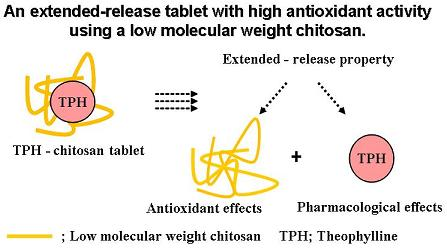
Useful Extend-release Chitosan Tablets with High Antioxidant Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
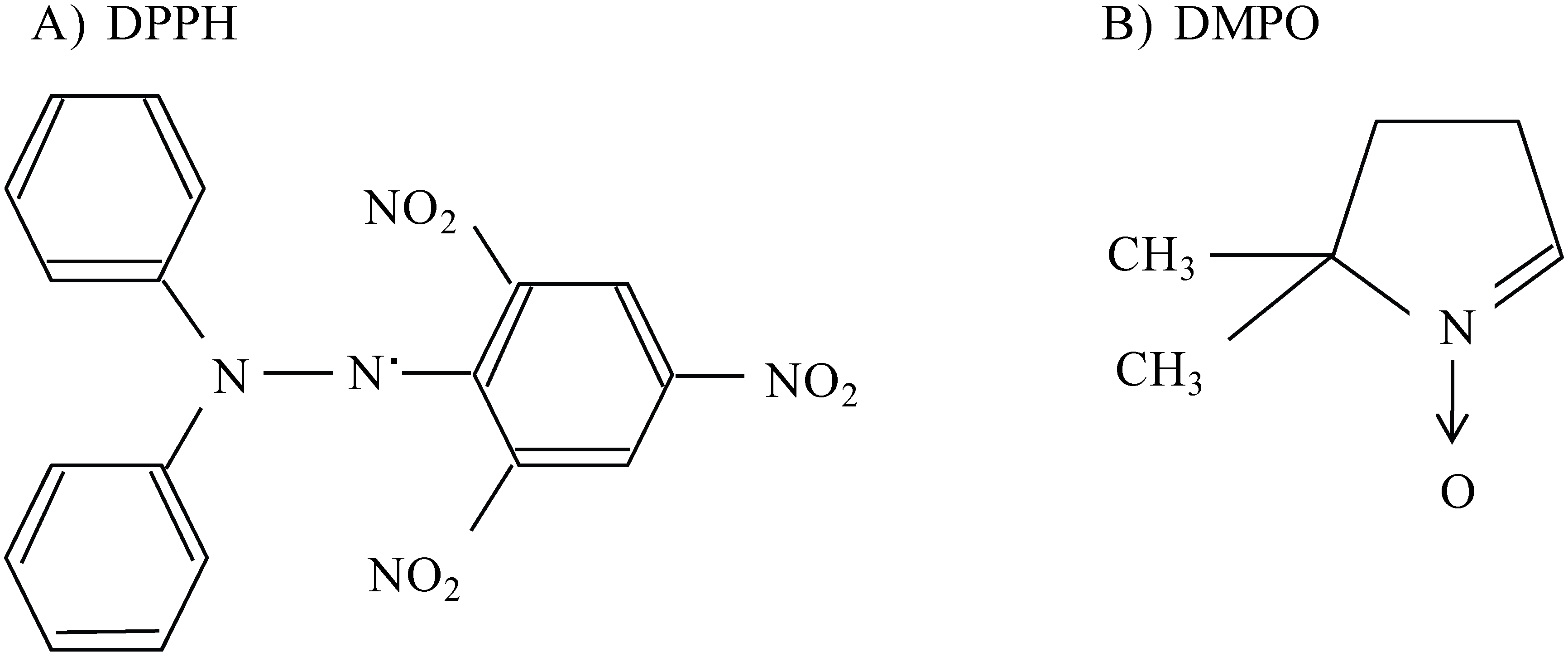
2.2. Scavenging activity of LMW chitosans on DPPH radicals
2.3. Reducing power by using the TPAC test
2.4. ESR spectroscopy
2.5. Hydroxyl radical assay
2.6. Superoxide radical assay
2.7. Preparation of extended-release matrix tablet
2.8. Dissolution of TPH from tablets and scavenging activity of chitosans-TPH tablets on ABTS radicals
2.9. Statistics
3. Results and Discussion
3.1. Scavenging activity of LMW chitosans on DPPH radicals
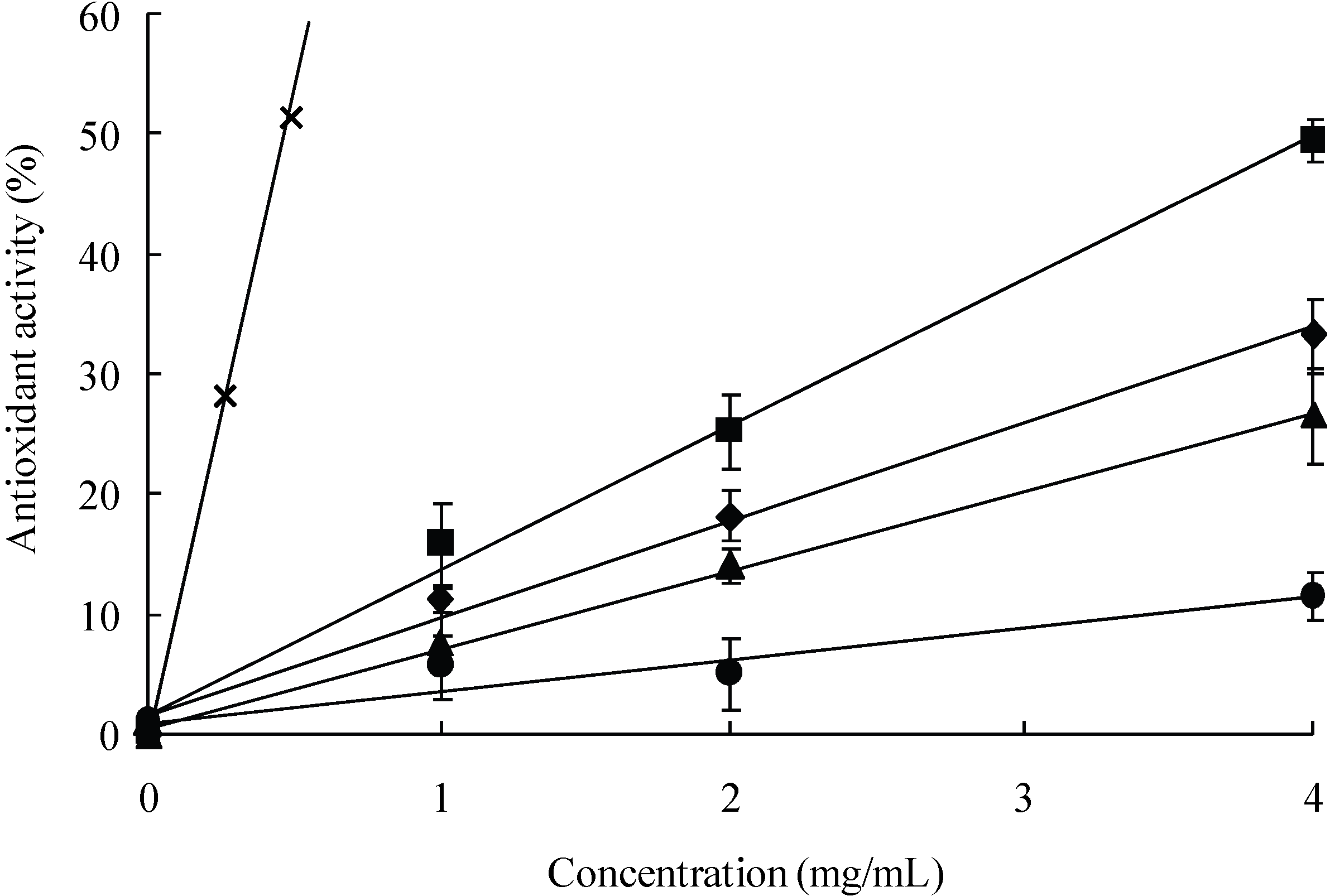
| Antioxidant | DPPH |
|---|---|
| IC50 (mg/mL) a | |
| CS1 | 6 |
| CS2 | 4.02 |
| CS3 | 7.54 |
| CS4 | >10 |
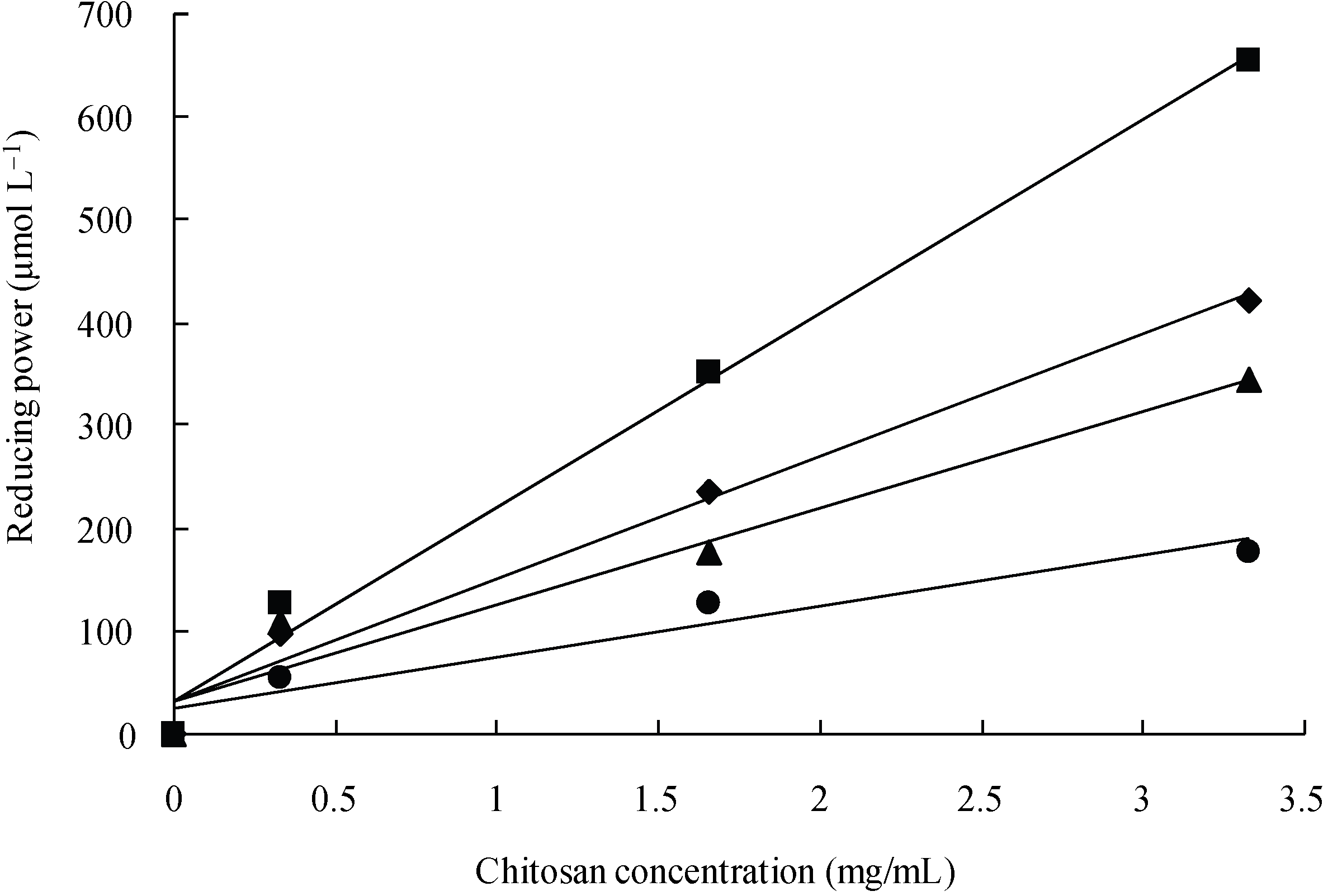
3.2. Reducing power of LMW chitosans by using the TPAC test
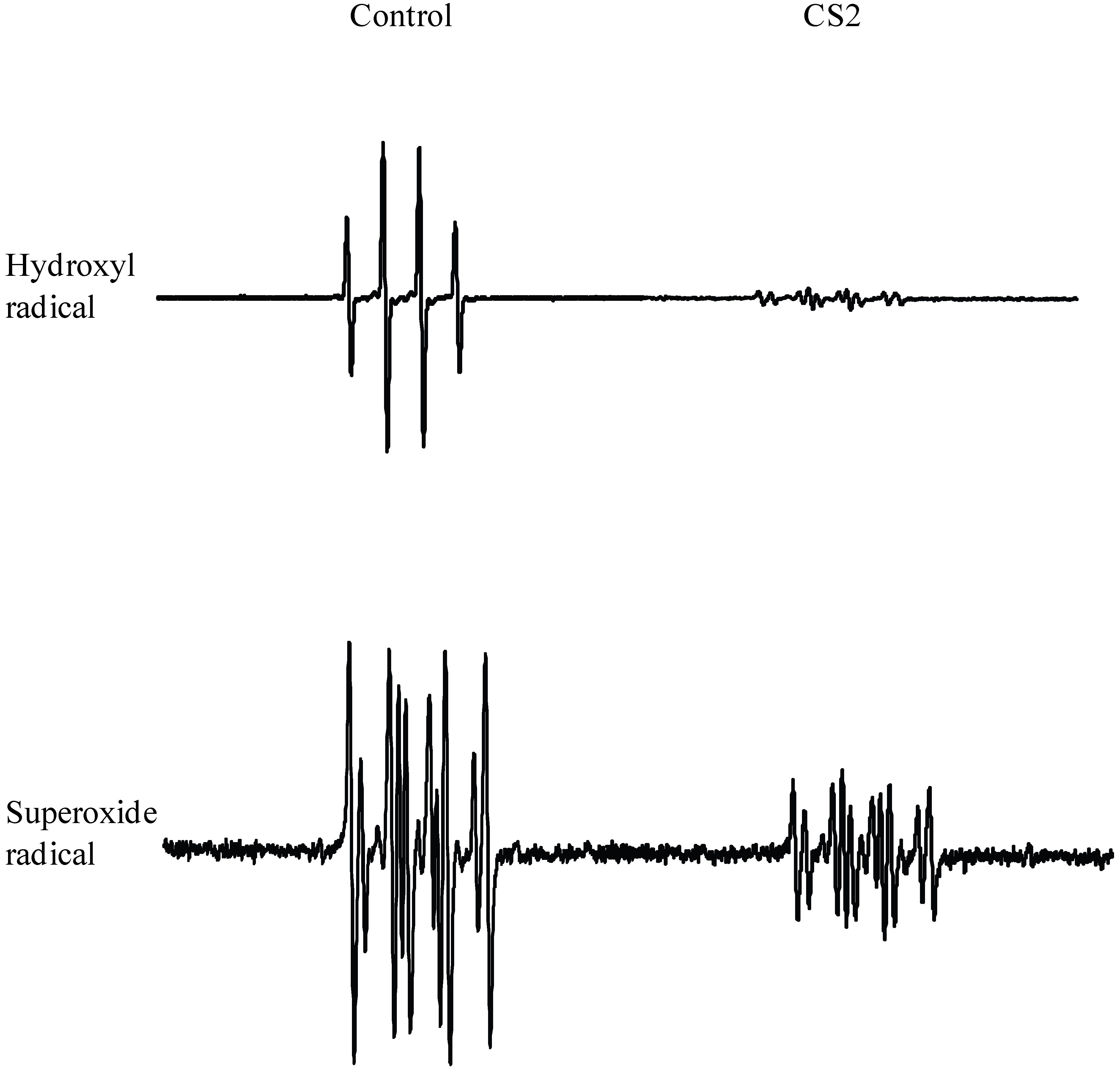
3.3. Radical scavenging potency of LMW chitosans by ESR spectroscopy
| Conc. (g/mL) | Scavenging percentage (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Hydroxyl radical | Superoxide radical | ||||||||
| CS1 | CS2 | CS3 | CS4 | CS1 | CS2 | CS3 | CS4 | ||
| 100 | 53.7 ± 3.5 | 65.1 ± 4.5* | 50.5 ± 3.8 | 49.5 ± 4.7 | 39.7 ± 4.8 | 48.2 ± 5.8* | 33.5 ± 5.4 | 30.8 ± 5.2 | |
| 200 | 80.7 ± 4.5 | 92.1 ± 4.6* | 78.1 ± 4.6 | 75.1 ± 3.1 | 65.1 ± 4.6 | 80.1 ± 5.6* | 56.1 ± 5.8 | 50.1 ± 5.6 | |
3.4. Release study of TPH and scavenging activity of chitosans-TPH tablet
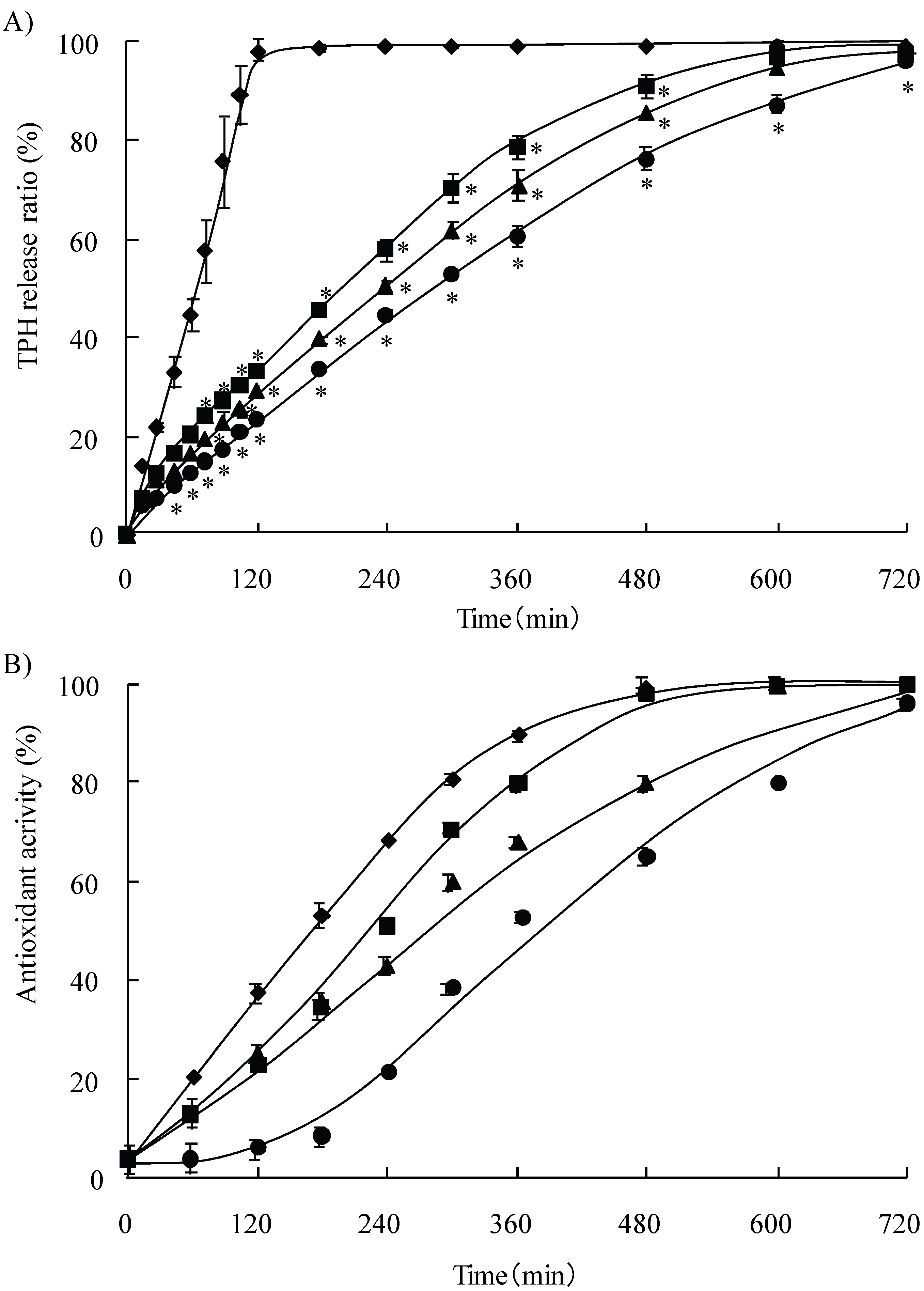
3.5. Discussion
4. Conclusions
References and Notes
- Miyazaki, Y.; Tanaka, Y.; Yakou, S.; Takayama, K. In vivo drug release from hydrophilic dextran tablets capable of forming polyion complex. J. Controlled Release 2006, 114, 47–52. [Google Scholar] [CrossRef]
- Liew, C.V.; Chan, L.W.; Ching, L.; Heng, P.W. Evaluation of sodium alginate as drug release modifier in matrix tablets. Int. J. Pharm. 2006, 309, 25–37. [Google Scholar] [CrossRef]
- Pitt, C.G. The controlled parenteral delivery of peptides and protein. Int. J. Pharm. 1990, 59, 173–196. [Google Scholar] [CrossRef]
- Aiedeh, K.; Taha, M.O. Synthesis of iron-crosslinked chitosan succinate and iron-crosslinked hydroxamated chitosan succinate and their in vitro evaluation as potential matrix materials for oral theophylline sustained-release beads. Eur. J. Pharm. Sci. 2001, 13, 159–168. [Google Scholar] [CrossRef]
- Patel, V.R.; Amiji, M.M. Preparation and characterization of freeze-dried chitosan-poly(ethylene oxide) hydrogels for site-specific antibiotic delivery in the stomach. Pharm. Res. 1996, 13, 588–593. [Google Scholar] [CrossRef]
- Puttipipatkhachorn, S.; Nunthanid, J.; Yamamoto, K.; Peck, G.E. Drug physical state and drug-polymer interaction on drug release from chitosan matrix films. J. Controlled Release 2001, 75, 143–153. [Google Scholar] [CrossRef]
- Oungbho, K.; Muller, B.W. Chitosan sponges as sustained release drug carriers. Int. J. Pharm. 1997, 156, 229–237. [Google Scholar] [CrossRef]
- He, P.; Davis, S.S.; Illum, L. Chitosan microspheres prepared by spray drying. Int. J. Pharm. 1999, 187, 53–65. [Google Scholar] [CrossRef]
- Xue, C.; Yu, G.; Hirata, T.; Terao, J.; Lin, H. Antioxidative activities of several marine polysaccharides evaluated in a phosphatidylcholine-liposomal suspension and organic solvents. Biosci. Biotechnol. Biochem. 1998, 62, 206–209. [Google Scholar] [CrossRef]
- Chiang, M.T.; Yao, H.T.; Chen, H.C. Effect of dietary chitosans with different viscosity on plasma lipids and lipid peroxidation in rats fed on a diet enriched with cholesterol. Biosci. Biotechnol. Biochem. 2000, 64, 965–971. [Google Scholar] [CrossRef]
- Anraku, M.; Kabashima, M.; Namura, H.; Maruyama, T.; Otagiri, M.; Gebicki, J.M.; Furutani, N.; Tomida, H. Antioxidant protection of human serum albumin by chitosan. Int. J. Biol. Macromol. 2008, 43, 159–164. [Google Scholar] [CrossRef]
- Santhosh, S.; Sini, T.K.; Anandan, R.; Mathew, P.T. Effect of chitosan supplementation on antitubercular drugs-induced hepatotoxicity in rats. Toxicology 2006, 219, 53–59. [Google Scholar] [CrossRef]
- Yoon, H.J.; Moon, M.E.; Park, H.S.; Kim, H.W.; Im, S.Y.; Lee, J.H.; Kim, Y.H. Effects of chitosan oligosaccharide (COS) on the glycerol-induced acute renal failure in vitro and in vivo. Food Chem. Toxicol. 2008, 46, 710–716. [Google Scholar] [CrossRef]
- Anraku, M.; Fujii, T.; Furutani, N.; Kadowaki, D.; Maruyama, T.; Otagiri, M.; Gebicki, J.M.; Tomida, H. Antioxidant effects of a dietary supplement: reduction of indices of oxidative stress in normal subjects by water-soluble chitosan. Food Chem. Toxicol. 2009, 47, 104–109. [Google Scholar] [CrossRef]
- Kogure, K.; Goto, S.; Abe, K.; Ohiwa, C.; Akasu, M.; Terada, H. Potent antiperoxidation activity of the bisbenzylisoquinoline alkaloid cepharanthine: the amine moiety is responsible for its pH-dependent radical scavenge activity. Biochim. Biophys. Acta. 1999, 1426, 133–142. [Google Scholar] [CrossRef]
- Rosen, G.M.; Rauckman, E.J. Spin trapping of superoxide and hydroxyl radicals. In Methods in enzymology; Packer, L., Ed.; Academic Press: Orlando, F.L., USA, 1984; Volume 105, pp. 189–209. [Google Scholar]
- Guo, Q.; Zhao, B.; Shen, S.; Hou, J.; Hu, J.; Xin, W. ESR study on the structure-antioxidant activity relationship of tea catechins and their epimers. Biochim. Biophys. Acta. 1999, 1427, 13–23. [Google Scholar] [CrossRef]
- Leelarungrayub, N.; Rattanapanone, V.; Chanarat, N.; Gebicki, J.M. Quantitative evaluation of the antioxidant properties of garlic and shallot preparations. Nutrition 2006, 22, 266–274. [Google Scholar] [CrossRef]
- Butterfield, D.A.; Lauderback, C.M. Lipid peroxidation and protein oxidation in Alzheimer's disease brain: potential causes and consequences involving amyloid beta-peptide-associated free radical oxidative stress. Free Radical Biol. Med. 2002, 2, 1050–1060. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M. The antioxidants of human extracellular fluids. Arch. Biochem. Biophys. 1990, 280, 1–8. [Google Scholar] [CrossRef]
- Péterszegi, G.; Robert, A.M.; Robert, L. Protection by L-fucose and fucose-rich polysaccharides against ROS-produced cell death in presence of ascorbate. Biomed. Pharmacother. 2003, 57, 130–133. [Google Scholar] [CrossRef]
- Kim, S.K.; Rajapakse, N. Enzymatic production and biological activities of chitosan oligosaccharides. Carbohydr. Polym. 2005, 62, 357–368. [Google Scholar] [CrossRef]
- Soares, J.R.; Dins, T.C.P.; Cunha, A.P.; Almeida, L.M. Antioxidant activities of some extracts of Thymus zygis. Free Radical Res. 1997, 26, 469–478. [Google Scholar] [CrossRef]
- Park, P.J.; Je, J.Y.; Kim, S.K. Free radical scavenging activity of chitooligosaccharides by electron spin resonance spectrometry. J. Agric. Food Chem. 2003, 51, 4624–4627. [Google Scholar] [CrossRef]
- Xie, W.; Xu, P.; Liu, Q. Antioxidant activity of watersoluble chitosan derivatives. Bioorg. Med. Chem. Lett. 2001, 11, 1699–1701. [Google Scholar] [CrossRef]
- Nellore, R.V.; Rekhi, G.S.; Hussain, A.S.; Tillman, L.G.; Augsburger, L.L. Development of metoprolol tartrate extended-release matrix tablet formulations for regulatory policy consideration. J. Controlled Release 1998, 50, 247–256. [Google Scholar] [CrossRef]
- Kranz, H.; Guthmann, C.; Wagner, T.; Lipp, R.; Reiinhard, J. Development of a single unit extended release formulation for ZK 811 752, a weakly basic drug. Eur. J. Pharm. Sci. 2005, 26, 47–53. [Google Scholar] [CrossRef]
- Nigalaye, A.G.; Adusumili, P.; Bolton, S. Investigation of prolonged drug release from matrix formulations of chitosan. Drug Dev. Ind. Pharm. 1990, 16, 449–467. [Google Scholar] [CrossRef]
- Sawayanagi, Y.; Nambu, N.; Nagai, T. Direct compressed tablets containing chitin. Chem. Pharm. Bull. 1982, 30, 4216–4218. [Google Scholar] [CrossRef]
- Knapczyk, J. Excipient ability of chitosan for direct tableting. Int. J. Pharm. 1993, 89, 1–7. [Google Scholar] [CrossRef]
- Takeuchi, H.; Yasuji, T.; Yamamoto, H.; Kawashima, Y. Spray dried lactose composite particles containing an ion complex of alginate-chitosan for designing a dry coated tablet having a time controlled releasing function. Pharm. Res. 2000, 17, 94–99. [Google Scholar] [CrossRef]
- Sukaya, T.; Murakami, H.; Katayama, H. Preparation of minute particles of chitosan. J.P. Patent JP 632,210,101 A2, 25 January 1988. [Google Scholar]
- Yabe, H.; Itoyama, M.; Kawamura, Y. Manufacture of chitosan and chitin porous microparticles. J.P. Patent JP 632,210,101 A2, 31 August 1988. [Google Scholar]
- Yamanami, T.; Tsuchida, S.; Seki, M. Preparation of chitosan powder for pharmaceutics. J.P. Patent JP 63,017,902 A2, 25 January 1988. [Google Scholar]
- Tohda, Y.; Muraki, M.; Iwanaga, T.; Kubo, H.; Fukuoka, M.; Nakajima, S. The effect of theophylline on blood and sputum eosinophils and ECP in patients with bronchial asthma. Int. J. Immunopharm. 1998, 20, 173–181. [Google Scholar]
- Finnerty, J.P.; Lee, C.; Wilson, S.; Madden, J.; Djukanovic, R.; Holgate, S.T. Effects of theophylline on inflammatory cells and cytokines in asthmatic subjects: a placebo-controlled parallel group study. Eur. Resp. J. 1996, 9, 1672–1677. [Google Scholar] [CrossRef]
- Mellstrand, T.; Svedmyr, N.; Fagerström, P.O. Absorption of theophylline from conventional and sustained-release tablets. Eur. J. Resp. Dis. Suppl. 1980, 109, 54–60. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yasufuku, T.; Anraku, M.; Kondo, Y.; Hata, T.; Hirose, J.; Kobayashi, N.; Tomida, H. Useful Extend-release Chitosan Tablets with High Antioxidant Activity. Pharmaceutics 2010, 2, 245-257. https://doi.org/10.3390/pharmaceutics2020245
Yasufuku T, Anraku M, Kondo Y, Hata T, Hirose J, Kobayashi N, Tomida H. Useful Extend-release Chitosan Tablets with High Antioxidant Activity. Pharmaceutics. 2010; 2(2):245-257. https://doi.org/10.3390/pharmaceutics2020245
Chicago/Turabian StyleYasufuku, Taira, Makoto Anraku, Yuko Kondo, Toshiyuki Hata, Junzo Hirose, Nobuyuki Kobayashi, and Hisao Tomida. 2010. "Useful Extend-release Chitosan Tablets with High Antioxidant Activity" Pharmaceutics 2, no. 2: 245-257. https://doi.org/10.3390/pharmaceutics2020245