QSAR and Chemical Read-Across Analysis of 370 Potential MGMT Inactivators to Identify the Structural Features Influencing Inactivation Potency
Abstract
:1. Introduction
2. Materials and Methods
2.1. Compound Design and Synthesis
2.2. MGMT Activity Assay
2.3. Dataset Preparation
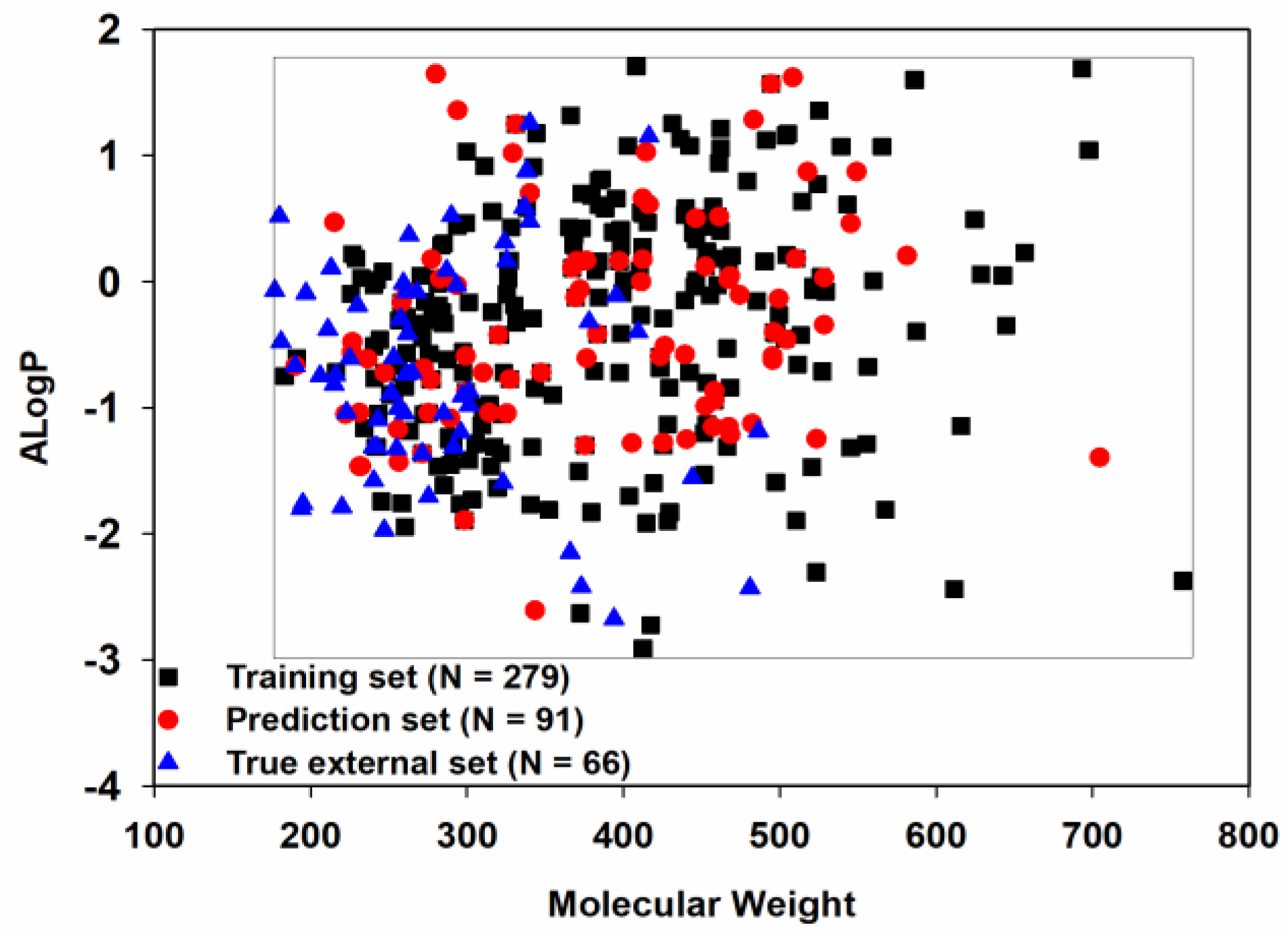
2.4. Descriptor Calculation and Dataset Splitting
2.5. Model Development and Validation
2.6. Best Model Selection by Multiple-Criteria Decision Making
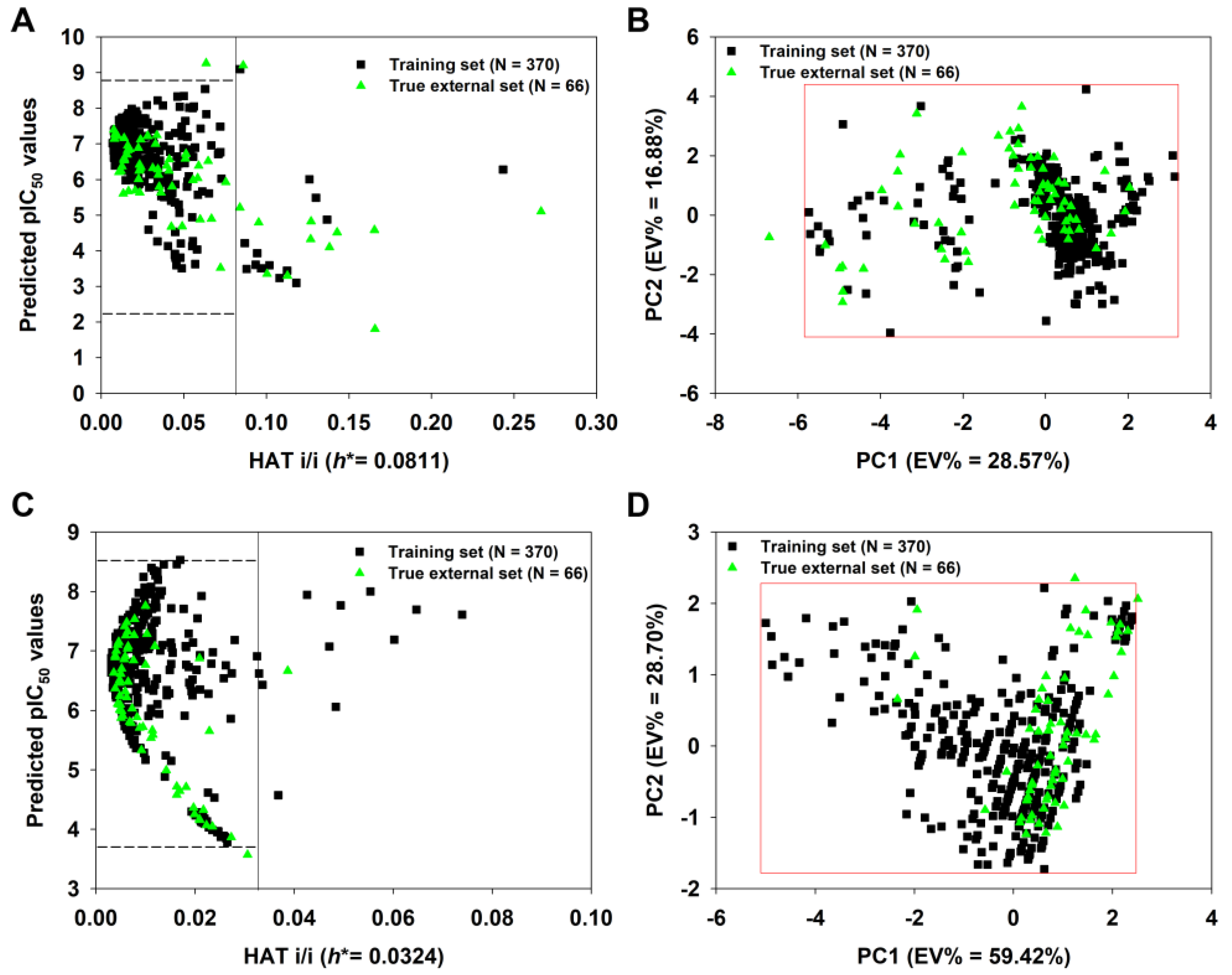
2.7. Applicability Domain (AD) Analysis
2.8. Prediction Using a Similarity-Based Chemical Read-Across Technique
3. Results and Discussion
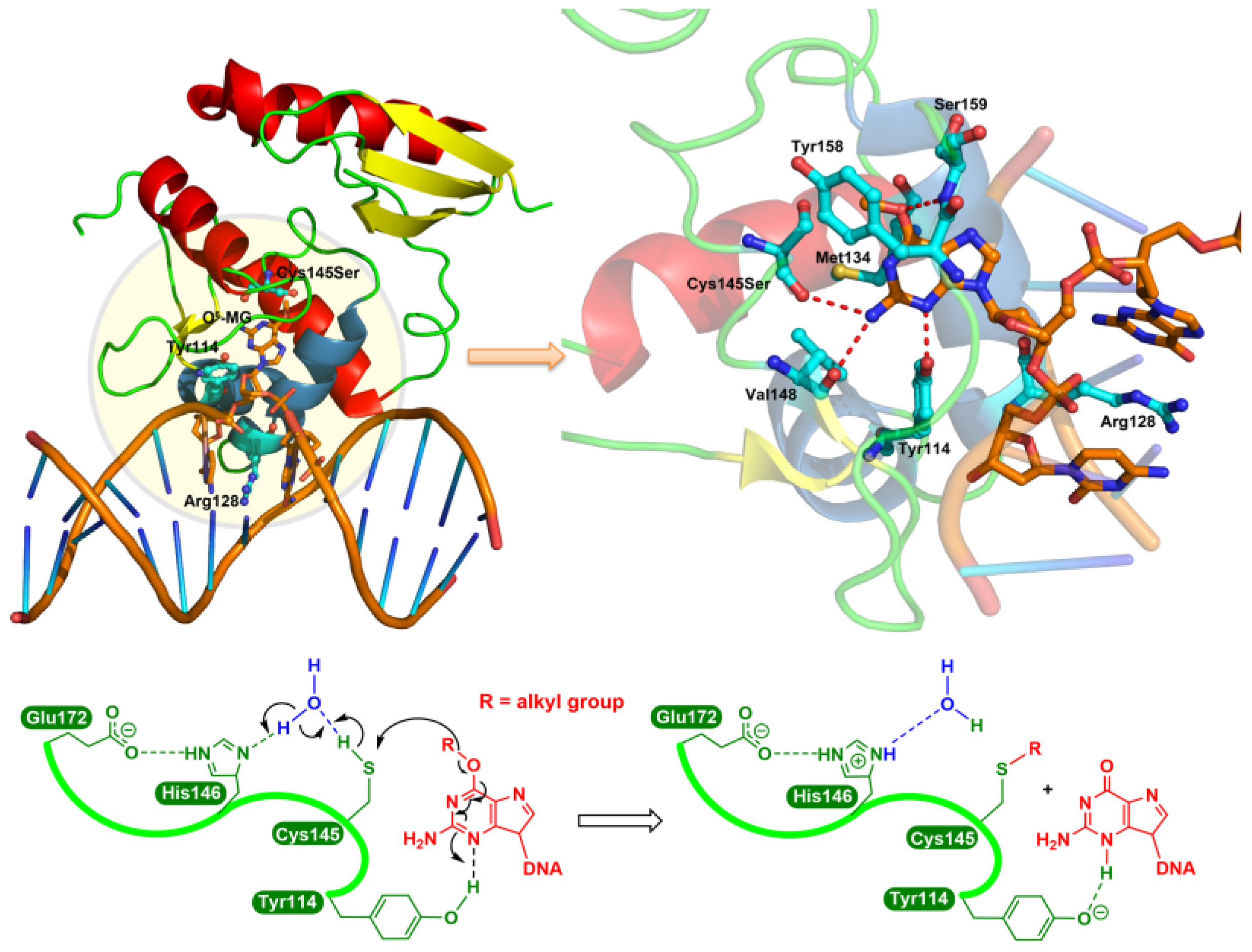
3.1. MGMT Inactivation
3.2. Chemical Space Distribution
3.3. QSAR Modeling of Potential MGMT Inactivators
3.3.1. Model Selection and Evaluation
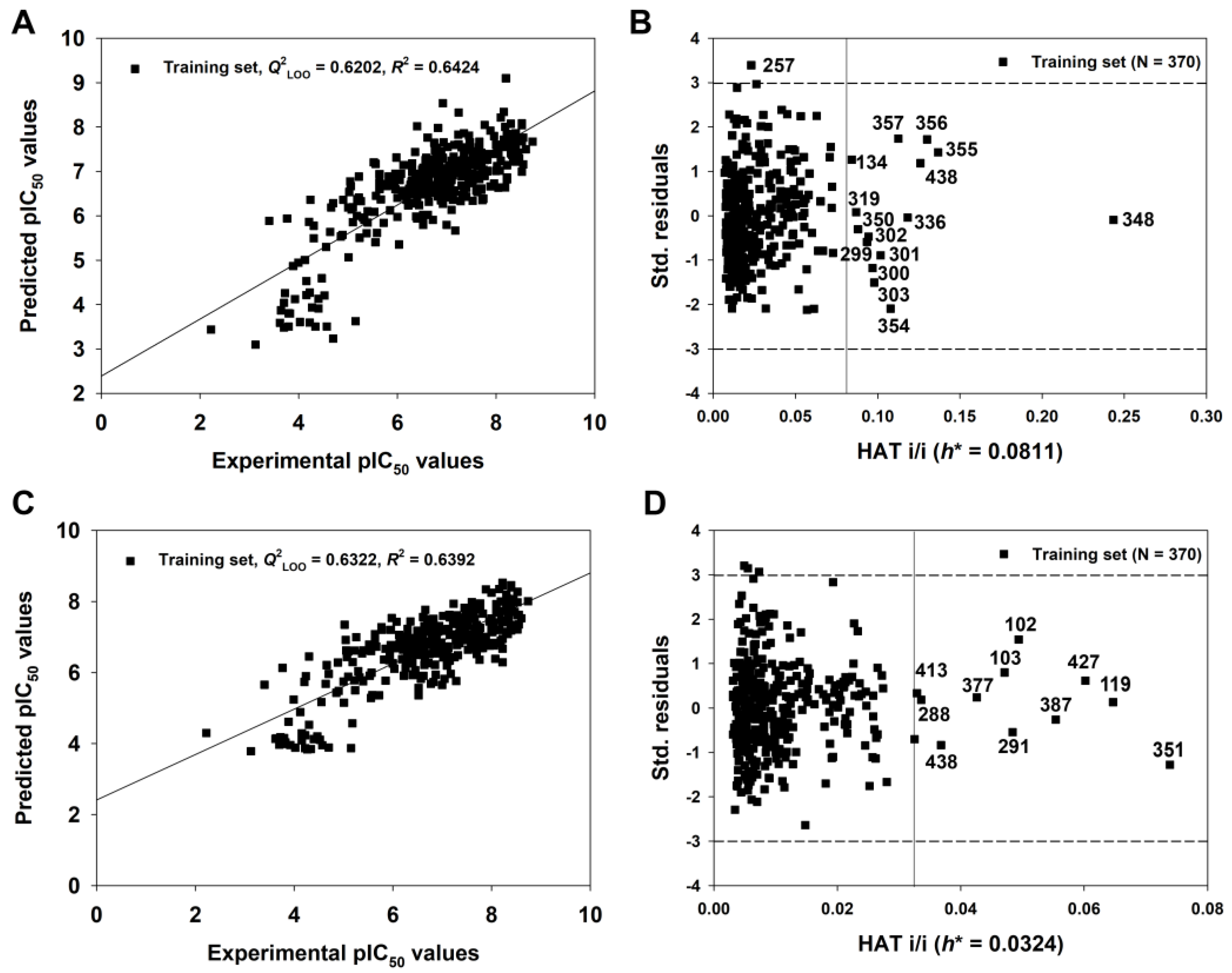
3.3.2. Full Model
3.4. q-RASAR Analysis
3.5. Application of the 2D-QSAR-Based Full Model and q-RASAR-Full Model
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaina, B.; Margison, G.P.; Christmann, M. Targeting O6-methylguanine-DNA methyltransferase with specific inhibitors as a strategy in cancer therapy. Cell. Mol. Life Sci. 2010, 67, 3663–3681. [Google Scholar] [CrossRef]
- Nikolova, T.; Roos, W.P.; Kramer, O.H.; Strik, H.M.; Kaina, B. Chloroethylating nitrosoureas in cancer therapy: DNA damage, repair and cell death signaling. Biochim. Biophys. Acta-Rev. Cancer 2017, 1868, 29–39. [Google Scholar] [CrossRef]
- Sun, G.H.; Zhao, L.J.; Zhong, R.G.; Peng, Y.Z. The specific role of O-6-methylguanine-DNA methyltransferase inhibitors in cancer chemotherapy. Future Med. Chem. 2018, 10, 1971–1996. [Google Scholar] [CrossRef]
- Gnewuch, C.T.; Sosnovsky, G. A critical appraisal of the evolution of N-nitrosoureas as anticancer drugs. Chem. Rev. 1997, 97, 829–1013. [Google Scholar] [CrossRef]
- Kaina, B.; Christmann, M. DNA repair in personalized brain cancer therapy with temozolomide and nitrosoureas. DNA Repair 2019, 78, 128–141. [Google Scholar] [CrossRef]
- Pegg, A.E. Multifaceted roles of alkyltransferase and related proteins in DNA repair, DNA damage, resistance to chemotherapy, and research tools. Chem. Res. Toxicol. 2011, 24, 618–639. [Google Scholar] [CrossRef]
- Lindahl, T.; Demple, B.; Robins, P. Suicide inactivation of the Escherichia-coli O6-methylguanine-DNA methyltransferase. EMBO J. 1982, 1, 1359–1363. [Google Scholar] [CrossRef]
- Daniels, D.S.; Woo, T.T.; Luu, K.X.; Noll, D.M.; Clarke, N.D.; Pegg, A.E.; Tainer, J.A. DNA binding and nucleotide flipping by the human DNA repair protein AGT. Nat. Struct. Mol. Biol. 2004, 11, 714–720. [Google Scholar] [CrossRef]
- Daniels, D.S.; Mol, C.D.; Arvai, A.S.; Kanugula, S.; Pegg, A.E.; Tainer, J.A. Active and alkylated human AGT structures: A novel zinc site, inhibitor and extrahelical base binding. EMBO J. 2000, 19, 1719–1730. [Google Scholar] [CrossRef]
- Xu-Welliver, M.; Pegg, A.E. Degradation of the alkylated form of the DNA repair protein, O6-alkylguanine-DNA alkyltransferase. Carcinogenesis 2002, 23, 823–830. [Google Scholar] [CrossRef]
- Dolan, M.E.; Morimoto, K.; Pegg, A.E. Reduction of O6-alkylguanine-DNA alkyltransferase activity in HeLa-cells treated with O6-alkylguanines. Cancer Res. 1985, 45, 6413–6417. [Google Scholar]
- Dolan, M.E.; Moschel, R.C.; Pegg, A.E. Depletion of mammalian O6-alkylguanine-DNA alkyltransferase activity by O6-benzylguanine provides a means to evaluate the role of this protein in protection against carcinogenic and therapeutic alkylating-agents. Proc. Natl. Acad. Sci. USA 1990, 87, 5368–5372. [Google Scholar] [CrossRef]
- Moschel, R.C.; McDougall, M.G.; Dolan, M.E.; Stine, L.; Pegg, A.E. Structural features of substituted purine derivatives compatible with depletion of human O6-alkylguanine-DNA alkyltransferase. J. Med. Chem. 1992, 35, 4486–4491. [Google Scholar] [CrossRef]
- Chae, M.Y.; McDougall, M.G.; Dolan, M.E.; Swenn, K.; Pegg, A.E.; Moschel, R.C. Substituted O6-benzylguanine derivatives and their inactivation of human O6-alkylguanine-DNA alkyltransferase. J. Med. Chem. 1994, 37, 342–347. [Google Scholar] [CrossRef]
- Chae, M.Y.; Swenn, K.; Kanugula, S.; Dolan, M.E.; Pegg, A.E.; Moschel, R.C. 8-Substituted O6-benzylguanine, substituted 6(4)-(benzyloxy)pyrimidine, and related derivatives as inactivators of human O6-alkylguanine-DNA alkyltransferase. J. Med. Chem. 1995, 38, 359–365. [Google Scholar] [CrossRef]
- Sun, G.H.; Fan, T.J.; Sun, X.D.; Hao, Y.X.; Cui, X.; Zhao, L.J.; Ren, T.; Zhou, Y.; Zhong, R.G.; Peng, Y.Z. In silico prediction of O6-methylguanine-dna methyltransferase inhibitory potency of base analogs with QSAR and machine learning methods. Molecules 2018, 23, 2892. [Google Scholar] [CrossRef]
- Dolan, M.E.; Roy, S.K.; Garbiras, B.J.; Helft, P.; Paras, P.; Chae, M.Y.; Moschel, R.C.; Pegg, A.E. O6-alkylguanine-DNA alkyltransferase inactivation by ester prodrugs of O-6-benzylguanine derivatives and their rate of hydrolysis by cellular esterases. Biochem. Pharmacol. 1998, 55, 1701–1709. [Google Scholar] [CrossRef]
- McElhinney, R.S.; Donnelly, D.J.; McCormick, J.E.; Kelly, J.; Watson, A.J.; Rafferty, J.A.; Elder, R.H.; Middleton, M.R.; Willington, M.A.; McMurry, T.B.H.; et al. Inactivation of O6-alkylguanine-DNA alkyltransferase. 1. Novel O-6-(hetarylmethyl)guanines having basic rings in the side chain. J. Med. Chem. 1998, 41, 5265–5271. [Google Scholar] [CrossRef]
- Terashima, I.; Kohda, K. Inhibition of human O6-alkylguanine-DNA alkyltransferase and potentiation of the cytotoxicity of chloroethylnitrosourea by 4(6)-(benzyloxy)-2,6(4)-diamino-5-(nitro or nitroso)pyrimidine derivatives and analogues. J. Med. Chem. 1998, 41, 503–508. [Google Scholar] [CrossRef]
- Griffin, R.J.; Arris, C.E.; Bleasdale, C.; Boyle, F.T.; Calvert, A.H.; Curtin, N.J.; Dalby, C.; Kanugula, S.; Lembicz, N.K.; Newell, D.R.; et al. Resistance-modifying agents. 8. Inhibition of O6-alkylguanine-DNA alkyltransferase by O6-alkenyl-, O6-cycloalkenyl-, and O6-(2-oxoalkyl)guanines and potentiation of temozolomide cytotoxicity in vitro by O-6-(1-cyclopentenylmethyl)guanine. J. Med. Chem. 2000, 43, 4071–4083. [Google Scholar] [CrossRef]
- Reinhard, J.; Hull, W.E.; von der Lieth, C.W.; Eichhorn, U.; Kliem, H.C.; Kaina, B.; Wiessler, M. Monosaccharide-linked inhibitors of O-6-methylguanine-DNA methyltransferase (MGMT): Synthesis, molecular modeling, and structure-activity relationships. J. Med. Chem. 2001, 44, 4050–4061. [Google Scholar] [CrossRef] [PubMed]
- Pauly, G.T.; Loktionova, N.A.; Fang, Q.M.; Vankayala, S.L.; Guida, W.C.; Pegg, A.E. Substitution of aminomethyl at the meta-position enhances the inactivation of O6-alkylguanine-DNA alkyltransferase by O6-benzylguanine. J. Med. Chem. 2008, 51, 7144–7153. [Google Scholar] [CrossRef] [PubMed]
- Ranson, M.; Middleton, M.R.; Bridgewater, J.; Lee, S.M.; Dawson, M.; Jowle, D.; Halbert, G.; Waller, S.; McGrath, H.; Gumbrell, L.; et al. Lomeguatrib, a potent inhibitor of O-6-alkylguanine-DNA-alkyltransferase: Phase I safety, pharmacodynamic, and pharmacokinetic trial and evaluation in combination with temozolomide in patients with advanced solid tumors. Clin. Cancer Res. 2006, 12, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Warren, K.E.; Gururangan, S.; Geyer, J.R.; McLendon, R.E.; Poussaint, T.Y.; Wallace, D.; Balis, F.M.; Berg, S.L.; Packer, R.J.; Goldman, S.; et al. A phase II study of O6-benzylguanine and temozolomide in pediatric patients with recurrent or progressive high-grade gliomas and brainstem gliomas: A Pediatric Brain Tumor Consortium study. J. Neuro-Oncol. 2012, 106, 643–649. [Google Scholar] [CrossRef]
- Quinn, J.A.; Jiang, S.X.; Reardon, D.A.; Desjardins, A.; Vredenburgh, J.J.; Rich, J.N.; Gururangan, S.; Friedman, A.H.; Bigner, D.D.; Sampson, J.H.; et al. Phase II trial of temozolomide plus O6-benzylguanine in adults with recurrent, temozolomide-resistant malignant glioma. J. Clin. Oncol. 2009, 27, 1262–1267. [Google Scholar] [CrossRef]
- Watson, A.J.; Sabharwal, A.; Thorncroft, M.; McGown, G.; Kerr, R.; Bojanic, S.; Soonawalla, Z.; King, A.; Miller, A.; Waller, S.; et al. Tumor O(6)-methylguanine-DNA methyltransferase inactivation by oral lomeguatrib. Clin. Cancer Res. 2010, 16, 743–749. [Google Scholar] [CrossRef]
- Pegg, A.E.; Kanugula, S.; Edara, S.; Pauly, G.T.; Moschel, R.C.; Goodtzova, K. Reaction of O6-benzylguanine-resistant mutants of human O6-alkylguanine-DNA alkyltransferase with O6-benzylguanine in oligodeoxyribonucleotides. J. Biol. Chem. 1998, 273, 10863–10867. [Google Scholar] [CrossRef]
- Dolan, M.E.; Chae, M.Y.; Pegg, A.E.; Mullen, J.H.; Friedman, H.S.; Moschel, R.C. Metabolism of O6-benzylguanine, an inactivator of O6-alkylguanine-dna alkyltransferase. Cancer Res. 1994, 54, 5123–5130. [Google Scholar]
- Zhu, R.; Liu, M.C.; Luo, M.Z.; Penketh, P.G.; Baumann, R.P.; Shyam, K.; Sartorelli, A.C. 4-Nitrobenzyloxycarbonyl derivatives of O6-benzylguanine as hypoxia-activated prodrug inhibitors of O6-alkylguanine-DNA alkyltransferase (AGT), which produces resistance to agents targeting the O6 position of DNA guanine. J. Med. Chem. 2011, 54, 7720–7728. [Google Scholar] [CrossRef]
- Zhu, R.; Seow, H.A.; Baumann, R.P.; Ishiguro, K.; Penketh, P.G.; Shyam, K.; Sartorelli, A.C. Design of a hypoxia-activated prodrug inhibitor of O-6-alkylguanine-DNA alkyltransferase. Bioorg. Med. Chem. Lett. 2012, 22, 6242–6247. [Google Scholar] [CrossRef]
- Huang, T.; Sun, G.; Zhao, L.; Zhang, N.; Zhong, R.; Peng, Y. Quantitative structure-activity relationship (QSAR) studies on the toxic effects of nitroaromatic compounds (NACs): A Systematic Review. Int. J. Mol. Sci. 2021, 22, 8557. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.; Roy, K. Ecotoxicological risk assessment of organic compounds against various aquatic and terrestrial species: Application of interspecies i-QSTTR and species sensitivity distribution techniques. Green Chem. 2022, 24, 1458–1516. [Google Scholar] [CrossRef]
- Gramatica, P. Principles of QSAR modeling: Comments and suggestions from personal experience. Int. J. Quant. Struct. Prop. Relatsh. 2020, 5, 61–97. [Google Scholar] [CrossRef]
- Li, F.; Sun, G.; Fan, T.; Zhang, N.; Zhao, L.; Zhong, R.; Peng, Y. Ecotoxicological QSAR modelling of the acute toxicity of fused and non-fused polycyclic aromatic hydrocarbons (FNFPAHs) against two aquatic organisms: Consensus modelling and comparison with ECOSAR. Aquat. Toxicol. 2023, 255, 106393. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Sun, G.; Fan, T.; Li, F.; Xu, Y.; Zhang, N.; Zhao, L.; Zhong, R. Ecotoxicological QSAR study of fused/non-fused polycyclic aromatic hydrocarbons (FNFPAHs): Assessment and priority ranking of the acute toxicity to Pimephales promelas by QSAR and consensus modeling methods. Sci. Total Environ. 2023, 876, 162736. [Google Scholar] [CrossRef]
- Kode Srl. Dragon Software for Molecular Descriptor Calculation V 7.0.6. Available online: https://chm.kode-solutions.net/ (accessed on 3 September 2017).
- Yap, C.W. PaDEL-Descriptor: An open source software to calculate molecular descriptors and fingerprints. J. Comput. Chem. 2011, 32, 1466–1474. [Google Scholar] [CrossRef]
- Gramatica, P.; Chirico, N.; Papa, E.; Cassani, S.; Kovarich, S. QSARINS: A new software for the development, analysis, and validation of QSAR MLR models. J. Comput. Chem. 2013, 34, 2121–2132. [Google Scholar] [CrossRef]
- Gramatica, P.; Cassani, S.; Chirico, N. QSARINS-Chem: Insubria datasets and new QSAR/QSPR models for environmental pollutants in QSARINS. J. Comput. Chem. 2014, 35, 1036–1044. [Google Scholar] [CrossRef]
- Gramatica, P.; Cassani, S.; Sangion, A. Aquatic ecotoxicity of personal care products: QSAR models and ranking for prioritization and safer alternatives’ design. Green Chem. 2016, 18, 4393–4406. [Google Scholar] [CrossRef]
- Sangion, A.; Gramatica, P. Hazard of pharmaceuticals for aquatic environment: Prioritization by structural approaches and prediction of ecotoxicity. Environ. Int. 2016, 95, 131–143. [Google Scholar] [CrossRef]
- OECD (Organization for Economic Co-Operation and Development). Guidance Document on the Validation of (Quantitative) Structure-Activity Relationships [(Q) SAR] Models; OECD Environment Health and Safety Publications Series on Testing and Assessment No. 69; OECD: Paris, France, 2007; Available online: http://www.oecd.org/env/guidance-document-onthe-validation-of-quantitative-structure-activity-relationship-q-sar-models-9789264085442-en.htm (accessed on 12 March 2021).
- Golbraikh, A.; Tropsha, A. Beware of q(2)! J. Mol. Graph. 2002, 20, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Gramatica, P.; Sangion, A. A historical excursus on the statistical validation parameters for QSAR models: A clarification concerning metrics and terminology. J. Chem. Inf. Model. 2016, 56, 1127–1131. [Google Scholar] [CrossRef] [PubMed]
- Todeschini, R.; Consonni, V.; Maiocchi, A. The K correlation index: Theory development and its application in chemometrics. Chemometr. Intell. Lab. Syst. 1999, 46, 13–29. [Google Scholar] [CrossRef]
- Roy, K.; Ambure, P.; Kar, S.; Ojha, P.K. Is it possible to improve the quality of predictions from an "intelligent" use of multiple QSAR/QSPR/QSTR models? J. Chemom. 2018, 32, e2992. [Google Scholar] [CrossRef]
- Chatterjee, M.; Banerjee, A.; De, P.; Gajewicz-Skretna, A.; Roy, K. A novel quantitative read-across tool designed purposefully to fill the existing gaps in nanosafety data. Environ. Sci. Nano 2022, 9, 189–203. [Google Scholar] [CrossRef]
- Banerjee, A.; Chatterjee, M.; De, P.; Roy, K. Quantitative predictions from chemical read-across and their confidence measures. Chemometr. Intell. Lab. 2022, 227, 104613. [Google Scholar] [CrossRef]
- Banerjee, A.; Roy, K. First report of q-RASAR modeling toward an approach of easy interpretability and efficient transferability. Mol. Divers. 2022, 26, 2847–2862. [Google Scholar] [CrossRef]
- Banerjee, A.; De, P.; Kumar, V.; Kar, S.; Roy, K. Quick and efficient quantitative predictions of androgen receptor binding affinity for screening endocrine disruptor chemicals using 2D-QSAR and chemical read-across. Chemosphere 2022, 309, 136579. [Google Scholar] [CrossRef]
- Cruz-Monteagudo, M.; Medina-Franco, J.L.; Pérez-Castillo, Y.; Nicolotti, O.; Cordeiro, M.N.D.S.; Borges, F. Activity cliffs in drug discovery: Dr Jekyll or Mr Hyde? Drug Discov. Today 2014, 19, 1069–1080. [Google Scholar] [CrossRef]
- Cruz-Monteagudo, M.; Medina-Franco, J.L.; Perera-Sardiña, Y.; Borges, F.; Tejera, E.; Paz-Y-Miño, C.; Pérez-Castillo, Y.; Sánchez-Rodríguez, A.; Contreras-Posada, Z.; Cordeiro, M.N. Probing the hypothesis of SAR continuity restoration by the removal of activity cliffs generators in QSAR. Curr. Pharm. Des. 2016, 22, 5043–5056. [Google Scholar] [CrossRef]
- Sun, G.; Zhang, Y.; Pei, L.; Lou, Y.; Mu, Y.; Yun, J.; Li, F.; Wang, Y.; Hao, Z.; Xi, S.; et al. Chemometric QSAR modeling of acute oral toxicity of Polycyclic Aromatic Hydrocarbons (PAHs) to rat using simple 2D descriptors and interspecies toxicity modeling with mouse. Ecotoxicol. Environ. Saf. 2021, 222, 112525. [Google Scholar] [CrossRef] [PubMed]
- Li, F.F.; Fan, T.J.; Sun, G.H.; Zhao, L.J.; Zhong, R.G.; Peng, Y.Z. Systematic QSAR and iQCCR modelling of fused/non-fused aromatic hydrocarbons (FNFAHs) carcinogenicity to rodents: Reducing unnecessary chemical synthesis and animal testing. Green Chem. 2022, 24, 5304–5319. [Google Scholar] [CrossRef]
- Hao, Y.X.; Sun, G.H.; Fan, T.J.; Tang, X.Y.; Zhang, J.; Liu, Y.D.; Zhang, N.; Zhao, L.J.; Zhong, R.G.; Peng, Y.Z. In vivo toxicity of nitroaromatic compounds to rats: QSTR modelling and interspecies toxicity relationship with mouse. J. Hazard. Mater. 2020, 399, 122981. [Google Scholar] [CrossRef] [PubMed]
- Ghose, A.K.; Crippen, G.M. Atomic physicochemical parameters for 3-dimensional structure-directed quantitative structure-activity-relationships I. Partition-coefficients as a measure of hydrophobicity. J. Comput. Chem. 1986, 7, 565–577. [Google Scholar] [CrossRef]
- Roy, K.; Ambure, P. The “double cross-validation” software tool for MLR QSAR model development. Chemometr. Intell. Lab. Syst. 2016, 159, 108–126. [Google Scholar] [CrossRef]
- Todeschini, R.; Consonni, V. Molecular Descriptors for Chemoinformatics, Second, Revised and Enlarged Edition; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Sun, G.H.; Fan, T.J.; Zhang, N.; Ren, T.; Zhao, L.J.; Zhong, R.G. Identification of the structural features of guanine derivatives as MGMT inhibitors using 3D-QSAR modeling combined with molecular docking. Molecules 2016, 21, 823. [Google Scholar] [CrossRef]
- Hall, L.H.; Kier, L.B. Electrotopological state indices for atom types: A novel combination of electronic, topological, and valence state information. J. Chem. Inf. Comput. Sci. 1995, 35, 1039–1045. [Google Scholar] [CrossRef]
- Galimberti, F.; Moretto, A.; Papa, E. Application of chemometric methods and QSAR models to support pesticide risk assessment starting from ecotoxicological datasets. Water Res. 2020, 174, 115583. [Google Scholar] [CrossRef]
- Roy, K.; Ambure, P.; Kar, S. How precise are our quantitative structure-activity relationship derived predictions for new query chemicals? ACS Omega 2018, 3, 11392–11406. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, A.; Pérez-Castillo, Y.; Schürer, S.C.; Nicolotti, O.; Mangiatordi, G.F.; Borges, F.; Cordeiro, M.N.D.S.; Tejera, E.; Medina-Franco, J.L.; Cruz-Monteagudo, M. From flamingo dance to (desirable) drug discovery: A nature-inspired approach. Drug Discov. Today 2017, 22, 1489–1502. [Google Scholar] [CrossRef]
- Lambrinidis, G.; Tsantili-Kakoulidou, A. Challenges with multi-objective QSAR in drug discovery. Expert. Opin. Drug Discov. 2018, 13, 851–859. [Google Scholar] [CrossRef] [PubMed]




| Division | Fitting | Robustness | Chance Correlation | External Validation | Accuracy | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Scheme | Ntr | Npr | R2 | Q2LOO | Q2LMO | Q2Yscr | R2Yscr | R2pr | Q2F1 | Q2F2 | Q2F3 | CCCpr | RMSEtr | RMSEpr | MAEtr | MAEpr |
| ORes | 278 | 92 | 0.5098 | 0.4968 | 0.4952 | −0.0186 | 0.0108 | 0.5319 | 0.5271 | 0.5266 | 0.5658 | 0.6891 | 0.8669 | 0.8151 | 0.6666 | 0.6440 |
| pIC50 = 3.9243 + 0.6729F09[O-S] + 0.121SaaN + 1.005MDEN-12 (k = 1.0, |R20 − R′20| = 0.4582) | (1) | |||||||||||||||
| OStr | 278 | 92 | 0.6826 | 0.6496 | 0.6451 | −0.0544 | 0.0432 | 0.5882 | 0.5721 | 0.5712 | 0.5814 | 0.7617 | 0.6917 | 0.7943 | 0.5514 | 0.5982 |
| pIC50 = 7.1639 − 47.2151VE2sign_B(m) + 2.8662MATS6i − 1.5036GATS7p − 0.1826H-048 + 0.5367O-060 − 0.3698B08[N-O] + 0.3948F06[C-S] − 0.1856SsNH2 + 0.0537minHBint6 + 1.8451MDEN-12 + 0.1755MDEN-22 − 0.8906minaaCH (k = 1.0, |R20 − R′20| = 0.1428) | (2) | |||||||||||||||
| Random (2D-QSAR) | 279 | 91 | 0.6086 | 0.5743 | 0.5648 | −0.0424 | 0.0324 | 0.7474 | 0.7377 | 0.7375 | 0.7437 | 0.8530 | 0.7682 | 0.6215 | 0.6114 | 0.5224 |
| pIC50 = 4.5562 + 2.5829MATS6i − 0.191nCp + 0.3196O-060 + 0.6746B03[O-S] − 0.2499SsNH2 + 2.4853maxHBd − 2.3712hmin + 1.1784MDEN-12 − 0.6509minaaCH (k = 1.0, |R20 − R′20| = 0.2436) | (3) | |||||||||||||||
| 2D-QSAR-Full model | 370 | — | 0.6426 | 0.6202 | 0.6127 | −0.0309 | 0.0248 | — | — | — | — | — | 0.7320 | — | 0.5855 | — |
| pIC50 = 4.7334 + 2.3826MATS6i − 0.2387nCp + 0.3401O-060 + 0.6301B03[O-S] − 0.248SsNH2 + 2.1364maxHBd − 2.3442hmin + 1.8332MDEN-12 − 0.6324minaaCH | (4) | |||||||||||||||
| q-RASAR | 279 | 91 | 0.6059 | 0.5957 | 0.5926 | −0.0189 | 0.0103 | 0.7528 | 0.7389 | 0.7387 | 0.7449 | 0.8560 | 0.7708 | 0.6201 | 0.6144 | 0.4812 |
| pIC50 = −1.1683 + 0.9192RA function (ED) + 0.0718CATS2D_07_AL + 1.2875LLS_02 | (5) | |||||||||||||||
| q-RASAR-Full model | 370 | 0.6392 | 0.6322 | 0.6305 | −0.0136 | 0.0083 | — | — | — | — | — | 0.7354 | — | 0.5799 | — | |
| pIC50 = −1.1089 + 0.9149RA function (ED) + 0.0667CATS2D_07_AL + 1.3166LLS_02 | (6) | |||||||||||||||
| Descriptors | Std. Coefficient | Range | Definition | |
|---|---|---|---|---|
| (Full Model) | Min | Max | ||
| MATS6i | 0.2152 (0.1946) | −0.212 | 0.371 | Moran autocorrelation of lag 6 weighted by ionization potential (DRAGON) |
| nCp | −0.106 (−0.1331) | 0 | 6 | number of terminal primary C(sp3) (DRAGON) |
| O-060 | 0.1908 (0.2012) | 0 | 4 | Al-O-Ar/Ar-O-Ar/R..O..R/R-O-C=X (Atom-centered fragments, Basic descriptors) (DRAGON) |
| B03[O-S] | 0.2623 (0.2452) | 0 | 1 | Presence/absence of O-S at topological distance 3 (DRAGON) |
| SsNH2 | −0.4636 (−0.4582) | 0 | 11.662 | Sum of atom-type E-State: –NH2 (DRAGON) |
| maxHBd | 0.2085 (0.185) | 0 | 0.764 | Maximum E-States for (strong) Hydrogen Bond donors (PaDEL) |
| hmin | −0.1766 (−0.1792) | −0.447 | 0.425 | Minimum H E-State (PaDEL) |
| MDEN-12 | 0.6452 (0.6567) | 0 | 2.515 | Molecular distance edge between all primary and secondary nitrogens (PaDEL) |
| minaaCH | −0.1103 (−0.1063) | 1.075 | 2.329 | Minimum atom-type E-State: CH: (PaDEL) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, G.; Bai, P.; Fan, T.; Zhao, L.; Zhong, R.; McElhinney, R.S.; McMurry, T.B.H.; Donnelly, D.J.; McCormick, J.E.; Kelly, J.; et al. QSAR and Chemical Read-Across Analysis of 370 Potential MGMT Inactivators to Identify the Structural Features Influencing Inactivation Potency. Pharmaceutics 2023, 15, 2170. https://doi.org/10.3390/pharmaceutics15082170
Sun G, Bai P, Fan T, Zhao L, Zhong R, McElhinney RS, McMurry TBH, Donnelly DJ, McCormick JE, Kelly J, et al. QSAR and Chemical Read-Across Analysis of 370 Potential MGMT Inactivators to Identify the Structural Features Influencing Inactivation Potency. Pharmaceutics. 2023; 15(8):2170. https://doi.org/10.3390/pharmaceutics15082170
Chicago/Turabian StyleSun, Guohui, Peiying Bai, Tengjiao Fan, Lijiao Zhao, Rugang Zhong, R. Stanley McElhinney, T. Brian H. McMurry, Dorothy J. Donnelly, Joan E. McCormick, Jane Kelly, and et al. 2023. "QSAR and Chemical Read-Across Analysis of 370 Potential MGMT Inactivators to Identify the Structural Features Influencing Inactivation Potency" Pharmaceutics 15, no. 8: 2170. https://doi.org/10.3390/pharmaceutics15082170
APA StyleSun, G., Bai, P., Fan, T., Zhao, L., Zhong, R., McElhinney, R. S., McMurry, T. B. H., Donnelly, D. J., McCormick, J. E., Kelly, J., & Margison, G. P. (2023). QSAR and Chemical Read-Across Analysis of 370 Potential MGMT Inactivators to Identify the Structural Features Influencing Inactivation Potency. Pharmaceutics, 15(8), 2170. https://doi.org/10.3390/pharmaceutics15082170







