Virus-like Particle Vaccine Expressing the Respiratory Syncytial Virus Pre-Fusion and G Proteins Confers Protection against RSV Challenge Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Cells, Antibodies, and Viruses
2.3. Generation of rBV and VLPs
2.4. Characterization of RSV VLPs
2.5. Immunization and Challenge Infection
2.6. Virus-Specific Antibody Responses
2.7. Flow Cytometry and Cytokine Assays
2.8. Pulmonary Histopathology
2.9. Neutralizing Activity Analysis and Virus Titer
2.10. Statistical Analysis
3. Results
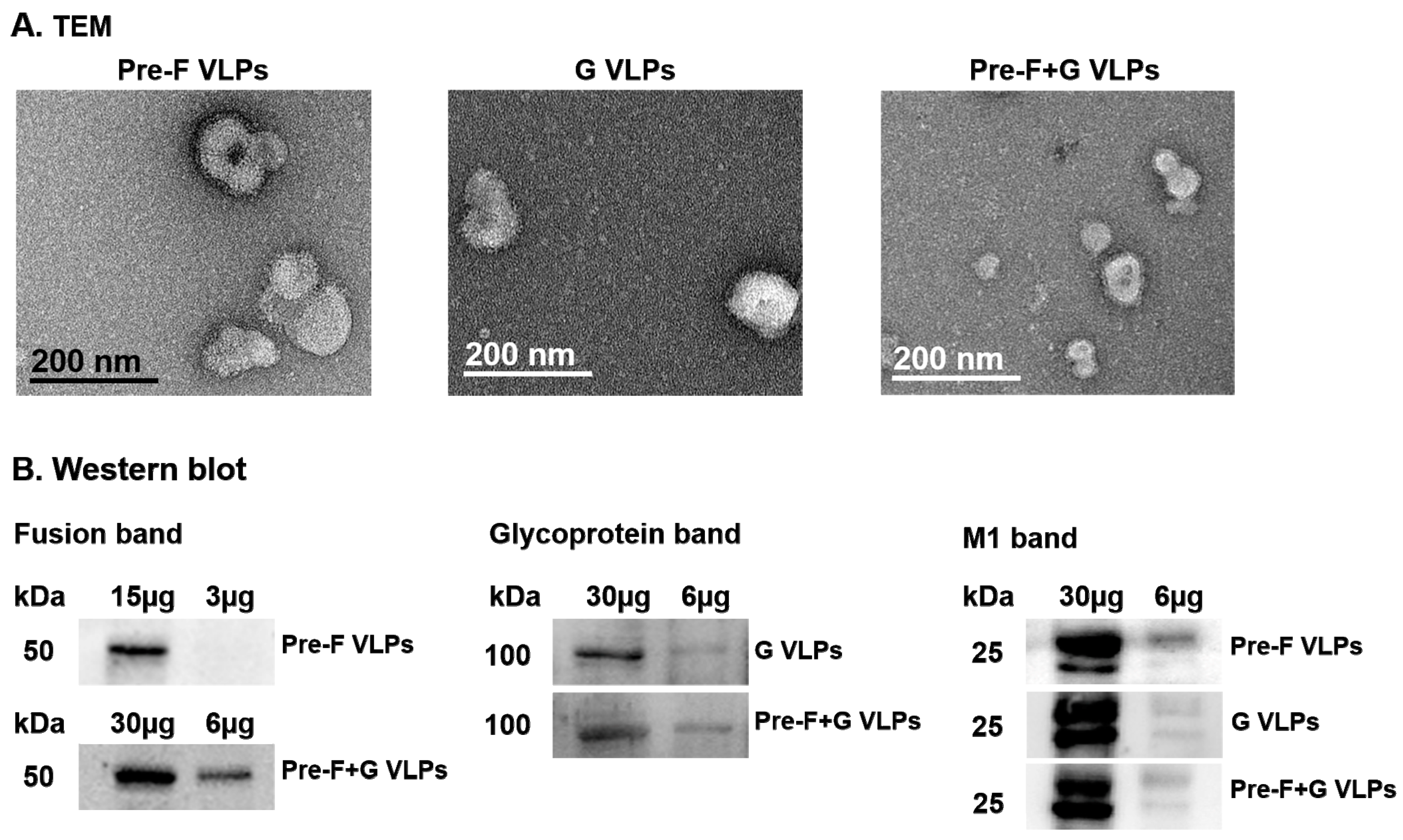
3.1. Immunoblotting and Electron Microscopy Characterization of VLPs
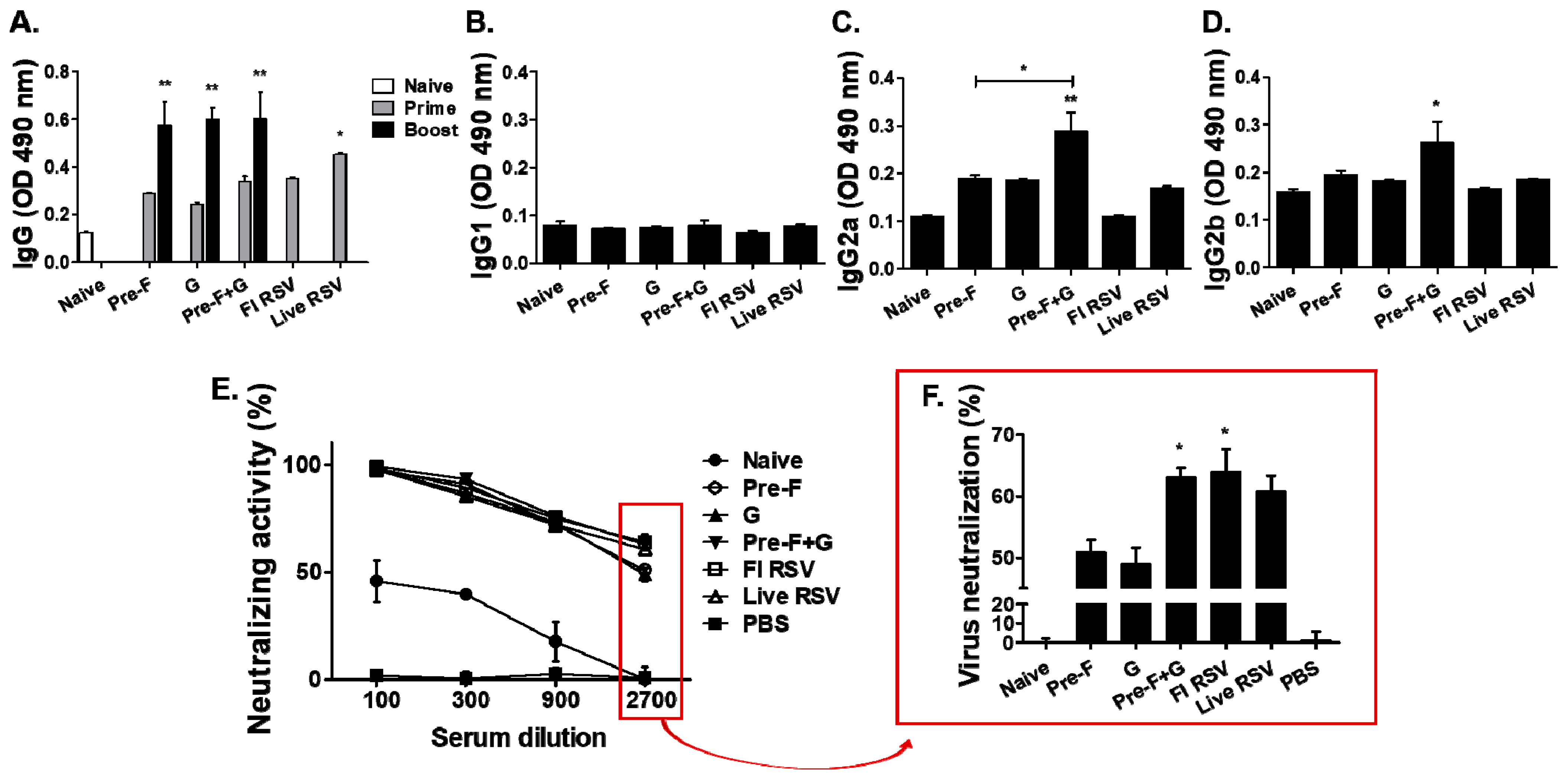
3.2. Antibody and Neutralization Responses in Serum
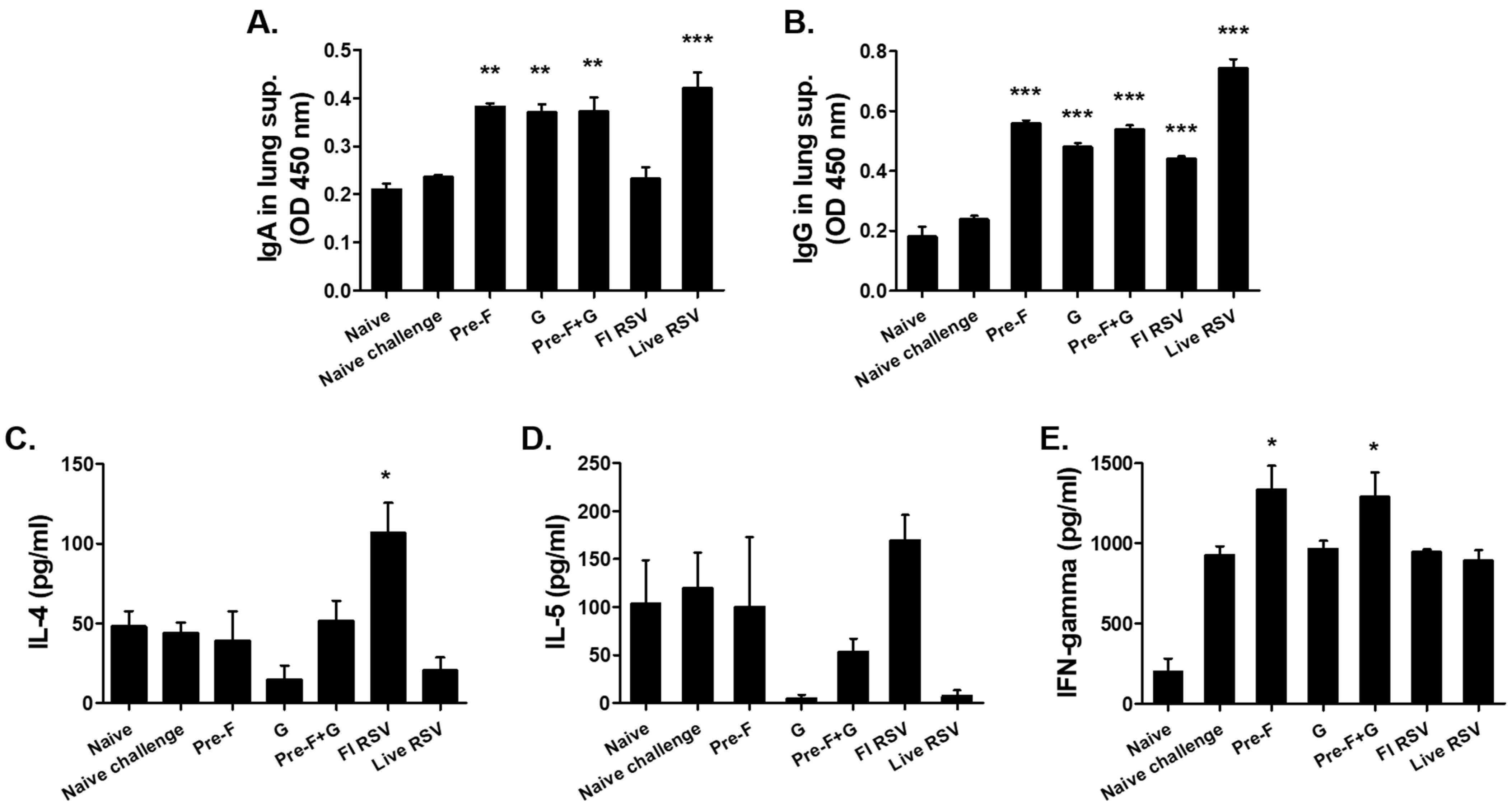
3.3. VLP Immunization Induced Antibody Response in the Lungs and Splenic Cytokine Responses
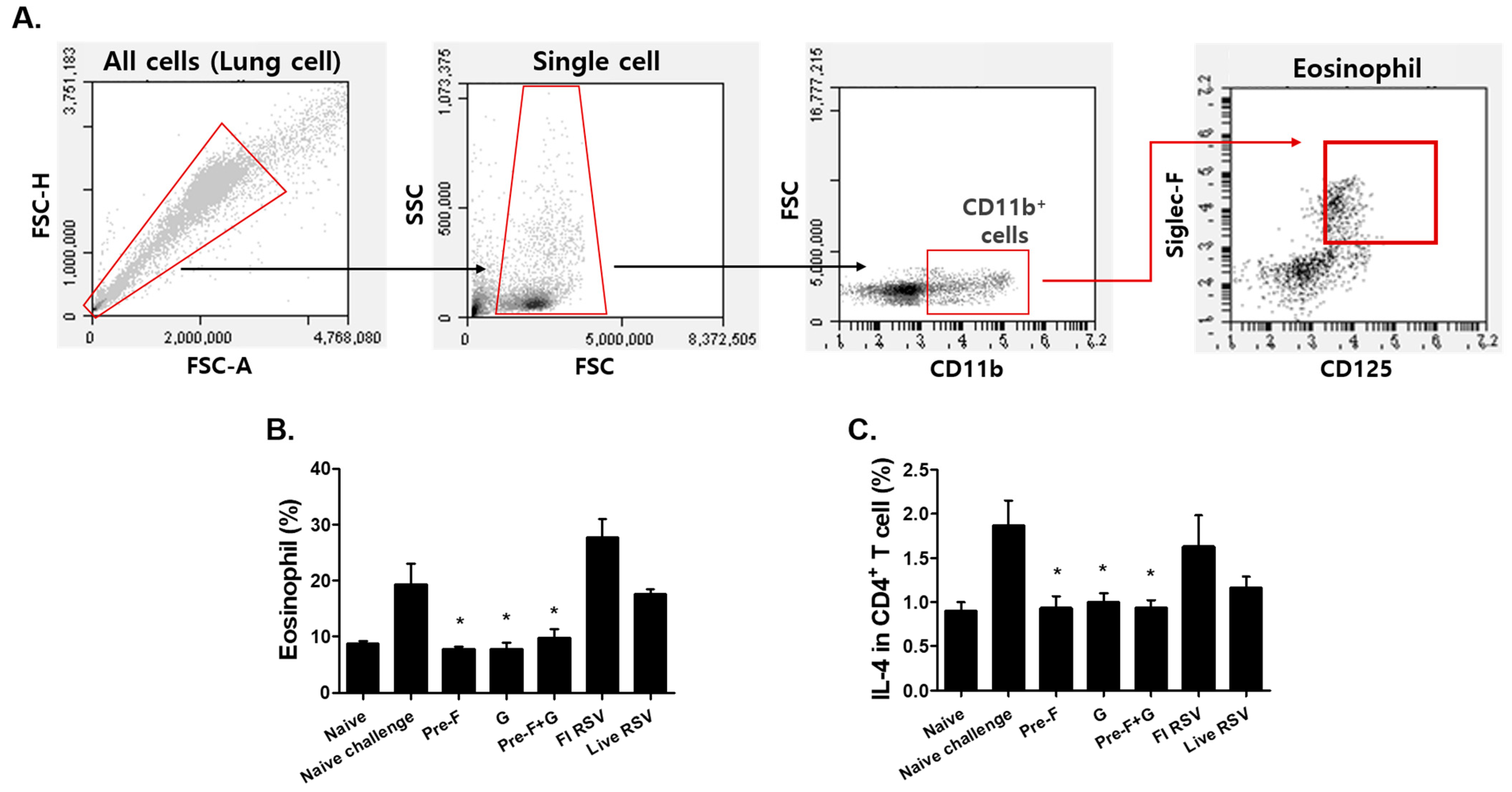
3.4. VLP Immunization Reduced Eosinophil and IL-4-Producing CD4+ T Cell Counts
3.5. Pre-F+G VLP Immunization Induced Cellular Immune Responses
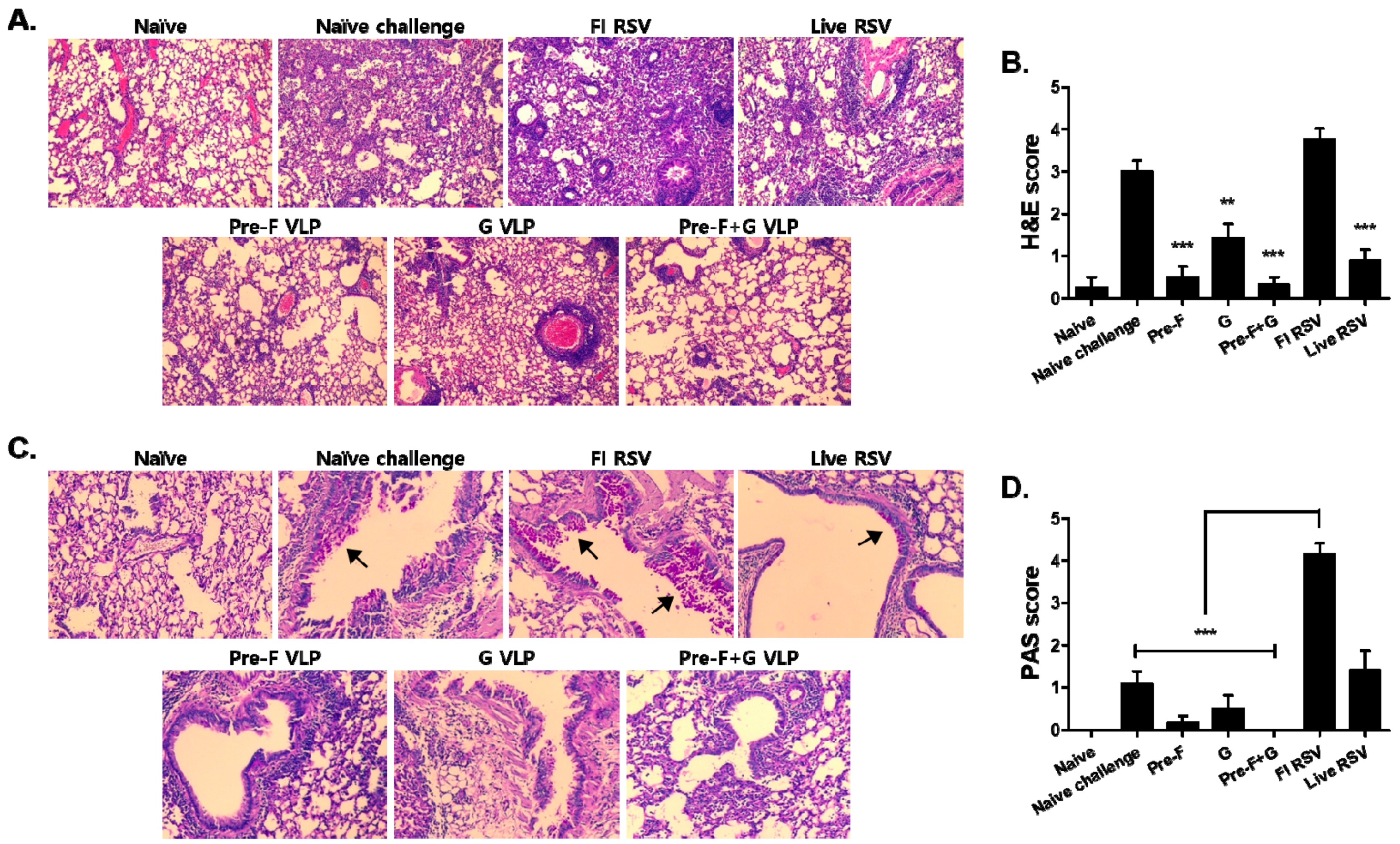
3.6. VLP Immunization Reduced Lung Inflammation
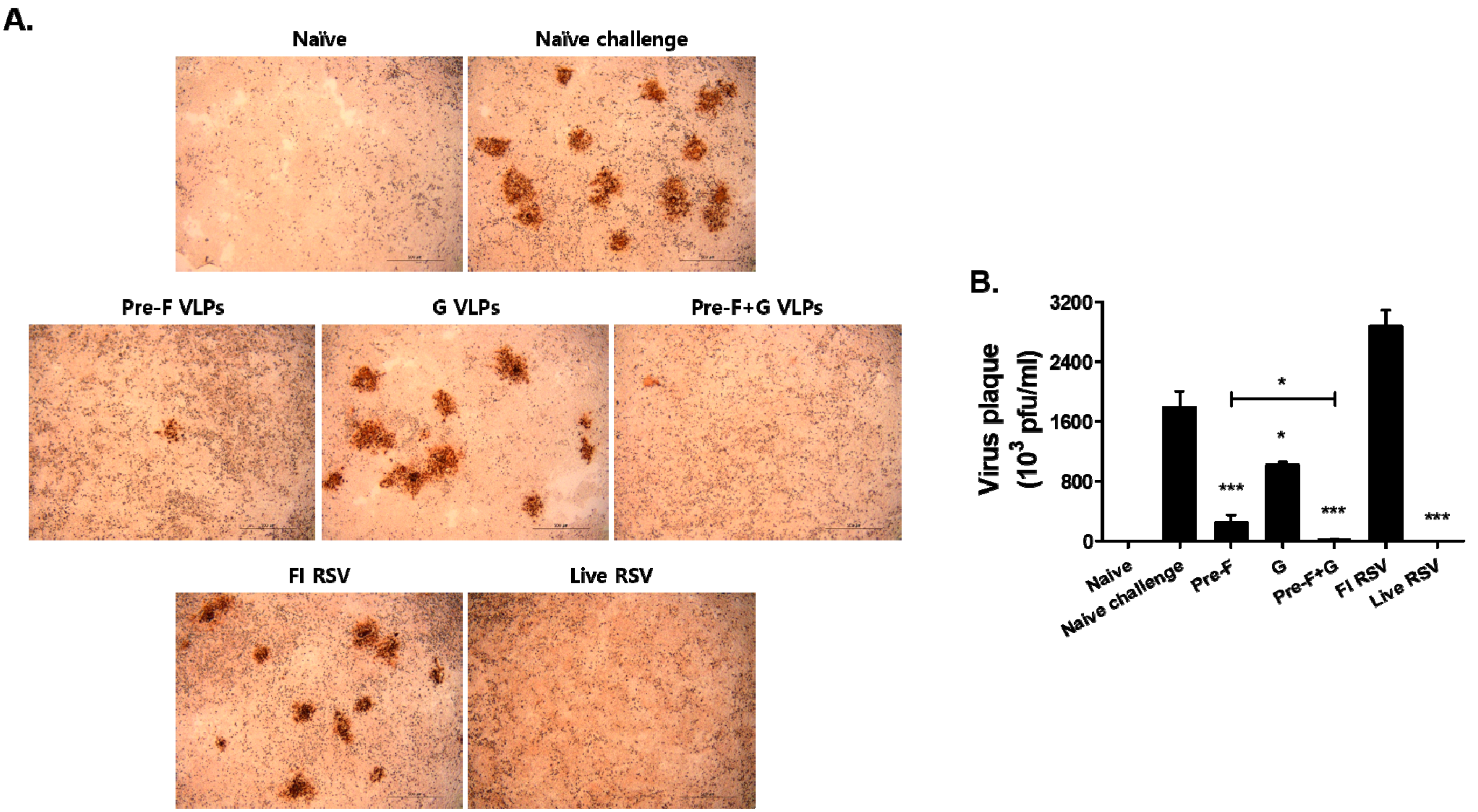
3.7. VLP Immunization Reduced Lung Viral Titers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Collins, P.L.; Fearns, R.; Graham, B.S. Respiratory syncytial virus: Virology, reverse genetics, and pathogenesis of disease. In Challenges and Opportunities for Respiratory Syncytial Virus Vaccines; Springer: Berlin/Heidelberg, Germany, 2013; pp. 3–38. [Google Scholar]
- Torsvik, V.; Øvreås, L.; Thingstad, T.F. Prokaryotic Diversity--Magnitude, Dynamics, and Controlling Factors. Science 2002, 296, 1064–1066. [Google Scholar] [CrossRef]
- Hall, C.B.; Simőes, E.A.; Anderson, L.J. Clinical and epidemiologic features of respiratory syncytial virus. In Challenges and Opportunities for Respiratory Syncytial Virus Vaccines; Springer: Berlin/Heidelberg, Germany, 2013; pp. 39–57. [Google Scholar]
- Kim, H.W.; Arrobio, J.O.; Brandt, C.D.; Jeffries, B.C.; Pyles, G.; Reid, J.L.; Chanock, R.M.; Parrott, R.H. Epidemiology of Respiratory Syncytial Virus Infection in Washington, DC: I. Importance of the Virus in Different Respiratory Tract Disease Syndromes and Temporal Distribution of Infection. Am. J. Epidemiol. 1973, 98, 216–225. [Google Scholar] [CrossRef]
- Shi, T.; McAllister, D.A.; O’Brien, K.L.; Simoes, E.A.; Madhi, S.A.; Gessner, B.D.; Polack, F.P.; Balsells, E.; Acacio, S.; Aguayo, C. Global, Regional, and National Disease Burden Estimates of Acute Lower Respiratory Infections due to Respiratory Syncytial Virus in Young Children in 2015: A Systematic Review and Modelling Study. Lancet 2017, 390, 946–958. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Blau, D.M.; Caballero, M.T.; Feikin, D.R.; Gill, C.J.; Madhi, S.A.; Omer, S.B.; Simões, E.A.; Campbell, H. Global, Regional, and National Disease Burden Estimates of Acute Lower Respiratory Infections due to Respiratory Syncytial Virus in Children Younger than 5 Years in 2019: A Systematic Analysis. Lancet 2022, 399, 2047–2064. [Google Scholar] [CrossRef]
- O’Brien, K.L.; Baggett, H.C.; Brooks, W.A.; Feikin, D.R.; Hammitt, L.L.; Higdon, M.M.; Howie, S.R.; Knoll, M.D.; Kotloff, K.L.; Levine, O.S. Causes of Severe Pneumonia Requiring Hospital Admission in Children without HIV Infection from Africa and Asia: The PERCH Multi-Country Case-Control Study. Lancet 2019, 394, 757–779. [Google Scholar] [CrossRef]
- Kim, H.W.; CANCHOLA, J.G.; Brandt, C.D.; Pyles, G.; Chanock, R.M.; Jensen, K.; Parrott, R.H. Respiratory Syncytial Virus Disease in Infants Despite Prior Administration of Antigenic Inactivated Vaccine. Am. J. Epidemiol. 1969, 89, 422–434. [Google Scholar] [CrossRef]
- Openshaw, P.J.; Culley, F.J.; Olszewska, W. Immunopathogenesis of Vaccine-Enhanced RSV Disease. Vaccine 2001, 20, S27–S31. [Google Scholar] [CrossRef]
- Chang, L.A.; Phung, E.; Crank, M.C.; Morabito, K.M.; Villafana, T.; Dubovsky, F.; Falloon, J.; Esser, M.T.; Lin, B.C.; Chen, G.L. A Prefusion-Stabilized RSV F Subunit Vaccine Elicits B Cell Responses with Greater Breadth and Potency than a Postfusion F Vaccine. Sci. Transl. Med. 2022, 14, eade0424. [Google Scholar] [CrossRef]
- McLellan, J.S.; Chen, M.; Joyce, M.G.; Sastry, M.; Stewart-Jones, G.B.; Yang, Y.; Zhang, B.; Chen, L.; Srivatsan, S.; Zheng, A. Structure-Based Design of a Fusion Glycoprotein Vaccine for Respiratory Syncytial Virus. Science 2013, 342, 592–598. [Google Scholar] [CrossRef]
- Walsh, E.E. Humoral, Mucosal, and Cellular Immune Response to Topical Immunization with a Subunit Respiratory Syncytial Virus Vaccine. J. Infect. Dis. 1994, 170, 345–350. [Google Scholar] [CrossRef]
- Graham, B.S. Vaccine Development for Respiratory Syncytial Virus. Curr. Opin. Virol. 2017, 23, 107–112. [Google Scholar] [CrossRef]
- Barik, S. Respiratory Syncytial Virus Mechanisms to Interfere with Type 1 Interferons. Chall. Oppor. Respir. Syncytial Virus Vaccines 2013, 372, 173–191. [Google Scholar]
- Murawski, M.R.; McGinnes, L.W.; Finberg, R.W.; Kurt-Jones, E.A.; Massare, M.J.; Smith, G.; Heaton, P.M.; Fraire, A.E.; Morrison, T.G. Newcastle Disease Virus-Like Particles Containing Respiratory Syncytial Virus G Protein Induced Protection in BALB/C Mice, with no Evidence of Immunopathology. J. Virol. 2010, 84, 1110–1123. [Google Scholar] [CrossRef]
- Johnson, T.R.; McLellan, J.S.; Graham, B.S. Respiratory Syncytial Virus Glycoprotein G Interacts with DC-SIGN and L-SIGN to Activate ERK1 and ERK2. J. Virol. 2012, 86, 1339–1347. [Google Scholar] [CrossRef]
- Bakre, A.A.; Harcourt, J.L.; Haynes, L.M.; Anderson, L.J.; Tripp, R.A. The Central Conserved Region (CCR) of Respiratory Syncytial Virus (RSV) G Protein Modulates Host miRNA Expression and Alters the Cellular Response to Infection. Vaccines 2017, 5, 16. [Google Scholar] [CrossRef]
- Anderson, L.J.; Bingham, P.; Hierholzer, J. Neutralization of Respiratory Syncytial Virus by Individual and Mixtures of F and G Protein Monoclonal Antibodies. J. Virol. 1988, 62, 4232–4238. [Google Scholar] [CrossRef]
- Zhan, X.; Hurwitz, J.L.; Krishnamurthy, S.; Takimoto, T.; Boyd, K.; Scroggs, R.A.; Surman, S.; Portner, A.; Slobod, K.S. Respiratory Syncytial Virus (RSV) Fusion Protein Expressed by Recombinant Sendai Virus Elicits B-Cell and T-Cell Responses in Cotton Rats and Confers Protection Against RSV Subtypes A and B. Vaccine 2007, 25, 8782–8793. [Google Scholar] [CrossRef]
- Yu, J.; Kim, S.; Lee, J.; Chang, J. Single Intranasal Immunization with Recombinant Adenovirus-Based Vaccine Induces Protective Immunity Against Respiratory Syncytial Virus Infection. J. Virol. 2008, 82, 2350–2357. [Google Scholar] [CrossRef]
- Cullen, L.M.; Schmidt, M.R.; Morrison, T.G. The Importance of RSV F Protein Conformation in VLPs in Stimulation of Neutralizing Antibody Titers in Mice Previously Infected with RSV. Hum. Vaccines Immunother. 2017, 13, 2814–2823. [Google Scholar] [CrossRef]
- Kim, A.; Lee, D.; Lee, S.; Rubino, I.; Choi, H.; Quan, F. Protection Induced by Virus-Like Particle Vaccine Containing Tandem Repeat Gene of Respiratory Syncytial Virus G Protein. PLoS ONE 2018, 13, e0191277. [Google Scholar] [CrossRef]
- Chu, K.; Lee, S.; Kim, M.; Kim, A.; Moon, E.; Quan, F. Virus-Like Particles Coexpressing the PreF and Gt Antigens of Respiratory Syncytial Virus Confer Protection in Mice. Nanomedicine 2022, 17, 1159–1171. [Google Scholar] [CrossRef]
- Leyrat, C.; Paesen, G.C.; Charleston, J.; Renner, M.; Grimes, J.M. Structural Insights into the Human Metapneumovirus Glycoprotein Ectodomain. J. Virol. 2014, 88, 11611–11616. [Google Scholar] [CrossRef]
- McGinnes Cullen, L.; Schmidt, M.R.; Morrison, T.G. Effect of Previous Respiratory Syncytial Virus Infection on Murine Immune Responses to F and G Protein-Containing Virus-Like Particles. J. Virol. 2019, 93, e00087-19. [Google Scholar] [CrossRef]
- Ha, B.; Yang, J.E.; Chen, X.; Jadhao, S.J.; Wright, E.R.; Anderson, L.J. Two Rsv Platforms for G, F, Or G F Proteins Vlps. Viruses 2020, 12, 906. [Google Scholar] [CrossRef]
- Blanco, J.C.; Cullen, L.M.; Kamali, A.; Sylla, F.Y.; Chinmoun, Z.; Boukhvalova, M.S.; Morrison, T.G. Correlative Outcomes of Maternal Immunization Against RSV in Cotton Rats. Hum. Vaccines Immunother. 2022, 18, 2148499. [Google Scholar] [CrossRef]
- McGinnes Cullen, L.; Luo, B.; Wen, Z.; Zhang, L.; Durr, E.; Morrison, T.G. The Respiratory Syncytial Virus (RSV) G Protein Enhances the Immune Responses to the RSV F Protein in an Enveloped Virus-Like Particle Vaccine Candidate. J. Virol. 2023, 97, e01900-22. [Google Scholar] [CrossRef]
- Quan, F.; Kim, Y.; Lee, S.; Yi, H.; Kang, S.; Bozja, J.; Moore, M.L.; Compans, R.W. Viruslike Particle Vaccine Induces Protection Against Respiratory Syncytial Virus Infection in Mice. J. Infect. Dis. 2011, 204, 987–995. [Google Scholar] [CrossRef]
- Peebles Jr, R.S.; Sheller, J.R.; Collins, R.D.; Jarzecka, K.; Mitchell, D.B.; Graham, B.S. Respiratory Syncytial Virus (RSV)—Induced Airway Hyperresponsiveness in Allergically Sensitized Mice is Inhibited by Live RSV and Exacerbated by Formalin-Inactivated RSV. J. Infect. Dis. 2000, 182, 671–677. [Google Scholar] [CrossRef]
- Patel, N.; Tian, J.; Flores, R.; Jacobson, K.; Walker, M.; Portnoff, A.; Gueber-Xabier, M.; Massare, M.J.; Glenn, G.; Ellingsworth, L. Flexible RSV Prefusogenic Fusion Glycoprotein Exposes Multiple Neutralizing Epitopes that may Collectively Contribute to Protective Immunity. Vaccines 2020, 8, 607. [Google Scholar] [CrossRef]
- Lee, S.; Kim, S.; Lee, D.; Kim, A.; Quan, F. Evaluation of Protective Efficacy Induced by Virus-Like Particles Containing a Trichinella Spiralis Excretory-Secretory (ES) Protein in Mice. Parasites Vectors 2016, 9, 384. [Google Scholar] [CrossRef]
- Lee, S.; Kim, A.; Lee, D.; Rubino, I.; Choi, H.; Quan, F. Protection Induced by Virus-Like Particles Containing Toxoplasma Gondii Microneme Protein 8 Against Highly Virulent RH Strain of Toxoplasma Gondii Infection. PLoS ONE 2017, 12, e0175644. [Google Scholar] [CrossRef]
- Lee, D.H.; Lee, S.H.; Kim, A.R.; Quan, F.S. Virus-Like Nanoparticle Vaccine Confers Protection Against Toxoplasma Gondii. PLoS ONE 2016, 11, e0161231. [Google Scholar] [CrossRef]
- Lee, S.; Kang, H.; Lee, D.; Quan, F. Protective Immunity Induced by Incorporating Multiple Antigenic Proteins of Toxoplasma Gondii into Influenza Virus-Like Particles. Front. Immunol. 2018, 9, 3073. [Google Scholar] [CrossRef]
- Mazur, N.I.; Higgins, D.; Nunes, M.C.; Melero, J.A.; Langedijk, A.C.; Horsley, N.; Buchholz, U.J.; Openshaw, P.J.; McLellan, J.S.; Englund, J.A. The Respiratory Syncytial Virus Vaccine Landscape: Lessons from the Graveyard and Promising Candidates. Lancet Infect. Dis. 2018, 18, e295–e311. [Google Scholar] [CrossRef]
- Blanco, J.C.; Pletneva, L.M.; McGinnes-Cullen, L.; Otoa, R.O.; Patel, M.C.; Fernando, L.R.; Boukhvalova, M.S.; Morrison, T.G. Efficacy of a Respiratory Syncytial Virus Vaccine Candidate in a Maternal Immunization Model. Nat. Commun. 2018, 9, 1904. [Google Scholar] [CrossRef]
- Blanco, J.C.; Fernando, L.R.; Zhang, W.; Kamali, A.; Boukhvalova, M.S.; McGinnes-Cullen, L.; Morrison, T.G. Alternative Virus-Like Particle-Associated Prefusion F Proteins as Maternal Vaccines for Respiratory Syncytial Virus. J. Virol. 2019, 93, e00914-19. [Google Scholar] [CrossRef]
- Zanin, M.; Baviskar, P.; Webster, R.; Webby, R. The Interaction between Respiratory Pathogens and Mucus. Cell Host Microbe 2016, 19, 159–168. [Google Scholar] [CrossRef]
- Persson, B.D.; Jaffe, A.B.; Fearns, R.; Danahay, H. Respiratory Syncytial Virus can Infect Basal Cells and Alter Human Airway Epithelial Differentiation. PLoS ONE 2014, 9, e102368. [Google Scholar] [CrossRef]
- Cose, S.; Brammer, C.; Khanna, K.M.; Masopust, D.; Lefrançois, L. Evidence that a Significant Number of Naive T Cells Enter Non-lymphoid Organs as Part of a Normal Migratory Pathway. Eur. J. Immunol. 2006, 36, 1423–1433. [Google Scholar] [CrossRef]
- Caucheteux, S.M.; Torabi-Parizi, P.; Paul, W.E. Analysis of Naive Lung CD4 T Cells Provides Evidence of Functional Lung to Lymph Node Migration. Proc. Natl. Acad. Sci. USA 2013, 110, 1821–1826. [Google Scholar] [CrossRef]
- Murphy, B.; Prince, G.; Walsh, E.; Kim, H.; Parrott, R.; Hemming, V.; Rodriguez, W.; Chanock, R. Dissociation between Serum Neutralizing and Glycoprotein Antibody Responses of Infants and Children Who Received Inactivated Respiratory Syncytial Virus Vaccine. J. Clin. Microbiol. 1986, 24, 197–202. [Google Scholar] [CrossRef]
- Corry, D.B.; Folkesson, H.G.; Warnock, M.L.; Erle, D.J.; Matthay, M.A.; Wiener-Kronish, J.P.; Locksley, R.M. Interleukin 4, but Not Interleukin 5 Or Eosinophils, is Required in a Murine Model of Acute Airway Hyperreactivity. J. Exp. Med. 1996, 183, 109–117. [Google Scholar] [CrossRef]
- Johnson, T.R.; Graham, B.S. Secreted Respiratory Syncytial Virus G Glycoprotein Induces Interleukin-5 (IL-5), IL-13, and Eosinophilia by an IL-4-Independent Mechanism. J. Virol. 1999, 73, 8485–8495. [Google Scholar] [CrossRef]
- Acosta, P.L.; Caballero, M.T.; Polack, F.P. Brief History and Characterization of Enhanced Respiratory Syncytial Virus Disease. Clin. Vaccine Immunol. 2016, 23, 189–195. [Google Scholar] [CrossRef]
- Chirkova, T.; Ha, B.; Rimawi, B.H.; Oomens, A.G.; Hartert, T.V.; Anderson, L.J. In Vitro Model for the Assessment of Human Immune Responses to Subunit RSV Vaccines. PLoS ONE 2020, 15, e0229660. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, Y.; Ko, E.; Kim, K.; Hwang, H.S.; Park, S.; Kwon, Y.; Kang, S.M. Soluble F Proteins Exacerbate Pulmonary Histopathology After Vaccination upon Respiratory Syncytial Virus Challenge but Not when Presented on Virus-Like Particles. Hum. Vaccines Immunother. 2017, 13, 2594–2605. [Google Scholar] [CrossRef]
- Xu, Y.; Yuen, P.; Lam, J.K. Intranasal DNA Vaccine for Protection Against Respiratory Infectious Diseases: The Delivery Perspectives. Pharmaceutics 2014, 6, 378–415. [Google Scholar] [CrossRef]
- Romagnani, S. Th1/Th2 Cells. Inflamm. Bowel Dis. 1999, 5, 285–294. [Google Scholar] [CrossRef]
- Capella, C.; Chaiwatpongsakorn, S.; Gorrell, E.; Risch, Z.A.; Ye, F.; Mertz, S.E.; Johnson, S.M.; Moore-Clingenpeel, M.; Ramilo, O.; Mejias, A. Prefusion F, Postfusion F, G Antibodies, and Disease Severity in Infants and Young Children with Acute Respiratory Syncytial Virus Infection. J. Infect. Dis. 2017, 216, 1398–1406. [Google Scholar] [CrossRef]
- Brakel, K.A.; Binjawadagi, B.; French-Kim, K.; Watts, M.; Harder, O.; Ma, Y.; Li, J.; Niewiesk, S. Coexpression of Respiratory Syncytial Virus (RSV) Fusion (F) Protein and Attachment Glycoprotein (G) in a Vesicular Stomatitis Virus (VSV) Vector System Provides Synergistic Effects Against RSV Infection in a Cotton Rat Model. Vaccine 2021, 39, 6817–6828. [Google Scholar] [CrossRef]
- Kuo, P.; Lee, C.; Wang, J.; Hsieh, S.; Huang, S.; Lam, C. Inhalation of Volatile Anesthetics Via a Laryngeal Mask is Associated with Lower Incidence of Intraoperative Awareness in Non-Critically Ill Patients. PLoS ONE 2017, 12, e0186337. [Google Scholar] [CrossRef] [PubMed]
- Ko, E.; Kwon, Y.; Lee, J.S.; Hwang, H.S.; Yoo, S.; Lee, Y.; Lee, Y.; Kim, M.; Cho, M.K.; Lee, Y.R. Virus-Like Nanoparticle and DNA Vaccination Confers Protection Against Respiratory Syncytial Virus by Modulating Innate and Adaptive Immune Cells. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 99–108. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.-H.; Chu, K.-B.; Kim, M.-J.; Mao, J.; Eom, G.-D.; Yoon, K.-W.; Ahmed, M.A.; Quan, F.-S. Virus-like Particle Vaccine Expressing the Respiratory Syncytial Virus Pre-Fusion and G Proteins Confers Protection against RSV Challenge Infection. Pharmaceutics 2023, 15, 782. https://doi.org/10.3390/pharmaceutics15030782
Lee S-H, Chu K-B, Kim M-J, Mao J, Eom G-D, Yoon K-W, Ahmed MA, Quan F-S. Virus-like Particle Vaccine Expressing the Respiratory Syncytial Virus Pre-Fusion and G Proteins Confers Protection against RSV Challenge Infection. Pharmaceutics. 2023; 15(3):782. https://doi.org/10.3390/pharmaceutics15030782
Chicago/Turabian StyleLee, Su-Hwa, Ki-Back Chu, Min-Ju Kim, Jie Mao, Gi-Deok Eom, Keon-Woong Yoon, Md Atique Ahmed, and Fu-Shi Quan. 2023. "Virus-like Particle Vaccine Expressing the Respiratory Syncytial Virus Pre-Fusion and G Proteins Confers Protection against RSV Challenge Infection" Pharmaceutics 15, no. 3: 782. https://doi.org/10.3390/pharmaceutics15030782
APA StyleLee, S.-H., Chu, K.-B., Kim, M.-J., Mao, J., Eom, G.-D., Yoon, K.-W., Ahmed, M. A., & Quan, F.-S. (2023). Virus-like Particle Vaccine Expressing the Respiratory Syncytial Virus Pre-Fusion and G Proteins Confers Protection against RSV Challenge Infection. Pharmaceutics, 15(3), 782. https://doi.org/10.3390/pharmaceutics15030782





