Therapeutic Strategies Aimed at Improving Neuroplasticity in Alzheimer Disease
Abstract
1. Introduction
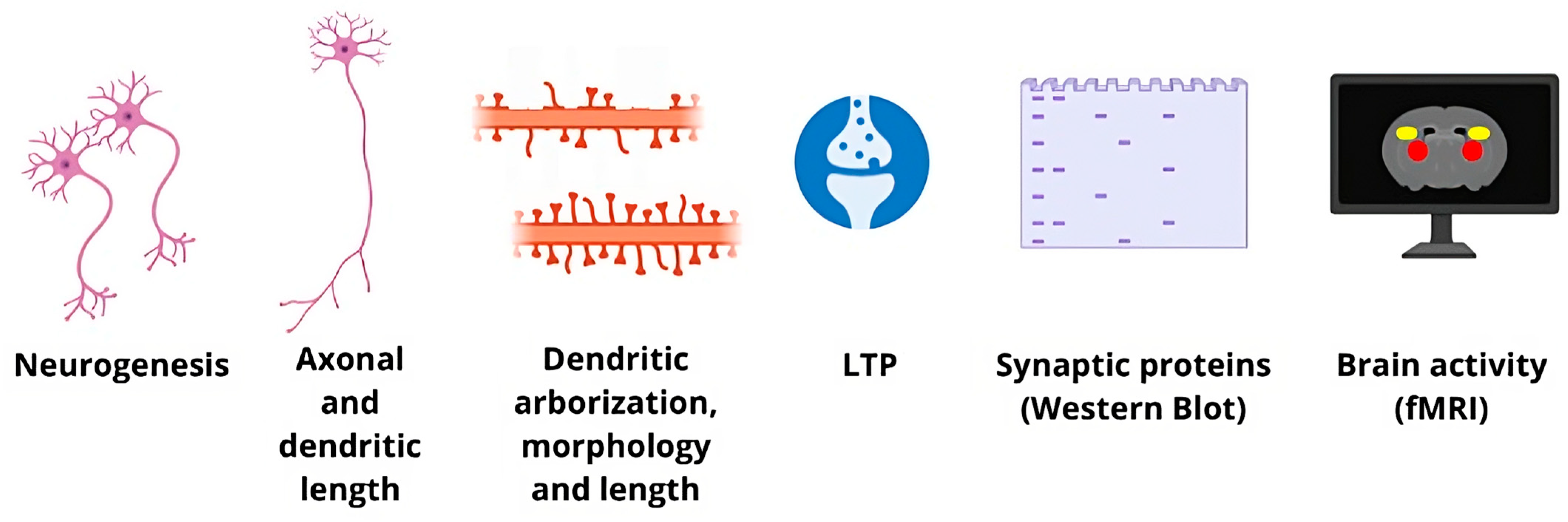
2. The Multiple Facets of Neuroplasticity
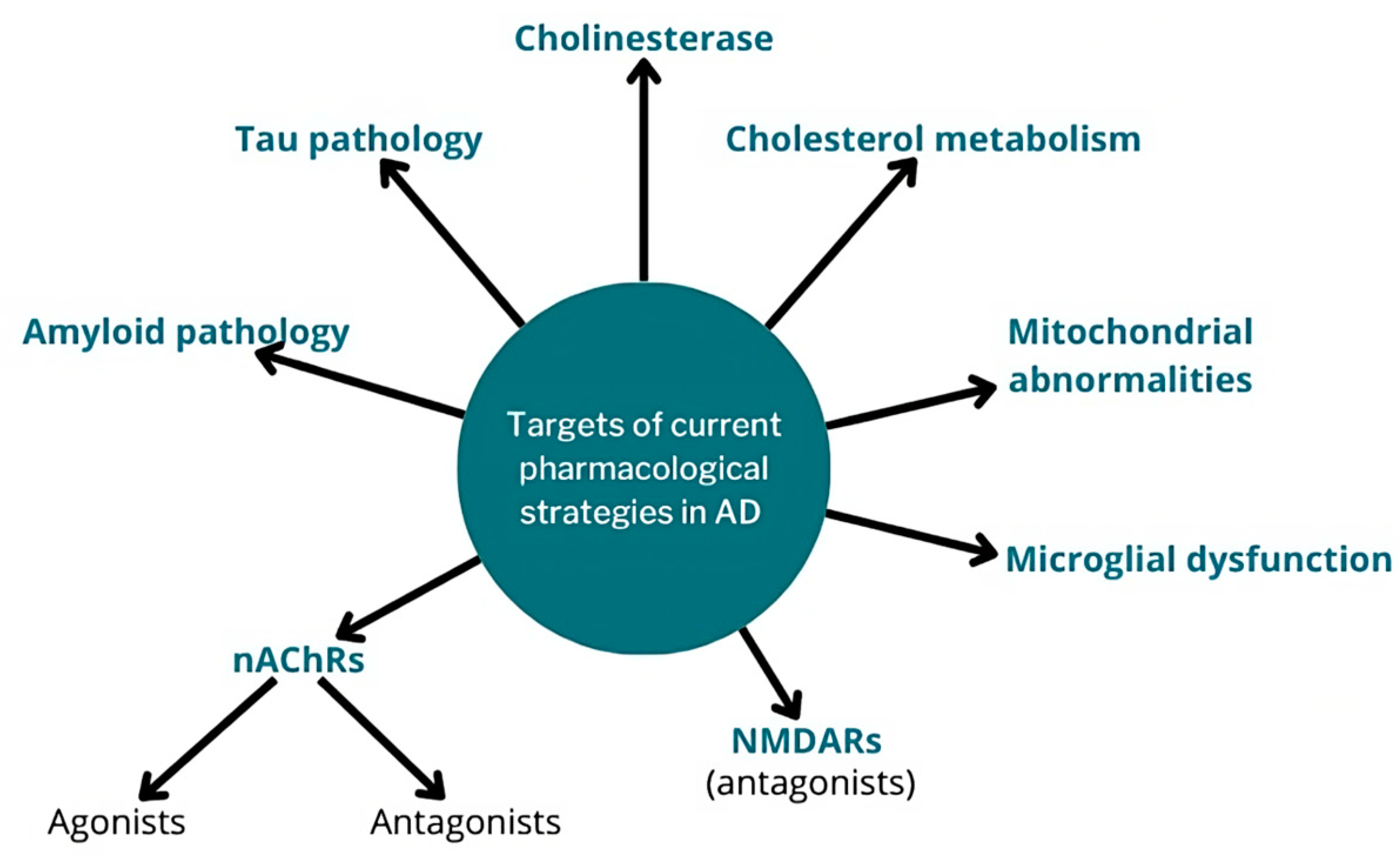
3. Current Pharmacological Strategies in AD
4. Clinical Trials Addressing Neuroplasticity in AD Patients
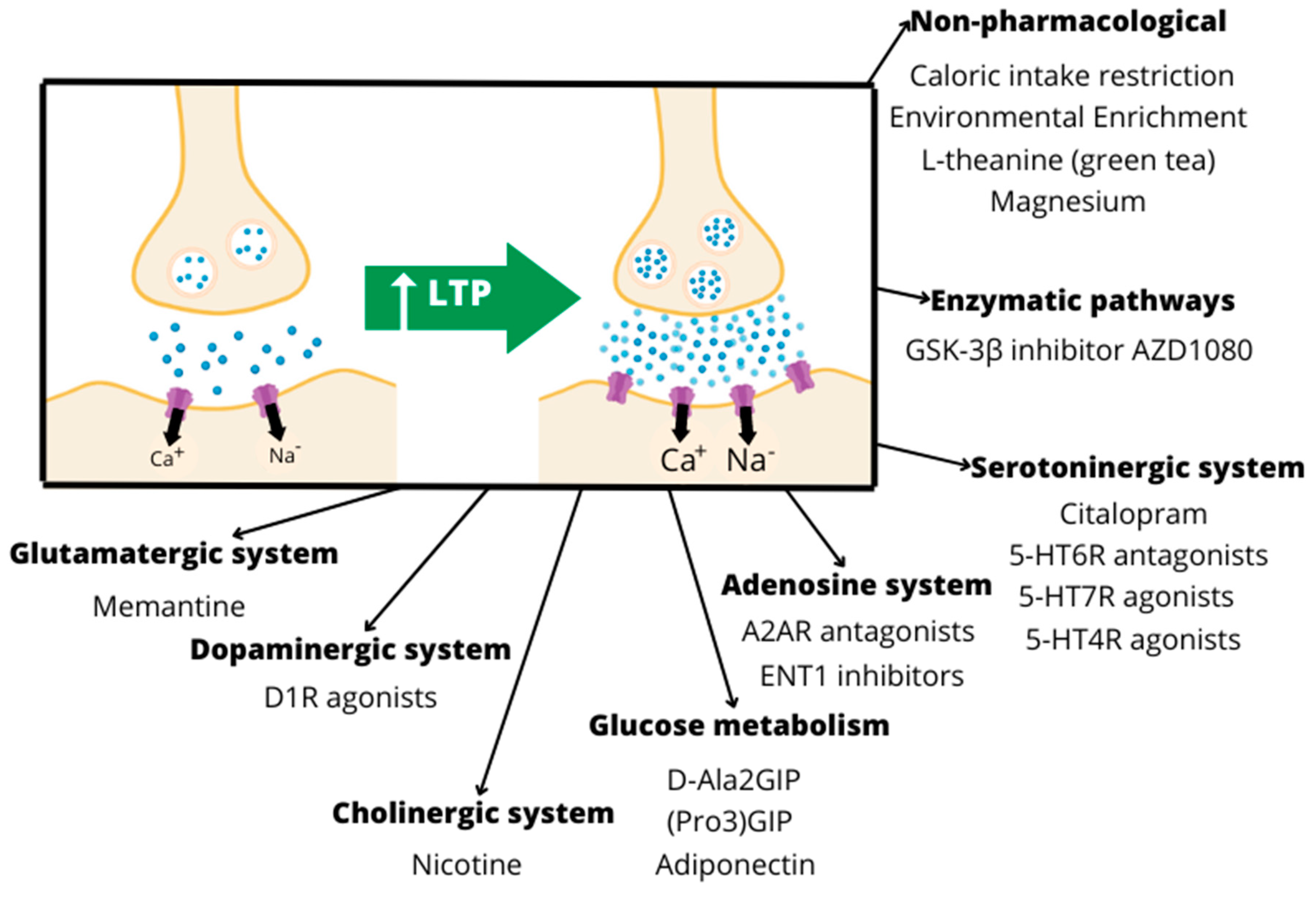
5. Interventions Targeting Neuroplasticity in Animal Models of AD
5.1. Glucose Metabolism
5.2. Neurotrophic Compounds
5.3. Glutamatergic System
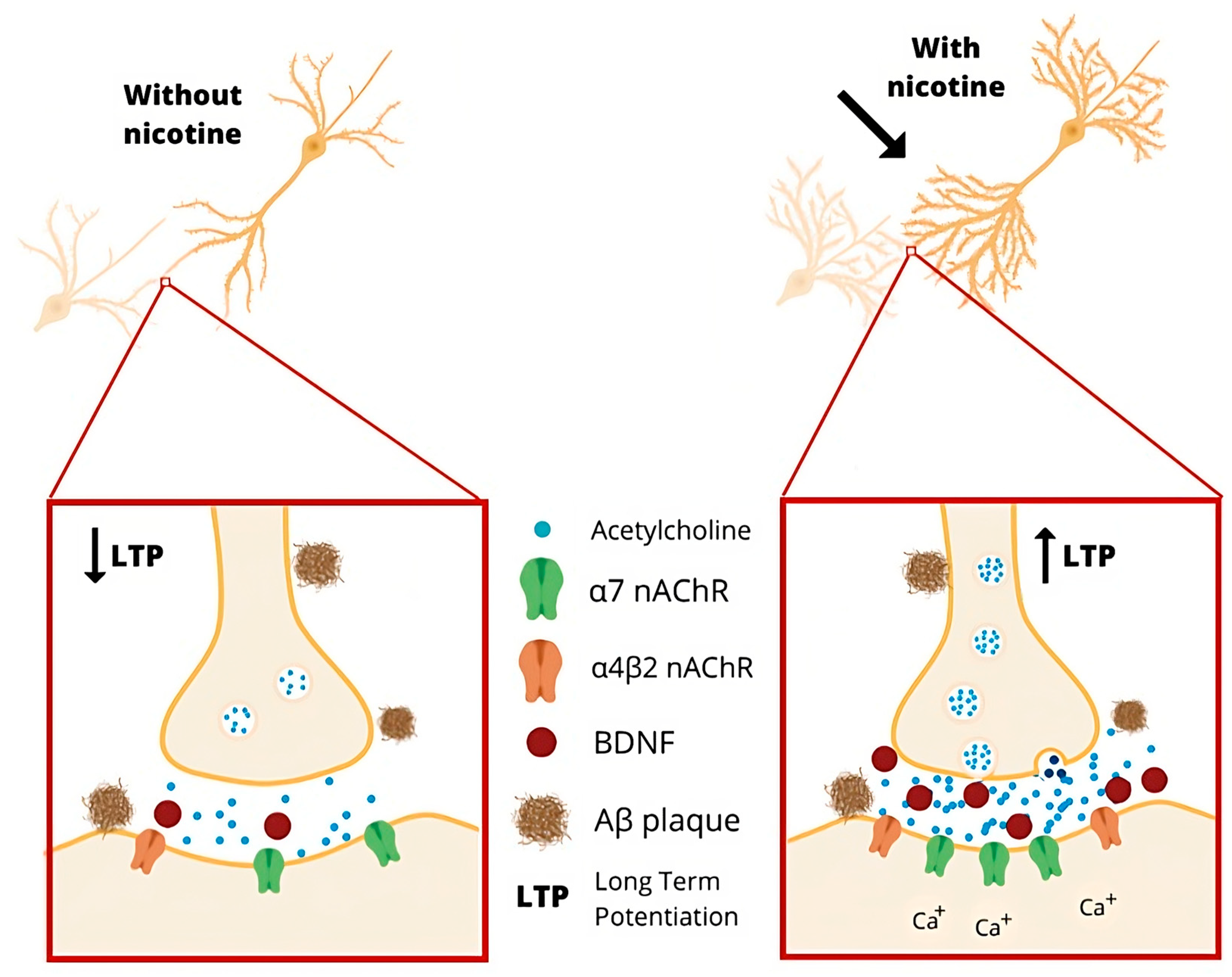
5.4. Cholinergic System
5.5. Serotoninergic System
5.6. Dopaminergic System
5.7. Adenosine System
5.8. Enzymatic Pathways
5.9. Neuroinflammation
5.10. Lipid Metabolism
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Psychiatry Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatry Association: Arlington, VA, USA, 2013; pp. 591–644. [Google Scholar]
- What is Alzheimer’s. Available online: https://www.alz.org/alzheimers-dementia/what-is-alzheimers (accessed on 15 March 2023).
- Trambauer, J.; Fukumori, A.; Steiner, H. Pathogenic Aβ generation in familial Alzheimer’s disease: Novel mechanistic insights and therapeutic implications. Curr. Opin. Neurobiol. 2020, 61, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Chávez-Gutiérrez, L.; Szaruga, M. Mechanisms of neurodegeneration—Insights from familial Alzheimer’s disease. Semin. Cell Dev. Biol. 2020, 105, 75–85. [Google Scholar] [CrossRef]
- Ulaganathan, S.; Pitchaimani, A. Spontaneous and familial models of Alzheimer’s disease: Challenges and advances in preclinical research. Life Sci. 2023, 328, 121918. [Google Scholar] [CrossRef] [PubMed]
- Farlow, M.; Arnold, S.; van Dyck, C.H.; Aisen, P.S.; Snider, B.; Porsteinsson, A.P.; Friedrich, S.; Dean, R.A.; Gonzales, C.; Sethuraman, G.; et al. Safety and biomarker effects of solanezumab in patients with Alzheimer’s disease. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2012, 8, 261–271. [Google Scholar] [CrossRef]
- Mills, S.M.; Mallmann, J.; Santacruz, A.M.; Fuqua, A.; Carril, M.; Aisen, P.S.; Althage, M.C.; Belyew, S.; Benzinger, T.L.; Brooks, W.S.; et al. Preclinical trials in autosomal dominant AD: Implementation of the DIAN-TU trial. Rev. Neurol. 2013, 169, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Pozo, A.; Frosch, M.P.; Masliah, E.; Hyman, B.T. Neuropathological alterations in Alzheimer disease. Cold Spring Harb. Perspect. Med. 2011, 1, a006189. [Google Scholar] [CrossRef]
- Numakawa, T.; Kajihara, R. Neurotrophins and Other Growth Factors in the Pathogenesis of Alzheimer’s Disease. Life 2023, 13, 647. [Google Scholar] [CrossRef] [PubMed]
- Eavri, R.; Shepherd, J.; Welsh, C.A.; Flanders, G.H.; Bear, M.F.; Nedivi, E. Interneuron Simplification and Loss of Structural Plasticity as Markers of Aging-Related Functional Decline. J. Neurosci. 2018, 38, 8421–8432. [Google Scholar] [CrossRef]
- Motley, S.E.; Grossman, Y.S.; Janssen, W.G.M.; Baxter, M.G.; Rapp, P.R.; Dumitriu, D.; Morrison, J.H. Selective Loss of Thin Spines in Area 7a of the Primate Intraparietal Sulcus Predicts Age-Related Working Memory Impairment. J. Neurosci. 2018, 38, 10467–10478. [Google Scholar] [CrossRef]
- Morrison, J.H.; Baxter, M.G. The ageing cortical synapse: Hallmarks and implications for cognitive decline. Nat. Rev. Neurosci. 2012, 13, 240–250. [Google Scholar] [CrossRef]
- Colavitta, M.F.; Grasso, L.; Barrantes, F.J. Environmental Enrichment in murine models and its translation to human factors improving conditions in Alzheimer disease. J. Prev. Alzheimers Dis. 2023, 10, 287–300. [Google Scholar] [CrossRef]
- Tzioras, M.; McGeachan, R.I.; Durrant, C.S.; Spires-Jones, T.L. Synaptic degeneration in Alzheimer disease. Nat. Rev. Neurol. 2023, 19, 19–38. [Google Scholar] [CrossRef]
- von Bernhardi, R.; Bernhardi, L.E.; Eugenín, J. What Is Neural Plasticity? Adv. Exp. Med. Biol. 2017, 1015, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Stern, Y.; Arenaza-Urquijo, E.M.; Bartrés-Faz, D.; Belleville, S.; Cantilon, M.; Chetelat, G.; Ewers, M.; Franzmeier, N.; Kempermann, G.; Kremen, W.S.; et al. Whitepaper: Defining and investigating cognitive reserve, brain reserve, and brain maintenance. Alzheimers Dement. 2020, 16, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
- Kühn, S.; Lindenberger, U. Chapter 6. Research on Human Plasticity in Adulthood; Academic Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Puderbaugh, M.; Emmady, P.D. Neuroplasticity. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Koch, G.; Spampinato, D. Alzheimer disease and neuroplasticity. Handb. Clin. Neurol. 2022, 184, 473–479. [Google Scholar] [CrossRef]
- Grafman, J. Conceptualizing functional neuroplasticity. J. Commun. Disord. 2000, 33, 345–355, quiz 355–346. [Google Scholar] [CrossRef]
- Mercerón-Martínez, D.; Ibaceta-González, C.; Salazar, C.; Almaguer-Melian, W.; Bergado-Rosado, J.A.; Palacios, A.G. Alzheimer’s Disease, Neural Plasticity, and Functional Recovery. J. Alzheimers Dis. 2021, 82, S37–S50. [Google Scholar] [CrossRef] [PubMed]
- Gage, F.H. Structural plasticity of the adult brain. Dialogues Clin. Neurosci. 2004, 6, 135–141. [Google Scholar] [CrossRef]
- Fuchs, E.; Flügge, G. Adult neuroplasticity: More than 40 years of research. Neural. Plast. 2014, 2014, 541870. [Google Scholar] [CrossRef]
- Jackson, J.; Jambrina, E.; Li, J.; Marston, H.; Menzies, F.; Phillips, K.; Gilmour, G. Targeting the Synapse in Alzheimer’s Disease. Front. Neurosci. 2019, 13, 735. [Google Scholar] [CrossRef] [PubMed]
- Fide, E.; Hünerli-Gündüz, D.; Öztura, İ.; Yener, G.G. Hyperconnectivity matters in early-onset Alzheimer’s disease: A resting-state EEG connectivity study. Neurophysiol. Clin. 2022, 52, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Glasgow, S.D.; McPhedrain, R.; Madranges, J.F.; Kennedy, T.E.; Ruthazer, E.S. Approaches and Limitations in the Investigation of Synaptic Transmission and Plasticity. Front. Synaptic Neurosci. 2019, 11, 20. [Google Scholar] [CrossRef] [PubMed]
- Smolen, P. A model of late long-term potentiation simulates aspects of memory maintenance. PLoS ONE 2007, 2, e445. [Google Scholar] [CrossRef] [PubMed]
- Massey, P.V.; Bashir, Z.I. Long-term depression: Multiple forms and implications for brain function. Trends Neurosci. 2007, 30, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Collingridge, G.L.; Peineau, S.; Howland, J.G.; Wang, Y.T. Long-term depression in the CNS. Nat. Rev. Neurosci. 2010, 11, 459–473. [Google Scholar] [CrossRef]
- Krebs, C.; Brill, E.; Minkova, L.; Federspiel, A.; Kellner-Weldon, F.; Wyss, P.; Teunissen, C.E.; van Harten, A.C.; Seydell-Greenwald, A.; Klink, K.; et al. Investigating Compensatory Brain Activity in Older Adults with Subjective Cognitive Decline. J. Alzheimers Dis. 2023, 93, 107–124. [Google Scholar] [CrossRef]
- Fernández-Ballesteros, R.; Zamarrón, M.D.; Tárraga, L.; Moya, R.; Iñiguez, J. Cognitive plasticity in healthy, mild cognitive impairment (MCI) subjects and Alzheimer’s disease patients: A research project in Spain. Eur. Psychol. 2003, 8, 148–159. [Google Scholar] [CrossRef]
- Wield, K.H.; Wienobost, J.; Schöttke, H. Estimating rehabilitation potential in schizophrenic subjects. In The Treatment of Schizophrenia: Status and Emerging Trends; Hogrefe & Huber Pub: Cambridge, MA, USA, 2001; pp. 88–103. [Google Scholar]
- Visser, P.J.; Reus, L.M.; Gobom, J.; Jansen, I.; Dicks, E.; van der Lee, S.J.; Tsolaki, M.; Verhey, F.R.J.; Popp, J.; Martinez-Lage, P.; et al. Cerebrospinal fluid tau levels are associated with abnormal neuronal plasticity markers in Alzheimer’s disease. Mol. Neurodegener. 2022, 17, 27. [Google Scholar] [CrossRef]
- Warpechowski, M.; Warpechowski, J.; Kulczyńska-Przybik, A.; Mroczko, B. Biomarkers of Activity-Dependent Plasticity and Persistent Enhancement of Synaptic Transmission in Alzheimer Disease: A Review of the Current Status. Med. Sci. Monit. 2023, 29, e938826. [Google Scholar] [CrossRef]
- Elahi, F.M.; Casaletto, K.B.; La Joie, R.; Walters, S.M.; Harvey, D.; Wolf, A.; Edwards, L.; Rivera-Contreras, W.; Karydas, A.; Cobigo, Y.; et al. Plasma biomarkers of astrocytic and neuronal dysfunction in early- and late-onset Alzheimer’s disease. Alzheimers Dement. 2020, 16, 681–695. [Google Scholar] [CrossRef]
- Karami, A.; Darreh-Shori, T.; Schultzberg, M.; Eriksdotter, M. CSF and Plasma Cholinergic Markers in Patients with Cognitive Impairment. Front. Aging Neurosci. 2021, 13, 704583. [Google Scholar] [CrossRef] [PubMed]
- Díez-Guerra, F.J. Neurogranin, a link between calcium/calmodulin and protein kinase C signaling in synaptic plasticity. IUBMB Life 2010, 62, 597–606. [Google Scholar] [CrossRef]
- Thorsell, A.; Bjerke, M.; Gobom, J.; Brunhage, E.; Vanmechelen, E.; Andreasen, N.; Hansson, O.; Minthon, L.; Zetterberg, H.; Blennow, K. Neurogranin in cerebrospinal fluid as a marker of synaptic degeneration in Alzheimer’s disease. Brain Res. 2010, 1362, 13–22. [Google Scholar] [CrossRef]
- Irfan, M.; Gopaul, K.R.; Miry, O.; Hökfelt, T.; Stanton, P.K.; Bark, C. SNAP-25 isoforms differentially regulate synaptic transmission and long-term synaptic plasticity at central synapses. Sci. Rep. 2019, 9, 6403. [Google Scholar] [CrossRef] [PubMed]
- Brinkmalm, A.; Brinkmalm, G.; Honer, W.; Frölich, L.; Hausner, L.; Minthon, L.; Hansson, O.; Wallin, A.; Zetterberg, H.; Blennow, K.; et al. SNAP-25 is a promising novel cerebrospinal fluid biomarker for synapse degeneration in Alzheimer’s disease. Mol. Neurodegener. 2014, 9, 53. [Google Scholar] [CrossRef]
- Tyler, W.J.; Alonso, M.; Bramham, C.R.; Pozzo-Miller, L.D. From acquisition to consolidation: On the role of brain-derived neurotrophic factor signaling in hippocampal-dependent learning. Learn. Mem. 2002, 9, 224–237. [Google Scholar] [CrossRef]
- Laske, C.; Stransky, E.; Leyhe, T.; Eschweiler, G.; Wittorf, A.; Richartz, E.; Bartels, M.; Buchkremer, G.; Schott, K. Stage-dependent BDNF serum concentrations in Alzheimer’s disease. J. Neural Transm. 2006, 113, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Yasutake, C.; Kuroda, K.; Yanagawa, T.; Okamura, T.; Yoneda, H. Serum BDNF, TNF-α and IL-1β levels in dementia patients: Comparison between Alzheimer’s disease and vascular dementia. Eur. Arch. Psychiatry Clin. Neurosci. 2006, 256, 402–406. [Google Scholar] [CrossRef]
- Matthews, P. Brain plasticity and clinical applications of fMRI. Riv. Neuroradiol. 2005, 18, 493–501. [Google Scholar] [CrossRef]
- Luo, Y.; Sun, T.; Ma, C.; Zhang, X.; Ji, Y.; Fu, X.; Ni, H. Alterations of Brain Networks in Alzheimer’s Disease and Mild Cognitive Impairment: A Resting State fMRI Study Based on a Population-specific Brain Template. Neuroscience 2021, 452, 192–207. [Google Scholar] [CrossRef]
- Holtzman, D.; Morris, J.; Goate, A. Alzheimer’s disease: The challenge of the second century. Sci. Transl. Med. 2011, 3, 77sr1. [Google Scholar] [CrossRef] [PubMed]
- Selkoe, D.J. Deciphering Alzheimer’s disease: The amyloid precursor protein yields new clues. Science 1990, 248, 1058–1060. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.A.; Higgins, G.A. Alzheimer’s disease: The amyloid cascade hypothesis. Science 1992, 256, 184–185. [Google Scholar] [CrossRef] [PubMed]
- Babic, T. The cholinergic hypothesis of Alzheimer’s disease: A review of progress. J. Neurol. Neurosurg. Psychiatry 1999, 67, 558. [Google Scholar] [CrossRef]
- Francis, P.T.; Palmer, A.M.; Snape, M.; Wilcock, G.K. The cholinergic hypothesis of Alzheimer’s disease: A review of progress. J. Neurol. Neurosurg. Psychiatry 1999, 66, 137–147. [Google Scholar] [CrossRef]
- Selkoe, D.J. Alzheimer’s disease is a synaptic failure. Science 2002, 298, 789–791. [Google Scholar] [CrossRef]
- Masliah, E.; Mallory, M.; Alford, M.; DeTeresa, R.; Hansen, L.; McKeel, D.; Morris, J. Altered expression of synaptic proteins occurs early during progression of Alzheimer’s disease. Neurology 2001, 56, 127–129. [Google Scholar] [CrossRef]
- Koffie, R.M.; Hyman, B.T.; Spires-Jones, T.L. Alzheimer’s disease: Synapses gone cold. Mol. Neurodegener. 2011, 6, 63. [Google Scholar] [CrossRef]
- Lanz, T.; Carter, D.; Merchant, K. Dendritic spine loss in the hippocampus of young PDAPP and Tg2576 mice and its prevention by the ApoE2 genotype. Neurobiol. Dis. 2003, 13, 246–253. [Google Scholar] [CrossRef]
- Walsh, D.M.; Klyubin, I.; Fadeeva, J.V.; Cullen, W.K.; Anwyl, R.; Wolfe, M.S.; Rowan, M.J.; Selkoe, D.J. Naturally secreted oligomers of amyloid β protein potently inhibit hippocampal long-term potentiation in vivo. Nature 2002, 416, 535–539. [Google Scholar] [CrossRef]
- Li, S.; Hong, S.; Shepardson, N.E.; Walsh, D.M.; Shankar, G.M.; Selkoe, D. Soluble oligomers of amyloid Beta protein facilitate hippocampal long-term depression by disrupting neuronal glutamate uptake. Neuron 2009, 62, 788–801. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Homma, K.J.; Poo, M.M. Shrinkage of dendritic spines associated with long-term depression of hippocampal synapses. Neuron 2004, 44, 749–757. [Google Scholar] [CrossRef]
- Selkoe, D.J. Soluble oligomers of the amyloid beta-protein impair synaptic plasticity and behavior. Behav. Brain Res. 2008, 192, 106–113. [Google Scholar] [CrossRef]
- Bäckman, L.; Andersson, J.; Nyberg, L.; Winblad, B.; Nordberg, A.; Almkvist, O. Brain regions associated with episodic retrieval in normal aging and Alzheimer’s disease. Neurology 1999, 52, 1861. [Google Scholar] [CrossRef]
- Terry, R.D.; Masliah, E.; Salmon, D.P.; Butters, N.; DeTeresa, R.; Hill, R.; Hansen, L.A.; Katzman, R. Physical basis of cognitive alterations in Alzheimer’s disease: Synapse loss is the major correlate of cognitive impairment. Ann. Neurol. Off. J. Am. Neurol. Assoc. Child Neurol. Soc. 1991, 30, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Bekdash, R.A. The Cholinergic System, the Adrenergic System and the Neuropathology of Alzheimer’s Disease. Int. J. Mol. Sci. 2021, 22, 1273. [Google Scholar] [CrossRef]
- de Aquino, C. Methodological issued in randomized clinical trials for prodromal Alzheimer’s and Parkinson’s disease. Front. Neurol. 2021, 12, 694329. [Google Scholar] [CrossRef]
- Medications for Memory, Cognition and Dementia-Related Behaviors. Available online: https://www.alz.org/alzheimers-dementia/treatments/medications-for-memory?lang=en-US (accessed on 7 July 2023).
- Fink, H.A.; Hemmy, L.S.; Linskens, E.J.; Silverman, P.C.; MacDonald, R.; McCarten, R.; Talley, K.M.; Desai, P.J.; Forte, M.L.; Miller, M.A.; et al. AHRQ comparative efectiveness reviews. In Diagnosis and Treatment of Clinical Alzheimer’s Type Dementia: A Systematic Review; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2020. [Google Scholar]
- Watermeyer, T.; Calia, C. Neuropsychological assessment in preclinical and prodromal Alzheimer disease: A global perspective. J. Glob. Health 2019, 9, 010317. [Google Scholar] [CrossRef] [PubMed]
- Glynn-Servedio, B.E.; Ranola, T.S. AChE Inhibitors and NMDA Receptor Antagonists in Advanced Alzheimer’s Disease. Consult. Pharm. J. Am. Soc. Consult. Pharm. 2017, 32, 511–518. [Google Scholar] [CrossRef]
- Athar, T.; Al Balushi, K.; Khan, S.A. Recent advances on drug development and emerging therapeutic agents for Alzheimer’s disease. Mol. Biol. Rep. 2021, 48, 5629–5645. [Google Scholar] [CrossRef]
- Nordberg, A.; Lilja, A.; Lundqvist, H.; Hartvig, P.; Amberla, K.; Viitanen, M.; Warpman, U.; Johansson, M.; Hellström-Lindahl, E.; Bjurling, P.; et al. Tacrine restores cholinergic nicotinic receptors and glucose metabolism in Alzheimer patients as visualized by positron emission tomography. Neurobiol. Aging 1992, 13, 747–758. [Google Scholar] [CrossRef] [PubMed]
- Auld, D.S.; Kornecook, T.J.; Bastianetto, S.; Quirion, R. Alzheimer’s disease and the basal forebrain cholinergic system: Relations to beta-amyloid peptides, cognition, and treatment strategies. Prog. Neurobiol. 2002, 68, 209–245. [Google Scholar] [CrossRef]
- Sevigny, J.; Chiao, P.; Bussière, T.; Weinreb, P.H.; Williams, L.; Maier, M.; Dunstan, R.; Salloway, S.; Chen, T.; Ling, Y.; et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature 2016, 537, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Reiman, E.M. Alzheimer’s disease: Attack on amyloid-[beta] protein. Nature 2016, 537, 36–37. [Google Scholar] [CrossRef]
- Musiek, E.S.; Morris, J.C. Possible Consequences of the Approval of a Disease-Modifying Therapy for Alzheimer Disease. JAMA Neurol. 2020, 78, 141–142. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, R.N.; Lambracht-Washington, D. Active Immunotherapy to Prevent Alzheimer Disease—A DNA Amyloid β 1-42 Trimer Vaccine. JAMA Neurol. 2020, 77, 289–290. [Google Scholar] [CrossRef]
- Cummings, J.; Lee, G.; Nahed, P.; Kambar, M.; Zhong, K.; Fonseca, J.; Taghva, K. Alzheimer’s disease drug development pipeline: 2022. Alzheimers Dement. 2022, 8, e12295. [Google Scholar] [CrossRef]
- Iliyasu, M.O.; Musa, S.A.; Oladele, S.B.; Iliya, A.I. Amyloid-beta aggregation implicates multiple pathways in Alzheimer’s disease: Understanding the mechanisms. Front. Neurosci. 2023, 17, 1081938. [Google Scholar] [CrossRef]
- Takada-Takatori, Y.; Kume, T.; Sugimoto, M.; Katsuki, H.; Sugimoto, H.; Akaike, A. Acetylcholinesterase inhibitors used in treatment of Alzheimer’s disease prevent glutamate neurotoxicity via nicotinic acetylcholine receptors and phosphatidylinositol 3-kinase cascade. Neuropharmacology 2006, 51, 474–486. [Google Scholar] [CrossRef]
- Wenk, G.L.; McGann, K.; Mencarelli, A.; Hauss-Wegrzyniak, B.; Del, S.P.; Fiorucci, S. Mechanisms to prevent the toxicity of chronic neuroinflammation on forebrain cholinergic neurons. Eur. J. Pharmacol. 2000, 402, 77–85. [Google Scholar] [CrossRef]
- Cheng, J.; Dong, Y.; Ma, J.; Pan, R.; Liao, Y.; Kong, X.; Li, X.; Li, S.; Chen, P.; Wang, L.; et al. Microglial Calhm2 regulates neuroinflammation and contributes to Alzheimer’s disease pathology. Sci. Adv. 2021, 7, eabe3600. [Google Scholar] [CrossRef]
- Guerrero, A.; De Strooper, B.; Arancibia-Cárcamo, I.L. Cellular senescence at the crossroads of inflammation and Alzheimer’s disease. Trends Neurosci. 2021, 44, 714–727. [Google Scholar] [CrossRef]
- Tyagi, E.; Agrawal, R.; Nath, C.; Shukla, R. Inhibitory role of cholinergic system mediated via {alpha}7 nicotinic acetylcholine receptor in LPS-induced neuro-inflammation. Innate Immun. 2009, 16, 3–13. [Google Scholar] [CrossRef]
- Borroni, V.; Barrantes, F.J. Homomeric and Heteromeric α7 Nicotinic Acetylcholine Receptors in Health and Some Central Nervous System Diseases. Membranes 2021, 11, 664. [Google Scholar] [CrossRef]
- Tyagi, E.; Agrawal, R.; Nath, C.; Shukla, R. Cholinergic protection via α7 nicotinic acetylcholine receptors and PI3K-Akt pathway in LPS-induced neuroinflammation. Neurochem. Int. 2010, 56, 135–142. [Google Scholar] [CrossRef]
- Perez-Lloret, S.; Peralta, M.C.; Barrantes, F.J. Pharmacotherapies for Parkinson’s disease symptoms related to cholinergic degeneration. Expert Opin. Pharmacother. 2016, 17, 2405–2415. [Google Scholar] [CrossRef]
- Dajas-Bailador, F.A.; Heimala, K.; Wonnacott, S. The allosteric potentiation of nicotinic acetylcholine receptors by galantamine is transduced into cellular responses in neurons: Ca2+ signals and neurotransmitter release. Mol. Pharmacol. 2003, 64, 1217–1226. [Google Scholar] [CrossRef]
- Pereira, E.F.; Hilmas, C.; Santos, M.D.; Alkondon, M.; Maelicke, A.; Albuquerque, E.X. Unconventional ligands and modulators of nicotinic receptors. J. Neurobiol. 2002, 53, 479–500. [Google Scholar] [CrossRef]
- Akk, G.; Steinbach, J.H. Galantamine activates muscle-type nicotinic acetylcholine receptors without binding to the acetylcholine-binding site. J. Neurosci. 2005, 25, 1992–2001. [Google Scholar] [CrossRef]
- Arias, E.; Alés, E.; Gabilan, N.H.; Cano-Abad, M.F.; Villarroya, M.; García, A.G.; López, M.G. Galantamine prevents apoptosis induced by β-amyloid and thapsigargin: Involvement of nicotinic acetylcholine receptors. Neuropharmacology 2004, 46, 103–114. [Google Scholar] [CrossRef]
- Wang, D.; Noda, Y.; Zhou, Y.; Mouri, A.; Mizoguchi, H.; Nitta, A.; Chen, W.; Nabeshima, T. The Allosteric Potentiation of Nicotinic Acetylcholine Receptors by Galantamine Ameliorates the Cognitive Dysfunction in Beta Amyloid(25-35) i.c.v.-Injected Mice: Involvement of Dopaminergic Systems. Neuropsychopharmacology 2006, 32, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Moriguchi, S.; Zhao, X.; Marszalec, W.; Yeh, J.Z.; Fukunaga, K.; Narahashi, T. Nefiracetam and galantamine modulation of excitatory and inhibitory synaptic transmission via stimulation of neuronal nicotinic acetylcholine receptors in rat cortical neurons. Neuroscience 2009, 160, 484–491. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.; Zheng, X.; Fang, T.; Yang, X.; Luo, X.; Guo, A.; Newell, K.A.; Huang, X.F.; Yu, Y. Galantamine improves cognition, hippocampal inflammation, and synaptic plasticity impairments induced by lipopolysaccharide in mice. J. Neuroinflammation 2018, 15, 112. [Google Scholar] [CrossRef]
- Macdonald, R.; Barnes, K.; Hastings, C.; Mortiboys, H. Mitochondrial abnormalities in Parkinson’s disease and Alzheimer’s disease: Can mitochondria be targeted therapeutically? Biochem. Soc. Trans. 2018, 46, 891–909. [Google Scholar] [CrossRef]
- Lewcock, J.W.; Schlepckow, K.; Di Paolo, G.; Tahirovic, S.; Monroe, K.M.; Haass, C. Emerging Microglia Biology Defines Novel Therapeutic Approaches for Alzheimer’s Disease. Neuron 2020, 108, 801–821. [Google Scholar] [CrossRef]
- Button, E.B.; Robert, J.; Caffrey, T.M.; Fan, J.; Zhao, W.; Wellington, C.L. HDL from an Alzheimer’s disease perspective. Curr. Opin. Lipidol. 2019, 30, 224–234. [Google Scholar] [CrossRef]
- Farlow, M.; Thompson, R.; Wei, L.-J.; Tuchman, A.; Grenier, E.; Crockford, D.; Wilke, S.; Benison, J.; Alkon, D. A Randomized, Double-Blind, Placebo-Controlled, Phase II Study Assessing Safety, Tolerability, and Efficacy of Bryostatin in the Treatment of Moderately Severe to Severe Alzheimer’s Disease. J. Alzheimer’s Dis. 2018, 67, 555–570. [Google Scholar] [CrossRef]
- Piscopo, P.; Crestini, A.; Carbone, E.; Rivabene, R.; Ancidoni, A.; Lo Giudice, M.; Corbo, M.; Vanacore, N.; Lacorte, E. A systematic review on drugs for synaptic plasticity in the treatment of dementia. Ageing Res. Rev. 2022, 81, 101726. [Google Scholar] [CrossRef]
- Hill, N.L.; Kolanowski, A.M.; Gill, D.J. Plasticity in Early Alzheimer’s Disease: An Opportunity for Intervention. Top. Geriatr. Rehabil. 2011, 27, 257–267. [Google Scholar] [CrossRef]
- Lazarov, O.; Hollands, C.; Bartolotti, N. Alzheimer’s disease and Hippocampal Adult Neurogenesis; Exploring Shared Mechanisms. Front. Neurosci. 2016, 10, 178. [Google Scholar] [CrossRef]
- De Felice, F.G.; Vieira, M.N.; Bomfim, T.R.; Decker, H.; Velasco, P.T.; Lambert, M.P.; Viola, K.L.; Zhao, W.Q.; Ferreira, S.T.; Klein, W.L. Protection of synapses against Alzheimer’s-linked toxins: Insulin signaling prevents the pathogenic binding of Abeta oligomers. Proc. Natl. Acad. Sci. USA 2009, 106, 1971–1976. [Google Scholar] [CrossRef] [PubMed]
- Edwards, G.A., III; Gamez, N.; Escobedo, G., Jr.; Calderon, O.; Moreno-Gonzalez, I. Modifiable Risk Factors for Alzheimer’s Disease. Front. Aging Neurosci. 2019, 11, 146. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Mahmood, Z.; Javed, A.; Hashmi, S.N.; Zerr, I.; Zafar, S.; Zahid, S. Effect of Metformin on Adult Hippocampal Neurogenesis: Comparison with Donepezil and Links to Cognition. J. Mol. Neurosci. 2017, 62, 88–98. [Google Scholar] [CrossRef]
- Ayaz, M.; Sadiq, A.; Junaid, M.; Ullah, F.; Subhan, F.; Ahmed, J. Neuroprotective and Anti-Aging Potentials of Essential Oils from Aromatic and Medicinal Plants. Front. Aging Neurosci. 2017, 9, 168. [Google Scholar] [CrossRef]
- Zhou, G.; Yuan-Lin Ma, J.; Chen, G. ARL6IP1 Mediates Small-molecule-induced Alleviation of Alzheimer Pathology Through FXR1-dependent BACE1 Translation Initiation. Proc. Natl. Acad. Sci. USA 2023, 120, e2220148120. [Google Scholar] [CrossRef] [PubMed]
- Brickman, A.; Yeung, L.; Alschuler, D.; Ottaviani, J.; Kuhnle, G.; Sloan, R.; Luttmann-Gibson, H.; Copeland, T.; Schroeter, H.; Sesso, H.; et al. Dietary flavonols restore hippocampal-dependent memory in older adults with lower diet quality and lower habitual flavonol consumption. Proc. Natl. Acad. Sci. USA 2023, 120, e2216932120. [Google Scholar] [CrossRef]
- Cummings, J.; Fox, N. Defining Disease Modifying Therapy for Alzheimer’s Disease. J. Prev. Alzheimers Dis. 2017, 4, 109–115. [Google Scholar] [CrossRef]
- Cummings, J.L.; Tong, G.; Ballard, C. Treatment Combinations for Alzheimer’s Disease: Current and Future Pharmacotherapy Options. J. Alzheimers Dis. 2019, 67, 779–794. [Google Scholar] [CrossRef]
- Tong, M.; Deochand, C.; Didsbury, J.; de la Monte, S.M. T3D-959: A Multi-Faceted Disease Remedial Drug Candidate for the Treatment of Alzheimer’s Disease. J. Alzheimers Dis. 2016, 51, 123–138. [Google Scholar] [CrossRef]
- Chamberlain, S.; Gabriel, H.; Strittmatter, W.; Didsbury, J. An Exploratory Phase IIa Study of the PPAR delta/gamma Agonist T3D-959 Assessing Metabolic and Cognitive Function in Subjects with Mild to Moderate Alzheimer’s Disease. J. Alzheimers Dis. 2020, 73, 1085–1103. [Google Scholar] [CrossRef]
- Li, S.; Selkoe, D.J. A mechanistic hypothesis for the impairment of synaptic plasticity by soluble Aβ oligomers from Alzheimer’s brain. J. Neurochem. 2020, 154, 583–597. [Google Scholar] [CrossRef]
- Cummings, J.; Ortiz, A.; Castellino, J.; Kinney, J. Diabetes: Risk factor and translational therapeutic implications for Alzheimer’s disease. Eur. J. Neurosci. 2022, 56, 5727–5757. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Reddy, P.H. Role of Glutamate and NMDA Receptors in Alzheimer’s Disease. J. Alzheimers Dis. 2017, 57, 1041–1048. [Google Scholar] [CrossRef] [PubMed]
- Al-Horani, R.A. Riluzole and its prodrugs for the treatment of Alzheimer’s disease. Pharm. Pat. Anal. 2023, 12, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.C.; Mao, X.; Dowd, K.; Tsakanikas, D.; Jiang, C.S.; Meuser, C.; Andrews, R.D.; Lukic, A.S.; Lee, J.; Hampilos, N.; et al. Riluzole, a glutamate modulator, slows cerebral glucose metabolism decline in patients with Alzheimer’s disease. Brain 2021, 144, 3742–3755. [Google Scholar] [CrossRef]
- Xiang, Y.; Xin, J.; Le, W.; Yang, Y. Neurogranin: A Potential Biomarker of Neurological and Mental Diseases. Front. Aging Neurosci. 2020, 12, 584743. [Google Scholar] [CrossRef]
- Wang, H.Y.; Pei, Z.; Lee, K.C.; Lopez-Brignoni, E.; Nikolov, B.; Crowley, C.A.; Marsman, M.R.; Barbier, R.; Friedmann, N.; Burns, L.H. PTI-125 Reduces Biomarkers of Alzheimer’s Disease in Patients. J. Prev. Alzheimers Dis. 2020, 7, 256–264. [Google Scholar] [CrossRef]
- Tormählen, N.M.; Martorelli, M.; Kuhn, A.; Maier, F.; Guezguez, J.; Burnet, M.; Albrecht, W.; Laufer, S.A.; Koch, P. Design and Synthesis of Highly Selective Brain Penetrant p38α Mitogen-Activated Protein Kinase Inhibitors. J. Med. Chem. 2022, 65, 1225–1242. [Google Scholar] [CrossRef]
- Prins, N.D.; Harrison, J.E.; Chu, H.M.; Blackburn, K.; Alam, J.J.; Scheltens, P. A phase 2 double-blind placebo-controlled 24-week treatment clinical study of the p38 alpha kinase inhibitor neflamapimod in mild Alzheimer’s disease. Alzheimers Res. Ther. 2021, 13, 106. [Google Scholar] [CrossRef]
- Schupp, M.; Malsam, J.; Ruiter, M.; Scheutzow, A.; Wierda, K.D.; Söllner, T.H.; Sørensen, J.B. Interactions Between SNAP-25 and Synaptotagmin-1 Are Involved in Vesicle Priming, Clamping Spontaneous and Stimulating Evoked Neurotransmission. J. Neurosci. 2016, 36, 11865–11880. [Google Scholar] [CrossRef]
- Zhang, F.-X.; Sun, Q.-J.; Zheng, X.-Y.; Lin, Y.-T.; Shang, W.; Wang, A.-H.; Duan, R.-S.; Chi, Z.-F. Abnormal Expression of Synaptophysin, SNAP-25, and Synaptotagmin 1 in the Hippocampus of Kainic Acid-Exposed Rats with Behavioral Deficits. Cell. Mol. Neurobiol. 2014, 34, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Nct. Effect of CT1812 Treatment on Brain Synaptic Density. 2018. Available online: https://clinicaltrials.gov/show/NCT03493282 (accessed on 23 June 2023).
- Catalano, S.M.; Schneider, L.S.; DeKosky, S.; Morgan, R.; Rehak, C.; Silky, C.; Mozzoni, K.; Izzo, N.J.; Grundman, M.; Schirm, M.; et al. CT1812 exhibits a synaptoprotective effect in Alzheimer’s disease patients. Neurotherapeutics 2018, 15, 819–820. [Google Scholar]
- Rishton, G.M.; Look, G.C.; Ni, Z.J.; Zhang, J.; Wang, Y.; Huang, Y.; Wu, X.; Izzo, N.J.; LaBarbera, K.M.; Limegrover, C.S.; et al. Discovery of Investigational Drug CT1812, an Antagonist of the Sigma-2 Receptor Complex for Alzheimer’s Disease. ACS Med. Chem. Lett. 2021, 12, 1389–1395. [Google Scholar] [CrossRef] [PubMed]
- Izzo, N.J.; Yuede, C.M.; LaBarbera, K.M.; Limegrover, C.S.; Rehak, C.; Yurko, R.; Waybright, L.; Look, G.; Rishton, G.; Safferstein, H.; et al. Preclinical and clinical biomarker studies of CT1812: A novel approach to Alzheimer’s disease modification. Alzheimers Dement. 2021, 17, 1365–1382. [Google Scholar] [CrossRef]
- Alam, J.; Blackburn, K.; Patrick, D. Neflamapimod: Clinical Phase 2b-Ready Oral Small Molecule Inhibitor of p38α to Reverse Synaptic Dysfunction in Early Alzheimer’s Disease. J. Prev. Alzheimers Dis. 2017, 4, 273–278. [Google Scholar] [CrossRef]
- Nussbaum, J.M.; Schilling, S.; Cynis, H.; Silva, A.; Swanson, E.; Wangsanut, T.; Tayler, K.; Wiltgen, B.; Hatami, A.; Rönicke, R.; et al. Prion-like behaviour and tau-dependent cytotoxicity of pyroglutamylated amyloid-β. Nature 2012, 485, 651–655. [Google Scholar] [CrossRef]
- Scheltens, P.; Hallikainen, M.; Grimmer, T.; Duning, T.; Gouw, A.A.; Teunissen, C.E.; Wink, A.M.; Maruff, P.; Harrison, J.; van Baal, C.M.; et al. Safety, tolerability and efficacy of the glutaminyl cyclase inhibitor PQ912 in Alzheimer’s disease: Results of a randomized, double-blind, placebo-controlled phase 2a study. Alzheimers Res. Ther. 2018, 10, 107. [Google Scholar] [CrossRef]
- Kempermann, G. Environmental enrichment, new neurons and the neurobiology of individuality. Nat. Rev. Neurosci. 2019, 20, 235–245. [Google Scholar] [CrossRef]
- Lauretti, E.; Li, J.G.; Di Meco, A.; Praticò, D. Glucose deficit triggers tau pathology and synaptic dysfunction in a tauopathy mouse model. Transl. Psychiatry 2017, 7, e1020. [Google Scholar] [CrossRef]
- de la Monte, S.M. Insulin resistance and Alzheimer’s disease. BMB Rep. 2009, 42, 475–481. [Google Scholar] [CrossRef]
- Batista, A.F.; Forny-Germano, L.; Clarke, J.R.; Lyra, E.S.N.M.; Brito-Moreira, J.; Boehnke, S.E.; Winterborn, A.; Coe, B.C.; Lablans, A.; Vital, J.F.; et al. The diabetes drug liraglutide reverses cognitive impairment in mice and attenuates insulin receptor and synaptic pathology in a non-human primate model of Alzheimer’s disease. J. Pathol. 2018, 245, 85–100. [Google Scholar] [CrossRef] [PubMed]
- Pilipenko, V.; Narbute, K.; Pupure, J.; Langrate, I.K.; Muceniece, R.; Kluša, V. Neuroprotective potential of antihyperglycemic drug metformin in streptozocin-induced rat model of sporadic Alzheimer’s disease. Eur. J. Pharmacol. 2020, 881, 173290. [Google Scholar] [CrossRef] [PubMed]
- Dong, Q.; Teng, S.W.; Wang, Y.; Qin, F.; Li, Y.; Ai, L.L.; Yu, H. Sitagliptin protects the cognition function of the Alzheimer’s disease mice through activating glucagon-like peptide-1 and BDNF-TrkB signalings. Neurosci. Lett. 2019, 696, 184–190. [Google Scholar] [CrossRef]
- Bomba, M.; Granzotto, A.; Castelli, V.; Onofrj, M.; Lattanzio, R.; Cimini, A.; Sensi, S.L. Exenatide Reverts the High-Fat-Diet-Induced Impairment of BDNF Signaling and Inflammatory Response in an Animal Model of Alzheimer’s Disease. J. Alzheimers Dis. 2019, 70, 793–810. [Google Scholar] [CrossRef]
- Yan, W.; Guo, T.; Liu, N.; Cui, X.; Wei, X.; Sun, Y.; Hu, H.; Chen, L. Erythropoietin ameliorates cognitive deficits by improving hippocampal and synaptic damage in streptozotocin-induced diabetic mice. Cell Signal. 2023, 106, 110614. [Google Scholar] [CrossRef]
- Zhang, Z.Q.; Hölscher, C. GIP has neuroprotective effects in Alzheimer and Parkinson’s disease models. Peptides 2020, 125, 170184. [Google Scholar] [CrossRef]
- Yuan, L.; Zhang, J.; Guo, J.H.; Holscher, C.; Yang, J.T.; Wu, M.N.; Wang, Z.J.; Cai, H.Y.; Han, L.N.; Shi, H.; et al. DAla2-GIP-GLU-PAL Protects Against Cognitive Deficits and Pathology in APP/PS1 Mice by Inhibiting Neuroinflammation and Upregulating cAMP/PKA/CREB Signaling Pathways. J. Alzheimers Dis. 2021, 80, 695–713. [Google Scholar] [CrossRef] [PubMed]
- Faivre, E.; Hölscher, C. D-Ala2GIP facilitated synaptic plasticity and reduces plaque load in aged wild type mice and in an Alzheimer’s disease mouse model. J. Alzheimers Dis. 2013, 35, 267–283. [Google Scholar] [CrossRef]
- Faivre, E.; Hamilton, A.; Hölscher, C. Effects of acute and chronic administration of GIP analogues on cognition, synaptic plasticity and neurogenesis in mice. Eur. J. Pharmacol. 2012, 674, 294–306. [Google Scholar] [CrossRef]
- Deng, C.; Meng, Z.; Chen, H.; Meng, S. Tetramethylpyrazine ameliorates systemic streptozotocin-induced Alzheimer-like pathology. J. Chem. Neuroanat. 2023, 127, 102207. [Google Scholar] [CrossRef]
- Panja, D.; Kenney, J.W.; D’Andrea, L.; Zalfa, F.; Vedeler, A.; Wibrand, K.; Fukunaga, R.; Bagni, C.; Proud, C.G.; Bramham, C.R. Two-stage translational control of dentate gyrus LTP consolidation is mediated by sustained BDNF-TrkB signaling to MNK. Cell Rep. 2014, 9, 1430–1445. [Google Scholar] [CrossRef] [PubMed]
- Fortin, D.A.; Srivastava, T.; Dwarakanath, D.; Pierre, P.; Nygaard, S.; Derkach, V.A.; Soderling, T.R. Brain-derived neurotrophic factor activation of CaM-kinase kinase via transient receptor potential canonical channels induces the translation and synaptic incorporation of GluA1-containing calcium-permeable AMPA receptors. J. Neurosci. 2012, 32, 8127–8137. [Google Scholar] [CrossRef] [PubMed]
- Briz, V.; Liu, Y.; Zhu, G.; Bi, X.; Baudry, M. A novel form of synaptic plasticity in field CA3 of hippocampus requires GPER1 activation and BDNF release. J. Cell Biol. 2015, 210, 1225–1237. [Google Scholar] [CrossRef]
- Vallés, A.S.; Borroni, M.V.; Barrantes, F.J. Targeting brain α7 nicotinic acetylcholine receptors in Alzheimer’s disease: Rationale and current status. CNS Drugs 2014, 28, 975–987. [Google Scholar] [CrossRef] [PubMed]
- Vallés, A.S.; Barrantes, F.J. Dysregulation of neuronal nicotinic acetylcholine receptor–cholesterol crosstalk in autism spectrum disorder. Front. Mol. Neurosci. 2021, 14, 744597. [Google Scholar] [CrossRef]
- Baazaoui, N.; Iqbal, K. Alzheimer’s Disease: Challenges and a Therapeutic Opportunity to Treat It with a Neurotrophic Compound. Biomolecules 2022, 12, 1409. [Google Scholar] [CrossRef]
- Sangiovanni, E.; Brivio, P.; Dell’Agli, M.; Calabrese, F. Botanicals as Modulators of Neuroplasticity: Focus on BDNF. Neural Plast. 2017, 2017, 5965371. [Google Scholar] [CrossRef]
- Flores, I.O.; Treviño, S.; Díaz, A. Neurotrophic fragments as therapeutic alternatives to ameliorate brain aging. Neural Regen. Res. 2023, 18, 51–56. [Google Scholar] [CrossRef]
- Gao, L.; Zhang, Y.; Sterling, K.; Song, W. Brain-derived neurotrophic factor in Alzheimer’s disease and its pharmaceutical potential. Transl. Neurodegener. 2022, 11, 4. [Google Scholar] [CrossRef]
- Kazim, S.F.; Blanchard, J.; Dai, C.L.; Tung, Y.C.; LaFerla, F.M.; Iqbal, I.G.; Iqbal, K. Disease modifying effect of chronic oral treatment with a neurotrophic peptidergic compound in a triple transgenic mouse model of Alzheimer’s disease. Neurobiol. Dis. 2014, 71, 110–130. [Google Scholar] [CrossRef]
- Wei, W.; Liu, Y.; Dai, C.L.; Baazaoui, N.; Tung, Y.C.; Liu, F.; Iqbal, K. Neurotrophic Treatment Initiated During Early Postnatal Development Prevents the Alzheimer-Like Behavior and Synaptic Dysfunction. J. Alzheimers Dis. 2021, 82, 631–646. [Google Scholar] [CrossRef]
- Bolognin, S.; Buffelli, M.; Puoliväli, J.; Iqbal, K. Rescue of cognitive-aging by administration of a neurogenic and/or neurotrophic compound. Neurobiol. Aging 2014, 35, 2134–2146. [Google Scholar] [CrossRef] [PubMed]
- Rockenstein, E.; Mante, M.; Adame, A.; Crews, L.; Moessler, H.; Masliah, E. Effects of Cerebrolysin on neurogenesis in an APP transgenic model of Alzheimer’s disease. Acta Neuropathol. 2007, 113, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Rockenstein, E.; Adame, A.; Mante, M.; Moessler, H.; Windisch, M.; Masliah, E. The neuroprotective effects of Cerebrolysin in a transgenic model of Alzheimer’s disease are associated with improved behavioral performance. J. Neural Transm. 2003, 110, 1313–1327. [Google Scholar] [CrossRef] [PubMed]
- Parrado-Fernandez, C.; Juric, S.; Backlund, M.; Dahlström, M.; Madjid, N.; Lidell, V.; Rasti, A.; Sandin, J.; Nordvall, G.; Forsell, P. Neuroprotective and Disease Modifying Effects of the Triazinetrione ACD856, a Positive Allosteric Modulator of Trk-Receptors for the Treatment of Cognitive Dysfunction in Alzheimer’s Disease. Int. J. Mol. Sci. 2023, 24, 11159. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zou, Y.; Wang, L. Neurotransmitters in Prevention and Treatment of Alzheimer’s Disease. Int. J. Mol. Sci. 2023, 24, 3841. [Google Scholar] [CrossRef]
- Anand, R.; Gill, K.D.; Mahdi, A.A. Therapeutics of Alzheimer’s disease: Past, present and future. Neuropharmacology 2014, 76 Pt A, 27–50. [Google Scholar] [CrossRef]
- Kandimalla, R.; Reddy, P.H. Therapeutics of Neurotransmitters in Alzheimer’s Disease. J. Alzheimer’s Dis. JAD 2017, 57, 1049–1069. [Google Scholar] [CrossRef]
- Kabir, M.T.; Sufian, M.A.; Uddin, M.S.; Begum, M.M.; Akhter, S.; Islam, A.; Mathew, B.; Islam, M.S.; Amran, M.S.; Md Ashraf, G. NMDA Receptor Antagonists: Repositioning of Memantine as a Multitargeting Agent for Alzheimer’s Therapy. Curr. Pharm. Des. 2019, 25, 3506–3518. [Google Scholar] [CrossRef]
- Lipton, S.A. Pathologically activated therapeutics for neuroprotection. Nat. Rev. Neurosci. 2007, 8, 803–808. [Google Scholar] [CrossRef]
- Creeley, C.; Wozniak, D.F.; Labruyere, J.; Taylor, G.T.; Olney, J.W. Low doses of memantine disrupt memory in adult rats. J. Neurosci. Off. J. Soc. Neurosci. 2006, 26, 3923–3932. [Google Scholar] [CrossRef]
- Morè, L.; Gravius, A.; Nagel, J.; Valastro, B.; Greco, S.; Danysz, W. Therapeutically relevant plasma concentrations of memantine produce significant L-N-methyl-D-aspartate receptor occupation and do not impair learning in rats. Behav. Pharmacol. 2008, 19, 724–734. [Google Scholar] [CrossRef]
- Klyubin, I.; Wang, Q.; Reed, M.N.; Irving, E.A.; Upton, N.; Hofmeister, J.; Cleary, J.P.; Anwyl, R.; Rowan, M.J. Protection against Aβ-mediated rapid disruption of synaptic plasticity and memory by memantine. Neurobiol. Aging 2011, 32, 614–623. [Google Scholar] [CrossRef]
- Hu, N.W.; Klyubin, I.; Anwyl, R.; Rowan, M.J. GluN2B subunit-containing NMDA receptor antagonists prevent Abeta-mediated synaptic plasticity disruption in vivo. Proc. Natl. Acad. Sci. USA 2009, 106, 20504–20509. [Google Scholar] [CrossRef]
- Kume, T.; Sugimoto, M.; Takada, Y.; Yamaguchi, T.; Yonezawa, A.; Katsuki, H.; Sugimoto, H.; Akaike, A. Up-regulation of nicotinic acetylcholine receptors by central-type acetylcholinesterase inhibitors in rat cortical neurons. Eur. J. Pharmacol. 2005, 527, 77–85. [Google Scholar] [CrossRef]
- Unzeta, M.; Esteban, G.; Bolea, I.; Fogel, W.A.; Ramsay, R.R.; Youdim, M.B.H.; Tipton, K.F.; Marco-Contelles, J. Multi-Target Directed Donepezil-Like Ligands for Alzheimer’s Disease. Front. Neurosci. 2016, 10, 205. [Google Scholar] [CrossRef]
- Aranda-Abreu, G.E.; Hernández-Aguilar, M.E.; Manzo Denes, J.; García Hernández, L.I.; Herrera Rivero, M. Rehabilitating a brain with Alzheimer’s: A proposal. Clin. Interv. Aging 2011, 6, 53–59. [Google Scholar] [CrossRef]
- Muñoz-Torrero, D. Acetylcholinesterase inhibitors as disease-modifying therapies for Alzheimer’s disease. Curr. Med. Chem. 2008, 15, 2433–2455. [Google Scholar] [CrossRef]
- Cummings, J.L.; Morstorf, T.; Zhong, K. Alzheimer’s disease drug-development pipeline: Few candidates, frequent failures. Alzheimers Res. Ther. 2014, 6, 37. [Google Scholar] [CrossRef]
- Li, K.; Jiang, Y.; Li, G.; Liu, T.; Yang, Z. Novel Multitarget Directed Tacrine Hybrids as Anti-Alzheimer’s Compounds Improved Synaptic Plasticity and Cognitive Impairment in APP/PS1 Transgenic Mice. ACS Chem. Neurosci. 2020, 11, 4316–4328. [Google Scholar] [CrossRef]
- Alcántara-González, F.; Mendoza-Perez, C.R.; Zaragoza, N.; Juarez, I.; Arroyo-García, L.E.; Gamboa, C.; De La Cruz, F.; Zamudio, S.; Garcia-Dolores, F.; Flores, G. Combined administration of cerebrolysin and donepezil induces plastic changes in prefrontal cortex in aged mice. Synapse 2012, 66, 938–949. [Google Scholar] [CrossRef]
- Srivareerat, M.; Tran, T.T.; Salim, S.; Aleisa, A.M.; Alkadhi, K.A. Chronic nicotine restores normal Aβ levels and prevents short-term memory and E-LTP impairment in Aβ rat model of Alzheimer’s disease. Neurobiol. Aging 2011, 32, 834–844. [Google Scholar] [CrossRef]
- Esteves, I.M.; Lopes-Aguiar, C.; Rossignoli, M.T.; Ruggiero, R.N.; Broggini, A.C.S.; Bueno-Junior, L.S.; Kandratavicius, L.; Monteiro, M.R.; Romcy-Pereira, R.N.; Leite, J.P. Chronic nicotine attenuates behavioral and synaptic plasticity impairments in a streptozotocin model of Alzheimer’s disease. Neuroscience 2017, 353, 87–97. [Google Scholar] [CrossRef]
- Dinklo, T.; Shaban, H.; Thuring, J.W.; Lavreysen, H.; Stevens, K.E.; Zheng, L.; Mackie, C.; Grantham, C.; Vandenberk, I.; Meulders, G.; et al. Characterization of 2-[[4-fluoro-3-(trifluoromethyl)phenyl]amino]-4-(4-pyridinyl)-5-thiazolemethanol (JNJ-1930942), a novel positive allosteric modulator of the {alpha}7 nicotinic acetylcholine receptor. J. Pharmacol. Exp. Ther. 2011, 336, 560–574. [Google Scholar] [CrossRef]
- Chen, L.; Yamada, K.; Nabeshima, T.; Sokabe, M. alpha7 Nicotinic acetylcholine receptor as a target to rescue deficit in hippocampal LTP induction in beta-amyloid infused rats. Neuropharmacology 2006, 50, 254–268. [Google Scholar] [CrossRef]
- Hashemi-Firouzi, N.; Shahidi, S.; Soleimani Asl, S. Chronic stimulation of the serotonergic 5-HT4 receptor modulates amyloid-beta-related impairments in synaptic plasticity and memory deficits in male rats. Brain Res. 2021, 1773, 147701. [Google Scholar] [CrossRef]
- Levy, M.J.F.; Boulle, F.; Emerit, M.B.; Poilbout, C.; Steinbusch, H.W.M.; Van den Hove, D.L.A.; Kenis, G.; Lanfumey, L. 5-HTT independent effects of fluoxetine on neuroplasticity. Sci. Rep. 2019, 9, 6311. [Google Scholar] [CrossRef]
- Sun, D.S.; Gao, L.F.; Jin, L.; Wu, H.; Wang, Q.; Zhou, Y.; Fan, S.; Jiang, X.; Ke, D.; Lei, H.; et al. Fluoxetine administration during adolescence attenuates cognitive and synaptic deficits in adult 3 × TgAD mice. Neuropharmacology 2017, 126, 200–212. [Google Scholar] [CrossRef]
- Wei, Z.; Junhong, G.; Xiaoyuan, N.; Jie, W.; Zhaojun, W.; Meina, W.; Wei, Y.; Jun, Z.; Jinshun, Q. Citalopram Ameliorates Impairments in Spatial Memory and Synaptic Plasticity in Female 3xTgAD Mice. BioMed Res. Int. 2017, 2017, 1238687. [Google Scholar] [CrossRef]
- Shahidi, S.; Hashemi-Firouzi, N.; Asl, S.S.; Komaki, A. Serotonin type 6 receptor antagonist attenuates the impairment of long-term potentiation and memory induced by Abeta. Behav. Brain Res. 2019, 364, 205–212. [Google Scholar] [CrossRef]
- Shahidi, S.; Asl, S.S.; Komaki, A.; Hashemi-Firouzi, N. The effect of chronic stimulation of serotonin receptor type 7 on recognition, passive avoidance memory, hippocampal long-term potentiation, and neuronal apoptosis in the amyloid β protein treated rat. Psychopharmacology 2018, 235, 1513–1525. [Google Scholar] [CrossRef]
- Moreno-Castilla, P.; Rodriguez-Duran, L.F.; Guzman-Ramos, K.; Barcenas-Femat, A.; Escobar, M.L.; Bermudez-Rattoni, F. Dopaminergic neurotransmission dysfunction induced by amyloid-β transforms cortical long-term potentiation into long-term depression and produces memory impairment. Neurobiol. Aging 2016, 41, 187–199. [Google Scholar] [CrossRef]
- Hao, J.R.; Sun, N.; Lei, L.; Li, X.Y.; Yao, B.; Sun, K.; Hu, R.; Zhang, X.; Shi, X.D.; Gao, C. L-Stepholidine rescues memory deficit and synaptic plasticity in models of Alzheimer’s disease via activating dopamine D1 receptor/PKA signaling pathway. Cell Death Dis. 2015, 6, e1965. [Google Scholar] [CrossRef]
- Kouhnavardi, S.; Ecevitoglu, A.; Dragačević, V.; Sanna, F.; Arias-Sandoval, E.; Kalaba, P.; Kirchhofer, M.; Lubec, J.; Niello, M.; Holy, M.; et al. A Novel and Selective Dopamine Transporter Inhibitor, (S)-MK-26, Promotes Hippocampal Synaptic Plasticity and Restores Effort-Related Motivational Dysfunctions. Biomolecules 2022, 12, 881. [Google Scholar] [CrossRef]
- Rahman, A. The role of adenosine in Alzheimer’s disease. Curr. Neuropharmacol. 2009, 7, 207–216. [Google Scholar] [CrossRef]
- Costenla, A.R.; Diógenes, M.J.; Canas, P.M.; Rodrigues, R.J.; Nogueira, C.; Maroco, J.; Agostinho, P.M.; Ribeiro, J.A.; Cunha, R.A.; de Mendonça, A. Enhanced role of adenosine A(2A) receptors in the modulation of LTP in the rat hippocampus upon ageing. Eur. J. Neurosci. 2011, 34, 12–21. [Google Scholar] [CrossRef]
- Rebola, N.; Lujan, R.; Cunha, R.A.; Mulle, C. Adenosine A2A receptors are essential for long-term potentiation of NMDA-EPSCs at hippocampal mossy fiber synapses. Neuron 2008, 57, 121–134. [Google Scholar] [CrossRef]
- Gessi, S.; Poloni, T.E.; Negro, G.; Varani, K.; Pasquini, S.; Vincenzi, F.; Borea, P.A.; Merighi, S. A(2A) Adenosine Receptor as a Potential Biomarker and a Possible Therapeutic Target in Alzheimer’s Disease. Cells 2021, 10, 2344. [Google Scholar] [CrossRef]
- Silva, A.C.; Lemos, C.; Gonçalves, F.Q.; Pliássova, A.V.; Machado, N.J.; Silva, H.B.; Canas, P.M.; Cunha, R.A.; Lopes, J.P.; Agostinho, P. Blockade of adenosine A(2A) receptors recovers early deficits of memory and plasticity in the triple transgenic mouse model of Alzheimer’s disease. Neurobiol. Dis. 2018, 117, 72–81. [Google Scholar] [CrossRef]
- Jerónimo-Santos, A.; Batalha, V.L.; Müller, C.E.; Baqi, Y.; Sebastião, A.M.; Lopes, L.V.; Diógenes, M.J. Impact of in vivo chronic blockade of adenosine A2A receptors on the BDNF-mediated facilitation of LTP. Neuropharmacology 2014, 83, 99–106. [Google Scholar] [CrossRef]
- Lee, C.C.; Chang, C.P.; Lin, C.J.; Lai, H.L.; Kao, Y.H.; Cheng, S.J.; Chen, H.M.; Liao, Y.P.; Faivre, E.; Buée, L.; et al. Adenosine Augmentation Evoked by an ENT1 Inhibitor Improves Memory Impairment and Neuronal Plasticity in the APP/PS1 Mouse Model of Alzheimer’s Disease. Mol. Neurobiol. 2018, 55, 8936–8952. [Google Scholar] [CrossRef]
- Alkon, D.L.; Sun, M.K.; Nelson, T.J. PKC signaling deficits: A mechanistic hypothesis for the origins of Alzheimer’s disease. Trends Pharmacol. Sci. 2007, 28, 51–60. [Google Scholar] [CrossRef]
- Hongpaisan, J.; Sun, M.K.; Alkon, D.L. PKC ε activation prevents synaptic loss, Aβ elevation, and cognitive deficits in Alzheimer’s disease transgenic mice. J. Neurosci. 2011, 31, 630–643. [Google Scholar] [CrossRef]
- Navarrete, M.; Cuartero, M.I.; Palenzuela, R.; Draffin, J.E.; Konomi, A.; Serra, I.; Colié, S.; Castaño-Castaño, S.; Hasan, M.T.; Nebreda, Á.R.; et al. Astrocytic p38α MAPK drives NMDA receptor-dependent long-term depression and modulates long-term memory. Nat. Commun. 2019, 10, 2968. [Google Scholar] [CrossRef]
- Huentelman, M.J.; Piras, I.S.; Siniard, A.L.; De Both, M.D.; Richholt, R.F.; Balak, C.D.; Jamshidi, P.; Bigio, E.H.; Weintraub, S.; Loyer, E.T.; et al. Associations of MAP2K3 Gene Variants with Superior Memory in SuperAgers. Front. Aging Neurosci. 2018, 10, 155. [Google Scholar] [CrossRef]
- Alam, J.J. Selective Brain-Targeted Antagonism of p38 MAPKα Reduces Hippocampal IL-1β Levels and Improves Morris Water Maze Performance in Aged Rats. J. Alzheimers Dis. 2015, 48, 219–227. [Google Scholar] [CrossRef]
- Kheiri, G.; Dolatshahi, M.; Rahmani, F.; Rezaei, N. Role of p38/MAPKs in Alzheimer’s disease: Implications for amyloid beta toxicity targeted therapy. Rev. Neurosci. 2018, 30, 9–30. [Google Scholar] [CrossRef]
- Gee, M.S.; Son, S.H.; Jeon, S.H.; Do, J.; Kim, N.; Ju, Y.J.; Lee, S.J.; Chung, E.K.; Inn, K.S.; Kim, N.J.; et al. A selective p38α/β MAPK inhibitor alleviates neuropathology and cognitive impairment, and modulates microglia function in 5XFAD mouse. Alzheimers Res. Ther. 2020, 12, 45. [Google Scholar] [CrossRef]
- Roy, S.M.; Grum-Tokars, V.L.; Schavocky, J.P.; Saeed, F.; Staniszewski, A.; Teich, A.F.; Arancio, O.; Bachstetter, A.D.; Webster, S.J.; Van Eldik, L.J.; et al. Targeting human central nervous system protein kinases: An isoform selective p38αMAPK inhibitor that attenuates disease progression in Alzheimer’s disease mouse models. ACS Chem. Neurosci. 2015, 6, 666–680. [Google Scholar] [CrossRef]
- Georgievska, B.; Sandin, J.; Doherty, J.; Mörtberg, A.; Neelissen, J.; Andersson, A.; Gruber, S.; Nilsson, Y.; Schött, P.; Arvidsson, P.I.; et al. AZD1080, a novel GSK3 inhibitor, rescues synaptic plasticity deficits in rodent brain and exhibits peripheral target engagement in humans. J. Neurochem. 2013, 125, 446–456. [Google Scholar] [CrossRef]
- Yang, M.; Wang, J. Berberine Ameliorates Cognitive Disorder via GSK3β/PGC-1α Signaling in APP/PS1 Mice. J. Nutr. Sci. Vitaminol. 2022, 68, 228–235. [Google Scholar] [CrossRef]
- Singh, A.K.; Singh, S.K.; Nandi, M.K.; Mishra, G.; Maurya, A.; Rai, A.; Rai, G.K.; Awasthi, R.; Sharma, B.; Kulkarni, G.T. Berberine: A Plant-derived Alkaloid with Therapeutic Potential to Combat Alzheimer’s disease. Cent. Nerv. Syst. Agents Med. Chem. 2019, 19, 154–170. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Yan, Q.; Huang, Z.T.; Zou, Q.; Li, J.; Yuan, M.H.; Wu, L.Q.; Cai, Z.Y. Ameliorating Ribosylation-Induced Amyloid-β Pathology by Berberine via Inhibiting mTOR/p70S6K Signaling. J. Alzheimer’s Dis. JAD 2021, 79, 833–844. [Google Scholar] [CrossRef]
- Wei, W.; Yao, J.-X.; Zhang, T.-T.; Wen, J.-Y.; Zhang, Z.; Luo, Y.-M.; Cao, Y.; Li, H. Network pharmacology reveals that Berberine may function against Alzheimer’s disease via the AKT signaling pathway. Front. Neurosic. 2023, 17, 1059496. [Google Scholar] [CrossRef]
- Akhondzadeh, S.; Noroozian, M.; Mohammadi, M.; Ohadinia, S.; Jamshidi, A.H.; Khani, M. Salvia officinalis extract in the treatment of patients with mild to moderate Alzheimer’s disease: A double blind, randomized and placebo-controlled trial. J. Clin. Pharm. Ther. 2003, 28, 53–59. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, X.; Teng, Z.; Zhang, T.; Li, Y. Downregulation of PI3K/Akt/mTOR signaling pathway in curcumin-induced autophagy in APP/PS1 double transgenic mice. Eur. J. Pharmacol. 2014, 740, 312–320. [Google Scholar] [CrossRef]
- Zeng, Y.; Zhang, J.; Zhu, Y.; Zhang, J.; Shen, H.; Lu, J.; Pan, X.; Lin, N.; Dai, X.; Zhou, M.; et al. Tripchlorolide improves cognitive deficits by reducing amyloid β and upregulating synapse-related proteins in a transgenic model of Alzheimer’s Disease. J. Neurochem. 2015, 133, 38–52. [Google Scholar] [CrossRef]
- Yi, J.H.; Baek, S.J.; Heo, S.; Park, H.J.; Kwon, H.; Lee, S.; Jung, J.; Park, S.J.; Kim, B.C.; Lee, Y.C.; et al. Direct pharmacological Akt activation rescues Alzheimer’s disease like memory impairments and aberrant synaptic plasticity. Neuropharmacology 2018, 128, 282–292. [Google Scholar] [CrossRef]
- Yang, S.; Du, Y.; Zhao, X.; Wu, C.; Yu, P. Reducing PDK1/Akt Activity: An Effective Therapeutic Target in the Treatment of Alzheimer’s Disease. Cells 2022, 11, 1735. [Google Scholar] [CrossRef]
- Yang, A.J.T.; Bagit, A.; MacPherson, R.E.K. Resveratrol, Metabolic Dysregulation, and Alzheimer’s Disease: Considerations for Neurogenerative Disease. Int. J. Mol. Sci. 2021, 22, 4628. [Google Scholar] [CrossRef]
- Amato, S.; Man, H.Y. Bioenergy sensing in the brain: The role of AMP-activated protein kinase in neuronal metabolism, development and neurological diseases. Cell Cycle 2011, 10, 3452–3460. [Google Scholar] [CrossRef]
- Ma, T.; Chen, Y.; Vingtdeux, V.; Zhao, H.; Viollet, B.; Marambaud, P.; Klann, E. Inhibition of AMP-activated protein kinase signaling alleviates impairments in hippocampal synaptic plasticity induced by amyloid β. J. Neurosci. 2014, 34, 12230–12238. [Google Scholar] [CrossRef]
- Tweedie, D.; Ferguson, R.A.; Fishman, K.; Frankola, K.A.; Van Praag, H.; Holloway, H.W.; Luo, W.; Li, Y.; Caracciolo, L.; Russo, I.; et al. Tumor necrosis factor-α synthesis inhibitor 3,6’-dithiothalidomide attenuates markers of inflammation, Alzheimer pathology and behavioral deficits in animal models of neuroinflammation and Alzheimer’s disease. J. Neuroinflammation 2012, 9, 106. [Google Scholar] [CrossRef]
- Lonnemann, N.; Hosseini, S.; Marchetti, C.; Skouras, D.B.; Stefanoni, D.; D’Alessandro, A.; Dinarello, C.A.; Korte, M. The NLRP3 inflammasome inhibitor OLT1177 rescues cognitive impairment in a mouse model of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 2020, 117, 32145–32154. [Google Scholar] [CrossRef]
- Alves, S.; Churlaud, G.; Audrain, M.; Michaelsen-Preusse, K.; Fol, R.; Souchet, B.; Braudeau, J.; Korte, M.; Klatzmann, D.; Cartier, N. Interleukin-2 improves amyloid pathology, synaptic failure and memory in Alzheimer’s disease mice. Brain 2017, 140, 826–842. [Google Scholar] [CrossRef]
- Jiang, Y.; Li, K.; Li, X.; Xu, L.; Yang, Z. Sodium butyrate ameliorates the impairment of synaptic plasticity by inhibiting the neuroinflammation in 5XFAD mice. Chem. Biol. Interact. 2021, 341, 109452. [Google Scholar] [CrossRef]
- Serrano, E.; Sanz, P.; Barrantes, F.J. The impact of apolipoprotein E allelic variants on Alzheimer disease. In Psychiatry and Neuroscience Update; Gargiulo, P.A., Mesones, H.L., Eds.; Springer International Publishing AG: New York, NY, USA, 2021. [Google Scholar]
- Barrantes, F.J.; Borroni, V.; Vallés, S. Neuronal nicotinic acetylcholine receptor–cholesterol crosstalk in Alzheimer’s disease. FEBS Lett. 2010, 584, 1856–1863. [Google Scholar] [CrossRef]
- Kanekiyo, T.; Xu, H.; Bu, G. ApoE and Aβ in Alzheimer’s disease: Accidental encounters or partners? Neuron 2014, 81, 740–754. [Google Scholar] [CrossRef]
- Liu, D.S.; Pan, X.D.; Zhang, J.; Shen, H.; Collins, N.C.; Cole, A.M.; Koster, K.P.; Ben Aissa, M.; Dai, X.M.; Zhou, M.; et al. APOE4 enhances age-dependent decline in cognitive function by down-regulating an NMDA receptor pathway in EFAD-Tg mice. Mol. Neurodegener. 2015, 10, 7. [Google Scholar] [CrossRef]
- Zhao, N.; Liu, C.C.; Qiao, W.; Bu, G. Apolipoprotein E, Receptors, and Modulation of Alzheimer’s Disease. Biol. Psychiatry 2018, 83, 347–357. [Google Scholar] [CrossRef]
- Teter, B. ApoE-dependent plasticity in Alzheimer’s disease. J. Mol. Neurosci. 2004, 23, 167–179. [Google Scholar] [CrossRef]
- Michikawa, M. The role of cholesterol in pathogenesis of Alzheimer’s disease: Dual metabolic interaction between amyloid beta-protein and cholesterol. Mol. Neurobiol. 2003, 27, 1–12. [Google Scholar] [CrossRef]
- Champagne, D.; Pearson, D.; Dea, D.; Rochford, J.; Poirier, J. The cholesterol-lowering drug probucol increases apolipoprotein E production in the hippocampus of aged rats: Implications for Alzheimer’s disease. Neuroscience 2003, 121, 99–110. [Google Scholar] [CrossRef]
- 224 Oliveira, T.G.; Chan, R.B.; Tian, H.; Laredo, M.; Shui, G.; Staniszewski, A.; Zhang, H.; Wang, L.; Kim, T.W.; Duff, K.E.; et al. Phospholipase d2 ablation ameliorates Alzheimer’s disease-linked synaptic dysfunction and cognitive deficits. J. Neurosci. 2010, 30, 16419–16428. [Google Scholar] [CrossRef]
- Schaeffer, E.L.; Forlenza, O.V.; Gattaz, W.F. Phospholipase A2 activation as a therapeutic approach for cognitive enhancement in early-stage Alzheimer disease. Psychopharmacology 2009, 202, 37–51. [Google Scholar] [CrossRef]
- Bernard, J.; Lahsaini, A.; Massicotte, G. Potassium-induced long-term potentiation in area CA1 of the hippocampus involves phospholipase activation. Hippocampus 1994, 4, 447–453. [Google Scholar] [CrossRef]
- Zhang, W.; Huang, Y.; Guo, X.; Zhang, M.; Yuan, X.; Zu, H. DHCR24 Reverses Alzheimer’s Disease-related Pahtology and Cognitive Impairment Via Increasing Hippocampal Cholesterol Levels in 5 × FAD Mice. Acta Neuropathol. 2023, 11, 102. [Google Scholar] [CrossRef]
- Borroni, V.; Kamerbeek, C.; Pediconi, M.F.; Barrantes, F.J. Lovastatin Differentially Regulates α7 and α4 Neuronal Nicotinic Acetylcholine Receptor Levels in Rat Hippocampal Neurons. Molecules 2020, 25, 4838. [Google Scholar] [CrossRef]
- Sodero, A.O.; Barrantes, F.J. Pleiotropic effects of statins on brain cells. Biochim. Biophys. Acta Biomembr. 2020, 1862, 183340. [Google Scholar] [CrossRef]
- Kwak, B.; Mulhaupt, F.; Myit, S.; Mach, F. Statins as a newly recognized type of immunomodulator. Nat. Med. 2000, 6, 1399–1402. [Google Scholar] [CrossRef]
- Waragai, M.; Adame, A.; Trinh, I.; Sekiyama, K.; Takamatsu, Y.; Une, K.; Masliah, E.; Hashimoto, M. Possible involvement of adiponectin, the anti-diabetes molecule, in the pathogenesis of Alzheimer’s disease. J. Alzheimer’s Dis. 2016, 52, 1453–1459. [Google Scholar] [CrossRef]
- García-Casares, N.; García-Arnés, J.A.; Rioja, J.; Ariza, M.J.; Gutiérrez, A.; Alfaro, F.; Nabrozidis, A.; González-Alegre, P.; González-Santos, P. Alzheimer’s like brain changes correlate with low adiponectin plasma levels in type 2 diabetic patients. J. Diabetes Its Complicat. 2016, 30, 281–286. [Google Scholar] [CrossRef]
- Yan, X.D.; Qu, X.S.; Yin, J.; Qiao, J.; Zhang, J.; Qi, J.S.; Wu, M.N. Adiponectin Ameliorates Cognitive Behaviors and in vivo Synaptic Plasticity Impairments in 3xTg-AD Mice. J. Alzheimers Dis. 2022, 85, 343–357. [Google Scholar] [CrossRef]
- Wang, M.; Jo, J.; Song, J. Adiponectin improves long-term potentiation in the 5XFAD mouse brain. Sci. Rep. 2019, 9, 8918. [Google Scholar] [CrossRef]
| Protein | Process in Which It Is Involved | Mode of Presentation in AD |
|---|---|---|
| Neurogranin | Promotion of synaptic plasticity and LTP | Increased |
| SNAP-25 | Control of synaptic plasticity | Increased |
| BDNF | Structural plasticity and cognitive plasticity/learning | Decreased |
| VEGF | Neurogenesis and synaptic plasticity | Decreased |
| Target | Drug | Outcome |
|---|---|---|
| Glucose metabolism | T3D-959 (anti-diabetic) | Improved functional connectivity of the hippocampus |
| Glucose metabolism | empagliflozin | Alleged neuroplasticity improvement through glutamate activity regulation |
| Glutamatergic toxicity | Riluzole | Inhibition of the presynaptic release of glutamate and associated glutamatergic toxicity |
| Amyloid and tau pathologies | Simulifam | Decrease in synaptic neurogranin levels |
| Mitogen-activated protein kinase p38α | Neflamapinod | Decrease in neurogranin levels/ Enhanced memory performance |
| Sigma2 receptor | CT1812 | Prevention of synaptotoxicity induced by Aβ, increased levels of synaptic proteins related to plasticity, and synaptic density |
| Protein kinase C | Bryostatin | Enhanced cognitive performance |
| Glutaminyl cyclase | PQ912 | Decrease in neurogranin levels |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Colavitta, M.F.; Barrantes, F.J. Therapeutic Strategies Aimed at Improving Neuroplasticity in Alzheimer Disease. Pharmaceutics 2023, 15, 2052. https://doi.org/10.3390/pharmaceutics15082052
Colavitta MF, Barrantes FJ. Therapeutic Strategies Aimed at Improving Neuroplasticity in Alzheimer Disease. Pharmaceutics. 2023; 15(8):2052. https://doi.org/10.3390/pharmaceutics15082052
Chicago/Turabian StyleColavitta, María F., and Francisco J. Barrantes. 2023. "Therapeutic Strategies Aimed at Improving Neuroplasticity in Alzheimer Disease" Pharmaceutics 15, no. 8: 2052. https://doi.org/10.3390/pharmaceutics15082052
APA StyleColavitta, M. F., & Barrantes, F. J. (2023). Therapeutic Strategies Aimed at Improving Neuroplasticity in Alzheimer Disease. Pharmaceutics, 15(8), 2052. https://doi.org/10.3390/pharmaceutics15082052






