Development of Nifedipine Phytantriol-Based Cubosomes and In Vitro Simulation of Administration Through Pediatric Feeding Tubes
Abstract
1. Introduction
2. Materials and Methods
2.1. The Materials
2.2. Preparation of the NIF-Loaded Nanocarrier
2.3. Physicochemical Characterization of the Nanocarriers
2.3.1. Particle Size and Distribution
2.3.2. The Zeta Potential
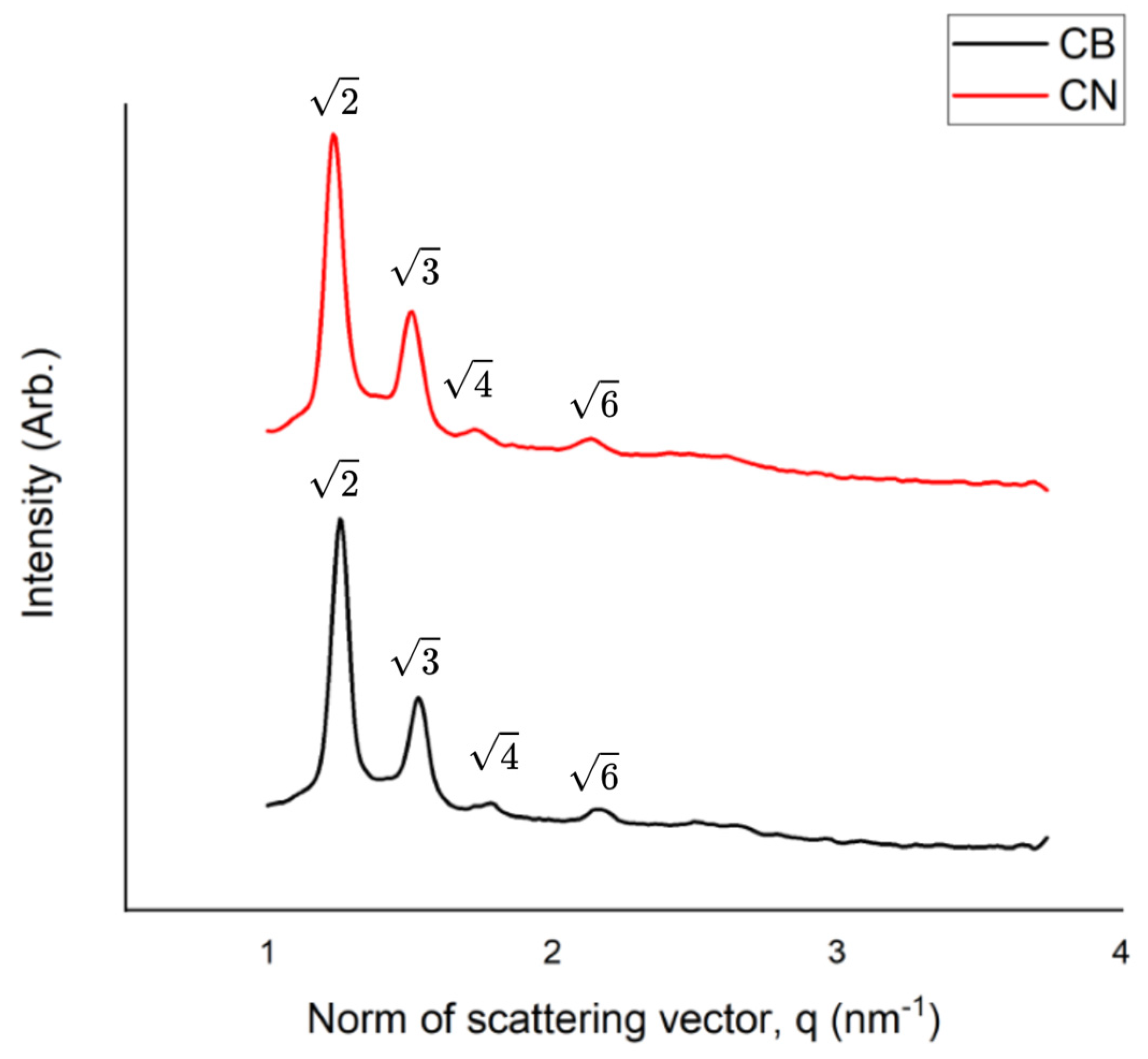
2.3.3. Small-Angle X-Ray Scattering Measurements
2.3.4. The NIF Content
2.3.5. The Drug Incorporation Efficiency
2.3.6. Determination of pH and Density
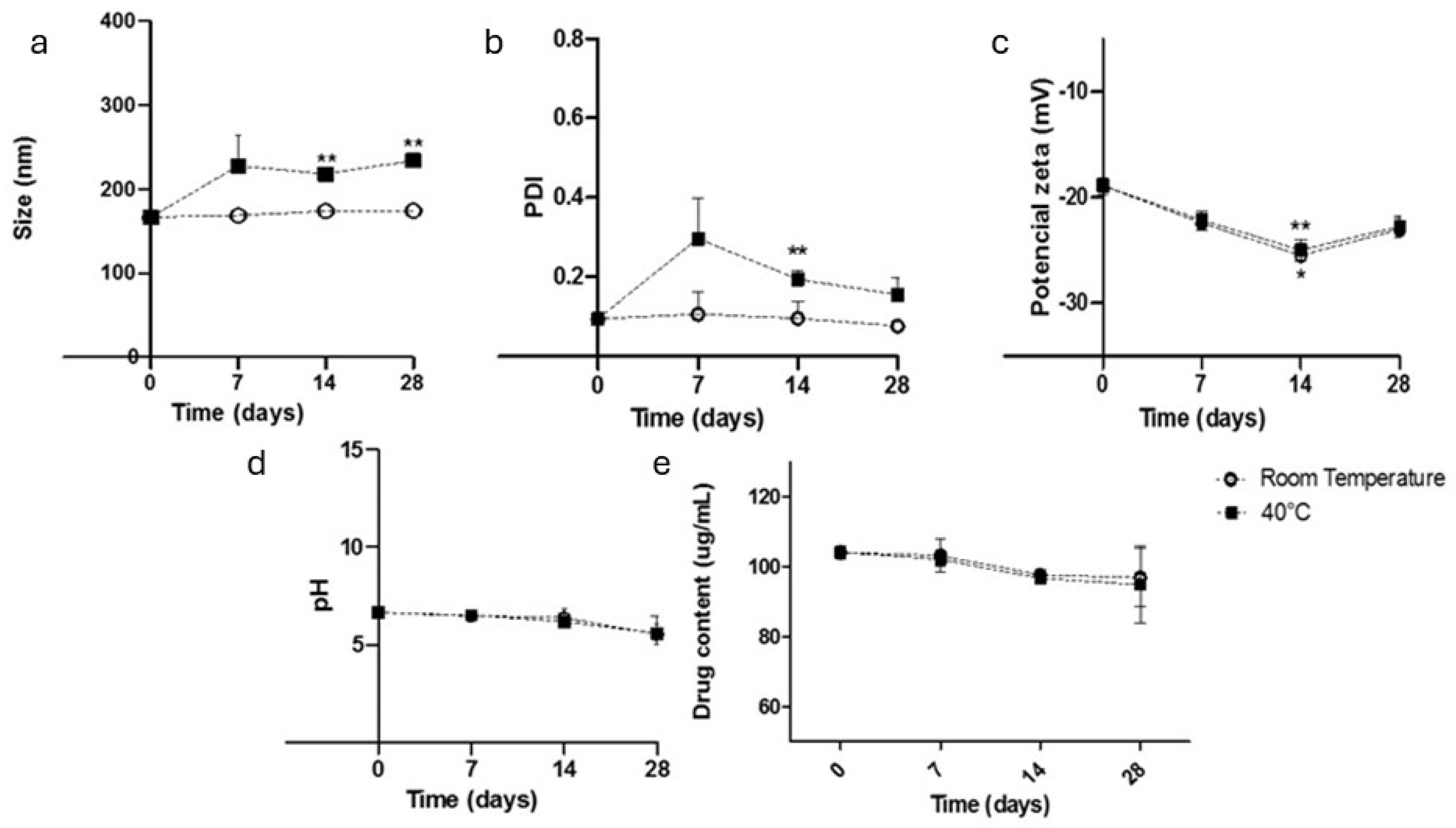
2.3.7. The Preliminary Stability Analysis
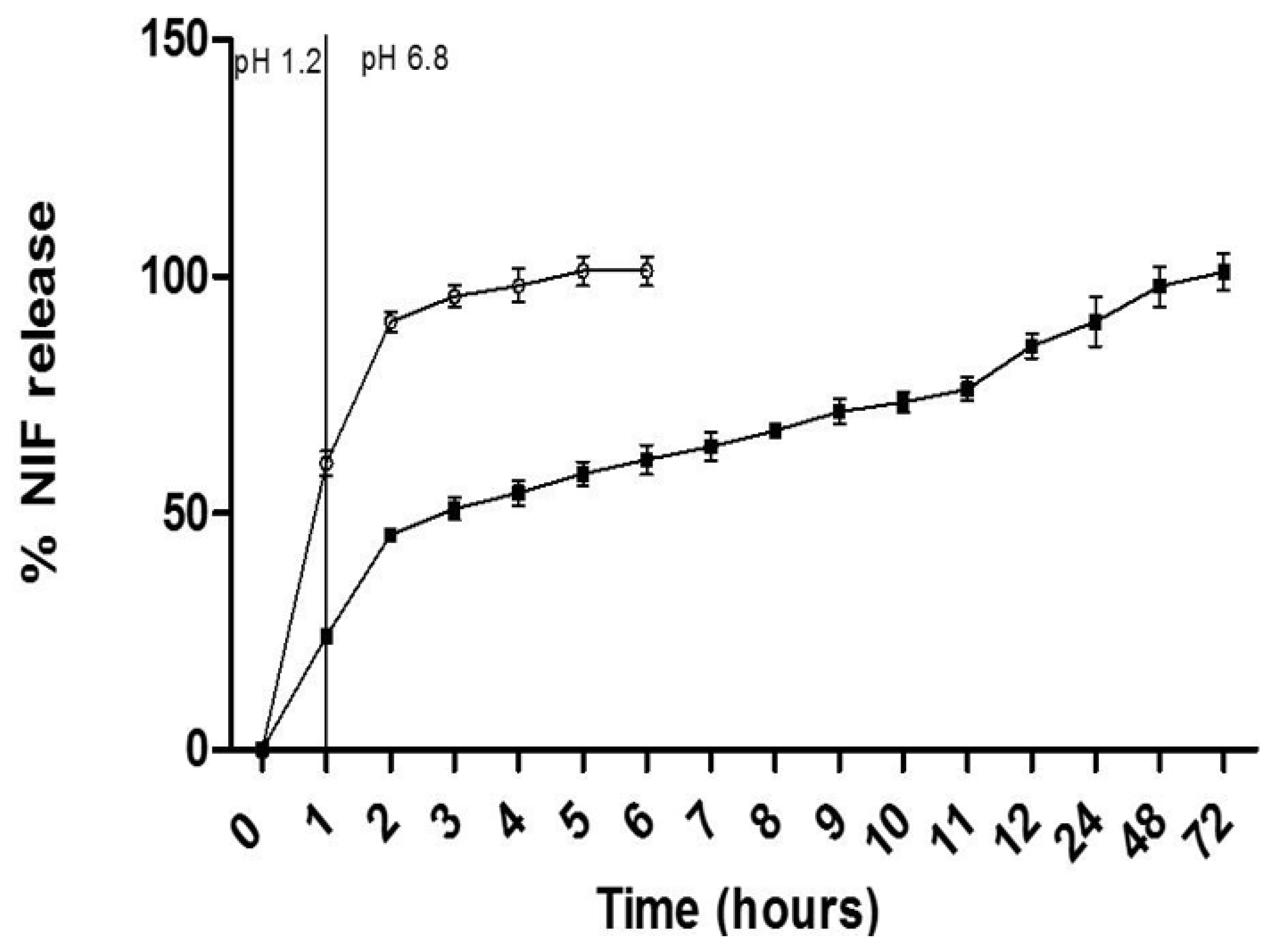
2.4. The In Vitro Release Profile in Simulated Gastric Fluid
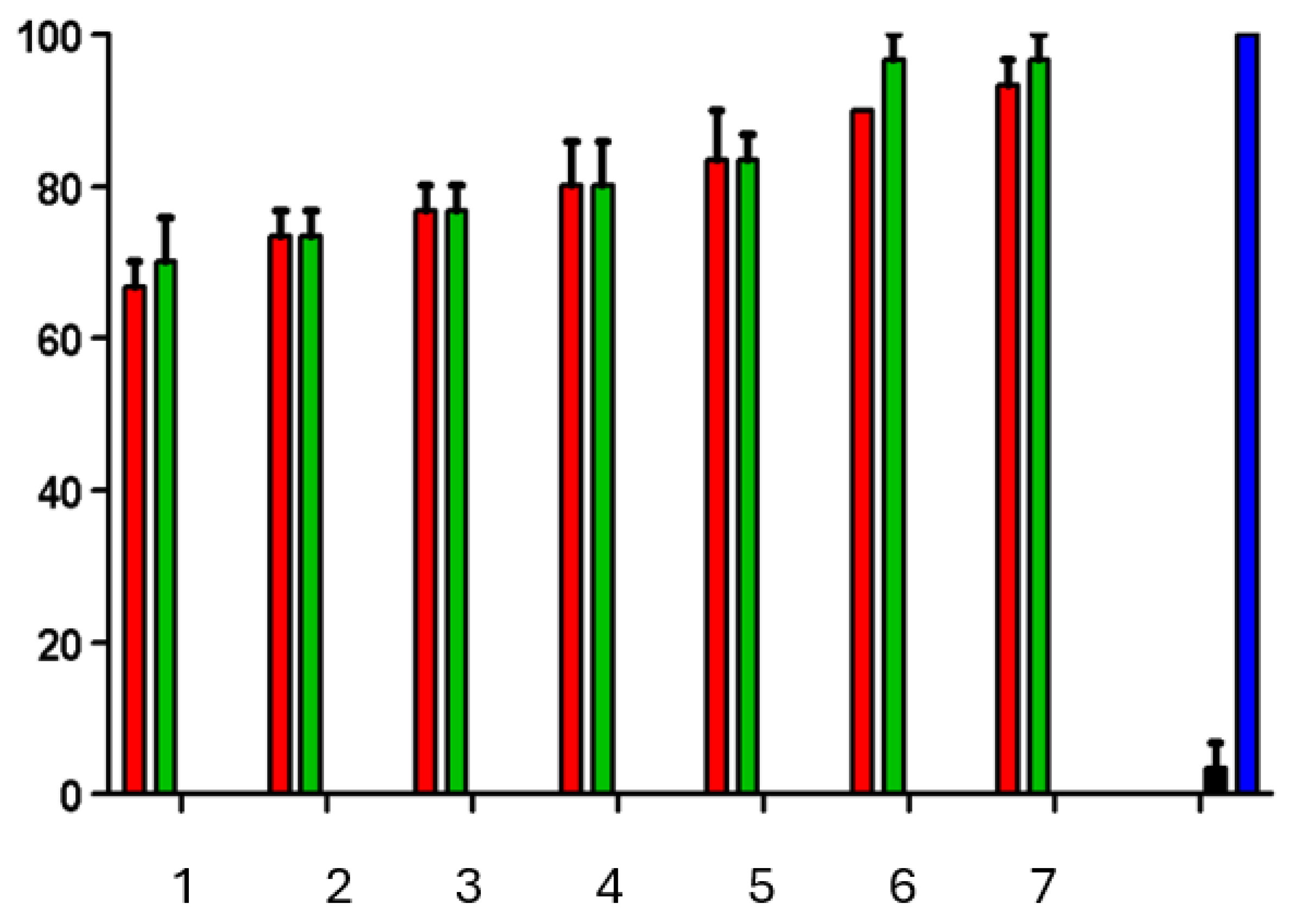
2.5. The Brine Shrimp Mortality Assay
2.6. Preparation of the Extemporaneous Nifedipine Suspensions
2.7. Evaluation of the Passage of the Formulations Through Pediatric Enteral Nutrition Tubes
2.8. The Statistical Analysis
3. Results
3.1. Physicochemical Characterization
3.2. In Vitro Release
3.3. The Passage of the Formulations Through Pediatric Enteral Nutrition Tubes
3.4. The Brine Shrimp Mortality Assay
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CNs | Cubosomes with Nifedipine |
| CB | Control Blank |
| FE-NIF | Extemporaneous Formulation of Nifedipine |
| NGT | Nasogastric Tube |
| IE (%) | Incorporation Efficiency |
| PDI | Polydispersity Index |
| SAXS | Small-Angle X-Ray Scattering |
| CL50 | Lethal Concentration for 50% |
| CTL+ | Positive Control |
| CTL− | Negative Control |
| FR | French |
References
- Garbacz, G.; Golke, B.; Wedemeyer, R.-S.; Axell, M.; Söderlind, E.; Abrahamsson, B.; Weitschies, W. Comparison of dissolution profiles obtained from nifedipine extended release once a day products using different dissolution test apparatuses. Eur. J. Pharm. Sci. 2009, 38, 147–155. [Google Scholar] [CrossRef]
- Huang, J.; Li, Y.; Wigent, R.J.; Malick, W.A.; Sandhu, H.K.; Singhal, D.; Shah, N.H. Interplay of formulation and process methodology on the extent of nifedipine molecular dispersion in polymers. Int. J. Pharm. 2011, 420, 59–67. [Google Scholar] [CrossRef]
- Sakurai, M.; Naruto, I.; Matsuyama, K. Evaluation for zero-order controlled release preparations of nifedipine tablet on dissolution test, together with cost benefit point of views. Yakugaku Zasshi-J. Pharm. Soc. Jpn. 2008, 128, 819–826. [Google Scholar] [CrossRef]
- Beig, A.; Miller, J.M.; Dahan, A. The interaction of nifedipine with selected cyclodextrins and the subsequent solubility–permeability trade-off. Eur. J. Pharm. Biopharm. 2013, 85, 1293–1299. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-Y.; Jin, S.-E.; Park, Y.; Lee, H.-J.; Park, Y.; Maeng, H.-J.; Kim, C.-K. Development of coated nifedipine dry elixir as a long acting oral delivery with bioavailability enhancement. Arch. Pharm. Res. 2011, 34, 1711–1717. [Google Scholar] [CrossRef] [PubMed]
- Blowey, D.L.; Duda, P.J.; Stokes, P.; Hall, M. Incidence and treatment of hypertension in the neonatal intensive care unit. J. Am. Soc. Hypertens. 2011, 5, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Mfoafo, K.A.; Omidian, M.; Bertol, C.D.; Omidi, Y.; Omidian, H. Neonatal and pediatric oral drug delivery: Hopes and hurdles. Int. J. Pharm. 2021, 597, 120296. [Google Scholar] [CrossRef]
- Thomas, J.; Stonebrook, E.; Kallash, M. Pediatric hypertension: Review of the definition, diagnosis, and initial management. Int. J. Pediatr. Adolesc. Med. 2022, 9, 1–6. [Google Scholar] [CrossRef]
- WHO. Nifedipine (Nifedipinum). In The International Pharmacopoeia [Internet], 11th ed.; WHO: Geneva, Switzerland, 2022. [Google Scholar]
- Salmon, D.; Pont, E.; Chevallard, H.; Diouf, E.; Tall, M.-L.; Pivot, C.; Pirot, F. Pharmaceutical and safety considerations of tablet crushing in patients undergoing enteral intubation. Int. J. Pharm. 2013, 443, 146–153. [Google Scholar] [CrossRef]
- Ponrouch, M.; Sautou-Miranda, V.; Boyer, A.; Bourdeaux, D.; Montagner, A.; Chopineau, J. Proton pump inhibitor administration via nasogastric tube in pediatric practice: Comparative analysis with protocol optimization. Int. J. Pharm. 2010, 390, 160–164. [Google Scholar] [CrossRef]
- Hallouard, F.; Dollo, G.; Brandhonneur, N.; Grasset, F.; Le Corre, P. Preparation and characterization of spironolactone-loaded nanoemulsions for extemporaneous applications. Int. J. Pharm. 2015, 478, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Jog, R.; Unachukwu, K.; Burgess, D. Formulation design space for stable, pH sensitive crystalline nifedipine nanoparticles. Int. J. Pharm. 2016, 514, 81–92. [Google Scholar] [CrossRef]
- Javed, I.; Ranjha, N.; Mahmood, K.; Kashif, S.; Rehman, M.; Usman, F. Drug release optimization from microparticles of poly(ε-caprolactone) and hydroxypropyl methylcellulose polymeric blends: Formulation and characterization. J. Drug Deliv. Sci. Technol. 2014, 24, 607–612. [Google Scholar] [CrossRef]
- Tagliari, M.P.; Granada, A.; Silva, M.A.S.; Stulzer, H.K.; Zanetti-Ramos, B.G.; Fernandes, D.; Silva, I.T.; Simões, C.M.O.; Sordi, R.; Assreuy, J.; et al. Development of oral nifedipine-loaded polymeric nanocapsules: Physicochemical characterisation, photostability studies, in vitro and in vivo evaluation. Quim. Nova 2015, 38, 781–786. [Google Scholar]
- Bordallo, E.; Torneiro, M.; Lazzari, M. Dissolution of amorphous nifedipine from micelle-forming carboxymethylcellulose derivatives. Carbohydr. Polym. 2020, 247, 116699. [Google Scholar] [CrossRef]
- Bi, Y.; Lv, B.; Li, L.; Lee, R.J.; Xie, J.; Qiu, Z.; Teng, L. A Liposomal Formulation for Improving Solubility and Oral Bioavailability of Nifedipine. Molecules 2020, 25, 338. [Google Scholar] [CrossRef]
- Ali, A.; Kataoka, N.; Ranneh, A.-H.; Iwao, Y.; Noguchi, S.; Oka, T.; Itai, S. Enhancing the Solubility and Oral Bioavailability of Poorly Water-Soluble Drugs Using Monoolein Cubosomes. Chem. Pharm. Bull. 2017, 65, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Barriga, H.; Holme, M.; Stevens, M. Cubosomes: The Next Generation of Smart Lipid Nanoparticles? Angew. Chem.-Int. Edit. 2019, 58, 2958–2978. [Google Scholar] [CrossRef] [PubMed]
- Azmi, I.D.M.; Moghimi, S.M.; Yaghmur, A. Cubosomes and Hexosomes as Versatile Platforms for Drug Delivery. Ther. Deliv. 2015, 6, 1347–1364. [Google Scholar] [CrossRef]
- Karami, Z.; Hamidi, M. Cubosomes: Remarkable drug delivery potential. Drug Discov. Today 2016, 21, 789–801. [Google Scholar] [CrossRef]
- Nguyen, T.; Hanley, T.; Porter, C.; Boyd, B. Nanostructured liquid crystalline particles provide long duration sustained-release effect for a poorly water soluble drug after oral administration. J. Control. Release 2011, 153, 180–186. [Google Scholar] [CrossRef]
- Akhlaghi, S.; Ribeiro, I.; Boyd, B.; Loh, W. Impact of preparation method and variables on the internal structure, morphology, and presence of liposomes in phytantriol-Pluronic® F127 cubosomes. Colloid Surf. B-Biointerfaces 2016, 145, 845–853. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Shen, J.Q.; Gan, Y.; Geng, H.M.; Zhang, X.X.; Zhu, C.L.; Gan, L. Novel vehicle based on cubosomes for ophthalmic delivery of flurbiprofen with low irritancy and high bioavailability. Acta Pharmacol. Sin. 2010, 31, 990–998. [Google Scholar] [CrossRef]
- Marques, S.S.; Ramos, I.I.; Fernandes, S.R.; Barreiros, L.; Lima, S.A.; Reis, S.; Domingues, M.R.M.; Segundo, M.A. Insights on Ultrafiltration-Based Separation for the Purification and Quantification of Methotrexate in Nanocarriers. Molecules 2020, 25, 1879. [Google Scholar] [CrossRef] [PubMed]
- Guedes, M.D.V.; Marques, M.S.; Berlitz, S.J.; Facure, M.H.M.; Correa, D.S.; Steffens, C.; Contri, R.V.; Külkamp-Guerreiro, I.C. Lamivudine and Zidovudine-Loaded Nanostructures: Green Chemistry Preparation for Pediatric Oral Administration. Nanomaterials 2023, 13, 770. [Google Scholar] [CrossRef]
- Mudie, D.M.; Murray, K.; Hoad, C.L.; Pritchard, S.E.; Garnett, M.C.; Amidon, G.L.; Gowland, P.A.; Spiller, R.C.; Amidon, G.E.; Marciani, L. Quantification of Gastrointestinal Liquid Volumes and Distribution Following a 240 mL Dose of Water in the Fasted State. Mol. Pharm. 2014, 11, 3039–3047. [Google Scholar] [CrossRef] [PubMed]
- Diefenthaeler, H.S.; Bianchin, M.D.; Marques, M.S.; Nonnenmacher, J.L.; Bender, E.T.; Bender, J.G.; Nery, S.F.; Cichota, L.C.; Külkamp-Guerreiro, I.C. Omeprazole nanoparticles suspension: Development of a stable liquid formulation with a view to pediatric administration. Int. J. Pharm. 2020, 589, 119818. [Google Scholar] [CrossRef]
- Meyer, B.; Ferrigni, N.; Putnam, J.; Jacobsen, L.; Nichols, D.; Mclaughlin, J. Brine shrimp—A convenient general bioassay for active-plant constituents. Planta Med. 1982, 45, 31–34. [Google Scholar] [CrossRef]
- Leng, K.; Vijayarathna, S.; Jothy, S.; Sasidharan, S.; Kanwar, J. In vitro and in vivo toxicity assessment of alginate/eudragit S 100-enclosed chitosan-calcium phosphate-loaded iron saturated bovine lactoferrin nanocapsules (Fe-bLf NCs). Biomed. Pharmacother. 2018, 97, 26–37. [Google Scholar] [CrossRef]
- McLaughlin, J.; Rogers, L. The use of biological assays to evaluate botanicals. Drug Inf. J. 1998, 32, 513–524. [Google Scholar] [CrossRef]
- Messaouik, D.; Sautou-Miranda, V.; Bagel-Boithias, S.; Chopineau, J. Comparative study and optimization of the administration mode of three proton pump inhibitors by nasogastric tube. Int. J. Pharm. 2005, 299, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Maria do Rosário, D.L.; Marchini, J.S. Sonda nasogástrica/nasoentérica: Cuidados na instalação, na administração da dieta e prevenção de complicações. Medicina 2002, 35, 95–101. [Google Scholar] [CrossRef][Green Version]
- van den Anker, J.N.; Schwab, M.; Kearns, G.L. Developmental Pharmacokinetics. In Pediatric Clinical Pharmacology [Internet]; Seyberth, H.W., Rane, A., Schwab, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 51–75. [Google Scholar] [CrossRef]
- Barradas, T.N.; de Campos, V.E.B.; Senna, J.P.; Coutinho, C.d.S.C.; Tebaldi, B.S.; e Silva, K.G.d.H.; Mansur, C.R.E. Development and characterization of promising o/w nanoemulsions containing sweet fennel essential oil and nonionic sufactants. Colloid Surf. A-Physicochem. Eng. Asp. 2015, 480, 214–221. [Google Scholar] [CrossRef]
- Avachat, A.; Parpani, S. Formulation and development of bicontinuous nanostructured liquid crystalline particles of efavirenz. Colloid Surf. B-Biointerfaces 2015, 126, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Basniwal, P.; Shrivastava, P.; Jain, D. Hydrolytic degradation profile and RP-HPLC estimation of cilostazol in tablet dosage form. Indian J. Pharm. Sci. 2008, 70, 222. [Google Scholar] [CrossRef] [PubMed]
- Waterman, K.C.; Adami, R.C.; Alsante, K.M.; Antipas, A.S.; Arenson, D.R.; Carrier, R.; Hong, J.; Landis, M.S.; Lombardo, F.; Shah, J.C.; et al. Hydrolysis in Pharmaceutical Formulations. Pharm. Dev. Technol. 2002, 7, 113–146. [Google Scholar] [CrossRef]
- Kazemi, I.; Sefid, M.; Afrand, M. A novel comparative experimental study on rheological behavior of mono & hybrid nanofluids concerned graphene and silica nanopowders: Characterization, stability and viscosity measurements. Powder Technol. 2020, 366, 216–229. [Google Scholar]
- Elkomy, M.H.; Khallaf, R.A.; Mahmoud, M.O.; Hussein, R.R.S.; El-Kalaawy, A.M.; Abdel-Razik, A.-R.H.; Aboud, H.M. Intratracheally Inhalable Nifedipine-Loaded Chitosan-PLGA Nanocomposites as a Promising Nanoplatform for Lung Targeting: Snowballed Protection via Regulation of TGF-β/β-Catenin Pathway in Bleomycin-Induced Pulmonary Fibrosis. Pharmaceuticals 2021, 14, 1225. [Google Scholar] [CrossRef]
- Hyde, S.T. Identification of Lyotropic Liquid Crystalline Mesophases. In Handbook of Applied Surface and Colloid Chemistry; Wiley: New York, NY, USA, 2001; pp. 299–332. [Google Scholar]
- Ali, M.; Noguchi, S.; Iwao, Y.; Oka, T.; Itai, S. Preparation and Characterization of SN-38-Encapsulated Phytantriol Cubosomes Containing α-Monoglyceride Additives. Chem. Pharm. Bull. 2016, 64, 577–584. [Google Scholar] [CrossRef]
- Ruzsíková, A.; Soucková, L.; Suk, P.; Opatrilová, R.; Kejdusová, M.; Srámek, V. Quantitative analysis of drug losses administered via nasogastric tube—In vitro study. Int. J. Pharm. 2015, 478, 368–371. [Google Scholar] [CrossRef]
- Tawfic, Q.A.; Bhakta, P.; Date, R.R.; Sharma, P.K. Esophageal bezoar formation due to solidification of enteral feed administered through a malpositioned nasogastric tube: Case report and review of the literature. Acta Anesthesiol. Taiwanica 2012, 50, 188–190. [Google Scholar] [CrossRef] [PubMed]
- Pecoraro, R.; Scalisi, E.M.; Messina, G.; Fragalà, G.; Ignoto, S.; Salvaggio, A.; Zimbone, M.; Impellizzeri, G.; Brundo, M.V. Artemia salina: A microcrustacean to assess engineered nanoparticles toxicity. Microsc. Res. Tech. 2020, 84, 531–536. [Google Scholar] [CrossRef] [PubMed]
| Sample | ||
|---|---|---|
| CB | CN | |
| D [4, 3] (v) (nm) | 145 ± 6.1 a | 152 ± 5.5 a |
| Span (v) | 1.22 ± 0.02 a | 1.22 ± 0.02 a |
| Size (nm) | 156 ± 0.17 a | 159 ± 2.8 a |
| PDI | 0.099 ± 0.001 a | 0.094 ± 0.018 a |
| Zeta potential (mV) | −17.3 ± 0.55 a | −18.9 ± 0.78 a |
| pH | 6.79 ± 0.07 a | 6.64 ± 0.12 a |
| Density (g/mL) | 1.050 ± 0.0014 a | 1.020 ± 0.001 a |
| Drug content (%) | - | 99 ± 0.69 |
| IE (%) | - | 94.73 ± 4.9 |
| Total Drug Recovered (%) After Passing Through the Probe | ||||||
|---|---|---|---|---|---|---|
| Probe Caliber | Sample | Original Drug Content (%) | Probe 1 (1 mL Rinse) | Probe 2 (2 mL Rinse) | Probe 3 (5 mL Rinse) | Probe 4 (10 mL Rinse) |
| 4 FR | FE-NIF | 101.1 ± 1.8 a | 62.5 ± 4.2 b | 67.6 ± 5.6 b | — | — |
| CN | 102.5 ± 4 a | 103.9 ± 4 a | 100.3 ± 4 a | — | — | |
| 6 FR | FE-NIF | 101.1 ± 1.8 a | — | 65.0 ± 12 b | 72.6 ± 6 b | 82.9 ± 9 b |
| CN | 102.5 ± 4 a | — | 103.9 ± 4 a | 100.3 ± 4 a | 100.8 ± 5 a | |
| 8 FR | FE-NIF | 101.1 ± 1.8 a | — | 70.2 ± 9.3 b | 78.1 ± 8.9 b | 91.1 ± 10 a,b |
| CN | 102.5 ± 4 a | — | 103.9 ± 4 a | 100.3 ± 4 a | 100.8 ± 5 a | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima, L.A.; Moura, E.E.L.d.; Fraga, S.F.; Contri, R.V.; Külkamp-Guerreiro, I.C. Development of Nifedipine Phytantriol-Based Cubosomes and In Vitro Simulation of Administration Through Pediatric Feeding Tubes. Pharmaceutics 2025, 17, 828. https://doi.org/10.3390/pharmaceutics17070828
Lima LA, Moura EELd, Fraga SF, Contri RV, Külkamp-Guerreiro IC. Development of Nifedipine Phytantriol-Based Cubosomes and In Vitro Simulation of Administration Through Pediatric Feeding Tubes. Pharmaceutics. 2025; 17(7):828. https://doi.org/10.3390/pharmaceutics17070828
Chicago/Turabian StyleLima, Lorena Almeida, Euler Eduardo Lisboa de Moura, Schauana Freitas Fraga, Renata Vidor Contri, and Irene Clemes Külkamp-Guerreiro. 2025. "Development of Nifedipine Phytantriol-Based Cubosomes and In Vitro Simulation of Administration Through Pediatric Feeding Tubes" Pharmaceutics 17, no. 7: 828. https://doi.org/10.3390/pharmaceutics17070828
APA StyleLima, L. A., Moura, E. E. L. d., Fraga, S. F., Contri, R. V., & Külkamp-Guerreiro, I. C. (2025). Development of Nifedipine Phytantriol-Based Cubosomes and In Vitro Simulation of Administration Through Pediatric Feeding Tubes. Pharmaceutics, 17(7), 828. https://doi.org/10.3390/pharmaceutics17070828










