Overcoming the Blood–Brain Barrier: Advanced Strategies in Targeted Drug Delivery for Neurodegenerative Diseases
Abstract
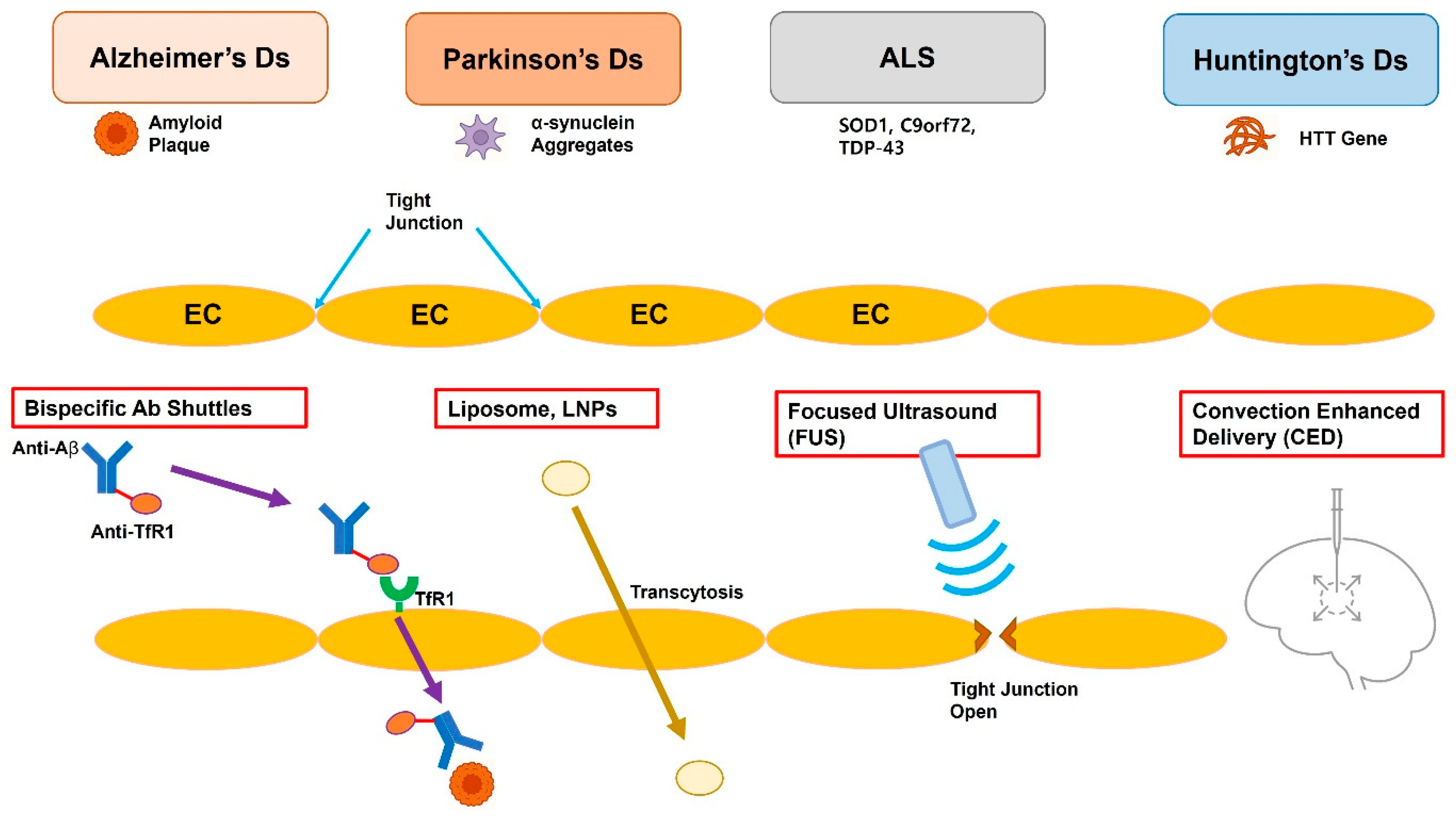
1. Introduction
2. Pathophysiology and Therapeutic Targets
2.1. Alzheimer’s Disease (AD)
2.2. Parkinson’s Disease (PD)
2.3. Amyotrophic Lateral Sclerosis (ALS)
2.4. Huntington’s Disease (HD)
2.5. Other Neurodegenerative Diseases
3. Barriers to CNS Drug Delivery
3.1. Blood–Brain Barrier (BBB)
3.2. Blood–CSF Barrier and Glymphatic Pathways
3.3. Disease-Specific Barrier Modulation
3.4. Physicochemical Constraints
4. Targeted Drug-Delivery Strategies
4.1. Medicinal Chemistry Optimization
4.2. Nanoparticle Platforms
4.2.1. Lipid Nanoparticles (LNPs)
4.2.2. Polymeric Nanoparticles
4.2.3. Inorganic and Hybrid Carriers
4.2.4. Exosomes and Biomimetic Vesicles
4.2.5. Nanogels and Hydrogel-Derived Carriers
4.2.6. Stimuli-Responsive Composite Systems
4.2.7. Theranostic and Multimodal Nanocarriers
4.3. Ligand-Directed and Bispecific Antibody Shuttles
4.4. Gene and RNA Therapies
4.4.1. Recombinant AAV Vectors
4.4.2. Non-Viral and Synthetic Vectors
4.4.3. Self-Amplifying RNA and Circular RNA
4.4.4. Genome and Epigenome Editing
4.5. Cell-Mediated “Trojan Horse” Delivery
4.6. Physical and Regional Techniques
4.6.1. Focused Ultrasound (FUS) + Microbubbles
4.6.2. Convection-Enhanced Delivery (CED)
4.6.3. Electromagnetic, Photothermal, and Magnetothermal Modalities
4.7. Integrative Analysis of Delivery Approaches
4.8. Emerging Combination Strategies and Platform Integration
5. Clinical Translation and Regulatory Landscape
5.1. Approved or Conditionally Approved Agents
5.2. Late-Stage Pipeline
5.3. Safety, Immunogenicity, and CMC Considerations
5.3.1. Immunogenicity
5.3.2. Genotoxicity
5.3.3. Chemistry, Controls, and Manufacturing (CCM)
5.4. Scaling Up Manufacturing and Supply Chains
5.5. Health Economics and Reimbursement
5.6. Evolution of Regulatory Frameworks
5.7. Digital Health Integration and Real-World Evidence
6. Future Perspectives and Integrative Approaches
6.1. AI-Guided Carrier Design
6.2. Multiplexed, “All-in-One” Nanomedicine
6.3. Personalized Exosome Therapy
6.4. Regenerative Genome Editing
6.5. Neuro-Immune Modulation
7. Conclusions
Funding
Conflicts of Interest
Abbreviations
| WHO | World Health Organization |
| PD | Parkinson’s disease |
| CNS | central nervous system |
| BBB | blood–brain barrier |
| AD | Alzheimer’s disease |
| Aβ | β-amyloid |
| GSK-3β | glycogen synthase kinase-3β |
| CDK5 | cyclin-dependent kinase 5 |
| TREM2 | Triggering Receptor Expressed on Myeloid cells 2 |
| NLRP3 | NOD-, LRR-, and pyrin domain-containing protein 3 |
| SNCA | synuclein alpha gene |
| PINK1 | PTEN-induced kinase 1 |
| GBA1 | Glucocerebrosidase 1 |
| ASOs | antisense oligonucleotides |
| GDNF | glial cell line-derived neurotrophic factor |
| ALS | amyotrophic lateral sclerosis |
| C9orf72 | chromosome 9 open reading frame 72 |
| SOD1 | superoxide dismutase 1 |
| TDP-43 | TAR DNA-binding protein 43 |
| HD | Huntington’s disease |
| HTT | huntingtin |
| AAV | adeno-associated virus |
| P-gp | P-glycoprotein |
| BCRP | breast cancer resistance protein |
| TfR | transferrin receptor |
| IR | insulin receptor |
| LRP1 | low-density lipoprotein receptor-related protein 1 |
| CSF | cerebrospinal fluid |
| clogP | calculated logP |
| PSA | polar surface area |
| MPO | multiparameter optimization |
| PET | positron emission tomography |
| LAT1 | L-type amino acid transporter 1 |
| LNPs | lipid nanoparticles |
| pKa | acid dissociation constant |
| GMP | Good Manufacturing Practices |
| PLGA-PEG | poly(lactic-co-glycolic acid)–polyethylene glycol |
| RVG29 | rabies virus glycoprotein 29 |
| MSNs | mesoporous silica nanoparticles |
| BDNF | brain-derived neurotrophic factor |
| iPSC | induced pluripotent stem cell |
| Fe3O4 | iron(II,III) oxide |
| MSC | mesenchymal stem cell |
| ROS | reactive oxygen species |
| RGDyK | arginylglycylaspartic acid-tyrosine-lysine |
| tMCAO | transient middle cerebral artery occlusion |
| MMP-9 | matrix metalloproteinase-9 |
| Rx | diagnostic and therapeutic |
| MRI | magnetic resonance imaging |
| FcRn | neonatal Fc receptor |
| AUC | area-under-the-curve |
| PrP | prion protein |
| siRNA | small interfering RNA |
| CED | Convection-Enhanced Delivery |
| VEE | Venezuelan equine encephalitis |
| saRNA | self-amplifying RNA |
| circRNA | circular RNA |
| SpCas9-HF1 | Streptococcus pyogenes Cas9-High Fidelity 1 |
| ABE8e | adenine base editor 8e |
| dCas9-KRAB-MeCP2 | deactivated Cas9 fused to a Krüppel-associated box and methyl-CpG-binding protein 2 |
| CAR | Chimeric Antigen Receptor |
| IL-10 | Interleukin-10 |
| TNF-α | tumor necrosis factor-alpha |
| IL-1β | Interleukin-1 beta |
| CD14 | Cluster of Differentiation 14 |
| NSC | neural stem cell |
| TK/GCV | thymidine kinase/ganciclovir |
| FUS | focused ultrasound |
| AADC | aromatic L-amino acid decarboxylase |
| UPDRS | Unified Parkinson’s Disease Rating Scale |
| AMF | alternating magnetic field |
| TRPV1 | transient receptor potential vanilloid 1 |
| PCI | photochemical internalization |
| FDA | U.S. Food and Drug Administration |
| ARIA | amyloid-related imaging abnormalities |
| PK–PD | pharmacokinetic–pharmacodynamic |
| CARPA | complement activation-related pseudo-allergy |
| pHEA | poly(2-hydroxyethyl aspartamide) |
| CCM | Chemistry, Controls, and Manufacturing |
| PDI | polydispersity index |
| vg | vector genomes |
| CEAs | cost-effectiveness analyses |
| ICER | incremental cost-effectiveness ratio |
| QALY | quality-adjusted life-year |
| EMA | European Medicines Agency |
| ATMP | Advanced Therapy Medicinal Product |
| AI | artificial intelligence |
References
- Dugger, B.N.; Dickson, D.W. Pathology of neurodegenerative diseases. Cold Spring Harb. Perspect. Biol. 2017, 9, a028035. [Google Scholar] [CrossRef]
- Nichols, E.; Steinmetz, J.D.; Vollset, S.E. Estimation of the Global Prevalence of Dementia in 2019 and Forecasted Prevalence in 2050. Lancet Public Health 2022, 7, e105–e125. [Google Scholar] [CrossRef]
- Dorsey, E.R.; Sherer, T.; Okun, M.S.; Bloem, B.R. The emerging evidence of the Parkinson pandemic. J. Parkinsons Dis. 2018, 8, S3–S8. [Google Scholar] [CrossRef] [PubMed]
- Kalia, L.V.; Lang, A.E. Parkinson’s Disease. Lancet 2015, 386, 896–912. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. Drug Transport across the Blood-Brain Barrier. J. Cereb. Blood Flow Metab. 2012, 32, 1959–1972. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A. From Blood-Brain Barrier to Blood-Brain Interface: New Opportunities for CNS Drug Delivery. Nat. Rev. Drug Discov. 2016, 15, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Hitchcock, S.A.; Pennington, L.D. Structure-Brain Exposure Relationships. J. Med. Chem. 2006, 49, 7559–7583. [Google Scholar] [CrossRef] [PubMed]
- Lajoie, J.M.; Shusta, E.V. Targeting Receptors for Drug Delivery Across the Blood-Brain Barrier. Annu. Rev. Pharmacol. Toxicol. 2015, 55, 613–631. [Google Scholar] [CrossRef]
- Meng, Y.; Pople, C.B.; Huang, Y. Noninvasive Brain Drug Delivery with Focused Ultrasound. Nat. Rev. Neurol. 2021, 17, 7–22. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Kauffman, K.J.; Anderson, D.G. Delivery technologies for genome editing. Nat. Rev. Drug Discov. 2017, 16, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.L.; Ritter, A.; Zhong, K. Clinical Trial Design for Neurodegenerative Diseases: A New Era. Alzheimers Dement. 2022, 18, 1092–1102. [Google Scholar]
- Walsh, D.M.; Selkoe, D.J. Deciphering the molecular basis of memory failure in Alzheimer’s disease. Neuron 2004, 44, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Medina, M.; Urrialde, L. Targeting Tau Pathologies in Alzheimer’s Disease and Other Tauopathies. Expert Rev. Neurother. 2023, 23, 113–123. [Google Scholar]
- Gao, Y.; Yang, G.; Zhang, S. TREM2 in Neurodegenerative Diseases. J. Neuroinflamm. 2023, 20, 112. [Google Scholar]
- Ulland, T.K.; Colonna, M. The NLRP3 Inflammasome in the Central Nervous System. Trends Immunol. 2018, 39, 513–524. [Google Scholar]
- van Dyck, C.H.; Swanson, C.J.; Aisen, P. Lecanemab in Early Alzheimer’s Disease. N. Engl. J. Med. 2023, 388, 9–21. [Google Scholar] [CrossRef]
- Swanson, C.J.; Zhang, Y.; Dhadda, S. A Randomized, Double-Blind, Placebo-Controlled Study of Donanemab in Early Symptomatic Alzheimer’s Disease (TRAILBLAZER-ALZ). N. Engl. J. Med. 2021, 384, 1691–1704. [Google Scholar]
- Wong, Y.C.; Krainc, D. α-Synuclein, Mitochondria, and Parkinson’s Disease. Trends Mol. Med. 2017, 23, 624–634. [Google Scholar]
- Mullin, S.; Schapira, A. Glucocerebrosidase and Parkinson Disease. Curr. Neurol. Neurosci. Rep. 2015, 15, 42. [Google Scholar]
- Mazzulli, J.R.; Zunke, F.; Isacson, O. GBA1 mutations cause synucleinopathy by impairing glucocerebrosidase activity and autophagic-lysosomal function. Cell 2016, 165, 156–168. [Google Scholar]
- Alarcón-Arís, D.; Morris, G.P.; Smith, K.D. Antisense Oligonucleotides for the Treatment of Neurodegenerative Diseases. Mov. Disord. 2023, 38, 445–459. [Google Scholar]
- Bartus, R.T.; Weinberg, M.S.; Samulski, R.J. AAV-Based Gene Therapy for Neurodegenerative Disorders of the CNS. Mov. Disord. 2020, 35, 115–130. [Google Scholar]
- Taylor, J.P.; Brown, R.H., Jr.; Cleveland, D.W. Decoding ALS: From Genes to Mechanism. Nature 2016, 539, 197–206. [Google Scholar] [CrossRef]
- Miller, R.G.; Jackson, C.E.; Kasarskis, E.J. Riluzole for Amyotrophic Lateral Sclerosis (ALS)/Motor Neuron Disease (MND). Neurology 2009, 73, 1218–1224. [Google Scholar] [CrossRef]
- Miller, T.M.; Cudkowicz, M.E.; Genge, A. Trial of Antisense Oligonucleotide Tofersen for SOD1 ALS. N. Engl. J. Med. 2022, 387, 1099–1110. [Google Scholar] [CrossRef]
- Wainger, B.J.; Buttermore, E.D.; Kiskinis, E. Intrinsic Membrane Hyperexcitability of ALS Patient-Derived Motor Neurons. Cell Rep. 2014, 7, 1–11. [Google Scholar] [CrossRef]
- Liu, K.; Muse, E.D.; Hartigan, C.R. RNA-targeting CRISPR-Cas13d systems for C9orf72-associated ALS and FTD. Cell 2022, 185, 2897–2912.e18. [Google Scholar]
- Ross, C.A.; Tabrizi, S.J. Huntington’s disease. Lancet 2011, 379, 113–128. [Google Scholar]
- Spronck, E.A.; Kent, T.J.; Howson, A. Preclinical evaluation of WVE-003, a stereopure antisense oligonucleotide targeting mutant huntingtin. Mol. Ther. Nucleic Acids 2023, 31, 467–477. [Google Scholar]
- Yang, S.; Chang, R.; Yang, H. CRISPR/Cas9-mediated gene editing in vitro and in vivo corrects structural and functional defects in a mouse model of Huntington’s disease. Mol. Ther. 2017, 25, 1966–1977. [Google Scholar]
- Przedborski, S. Neurodegeneration: What is it and where are we? Nat. Med. 2017, 23, 166–169. [Google Scholar]
- Jones, A.R.; Shusta, E.V. Blood-brain barrier transport of therapeutics via receptor-mediation. Pharm. Res. 2007, 24, 1759–1771. [Google Scholar] [CrossRef]
- Keep, R.F.; Jones, H.C.; Drewes, L.R. The choroid plexus as a therapeutic target. Adv. Drug Deliv. Rev. 2014, 71, 2–12. [Google Scholar]
- Louveau, A.; Smirnov, I.; Keyes, T.J. Structural and functional features of central nervous system lymphatic vessels. Nature 2015, 523, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Boland, B.; Yu, W.H.; Corti, O.; Mollereau, B.; Henriques, A.; Bezard, E.; Pastores, G.M.; Rubinsztein, D.C.; Nixon, R.A.; Duchen, M.R.; et al. Promoting the clearance of neurotoxic proteins in neurodegenerative disorders of ageing. Nat. Rev. Drug Discov. 2018, 17, 660–688. [Google Scholar] [CrossRef]
- Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.V. Blood-brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat. Rev. Neurol. 2018, 14, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Cramer, S.P.; Simonsen, H.; Frederiksen, J.L.; Rostrup, E.; Larsson, H.B. Abnormal blood-brain barrier permeability in normal appearing white matter in multiple sclerosis investigated by MRI. Neuroimage Clin. 2014, 4, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Al-Bachari, S.; Naish, J.H.; Parker, G.J.M.; Emsley, H.C.A.; Parkes, L.M. Blood-Brain Barrier Leakage Is Increased in Parkinson’s Disease. Front. Physiol. 2020, 11, 593026. [Google Scholar] [CrossRef]
- Kulkarni, J.A.; Darjuan, M.M.; Mercer, J.E.; Chen, S.; Van Der Meel, R.; Thewalt, J.L.; Tam, Y.Y.; Cullis, P.R. On the formation and morphology of lipid nanoparticles containing ionizable cationic lipids and siRNA. ACS Nano 2018, 12, 4787–4795. [Google Scholar] [CrossRef]
- Fernandes, T.B.; Segretti, M.C.F.; Polli, M.C.; Parise, R. Analysis of the Applicability and Use of Lipinski’s Rule for Central Nervous System Drugs. Lett. Drug Des. Discov. 2016, 13, 999–1006. [Google Scholar] [CrossRef]
- Pajouhesh, H.; Lenz, G.R. Medicinal Chemical Properties of Successful Central Nervous System Drugs. NeuroRx 2005, 2, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Wager, T.T.; Hou, X.; Verhoest, P.R. Central Nervous System Multiparameter Optimization of Drug-Like Properties. ACS Chem. Neurosci. 2010, 1, 435–449. [Google Scholar] [CrossRef]
- Bodor, N.; Buchwald, P. Soft drug design: Past, present, and future. Adv. Drug Deliv. Rev. 2000, 41, 5–22. [Google Scholar]
- Grunewald, G.L.; Dahan, A.M.; Pediaditakis, P. Nepicastat Prodrugs with Improved Oral Bioavailability. J. Med. Chem. 2004, 47, 5649–5652. [Google Scholar]
- Rautio, J.; Kumpulainen, H.; Heimbach, T. Prodrugs: Design and clinical applications. Nat. Rev. Drug Discov. 2008, 7, 255–270. [Google Scholar] [CrossRef]
- Wu, H.Q.; Ferreira, R.S.; Tella, J.G. Brain-Targeted Prodrug of a Kynurenine-3-monooxygenase Inhibitor. Mol. Pharm. 2014, 11, 1565–1574. [Google Scholar]
- Dong, Y.; Siegwart, D.J.; Anderson, D.G. Strategies for Designing Brain-Targeted Lipid Nanoparticles. Adv. Drug Deliv. Rev. 2019, 148, 133–147. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, J.A.; Witzigmann, D.; Thomson, S.B. The Current Landscape of Nucleic Acid Therapeutics. Nat. Nanotechnol. 2021, 16, 630–643. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Zeng, F.; Fu, D. Surfactant-free poly(lactide-co-glycolide) nanoparticles for improving in vitro anticancer efficacy of tetrandrine. J. Microencapsul. 2016, 33, 249–256. [Google Scholar] [CrossRef]
- Pardridge, W.M. Brain Delivery of Nanomedicines: Trojan Horse Liposomes for Plasmid DNA Gene Therapy of the Brain. Front. Med. Technol. 2020, 2, 602236. [Google Scholar] [CrossRef]
- Salvati, E.; Re, F.; Sesana, S.; Cambianica, I.; Sancini, G.; Masserini, M.; Gregori, M. Liposomes functionalized to overcome the blood-brain barrier and to target amyloid-beta peptide: The chemical design affects the permeability across an in vitro model. Int. J. Nanomed. 2013, 8, 1749–1758. [Google Scholar]
- Yang, F.; Lim, G.P.; Begum, A.N.; Ubeda, O.J.; Simmons, M.R.; Ambegaokar, S.S.; Chen, P.P.; Kayed, R.; Glabe, C.G.; Frautschy, S.A.; et al. Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J. Biol. Chem. 2005, 280, 5892–5901. [Google Scholar] [CrossRef]
- Tosi, G.; Duskey, J.T.; Kreuter, J. Nanoparticles as carriers for drug delivery of macromolecules across the blood-brain barrier. Expert Opin. Drug Deliv. 2020, 17, 23–32. [Google Scholar] [CrossRef]
- Hirsjärvi, S.; Sneck, A.; Fagerholm, U. Challenges and opportunities of polymeric nanoparticles for CNS drug delivery. Ther. Deliv. 2011, 2, 1035–1043. [Google Scholar]
- Zhou, R.; Zhu, L.; Zeng, Z.; Luo, R.; Zhang, J.; Guo, R.; Zhang, L.; Zhang, Q.; Bi, W. Targeted brain delivery of RVG29-modified rifampicin-loaded nanoparticles for Alzheimer’s disease treatment and diagnosis. Bioeng. Transl. Med. 2022, 7, e10395. [Google Scholar] [CrossRef] [PubMed]
- Fornaguera, C.; Dols-Perez, A.; Calderó, G.; García-Celma, M.J.; Camarasa, J.; Solans, C. PLGA nanoparticles pre-pared by nano-emulsion templating using low-energy methods as efficient nanocarriers for drug delivery across the blood-brain barrier. J. Control. Release 2015, 211, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, X.; Zhou, F. Mesoporous Silica Nanoparticles for Targeted Drug Delivery to the Central Nervous System. Adv. Mater. 2020, 32, 1902607. [Google Scholar]
- Chen, H.; Tian, J.; He, W. Gold Nanorod-Based Photothermal Therapy for Alzheimer’s Disease. Adv. Funct. Mater. 2023, 33, 2208794. [Google Scholar]
- Ho, D.; Lee, H.; Park, W. Nanoparticle-mediated neuromodulation. Nat. Biomed. Eng. 2019, 3, 862–876. [Google Scholar]
- de Almeida, F.M.; Inada, J.K. The “don’t eat me” signal, CD47, is a marker of self on the surface of several cell types. Stem Cells 2016, 34, 1967–1975. [Google Scholar]
- Haney, M.J.; Zhao, Y.; Harrison, E.B. Macrophage-Mediated Delivery of Catalase to the CNS for Neuroprotection. J. Control. Release 2015, 207, 177–186. [Google Scholar]
- Luan, X.; Sansanaphongpricha, K.; Myers, I. Engineering exosomes as refined drug delivery systems. Nat. Rev. Mater. 2021, 6, 1033–1050. [Google Scholar]
- Kim, M.S.; Haney, M.J.; Zhao, Y. RGD-Modified Exosomes for Targeted Delivery to Brain Ischemic Lesions. ACS Nano 2022, 16, 10832–10845. [Google Scholar]
- Wei, M.; Gao, Y.; Li, X.; Serpe, M.J. Stimuli-responsive polymers and their applications. Polym. Chem. 2017, 8, 127–143. [Google Scholar] [CrossRef]
- Sonoda, H.; Morimoto, H.; Yano, S. Nanogel-based delivery of antisense oligonucleotides to the brain for Alzheimer’s disease. J. Control. Release 2023, 355, 220–231. [Google Scholar]
- Wei, T.; Cheng, Q.; Min, Y.L. Systemic nanoparticle delivery of CRISPR-Cas9 ribonucleoproteins for effective tissue specific genome editing. Nat. Commun. 2020, 11, 3232. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.J.; Cha, M.Y.; Kim, D.; Kim, D.K.; Soh, M.; Shin, K.; Hyeon, T.; Mook-Jung, I. Mitochondria-Targeting Ceria Nanoparticles as Antioxidants for Alzheimer’s Disease. ACS Nano 2016, 10, 2860–2870. [Google Scholar] [CrossRef] [PubMed]
- Kelkar, S.S.; Reineke, T.M. Theranostics: A new paradigm for drug delivery. Bioconjug. Chem. 2011, 22, 1879–1903. [Google Scholar] [CrossRef]
- Terreno, E.; Delli Castelli, D.; Viale, A. Challenges for molecular magnetic resonance imaging. Chem. Rev. 2010, 110, 3019–3042. [Google Scholar] [CrossRef] [PubMed]
- Park, B.; Oh, D.; Kim, J.; Kim, C. Functional photoacoustic imaging: From nano- and micro- to macro-scale. Nano Converg. 2023, 10, 29. [Google Scholar] [CrossRef]
- Sonoda, H.; Morimoto, H.; Yano, S. A Transferrin Receptor Antibody-Iduronate-2-Sulfatase Fusion Protein for Neuron-Targeted Enzyme Delivery. Mol. Ther. 2018, 26, 1122–1129. [Google Scholar]
- Niewoehner, J.; Bohrmann, B.; Collin, L. Increased Brain Penetration and Potency of a Therapeutic Antibody Using a Monovalent Molecular Shuttle. Neuron 2014, 81, 49–60. [Google Scholar] [CrossRef]
- Zale, S.E.; Miles, S.D.; Balthasar, J.P. FcRn Optimization for Antibody Half-Life Extension. mAbs 2022, 14, 2094933. [Google Scholar]
- Lehmann, M.J. Aptamer-siRNA chimeras for targeted delivery of siRNA to the prion protein. Nucleic Acid Ther. 2016, 26, 169–180. [Google Scholar]
- Pardridge, W.M. Blood-brain barrier drug delivery of peptides and protein therapeutics. Bioconjug. Chem. 2015, 26, 2213–2222. [Google Scholar]
- Hordeaux, J.; Hinderer, C.; Goode, T. AAV-LK03 and AAV-MYO5: Novel AAV capsids with enhanced neuronal tropism. Hum. Gene Ther. 2021, 32, 257–263. [Google Scholar]
- LeWitt, P.A.; Rezai, A.R.; Leehey, M.A. AAV2-GAD Gene Therapy for Parkinson’s Disease: A Randomized Trial. N. Engl. J. Med. 2023, 388, 195–204. [Google Scholar]
- Christine, C.W.; Starr, P.A.; Larson, P.S. AAV2-AADC Gene Therapy for Parkinson’s Disease. Lancet Neurol. 2019, 18, 1112–1118. [Google Scholar]
- Wang, W.; Li, W.; Ma, N. Non-viral gene delivery methods. Nat. Rev. Mater. 2017, 2, 17024. [Google Scholar]
- Blakney, A.K.; Ip, S.; Geall, A.J. An Update on Self-Amplifying mRNA Vaccine Development. Vaccines 2021, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- Per-Olof, E. Self-amplifying RNA vaccines. Nat. Rev. Drug Discov. 2022, 21, 447–463. [Google Scholar]
- Dilliard, S.A.; Cheng, Q.; Siegwart, D.J. On the Mechanism of Tissue-Specific mRNA Delivery by Selective Organ Targeting Nanoparticles. Proc. Natl. Acad. Sci. USA 2021, 118, e2109256118. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Garcia, L.; Moreno-Martinez, L.; de la Torre, M.; Macias-Redondo, S.; Garcia-Redondo, A.; Osta, R.; Toivonen, J.M.; Calvo, A.C. Circular RNA expression in ALS is progressively deregulated and tissue-dependent. BMC Genom. 2025, 26, 576. [Google Scholar] [CrossRef]
- Levy, J.M.; Yeh, W.H.; Pendse, N. Cytosine Base Editing in Vivo Using Engineered CRISPR-Cas9. Nat. Biotechnol. 2020, 38, 44–48. [Google Scholar]
- Yang, Y.; Xu, J.; Wang, G. Epigenome editing by dCas9-KRAB-MeCP2 fusion protein for targeted gene silencing. Mol. Ther. 2019, 27, 1746–1758. [Google Scholar]
- Hickman, S.E.; Allison, E.K.; El Khoury, J. Microglial dysfunction and defective β-amyloid clearance pathways in aging Alzheimer’s disease mice. J. Neurosci. 2008, 28, 8354–8360. [Google Scholar] [CrossRef] [PubMed]
- Kordower, J.H.; Olanow, C.W.; Dodiya, H.B. Disease Duration and the Integrity of the Nigrostriatal System in Parkinson’s Disease. Brain 2013, 136, 2419–2431. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Lee, K.; Kim, S.H. Neural Stem Cell-Based Therapy for Neurodegenerative Diseases. Mol. Cells 2013, 35, 187–194. [Google Scholar]
- D’Souza, S.; Murthy, V.; Kasi, A. Suicide Gene Therapy in Cancer: A Review. Stem Cell Rev. Rep. 2022, 18, 119–130. [Google Scholar]
- Meng, Y.; Hynynen, K. Focused ultrasound for blood-brain barrier opening: A review of physical mechanisms. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2021, 68, 556–574. [Google Scholar]
- Rezai, A.R.; Ranjan, M.; D’Haese, P.F. Focused Ultrasound for Blood-Brain Barrier Opening in Alzheimer’s Disease. Nat. Med. 2023, 29, 1580–1585. [Google Scholar]
- Lipsman, N.; Meng, Y.; Bethune, A.J. Blood-Brain Barrier Opening in Alzheimer’s Disease Using MR-Guided Focused Ultrasound. Nat. Commun. 2018, 9, 2336. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, Z.I.; Kim, S.; Jikaria, N. Disrupting the blood-brain barrier by focused ultrasound induces sterile inflammation. Proc. Natl. Acad. Sci. USA 2017, 114, E75–E84. [Google Scholar] [CrossRef] [PubMed]
- Downs, M.E.; Buch, A.; Sierra, C. Long-term safety of repeated blood-brain barrier opening with focused ultrasound. J. Neurosurg. 2017, 127, 1419–1427. [Google Scholar]
- Krauze, M.T.; Saito, R.; Noble, C. Reflux-free convection-enhanced delivery. J. Neurosurg. 2005, 103, 901–906. [Google Scholar] [CrossRef]
- Yin, D.; Richardson, R.M. Convection-enhanced delivery for the treatment of brain disease. Expert Opin. Drug Deliv. 2016, 13, 677–686. [Google Scholar]
- Christine, C.W.; Bankiewicz, K.S.; Van Laar, A.D. AAV2-AADC gene therapy for Parkinson’s disease: Five-year data from a Phase 1b trial. J. Neurol. Neurosurg. Psychiatry 2022, 93, 56–63. [Google Scholar]
- Miller, T.M.; Pestronk, A.; David, W. An antisense oligonucleotide against SOD1 delivered intrathecally for ALS. N. Engl. J. Med. 2013, 369, 819–829. [Google Scholar]
- Chen, R.; Romero, G.; Christiansen, M.G. Wireless Magnetothermal Deep Brain Stimulation. Science 2021, 374, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Berg, K.; Selbo, P.K.; Prasmickaite, L. Photochemical internalization: A novel technology for delivery of macromolecules. Cancer Res. 2007, 67, 9979–9982. [Google Scholar]
- Timbie, K.F.; Mead, B.P.; Price, R.J. Drug and gene delivery across the blood-brain barrier with focused ultrasound. J. Control. Release 2015, 219, 61–75. [Google Scholar] [CrossRef]
- Mead, B.P.; Mastorakos, P.; Suk, J.S.; Klibanov, A.L.; Hanes, J.; Price, R.J. Targeted gene transfer to the brain via the delivery of brain-penetrating DNA nanoparticles with focused ultrasound. J. Control. Release 2016, 223, 109–117. [Google Scholar] [CrossRef]
- Liu, H.L.; Hua, M.Y.; Yang, H.W.; Huang, C.Y.; Chu, P.C.; Wu, J.S.; Tseng, I.C.; Wang, J.J.; Yen, T.C.; Chen, P.Y.; et al. Magnetic resonance monitoring of focused ultrasound/magnetic nanoparticle targeting delivery of therapeutic agents to the brain. Proc. Natl. Acad. Sci. USA 2010, 107, 15205–15210. [Google Scholar] [CrossRef]
- McDannold, N.; Arvanitis, C.D.; Vykhodtseva, N.; Livingstone, M.S. Temporary disruption of the blood-brain barrier by use of ultrasound and microbubbles: Safety and efficacy evaluation in rhesus macaques. Cancer Res. 2012, 72, 3652–3663. [Google Scholar] [CrossRef]
- Thanou, M.; Gedroyc, W. MRI-Guided Focused Ultrasound as a New Method of Drug Delivery. J. Drug Deliv. 2013, 2013, 616197. [Google Scholar] [CrossRef]
- Mainprize, T.; Lipsman, N.; Huang, Y.; Meng, Y.; Bethune, A.; Ironside, S.; Heyn, C.; Alkins, R.; Trudeau, M.; Sahgal, A.; et al. Blood-Brain Barrier Opening in Primary Brain Tumors with Non-invasive MR-Guided Focused Ultrasound: A Clinical Safety and Feasibility Study. Sci. Rep. 2019, 9, 321. [Google Scholar] [CrossRef]
- Burgess, A.; Hynynen, K. Drug delivery across the blood-brain barrier using focused ultrasound. Expert Opin. Drug Deliv. 2014, 11, 711–721. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Early Alzheimer’s Disease: Developing Drugs for Treatment; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2024.
- Minikel, E.V.; Janssens, A.C.J.W.; Sattar, N. ARIA Safety in Anti-Amyloid Immunotherapy Trials. Alzheimers Dement. 2023, 19, 1425–1437. [Google Scholar]
- Cole, T. A novel AAV capsid with enhanced CNS tropism. Mol. Ther. Methods Clin. Dev. 2021, 23, 211–224. [Google Scholar]
- Calcedo, R.; Vandenberghe, L.H.; Gao, G.; Lin, J.; Wilson, J.M. Worldwide epidemiology of neutralizing antibodies to adeno-associated viruses. J. Infect. Dis. 2009, 199, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Szebeni, J.; Storm, G. Complement activation-related pseudoallergy. Mol. Ther. 2021, 29, 2209–2219. [Google Scholar]
- Lino, C.A.; Harper, J.C.; Carney, J.P. Delivering CRISPR: A review of the challenges and approaches. Drug Deliv. 2018, 25, 1234–1257. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Human Gene Therapy Products Incorporating Human Genome Editing; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2024.
- Hou, X.; Zaks, T.; Langer, R. Lipid Nanoparticles for mRNA Delivery. Nat. Rev. Mater. 2022, 7, 265–280. [Google Scholar] [CrossRef] [PubMed]
- Clément, N.; Grieger, J.C. Manufacturing of AAV for clinical trials and commercial supply. Mol. Ther. Methods Clin. Dev. 2016, 3, 16002. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.D.; van der Geest, R.; Ratanji, K.D. Stability of lyophilized biologics. J. Pharm. Sci. 2018, 107, 1569–1581. [Google Scholar]
- Lin, P.J.; D’Abreu, A.; Neumann, P.J. The Cost Effectiveness of Lecanemab for Early Alzheimer’s Disease. Ann. Intern. Med. 2023, 176, 850–851. [Google Scholar]
- Garrison, L.P., Jr.; Neumann, P.J.; Willke, R.J. A Health Economics Approach to US Value Assessment Frameworks. Value Health 2018, 21, 161–165. [Google Scholar] [CrossRef]
- Hettle, R.; Corbett, M.; Hinde, S. The assessment and appraisal of regenerative medicines and cell therapy products. Health Policy 2017, 121, 467–476. [Google Scholar]
- European Medicines Agency. Guideline on the Quality, Non-Clinical and Clinical Aspects of Gene Therapy Medicinal Products; European Medicines Agency: Amsterdam, The Netherlands, 2024.
- Arora, S.; Venkataraman, V.; Zhan, A.; Donohue, S.; Biglan, K.M.; Dorsey, E.R.; Little, M.A. Detecting and monitoring the symptoms of Parkinson’s disease using smartphones: A pilot study. Park. Relat. Disord. 2015, 21, 650–653. [Google Scholar] [CrossRef] [PubMed]
- Rusz, J.; Hlavnicka, J.; Tykalova, T.; Buskova, J.; Ulmanova, O.; Ruzicka, E.; Sonka, K. Quantitative assessment of motor speech abnormalities in idiopathic rapid eye movement sleep behaviour disorder. Sleep Med. 2016, 19, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Henson, P.; Barnett, I.; Keshavan, M.; Torous, J. Towards clinically actionable digital phenotyping targets in schizophrenia. NPJ Schizophr. 2020, 6, 13. [Google Scholar] [CrossRef] [PubMed]
- Mathews, S.C.; McShea, M.J.; Hanley, C.L.; Ravitz, A.; Labrique, A.B.; Cohen, A.B. Digital health: A path to validation. NPJ Digit. Med. 2019, 2, 38. [Google Scholar] [CrossRef] [PubMed]
- Blonde, L.; Khunti, K.; Harris, S.B.; Meizinger, C.; Skolnik, N.S. Interpretation and Impact of Real-World Clinical Data for the Practicing Clinician. Adv. Ther. 2018, 35, 1763–1774. [Google Scholar] [CrossRef] [PubMed]
- Schneider, G.; Clark, D.E. Automated Design of Targeted Drug-like Molecules. Drug Discov. Today 2019, 24, 1182–1188. [Google Scholar]
- Kalani, A.; Tyagi, A.; Tyagi, N. Exosomes in Neurological Disease, Neuroprotection, and Therapy. Mol. Neurobiol. 2014, 49, 590–600. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; Kummer, M.P.; Stutz, A. NLRP3 is activated in Alzheimer’s disease and contributes to pathology. Nature 2013, 493, 674–678. [Google Scholar] [CrossRef]
| Disease | Therapeutic Target(s) | Representative Agent/Strategy | Key Limitation(s) | Ref. |
|---|---|---|---|---|
| AD | • Extracellular Aβ Plaques | Lecanemab, donanemab (anti-Aβ monoclonal antibodies) | • Modest cognitive benefit | [16,17] |
| • Risk of ARIA | ||||
| • Intracellular Tau Tangles | ASOs targeting Tau (e.g., BIIB080/IONIS-MAPTRx) | • Requires intrathecal delivery | [13] | |
| • Long-term efficacy unknown | ||||
| PD | • α-Synuclein Aggregates | ASOs (antisense oligonucleotides) targeting SNCA | • Unable to deliver to deep brain nuclei | [21] |
| • GBA1 Enzyme Deficiency | • Systemic side effects | |||
| • GDNF Replacement | AAV2-GDNF (gene therapy via CED) | • Invasive delivery (CED) | [22] | |
| • Pre-existing AAV immunity | ||||
| ALS | • Mutant SOD1 Protein | Tofersen (SOD1-targeting ASO) | • Benefits limited to specific genetic subtype | [25] |
| • Modest survival extension | ||||
| • C9orf72 Repeat Expansions | CRISPR-Cas13d (RNA editing) | • Off-target editing risks | [27] | |
| • In vivo delivery challenges | ||||
| HD | • Mutant Huntingtin (mHTT) | WVE-003 (allele-selective ASO) | • Difficulty balancing mHTT knockdown and normal HTT preservation | [29] |
| • HTT Gene | CRISPR/Cas9 (dual-AAV gene editing) | • Risk of permanent off-target DNA changes | [30] | |
| • AAV delivery limitations |
| Nanoparticle Platform | Key Features and Payloads | Advantages | Key Challenges | Refs. |
|---|---|---|---|---|
| Lipid Nanoparticles (LNPs) | • Ionizable lipids, PEGylation • Payloads: mRNA, siRNA, ASOs | • High encapsulation efficiency • Clinically validated and scalable | • Potential lipid toxicity • Moderate brain penetration | [39,47,48] |
| Polymeric Nanoparticles | • PLGA, PLA cores; surface ligands • Payloads: Small molecules, proteins | • Tunable drug release • High biocompatibility | • Lower payload capacity compared to LNPs • Potential for complement activation | [54,56] |
| Exosomes/Biomimetic | • Natural cell-derived vesicles • Payloads: Endogenous cargo, loaded drugs | • Low immunogenicity • Natural BBB-crossing ability | • Low production yield and purity • Batch-to-batch variability | [60,62] |
| Inorganic/Hybrid Carriers | • Gold, silica, iron oxide cores • Payloads: Drugs, imaging agents | • Theranostic capabilities • Externally triggerable release | • Long-term tissue accumulation • Complex, non-scalable synthesis | [57,59] |
| Technique | Mechanism of Action | Key Advantages | Key Challenges | Ref(s). |
|---|---|---|---|---|
| Focused Ultrasound (FUS) | • Microbubble-assisted transient opening of tight junctions | • Non-invasive and targeted • Reversible and repeatable | • Risk of microhemorrhage • High equipment cost | [90,91,92] |
| Convection-Enhanced Delivery (CED) | • Direct, pressure-driven infusion into brain parenchyma | • Bypasses the BBB entirely • High local drug concentration | • Highly invasive (surgical) • Limited to focal areas | [95,97] |
| Intranasal Delivery | • Bypasses BBB via olfactory and trigeminal neural pathways | • Non-invasive, patient-friendly • Rapid onset of action | • Low and variable bioavailability (10–30%) • Limited to specific molecules | [61] |
| Magnetothermal/Photothermal | • Nanoparticle-mediated local heating to increase permeability or trigger release | • High spatiotemporal control • On-demand drug release | • Requires co-delivered nanoparticles • Risk of tissue heating/damage | [99,100] |
| Delivery Platform | Primary BBB-Crossing Mechanism | Typical Brain Penetration | Main Strength | Primary Weakness | Refs. |
|---|---|---|---|---|---|
| Lipid/Polymeric Nanoparticles | Receptor-Mediated Transcytosis | Low to moderate | Versatility: Can carry diverse payloads (RNA, small molecules) | Efficiency: Low percentage of injected dose reaches brain | [47,54] |
| Bispecific Antibodies | Receptor-Mediated Transcytosis | Moderate to high | Specificity: High-affinity targeting of both BBB receptors and brain target | Complexity: High production cost and potential immunogenicity | [71,72] |
| AAV Gene Therapy | Transduction of Endothelial Cells/Neurons | High (with direct injection) | Durability: Potential for long-term or one-time treatment | Safety: Pre-existing immunity and genotoxicity risks | [76] |
| Focused Ultrasound (FUS) | Physical Disruption of Tight Junctions | High (in targeted region) | Universality: Enables delivery of nearly any systemic agent | Safety/Cost: Risk of hemorrhage and high equipment cost | [90,92] |
| Cell-Mediated Delivery | Active Migration Across BBB | Variable | Bio-integration: “Living drugs” can respond to microenvironment | Control: Difficult to control cell fate, distribution, and safety | [86,88] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, H.-M. Overcoming the Blood–Brain Barrier: Advanced Strategies in Targeted Drug Delivery for Neurodegenerative Diseases. Pharmaceutics 2025, 17, 1041. https://doi.org/10.3390/pharmaceutics17081041
Yang H-M. Overcoming the Blood–Brain Barrier: Advanced Strategies in Targeted Drug Delivery for Neurodegenerative Diseases. Pharmaceutics. 2025; 17(8):1041. https://doi.org/10.3390/pharmaceutics17081041
Chicago/Turabian StyleYang, Han-Mo. 2025. "Overcoming the Blood–Brain Barrier: Advanced Strategies in Targeted Drug Delivery for Neurodegenerative Diseases" Pharmaceutics 17, no. 8: 1041. https://doi.org/10.3390/pharmaceutics17081041
APA StyleYang, H.-M. (2025). Overcoming the Blood–Brain Barrier: Advanced Strategies in Targeted Drug Delivery for Neurodegenerative Diseases. Pharmaceutics, 17(8), 1041. https://doi.org/10.3390/pharmaceutics17081041







