Calcium Electrochemotherapy and Challenges in Combined Treatment with Dendritic Cell Vaccination
Abstract
1. Introduction
2. Materials and Methods
2.1. Mice and Tumor Induction
2.2. Electroporation Procedure
2.3. Dendritic Cell Vaccine Preparation
2.4. Experimental Scheme
2.5. Flow Cytometry
2.6. Determination of Antitumor Antibodies
2.7. Methods of Statistical Analysis
3. Results
3.1. Evaluation of the Phenotype of Prepared DCV
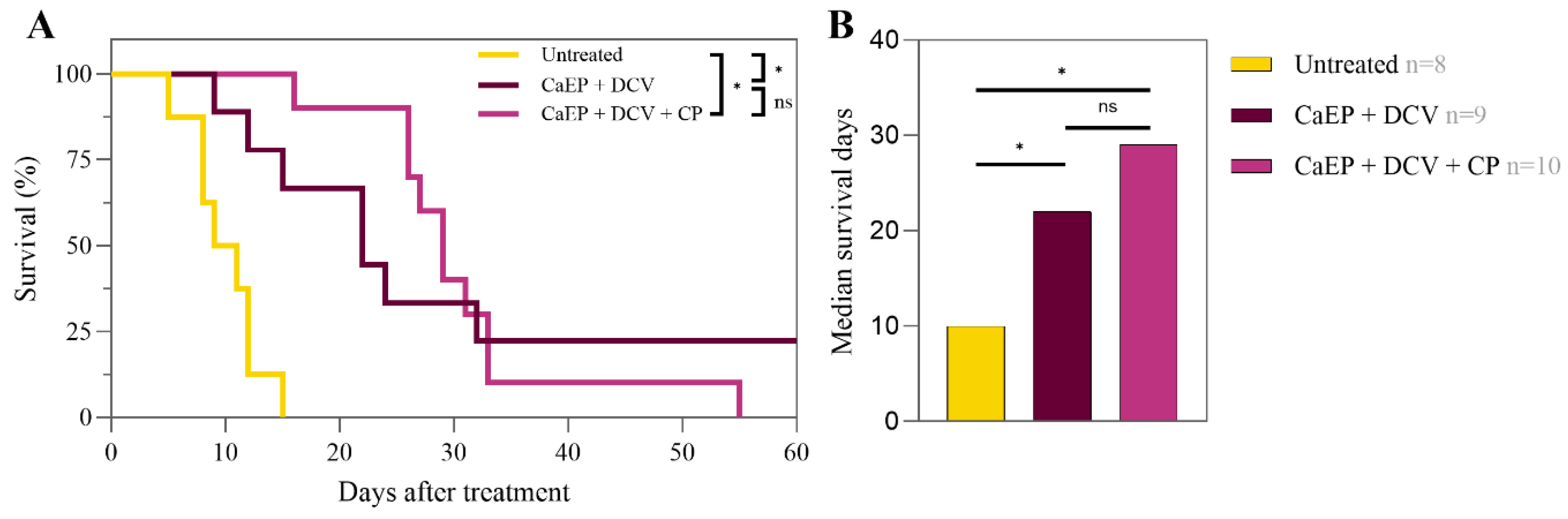
3.2. Survival Rates
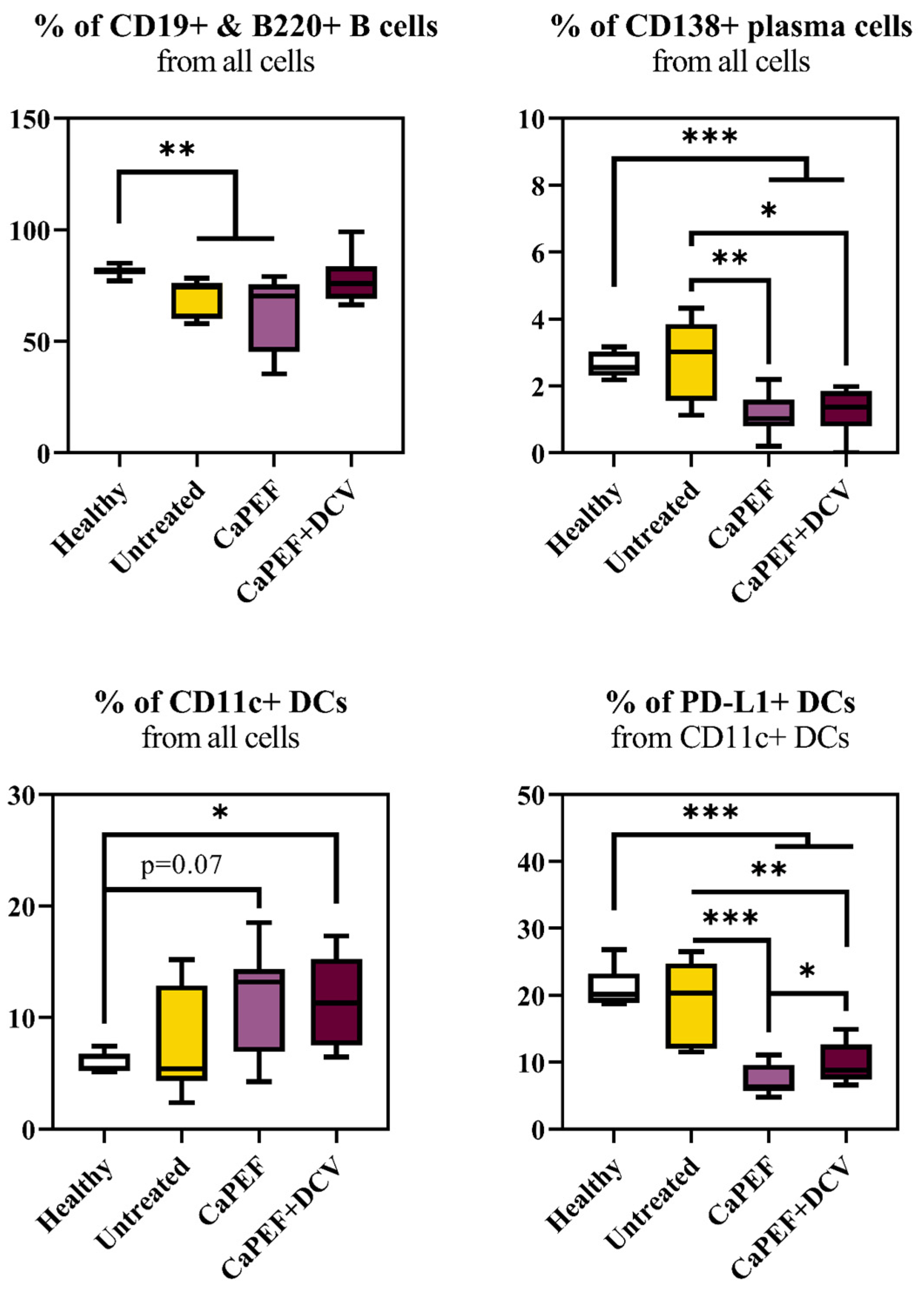
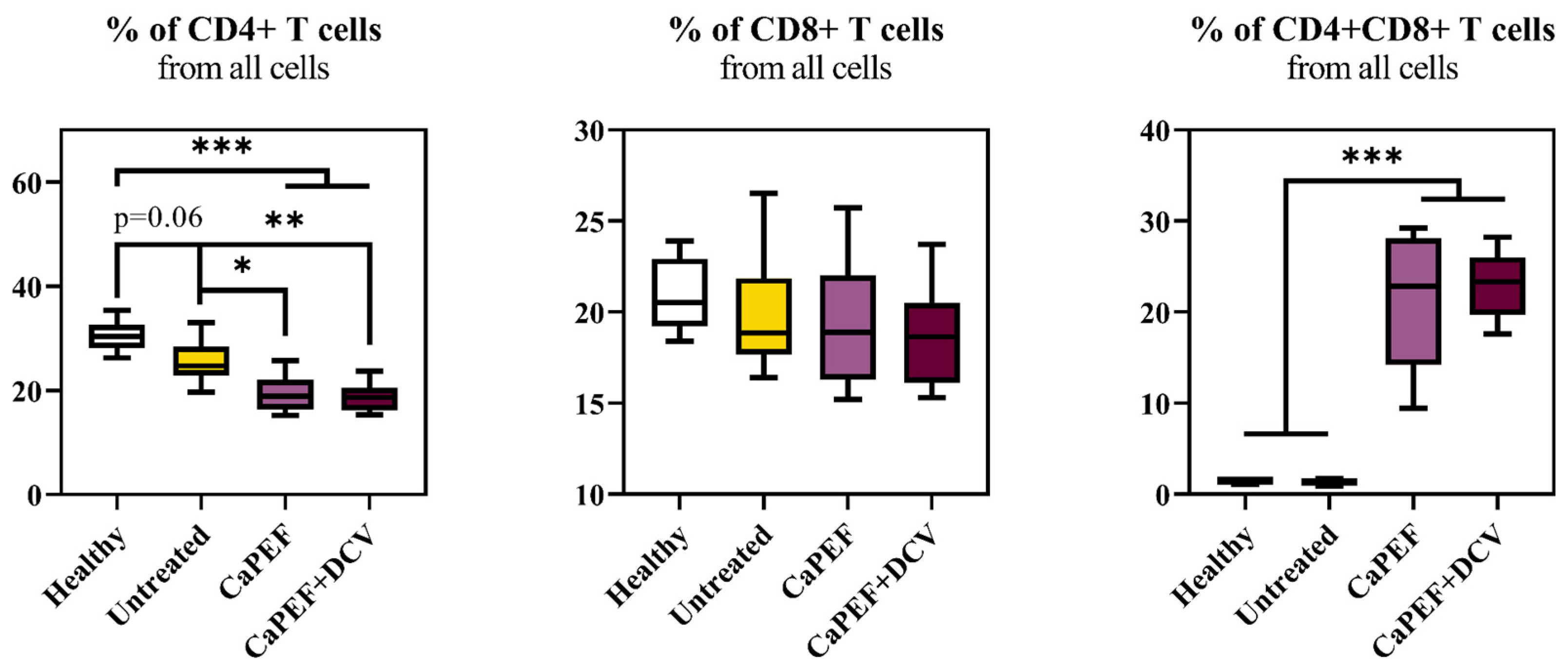
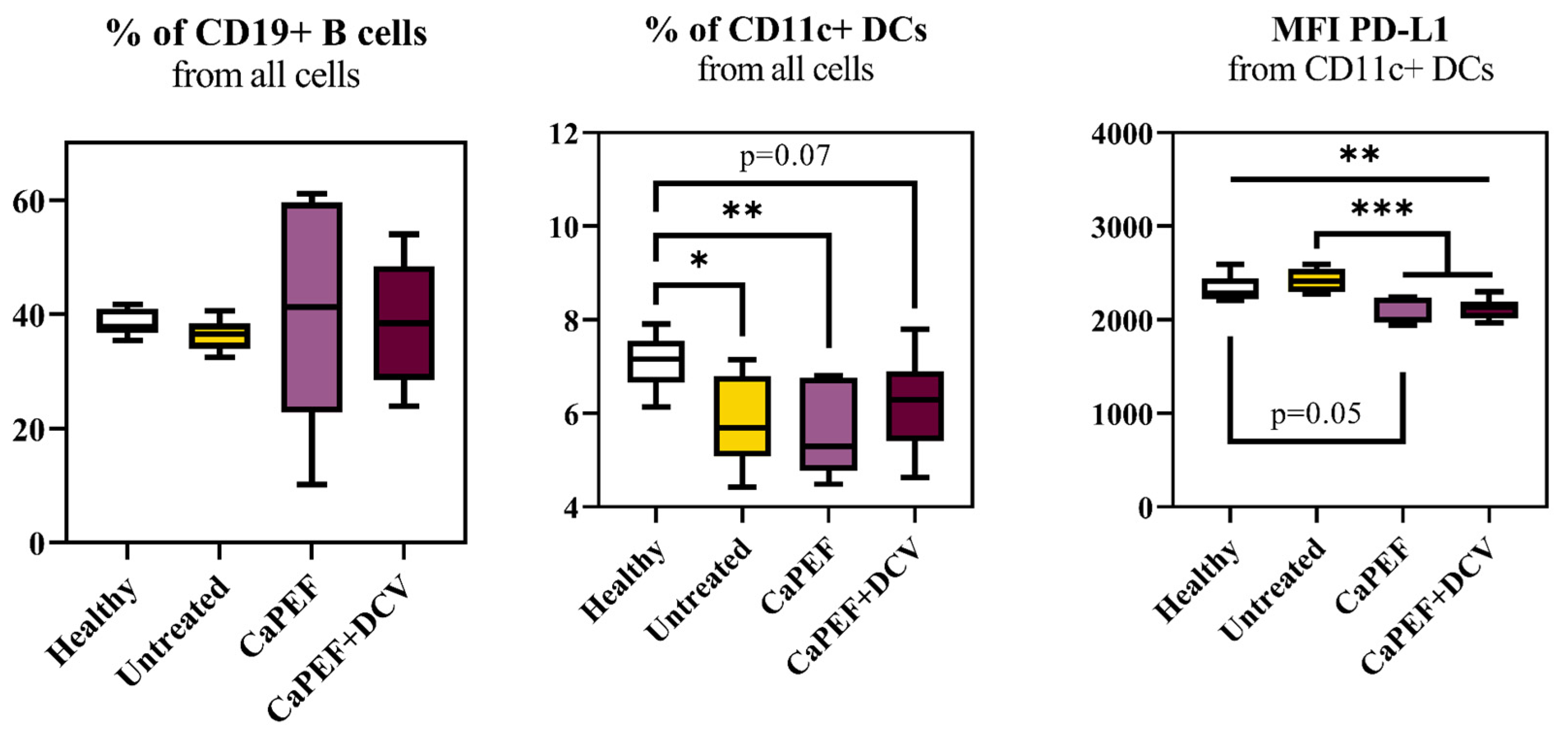
3.3. Lymphocyte Subsets in Spleen and Lymph Nodes
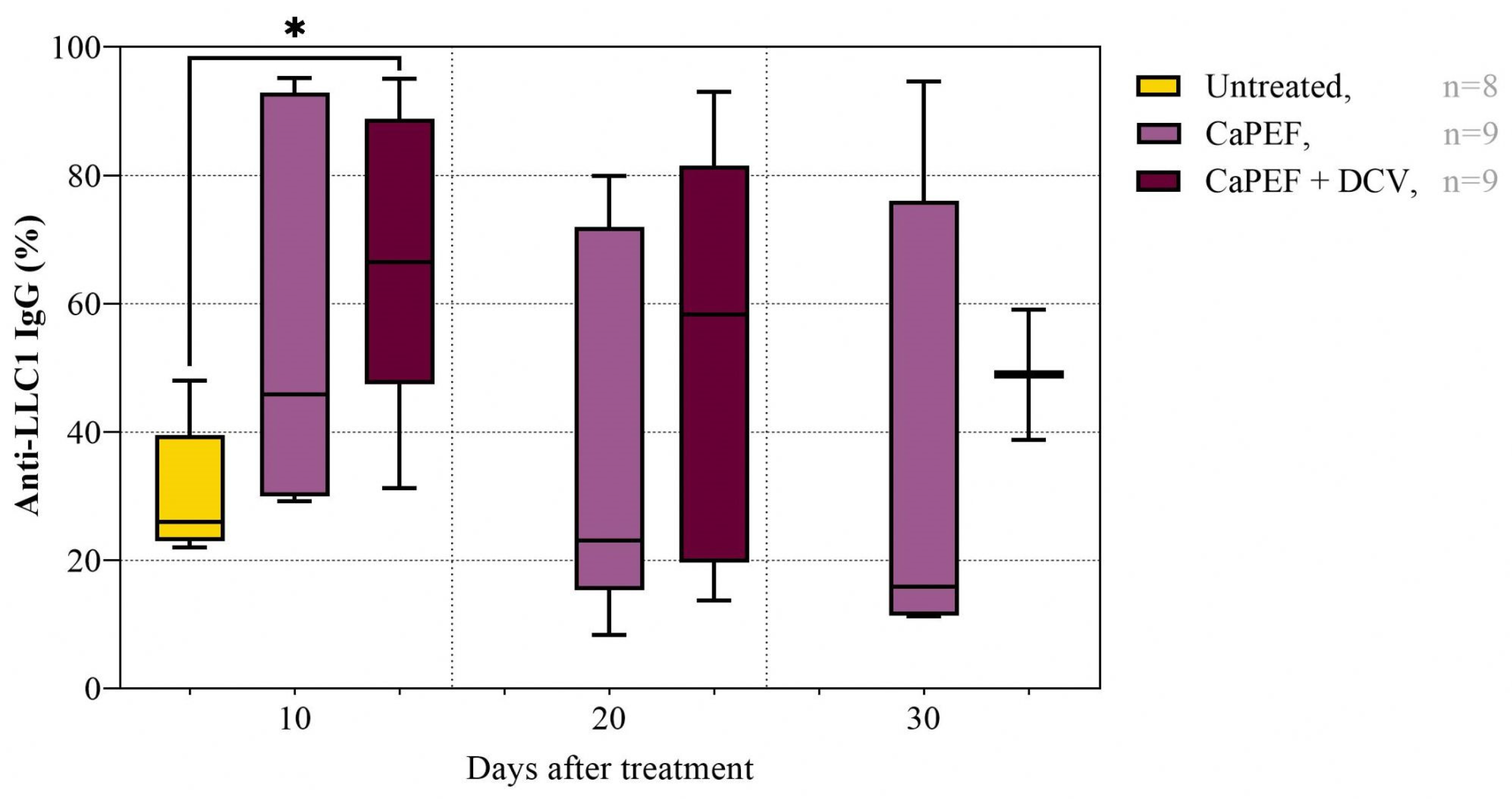
3.4. Antitumor Immune Response
3.5. Survival Rates of the Second Experiment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mir, L.M.; Banoun, H.; Paoletti, C. Introduction of definite amounts of nonpermeant molecules into living cells after electropermeabilization: Direct access to the cytosol. Exp. Cell Res. 1988, 175, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Jaroszeski, M.J.; Dang, V.; Pottinger, C.; Hickey, J.; Gilbert, R.; Heller, R. Toxicity of anticancer agents mediated by electroporation in vitro. Anticancer Drugs 2000, 11, 201–208. [Google Scholar] [CrossRef]
- Sersa, G.; Cemazar, M.; Miklavcic, D. Antitumor effectiveness of electrochemotherapy with cis-diamminedichloroplatinum(II) in mice. Cancer Res. 1995, 55, 3450–3455. [Google Scholar] [PubMed]
- Rols, M.-P.; Delteil, C.; Golzio, M.; Dumond, P.; Cros, S.; Teissie, J. In vivo electrically mediated protein and gene transfer in murine melanoma. Nat. Biotechnol. 1998, 16, 168–171. [Google Scholar] [CrossRef]
- Venslauskas, M.S.; Šatkauskas, S. Mechanisms of transfer of bioactive molecules through the cell membrane by electroporation. Eur. Biophys. J. 2015, 44, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Markelc, B.; Čemažar, M.; Serša, G. Effects of Reversible and Irreversible Electroporation on Endothelial Cells and Tissue Blood Flow. In Handbook of Electroporation; Miklavčič, D., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 607–620. [Google Scholar] [CrossRef]
- Rotunno, R.; Campana, L.G.; Quaglino, P.; de Terlizzi, F.; Kunte, C.; Odili, J.; Gehl, J.; Ribero, S.; Liew, S.; Marconato, R.; et al. Electrochemotherapy of unresectable cutaneous tumours with reduced dosages of intravenous bleomycin: Analysis of 57 patients from the International Network for Sharing Practices of Electrochemotherapy registry. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 1147–1154. [Google Scholar] [CrossRef]
- Groselj, A.; Bosnjak, M.; Strojan, P.; Krzan, M.; Cemazar, M.; Sersa, G. Efficiency of electrochemotherapy with reduced bleomycin dose in the treatment of nonmelanoma head and neck skin cancer: Preliminary results. Head Neck 2018, 40, 120–125. [Google Scholar] [CrossRef]
- Marty, M.; Sersa, G.; Garbay, J.R.; Gehl, J.; Collins, C.G.; Snoj, M.; Billard, V.; Geertsen, P.F.; Larkin, J.O.; Miklavcic, D.; et al. Electrochemotherapy—An easy, highly effective and safe treatment of cutaneous and subcutaneous metastases: Results of ESOPE (European Standard Operating Procedures of Electrochemotherapy) study. Eur. J. Cancer Suppl. 2006, 4, 3–13. [Google Scholar] [CrossRef]
- Gehl, J.; Serša, G. Electrochemotherapy and Its Clinical Applications. In Handbook of Electroporation; Miklavčič, D., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 1771–1786. [Google Scholar] [CrossRef]
- Di Monta, G.; Caracò, C.; Simeone, E.; Grimaldi, A.M.; Marone, U.; Di Marzo, M.; Vanella, V.; Festino, L.; Palla, M.; Mori, S.; et al. Electrochemotherapy efficacy evaluation for treatment of locally advanced stage III cutaneous squamous cell carcinoma: A 22-cases retrospective analysis. J. Transl. Med. 2017, 15, 82. [Google Scholar] [CrossRef]
- Mir, L.M.; Belehradek, M.; Domenge, C.; Orlowski, S.; Poddevin, B.; Belehradek, J., Jr.; Schwaab, G.; Luboinski, B.; Paoletti, C. Electrochemotherapy, a new antitumor treatment: First clinical trial. C. R. Acad. Sci. III 1991, 313, 613–618. [Google Scholar]
- Serša, G.; Štabuc, B.; Čemažar, M.; Jančar, B.; Miklavčič, D.; Rudolf, Z. Electrochemotherapy with cisplatin: Potentiation of local cisplatin antitumour effectiveness by application of electric pulses in cancer patients. Eur. J. Cancer 1998, 34, 1213–1218. [Google Scholar] [CrossRef] [PubMed]
- Cucu, C.I.; Giurcăneanu, C.; Popa, L.G.; Orzan, O.A.; Beiu, C.; Holban, A.M.; Grumezescu, A.M.; Matei, B.M.; Popescu, M.N.; Căruntu, C.; et al. Electrochemotherapy and Other Clinical Applications of Electroporation for the Targeted Therapy of Metastatic Melanoma. Materials 2021, 14, 3985. [Google Scholar] [CrossRef]
- Frandsen, S.K.; Gissel, H.; Hojman, P.; Tramm, T.; Eriksen, J.; Gehl, J. Direct Therapeutic Applications of Calcium Electroporation to Effectively Induce Tumor Necrosis. Cancer Res. 2012, 72, 1336–1341. [Google Scholar] [CrossRef]
- Frandsen, S.K.; Gehl, J.; Tramm, T.; Thoefner, M.S. Calcium Electroporation of Equine Sarcoids. Animals 2020, 10, 517. [Google Scholar] [CrossRef]
- Anastasova, R.; Fiorentzis, M.; Liu, H.; Dalbah, S.; Bechrakis, N.E.; Seitz, B.; Berchner-Pfannschmidt, U.; Tsimpaki, T. Electroporation with Calcium or Bleomycin: First Application in an In Vivo Uveal Melanoma Patient-Derived Xenograft Model. Pharmaceuticals 2024, 17, 905. [Google Scholar] [CrossRef] [PubMed]
- Łapińska, Z.; Szwedowicz, U.; Choromańska, A.; Saczko, J. Electroporation and Electrochemotherapy in Gynecological and Breast Cancer Treatment. Molecules 2022, 27, 2476. [Google Scholar] [CrossRef]
- Hansen, E.L.; Sozer, E.B.; Romeo, S.; Frandsen, S.K.; Vernier, P.T.; Gehl, J. Correction: Dose-Dependent ATP Depletion and Cancer Cell Death following Calcium Electroporation, Relative Effect of Calcium Concentration and Electric Field Strength. PLoS ONE 2015, 10, e0128034. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, M.M.; Tsimpaki, T.; Berchner-Pfannschmidt, U.; Bechrakis, N.E.; Seitz, B.; Fiorentzis, M. Calcium Electroporation Reduces Viability and Proliferation Capacity of Four Uveal Melanoma Cell Lines in 2D and 3D Cultures. Cancers 2022, 14, 2889. [Google Scholar] [CrossRef]
- Lisec, B.; Markelc, B.; Valentinuzzi, K.U.; Sersa, G.; Cemazar, M. The effectiveness of calcium electroporation combined with gene electrotransfer of a plasmid encoding IL-12 is tumor type-dependent. Front. Immunol. 2023, 14, 1189960. [Google Scholar] [CrossRef]
- Tremble, L.F.; Heffron, C.C.B.B.; Forde, P.F. The effect of calcium electroporation on viability, phenotype and function of melanoma conditioned macrophages. Sci. Rep. 2020, 10, 20645. [Google Scholar] [CrossRef]
- Frandsen, S.K.; Gissel, H.; Hojman, P.; Eriksen, J.; Gehl, J. Calcium electroporation in three cell lines: A comparison of bleomycin and calcium, calcium compounds, and pulsing conditions. Biochim. Biophys. Acta 2014, 1840, 1204–1208. [Google Scholar] [CrossRef]
- Zhivotovsky, B.; Orrenius, S. Calcium and cell death mechanisms: A perspective from the cell death community. Cell Calcium 2011, 50, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Nuccitelli, R.; Lui, K.; Kreis, M.; Athos, B.; Nuccitelli, P. Nanosecond pulsed electric field stimulation of reactive oxygen species in human pancreatic cancer cells is Ca2+-dependent. Biochem. Biophys. Res. Commun. 2013, 435, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Falk, H.; Forde, P.F.; Bay, M.L.; Mangalanathan, U.M.; Hojman, P.; Soden, D.M.; Gehl, J. Calcium electroporation induces tumor eradication, long-lasting immunity and cytokine responses in the CT26 colon cancer mouse model. OncoImmunology 2017, 6, e1301332. [Google Scholar] [CrossRef]
- Brock, R.M.; Beitel-White, N.; Davalos, R.V.; Allen, I.C. Starting a Fire Without Flame: The Induction of Cell Death and Inflammation in Electroporation-Based Tumor Ablation Strategies. Front. Oncol. 2020, 10, 1235. [Google Scholar] [CrossRef]
- Novickij, V.; Čėsna, R.; Perminaitė, E.; Zinkevičienė, A.; Characiejus, D.; Novickij, J.; Šatkauskas, S.; Ruzgys, P.; Girkontaitė, I. Antitumor Response and Immunomodulatory Effects of Sub-Microsecond Irreversible Electroporation and Its Combination with Calcium Electroporation. Cancers 2019, 11, 1763. [Google Scholar] [CrossRef]
- Falk, H.; Lambaa, S.; Johannesen, H.H.; Wooler, G.; Venzo, A.; Gehl, J. Electrochemotherapy and calcium electroporation inducing a systemic immune response with local and distant remission of tumors in a patient with malignant melanoma—A case report. Acta Oncol. 2017, 56, 1126–1131. [Google Scholar] [CrossRef]
- Calvet, C.Y.; Famin, D.; André, F.M.; Mir, L.M. Electrochemotherapy with bleomycin induces hallmarks of immunogenic cell death in murine colon cancer cells. OncoImmunology 2014, 3, e28131. [Google Scholar] [CrossRef]
- Falk, H.; Matthiessen, L.; Wooler, G.; Gehl, J. Calcium electroporation for treatment of cutaneous metastases; a randomized double-blinded phase II study, comparing the effect of calcium electroporation with electrochemotherapy. Acta Oncol. 2017, 57, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Plaschke, C.C.; Gehl, J.; Johannesen, H.; Fischer, B.M.; Kjaer, A.; Lomholt, A.F.; Wessel, I. Calcium electroporation for recurrent head and neck cancer: A clinical phase I study. Laryngoscope 2019, 4, 49–56. [Google Scholar] [CrossRef]
- Zhao, J.; Wen, X.; Tian, L.; Li, T.; Xu, C.; Wen, X.; Melancon, M.P.; Gupta, S.; Shen, B.; Peng, W.; et al. Irreversible electroporation reverses resistance to immune checkpoint blockade in pancreatic cancer. Nat. Commun. 2019, 10, 899. [Google Scholar] [CrossRef]
- Heppt, M.V.; Eigentler, T.K.; Kähler, K.C.; Herbst, R.A.; Göppner, D.; Gambichler, T.; Ulrich, J.; Dippel, E.; Loquai, C.; Schell, B.; et al. Immune checkpoint blockade with concurrent electrochemotherapy in advanced melanoma: A retrospective multicenter analysis. Cancer Immunol. Immunother. 2016, 65, 951–959. [Google Scholar] [CrossRef]
- Brizio, M.; Fava, P.; Astrua, C.; Cavaliere, G.; Savoia, P. Complete regression of melanoma skin metastases after electrochemotherapy plus ipilimumab treatment: An unusual clinical presentation. Eur. J. Dermatol. 2015, 25, 271–272. [Google Scholar] [CrossRef] [PubMed]
- Mozzillo, N.; Simeone, E.; Benedetto, L.; Curvietto, M.; Giannarelli, D.; Gentilcore, G.; Camerlingo, R.; Capone, M.; Madonna, G.; Festino, L.; et al. Assessing a novel immuno-oncology-based combination therapy: Ipilimumab plus electrochemotherapy. OncoImmunology 2015, 4, e1008842. [Google Scholar] [CrossRef]
- Burbach, B.J.; O’fLanagan, S.D.; Shao, Q.; Young, K.M.; Slaughter, J.R.; Rollins, M.R.; Street, T.J.L.; Granger, V.E.; Beura, L.K.; Azarin, S.M.; et al. Irreversible electroporation augments checkpoint immunotherapy in prostate cancer and promotes tumor antigen-specific tissue-resident memory CD8+ T cells. Nat. Commun. 2021, 12, 3862. [Google Scholar] [CrossRef]
- Andersen, M.H.; Gehl, J.; Reker, S.; Pedersen, L.Ø.; Becker, J.C.; Geertsen, P.; Straten, P.T. Dynamic changes of specific T cell responses to melanoma correlate with IL-2 administration. Semin. Cancer Biol. 2003, 13, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Mir, L.M.; Roth, C.; Orlowski, S.; Quintin-Colonna, F.; Fradelizi, D.; Belehradek, J.J.; Kourilsky, P. Systemic Antitumor Effects of Electrochemotherapy Combined with Histoincompatible Cells Secreting Interleukin-2. J. Immunother. 1995, 17, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Cemazar, M.; Avgustin, J.A.; Pavlin, D.; Sersa, G.; Poli, A.; Levacic, A.K.; Tesic, N.; Tratar, U.L.; Rak, M.; Tozon, N. Efficacy and safety of electrochemotherapy combined with peritumoral IL-12 gene electrotransfer of canine mast cell tumours. Vet. Comp. Oncol. 2017, 15, 641–654. [Google Scholar] [CrossRef]
- Heller, L.; Pottinger, C.; Jaroszeski, M.J.; Gilbert, R.; Heller, R. In vivo electroporation of plasmids encoding GM-CSF or interleukin-2 into existing B16 melanomas combined with electrochemotherapy induces long-term antitumour immunity. Melanoma Res. 2000, 10, 577–583. [Google Scholar] [CrossRef]
- Go, E.-J.; Yang, H.; Chon, H.J.; Yang, D.; Ryu, W.; Kim, D.-H.; Han, D.K.; Kim, C.; Park, W. Combination of Irreversible Electroporation and STING Agonist for Effective Cancer Immunotherapy. Cancers 2020, 12, 3123. [Google Scholar] [CrossRef]
- Filin, I.Y.; Kitaeva, K.V.; Rutland, C.S.; Rizvanov, A.A.; Solovyeva, V.V. Recent Advances in Experimental Dendritic Cell Vaccines for Cancer. Front. Oncol. 2021, 11, 730824. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Sun, H.; Cao, W.; Song, Y.; Jiang, Z. Research progress on dendritic cell vaccines in cancer immunotherapy. Exp. Hematol. Oncol. 2022, 11, 3. [Google Scholar] [CrossRef]
- Najafi, S.; Mortezaee, K. Advances in dendritic cell vaccination therapy of cancer. Biomed. Pharmacother. 2023, 164, 114954. [Google Scholar] [CrossRef]
- Banchereau, J.; Briere, F.; Caux, C.; Davoust, J.; Lebecque, S.; Liu, Y.J.; Pulendran, B.; Palucka, K. Immunobiology of Dendritic Cells. Annu. Rev. Immunol. 2000, 18, 767–811. [Google Scholar] [CrossRef]
- Shurin, M.R. Dendritic cells presenting tumor antigen. Cancer Immunol. Immunother. 1996, 43, 158–164. [Google Scholar] [CrossRef]
- Banchereau, J.; Steinman, R.M. Dendritic cells and the control of immunity. Nature 1998, 392, 245–252. [Google Scholar] [CrossRef]
- Fang, P.; Li, X.; Dai, J.; Cole, L.; Camacho, J.A.; Zhang, Y.; Ji, Y.; Wang, J.; Yang, X.-F.; Wang, H. Immune cell subset differentiation and tissue inflammation. J. Hematol. Oncol. 2018, 11, 97. [Google Scholar] [CrossRef]
- Sprooten, J.; Ceusters, J.; Coosemans, A.; Agostinis, P.; De Vleeschouwer, S.; Zitvogel, L.; Kroemer, G.; Galluzzi, L.; Garg, A.D. Trial watch: Dendritic cell vaccination for cancer immunotherapy. OncoImmunology 2019, 8, e1638212. [Google Scholar] [CrossRef]
- Chen, M.Y.; Zhang, F.; Goedegebuure, S.P.; Gillanders, W.E. Dendritic cell subsets and implications for cancer immunotherapy. Front. Immunol. 2024, 15, 1393451. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.L.-L.; Kandalaft, L.E. In vivo cancer vaccination: Which dendritic cells to target and how? Cancer Treat. Rev. 2018, 71, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Ridolfi, L.; Gurrieri, L.; Riva, N.; Bulgarelli, J.; De Rosa, F.; Guidoboni, M.; Fausti, V.; Ranallo, N.; Calpona, S.; Tazzari, M.; et al. First step results from a phase II study of a dendritic cell vaccine in glioblastoma patients (CombiG-vax). Front. Immunol. 2024, 15, 1404861. [Google Scholar] [CrossRef] [PubMed]
- Nimanong, S.; Ostroumov, D.; Wingerath, J.; Knocke, S.; Woller, N.; Gürlevik, E.; Falk, C.S.; Manns, M.P.; Kühnel, F.; Wirth, T.C. CD40 Signaling Drives Potent Cellular Immune Responses in Heterologous Cancer Vaccinations. Cancer Res. 2017, 77, 1918–1926. [Google Scholar] [CrossRef] [PubMed]
- Moreno Ayala, M.A.; Gottardo, M.F.; Imsen, M.; Asad, A.S.; Bal de Kier Joffé, E.; Casares, N.; Lasarte, J.J.; Seilicovich, A.; Candolfi, M. Therapeutic blockade of Foxp3 in experimental breast cancer models. Breast Cancer Res. Treat. 2017, 166, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Moreno Ayala, M.A.; Gottardo, M.F.; Gori, M.S.; Nicola Candia, A.J.; Caruso, C.; De Laurentiis, A.; Imsen, M.; Klein, S.; Bal de Kier Joffé, E.; Salamone, G.; et al. Dual activation of Toll-like receptors 7 and 9 impairs the efficacy of antitumor vaccines in murine models of metastatic breast cancer. J. Cancer Res. Clin. Oncol. 2017, 143, 1713–1732. [Google Scholar] [CrossRef]
- Liu, H.; Chen, L.; Liu, J.; Meng, H.; Zhang, R.; Ma, L.; Wu, L.; Yu, S.; Shi, F.; Li, Y.; et al. Co-delivery of tumor-derived exosomes with alpha-galactosylceramide on dendritic cell-based immunotherapy for glioblastoma. Cancer Lett. 2017, 411, 182–190. [Google Scholar] [CrossRef]
- Escribà-Garcia, L.; Alvarez-Fernández, C.; Tellez-Gabriel, M.; Sierra, J.; Briones, J. Dendritic cells combined with tumor cells and α-galactosylceramide induce a potent, therapeutic and NK-cell dependent antitumor immunity in B cell lymphoma. J. Transl. Med. 2017, 15, 115. [Google Scholar] [CrossRef]
- Vo, M.-C.; Nguyen-Pham, T.-N.; Lee, H.-J.; Lakshmi, T.J.; Yang, S.; Jung, S.-H.; Kim, H.-J.; Lee, J.-J. Combination therapy with dendritic cells and lenalidomide is an effective approach to enhance antitumor immunity in a mouse colon cancer model. Oncotarget 2017, 8, 27252–27262. [Google Scholar] [CrossRef]
- Dammeijer, F.; Lievense, L.A.; Kaijen-Lambers, M.E.; van Nimwegen, M.; Bezemer, K.; Hegmans, J.P.; van Hall, T.; Hendriks, R.W.; Aerts, J.G. Depletion of Tumor-Associated Macrophages with a CSF-1R Kinase Inhibitor Enhances Antitumor Immunity and Survival Induced by DC Immunotherapy. Cancer Immunol. Res. 2017, 5, 535–546. [Google Scholar] [CrossRef]
- Stefanski, H.E.; Jonart, L.; Goren, E.; Mulé, J.J.; Blazar, B.R.; Labrecque, N. A novel approach to improve immune effector responses post transplant by restoration of CCL21 expression. PLoS ONE 2018, 13, e0193461. [Google Scholar] [CrossRef]
- Van Woensel, M.; Mathivet, T.; Wauthoz, N.; Rosière, R.; Garg, A.D.; Agostinis, P.; Mathieu, V.; Kiss, R.; Lefranc, F.; Boon, L. Sensitization of glioblastoma tumor micro-environment to chemo- and immunotherapy by Galectin-1 intranasal knock-down strategy. Sci. Rep. 2017, 7, 1217. [Google Scholar] [CrossRef]
- Ahlmann, M.; Hempel, G. The effect of cyclophosphamide on the immune system: Implications for clinical cancer therapy. Cancer Chemother. Pharmacol. 2016, 78, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Emadi, A.; Jones, R.J.; Brodsky, R.A. Cyclophosphamide and cancer: Golden anniversary. Nat. Rev. Clin. Oncol. 2009, 6, 638–647. [Google Scholar] [CrossRef]
- Gernaat, S.A.M.; von Stedingk, H.; Hassan, M.; Nilsson, H.P.; Rodriguez-Wallberg, K.A.; Hedayati, E.; Rydberg, P. Cyclophosphamide exposure assessed with the biomarker phosphoramide mustard-hemoglobin in breast cancer patients: The TailorDose I study. Sci. Rep. 2021, 11, 2707. [Google Scholar] [CrossRef]
- Jiang, M.; Wang, W.; Zhang, J.; Wang, C.; Bi, Y.; Li, P.; Yang, S.; Li, J.; Xu, Y.-T.; Wang, T. Protective Effects and Possible Mechanisms of Actions of Bushen Cuyun Recipe on Diminished Ovarian Reserve Induced by Cyclophosphamide in Rats. Front. Pharmacol. 2020, 11, 546. [Google Scholar] [CrossRef]
- Scurr, M.; Pembroke, T.; Bloom, A.; Roberts, D.; Thomson, A.; Smart, K.; Bridgeman, H.; Adams, R.; Brewster, A.; Jones, R.; et al. Low-Dose Cyclophosphamide Induces Antitumor T-Cell Responses, which Associate with Survival in Metastatic Colorectal Cancer. Clin. Cancer Res. 2017, 23, 6771–6780. [Google Scholar] [CrossRef]
- Huijts, C.M.; Werter, I.M.; Lougheed, S.M.; Goedegebuure, R.S.; van Herpen, C.M.; Hamberg, P.; Tascilar, M.; Haanen, J.B.; Verheul, H.M.; de Gruijl, T.D.; et al. Phase 1 study of everolimus and low-dose oral cyclophosphamide in patients with metastatic renal cell carcinoma. Cancer Immunol. Immunother. 2019, 68, 319–329. [Google Scholar] [CrossRef]
- Hughes, E.; Scurr, M.; Campbell, E.; Jones, E.; Godkin, A.; Gallimore, A. T-cell modulation by cyclophosphamide for tumour therapy. Immunology 2018, 154, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Dimeloe, S.; Frick, C.; Fischer, M.; Gubser, P.M.; Razik, L.; Bantug, G.R.; Ravon, M.; Langenkamp, A.; Hess, C. Human regulatory T cells lack the cyclophosphamide-extruding transporter ABCB1 and are more susceptible to cyclophosphamide-induced apoptosis. Eur. J. Immunol. 2014, 44, 3614–3620. [Google Scholar] [CrossRef] [PubMed]
- Gershan, J.A.; Barr, K.M.; Weber, J.J.; Jing, W.; Johnson, B.D. Immune modulating effects of cyclophosphamide and treatment with tumor lysate/CpG synergize to eliminate murine neuroblastoma. J. Immunother. Cancer 2015, 3, 24. [Google Scholar] [CrossRef]
- Webb, E.R.; Moreno-Vicente, J.; Easton, A.; Lanati, S.; Taylor, M.; James, S.; Williams, E.L.; English, V.; Penfold, C.; Beers, S.A.; et al. Cyclophosphamide depletes tumor infiltrating T regulatory cells and combined with anti-PD-1 therapy improves survival in murine neuroblastoma. iScience 2022, 25, 104995. [Google Scholar] [CrossRef]
- Radojcic, V.; Bezak, K.B.; Skarica, M.; Pletneva, M.A.; Yoshimura, K.; Schulick, R.D.; Luznik, L. Cyclophosphamide resets dendritic cell homeostasis and enhances antitumor immunity through effects that extend beyond regulatory T cell elimination. Cancer Immunol. Immunother. 2010, 59, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Bracci, L.; Moschella, F.; Sestili, P.; La Sorsa, V.; Valentini, M.; Canini, I.; Baccarini, S.; Maccari, S.; Ramoni, C.; Belardelli, F.; et al. Cyclophosphamide Enhances the Antitumor Efficacy of Adoptively Transferred Immune Cells through the Induction of Cytokine Expression, B-Cell and T-Cell Homeostatic Proliferation, and Specific Tumor Infiltration. Clin. Cancer Res. 2007, 13, 644–653. [Google Scholar] [CrossRef]
- Richtig, E.; Langmann, G.; Müllner, K.; Richtig, G.; Smolle, J. Calculated tumour volume as a prognostic parameter for survival in choroidal melanomas. Eye 2004, 18, 619–623. [Google Scholar] [CrossRef] [PubMed]
- Tomayko, M.M.; Reynolds, C.P. Determination of subcutaneous tumor size in athymic (nude) mice. Cancer Chemother. Pharmacol. 1989, 24, 148–154. [Google Scholar] [CrossRef]
- Lutz, M.B.; Kukutsch, N.; Ogilvie, A.L.; Rößner, S.; Koch, F.; Romani, N.; Schuler, G. An advanced culture method for generating large quantities of highly pure dendritic cells from mouse bone marrow. J. Immunol. Methods 1999, 223, 77–92. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Jordan, M.; Waxman, D.J. Metronomic cyclophosphamide activation of anti-tumor immunity: Tumor model, mouse host, and drug schedule dependence of gene responses and their upstream regulators. BMC Cancer 2016, 16, 623. [Google Scholar] [CrossRef]
- Balevičiūtė, A.; Radzevičiūtė, E.; Želvys, A.; Malyško-Ptašinskė, V.; Novickij, J.; Zinkevičienė, A.; Kašėta, V.; Novickij, V.; Girkontaitė, I. High-Frequency Nanosecond Bleomycin Electrochemotherapy and its Effects on Changes in the Immune System and Survival. Cancers 2022, 14, 6254. [Google Scholar] [CrossRef]
- Jiang, Y.; Chen, M.; Nie, H.; Yuan, Y. PD-1 and PD-L1 in cancer immunotherapy: Clinical implications and future considerations. Hum. Vaccines Immunother. 2019, 15, 1111–1122. [Google Scholar] [CrossRef]
- Chen, R.-Y.; Zhu, Y.; Shen, Y.-Y.; Xu, Q.-Y.; Tang, H.-Y.; Cui, N.-X.; Jiang, L.; Dai, X.-M.; Chen, W.-Q.; Lin, Q.; et al. The role of PD-1 signaling in health and immune-related diseases. Front. Immunol. 2023, 14, 1163633. [Google Scholar] [CrossRef]
- Kumar, A.; Khani, A.T.; Ortiz, A.S.; Swaminathan, S. GM-CSF: A Double-Edged Sword in Cancer Immunotherapy. Front. Immunol. 2022, 13, 901277. [Google Scholar] [CrossRef]
- Bassez, A.; Vos, H.; Van Dyck, L.; Floris, G.; Arijs, I.; Desmedt, C.; Boeckx, B.; Bempt, M.V.; Nevelsteen, I.; Lambein, K.; et al. A single-cell map of intratumoral changes during anti-PD1 treatment of patients with breast cancer. Nat. Med. 2021, 27, 820–832. [Google Scholar] [CrossRef]
- Lutz, M.B.; Ali, S.; Audiger, C.; Autenrieth, S.E.; Berod, L.; Bigley, V.; Cyran, L.; Dalod, M.; Dörrie, J.; Dudziak, D.; et al. Guidelines for mouse and human DC generation. Eur. J. Immunol. 2023, 53, e2249816. [Google Scholar] [CrossRef]
- Helft, J.; Böttcher, J.; Chakravarty, P.; Zelenay, S.; Huotari, J.; Schraml, B.U.; Goubau, D.; Sousa, C.R.E. GM-CSF Mouse Bone Marrow Cultures Comprise a Heterogeneous Population of CD11c+MHCII+ Macrophages and Dendritic Cells. Immunity 2015, 42, 1197–1211. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Sprent, J. GM-CSF Culture Revisited: Preparation of Bulk Populations of Highly Pure Dendritic Cells from Mouse Bone Marrow. J. Immunol. 2018, 201, 3129–3139. [Google Scholar] [CrossRef]
- Peng, Q.; Qiu, X.; Zhang, Z.; Zhang, S.; Zhang, Y.; Liang, Y.; Guo, J.; Peng, H.; Chen, M.; Fu, Y.-X.; et al. PD-L1 on dendritic cells attenuates T cell activation and regulates response to immune checkpoint blockade. Nat. Commun. 2020, 11, 4835. [Google Scholar] [CrossRef] [PubMed]
- Mayoux, M.; Roller, A.; Pulko, V.; Sammicheli, S.; Chen, S.; Sum, E.; Jost, C.; Fransen, M.F.; Buser, R.B.; Kowanetz, M.; et al. Dendritic cells dictate responses to PD-L1 blockade cancer immunotherapy. Sci. Transl. Med. 2020, 12, eaav7431. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.A.; Wu, D.-C.; Cheung, J.; Navarro, A.; Xiong, H.; Cubas, R.; Totpal, K.; Chiu, H.; Wu, Y.; Comps-Agrar, L.; et al. PD-L1 expression by dendritic cells is a key regulator of T-cell immunity in cancer. Nat. Cancer 2020, 1, 681–691. [Google Scholar] [CrossRef]
- Cohen, S.; Haimovich, J.; Hollander, N. Dendritic Cell-Based Therapeutic Vaccination against Myeloma: Vaccine Formulation Determines Efficacy against Light Chain Myeloma. J. Immunol. 2009, 182, 1667–1673. [Google Scholar] [CrossRef]
- Stevens, D.; Ingels, J.; Van Lint, S.; Vandekerckhove, B.; Vermaelen, K. Dendritic Cell-Based Immunotherapy in Lung Cancer. Front. Immunol. 2021, 11, 620374. [Google Scholar] [CrossRef]
- Verheye, E.; Melgar, J.B.; Deschoemaeker, S.; Raes, G.; Maes, A.; De Bruyne, E.; Menu, E.; Vanderkerken, K.; Laoui, D.; De Veirman, K. Dendritic Cell-Based Immunotherapy in Multiple Myeloma: Challenges, Opportunities, and Future Directions. Int. J. Mol. Sci. 2022, 23, 904. [Google Scholar] [CrossRef]
- Hoang, M.-D.; Jung, S.-H.; Lee, H.-J.; Lee, Y.-K.; Nguyen-Pham, T.-N.; Choi, N.-R.; Vo, M.-C.; Lee, S.-S.; Ahn, J.-S.; Yang, D.-H.; et al. Dendritic Cell-Based Cancer Immunotherapy against Multiple Myeloma: From Bench to Clinic. Chonnam. Med. J. 2015, 51, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Eresen, A.; Shangguan, J.; Ma, Q.; Zhang, Z.; Yaghmai, V. Effect of route of administration on the efficacy of dendritic cell vaccine in PDAC mice. Am. J. Cancer Res. 2020, 10, 3911. [Google Scholar] [PubMed]
- Lesterhuis, W.J.; de Vries, I.J.M.; Schreibelt, G.; Lambeck, A.J.A.; Aarntzen, E.H.J.G.; Jacobs, J.F.M.; Scharenborg, N.M.; van de Rakt, M.W.M.M.; de Boer, A.J.; Croockewit, S.; et al. Route of Administration Modulates the Induction of Dendritic Cell Vaccine–Induced Antigen-Specific T Cells in Advanced Melanoma Patients. Clin. Cancer Res. 2011, 17, 5725–5735. [Google Scholar] [CrossRef] [PubMed]
- Hintzen, R.Q.; de Jong, R.; Lens, S.M.; Brouwer, M.; Baars, P.; van Lier, R.A. Regulation of CD27 expression on subsets of mature T-lymphocytes. J. Immunol. 1993, 151, 2426–2435. [Google Scholar] [CrossRef]
- Hendriks, J.; Gravestein, L.A.; Tesselaar, K.; van Lier, R.A.W.; Schumacher, T.N.M.; Borst, J. CD27 is required for generation and long-term maintenance of T cell immunity. Nat. Immunol. 2000, 1, 433–440. [Google Scholar] [CrossRef]
- Bozdogan, G.; Dogu, F.; Güloglu, D.; Yuksek, M.; Aytekin, C.; İkinciogullari, A. CD27 expression on lymphocyte and sCD27 levels in children with asthma. Allergol. Immunopathol. 2010, 38, 327–332. [Google Scholar] [CrossRef]
- Li, O.; Zheng, P.; Liu, Y. CD24 Expression on T Cells Is Required for Optimal T Cell Proliferation in Lymphopenic Host. J. Exp. Med. 2004, 200, 1083–1089. [Google Scholar] [CrossRef]
- Liu, Y.; Zheng, P. CD24: A genetic checkpoint in T cell homeostasis and autoimmune diseases. Trends Immunol. 2007, 28, 315–320. [Google Scholar] [CrossRef]
- Kim, I.K.; Chung, Y.; Kang, C.Y. GITR drives TH9-mediated antitumor immunity. Oncoimmunology 2015, 5, e1122862. [Google Scholar] [CrossRef]
- Dupre’, S.A.; Hunter, K.W. Murine mammary carcinoma 4T1 induces a leukemoid reaction with splenomegaly: Association with tumor-derived growth factors. Exp. Mol. Pathol. 2007, 82, 12–24. [Google Scholar] [CrossRef]
- Liang, S.C.; Moskalenko, M.; Van Roey, M.; Jooss, K. Depletion of regulatory T cells by targeting folate receptor 4 enhances the potency of a GM-CSF-secreting tumor cell immunotherapy. Clin. Immunol. 2013, 148, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Hirota, K.; Nagahama, K.; Ohkawa, K.; Takahashi, T.; Nomura, T.; Sakaguchi, S. Control of Immune Responses by Antigen-Specific Regulatory T Cells Expressing the Folate Receptor. Immunity 2007, 27, 145–159. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhao, Q.; Cao, M.; Li, X.; Chen, X.; He, M.; Liu, Y.; Zhao, J.; Shi, Z. Folate Receptor 4-Expressing T cell Is Associated with Disease-Free Survival in Patients with Esophageal Squamous Cell Carcinoma. Dis. Markers 2022, 2022, 4351949. [Google Scholar] [CrossRef] [PubMed]
- Rush, C.M.; Millington, O.R.; Hutchison, S.; Bryson, K.; Brewer, J.M.; Garside, P. Characterization of CD4+ T-cell–dendritic cell interactions during secondary antigen exposure in tolerance and priming. Immunology 2009, 128, 463. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radzevičiūtė-Valčiukė, E.; Balevičiūtė, A.; Želvys, A.; Suveizdė, K.; Zinkevičienė, A.; Kašėta, V.; Malyško-Ptašinskė, V.; Dobrovolskienė, N.; Pašukonienė, V.; Novickij, J.; et al. Calcium Electrochemotherapy and Challenges in Combined Treatment with Dendritic Cell Vaccination. Pharmaceutics 2025, 17, 804. https://doi.org/10.3390/pharmaceutics17070804
Radzevičiūtė-Valčiukė E, Balevičiūtė A, Želvys A, Suveizdė K, Zinkevičienė A, Kašėta V, Malyško-Ptašinskė V, Dobrovolskienė N, Pašukonienė V, Novickij J, et al. Calcium Electrochemotherapy and Challenges in Combined Treatment with Dendritic Cell Vaccination. Pharmaceutics. 2025; 17(7):804. https://doi.org/10.3390/pharmaceutics17070804
Chicago/Turabian StyleRadzevičiūtė-Valčiukė, Eivina, Austėja Balevičiūtė, Augustinas Želvys, Karolina Suveizdė, Auksė Zinkevičienė, Vytautas Kašėta, Veronika Malyško-Ptašinskė, Neringa Dobrovolskienė, Vita Pašukonienė, Jurij Novickij, and et al. 2025. "Calcium Electrochemotherapy and Challenges in Combined Treatment with Dendritic Cell Vaccination" Pharmaceutics 17, no. 7: 804. https://doi.org/10.3390/pharmaceutics17070804
APA StyleRadzevičiūtė-Valčiukė, E., Balevičiūtė, A., Želvys, A., Suveizdė, K., Zinkevičienė, A., Kašėta, V., Malyško-Ptašinskė, V., Dobrovolskienė, N., Pašukonienė, V., Novickij, J., Girkontaitė, I., & Novickij, V. (2025). Calcium Electrochemotherapy and Challenges in Combined Treatment with Dendritic Cell Vaccination. Pharmaceutics, 17(7), 804. https://doi.org/10.3390/pharmaceutics17070804








