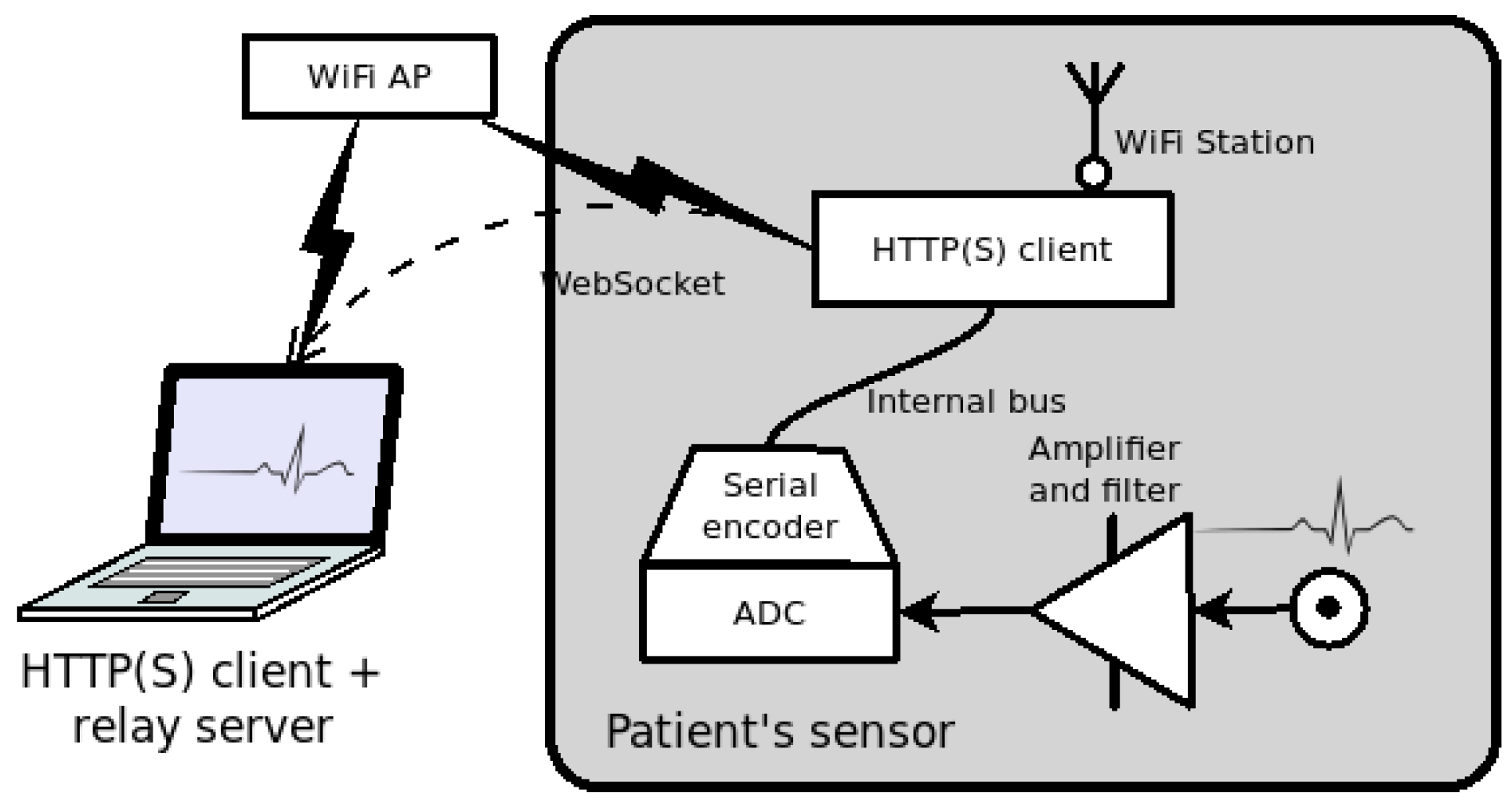
3. An Open Service for Remote ECG
3.1. Doctor’s Side Web Interface
3.2. Patient’s Side Sensor Device
3.3. Relay Server in the Cloud
3.4. Design Overview and Security Issues
4. A Prototype for a Remote ECG Service
- the analog-to-serial encoder in the MCU,
- the HTTP patient-side UA,
- the relay server hosting WebSocket endpoints and doctor’s access page.
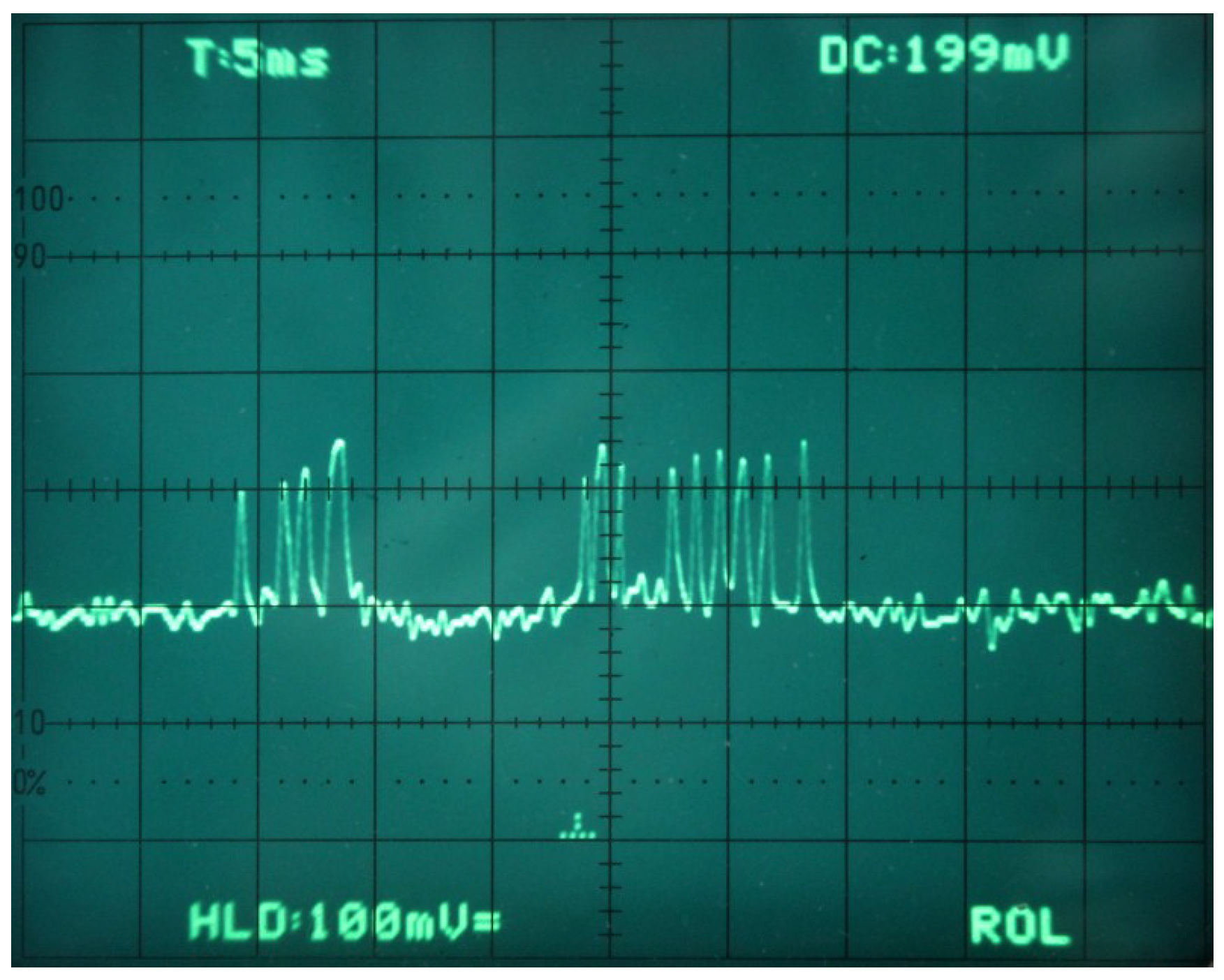
4.1. The Sensor: Amplifier and Filter
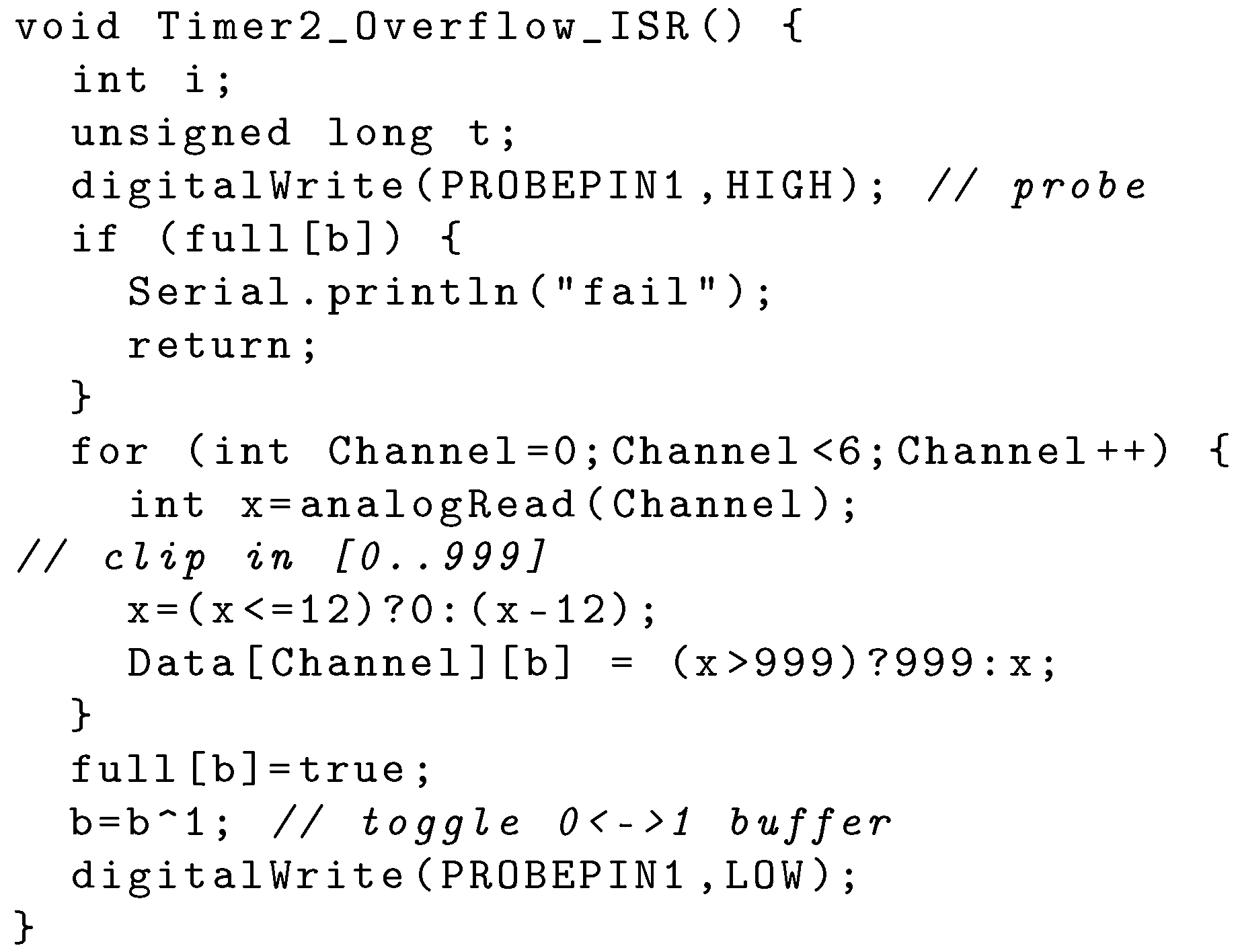
4.2. The MCU: ADC and Serial Encoder
<hh>:<mm>:<ss>.<ddd> <v1><v2><v3><v4><v5><v6>Q
4.3. MCU to HTTP User Agent Interface
4.4. Patient Side HTTP User-Agent and WiFi Interface
4.5. The Server
- / which returns a introductory page,
- /in/<id> used by patient’s UA WebSocket,
- /<id> used by doctor’s browser to open the ECG visualization page,
- /out/<id> used as endpoint for doctor’s WebSocket,
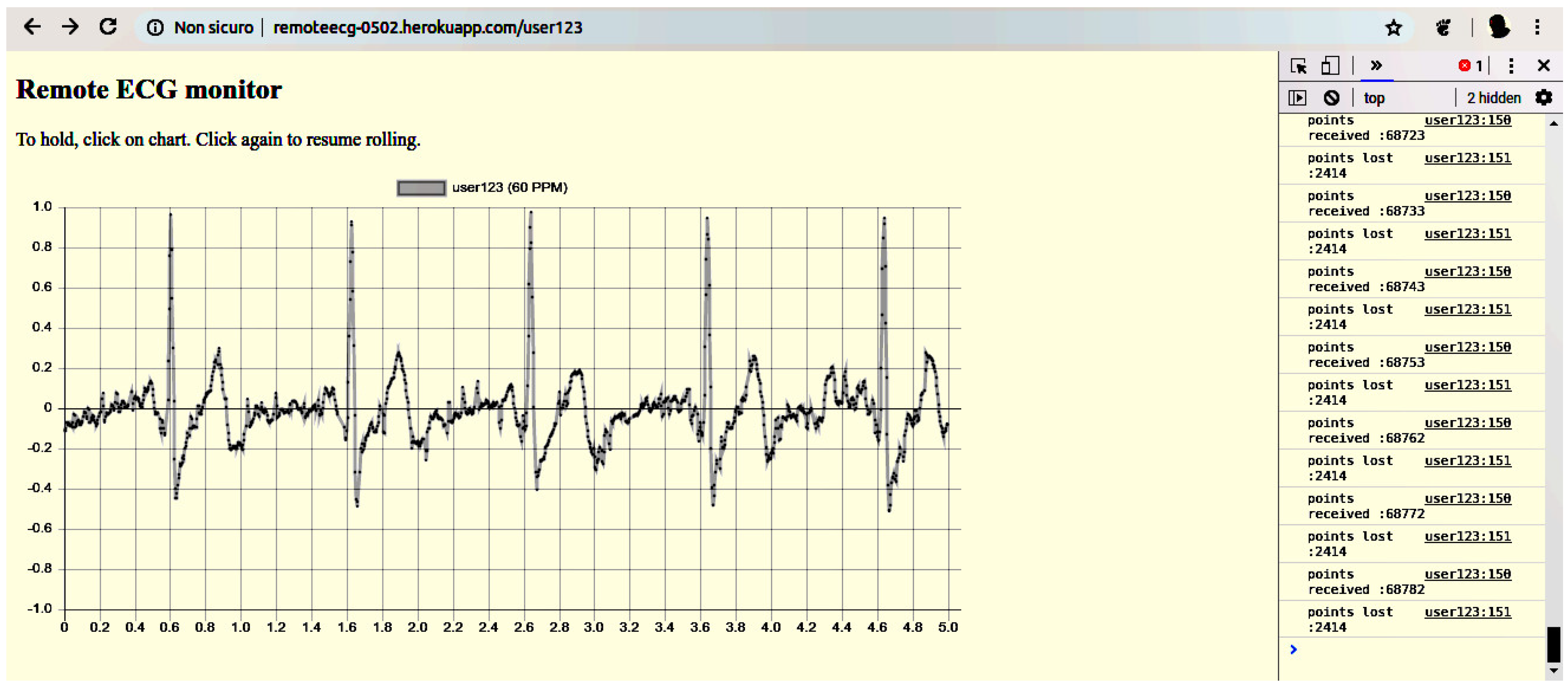
4.6. The Display UA
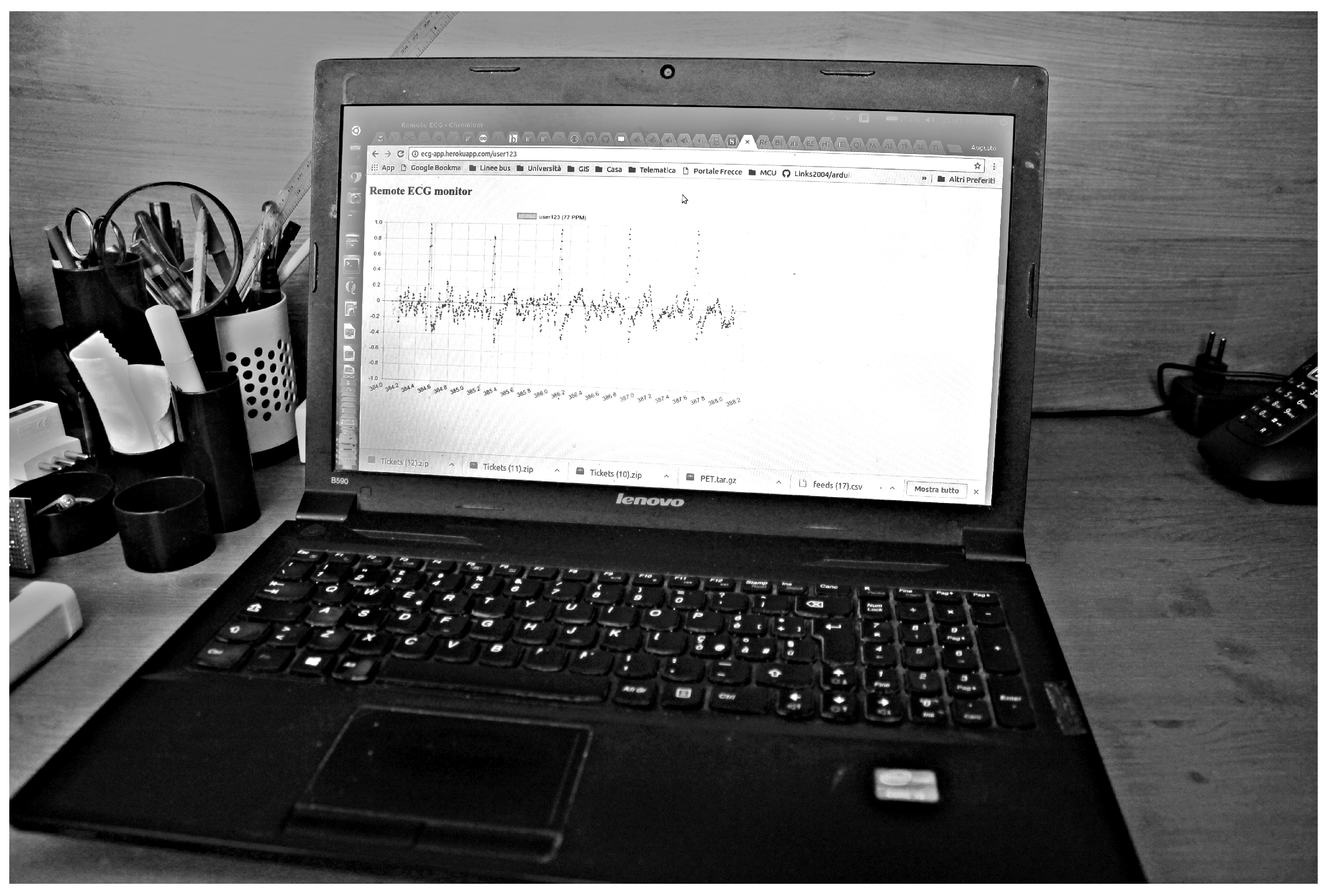
5. Experimental Results
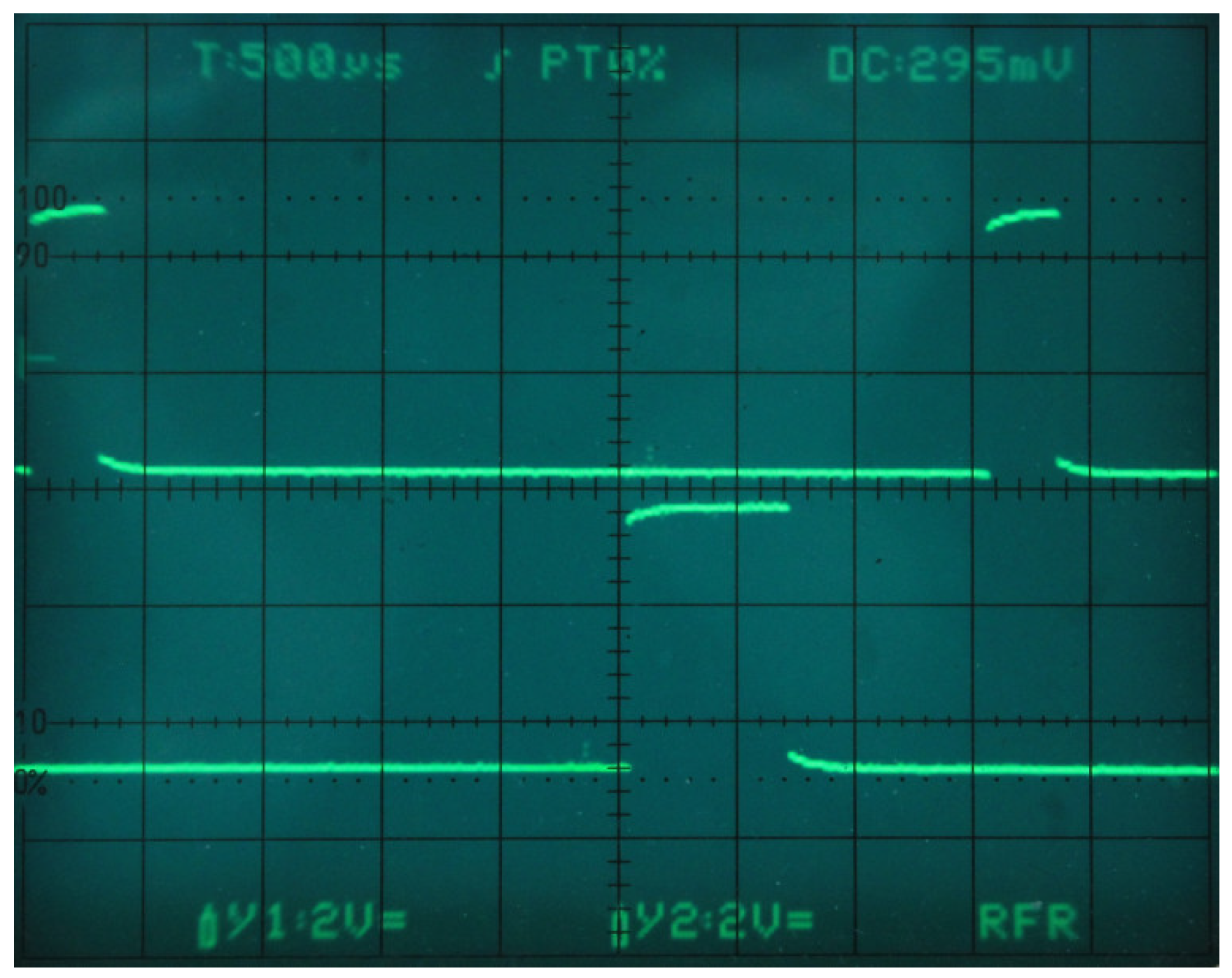
5.1. Data Acquisition
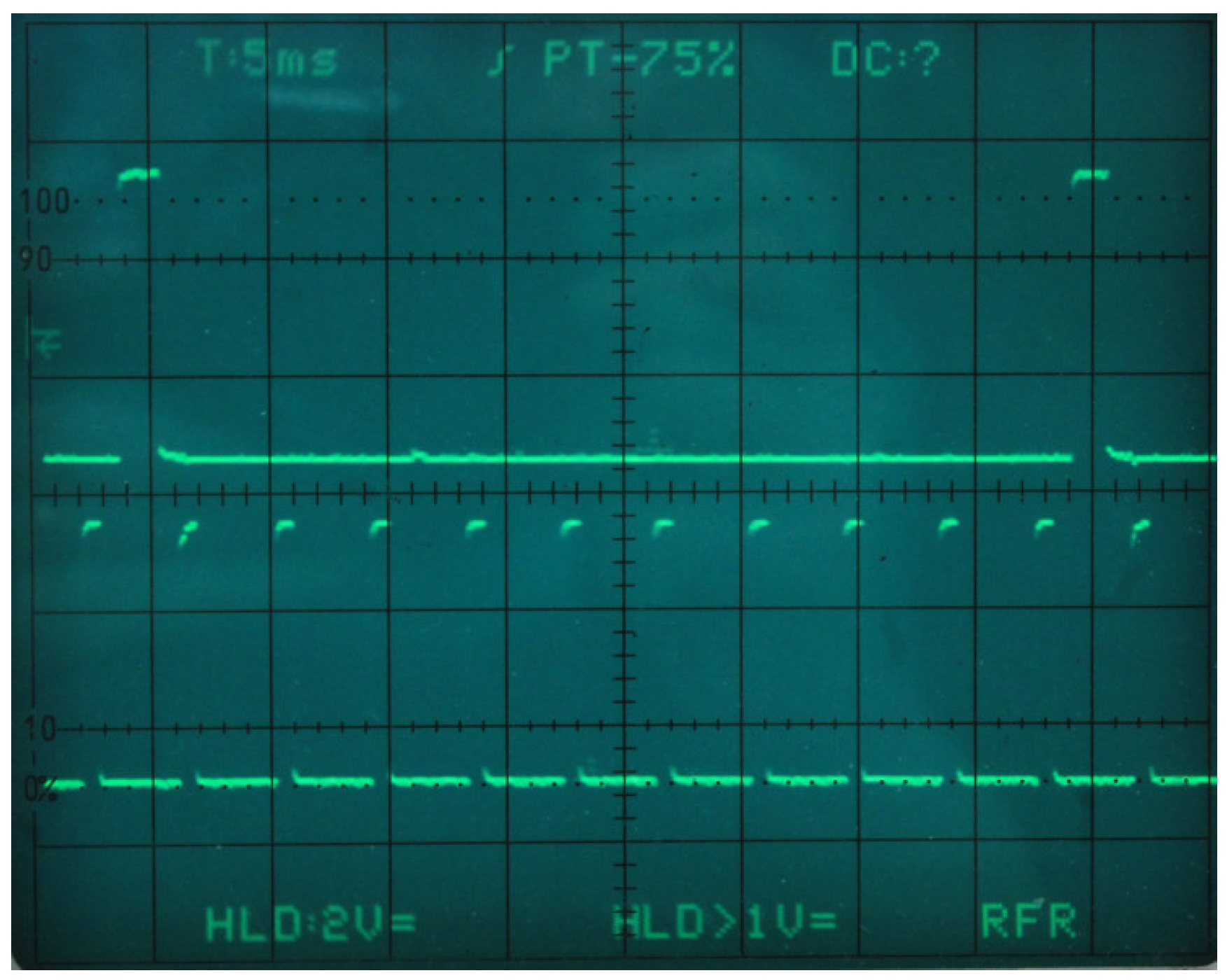
5.2. Data Frame Construction and Delivery
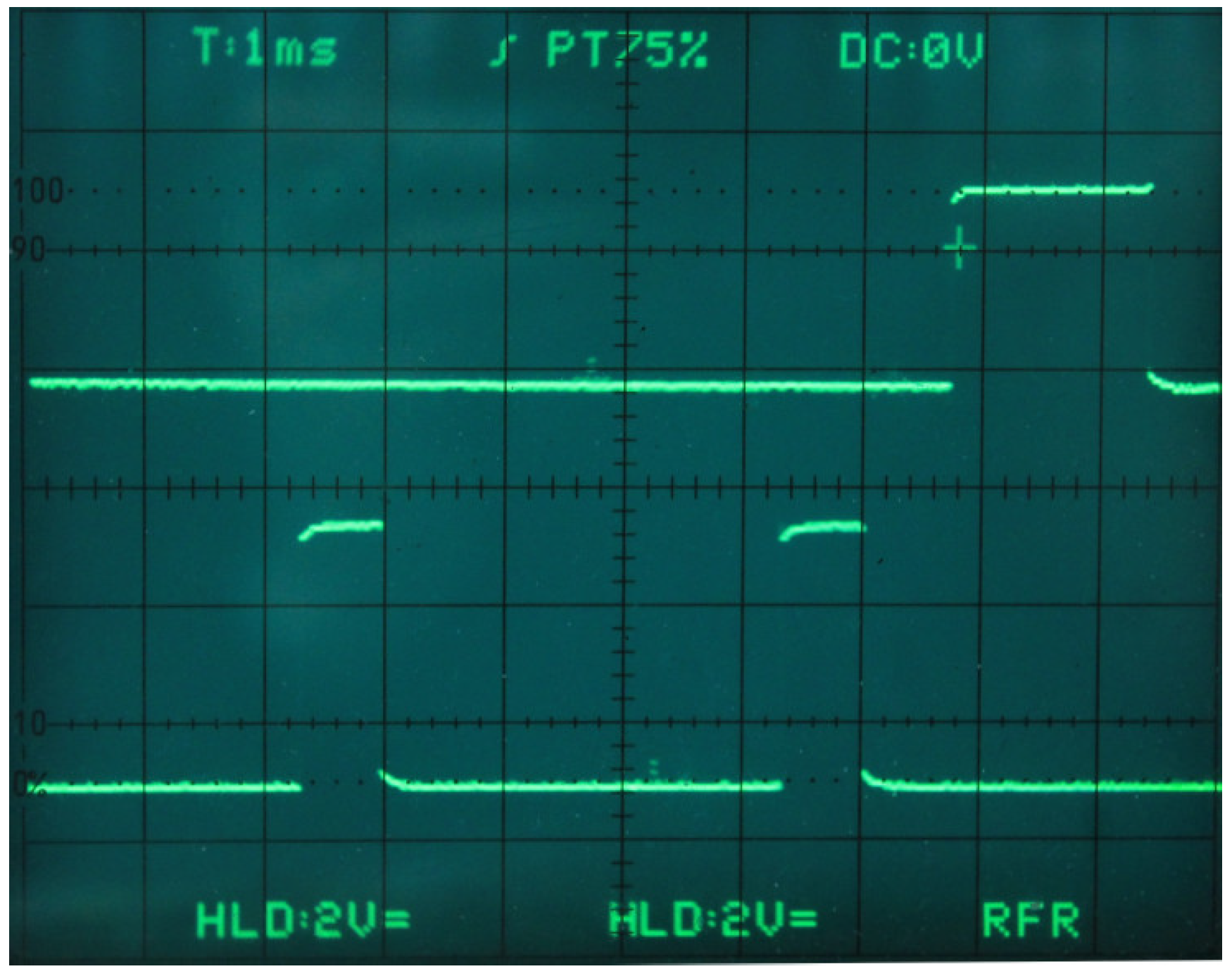
5.3. Server and Network Infrastructure
5.4. The Display
- a moving average filter that significantly attenuates the residual 50 Hz noise,
- the dynamic evaluation of the heartbeat rate,
- the possibility to put on hold the screen to analyze the ECG.
6. Conclusions and Future Work
Funding
Conflicts of Interest
Abbreviations
| WAN | Wide Area Network |
| CoAP | Constrained Application Protocol |
| ISP | Internet Service Provider |
| AF | Atrial Fibrillation |
| OpLoC | Open source, Low cost, Commercial |
| COTS | Commercial Off-the-shelf |
| ECG | electrocardiogram |
| UA | User Agent |
| FQDN | Fully Qualified Domain Name |
| HTTP | Hypertext Transfer Protocol |
| HTTPS | Secure Hypertext Transfer Protocol |
| IETF | Internet Engineering Task Force |
| URL | Uniform Resource Locator |
| ASCII | American Standard Code for Information Interchange |
| JSON | JavaScript Object Notation |
| WSGI | Web Server Gateway Interface |
| MCU | Micro-Controller Unit |
| SBC | Single Board Computer |
| PaaS | Platform as a Service |
| AP | Access Point |
| API | Application Programming Interface |
| WiFi | Wireless Fidelity |
| BAN | Body (or Personal) Data Network |
| MSN | Medical Sensor Network |
| PC | Personal Computer |
| AWS | Amazon Web Services |
| POP | Point Of Presence |
| NAT | Network Address Translation |
| TLS | Transport Level Security |
| ADC | Analog Digital Converter |
| UART | Universal Asynchronous Receiver-Transmitter |
| IP | Internet Protocol |
| TCP | Transport Control Protocol |
| UDP | User Datagram Protocol |
| LoRa | Long Range |
| IDE | Integrated Develpment Environment |
References
- Halcox, J.P.; Wareham, K.; Cardew, A.; Gilmore, M.; Barry, J.P.; Phillips, C.; Gravenor, M.B. Assessment of Remote Heart Rhythm Sampling Using the AliveCor Heart Monitor to Screen for Atrial Fibrillation. Circulation 2017, 136, 1784–1794. [Google Scholar] [CrossRef] [PubMed]
- Haritha, C.; Ganesan, M.; Sumesh, E.P. A survey on modern trends in ECG noise removal techniques. In Proceedings of the 2016 International Conference on Circuit, Power and Computing Technologies (ICCPCT), Nagercoil, India, 18–19 March 2016; pp. 1–7. [Google Scholar] [CrossRef]
- Naik, G.R.; Reddy, K.A. A new model for ECG signal filtering and feature extraction. In Proceedings of the 2016 2nd IEEE International Conference on Computer and Communications (ICCC), Chengdu, China, 14–17 October 2016; pp. 765–768. [Google Scholar] [CrossRef]
- Gutierrez-Rivas, R.; Garcia, J.J.; Marnane, W.P.; Hernandez, A. Novel Real-Time Low-Complexity QRS Complex Detector Based on Adaptive Thresholding. IEEE Sens. J. 2015, 15, 6036–6043. [Google Scholar] [CrossRef]
- Lugovaya, T.S. Biometric human identification based on electrocardiogram. Master’s Thesis, Faculty of Computing Technologies and Informatics, Electrotechnical University “LETI”, Saint-Petersburg, Russian, 2005. Available online: https://physionet.org/physiobank/database/ecgiddb/biometric.shtml (accessed on 23 April 2019).
- Sathya, D.; Kumar, P.G. Secured remote health monitoring system. Healthc.Technol. Lett. 2017, 4, 228–232. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5761311/ (accessed on 23 April 2019). [CrossRef] [PubMed]
- Baig, M.M.; GholamHosseini, H.; Moqeem, A.A.; Mirza, F.; Lindén, M. A Systematic Review of Wearable Patient Monitoring Systems—Current Challenges and Opportunities for Clinical Adoption. J. Med. Syst. 2017, 41, 115. [Google Scholar] [CrossRef] [PubMed]
- Jones, V.; Bults, R.; Konstantas, D.; Vierhout, P. Healthcare PANs: Personal Area Networks for trauma care and home care. In Proceedings of the Fourth International Symposium on Wireless Personal Multimedia Communications, Aalborg, Denmark, 9–12 September 2001; Volume 3, pp. 1369–1374. Available online: https://research.utwente.nl/files/6141897/paper51withheaderpage.pdf (accessed on 23 April 2019).
- Chen, M.; Gonzalez, S.; Vasilakos, A.; Cao, H.; Leung, V.C.M. Body Area Networks: A Survey. Mob. Netw. Appl. 2011, 16, 171–193. [Google Scholar] [CrossRef]
- Shnayder, V.; Chen, B.R.; Lorincz, K.; Fulford-Jones, T.R.F.; Welsh, M. Sensor Networks for Medical Care; Technical Report TR-08-05; Harvard Computer Science Group: Cambridge, MA, USA, 2005; Available online: http://ftp.deas.harvard.edu/techreports/tr-08-05.pdf (accessed on 23 April 2019).
- Plesnik, E.; Malgina, O.; Tasič, J.F.; Zajc, M. ECG signal acquisition and analysis for telemonitoring. In Proceedings of the Melecon 2010—2010 15th IEEE Mediterranean Electrotechnical Conference, Valletta, Malta, 26–28 April 2010; pp. 1350–1355. [Google Scholar] [CrossRef]
- Cristea, C.; Pasarica, A.; Andruseac, G.; Dionisie, B.; Rotariu, C. A wireless ECG acquisition device for remote monitoring of heart rate and arrhythmia detection. In Proceedings of the 2015 E-Health and Bioengineering Conference (EHB), Iasi, Romania, 19–21 November 2015; pp. 1–4. [Google Scholar] [CrossRef]
- Xia, H.; Asif, I.; Zhao, X. Cloud-ECG for real time ECG monitoring and analysis. Comput. Methods Programs Biomed. 2013, 110, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Page, A.; Kocabas, O.; Soyata, T.; Aktas, M.; Couderc, J.P. Cloud-Based Privacy-Preserving Remote ECG Monitoring and Surveillance. Ann. Noninvasive Electrocardiol. 2014, 20, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Welton, N.J.; McAleenan, A.; Thom, H.H.; Davies, P.; Hollingworth, W.; Higgins, J.P.; Okoli, G.; Sterne, J.A.; Feder, G.; Eaton, D.; et al. Screening strategies for atrial fibrillation: A systematic review and cost-effectiveness analysis. Health Technol. Assess. 2017, 21, 1–236. [Google Scholar] [CrossRef] [PubMed]
- Walker, B.A.; Khandoker, A.H.; Black, J. Low cost ECG monitor for developing countries. In Proceedings of the 2009 International Conference on Intelligent Sensors, Sensor Networks and Information Processing (ISSNIP), Melbourne, Australia, 7–10 December 2009; pp. 195–199. [Google Scholar] [CrossRef]
- Deb, S.; Islam, S.M.R.; RobaiatMou, J.; Islam, M.T. Design and implementation of low cost ECG monitoring system for the patient using smart device. In Proceedings of the 2017 International Conference on Electrical, Computer and Communication Engineering (ECCE), Cox’s Bazar, Bangladesh, 16–18 February 2017; pp. 774–778. [Google Scholar] [CrossRef]
- Guo, S.L.; Han, L.N.; Liu, H.W.; Si, Q.J.; Kong, D.F.; Guo1, F.S. The future of remote ECG monitoring systems. J. Geriatric Cardiol. 2016. [Google Scholar] [CrossRef]
- Ciuffoletti, A. Low-cost IoT: A holistic approach. J. Sens. Actuator Netw. 2018, 7, 19. [Google Scholar] [CrossRef]
- Fette, I.; Melnikov, A. The WebSocket Protocol. RFC 6455, RFC Editor. 2011. Available online: http://www.rfc-editor.org/rfc/rfc6455.txt (accessed on 23 April 2019).
- Ciuffoletti, A. On-line Remote EKG as a Web Service. arXiv 2019, arXiv:1901.00724. Available online: https://arxiv.org/abs/1901.00724 (accessed on 23 April 2019).
- Shelby, Z.; Hartke, K.; Bormann, C. The Constrained Application Protocol (CoAP). RFC 7252, RFC Editor. 2014. Available online: http://www.rfc-editor.org/rfc/rfc7252.txt (accessed on 23 April 2019).
- Newman, S. Building Microservices: Designing Fine-Grained Systems; O’Reilly Media: Sebastopol, CA, USA, 2015; Available online: http://shop.oreilly.com/product/0636920033158.do (accessed on 23 April 2019).
- Ciuffoletti, A. Prototype of an Open Remote ECG Service. 2018. Available online: https://bitbucket.org/augusto_ciuffoletti/prototype-of-an-open-remote-ecg-service (accessed on 23 April 2019).
- SHIELD EKG-EMG REV.B. Available online: https://www.olimex.com/Products/Duino/Shields/SHIELD-EKG-EMG/resources/SHIELD-EKG-EMG-REV-B-SCHEMATIC.pdf (accessed on 23 April 2019).
- Pizzuti, G.; Cifaldi, S.; Nolfe, G. Digital sampling rate and ECG analysis. J. Biomed. Eng. 1985, 7, 247–250. [Google Scholar] [CrossRef]
- Mdhaffar, A.; Chaari, T.; Larbi, K.; Jmaiel, M.; Freisleben, B. IoT-based health monitoring via LoRaWAN. In Proceedings of the IEEE EUROCON 2017-17th International Conference on Smart Technologies, Ohrid, Macedonia, 6–8 July 2017; pp. 519–524. [Google Scholar] [CrossRef]



© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciuffoletti, A. Design of an Open Remote Electrocardiogram (ECG) Service. Future Internet 2019, 11, 101. https://doi.org/10.3390/fi11040101
Ciuffoletti A. Design of an Open Remote Electrocardiogram (ECG) Service. Future Internet. 2019; 11(4):101. https://doi.org/10.3390/fi11040101
Chicago/Turabian StyleCiuffoletti, Augusto. 2019. "Design of an Open Remote Electrocardiogram (ECG) Service" Future Internet 11, no. 4: 101. https://doi.org/10.3390/fi11040101
APA StyleCiuffoletti, A. (2019). Design of an Open Remote Electrocardiogram (ECG) Service. Future Internet, 11(4), 101. https://doi.org/10.3390/fi11040101




