Combating Acute Myeloid Leukemia via Sphingosine Kinase 1 Inhibitor-Nanomedicine Combination Therapy with Cytarabine or Venetoclax
Abstract
1. Introduction
2. Materials and Methods
3. Results
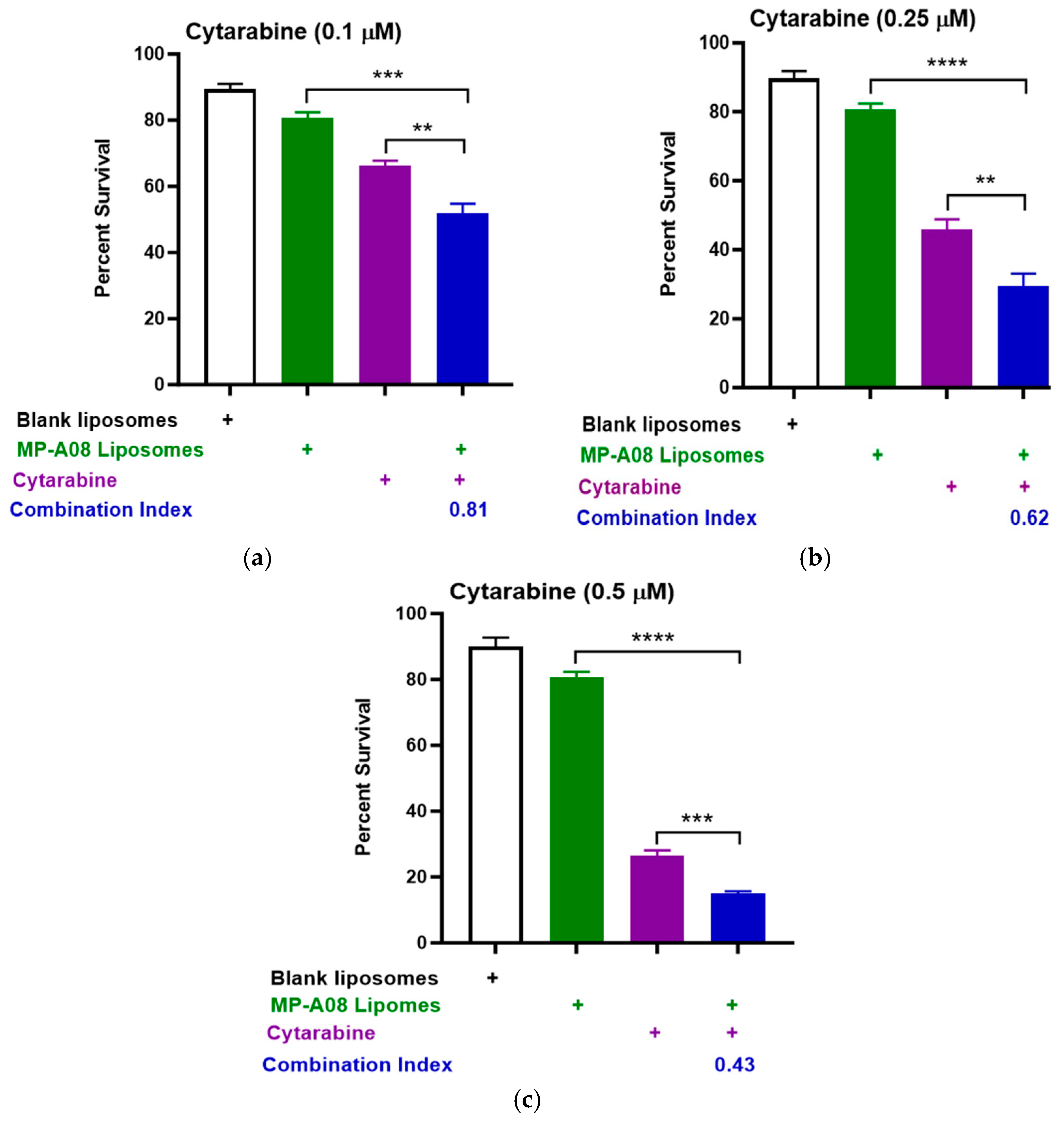
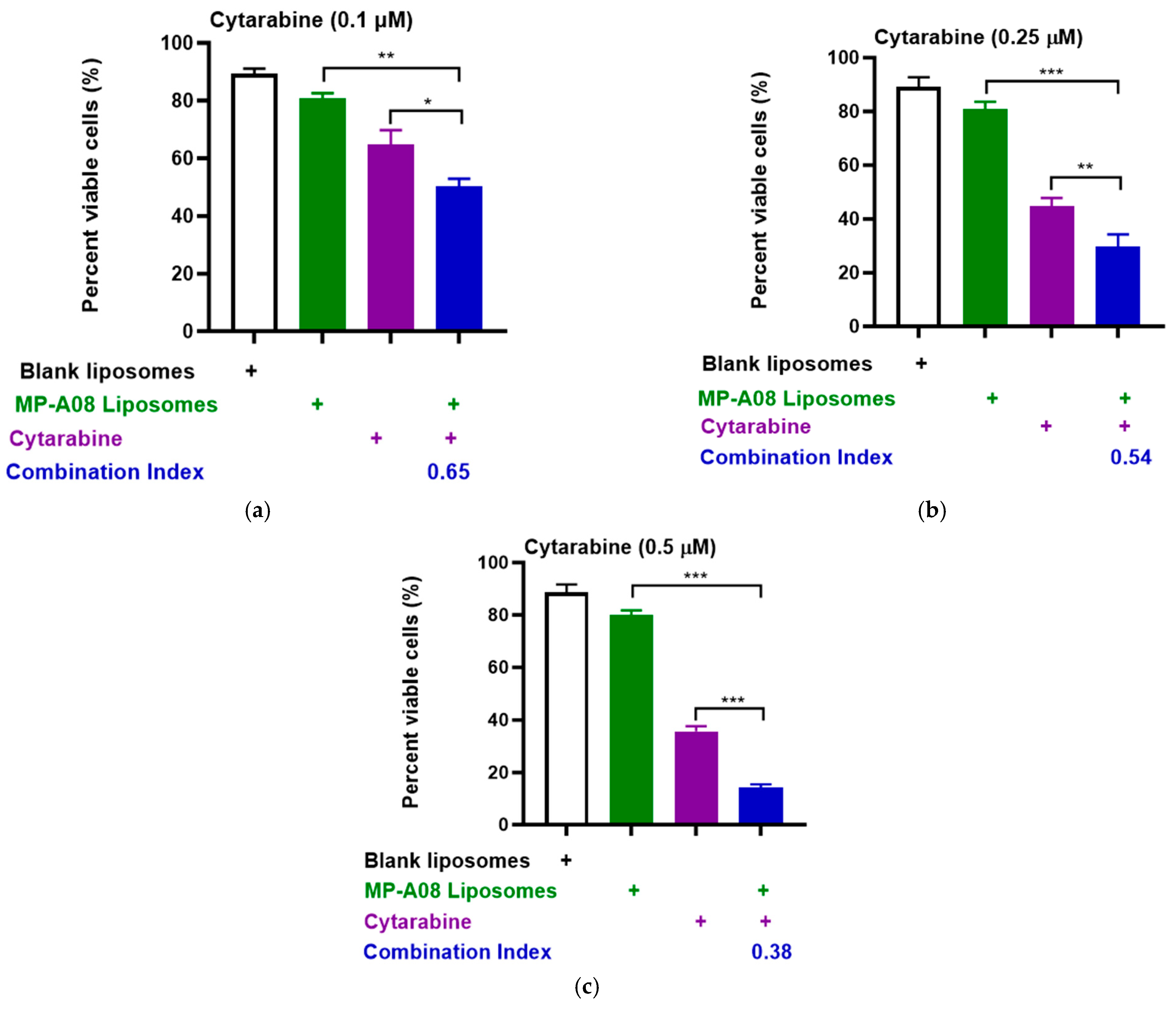
3.1. MP-A08-Loaded Liposomes Combined with Cytarabine Provide a Synergistic Effect against AML Cells
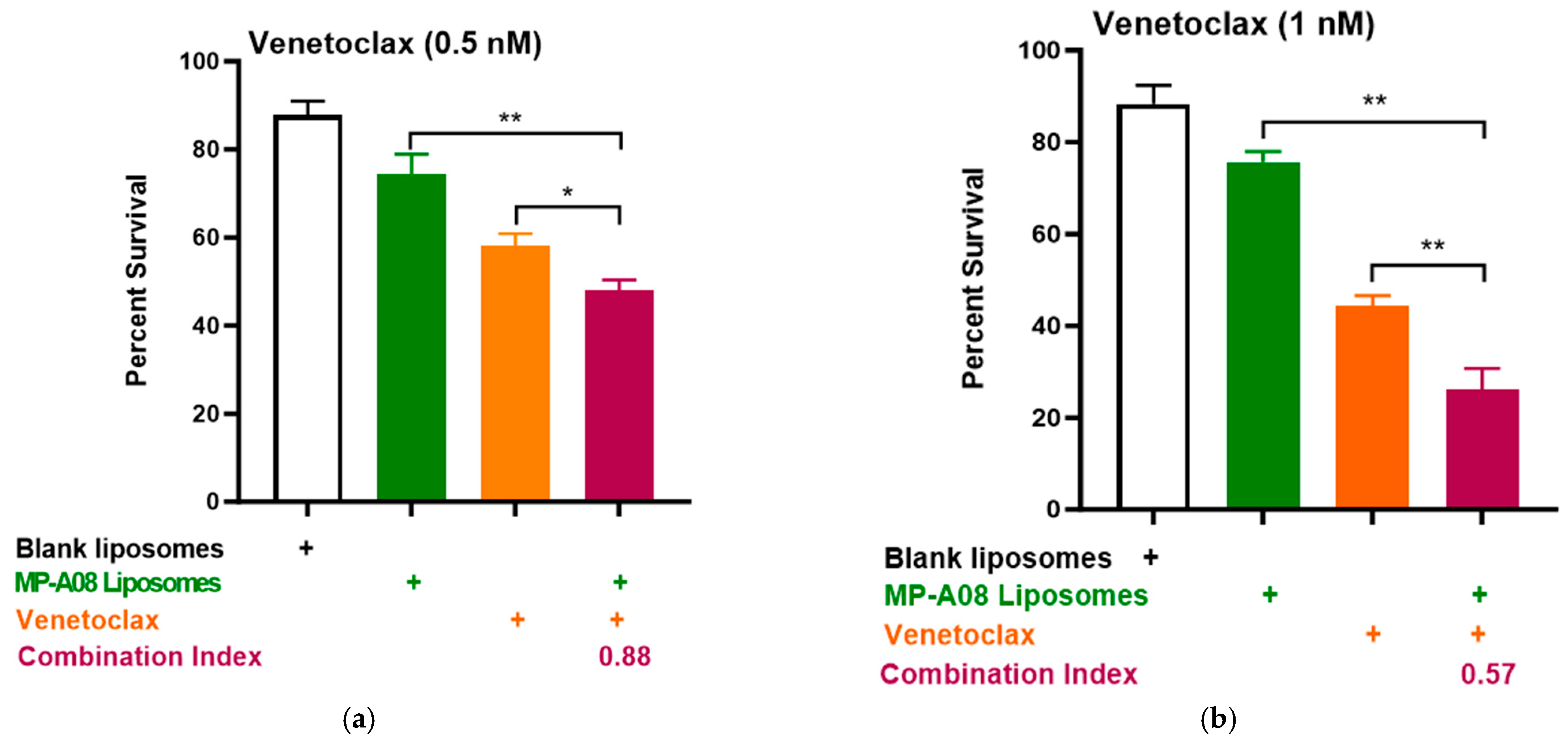
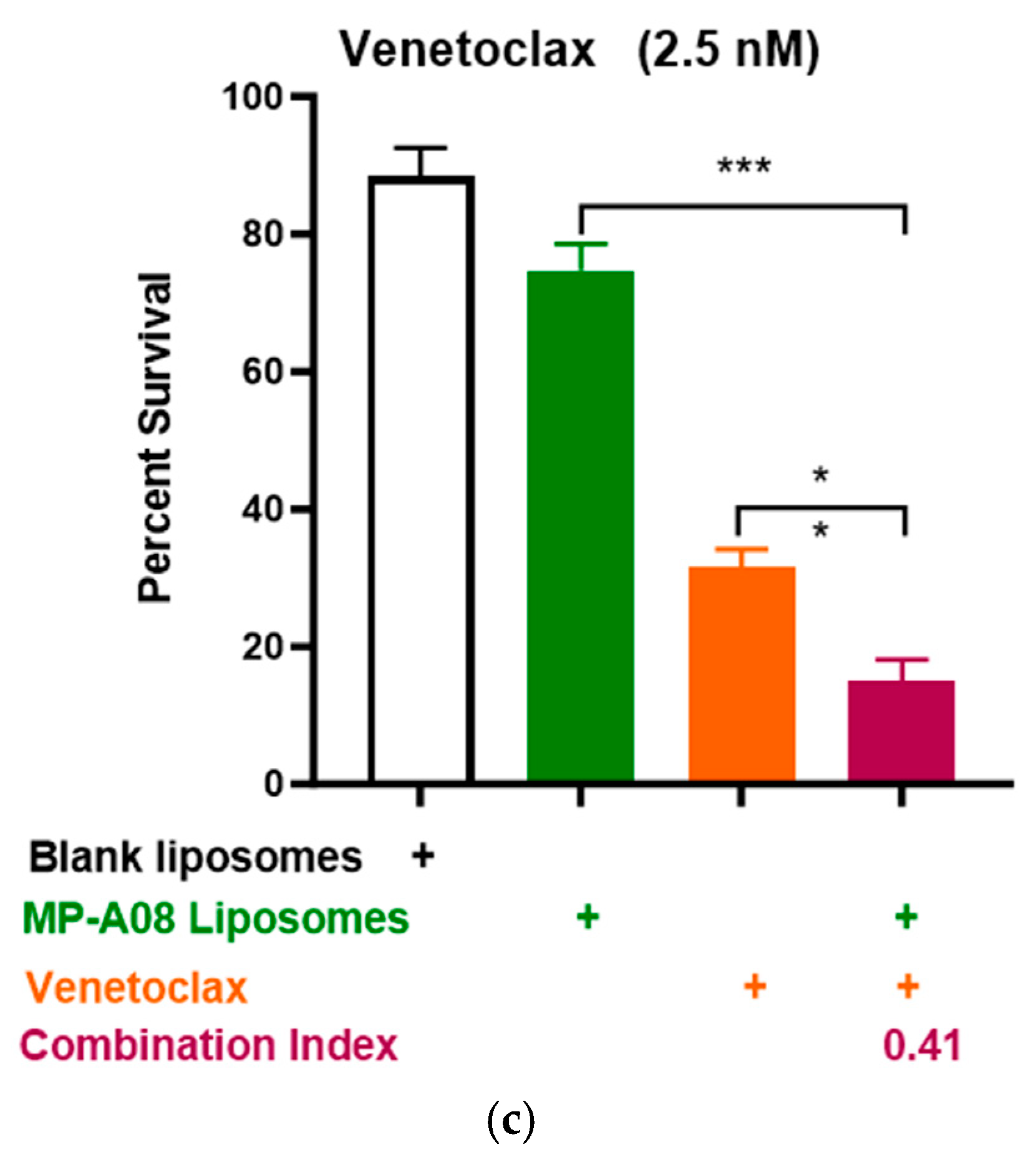
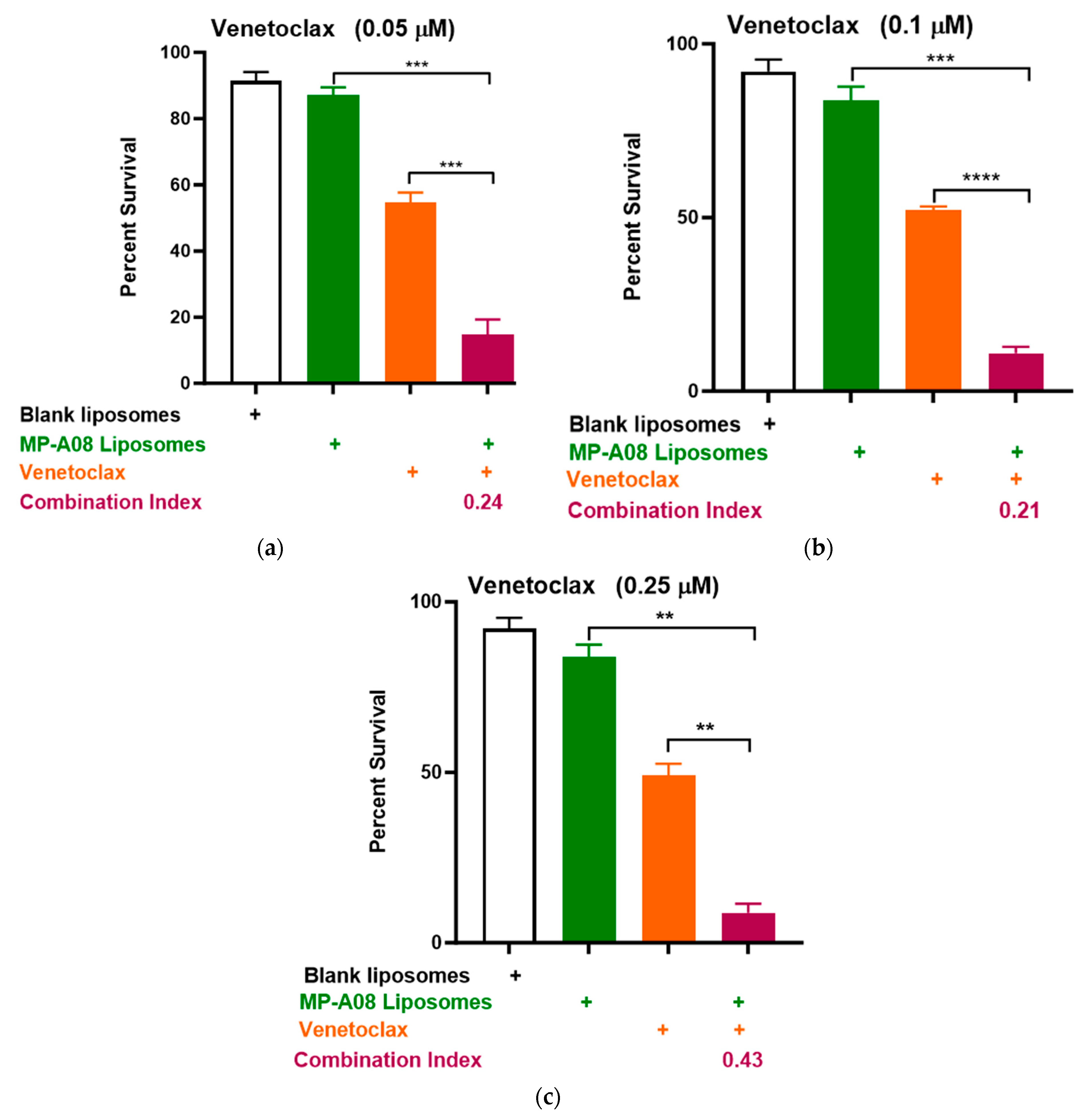
3.2. MP-A08-Loaded Liposomes Potentiated the Anti-Leukemic Activity of Venetoclax in AML Cells
3.3. MP-A08-Loaded Liposomes Overcome Venetoclax Resistance in Human AML Cells
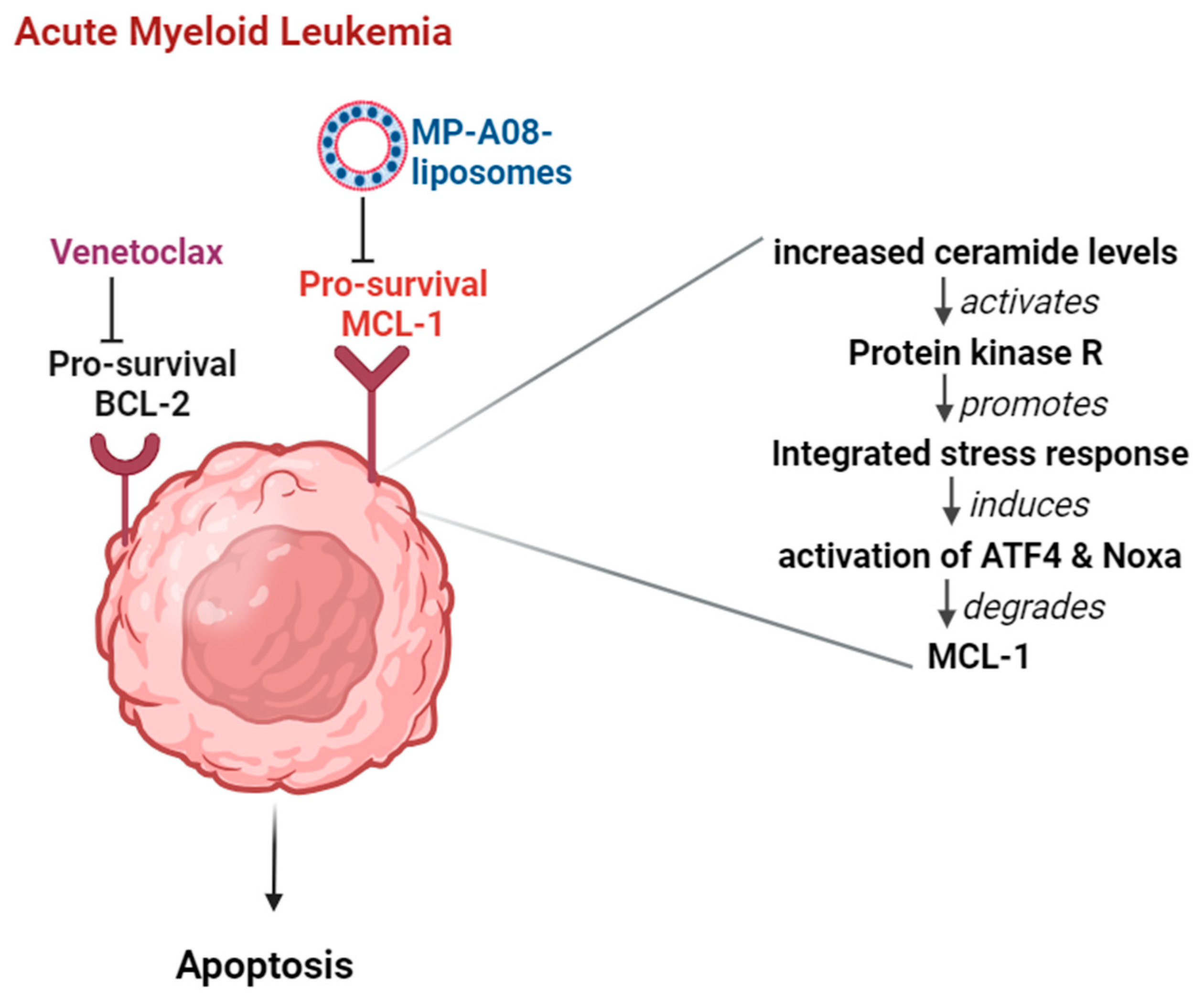
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Choi, J.H.; Bogenberger, J.M.; Tibes, R. Targeting Apoptosis in Acute Myeloid Leukemia: Current Status and Future Directions of BCL-2 Inhibition with Venetoclax and Beyond. Target. Oncol. 2020, 15, 147–162. [Google Scholar] [CrossRef]
- Chen, K.T.J.; Gilabert-Oriol, R.; Bally, M.B.; Leung, A.W.Y. Recent Treatment Advances and the Role of Nanotechnology, Combination Products, and Immunotherapy in Changing the Therapeutic Landscape of Acute Myeloid Leukemia. Pharm. Res. 2019, 36, 125. [Google Scholar] [CrossRef]
- Estey, E.H. Acute myeloid leukemia: 2021 update on risk-stratification and management. Am. J. Hematol. 2020, 95, 1368–1398. [Google Scholar] [CrossRef]
- Fleischmann, M.; Schnetzke, U.; Hochhaus, A.; Scholl, S. Management of Acute Myeloid Leukemia: Current Treatment Options and Future Perspectives. Cancers 2021, 13, 5722. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Lin, H.; Huang, F.; Xie, Y.; Wong, K.H.; Chen, X.; Wu, D.; Lu, A.; Yang, Z. Targeting Approaches of Nanomedicines in Acute Myeloid Leukemia. Dose Response 2019, 17. [Google Scholar] [CrossRef]
- Wei, A.H.; Roberts, A.W. BCL2 Inhibition: A New Paradigm for the Treatment of AML and Beyond. Hemasphere 2023, 7, e912. [Google Scholar] [CrossRef] [PubMed]
- DiNardo, C.D.; Jonas, B.A.; Pullarkat, V.; Thirman, M.J.; Garcia, J.S.; Wei, A.H.; Konopleva, M.; Döhner, H.; Letai, A.; Fenaux, P.; et al. Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia. N. Engl. J. Med. 2020, 383, 617–629. [Google Scholar] [CrossRef]
- Pitman, M.R.; Powell, J.A.; Coolen, C.; Moretti, P.A.; Zebol, J.R.; Pham, D.H.; Finnie, J.W.; Don, A.S.; Ebert, L.M.; Bonder, C.S.; et al. A selective ATP-competitive sphingosine kinase inhibitor demonstrates anti-cancer properties. Oncotarget 2015, 6, 7065–7083. [Google Scholar] [CrossRef] [PubMed]
- Pitman, M.R.; Costabile, M.; Pitson, S.M. Recent advances in the development of sphingosine kinase inhibitors. Cell Signal 2016, 28, 1349–1363. [Google Scholar] [CrossRef]
- Lewis, A.C.; Wallington-Beddoe, C.T.; Powell, J.A.; Pitson, S.M. Targeting sphingolipid metabolism as an approach for combination therapies in haematological malignancies. Cell Death Discov. 2018, 4, 72. [Google Scholar] [CrossRef]
- Maceyka, M.; Milstien, S.; Spiegel, S. Sphingosine-1-phosphate: The Swiss army knife of sphingolipid signaling. J. Lipid Res. 2009, 50, S272–S276. [Google Scholar] [CrossRef] [PubMed]
- Pitson, S.M. Regulation of sphingosine kinase and sphingolipid signaling. Trends Biochem. Sci. 2011, 36, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Dick, T.E.; Hengst, J.A.; Fox, T.E.; Colledge, A.L.; Kale, V.P.; Sung, S.S.; Sharma, A.; Amin, S.; Loughran, T.P., Jr.; Kester, M.; et al. The apoptotic mechanism of action of the sphingosine kinase 1 selective inhibitor SKI-178 in human acute myeloid leukemia cell lines. J. Pharmacol. Exp. Ther. 2015, 352, 494–508. [Google Scholar] [CrossRef] [PubMed]
- Xia, P.; Gamble, J.R.; Wang, L.; Pitson, S.M.; Moretti, P.A.; Wattenberg, B.W.; D’Andrea, R.J.; Vadas, M.A. An oncogenic role of sphingosine kinase. Curr. Biol. 2000, 10, 1527–1530. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.F.; Liu, X.; Fox, T.E.; Barth, B.M.; Sharma, A.; Turner, S.D.; Awwad, A.; Dewey, A.; Doi, K.; Spitzer, B.; et al. Acid ceramidase is upregulated in AML and represents a novel therapeutic target. Oncotarget 2016, 7, 83208–83222. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.C.; Pope, V.S.; Tea, M.N.; Li, M.; Nwosu, G.O.; Nguyen, T.M.; Wallington-Beddoe, C.T.; Moretti, P.A.B.; Anderson, D.; Creek, D.J.; et al. Ceramide-induced integrated stress response overcomes Bcl-2 inhibitor resistance in acute myeloid leukemia. Blood 2022, 139, 3737–3751. [Google Scholar] [CrossRef]
- Nguyen, T.M.; Jambhrunkar, M.; Wong, S.S.; Ross, D.M.; Joyce, P.; Finnie, J.W.; Manavis, J.; Bremmell, K.; Pitman, M.R.; Prestidge, C.A. Targeting Acute Myeloid Leukemia Using Sphingosine Kinase 1 Inhibitor-Loaded Liposomes. Mol. Pharm. 2023, 20, 3937–3946. [Google Scholar] [CrossRef]
- Powell, J.A.; Lewis, A.C.; Zhu, W.; Toubia, J.; Pitman, M.R.; Wallington-Beddoe, C.T.; Moretti, P.A.; Iarossi, D.; Samaraweera, S.E.; Cummings, N.; et al. Targeting sphingosine kinase 1 induces MCL1-dependent cell death in acute myeloid leukemia. Blood 2017, 129, 771–782. [Google Scholar] [CrossRef]
- Roberts, A.W.; Wei, A.H.; Huang, D.C.S. BCL2 and MCL1 inhibitors for hematologic malignancies. Blood 2021, 138, 1120–1136. [Google Scholar] [CrossRef]
- Moujalled, D.M.; Pomilio, G.; Ghiurau, C.; Ivey, A.; Salmon, J.; Rijal, S.; Macraild, S.; Zhang, L.; Teh, T.C.; Tiong, I.S.; et al. Combining BH3-mimetics to target both BCL-2 and MCL1 has potent activity in pre-clinical models of acute myeloid leukemia. Leukemia 2019, 33, 905–917. [Google Scholar] [CrossRef]
- Roberts, A.W. Therapeutic development and current uses of BCL-2 inhibition. Hematol. Am. Soc. Hematol. Educ. Program. 2020, 2020, 1–9. [Google Scholar] [CrossRef]
- Mayer, L.D.; Tardi, P.; Louie, A.C. CPX-351: A nanoscale liposomal co-formulation of daunorubicin and cytarabine with unique biodistribution and tumor cell uptake properties. Int. J. Nanomed. 2019, 14, 3819–3830. [Google Scholar] [CrossRef]
- Saxena, K.; Konopleva, M. New Treatment Options for Older Patients with Acute Myeloid Leukemia. Curr. Treat. Options Oncol. 2021, 22, 39. [Google Scholar] [CrossRef] [PubMed]
- Talati, C.; Lancet, J.E. CPX-351: Changing the landscape of treatment for patients with secondary acute myeloid leukemia. Future Oncol. 2018, 14, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Tardi, P.; Sadowski, N.; Xie, S.; Heller, D.; Mayer, L. Pharmacokinetics, drug metabolism, and tissue distribution of CPX-351 in animals. Nanomedicine 2020, 30, 102275. [Google Scholar] [CrossRef] [PubMed]
- Shukla, A.; Maiti, P. Nanomedicine and versatile therapies for cancer treatment. MedComm 2022, 3, e163. [Google Scholar] [CrossRef]
- Alfayez, M.; Kantarjian, H.; Kadia, T.; Ravandi-Kashani, F.; Daver, N. CPX-351 (vyxeos) in AML. Leuk. Lymphoma 2020, 61, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Zhou, R. Advances in the Application of Liposomal Nanosystems in Anticancer Therapy. Curr. Stem Cell Res. Ther. 2021, 16, 14–22. [Google Scholar] [CrossRef]
- Sun, W.; Chen, G.; Du, F.; Li, X. Targeted Drug Delivery to Cancer Stem Cells through Nanotechnological Approaches. Curr. Stem Cell Res. Ther. 2021, 16, 367–384. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Selamoglu, Z.; Mileski, K.S.; Pezzani, R.; Redaelli, M.; Cho, W.C.; Kobarfard, F.; Rajabi, S.; Martorell, M.; Kumar, P.; et al. Liposomal Cytarabine as Cancer Therapy: From Chemistry to Medicine. Biomolecules 2019, 9, 773. [Google Scholar] [CrossRef]
- Gurunathan, S.; Kang, M.H.; Qasim, M.; Kim, J.H. Nanoparticle-Mediated Combination Therapy: Two-in-One Approach for Cancer. Int. J. Mol. Sci. 2018, 19, 3264. [Google Scholar] [CrossRef]
- Thol, F. What to use to treat AML: The role of emerging therapies. Hematol. Am. Soc. Hematol. Educ. Program. 2021, 2021, 16–23. [Google Scholar] [CrossRef]
- Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef] [PubMed]
- Trumpp, A.; Haas, S. Cancer stem cells: The adventurous journey from hematopoietic to leukemic stem cells. Cell 2022, 185, 1266–1270. [Google Scholar] [CrossRef]
- Thakral, D.; Gupta, R.; Khan, A. Leukemic stem cell signatures in Acute myeloid leukemia- targeting the Guardians with novel approaches. Stem Cell Rev. Rep. 2022, 18, 1756–1773. [Google Scholar] [CrossRef] [PubMed]
- Cluzeau, T.; Lemoli, R.M.; McCloskey, J.; Cooper, T. Measurable Residual Disease in High-Risk Acute Myeloid Leukemia. Cancers 2022, 14, 1278. [Google Scholar] [CrossRef] [PubMed]
- Molica, M.; Perrone, S.; Mazzone, C.; Cesini, L.; Canichella, M.; de Fabritiis, P. CPX-351: An Old Scheme with a New Formulation in the Treatment of High-Risk AML. Cancers 2022, 14, 2843. [Google Scholar] [CrossRef]
- Lee, D.; Jain, A.G.; Deutsch, Y.; Eatrides, J.; Chan, O.; Padron, E.; Kuykendall, A.; Komrokji, R.; Lancet, J.; Sallman, D.; et al. CPX-351 Yields Similar Response and Survival Outcome in Younger and Older Patients With Secondary Acute Myeloid Leukemia. Clin. Lymphoma Myeloma Leuk. 2022, 22, 774–779. [Google Scholar] [CrossRef]
- Hu, Y.; Caldwell, K.J.; Onciu, M.; Federico, S.M.; Salek, M.; Lewis, S.; Lei, S.; Zhang, J.; Nichols, K.E.; Takemoto, C.M.; et al. CPX-351 induces remission in newly diagnosed pediatric secondary myeloid malignancies. Blood Adv. 2022, 6, 521–527. [Google Scholar] [CrossRef]
- Alotaibi, S.; Niederwieser, D.; Ahmed, S.O.; Sanz, J.; Mohty, M.; Aljurf, M. Current Status of CPX-351 Therapy in Acute Myeloid Leukemia and Myelodysplastic Syndrome. Clin. Lymphoma Myeloma Leuk. 2022, 22, 575–580. [Google Scholar] [CrossRef]
- Thomas, X.; Plesa, A. CPX-351: An attractive option for the treatment of older patients with high-risk or secondary acute myeloid leukaemia. Lancet Haematol. 2021, 8, e468–e469. [Google Scholar] [CrossRef]
- Rautenberg, C.; Stölzel, F.; Röllig, C.; Stelljes, M.; Gaidzik, V.; Lauseker, M.; Kriege, O.; Verbeek, M.; Unglaub, J.M.; Thol, F.; et al. Real-world experience of CPX-351 as first-line treatment for patients with acute myeloid leukemia. Blood Cancer J. 2021, 11, 164. [Google Scholar] [CrossRef]
- Rollig, C.; Serve, H.; Huttmann, A.; Noppeney, R.; Muller-Tidow, C.; Krug, U.; Baldus, C.D.; Brandts, C.H.; Kunzmann, V.; Einsele, H.; et al. Addition of sorafenib versus placebo to standard therapy in patients aged 60 years or younger with newly diagnosed acute myeloid leukaemia (SORAML): A multicentre, phase 2, randomised controlled trial. Lancet Oncol. 2015, 16, 1691–1699. [Google Scholar] [CrossRef]
- Stein, E.M.; DiNardo, C.D.; Fathi, A.T.; Mims, A.S.; Pratz, K.W.; Savona, M.R.; Stein, A.S.; Stone, R.M.; Winer, E.S.; Seet, C.S.; et al. Ivosidenib or enasidenib combined with intensive chemotherapy in patients with newly diagnosed AML: A phase 1 study. Blood 2021, 137, 1792–1803. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.E.; Douglas Smith, B.; Wang, E.S.; Merchant, A.; Oehler, V.G.; Arellano, M.; DeAngelo, D.J.; Pollyea, D.A.; Sekeres, M.A.; Robak, T.; et al. Glasdegib in combination with cytarabine and daunorubicin in patients with AML or high-risk MDS: Phase 2 study results. Am. J. Hematol. 2018, 93, 1301–1310. [Google Scholar] [CrossRef] [PubMed]
- Zeidner, J.F.; Foster, M.C.; Blackford, A.L.; Litzow, M.R.; Morris, L.E.; Strickland, S.A.; Lancet, J.E.; Bose, P.; Levy, M.Y.; Tibes, R.; et al. Randomized multicenter phase II study of flavopiridol (alvocidib), cytarabine, and mitoxantrone (FLAM) versus cytarabine/daunorubicin (7+3) in newly diagnosed acute myeloid leukemia. Haematologica 2015, 100, 1172–1179. [Google Scholar] [CrossRef] [PubMed]
- Stentoft, J. The toxicity of cytarabine. Drug Saf. 1990, 5, 7–27. [Google Scholar] [CrossRef]
- Wei, Y.; Cao, Y.; Sun, R.; Cheng, L.; Xiong, X.; Jin, X.; He, X.; Lu, W.; Zhao, M. Targeting Bcl-2 Proteins in Acute Myeloid Leukemia. Front. Oncol. 2020, 10, 584974. [Google Scholar] [CrossRef]
- Fiorentini, A.; Capelli, D.; Saraceni, F.; Menotti, D.; Poloni, A.; Olivieri, A. The Time Has Come for Targeted Therapies for AML: Lights and Shadows. Oncol. Ther. 2020, 8, 13–32. [Google Scholar] [CrossRef]
- Bogenberger, J.; Whatcott, C.; Hansen, N.; Delman, D.; Shi, C.X.; Kim, W.; Haws, H.; Soh, K.; Lee, Y.S.; Peterson, P.; et al. Combined venetoclax and alvocidib in acute myeloid leukemia. Oncotarget 2017, 8, 107206–107222. [Google Scholar] [CrossRef]
- Teh, T.C.; Nguyen, N.Y.; Moujalled, D.M.; Segal, D.; Pomilio, G.; Rijal, S.; Jabbour, A.; Cummins, K.; Lackovic, K.; Blombery, P.; et al. Enhancing venetoclax activity in acute myeloid leukemia by co-targeting MCL1. Leukemia 2018, 32, 303–312. [Google Scholar] [CrossRef]
- Campos, L.; Rouault, J.P.; Sabido, O.; Oriol, P.; Roubi, N.; Vasselon, C.; Archimbaud, E.; Magaud, J.P.; Guyotat, D. High expression of bcl-2 protein in acute myeloid leukemia cells is associated with poor response to chemotherapy. Blood 1993, 81, 3091–3096. [Google Scholar] [CrossRef]
- Glaser, S.P.; Lee, E.F.; Trounson, E.; Bouillet, P.; Wei, A.; Fairlie, W.D.; Izon, D.J.; Zuber, J.; Rappaport, A.R.; Herold, M.J.; et al. Anti-apoptotic Mcl-1 is essential for the development and sustained growth of acute myeloid leukemia. Genes. Dev. 2012, 26, 120–125. [Google Scholar] [CrossRef]
- Niu, X.; Zhao, J.; Ma, J.; Xie, C.; Edwards, H.; Wang, G.; Caldwell, J.T.; Xiang, S.; Zhang, X.; Chu, R.; et al. Binding of Released Bim to Mcl-1 is a Mechanism of Intrinsic Resistance to ABT-199 which can be Overcome by Combination with Daunorubicin or Cytarabine in AML Cells. Clin. Cancer Res. 2016, 22, 4440–4451. [Google Scholar] [CrossRef] [PubMed]
- Tahir, S.K.; Smith, M.L.; Hessler, P.; Rapp, L.R.; Idler, K.B.; Park, C.H.; Leverson, J.D.; Lam, L.T. Potential mechanisms of resistance to venetoclax and strategies to circumvent it. BMC Cancer 2017, 17, 399. [Google Scholar] [CrossRef]
- Han, L.; Zhang, Q.; Dail, M.; Shi, C.; Cavazos, A.; Ruvolo, V.R.; Zhao, Y.; Kim, E.; Rahmani, M.; Mak, D.H.; et al. Concomitant targeting of BCL2 with venetoclax and MAPK signaling with cobimetinib in acute myeloid leukemia models. Haematologica 2020, 105, 697–707. [Google Scholar] [CrossRef]
- Lin, K.H.; Winter, P.S.; Xie, A.; Roth, C.; Martz, C.A.; Stein, E.M.; Anderson, G.R.; Tingley, J.P.; Wood, K.C. Targeting MCL-1/BCL-XL Forestalls the Acquisition of Resistance to ABT-199 in Acute Myeloid Leukemia. Sci. Rep. 2016, 6, 27696. [Google Scholar] [CrossRef] [PubMed]
- Wei, A.H.; Strickland, S.A., Jr.; Hou, J.Z.; Fiedler, W.; Lin, T.L.; Walter, R.B.; Enjeti, A.; Tiong, I.S.; Savona, M.; Lee, S.; et al. Venetoclax Combined With Low-Dose Cytarabine for Previously Untreated Patients With Acute Myeloid Leukemia: Results From a Phase Ib/II Study. J. Clin. Oncol. 2019, 37, 1277–1284. [Google Scholar] [CrossRef]
- DiNardo, C.D.; Pratz, K.; Pullarkat, V.; Jonas, B.A.; Arellano, M.; Becker, P.S.; Frankfurt, O.; Konopleva, M.; Wei, A.H.; Kantarjian, H.M.; et al. Venetoclax combined with decitabine or azacitidine in treatment-naive, elderly patients with acute myeloid leukemia. Blood 2019, 133, 7–17. [Google Scholar] [CrossRef]
- Roeker, L.E.; Fox, C.P.; Eyre, T.A.; Brander, D.M.; Allan, J.N.; Schuster, S.J.; Nabhan, C.; Hill, B.T.; Shah, N.N.; Lansigan, F.; et al. Tumor Lysis, Adverse Events, and Dose Adjustments in 297 Venetoclax-Treated CLL Patients in Routine Clinical Practice. Clin. Cancer Res. 2019, 25, 4264–4270. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.S.; Baron, J. Management of toxicities associated with targeted therapies for acute myeloid leukemia: When to push through and when to stop. Hematology 2020, 2020, 57–66. [Google Scholar] [CrossRef]
- Finnes, H.D. Venetoclax (Venclexta™). Oncol. Times 2017, 39, 19, 31. [Google Scholar] [CrossRef]
- Lew, T.E.; Seymour, J.F. Clinical experiences with venetoclax and other pro-apoptotic agents in lymphoid malignancies: Lessons from monotherapy and chemotherapy combination. J. Hematol. Oncol. 2022, 15, 75. [Google Scholar] [CrossRef] [PubMed]
- Waggoner, M.; Katsetos, J.; Thomas, E.; Galinsky, I.; Fox, H. Practical Management of the Venetoclax-Treated Patient in Chronic Lymphocytic Leukemia and Acute Myeloid Leukemia. J. Adv. Pract. Oncol. 2022, 13, 400–415. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.M.; Joyce, P.; Ross, D.M.; Bremmell, K.; Jambhrunkar, M.; Wong, S.S.; Prestidge, C.A. Combating Acute Myeloid Leukemia via Sphingosine Kinase 1 Inhibitor-Nanomedicine Combination Therapy with Cytarabine or Venetoclax. Pharmaceutics 2024, 16, 209. https://doi.org/10.3390/pharmaceutics16020209
Nguyen TM, Joyce P, Ross DM, Bremmell K, Jambhrunkar M, Wong SS, Prestidge CA. Combating Acute Myeloid Leukemia via Sphingosine Kinase 1 Inhibitor-Nanomedicine Combination Therapy with Cytarabine or Venetoclax. Pharmaceutics. 2024; 16(2):209. https://doi.org/10.3390/pharmaceutics16020209
Chicago/Turabian StyleNguyen, Thao M., Paul Joyce, David M. Ross, Kristen Bremmell, Manasi Jambhrunkar, Sook S. Wong, and Clive A. Prestidge. 2024. "Combating Acute Myeloid Leukemia via Sphingosine Kinase 1 Inhibitor-Nanomedicine Combination Therapy with Cytarabine or Venetoclax" Pharmaceutics 16, no. 2: 209. https://doi.org/10.3390/pharmaceutics16020209
APA StyleNguyen, T. M., Joyce, P., Ross, D. M., Bremmell, K., Jambhrunkar, M., Wong, S. S., & Prestidge, C. A. (2024). Combating Acute Myeloid Leukemia via Sphingosine Kinase 1 Inhibitor-Nanomedicine Combination Therapy with Cytarabine or Venetoclax. Pharmaceutics, 16(2), 209. https://doi.org/10.3390/pharmaceutics16020209








