Design and Characterization of Carboplatin and Paclitaxel Loaded PCL Filaments for 3D Printed Controlled Release Intrauterine Implants
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation and Characterization of Drug:CD Inclusion Complex
2.2.2. Characterization of PCL Polymers
2.2.3. Preparation and Characterization of Filaments
2.2.4. In Vitro cell Culture Studies
2.3. Statistical Analysis
3. Results and Discussion
3.1. Preparation and Characterization of Drug:CD Inclusion Complex
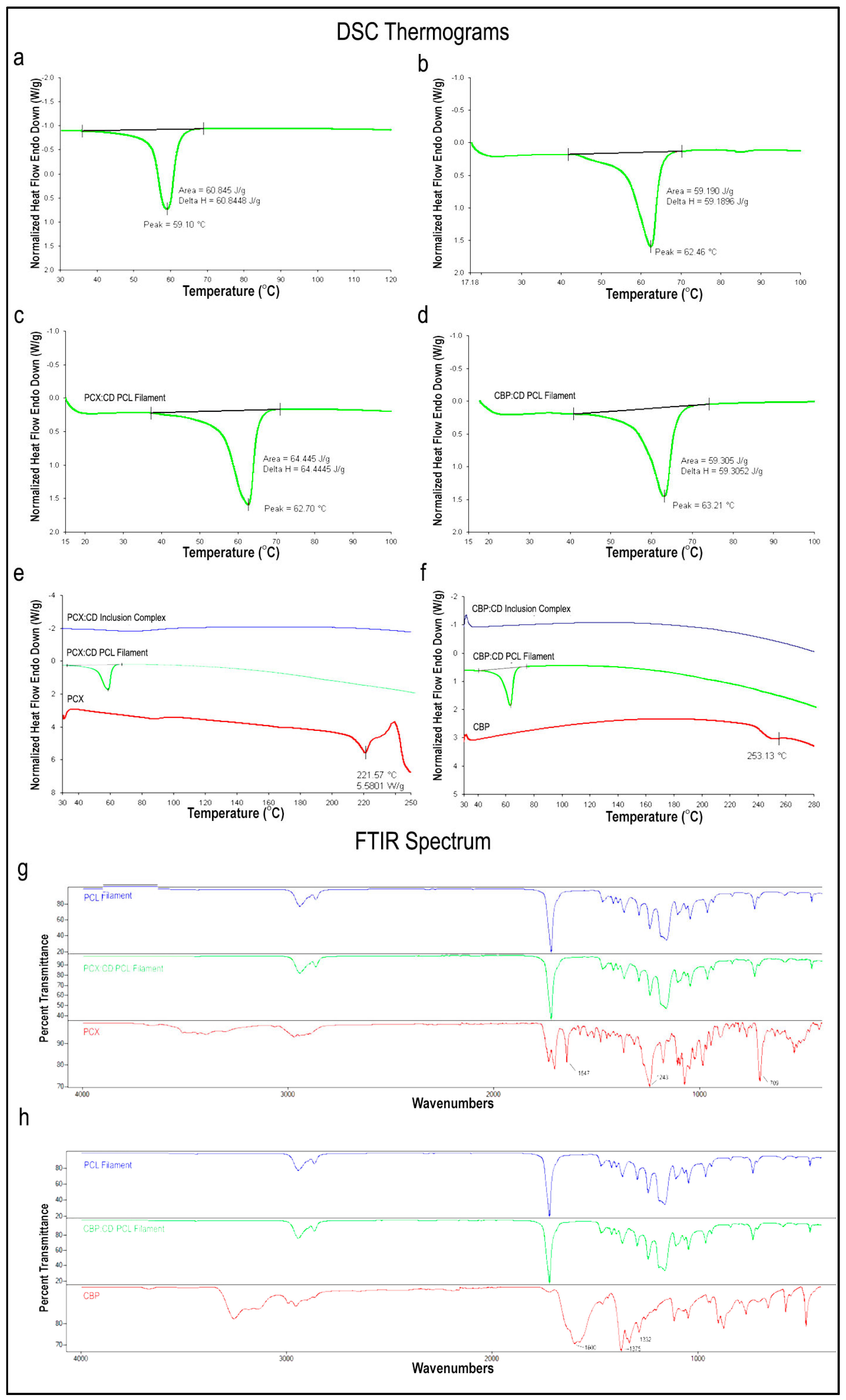
3.1.1. DSC Analysis
3.1.2. FTIR Analysis
3.1.3. SEM Images
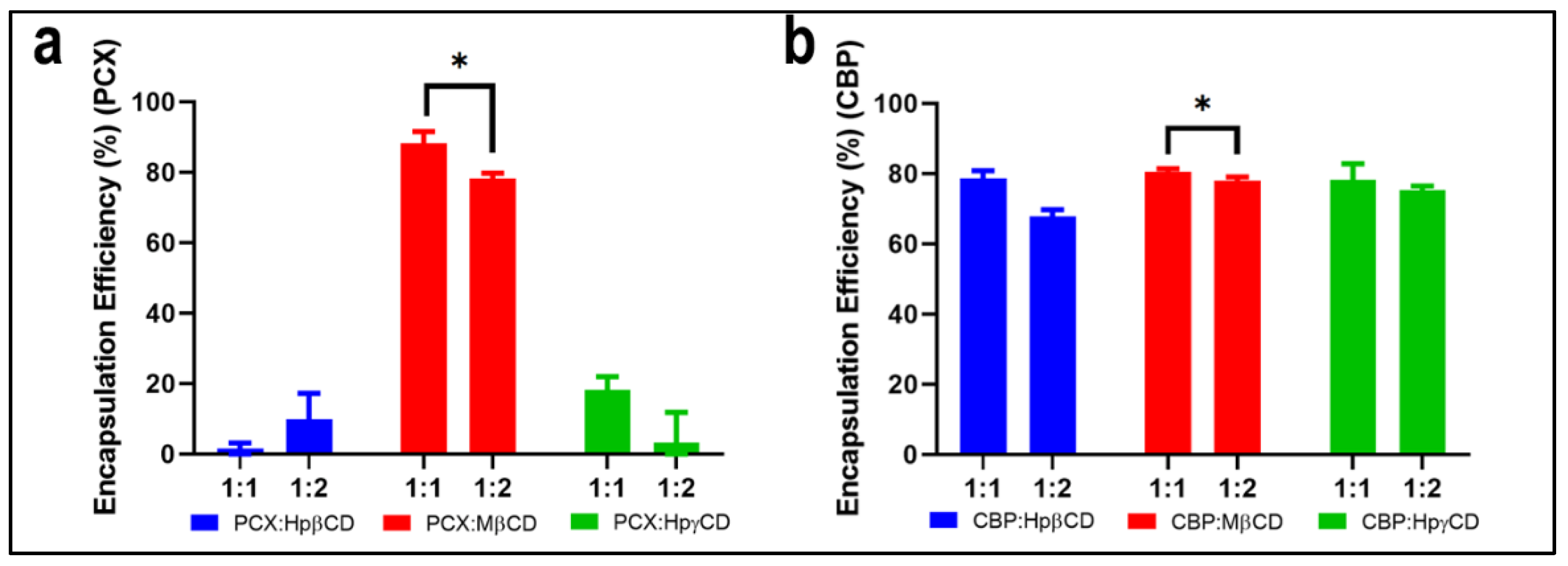
3.1.4. Encapsulation Efficiency
3.2. Characterization of PCL Polymers
3.2.1. Hardness and MFI Analysis
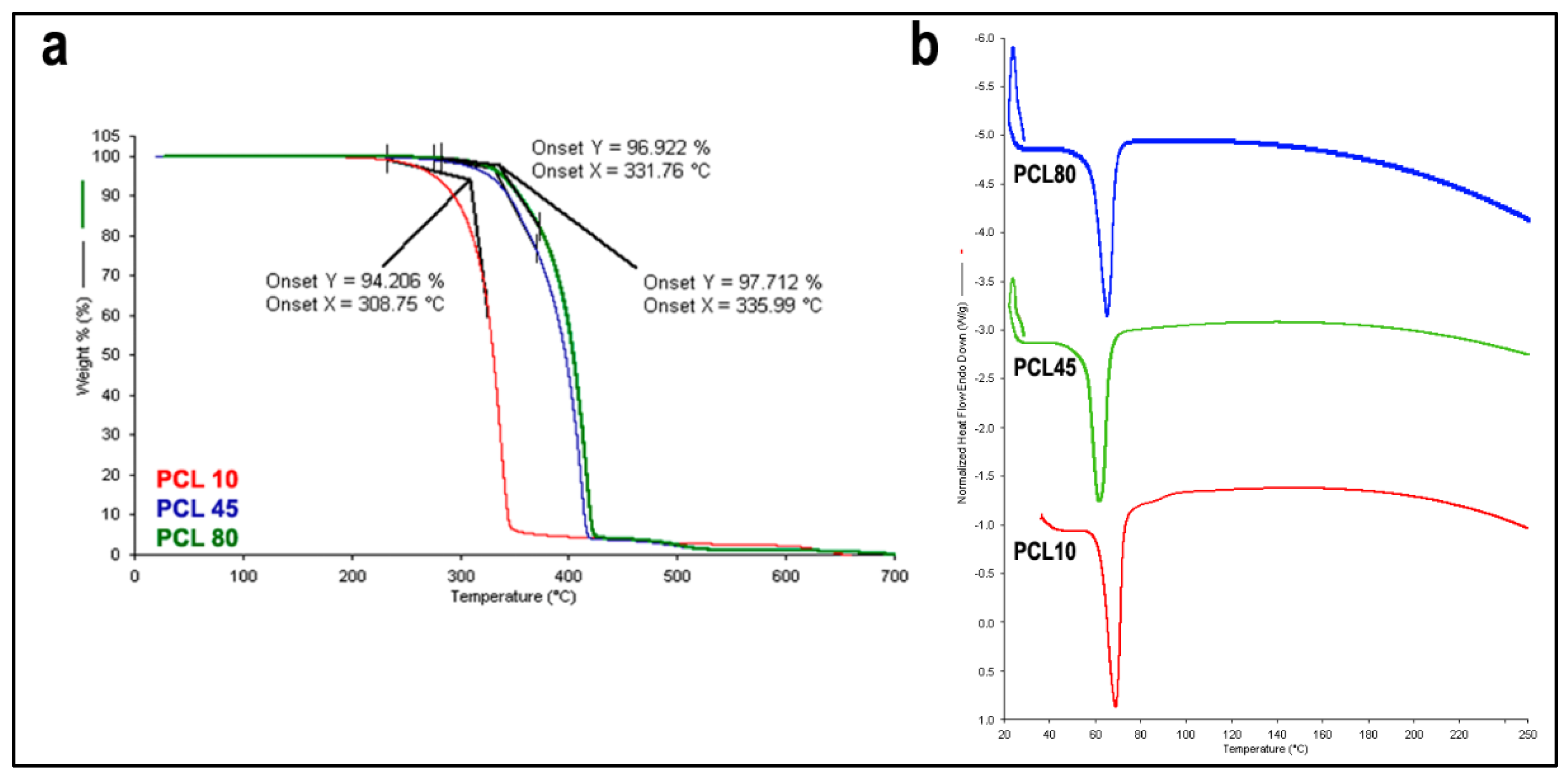
3.2.2. TGA and DSC Analysis
3.3. Preparation and Characterization of Filaments
3.3.1. DSC and FTIR Analysis
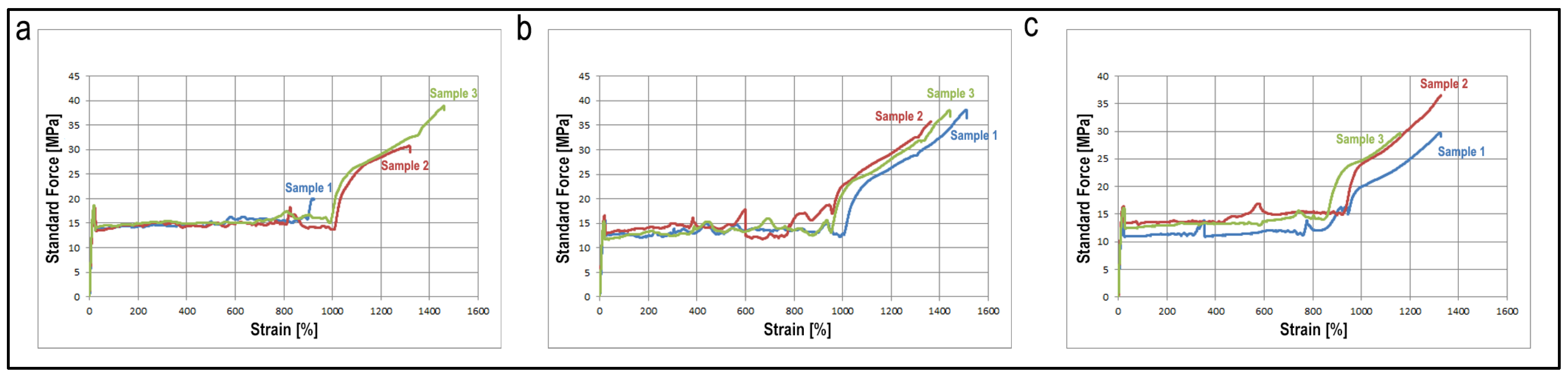
3.3.2. Mechanical Properties
3.3.3. Surface Energy
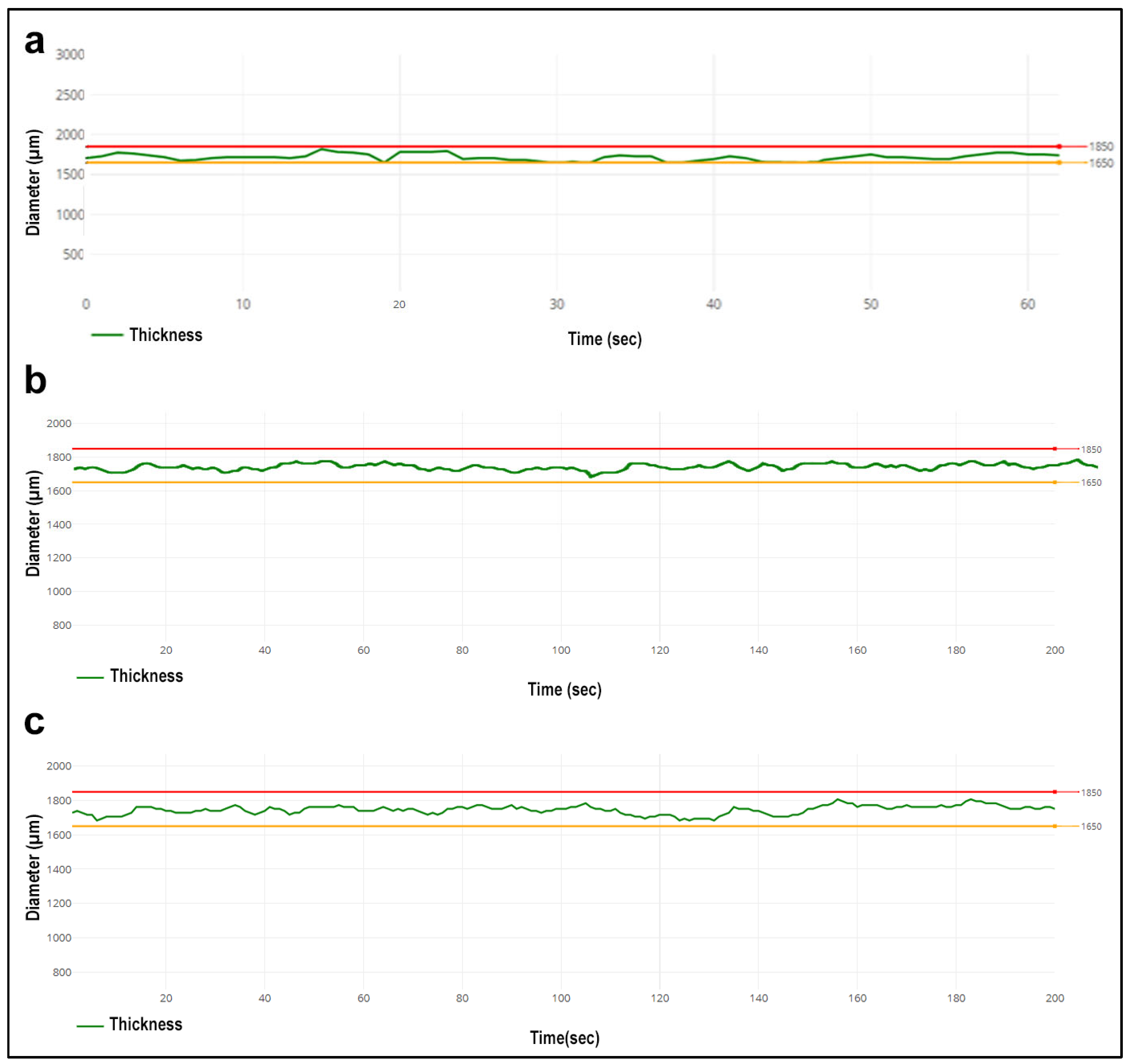
3.3.4. Nano-Level Gaps Analysis
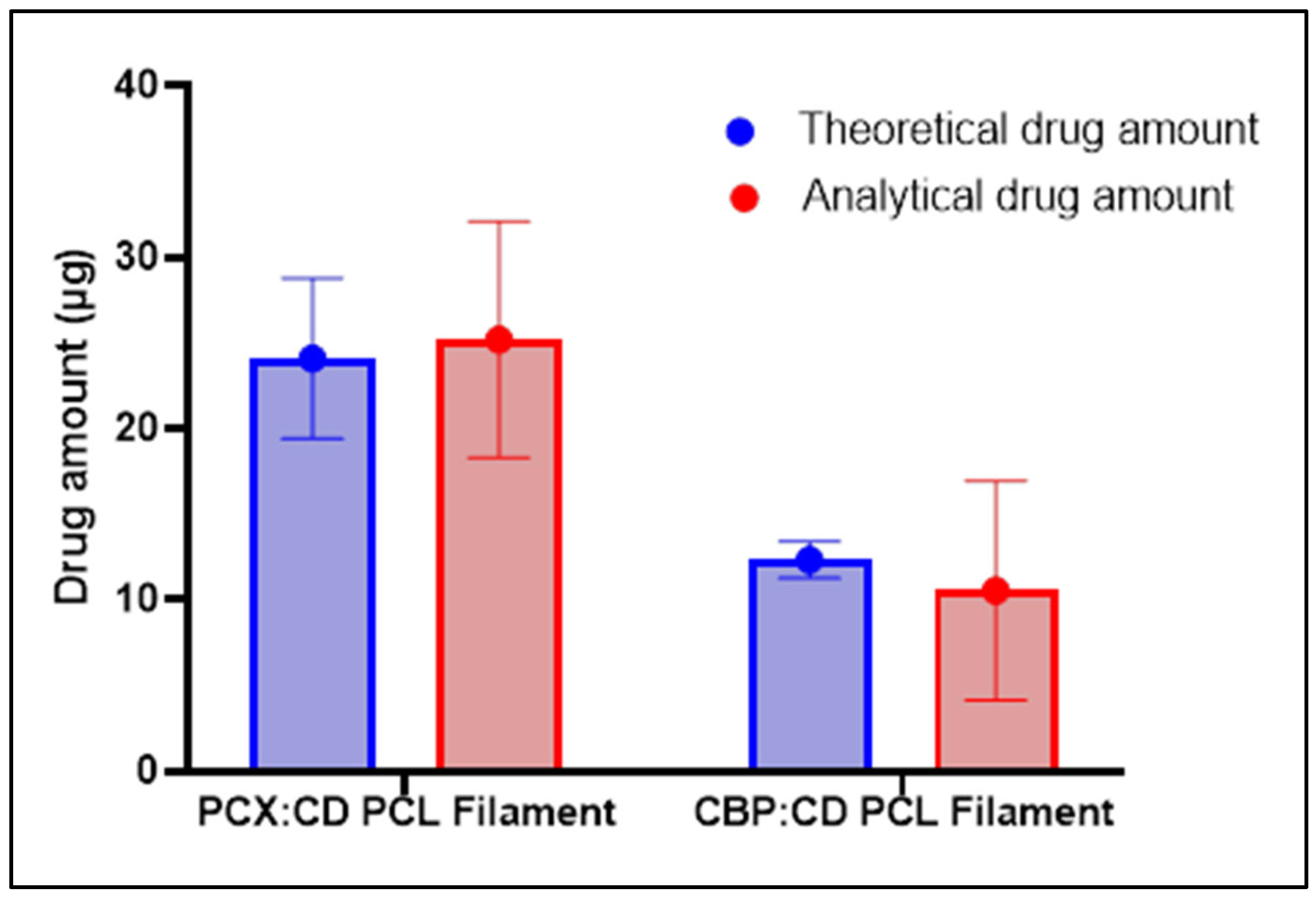
3.3.5. Theoretical and Analytical Drug Amount
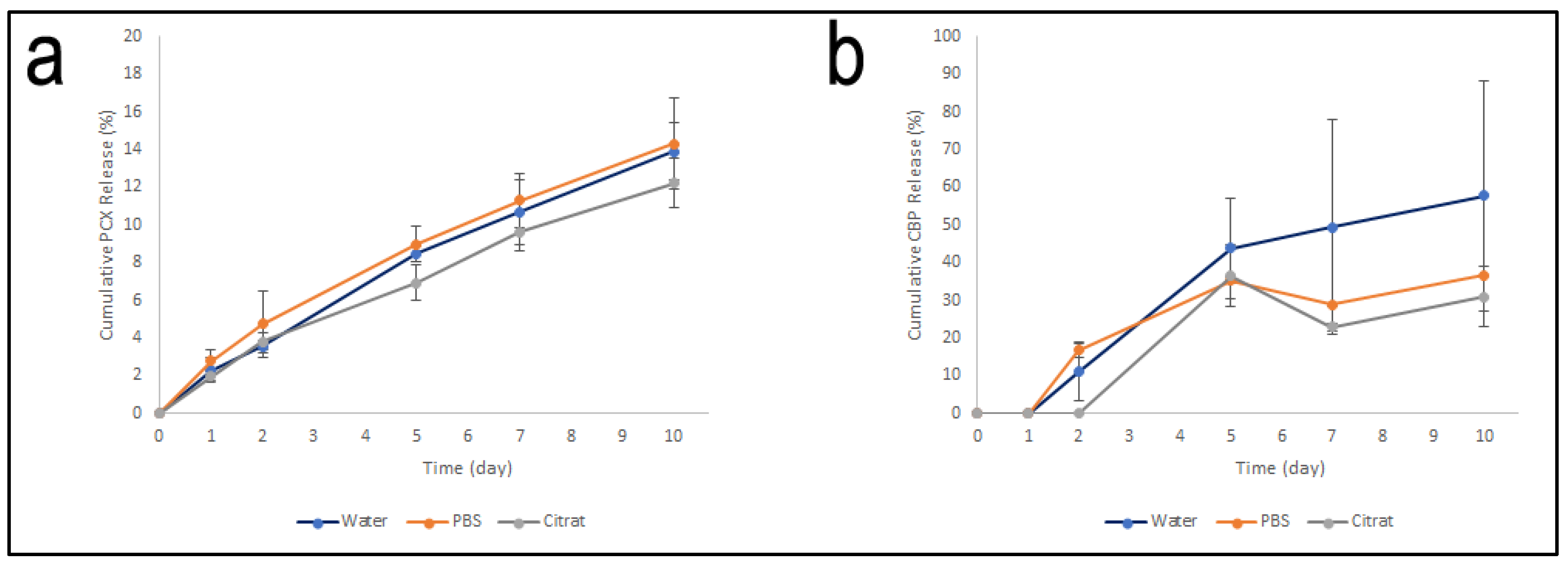
3.3.6. Drug Release Profile
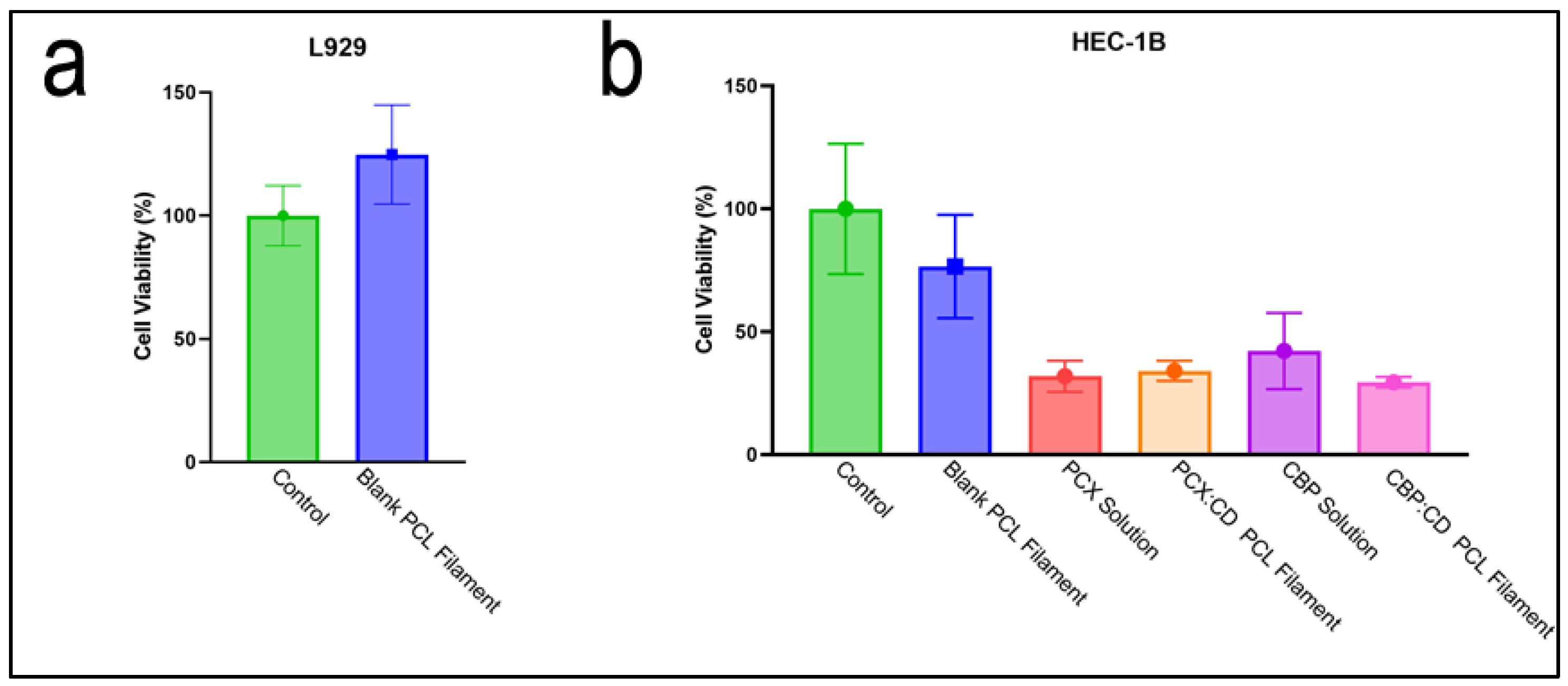
3.4. In Vitro Cell Culture Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA A Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Henley, S.J.; Ward, E.M.; Scott, S.; Ma, J.; Anderson, R.N.; Firth, A.U.; Thomas, C.C.; Islami, F.; Weir, H.K.; Lewis, D.R.; et al. Annual report to the nation on the status of cancer, part I: National cancer statistics. Cancer 2020, 126, 2225–2249. [Google Scholar] [CrossRef] [PubMed]
- Oaknin, A.; Bosse, T.J.; Creutzberg, C.L.; Giornelli, G.; Harter, P.; Joly, F.; Lorusso, D.; Marth, C.; Makker, V.; Mirza, M.R.; et al. Endometrial cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2022, 33, 860–877. [Google Scholar] [CrossRef] [PubMed]
- Bentivegna, E.; Maulard, A.; Miailhe, G.; Gouy, S.; Morice, P. Gynaecologic cancer surgery and preservation of fertility. J. Visc. Surg. 2018, 155, S23–S29. [Google Scholar] [CrossRef]
- Chambers, J.T.; Chambers, S.K.; Kohorn, E.I.; Carcangiu, M.L.; Schwartz, P.E. Uterine papillary serous carcinoma treated with intraperitoneal cisplatin and intravenous doxorubicin and cyclophosphamide. Gynecol. Oncol. 1996, 60, 438–442. [Google Scholar] [CrossRef]
- Ramondetta, L.; Burke, T.W.; Levenback, C.; Bevers, M.; Bodurka-Bevers, D.; Gershenson, D.M. Treatment of uterine papillary serous carcinoma with paclitaxel. Gynecol. Oncol. 2001, 82, 156–161. [Google Scholar] [CrossRef]
- Zanotti, K.M.; Belinson, J.L.; Kennedy, A.W.; Webster, K.D.; Markman, M. The use of paclitaxel and platinum-based chemotherapy in uterine papillary serous carcinoma. Gynecol. Oncol. 1999, 74, 272–277. [Google Scholar] [CrossRef]
- Amant, F.; Moerman, P.; Neven, P.; Timmerman, D.; Van Limbergen, E.; Vergote, I. Endometrial cancer. Lancet 2005, 366, 491–505. [Google Scholar] [CrossRef]
- Goyanes, A.; Kobayashi, M.; Martínez-Pacheco, R.; Gaisford, S.; Basit, A.W. Fused-filament 3D printing of drug products: Microstructure analysis and drug release characteristics of PVA-based caplets. Int. J. Pharm. 2016, 514, 290–295. [Google Scholar] [CrossRef]
- Smith, D.M.; Kapoor, Y.; Klinzing, G.R.; Procopio, A.T. Pharmaceutical 3D printing: Design and qualification of a single step print and fill capsule. Int. J. Pharm. 2018, 544, 21–30. [Google Scholar] [CrossRef]
- Okwuosa, T.C.; Soares, C.; Gollwitzer, V.; Habashy, R.; Timmins, P.; Alhnan, M.A. On demand manufacturing of patient-specific liquid capsules via co-ordinated 3D printing and liquid dispensing. Eur. J. Pharm. Sci. 2018, 118, 134–143. [Google Scholar] [CrossRef]
- Stephen Searle, E. The intrauterine device and the intrauterine system. Best Pract. Res. Clin. Obstet. Gynaecol. 2014, 28, 807–824. [Google Scholar] [CrossRef]
- Holländer, J.; Genina, N.; Jukarainen, H.; Khajeheian, M.; Rosling, A.; Mäkilä, E.; Sandler, N. Three-Dimensional Printed PCL-Based Implantable Prototypes of Medical Devices for Controlled Drug Delivery. J. Pharm. Sci. 2016, 105, 2665–2676. [Google Scholar] [CrossRef]
- Genina, N.; Holländer, J.; Jukarainen, H.; Mäkilä, E.; Salonen, J.; Sandler, N. Ethylene vinyl acetate (EVA) as a new drug carrier for 3D printed medical drug delivery devices. Eur. J. Pharm. Sci. 2016, 90, 53–63. [Google Scholar] [CrossRef]
- Woodruff, M.A.; Hutmacher, D.W. The return of a forgotten polymer—Polycaprolactone in the 21st century. Prog. Polym. Sci. 2010, 35, 1217–1256. [Google Scholar] [CrossRef]
- Wong, B.S.; Teoh, S.H.; Kang, L. Polycaprolactone scaffold as targeted drug delivery system and cell attachment scaffold for postsurgical care of limb salvage. Drug Deliv. Transl. Res. 2012, 2, 272–283. [Google Scholar] [CrossRef]
- Mondal, D.; Griffith, M.; Venkatraman, S.S. Polycaprolactone-based biomaterials for tissue engineering and drug delivery: Current scenario and challenges. Int. J. Polym. Mater. Polym. Biomater. 2016, 65, 255–265. [Google Scholar] [CrossRef]
- Bilensoy, E.; Gürkaynak, O.; Ertan, M.; Şen, M.; Hıncal, A.A. Development of Nonsurfactant Cyclodextrin Nanoparticles Loaded With Anticancer Drug Paclitaxel. J. Pharm. Sci. 2008, 97, 1519–1529. [Google Scholar] [CrossRef]
- Varan, C.; Wickström, H.; Sandler, N.; Aktaş, Y.; Bilensoy, E. Inkjet printing of antiviral PCL nanoparticles and anticancer cyclodextrin inclusion complexes on bioadhesive film for cervical administration. Int. J. Pharm. 2017, 531, 701–713. [Google Scholar] [CrossRef]
- Çirpanli, Y.; Bilensoy, E.; Lale Doğan, A.; Çaliş, S. Comparative evaluation of polymeric and amphiphilic cyclodextrin nanoparticles for effective camptothecin delivery. Eur. J. Pharm. Biopharm. 2009, 73, 82–89. [Google Scholar] [CrossRef]
- Pitha, J.; Milecki, J.; Fales, H.; Pannell, L.; Uekama, K. Hydroxypropyl-β-cyclodextrin: Preparation and characterization; effects on solubility of drugs. Int. J. Pharm. 1986, 29, 73–82. [Google Scholar] [CrossRef]
- Loftsson, T.; Jarho, P.; Másson, M.; Järvinen, T. Cyclodextrins in drug delivery. Expert Opin. Drug Deliv. 2005, 2, 335–351. [Google Scholar] [CrossRef] [PubMed]
- Cid-Samamed, A.; Rakmai, J.; Mejuto, J.C.; Simal-Gandara, J.; Astray, G. Cyclodextrins inclusion complex: Preparation methods, analytical techniques and food industry applications. Food Chem. 2022, 384, 132467. [Google Scholar] [CrossRef] [PubMed]
- Mastropaolo, D.; Camerman, A.; Luo, Y.; Brayer, G.D.; Camerman, N. Crystal and molecular structure of paclitaxel (taxol). Proc. Natl. Acad. Sci. USA 1995, 92, 6920–6924. [Google Scholar] [CrossRef]
- Pattekari, P.; Zheng, Z.; Zhang, X.; Levchenko, T.; Torchilin, V.; Lvov, Y. Top-down and bottom-up approaches in production of aqueous nanocolloids of low solubility drug paclitaxel. Phys. Chem. Chem. Phys. 2011, 13, 9014–9019. [Google Scholar] [CrossRef]
- Fu, J.; Yu, X.; Jin, Y. 3D printing of vaginal rings with personalized shapes for controlled release of progesterone. Int. J. Pharm. 2018, 539, 75–82. [Google Scholar] [CrossRef]
- Machado, R.M.; Palmeira-De-Oliveira, A.; Martinez-De-Oliveira, J.; Palmeira-De-Oliveira, R. Vaginal Films for Drug Delivery. J. Pharm. Sci. 2013, 102, 2069–2081. [Google Scholar] [CrossRef]
- Varan, C.; Şen, M.; Sandler, N.; Aktaş, Y.; Bilensoy, E. Mechanical characterization and ex vivo evaluation of anticancer and antiviral drug printed bioadhesive film for the treatment of cervical cancer. Eur. J. Pharm. Sci. 2019, 130, 114–123. [Google Scholar] [CrossRef]
- Kansy, J. Microcomputer program for analysis of positron annihilation lifetime spectra. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrometers Detect. Assoc. Equip. 1996, 374, 235–244. [Google Scholar] [CrossRef]
- Llorina Rañada, M.; Akbulut, M.; Abad, L.; Güven, O. Molecularly imprinted poly(N-vinyl imidazole) based polymers grafted onto nonwoven fabrics for recognition/removal of phloretic acid. Radiat. Phys. Chem. 2014, 94, 93–97. [Google Scholar] [CrossRef]
- Söylemez, M.A.; Güven, O. Detailed positron annihilation lifetime spectroscopic investigation of atrazine imprinted polymers grafted onto PE/PP non-woven fabrics. J. Mol. Recognit. 2018, 31, e2676. [Google Scholar] [CrossRef]
- Martins, K.F.; Messias, A.D.; Leite, F.L.; Duek, E.A.R. Preparation and characterization of paclitaxel-loaded PLDLA microspheres. Mater. Res. 2014, 17, 650–656. [Google Scholar] [CrossRef]
- Yu, K.; Zhao, J.; Yu, C.; Sun, F.; Liu, Y.; Zhang, Y.; Lee, R.J.; Teng, L.; Li, Y. Role of Four Different Kinds of Polyethylenimines (PEIs) in Preparation of Polymeric Lipid Nanoparticles and Their Anticancer Activity Study. J. Cancer 2016, 7, 872–882. [Google Scholar] [CrossRef]
- Alex, A.T.; Joseph, A.; Shavi, G.; Rao, J.V.; Udupa, N. Development and evaluation of carboplatin-loaded PCL nanoparticles for intranasal delivery. Drug Deliv. 2016, 23, 2144–2153. [Google Scholar] [CrossRef]
- Mahmud, F.; Chung, S.W.; Alam, F.; Choi, J.U.; Kim, S.W.; Kim, I.-S.; Kim, S.Y.; Lee, D.S.; Byun, Y. Metronomic chemotherapy using orally active carboplatin/deoxycholate complex to maintain drug concentration within a tolerable range for effective cancer management. J. Control. Release 2017, 249, 42–52. [Google Scholar] [CrossRef]
- Ye, Y.J.; Wang, Y.; Lou, K.Y.; Chen, Y.Z.; Chen, R.; Gao, F. The preparation, characterization, and pharmacokinetic studies of chitosan nanoparticles loaded with paclitaxel/dimethyl-β-cyclodextrin inclusion complexes. Int. J. Nanomed. 2015, 10, 4309–4319. [Google Scholar] [CrossRef]
- Ho, M.N.; Bach, L.G.; Nguyen, T.H.; Ho, M.H.; Nguyen, D.H.; Nguyen, C.K.; Nguyen, C.H.; Nguyen, N.V.; Hoang Thi, T.T. PEGylated poly(amidoamine) dendrimers-based drug loading vehicles for delivering carboplatin in treatment of various cancerous cells. J. Nanopart. Res. 2019, 21, 43. [Google Scholar] [CrossRef]
- Luna, C.B.B.; Siqueira, D.D.; Ferreira, E.d.S.B.; Araújo, E.M.; Wellen, R.M.R. Reactive compatilization of PCL/WP upon addition of PCL-MA. Smart option for recycling industry. Mater. Res. Express 2019, 6, 125317. [Google Scholar] [CrossRef]
- Alhijjaj, M.; Nasereddin, J.; Belton, P.; Qi, S. Impact of Processing Parameters on the Quality of Pharmaceutical Solid Dosage Forms Produced by Fused Deposition Modeling (FDM). Pharmaceutics 2019, 11, 633. [Google Scholar] [CrossRef]
- Gopi, S.; Kontopoulou, M. Investigation of thermoplastic melt flow and dimensionless groups in 3D bioplotting. Rheol. Acta 2020, 59, 83–93. [Google Scholar] [CrossRef]
- Alemán-Domínguez, M.E.; Ortega, Z.; Benítez, A.N.; Monzón, M.; Garzón, L.V.; Ajami, S.; Liu, C. Polycaprolactone–carboxymethyl cellulose composites for manufacturing porous scaffolds by material extrusion. Bio Des. Manuf. 2018, 1, 245–253. [Google Scholar] [CrossRef]
- Unger, M.; Vogel, C.; Siesler, H.W. Molecular weight dependence of the thermal degradation of poly(epsilon-caprolactone): A thermogravimetric differential thermal Fourier transform infrared spectroscopy study. Appl. Spectrosc. 2010, 64, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Asgreen, C.; Knopp, M.M.; Skytte, J.; Löbmann, K. Influence of the Polymer Glass Transition Temperature and Molecular Weight on Drug Amorphization Kinetics Using Ball Milling. Pharmaceutics 2020, 12, 483. [Google Scholar] [CrossRef] [PubMed]
- Carmona, V.B.; Corrêa, A.C.; Marconcini, J.M.; Mattoso, L.H.C. Properties of a Biodegradable Ternary Blend of Thermoplastic Starch (TPS), Poly(ε-Caprolactone) (PCL) and Poly(Lactic Acid) (PLA). J. Polym. Environ. 2015, 23, 83–89. [Google Scholar] [CrossRef]
- Jana, S.; Leung, M.; Chang, J.; Zhang, M. Effect of nano- and micro-scale topological features on alignment of muscle cells and commitment of myogenic differentiation. Biofabrication 2014, 6, 035012. [Google Scholar] [CrossRef]
- Lozano-Sánchez, L.M.; Bagudanch, I.; Sustaita, A.O.; Iturbe-Ek, J.; Elizalde, L.E.; Garcia-Romeu, M.L.; Elías-Zúñiga, A. Single-Point Incremental Forming of Two Biocompatible Polymers: An Insight into Their Thermal and Structural Properties. Polymers 2018, 10, 391. [Google Scholar] [CrossRef]
- Manoj Prabhakar, M.; Saravanan, A.K.; Haiter Lenin, A.; Jerin Leno, I.; Mayandi, K.; Sethu Ramalingam, P. A short review on 3D printing methods, process parameters and materials. Mater. Today Proc. 2021, 45, 6108–6114. [Google Scholar] [CrossRef]
- Goyanes, A.; Det-Amornrat, U.; Wang, J.; Basit, A.W.; Gaisford, S. 3D scanning and 3D printing as innovative technologies for fabricating personalized topical drug delivery systems. J. Control. Release 2016, 234, 41–48. [Google Scholar] [CrossRef]
- Weng, Z.; Wang, J.; Senthil, T.; Wu, L. Mechanical and thermal properties of ABS/montmorillonite nanocomposites for fused deposition modeling 3D printing. Mater. Des. 2016, 102, 276–283. [Google Scholar] [CrossRef]
- Çevik, Ü.; Kam, M. A Review Study on Mechanical Properties of Obtained Products by FDM Method and Metal/Polymer Composite Filament Production. J. Nanomater. 2020, 2020, 6187149. [Google Scholar] [CrossRef]
- Hamelers, I.H.L.; van Loenen, E.; Staffhorst, R.W.H.M.; de Kruijff, B.; de Kroon, A.I.P.M. Carboplatin nanocapsules: A highly cytotoxic, phospholipid-based formulation of carboplatin. Mol. Cancer Ther. 2006, 5, 2007–2012. [Google Scholar] [CrossRef]



| Shore D | MFI (g/10 min) | |
|---|---|---|
| PCL10 (Mn = 10,000) | 51.0 ± 1.0 | 265.2 ± 1.4 |
| PCL45 (Mn = 45,000) | 52.2 ± 0.8 | 15.4 ± 0.7 |
| PCL80 (Mn = 80,000) | 53.6 ± 0.5 | 5.7 ± 0.1 |
| Blank PCL Filament | PCX:CD PCL Filament | CBP:CD PCL Filament | |
|---|---|---|---|
| Emod (MPa) | 169.6 ± 17.8 | 144.3 ± 21.8 | 159.9 ± 11.7 |
| F at 0.2% plastic strain (N) | 11.3 ± 2.3 | 7.0 ± 0.7 | 8.4 ± 1.5 |
| Fbreak (MPa) | 29.2 ± 9.1 | 36.3 ± 0.5 | 33.4 ± 7.3 |
| dL at break (%) | 1234.8 ± 278.3 | 1440.2 ± 74.5 | 1314.0 ± 148.1 |
| Surface Free Energy of Filaments | ||||
|---|---|---|---|---|
| γSLW | γS+ | γS− | γSTotal | |
| Blank PCL Filament | 25.02 | 0.41 | 25.43 | 50.9 |
| PCX:CD PCL Filament | 24.51 | 1.01 | 7.02 | 32.5 |
| CBP:CD PCL Filament | 24.81 | 0.66 | 17.52 | 43.0 |
| Gaps (nm) | |
|---|---|
| Blank PCL Filament | 0.325 |
| PCX:CD PCL Filament | 0.509 |
| CBP:CD PCL Filament | 0.492 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varan, C.; Aksüt, D.; Şen, M.; Bilensoy, E. Design and Characterization of Carboplatin and Paclitaxel Loaded PCL Filaments for 3D Printed Controlled Release Intrauterine Implants. Pharmaceutics 2023, 15, 1154. https://doi.org/10.3390/pharmaceutics15041154
Varan C, Aksüt D, Şen M, Bilensoy E. Design and Characterization of Carboplatin and Paclitaxel Loaded PCL Filaments for 3D Printed Controlled Release Intrauterine Implants. Pharmaceutics. 2023; 15(4):1154. https://doi.org/10.3390/pharmaceutics15041154
Chicago/Turabian StyleVaran, Cem, Davut Aksüt, Murat Şen, and Erem Bilensoy. 2023. "Design and Characterization of Carboplatin and Paclitaxel Loaded PCL Filaments for 3D Printed Controlled Release Intrauterine Implants" Pharmaceutics 15, no. 4: 1154. https://doi.org/10.3390/pharmaceutics15041154
APA StyleVaran, C., Aksüt, D., Şen, M., & Bilensoy, E. (2023). Design and Characterization of Carboplatin and Paclitaxel Loaded PCL Filaments for 3D Printed Controlled Release Intrauterine Implants. Pharmaceutics, 15(4), 1154. https://doi.org/10.3390/pharmaceutics15041154