1. Introduction
In 2021 the prevalence of diabetes in adults ranging from 20 to 79 years worldwide was 9.8%, representing 536.6 million patients [
1]. Diabetic patients have elevated levels of glucose in their blood, which leads to a range of conditions. A subcategory of complications derived from diabetes is diabetic ocular diseases. Examples include diabetic retinopathy, diabetic keratopathy, or cataracts, which have been linked to the polyol pathway [
2]. These different illnesses affect the patient with loss of vision, culminating in blindness if left untreated. The current treatments for diabetic ocular diseases involve intravitreal injections [
3], laser treatment [
4], or even vitrectomy [
5].
The polyol pathway transforms glucose into sorbitol when hexokinases active in the Embden-Meyerhof pathway are saturated, and subsequently, glucose is oxidated to sorbitol and further transformed into fructose. It is important to mention that this pathway is rarely used by the body in a healthy state as it is only activated at high intracellular glucose concentrations [
6]. However, in the case of diabetes, the blood glucose levels can be high enough for the reaction balance to favor sorbitol production. Sorbitol is not permeable to cell membranes and accumulates inside the cells, and this excess creates osmotic stress [
7]. Moreover, the conversion of glucose to sorbitol consumes NADPH which depletes NADPH stock [
8] and competes with glutathione reduction, which in turn adds to oxidative damage [
9]. This induces a lack of antioxidants and therefore increases the number of reactive oxygen species (ROS), increasing the oxidative stress in the affected tissue [
10].
The polyol pathway can be blocked by inhibiting aldose reductase, an essential enzyme in the reduction of glucose to sorbitol [
7,
11]. In the case of diabetic retinopathy, the posterior segment of the eye is affected by sorbitol accumulation, and therefore, a carrier able to deliver the aldose reductase inhibitor to the inner eye tissues is needed. The aldose reductase inhibitor epalrestat is approved in Japan for oral administration thrice a day in 50 mg doses [
12] to treat diabetic neuropathy. Its IC
90 as an aldose reductase inhibitor was found to be 2.5 × 10
−2 mg/mL [
13] and an IC
50 of 1.41 × 10
−6 mg/mL in rat lens [
14]. Epalrestat was shown to reduce aldose reductase expression and vascular endothelial growth factor (VEGF) secretion in retinal pigment epithelial cells [
15]. Compared to methylcobalamine, epalrestat taken orally thrice daily for twelve weeks is better tolerated by patients and showed fewer adverse effects [
16].
Although the information on ocular delivery systems of aldose reductase inhibitors is limited, topical instillation of tolrestat in rats was successful in preventing cataract development [
17]. Biodegradable injectable implants for sustained delivery of N-4- (benzoylaminophenylsulfonyl glycine) also demonstrated inhibition of aldose reductase activity and VEGF expression in ARPE cells, as well as in galactose-fed rats [
18]. Similarly, 2-methylsorbinil suspensions were shown to be successful in inhibiting cataracts in galactose-fed rats [
19]. The only previous work attempting to deliver epalrestat to the posterior segment of the eye made use of contact lenses either through direct loading [
20] or previous encapsulation in PEGylated solid lipid nanoparticles [
21]. This work aims to show the viability of a nanoparticle system as a topical delivery system for epalrestat without the need for a contact lens.
In order to deliver epalrestat to the therapeutic site topically, it has to be encapsulated to cross different ocular barriers due to the high hydrophobicity of the molecule. Therefore, niosomes were prepared to encapsulate the drug and carry it to the posterior segment for it to reach the therapeutic site [
22]. Niosomes are vesicles of small size with a bilayer made of surfactants and supplementary molecules that self-assemble through hydrophobic interactions as the involved molecules are amphiphilic. The shape, size, and surface charge can be modified by adjusting the concentration and molar ratios of different reagents [
23,
24]. They are being investigated for topical administration in ocular and skin tissue [
25,
26,
27].
This work aims to formulate a niosomal carrier that fits the requirements to deliver epalrestat to the posterior segment of the eye through the scleral route. This was done by selecting polysorbate 60 (Tween 60), cholesterol, and 1,2-di-O-octadecenyl-3-trimethylammonium propane (DOTMA) and preparing niosomes with specific size, polydispersity, and surface charge. The obtained systems loaded with epalrestat were characterized in depth using various methods testing its physicochemical properties, encapsulation efficiency, release profile, permeation profile through the porcine cornea and sclera, ocular irritability potential, impact on blood glucose levels, and toxicity. Alternatives to animal testing were used according to the 3Rs principles: the HET-CAM [
28] model for the ocular irritability potential, an extension of this model (Gluc-HET) to monitor the effect of the loaded niosome on the blood glucose levels [
29], a zebrafish embryo model was used to assess the toxicity of the loaded niosomes [
30,
31,
32], and porcine eye tissues from the slaughterhouse for ex vivo permeability tests.
2. Materials and Methods
Materials. Polysorbate 60 MW 1311.7 g/mol (Tween 60, HLB 14.9, Sigma Aldrich, Buchs, Switzerland), polysorbate 80 MW 1310 g/mol (Tween 80, HLB 15, Sigma Aldrich, Switzerland), 1,2-di-O-octadecenyl-3-trimethylammonium propane (chloride salt) (DOTMA, 670.58 g/mol) (Avanti, Alabaster, AL, USA), epalrestat (319.4 g/mol) (TCI, Tokyo, Japan), cholesterol (386.7 g/mol) (Chemtrec, Madrid, Spain), ethanol (VWR Chemicals, Briare, France), dichloromethane (Fischer Scientific, Waltham, MA USA), chloroform (Cienytech, Santiago de Compostela, Spain), phosphate-buffered saline (Life Tecnologies Co., Carlsbad, CA, USA), sodium chloride (Labkem, Barcelona, Spain), potassium chloride (Panreac, Castellar del Vallès, Spain), sodium bicarbonate (Merck, St Louis, MO, USA), calcium dihydrochloride (Merck, Darmstadt, Germany), potassium dihydrogen phosphate 1-basic (Panreac, Castellar del Vallès, Spain), disodium hydrogen phosphate dihydrate (VWR Chemicals, Briare, France), phosphate-buffered saline solution (Sigma-Aldrich, Lyon, France), Hanks’ Balanced Salt Solution (HBSS) (Paisley, Scotland, UK), glibenclamide (Roche, Basel, Switzerland). Ultrapure water (resistivity > 18.2 MΩ cm) was obtained by reverse osmosis (Milli-Q®, Millipore Ibérica, Madrid, Spain).
Niosome formulation. The protocol was adapted from previous reports [
33,
34,
35]. Briefly, polysorbate 60 and cholesterol were dissolved in 2 mL ethanol in the presence or absence of DOTMA (at 1/0.42/0, 1/0.42/0.075 and 1/0.42/0.158 molar ratios of Tween 60/cholesterol/DOTMA as shown in
Table 1). The combined total amount was kept at 76.38 µmol. Epalrestat (2 mg) was dissolved in 500 µL ethanol and added to the flask. The organic solvent was evaporated in a round bottom flask with a rotary evaporator at 70 °C under 50 mbar pressure to create a film. The film was subsequently desiccated for 30 min. 10 mL of ultrapure water was added to the flask. The film was removed from the walls of the flask by ultrasonication for 30 min. In order to form the niosomes, the solution was sonified for 90 s at 20% amplitude on a Branson Digital Sonifier 450 (Marshall Scientific, Hampton, NH, USA). This yielded epalrestat-loaded niosomes in water. In order to remove the unencapsulated drug, dialysis was performed for 30 min in 500 mL ultrapure water with 1 vol% Tween 80 with a 14,000 Da dialysis tubing (Sigma-Aldrich, Milwaukee, WI, USA).
The formulations were named with a code TCDX based on the included components: T for Tween, C for cholesterol, D for DOTMA, and x for the molar percentage of DOTMA present in the niosome. The code FD (free drug) was used to refer to a solution of 10% ethanol in water containing epalrestat at the same concentration as the epalrestat in the niosome formulation. For storage, the niosomes were kept in 15 mL Falcon tubes at room temperature in the absence of light.
Niosome characterization. The particle size and zeta potential of the niosomes were measured with DLS using a Zetasizer Nano (Malvern Instruments, Herrenberg, Germany) in ultrapure water at 20 °C with 10 s equilibration time using backscatter. The values taken were measured by intensity. The pH of the formulation solution was measured using a GL22 pH & ion meter (Crison, Barcelona, Spain). Viscosity at 35 °C was recorded between 0.05 and 200 rad/s in a Rheolyst AR-1000N rheometer (TA Instruments, Newcastle, UK) equipped with a Peltier plate and a cone geometry (40 mm diameter, 2°).
Stability study. The stability of epalrestat-loaded niosomes over 7 days was assessed at 4 °C (fridge) and 25 °C (oven) in the absence of light. The size, polydispersity index, zeta-potential, and content in epalrestat were measured at time points 0 and after 7 days.
TEM. Drops of 5 µL of blank and epalrestat-loaded niosome dispersions were placed on carbon-coated grids, and the excess the solution was removed with filter paper. The samples were dyed with 1% phosphotungstic acid in water. The grid was allowed to dry and observed using a high-resolution JEM-1011 transmission electron microscope (JEOL USA Inc., Peabody, MA, USA).
HPLC. Quantitative analysis of epalrestat was performed on a Waters 717 plus Autosampler with a 4.6 × 250 mm C18 Symmetry column (Waters, Wexford, Ireland) with 5 µm pores. The mobile phase was acetonitrile:elution buffer 45:55 (
v/
v). The elution buffer was composed of 25 mM potassium dihydrogen phosphate and 25 mM disodium hydrogen phosphate dihydrate in ultrapure water adjusted to pH 6.5 with phosphoric acid. The flow rate was 0.85 mL/min, the detection wavelength was 295 nm, the temperature was maintained at 25 °C, and the injection volume was 40 µL. The calibration curve was prepared with concentrations ranging from 1 to 10 µg/mL with an increment step of 1 µg/mL. The retention time of epalrestat was 4.5 min. The HPLC quantification method was validated with regard to specificity, detection and quantitation limits, linearity, accuracy, precision, and range (
Supplementary Material Figure S1).
Encapsulation efficiency. The encapsulation efficiency (
EE%) was calculated by dialyzing the niosomes for 30 min in ultrapure water with 1 vol% Tween 80 [
36] and analyzing the medium with HPLC. The dialysis membrane had a molecular weight cutoff of 12,000 Da and an effective dialysis area of 4.2 cm
2. The concentration was confirmed by lysing the niosomes with methanol [
34] and measuring the concentration of the drug encapsulated by HPLC. The efficiency was then calculated with Equation (1) [
35]:
Release study. The release was tested by placing 5 mL of niosomes (0.20 ± 0.01 mg epalrestat/mL) in 14,000 MWCO dialysis tubing (Sigma-Aldrich, St Louis, MO, USA) and using 500 mL ultrapure water with 1% Tween 80 as receptor medium [
36]. The medium was left at 20 °C for 20 days or 37 °C for 24 h stirring at 400 rpm with a magnetic stirrer. 1 mL of the medium was taken and replaced with 1 mL of fresh medium every day for 8 days and then every two days until day 21. The concentration of epalrestat in the release medium was quantified with HPLC. The niosomes left in the dialysis bag were lysed, and the remaining epalrestat was quantified with HPLC.
HET-CAM. For the Hen’s Egg Test on Chorioallantoic Membrane (HET-CAM) assay [
28], fertilized eggs (15) were supplied by Coren (Ourense, Spain) and cleaned before incubation in a CCRS 0150 incubator (Ineltec, Tona, Spain) for 9 days at 37 °C and 60% relative humidity. On the day of the experiment, the shell of the eggs was pared off with a circular saw at the location of the air cell. The untouched inner membrane was moistened with a 0.9% NaCl solution, and the eggs were placed back in the incubator for 30 min. The 0.9% NaCl solution was subsequently removed, as well as the inner membrane, while being careful not to damage the blood vessels of the CAM underneath. Any non-viable egg was discarded. The positive control was NaOH 0.1M, and the negative control was 0.9% NaCl. The solutions tested were formulation TCD0, TCD5, and TCD10 in water loaded with 0.2 mg/mL epalrestat, unloaded niosomes of the same molar ratios, and epalrestat dissolved in ethanol: water 10/90
v/
v mixture. The 300 µL of the testing solution was then added to the eggs, and the effect on the blood vessels regarding hemorrhage, lysis, and coagulation was recorded. The ocular irritability potential score was calculated with Equation (2) [
28]:
with
H = hemorrhage time (s),
L = lysis time (s),
C = coagulation time (s).
Gluc-HET. For the Gluc-HET assay [
29], fertilized eggs (15) were supplied by Coren (Ourense, Spain) and cleaned before incubation in a CCRS 0150 incubator (Ineltec, Tona, Spain) for 11 days at 37 °C and 60% relative humidity. On the day of the experiment, the shell of the egg above the air pocket was pierced with a needle, and 300 µL of the testing solution was deposited inside the air compartment. The air compartment of the negative controls was pieced, but no solution was added [
29]. The positive control was a solution of 0.002 mg/mL glibenclamide solution in HBSS. The eggshells are then closed off with parafilm. After 2 h incubation, the eggshell above the air compartment was removed, and the chorioallantoic membrane was cut next to a blood vessel with a scalpel. The blood vessel was placed on a flat metal tong and dried with paper. Once no moisture was absorbed anymore by the paper, the vessel was cut, and the blood glucose level was measured with a glucose meter (Contour next, Ascensia Diabetes Care, Basel, Switzerland).
Zebrafish embryotoxicity test. Epalrestat-loaded niosome toxicity was assessed using zebrafish embryos (Danio rerio) and the Fish Embryo Acute Aquatic Toxicity (FET) Test. Zebrafish embryos were selected around 3 h post-fertilization (hpf). The test was considered valid if the mortality of fish embryos was at least 30% in the positive control (3,4-dichloroaniline) and lower than 10% in the negative control. In our experiment, the mortality of the negative control was 3.3%, and the mortality of the positive control was 100%. The larvae were grown in autoclaved osmosis water. The experiment was carried out by including 5 or 10 µL of formulation in 200 µL of medium (4.9 µg/mL and 9.5 µg/mL, respectively) and quantifying the mortality at 24, 48, 72, and 96 h. The formulations tested were TCD0, TCD5, and TCD10. The experiments were conducted in triplicate.
Corneal permeation. Porcine eyes were supplied by a slaughterhouse and transported to the laboratory in diluted PBS solution at 4 °C in an ice bath. The corneas were dissected with 2–3 mm of surrounding tissue and washed with 0.9% NaCl to remove any attached tissue. The corneas were mounted in Franz diffusion cells with the outer part of the cornea facing up. The area available for permeation was 0.785 cm
2. The receiving chamber was filled with 6 mL of Tween 80:water 10:90
v/
v solution while making sure no bubbles formed and then agitated with a magnetic stirring rod at 400 rpm. The donor chamber was filled with 2 mL of carbonated buffer (pH 7.2) and closed off with parafilm to prevent evaporation. The system was then left to equilibrate for 1 h at 37 °C. Once the system was balanced, the carbonated buffer in the donor chamber was replaced by 2 mL of either 0.2 mg/mL epalrestat solution in 10 mL of ethanol:water 10:90
v/
v or 0.2 mg/mL epalrestat encapsulated in the TCD0, TCD5, and TCD10 formulations. After 30 min, at 1 h and then every hour, 1 mL of the solution in the receiving chamber was removed and replaced with 1 mL of fresh Tween 80:water 10:90
v/
v solution. After 6 h, the last sample was taken, and the corneas were incubated in ethanol at 37 °C for 24 h. They were then sonicated at 37 °C in an ultrasonic bath for 90 min. The resulting mixture was centrifuged at 1000 rpm at 25 °C for 5 min, and the supernatant was centrifuged at 14,000 rpm at 25 °C for 20 min. After filtration through 0.22 µm pore syringe filters (Scharlab, Barcelona, Spain), all the samples from the receptor chamber as well as the supernatant from the tissue incubation, were analyzed with HPLC according to the protocol described above. All experiments were carried out in triplicate (
Supplementary Material Figure S2).
Scleral permeation. Scleral permeation was performed the same way as corneal permeation, except the tissue used to permeate through was the porcine sclera instead of the porcine cornea.
IR-Raman. Porcine cornea and sclera were permeated with TCD0, TCD5, and TCD10 niosomes for 6 h under the same conditions as the corneal and scleral permeation experiment described above. The IR-Raman study was performed by taking a minimum of 3 points and a maximum of 6 points per cornea and per sclera (
Supplementary Material Figures S3 and S4) and measuring the Raman scattering of the surface. Furthermore, a line scan was performed in the x-z plane (
Supplementary Material Figures S5–S7). The excitation wavelength was 532.188 nm, the sample was kept at a temperature of 8 °C for the duration of the experiment, the laser power was 3 mW, and each point was measured with 60 accumulations, with an integration time of 0.3 s and an objective of ×50 (Zeiss LD EC Epiplan-Neofluar Dic 50×/0.55). The measurement was done on the top and bottom part of the tissue, and the absolute height of the peak (
Supplementary Material Figure S8) (CCD cts) was compared between the top and bottom of each tissue. For the line scan, spectra were accumulated by taking 30 spectra per line and 15 lines per image, each spectrum at a distance of 1 µm from the previous point, both in the x and the z direction.
Statistical analysis. All conditions in the experiments were carried out in triplicate, and the data were shown as an average with a standard deviation when possible. Statistical analysis was carried out in Origin, making use of a one-way analysis of variance (one-way ANOVA) with Tukey’s comparison for evaluation. The difference between groups was statistically significant when the p-value was lower than 0.05.
4. Discussion
To the best of our knowledge, this study is the first of its kind encapsulating epalrestat in niosomes and characterizing them in depth. Niosomes were designed by first choosing an adapted ratio of cholesterol to Tween 60, then testing the effect of the introduction of DOTMA to change the surface charge and render the niosomes cationic.
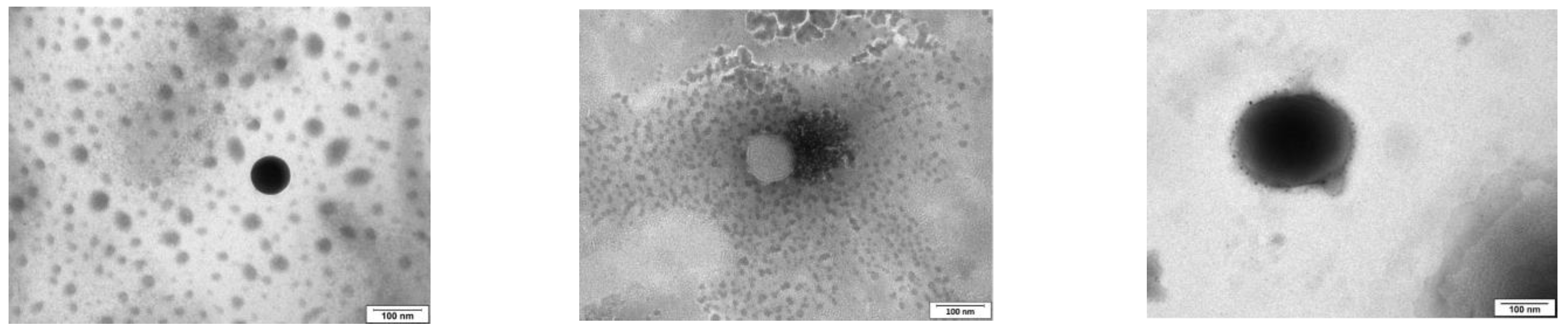
Three formulations were chosen, TCD0 as a control (−23 mV), TCD5 with low positive surface charge (+17 mV), and TCD10 as highly cationic (+40 mV). All three formulations exhibited sizes smaller than 100 nm, encapsulation efficiencies above 99%, spherical morphologies, and high stability over two months. When comparing the niosomes to other studies, the size of TCD0 niosomes was 20 nm bigger, and its polydispersity index was bigger than Tween 60/cholesterol prepared with the ethanol injection method [
56]. The ethanol injection method is known to give more uniform niosomes [
57] but is not appropriate for the encapsulation of all drugs. Two other studies made use of cationic niosomes and reported the size and zeta-potential, one substituting lycopene for cholesterol [
33] and one substituting Tween 20 for Tween 60 [
58]. In the first study, the size (101 nm) and the PDI (0.44) were bigger, and the zeta-potential (+33.8 mV) was lower than the TCD10 formulation. In the second study, the size (130 nm) was bigger, but the PDI (0.14) was smaller than the TCD10 formulation. Here the difference in non-ionic surfactant and helper lipid accounts for the difference in the physicochemical properties [
41], as the preparation method was similar. The high encapsulation efficiency of epalrestat may be explained by the highly hydrophobic nature of epalrestat (water solubility of 0.0467 mg/mL [
59]) that would tend to remain in the bilayer of the niosome, which is assembled through hydrophobic interactions. An encapsulating effect of hydrophobic molecules in the bilayer of the niosome was also seen in Span 80 niosomes with an added ionic surfactant [
60], blends of Tween60/Span60 anionic niosomes, or Tween 80/Span80 PEGylated systems [
61]. A complementary explanation is the length of the alkyl chains (stearyl C18), which usually leads to higher encapsulation efficiencies of hydrophobic cargo. Longer chains tend to display higher entrapment efficiency, with Tween 60 having a longer carbon tail than the lauryl chains (C12) of Tween 20 or Brij 35, for example [
62,
63]. With these encapsulation efficiencies, epalrestat concentration in the final niosome dispersion was close to 0.2 mg/mL. Niosome morphology (
Figure 1) was spherical with low aggregation, and the size agreed with the DLS data. These images were in line with TEM images of reports describing niosomes made using the thin film hydration method [
59].
The release of epalrestat from the niosomes reached 2.6%, 3.2%, and 4.0% after 12 h for the formulations TCD10, TCD5, and TCD0, respectively, which is around therapeutic levels for a tear film volume of 10 µL [
13,
14]. When releasing hydrophilic molecules, Tween 80/cholesterol niosomes displayed burst release behavior (90% in 1 h), as is the case with (−)-epigallocatechin gallate [
52]. Release of lipophilic molecules also showed to be faster with mixed Span 60/Tween 60 cholesterol niosomes loaded with α-tocopherol (40% in 4 h) [
64], as with Tween 60 niosomes containing meloxicam (90% in 7 h) [
65]. However, it must be noted that these two examples are not exactly similar to our system as the α-tocopherol was released in a simulated gastric environment, and the niosomes containing meloxicam were micrometer size. Furthermore, the behavior of sustained release clashes with previous findings where epalrestat was incorporated into solid-lipid nanoparticles-laden lenses [
21], and that showed a burst release. The epalrestat release looked similar to the sustained release obtained in lens delivery without nanoparticles after the initial release of adsorbed epalrestat [
20].
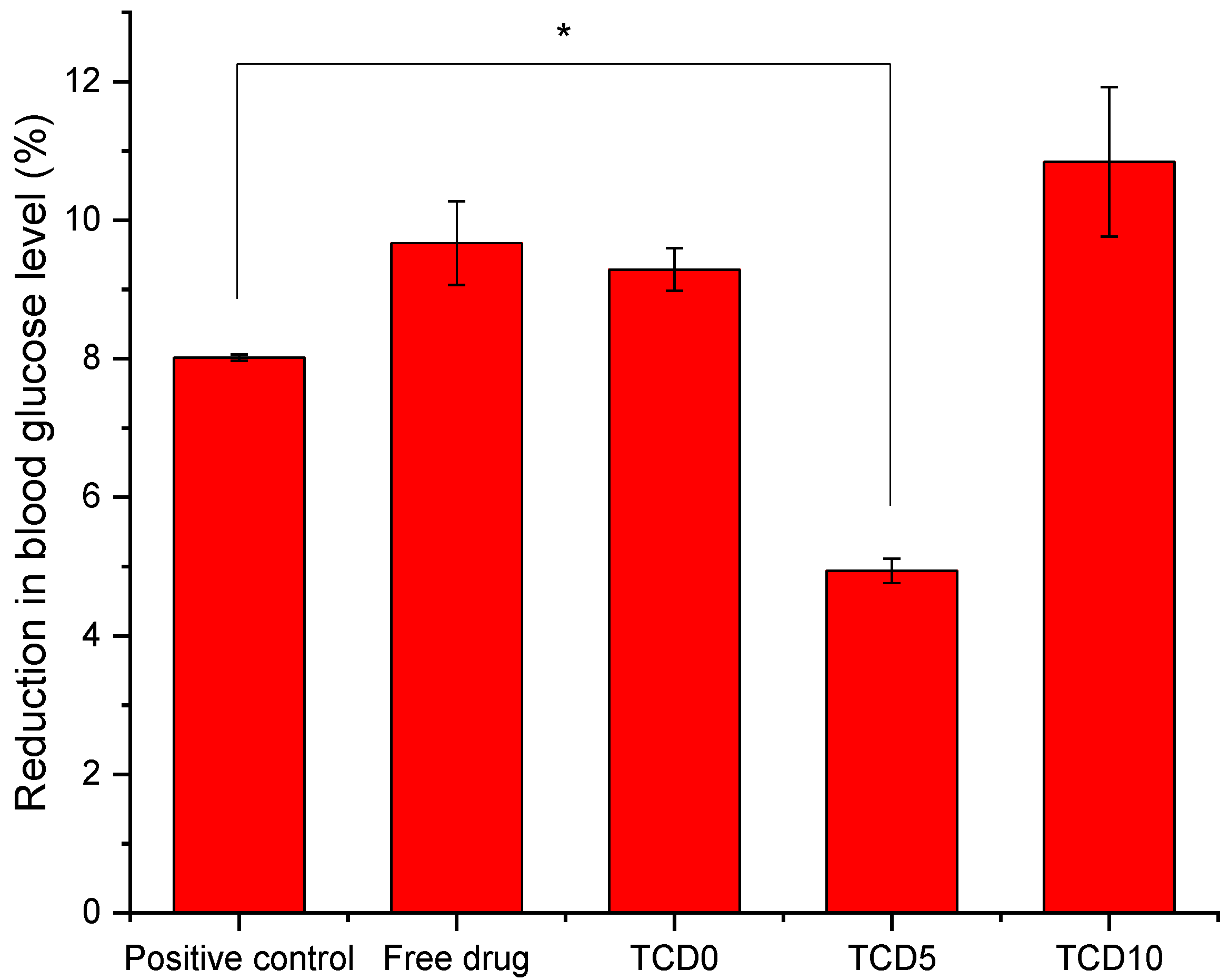
To screen that the formulations are able to be used in eye drop format, alternatives to in vivo experimentation were used: ocular irritability potential assessment with the HET-CAM model, the effect on blood glucose levels using its variation of the gluc-HET assay, and the toxicity using a zebrafish embryotoxicity assay. In the HET-CAM model, the encapsulation of epalrestat decreased ocular irritability by lowering the irritability score of free epalrestat in solution from 18.6 to 0. This demonstrated the ability of niosomes to protect the eye from the irritability potential of epalrestat. In order to make sure that the introduction of this drug/carrier complex would not have a worsening effect on the blood glucose levels of a diabetic patient, it is important to make sure that the blood glucose levels remain the same when epalrestat-loaded niosomes are incorporated into the body. In our case, the niosomal formulation decreased the blood glucose levels at the same level as the positive control. The gluc-HET assay showed that all components lower the blood glucose levels in a similar regard as the positive control (−8 to −10%) with the exception of formulation TCD5. Formulation TCD5, however, still maintained blood glucose level reduction (−5%). These results point toward a possible increased efficacy in the context of diabetic treatment. However, as the mechanism in which these formulations lower blood glucose levels in the chorioallantoic membrane, further investigation is required before imagining epalrestat-loaded cationic niosomes as a glucose reduction agent. Finally, the encapsulation of epalrestat in niosomes increased the survivability of zebrafish embryos from 57.5% to 96% when exposed to 5 µL of the solution. Together these results support the potential use of formulations TCD0, TCD5, and TCD10 topically. When compared to other nanoparticles, such as zinc phthalocyanine and aluminum phthalocyanine liposomes, our niosomes performed similarly or better; the liposomes achieved between 100% and 65% or 85% survival rates, respectively [
66]. Interestingly the results of zebrafish embryo survival with ionic-gradient liposomes go in the opposite direction, as the survival rate dropped when encapsulating bupivacaine in the liposomes [
67]. This can be explained by the difference in the release rate of the drugs when encapsulated in niosomes or ionic-gradient liposomes, as the niosome would release epalrestat slower into the zebrafish larvae (2.5–4% for epalrestat against 65–100% for bupivacaine). Furthermore, a positive correlation between DOTMA percentage and mortality was observed (
Table 6), which was to be expected as cationic nanoparticles are known to cause mortality in zebrafish embryos [
68]. Therefore, it was expected that formulations TCD0 and TCD5 would be safer than TCD10.
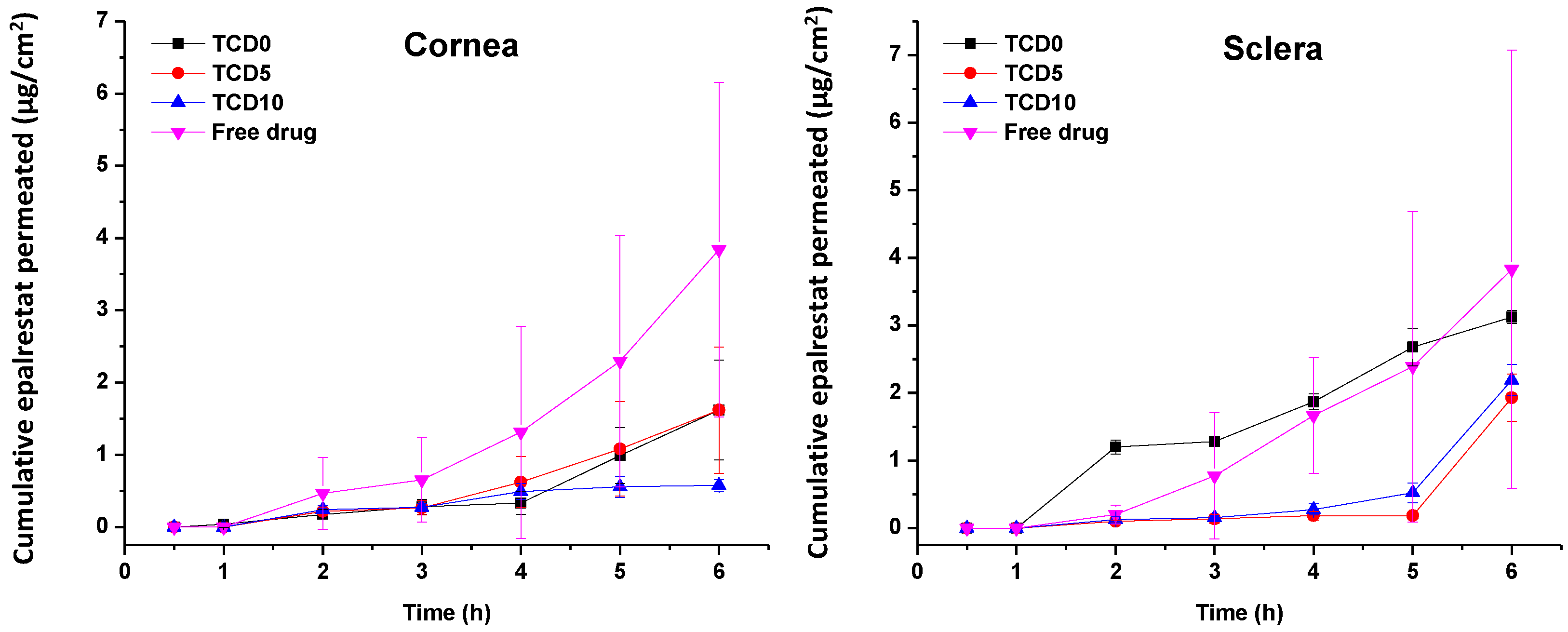
The permeation of epalrestat from the niosomal formulations through the porcine sclera and the cornea was also monitored, reaching 2.5 µg/cm
2 corneal permeation after 6 h in the case of TCD5. When compared to the study utilizing hydrogel lenses to deliver epalrestat [
20], the amount of epalrestat accumulated in the tissues is higher using niosomes. However, it must be noted that our experiment took place on porcine cornea instead of bovine cornea, which is thinner. Also, in the previous study with epalrestat loaded in contact lenses, no drug was detected in the receptor after the 6 h test [
20], which can be related to the lower drug concentration in the donor chamber that the contact lenses were able to supply. Indeed, in that previous study, the epalrestat control solution was 40 µg/mL, namely, five times lower than the control solution of free drug tested in our present study (200 µg/mL). That diluted free epalrestat solution facilitated the accumulation of only 10 µg drug in the bovine cornea [
20], which is approximately 5 times less than the accumulation achieved in the present study. The results indicate that there is a therapeutic amount of epalrestat [
14] that can be delivered to the cornea and the sclera with formulation TCD0. Further investigation needs to be done to confirm the accumulation of epalrestat in further tissues, but the results of our permeation experiments are positive as to the amounts of epalrestat able to traverse the cornea and sclera. The epalrestat steady-state flux obtained for the niosomal formulations is in the same order of magnitude as those recorded for skin formulations prepared with permeability enhancers [
69]. The permeability coefficients recorded for epalrestat in niosomes were also in the range of that recorded for epalrestat solution in the mucosal-to-serosal transport through jejunum [
70]. The lower permeability recorded for the most cationic niosomes through the cornea may be related to a higher binding to the cornea surface, which may hinder further diffusion of the niosomes and also of the drug encapsulated inside. Indeed, cationic liposomes have been demonstrated to prolong the retention time on the ocular surface [
39]. Therefore, the charge density of niosomes should be the result of a compromise between the extended time of permanence on the ocular surface and efficient delivery of epalrestat to the inner eye tissues. It should also be noted that the niosomes have a certain thickening capability increasing 3 to 4 times the viscosity compared to the aqueous medium.
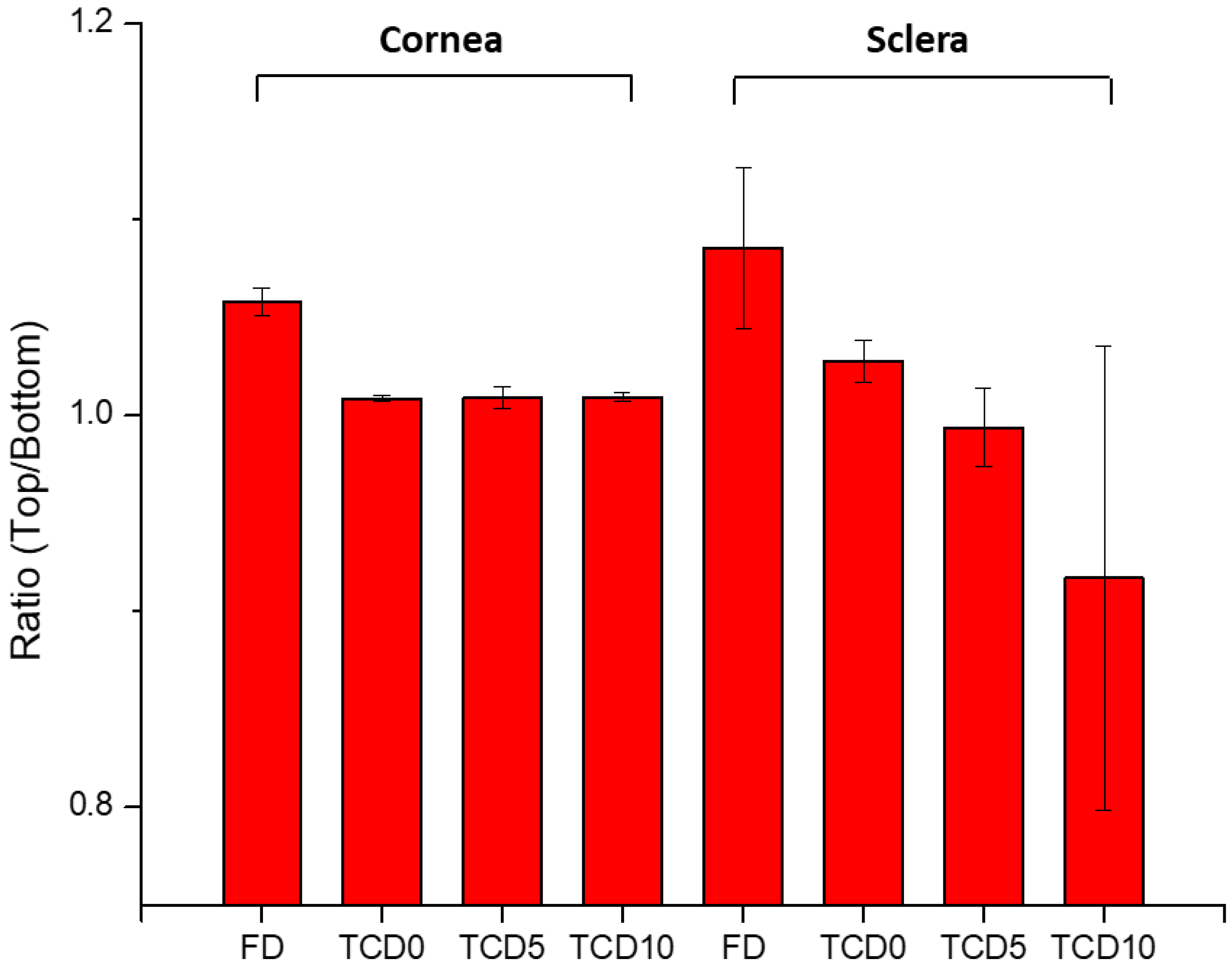
With the help of IR-Raman spectroscopy, drug permeation through the cornea and sclera was evidenced through the whole cross-sections of these tissues. A ratio of the Raman spectrum intensity taken from the top and bottom of the surface of the tissue equal 1, indicating that the concentration of epalrestat was similar on the tissue in contact with the donor as with the receiver chamber after 6 h of application of the formulation. The high values of the ratios of FD cornea (1.06) and FD sclera (1.09) can be explained by the faster access of unencapsulated epalrestat when it is free in solution. The staining of the tissue with the typical yellow color of epalrestat was also perceptible by the naked eye. The low value of the ratio for TCD10 sclera (0.92) is unusual but could be explained by interference from gasses liberated during the analysis process if the sample started to burn. Indeed, the peak of epalrestat situated at 1575 cm
−1 is close to that of carbon (1581 cm
−1) [
71]. This also would explain the great variance in the results between the four measuring points. The heat maps of the cornea and sclera show the presence of epalrestat inside the tissue, permeating in different channels, adding weight to the results obtained from the Franz cell permeation experiments. While Raman spectroscopy has been used on ocular media for the detection of ganciclovir in ocular media in vivo [
72] or for the detection of glutamate in tissues [
73], this is, to our knowledge, the first time IR-Raman was used to confirm drug permeation through ocular tissues.
Epalrestat is a highly hydrophobic drug, and we found that it can be efficiently encapsulated in niosomes. The niosomes increase the drug’s apparent solubility and stability in water, which makes the preparation of eyedrops possible. Moreover, niosomes protect from the irritancy that a pure drug solution causes on the ocular surface. According to our results, the niosomes do not release epalrestat during storage but only when subjected to a strong dilution in the presence of other surfactant molecules, which may resemble the conditions on the ocular surface. The formulation TCD5 seemed to be the most balanced, as it was cationic, had sustained release of epalrestat over a long period, was safe for the ocular surface, and permeated both the cornea and the sclera. The capability to sustain drug release in vitro means that most part of the drug dose remains in niosomes when topically placed on the eye, and niosomes have been reported to enhance drug permeability in vivo [
24]. Some previous reports on drug-loaded niosomes for ocular delivery have also shown in vitro sustained release in the frame of a few days for hydrophobic drugs such as fluconazole [
74] and natamycin [
75] or even mid-polarity drugs such as vancomycin [
76], doxycycline [
77] or naltrexone [
78]. In vivo, studies evidenced the success of the niosome approach compared to the free drug solution, indeed probably due to the more sustained release. Niosomes have been shown to remain on the ocular surface longer than the drug solution. Thus, a sustained release may prevent a very rapid washout from the ocular surface while still creating a drug concentration gradient that facilitates the diffusion through eye tissues [
24].