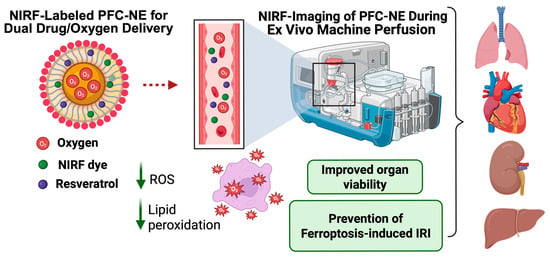
Perfluorocarbon Nanoemulsions for Simultaneous Delivery of Oxygen and Antioxidants During Machine Perfusion Supported Organ Preservation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Perfluorocarbon Nanoemulsion Perfusate Formulation and Manufacturing
2.2.2. Colloidal and Fluorescence Assessments of Nanoemulsions
2.2.3. Colloidal Stability Under Stress Conditions
2.2.4. Resveratrol Quantification and In Vitro Release Study
2.2.5. Nanoemulsion Stability Under Oxygenation Conditions
2.2.6. Oxygen Saturation and Off-Loading in an Ex Vivo Machine Perfusion Circuit
2.2.7. In-Line Perfusion NIRF Imaging of Nanoemulsion and UW Solution
2.2.8. Oxygen Radical Antioxidant Capacity (ORAC) of Nanoemulsions
2.2.9. Macrophage Cell Viability and Ferroptosis Inhibition Assay
2.2.10. Statistical Analyses
3. Results
3.1. Nanoemulsions Composition Optimization for Increased Oxygen Loading and Stability
3.2. Comparison of Production Scale and High Shear Processing Equipment
3.3. Evaluation of Drug-Free and Resveratrol-Loaded PFC-NEs for Oxygen Carrier Capacity
3.4. Drug Quantification and In Vitro Release Study of Resveratrol from PFC-NE
3.5. Oxygen-Loading and In-Line Perfusion NIRF Imaging of PFC-NEs
3.6. Oxygen Saturation and Off-Loading Under Machine Perfusion
3.7. Oxygen Radical Antioxidant Capacity (ORAC) of Resveratrol Loaded PFC-NE
3.8. Cell Viability and Ferroptosis Inhibition Study In Vitro
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Global Observatory on Donation and Transplantation. Available online: https://www.transplant-observatory.org/ (accessed on 14 July 2025).
- Scientific Registry of Transplant Recipients (SRTR). Available online: https://www.srtr.org/ (accessed on 20 July 2025).
- Dery, K.J.; Yao, S.; Cheng, B.; Kupiec-Weglinski, J.W. New therapeutic concepts against ischemia-reperfusion injury in organ transplantation. Expert Rev. Clin. Immunol. 2023, 19, 1205–1224. [Google Scholar] [CrossRef] [PubMed]
- Micó-Carnero, M.; Zaouali, M.A.; Rojano-Alfonso, C.; Maroto-Serrat, C.; Ben Abdennebi, H.; Peralta, C. A potential route to reduce ischemia/reperfusion injury in organ preservation. Cells 2022, 11, 2763. [Google Scholar] [CrossRef]
- Gazia, C.; Lenci, I.; Manzia, T.M.; Martina, M.; Tisone, G.; Angelico, R.; Abenavoli, L.; Grassi, G.; Signorello, A.; Baiocchi, L. Current strategies to minimize ischemia-reperfusion injury in liver transplantation: A systematic review. Rev. Recent Clin. Trials 2021, 16, 372–380. [Google Scholar] [CrossRef]
- Sun, R.; Wang, N.; Zheng, S.; Wang, H.; Xie, H. Nanotechnology-based Strategies for Molecular Imaging, Diagnosis, and Therapy of Organ Transplantation. Transplantation 2024, 108, 1730–1748. [Google Scholar] [CrossRef]
- Zhou, L.; Han, S.; Guo, J.; Qiu, T.; Zhou, J.; Shen, L. Ferroptosis—A new dawn in the treatment of organ ischemia–reperfusion injury. Cells 2022, 11, 3653. [Google Scholar] [CrossRef]
- Chen, Y.; Fan, H.; Wang, S.; Tang, G.; Zhai, C.; Shen, L. Ferroptosis: A novel therapeutic target for ischemia-reperfusion injury. Front. Cell Dev. Biol. 2021, 9, 688605. [Google Scholar] [CrossRef]
- Pan, Y.; Wang, X.; Liu, X.; Shen, L.; Chen, Q.; Shu, Q. Targeting ferroptosis as a promising therapeutic strategy for ischemia-reperfusion injury. Antioxidants 2022, 11, 2196. [Google Scholar] [CrossRef]
- Aufhauser, D.D.; Foley, D.P. Beyond ice and the cooler: Machine perfusion strategies in liver transplantation. Clin. Liver Dis. 2021, 25, 179–194. [Google Scholar] [CrossRef]
- Jing, L.; Yao, L.; Zhao, M.; Peng, L.-p.; Liu, M. Organ preservation: From the past to the future. Acta Pharmacol. Sin. 2018, 39, 845–857. [Google Scholar] [CrossRef] [PubMed]
- Hakim, G. Establishment and Assessment of Pancreas Preservation Using Oxygenated Hypothermic Machine Perfusion. Master’s Thesis, University of Oxford, Oxford, UK, 2022. [Google Scholar]
- Weissenbacher, A.; Vrakas, G.; Nasralla, D.; Ceresa, C.D. The future of organ perfusion and re-conditioning. Transpl. Int. 2019, 32, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Zulpaite, R.; Miknevicius, P.; Leber, B.; Strupas, K.; Stiegler, P.; Schemmer, P. Ex-vivo kidney machine perfusion: Therapeutic potential. Front. Med. 2021, 8, 808719. [Google Scholar] [CrossRef]
- Ferenz, K.B.; Steinbicker, A.U. Artificial oxygen carriers—Past, present, and future—A review of the most innovative and clinically relevant concepts. J. Pharmacol. Exp. Ther. 2019, 369, 300–310. [Google Scholar] [CrossRef]
- Riess, J.G. Oxygen carriers (“blood substitutes”) raison d’être, chemistry, and some physiology blut ist ein ganz besondrer saft. Chem. Rev. 2001, 101, 2797–2920. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yang, Z.; Liu, H. Hemoglobin-based oxygen carriers: Where are we now in 2023? Medicina 2023, 59, 396. [Google Scholar] [CrossRef] [PubMed]
- Jahr, J.S.; MacKinnon, K.; Baum, V.C.; Alayash, A.I. Hemoglobin-based oxygen carriers: Biochemical, biophysical differences, and safety. Transfusion 2025, 65, 386. [Google Scholar] [CrossRef]
- Kakaei, N.; Amirian, R.; Azadi, M.; Mohammadi, G.; Izadi, Z. Perfluorocarbons: A perspective of theranostic applications and challenges. Front. Bioeng. Biotechnol. 2023, 11, 1115254. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Jung, E.-A.; Kim, J.-E. Perfluorocarbon-based artificial oxygen carriers for red blood cell substitutes: Considerations and direction of technology. J. Pharm. Investig. 2024, 54, 267–282. [Google Scholar] [CrossRef]
- Vichare, R.; Janjic, J. Perfluorocarbon nanoemulsions in drug delivery: Design, development, and manufacturing. Theranostics 2025, 15, 3013. [Google Scholar] [CrossRef] [PubMed]
- Aburawi, M.M.; Fontan, F.M.; Karimian, N.; Eymard, C.; Cronin, S.; Pendexter, C.; Nagpal, S.; Banik, P.; Ozer, S.; Mahboub, P. Synthetic hemoglobin-based oxygen carriers are an acceptable alternative for packed red blood cells in normothermic kidney perfusion. Am. J. Transplant. 2019, 19, 2814–2824. [Google Scholar] [CrossRef]
- Laing, R.W.; Bhogal, R.H.; Wallace, L.; Boteon, Y.; Neil, D.A.; Smith, A.; Stephenson, B.T.; Schlegel, A.; Hübscher, S.G.; Mirza, D.F. The use of an acellular oxygen carrier in a human liver model of normothermic machine perfusion. Transplantation 2017, 101, 2746–2756. [Google Scholar] [CrossRef]
- Matton, A.P.; Burlage, L.C.; van Rijn, R.; de Vries, Y.; Karangwa, S.A.; Nijsten, M.W.; Gouw, A.S.; Wiersema-Buist, J.; Adelmeijer, J.; Westerkamp, A.C. Normothermic machine perfusion of donor livers without the need for human blood products. Liver Transplant. 2018, 24, 528–538. [Google Scholar] [CrossRef]
- Lalla, F.; Ning, J.; Chang, T. Effects of pyridoxalated polyhemoglobin and stroma-free hemoglobin on ADP-induced platelet aggregation. Biomater. Artif. Cells Artif. Organs 1989, 17, 363–369. [Google Scholar] [CrossRef]
- Ye, Q.; Zheng, D.; Chen, K.; Wu, J. Research progress in oxygen carrier design and application. Mol. Pharm. 2023, 20, 4373–4386. [Google Scholar] [CrossRef]
- Jägers, J.; Wrobeln, A.; Ferenz, K.B. Perfluorocarbon-based oxygen carriers: From physics to physiology. Pflügers Arch.-Eur. J. Physiol. 2021, 473, 139–150. [Google Scholar] [CrossRef]
- Castro, C.I.; Briceno, J.C. Perfluorocarbon-based oxygen carriers: Review of products and trials. Artif. Organs 2010, 34, 622–634. [Google Scholar] [CrossRef]
- Riess, J.G. Perfluorocarbon-based oxygen delivery. Artif. Cells Blood Substit. Biotechnol. 2006, 34, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Krafft, M.P.; Riess, J.G.; Weers, J.G. The design and engineering of oxygen-delivering fluorocarbon emulsions. In Submicron Emulsions in Drug Targeting and Delivery; CRC Press: London, UK, 2019; pp. 235–334. [Google Scholar]
- Riess, J.G. Understanding the fundamentals of perfluorocarbons and perfluorocarbon emulsions relevant to in vivo oxygen delivery. Artif. Cells Blood Substit. Biotechnol. 2005, 33, 47–63. [Google Scholar] [CrossRef] [PubMed]
- Riess, J.G. Fluorous materials for biomedical uses. In Handbook of Fluorous Chemistry; Wiley-VCH Verlag GmbH & Co. KgaA: Weinheim, Germany, 2004; pp. 521–573. [Google Scholar]
- Naito, H.; Nojima, T.; Fujisaki, N.; Tsukahara, K.; Yamamoto, H.; Yamada, T.; Aokage, T.; Yumoto, T.; Osako, T.; Nakao, A. Therapeutic strategies for ischemia reperfusion injury in emergency medicine. Acute Med. Surg. 2020, 7, e501. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef]
- Tang, L.-J.; Luo, X.-J.; Tu, H.; Chen, H.; Xiong, X.-M.; Li, N.-S.; Peng, J. Ferroptosis occurs in phase of reperfusion but not ischemia in rat heart following ischemia or ischemia/reperfusion. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2021, 394, 401–410. [Google Scholar] [CrossRef]
- Valgimigli, L. Lipid peroxidation and antioxidant protection. Biomolecules 2023, 13, 1291. [Google Scholar] [CrossRef]
- Kajarabille, N.; Latunde-Dada, G.O. Programmed cell-death by ferroptosis: Antioxidants as mitigators. Int. J. Mol. Sci. 2019, 20, 4968. [Google Scholar] [CrossRef] [PubMed]
- Živanović, N.; Lesjak, M.; Simin, N.; Srai, S.K. Beyond mortality: Exploring the influence of plant phenolics on modulating ferroptosis—A systematic review. Antioxidants 2024, 13, 334. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Tan, Y.; Ouyang, S.; He, J.; Liu, L. Resveratrol protects against myocardial ischemia-reperfusion injury via attenuating ferroptosis. Gene 2022, 808, 145968. [Google Scholar] [CrossRef]
- Wang, X.; Shen, T.; Lian, J.; Deng, K.; Qu, C.; Li, E.; Li, G.; Ren, Y.; Wang, Z.; Jiang, Z. Resveratrol reduces ROS-induced ferroptosis by activating SIRT3 and compensating the GSH/GPX4 pathway. Mol. Med. 2023, 29, 137. [Google Scholar] [CrossRef]
- Amri, A.; Chaumeil, J.; Sfar, S.; Charrueau, C. Administration of resveratrol: What formulation solutions to bioavailability limitations? J. Control. Release 2012, 158, 182–193. [Google Scholar] [CrossRef]
- Machado, N.D.; Fernández, M.A.; Díaz, D.D. Recent strategies in resveratrol delivery systems. ChemPlusChem 2019, 84, 951–973. [Google Scholar] [CrossRef] [PubMed]
- Herneisey, M.; Williams, J.; Mirtic, J.; Liu, L.; Potdar, S.; Bagia, C.; Cavanaugh, J.E.; Janjic, J.M. Development and characterization of resveratrol nanoemulsions carrying dual-imaging agents. Ther. Deliv. 2016, 7, 795–808. [Google Scholar] [CrossRef]
- Kotta, S.; Aldawsari, H.M.; Badr-Eldin, S.M.; Alhakamy, N.A.; Md, S. Coconut oil-based resveratrol nanoemulsion: Optimization using response surface methodology, stability assessment and pharmacokinetic evaluation. Food Chem. 2021, 357, 129721. [Google Scholar] [CrossRef]
- Nastiti, C.M.; Ponto, T.; Mohammed, Y.; Roberts, M.S.; Benson, H.A. Novel nanocarriers for targeted topical skin delivery of the antioxidant resveratrol. Pharmaceutics 2020, 12, 108. [Google Scholar] [CrossRef]
- Pangeni, R.; Sharma, S.; Mustafa, G.; Ali, J.; Baboota, S. Vitamin E loaded resveratrol nanoemulsion for brain targeting for the treatment of Parkinson’s disease by reducing oxidative stress. Nanotechnology 2014, 25, 485102. [Google Scholar] [CrossRef]
- Rinaldi, F.; Maurizi, L.; Forte, J.; Marazzato, M.; Hanieh, P.N.; Conte, A.L.; Ammendolia, M.G.; Marianecci, C.; Carafa, M.; Longhi, C. Resveratrol-loaded nanoemulsions: In vitro activity on human T24 bladder cancer cells. Nanomaterials 2021, 11, 1569. [Google Scholar] [CrossRef]
- Lambert, E.; Janjic, J.M. Quality by design approach identifies critical parameters driving oxygen delivery performance in vitro for perfluorocarbon based artificial oxygen carriers. Sci. Rep. 2021, 11, 5569. [Google Scholar] [CrossRef]
- Vichare, R.; Crelli, C.; Liu, L.; McCallin, R.; Cowan, A.; Stratimirovic, S.; Herneisey, M.; Pollock, J.A.; Janjic, J.M. Folate-conjugated near-infrared fluorescent perfluorocarbon nanoemulsions as theranostics for activated macrophage COX-2 inhibition. Sci. Rep. 2023, 13, 15229. [Google Scholar] [CrossRef] [PubMed]
- Herneisey, M.; Janjic, J.M. Multiple linear regression predictive modeling of colloidal and fluorescence stability of theranostic perfluorocarbon nanoemulsions. Pharmaceutics 2023, 15, 1103. [Google Scholar] [CrossRef]
- Lambert, E.; Gorantla, V.S.; Janjic, J.M. Pharmaceutical design and development of perfluorocarbon nanocolloids for oxygen delivery in regenerative medicine. Nanomedicine 2019, 14, 2697–2712. [Google Scholar] [CrossRef]
- Das, A.C.; Reddy, G.A.; Newaj, S.M.; Patel, S.; Vichare, R.; Liu, L.; Janjic, J.M. Design of Experiments Leads to Scalable Analgesic Near-Infrared Fluorescent Coconut Nanoemulsions. Pharmaceutics 2025, 17, 1010. [Google Scholar] [CrossRef] [PubMed]
- OxiSelect™ Oxygen Radical Antioxidant Capacity (ORAC) Activity Assay; Product Manual; Cell Biolabs, Inc.: San Diego, CA, USA.
- Schaafsma, B.E.; Mieog, J.S.D.; Hutteman, M.; Van der Vorst, J.R.; Kuppen, P.J.; Löwik, C.W.; Frangioni, J.V.; Van de Velde, C.J.; Vahrmeijer, A.L. The clinical use of indocyanine green as a near-infrared fluorescent contrast agent for image-guided oncologic surgery. J. Surg. Oncol. 2011, 104, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Belia, F.; Biondi, A.; Agnes, A.; Santocchi, P.; Laurino, A.; Lorenzon, L.; Pezzuto, R.; Tirelli, F.; Ferri, L.; D’Ugo, D. The use of indocyanine green (ICG) and near-infrared (NIR) fluorescence-guided imaging in gastric cancer surgery: A narrative review. Front. Surg. 2022, 9, 880773. [Google Scholar] [CrossRef]
- Vasudeva, K.; Andersen, K.; Zeyzus-Johns, B.; Hitchens, T.K.; Patel, S.K.; Balducci, A.; Janjic, J.M.; Pollock, J.A. Imaging neuroinflammation in vivo in a neuropathic pain rat model with near-infrared fluorescence and (1)(9)F magnetic resonance. PLoS ONE 2014, 9, e90589. [Google Scholar] [CrossRef]
- Janjic, J.M.; Shao, P.; Zhang, S.; Yang, X.; Patel, S.K.; Bai, M. Perfluorocarbon nanoemulsions with fluorescent, colloidal and magnetic properties. Biomaterials 2014, 35, 4958–4968. [Google Scholar] [CrossRef] [PubMed]
- Balducci, A.; Wen, Y.; Zhang, Y.; Helfer, B.M.; Hitchens, T.K.; Meng, W.S.; Wesa, A.K.; Janjic, J.M. A novel probe for the non-invasive detection of tumor-associated inflammation. Oncoimmunology 2013, 2, e23034. [Google Scholar] [CrossRef]
- Patrick, M.J.; Janjic, J.M.; Teng, H.; O’Hear, M.R.; Brown, C.W.; Stokum, J.A.; Schmidt, B.F.; Ahrens, E.T.; Waggoner, A.S. Intracellular pH measurements using perfluorocarbon nanoemulsions. J. Am. Chem. Soc. 2013, 135, 18445–18457. [Google Scholar] [CrossRef]
- Patel, S.K.; Patrick, M.J.; Pollock, J.A.; Janjic, J.M. Two-color fluorescent (near-infrared and visible) triphasic perfluorocarbon nanoemuslions. J. Biomed. Opt. 2013, 18, 101312. [Google Scholar] [CrossRef]
- Patel, S.K.; Williams, J.; Janjic, J.M. Cell Labeling for 19F MRI: New and Improved Approach to Perfluorocarbon Nanoemulsion Design. Biosensors 2013, 3, 341–359. [Google Scholar] [CrossRef]
- Wu, Y.; Suo, Y.; Wang, Z.; Yu, Y.; Duan, S.; Liu, H.; Qi, B.; Jian, C.; Hu, X.; Zhang, D.; et al. First clinical applications for the NIR-II imaging with ICG in microsurgery. Front. Bioeng. Biotechnol. 2022, 10, 1042546. [Google Scholar] [CrossRef]
- Yamamichi, T.; Oue, T.; Yonekura, T.; Owari, M.; Nakahata, K.; Umeda, S.; Nara, K.; Ueno, T.; Uehara, S.; Usui, N. Clinical application of indocyanine green (ICG) fluorescent imaging of hepatoblastoma. J. Pediatr. Surg. 2015, 50, 833–836. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Niu, F.; Ren, N.; Wang, X.; Zhong, H.; Zhu, J.; Li, B. Hyperbaric Oxygen Improves Cerebral Ischemia/Reperfusion Injury in Rats Probably via Inhibition of Autophagy Triggered by the Downregulation of Hypoxia-Inducing Factor-1 Alpha. BioMed Res. Int. 2021, 2021, 6615685. [Google Scholar] [CrossRef]
- Wigman, L.; Ooi, D. ICH Q10 Quality Systems. In ICH Quality Guidelines: An Implementation Guide; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018; p. 611. [Google Scholar]
- Xu, J.; Buchwald, J.E.; Martins, P.N. Review of current machine perfusion therapeutics for organ preservation. Transplantation 2020, 104, 1792–1803. [Google Scholar] [CrossRef]
- Chatzidaki, M.D.; Mitsou, E. Advancements in nanoemulsion-based drug delivery across different administration routes. Pharmaceutics 2025, 17, 337. [Google Scholar] [CrossRef] [PubMed]
- Darius, T.; Vergauwen, M.; Mueller, M.; Aydin, S.; Dutkowski, P.; Gianello, P.; Mourad, M. Brief bubble and intermittent surface oxygenation is a simple and effective alternative for membrane oxygenation during hypothermic machine perfusion in kidneys. Transplant. Direct 2020, 6, e571. [Google Scholar] [CrossRef]
- Lee, C.Y.; Mangino, M.J. Preservation methods for kidney and liver. Organogenesis 2009, 5, 105–112. [Google Scholar] [CrossRef]
- Alves, F.; Lane, D.; Nguyen, T.P.M.; Bush, A.I.; Ayton, S. In defence of ferroptosis. Signal Transduct. Target. Ther. 2025, 10, 2. [Google Scholar] [CrossRef]
- Lowe, K.C. Perfluorochemicals in vascular medicine. Vasc. Med. Rev. 1994, 5, 15–32. [Google Scholar] [CrossRef]
- Cabrales, P.; Intaglietta, M. Blood substitutes: Evolution from noncarrying to oxygen-and gas-carrying fluids. ASAIO J. 2013, 59, 337–354. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rother, T.; Horgby, C.; Schmalkuche, K.; Burgmann, J.M.; Nocke, F.; Jägers, J.; Schmitz, J.; Bräsen, J.H.; Cantore, M.; Zal, F. Oxygen carriers affect kidney immunogenicity during ex-vivo machine perfusion. Front. Transplant. 2023, 2, 1183908. [Google Scholar] [CrossRef] [PubMed]
- Pinnelas, R.; Kobashigawa, J.A. Ex vivo normothermic perfusion in heart transplantation: A review of the TransMedics® Organ Care System. Future Cardiol. 2022, 18, 5–15. [Google Scholar] [CrossRef]
- Tchouta, L.N.; Alghanem, F.; Rojas-Pena, A.; Bartlett, R.H. Prolonged (≥24 h) normothermic (≥32 °C) ex vivo organ perfusion: Lessons from the literature. Transplantation 2021, 105, 986–998. [Google Scholar] [CrossRef]
- Kawamura, M.; Parmentier, C.; Ray, S.; Clotet-Freixas, S.; Leung, S.; John, R.; Mazilescu, L.; Nogueira, E.; Noguchi, Y.; Goto, T. Normothermic ex vivo kidney perfusion preserves mitochondrial and graft function after warm ischemia and is further enhanced by AP39. Nat. Commun. 2024, 15, 8086. [Google Scholar] [CrossRef]








| Batch No. | RSV (mg/mL) | Dye | Dispersant | Batch Size (mL) | Processor | N Passes | Pressure (psi) | Size (nm) | PDI | Mean ± SD (nm) | Coefficient of Variation (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | - | DiI | DI water | 25 | M-110S | 6 | 18,640 | 99 | 0.2 | 102 ± 4.0 | 3.7 |
| 2 | 1.23 | DiI and DiR | 97 | 0.239 | |||||||
| 3 | - | DiI and DiR | 105 | 0.237 | |||||||
| 4 | 0.685 | DiI and DiR | 105 | 0.193 | |||||||
| 5 | - | - | 104 | 0.231 | |||||||
| 6 | 0.685 | - | 1× PBS | 125 | LM20 | 5 | 16,500 | 93 | 0.225 | 96 ± 2.5 | 2.6 |
| 7 | - | - | 96 | 0.228 | |||||||
| 8 | 0.685 | - | 98 | 0.223 | |||||||
| 9 | - | ICG | 1.68× PBS | 250 | LM20 | 4 | 20,000 | 83 | 0.179 | 80 ± 2.1 | 2.6 |
| 10 | 0.69 | 80 | 0.175 | ||||||||
| 11 | - | ICG | 1.68× PBS | 600 | LM20 | 5 | 18,500 | 79 | 0.182 | 94 ± 3.5 | 3.7 |
| 12 | - | 97 | 0.222 | ||||||||
| 13 | - | 81 | 0.216 | ||||||||
| 14 | - | ICG | 1.68× PBS | 600 | M-110P | 6 | 18,500 | 94 | 0.131 | 90 ± 8.4 | 9.4 |
| 15 | - | 80 | 0.208 | ||||||||
| 16 | - | 98 | 0.123 | ||||||||
| 17 | - | ICG | 1.68× PBS | 1000 | M-110P | 4 | 18,500 | 90 | 0.217 | 90 ± 2.0 | 2.2 |
| 18 | 0.69 | 88 | 0.189 | ||||||||
| 19 | - | 92 | 0.214 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Patel, S.; Pinky, P.P.; Das, A.C.; Copus, J.S.; Aardema, C.; Crelli, C.; Troidle, A.; Lambert, E.; McCallin, R.; Surti, V.; et al. Perfluorocarbon Nanoemulsions for Simultaneous Delivery of Oxygen and Antioxidants During Machine Perfusion Supported Organ Preservation. Pharmaceutics 2026, 18, 143. https://doi.org/10.3390/pharmaceutics18020143
Patel S, Pinky PP, Das AC, Copus JS, Aardema C, Crelli C, Troidle A, Lambert E, McCallin R, Surti V, et al. Perfluorocarbon Nanoemulsions for Simultaneous Delivery of Oxygen and Antioxidants During Machine Perfusion Supported Organ Preservation. Pharmaceutics. 2026; 18(2):143. https://doi.org/10.3390/pharmaceutics18020143
Chicago/Turabian StylePatel, Smith, Paromita Paul Pinky, Amit Chandra Das, Joshua S. Copus, Chip Aardema, Caitlin Crelli, Anneliese Troidle, Eric Lambert, Rebecca McCallin, Vidya Surti, and et al. 2026. "Perfluorocarbon Nanoemulsions for Simultaneous Delivery of Oxygen and Antioxidants During Machine Perfusion Supported Organ Preservation" Pharmaceutics 18, no. 2: 143. https://doi.org/10.3390/pharmaceutics18020143
APA StylePatel, S., Pinky, P. P., Das, A. C., Copus, J. S., Aardema, C., Crelli, C., Troidle, A., Lambert, E., McCallin, R., Surti, V., DiMarzio, C., Kopparthy, V., & Janjic, J. M. (2026). Perfluorocarbon Nanoemulsions for Simultaneous Delivery of Oxygen and Antioxidants During Machine Perfusion Supported Organ Preservation. Pharmaceutics, 18(2), 143. https://doi.org/10.3390/pharmaceutics18020143