Microneedles for Efficient and Precise Drug Delivery in Cancer Therapy
Abstract
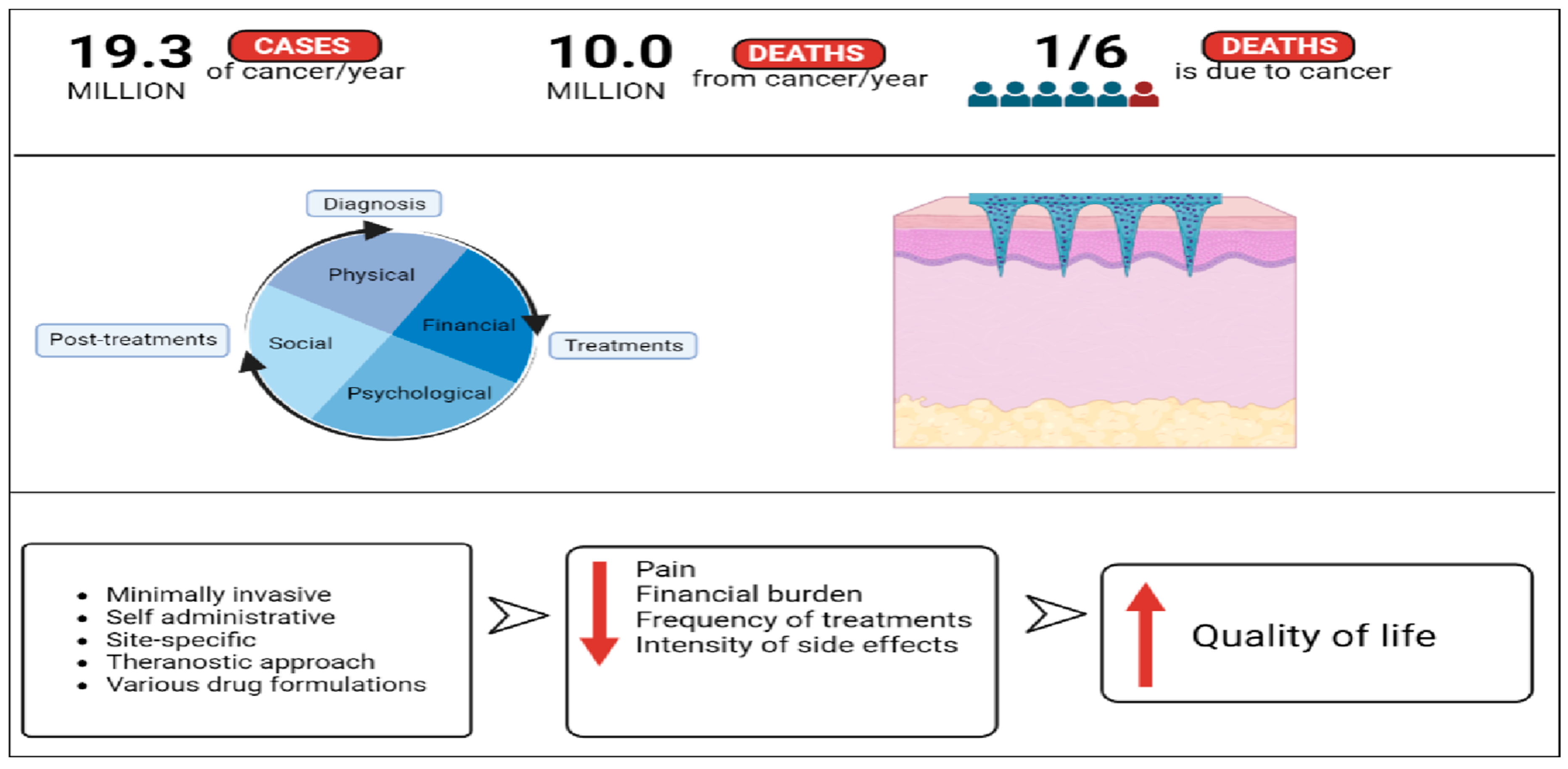
1. Introduction
2. General Properties and Classification of Microneedles
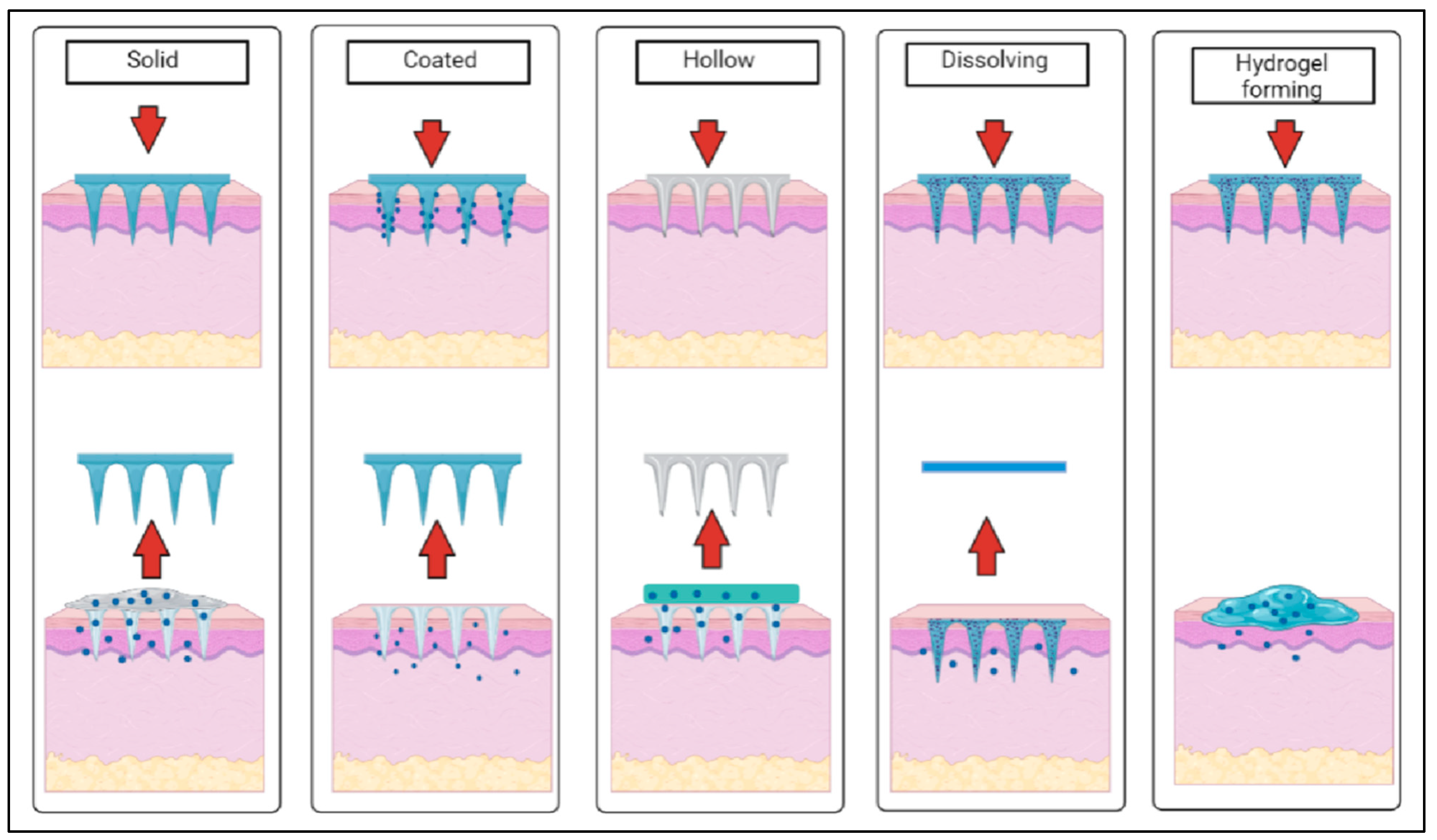
2.1. Types of Microneedles
2.2. Type of Materials and Fabrication Methods
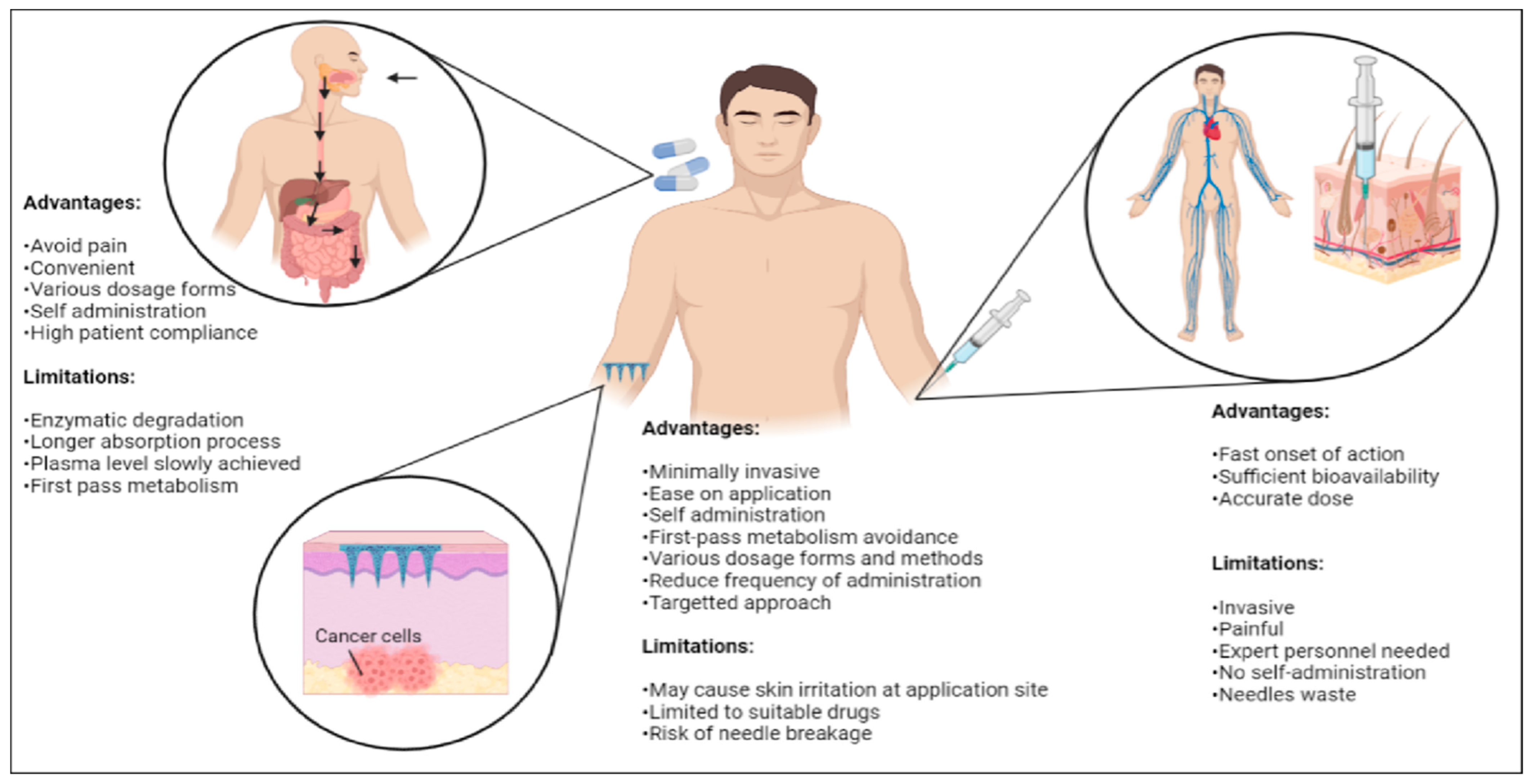
3. Advantages and Limitations of Microneedle
3.1. Transdermal Route of Administration
3.2. Self-Administration
3.3. Painless Treatment Approach
3.4. Economical
3.5. Environmental Sustainability
4. Breaking the Barriers: Microneedles in Cancer Diagnostic, Treatment and Theranostics
| Type of Treatments | Recent Studies |
|---|---|
| Combinatory treatments | A light-activatable rapidly separable microneedle patch made of polymer containing photosensitive nanomaterials (lanthanum hexaboride) that could quickly deliver doxorubicin (DOX) drug to the skin was used as photo-thermal transducers to repeatedly provide chemotherapy and photothermal therapy to superficial tumors [90]. |
| Bhatnagar and coworkers developed a polyvinylpyrrolidone/polyvinyl alcohol microneedle patch for the combinatorial delivery of doxorubicin and docetaxel to treat breast cancer tumors where the in vivo studies performed on 4T1 breast tumors bearing mice in this study proved to be more efficient than the single treatment approaches, leading to impaired tumor growth [93]. | |
| Self-assembled nano-dissolving microneedles drug delivery system was successfully constructed for chemo-photothermal combination therapy against melanoma [94]. | |
| Hao et al. [95] combined chemotherapy and photothermal therapy for the development of a PEGylated gold nanorod coated poly(l-lactide) microneedle system in order to enhance the antitumor efficiency of docetaxel-loaded MPEG-PDLLA micelles for the treatment of A431 tumors [95,96]. | |
| Immunotherapy | Encapsulation of DNA vaccines within microneedles as delivery vehicles protected the vaccines from the hostile environment in vivo, including omnipresent nucleases, increasing the half-life of vaccines while generating long-lasting immune stimulatory effects [84]. |
| Microneedle administration of antibodies showed that the concentration of antibodies was 2 times higher than that of the control, and the T cells were more responsive to HPV-16 oncogenic antigen expressing cells (TC-1) (IFN-γ levels in control ≈ 250 pg/mL and ≈530 pg/mL. This enhanced immune response prevented the establishment of cervical tumors in 4 of the 9 mice treated with microneedles [92]. | |
| Gene therapy | A polyvinylpyrrolidone microneedle patch loaded with E6/E7 pDNA RALA nanoparticles for the gene therapy of cervical cancer was developed by Ali et al. [97]. |
| A hyaluronic acid-based microneedle array for mediating the delivery of anti-PD1 antibody (aPD1), and -methyl-DL-tryptophan (1-MT) to B16F10 melanoma tumors was also produced, in which the aPD1 targets the PD-1 receptors expressed by T cells, and therefore avoids the cancer cells inhibitory signaling that prevent the T cells activation [98]. |
5. Future Direction of Microneedles in Cancer Care
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO. Breast Cancer. World Health Organization. 26 March 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/breast-cancer (accessed on 25 May 2022).
- WCRF. Breast Cancer Statistics: World Cancer Research Fund International. 14 April 2022. WCRF International. Available online: https://www.wcrf.org/cancer-trends/breast-cancer-statistics/ (accessed on 10 July 2022).
- WHO. Globocan 2020: New Global Cancer Data. UICC. 27 June 2022. Available online: https://www.uicc.org/news/globocan-2020-new-global-cancer-data (accessed on 4 January 2023).
- Elzohry, A.A.M.; Fikry, N.M.; Elhamed, M.F.A.; Mohammed, K.F. Updates in management of gastro-oesophageal cancer related pain. World News Nat. Sci. 2019, 23, 94–103. [Google Scholar]
- NCI. Cancer Statistics. National Cancer Institute. 25 September 2020. Available online: https://www.cancer.gov/about-cancer/understanding/statistics (accessed on 10 July 2022).
- Nersesyan, H.; Mucksavage, J.J.; Tesoro, E.; Slavin, K.V. Pain Management in Cancer Patients. In Cancer Management in Man: Chemotherapy, Biological Therapy, Hyperthermia and Supporting Measures; Springer: Dordrecht, The Netherlands, 2011; pp. 437–452. [Google Scholar]
- Alonazi, W.B.; Thomas, S.A. Quality of care and quality of life: Convergence or divergence? Health Serv. Insights 2014, 7, HSI-S13283. [Google Scholar] [CrossRef] [PubMed]
- Haleem, A.; Javaid, M.; Singh, R.P.; Suman, R. Medical 4.0 technologies for healthcare: Features, capabilities, and applications. Internet Things Cyber-Phys. Syst. 2022, 2, 12–30. [Google Scholar] [CrossRef]
- McGuirea, A.L.; Aulisiob, M.P.; Davisc, F.D.; Erwind, C.; Hartere, T.D. Ethical Challenges Arising in the COVID-19 Pandemic: An Overview from the Association of Bioethics Program Directors (ABPD) Task Force. Am. J. Bioeth. 2020, 20, 15–27. [Google Scholar] [CrossRef]
- Patt, D.; Gordan, L.; Diaz, M.; Okon, T.; Grady, L.; Harmison, M.; Markward, N.; Sullivan, M.; Peng, J.; Zhou, A. Impact of COVID-19 on cancer care: How the pandemic is delaying cancer diagnosis and treatment for American seniors. JCO Clin. Cancer Inform. 2020, 4, 1059–1071. [Google Scholar] [CrossRef]
- Riera, R.; Bagattini Â, M.; Pacheco, R.L.; Pachito, D.V.; Roitberg, F.; Ilbawi, A. Delays and disruptions in cancer health care due to COVID-19 pandemic: Systematic review. JCO Glob. Oncol. 2021, 7, 311–323. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- IARC. (n.d.). Cancer Tomorrow. Cancer Tomorrow. Available online: https://gco.iarc.fr/tomorrow/en (accessed on 10 July 2022).
- Rajput, A.; Kulkarni, M.; Deshmukh, P.; Pingale, P.; Garkal, A.; Gandhi, S.; Butani, S. A key role by polymers in microneedle technology: A new era. Drug Dev. Ind. Pharm. 2021, 47, 1713–1732. [Google Scholar] [CrossRef]
- Yang, J.; Liu, X.; Fu, Y.; Song, Y. Recent advances of microneedles for biomedical applications: Drug delivery and beyond. Acta Pharm. Sin. B 2019, 9, 469–483. [Google Scholar] [CrossRef]
- Singh, V.; Kesharwani, P. Recent advances in microneedles-based drug delivery device in the diagnosis and treatment of cancer. J. Control. Release 2021, 338, 394–409. [Google Scholar] [CrossRef]
- Gerstel, M.S.; Place, V.A. Drug Delivery Device. U.S. Patent No. 3964482, 22 June 1976. [Google Scholar]
- Permana, A.D.; Nainu, F.; Moffatt, K.; Larrañeta, E.; Donnelly, R.F. Recent advances in combination of microneedles and nanomedicines for lymphatic targeted drug delivery. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2021, 13, e1690. [Google Scholar] [CrossRef] [PubMed]
- Waghule, T.; Singhvi, G.; Dubey, S.K.; Pandey, M.M.; Gupta, G.; Singh, M.; Dua, K. Microneedles: A smart approach and increasing potential for transdermal drug delivery system. Biomed. Pharmacother. 2019, 109, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Halder, J.; Gupta, S.; Kumari, R.; Gupta, G.D.; Rai, V.K. Microneedle array: Applications, recent advances, and clinical pertinence in transdermal drug delivery. J. Pharm. Innov. 2020, 16, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Mehmood, S.; Raza, A.; Hayat, U.; Rasheed, T.; Iqbal, H.M. Microneedles in smart drug delivery. Adv. Wound Care 2021, 10, 204–219. [Google Scholar] [CrossRef] [PubMed]
- TheraJect n.d. Available online: https://www.theraject.com (accessed on 10 December 2022).
- Bariya, S.H.; Gohel, M.C.; Mehta, T.A.; Sharma, O.P. Microneedles: An emerging transdermal drug delivery system. J. Pharm. Pharmacol. 2012, 64, 11–29. [Google Scholar] [CrossRef]
- Fierce Biotech, n.d. Available online: https://www.diabetesincontrol.com/glucagon-patch-info/ (accessed on 10 December 2022).
- Nguyen, T.T.; Oh, Y.; Kim, Y.; Shin, Y.; Baek, S.K.; Park, J.H. Progress in microneedle array patch (MAP) for vaccine delivery. Hum. Vaccin. Immunother. 2021, 17, 316–327. [Google Scholar] [CrossRef]
- Our Proprietary Platforms, n.d. Available online: https://corium.com/propietary-platforms.html (accessed on 10 December 2022).
- Jung, J.H.; Jin, S.G. Microneedle for transdermal drug delivery: Current trends and fabrication. J. Pharm. Investig. 2021, 51, 503–517. [Google Scholar] [CrossRef]
- Aldawood, F.K.; Andar, A.; Desai, S. A Comprehensive Review of Microneedles: Types, Materials, Processes, Characterizations and Applications. Polymers 2021, 13, 2815. [Google Scholar] [CrossRef]
- Mdanda, S.; Ubanako, P.; Kondiah, P.P.D.; Kumar, P.; Choonara, Y.E. Recent advances in microneedle platforms for transdermal drug delivery technologies. Polymers 2021, 13, 2405. [Google Scholar] [CrossRef]
- Menon, I.; Bagwe, P.; Gomes, K.B.; Bajaj, L.; Gala, R.; Uddin, M.N.; D’Souza, M.J.; Zughaier, S.M. Microneedles: A new generation vaccine delivery system. Micromachines 2021, 12, 435. [Google Scholar] [CrossRef]
- Vinayakumar, K.B.; Kulkarni, P.G.; Nayak, M.M.; Dinesh, N.S.; Hegde, G.M.; Ramachandra, S.G.; Rajanna, K. A hollow stainless steel microneedle array to deliver insulin to a diabetic rat. J. Micromech. Microeng. 2016, 26, 065013. [Google Scholar] [CrossRef]
- Li, J.; Liu, B.; Zhou, Y.; Chen, Z.; Jiang, L.; Yuan, W.; Liang, L. Fabrication of a Ti porous microneedle array by metal injection molding for transdermal drug delivery. PLoS ONE 2017, 12, 0172043. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, H.; Lai, L.; Li, Y. Preparation of microneedle array mold based on MEMS lithography technology. Micromachines 2020, 12, 23. [Google Scholar] [CrossRef]
- Yu, W.; Jiang, G.; Liu, D.; Li, L.; Tong, Z.; Yao, J.; Kong, X. Transdermal delivery of insulin with bioceramic composite microneedles fabricated by gelatin and hydroxyapatite. Mater. Sci. Eng. C 2017, 73, 425–428. [Google Scholar] [CrossRef]
- Ita, K. Ceramic microneedles and hollow microneedles for transdermal drug delivery: Two decades of research. J. Drug Deliv. Sci. Technol. 2018, 44, 314–322. [Google Scholar] [CrossRef]
- Vallhov, H.; Xia, W.; Engqvist, H.; Scheynius, A. Bioceramic microneedle arrays are able to deliver OVA to dendritic cells in human skin. J. Mater. Chem. B 2018, 6, 6808–6816. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, H.; Yang, R.; Laffitte, Y.; Schmill, U.; Hu, W.; Kaddoura, M.; Blondeel, E.J.M.; Cui, B. Fabrication of sharp silicon hollow microneedles by deep-reactive ion etching towards minimally invasive diagnostics. Microsyst. Nanoeng. 2019, 5, 41. [Google Scholar] [CrossRef]
- Chu, L.Y.; Ye, L.; Dong, K.; Compans, R.W.; Yang, C.; Prausnitz, M.R. Enhanced stability of inactivated influenza vaccine encapsulated in dissolving microneedle patches. Pharm. Res. 2016, 33, 868–878. [Google Scholar] [CrossRef]
- Hong, J.Y.; Ko, E.J.; Choi, S.Y.; Li, K.; Kim, A.R.; Park, J.O.; Kim, B.J. Efficacy and safety of a novel, soluble microneedle patch for the improvement of facial wrinkle. J. Cosmet. Dermatol. 2018, 17, 235–241. [Google Scholar] [CrossRef]
- Yalcintas, E.P.; Ackerman, D.S.; Korkmaz, E.; Telmer, C.A.; Jarvik, J.W.; Campbell, P.G.; Bruchez, M.P.; Ozdoganlar, O.B. Analysis of in vitro cytotoxicity of carbohydrate-based materials used for dissolvable microneedle arrays. Pharm. Res. 2020, 37, 33. [Google Scholar] [CrossRef]
- Bhadale, R.S.; Londhe, V.Y. A systematic review of carbohydrate-based microneedles: Current status and future prospects. J. Mater. Sci. Mater. Med. 2021, 32, 89. [Google Scholar] [CrossRef] [PubMed]
- Dabholkar, N.; Gorantla, S.; Waghule, T.; Rapalli, V.K.; Kothuru, A.; Goel, S.; Singhvi, G. Biodegradable microneedles fabricated with carbohydrates and proteins: Revolutionary approach for transdermal drug delivery. Int. J. Biol. Macromol. 2021, 170, 602–621. [Google Scholar] [CrossRef]
- Arya, J.; Henry, S.; Kalluri, H.; McAllister, D.V.; Pewin, W.P.; Prausnitz, M.R. Tolerability, usability and acceptability of dissolving microneedle patch administration in human subjects. Biomaterials 2017, 128, 1–7. [Google Scholar] [CrossRef]
- Baek, S.-H.; Shin, J.-H.; Kim, Y.-C. Drug-coated microneedles for rapid and painless local anesthesia. Biomed. Microdevices 2017, 19, 2. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.M.; Lee, C.; Lahiji, S.F.; Jung, U.W.; Chung, G.; Jung, H. Dissolving microneedles for rapid and painless local anesthesia. Pharmaceutics 2020, 12, 366. [Google Scholar] [CrossRef]
- Eum, J.; Kim, Y.; Um, D.J.; Shin, J.; Yang, H.; Jung, H. Solvent-free polycaprolactone dissolving microneedles generated via the thermal melting method for the sustained release of capsaicin. Micromachines 2021, 12, 167. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Rivera, O.A.; Shin, M.D.; Chen, A.; Beiss, V.; Moreno-Gonzalez, M.A.; Lopez-Ramirez, M.A.; Reynoso, M.; Wang, H.; Hurst, B.L.; Wang, J.; et al. Trivalent subunit vaccine candidates for COVID-19 and their delivery devices. J. Am. Chem. Soc. 2021, 143, 14748–14765. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Kim, S.; Kang, G.; Lahiji, S.F.; Jang, M.; Kim, Y.M.; Kim, J.M.; Cho, S.N.; Jung, H. Centrifugal lithography: Self-shaping of polymer microstructures encapsulating biopharmaceutics by centrifuging polymer drops. Adv. Healthc. Mater. 2017, 6, 1700326. [Google Scholar] [CrossRef]
- Khan, S.; Hasan, A.; Attar, F.; Babadaei, M.M.N.; Zeinabad, H.A.; Salehi, M.; Alizadeh, M.; Hassan, M.; Derakhshankhah, H.; Hamblin, M.R.; et al. Diagnostic and drug release systems based on microneedle arrays in breast cancer therapy. J. Control. Release 2021, 338, 341–357. [Google Scholar] [CrossRef]
- Faezah, A.N.; Rahayu, A.; Vigneswari SMajid, M.I.A.; Amirul, A.A. Regulating the molar fraction of 4-hydroxybutyrate in poly(3-hydroxybutyrate-co-4-hydroxybutyrate) by biological fermentation and enzymatic degradation. World J. Microbiol. Biotechnol. 2011, 27, 2455–2459. [Google Scholar] [CrossRef]
- Ramadon, D.; McCrudden, M.T.; Courtenay, A.J.; Donnelly, R.F. Enhancement strategies for transdermal drug delivery systems: Current trends and applications. Drug Deliv. Transl. Res. 2022, 12, 758–791. [Google Scholar] [CrossRef]
- Laracuente, M.L.; Marina, H.Y.; McHugh, K.J. Zero-order drug delivery: State of the art and future prospects. J. Control. Release 2020, 327, 834–856. [Google Scholar] [CrossRef] [PubMed]
- Bellefroid, C.; Lechanteur, A.; Evrard, B.; Mottet, D.; Debacq-Chainiaux, F.; Piel, G. In vitro skin penetration enhancement techniques: A combined approach of ethosomes and microneedles. Int. J. Pharm. 2019, 572, 118793. [Google Scholar] [CrossRef] [PubMed]
- Seetharam, A.A.; Choudhry, H.; Bakhrebah, M.A.; Abdulaal, W.H.; Gupta, M.S.; Rizvi, S.M.D.; Alam, Q.; Gowda, D.V.; Moin, A. Microneedles drug delivery systems for treatment of cancer: A recent update. Pharmaceutics 2020, 12, 1101. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.C.; Ling, M.H.; Kusuma, S.J. Poly-γ-glutamic acid microneedles with a supporting structure design as a potential tool for transdermal delivery of insulin. Acta Biomater. 2015, 24, 106–116. [Google Scholar] [CrossRef]
- Min, H.S.; Kim, Y.; Kang, G.; Jang, M.; Yang, H.; Jung, H. Shape of dissolving microneedles determines skin penetration ability and efficacy of drug delivery. Biomater. Adv. 2022, 145, 213248. [Google Scholar] [CrossRef]
- García-Alfonso, P.; Muñoz Martín, A.J.; Ortega Morán, L.; Soto Alsar, J.; Torres Perez-Solero, G.; Blanco Codesido, M.; Calvo Ferrandiz, P.A.; Grasso Cicala, S. Oral drugs in the treatment of metastatic colorectal cancer. Ther. Adv. Med. Oncol. 2021, 13, 17588359211009001. [Google Scholar] [CrossRef]
- Kim, J.; Jesus, O.D. Medication Routes of Administration—Statpearls—NCBI Bookshelf. Medication Routes of Administration. Updated 17 February 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK568677/ (accessed on 14 July 2022).
- Ripolin, A.; Quinn, J.; Larrañeta, E.; Vicente-Perez, E.M.; Barry, J.; Donnelly, R.F. Successful application of large microneedle patches by human volunteers. Int. J. Pharm. 2017, 521, 92–101. [Google Scholar] [CrossRef]
- Rodriguez, C.; Ji, M.; Wang, H.L.; Padhya, T.; McMillan, S.C. Cancer pain and quality of life. J. Hosp. Palliat. Nurs. 2019, 21, 116–123. [Google Scholar] [CrossRef]
- Scarborough, B.M.; Smith, C.B. Optimal pain management for patients with cancer in the modern era. CA A Cancer J. Clin. 2018, 68, 182–196. [Google Scholar] [CrossRef]
- Dawson, S.; Koneti, K.K. The Role of Pain Management in Cancer Prehabilitation. In Prehabilitation for Cancer Surgery; Springer: Singapore, 2022; pp. 217–247. [Google Scholar]
- Zielińska, A.; Włodarczyk, M.; Makaro, A.; Sałaga, M.; Fichna, J. Management of pain in colorectal cancer patients. Crit. Rev. Oncol. /Hematol. 2021, 157, 103122. [Google Scholar] [CrossRef] [PubMed]
- Eaton, L.H.; Hulett, J.M. Mind-body interventions in the management of chronic cancer pain. In Seminars in Oncology Nursing; WB Saunders: Philadelphia, PA, USA, 2019; Volume 35, pp. 241–252. [Google Scholar]
- Dhadda, S.; Raigar, A.K.; Saini, K.; Guleria, A. Benzothiazoles: From recent advances in green synthesis to anti-cancer potential. Sustain. Chem. Pharm. 2021, 24, 100521. [Google Scholar] [CrossRef]
- Mao, J.J.; Pillai, G.G.; Andrade, C.J.; Ligibel, J.A.; Basu, P.; Cohen, L.; Khan, I.A.; Mustian, K.M.; Puthiyedath, R.; Dhiman, K.S.; et al. Integrative oncology: Addressing the global challenges of cancer prevention and treatment. CA A Cancer J. Clin. 2022, 72, 144–164. [Google Scholar] [CrossRef] [PubMed]
- UNEP. Why Optimized Cold-Chains Could Save a Billion COVID Vaccines; UNEP: Nairobi, Kenya, 2022. [Google Scholar]
- WHO. Health-Care Waste. World Health Organization. 8 February 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/health-care-waste (accessed on 31 October 2022).
- Greene, J.; Skolnik, C.L.; Merritt, M.W. How medicine becomes trash: Disposability in health care. Lancet 2022, 400, 1298–1299. [Google Scholar] [CrossRef]
- Luhar, I.; Luhar, S.; Abdullah, M.M.A.B. Challenges and Impacts of COVID-19 Pandemic on Global Waste Management Systems: A Review. J. Compos. Sci. 2022, 6, 271. [Google Scholar] [CrossRef]
- Valdivia, S.; Backes, J.G.; Traverso, M.; Sonnemann, G.; Cucurachi, S.; Guinée, J.B.; Schaubroeck, T.; Finkbeiner, M.; Leroy-Parmentier, N.; Ugaya, C.; et al. Principles for the application of life cycle sustainability assessment. Int. J. Life Cycle Assess. 2021, 26, 1900–1905. [Google Scholar] [CrossRef]
- Joseph, B.; James, J.; Kalarikkal, N.; Thomas, S. Recycling of medical plastics. Adv. Ind. Eng. Polym. Res. 2021, 4, 199–208. [Google Scholar] [CrossRef]
- Yahya, E.B.; Alqadhi, A.M. Recent trends in cancer therapy: A review on the current state of gene delivery. Life Sci. 2021, 269, 119087. [Google Scholar] [CrossRef]
- Kumari, S.; Choudhary, P.K.; Shukla, R.; Sahebkar, A.; Kesharwani, P. Recent advances in nanotechnology-based combination drug therapy for skin cancer. J. Biomater. Sci. Polym. Ed. 2022, 31, 1435–1468. [Google Scholar] [CrossRef]
- Madden, J.; O’Mahony, C.; Thompson, M.; O’Riordan, A.; Galvin, P. Biosensing in dermal interstitial fluid using microneedle based electrochemical devices. Sens. Bio-Sens. Res. 2020, 29, 100348. [Google Scholar] [CrossRef]
- Twycross, A.; Parker, R.; Williams, A.; Gibson, F. Cancer-related pain and pain management: Sources, prevalence, and the experiences of children and parents. J. Pediatr. Oncol. Nurs. 2015, 32, 369–384. [Google Scholar] [CrossRef] [PubMed]
- Gentry, S. Breast Cancer Treatments—Conquer: The Patient Voice. CONQUER. 8 October 2020. Available online: https://conquer-magazine.com/issues/special-issues/october-2020-breast-cancer/1364-breast-cancer-treatments (accessed on 31 October 2022).
- Allahverdiyev, A.M.; Parlar, E.; Dinparvar, S.; Bagirova, M.; Abamor, E.Ş. Current aspects in treatment of breast cancer based of nanodrug delivery systems and future prospects. Artif. Cells Nanomed. Biotechnol. 2018, 46 (Suppl. S3), S755–S762. [Google Scholar] [CrossRef] [PubMed]
- Kalaydina, R.V.; Bajwa, K.; Qorri, B.; Decarlo, A.; Szewczuk, M.R. Recent advances in “smart” delivery systems for extended drug release in cancer therapy. Int. J. Nanomed. 2018, 13, 4727. [Google Scholar] [CrossRef]
- Lin, G.; Zhang, H.; Huang, L. Smart polymeric nanoparticles for cancer gene delivery. Mol. Pharm. 2015, 12, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Liyanage, P.Y.; Hettiarachchi, S.D.; Zhou, Y.; Ouhtit, A.; Seven, E.S.; Oztan, C.Y.; Celik, E.; Leblanc, R.M. Nanoparticle-mediated targeted drug delivery for breast cancer treatment. Biochim. Biophys. Acta (BBA)-Rev. Cancer 2019, 1871, 419–433. [Google Scholar] [CrossRef]
- Wang, K.; Yee, C.; Tam, S.; Drost, L.; Chan, S.; Zaki, P.; Rico, V.; Ariello, K.; Dasios, M.; Lam, H.; et al. Prevalence of pain in patients with breast cancer post-treatment: A systematic review. Breast 2018, 42, 113–127. [Google Scholar] [CrossRef]
- Chen, M.C.; Lin, Z.W.; Ling, M.H. Near-infrared light-activatable microneedle system for treating superficial tumors by combination of chemotherapy and photothermal therapy. ACS Nano 2016, 10, 93–101. [Google Scholar] [CrossRef]
- Duong, H.T.T.; Thambi, T.; Yin, Y.; Kim, S.H.; Nguyen, T.L.; Phan, V.G.; Kim, J.; Jeong, J.H.; Lee, D.S. Degradation-regulated architecture of injectable smart hydrogels enhances humoral immune response and potentiates antitumor activity in human lung carcinoma. Biomaterials 2020, 230, 119599. [Google Scholar] [CrossRef]
- Cai, L.; Xu, J.; Yang, Z.; Tong, R.; Dong, Z.; Wang, C.; Leong, K.W. Engineered biomaterials for cancer immunotherapy. MedComm 2020, 1, 35–46. [Google Scholar] [CrossRef]
- Hao, Y.; Chung, C.K.; Yu, Z.; Huis in ‘t Veld, R.V.; Ossendorp, F.A.; Ten Dijke, P.; Cruz, L.J. Combinatorial therapeutic approaches with nanomaterial-based photodynamic cancer therapy. Pharmaceutics 2022, 14, 120. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, C.; Xiao, J.; You, J.; Zhang, W.; Liu, Y.; Xu, L.; Liu, A.; Xin, H.; Wang, X. Local extraction and detection of early stage breast cancers through a microneedle and nano-Ag/MBL film based painless and blood-free strategy. Mater. Sci. Eng. C 2020, 109, 110402. [Google Scholar] [CrossRef]
- Ahles, T.A.; Root, J.C. Cognitive effects of cancer and cancer treatments. Annu. Rev. Clin. Psychol. 2018, 14, 425. [Google Scholar] [CrossRef]
- Waks, A.G.; Winer, E.P. Breast cancer treatment: A review. JAMA 2019, 321, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Jiang, X.; Zeng, Y.; Terry, R.N.; Li, W. Rapidly separable microneedle patches for controlled release of therapeutics for long-acting therapies. Med. Drug Discov. 2021, 13, 100118. [Google Scholar] [CrossRef]
- Pires, L.R.; Vinayakumar, K.B.; Turos, M.; Miguel, V.; Gaspar, J. A perspective on microneedle-based drug delivery and diagnostics in paediatrics. J. Pers. Med. 2019, 9, 49. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.F.; Rodrigues, C.F.; Jacinto, T.A.; Miguel, S.P.; Costa, E.C.; Correia, I.J. Microneedle-based delivery devices for cancer therapy: A review. Pharmacol. Res. 2019, 148, 104438. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, S.; Bankar, N.G.; Kulkarni, M.V.; Venuganti, V.V.K. Dissolvable microneedle patch containing doxorubicin and docetaxel is effective in 4T1 xenografted breast cancer mouse model. Int. J. Pharm. 2019, 556, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Chen, M.; Yang, D.; Qin, W.; Quan, G.; Wu, C.; Pan, X. Self-assembly nanomicelle-microneedle patches with enhanced tumor penetration for superior chemo-photothermal therapy. Nano Res. 2022, 15, 2335–2346. [Google Scholar] [CrossRef]
- Hao, Y.; Dong, M.; Zhang, T.; Peng, J.; Jia, Y.; Cao, Y.; Qian, Z. Novel approach of using near-Infrared Responsive PEGylated Gold Nanorod Coated Poly (L-lactide) microneedles to enhance the antitumor efficiency of Docetaxel-Loaded MPEG-PDLLA Micelles for Treating an A431 tumor. ACS Appl. Mater. Interfaces 2017, 9, 15317–15327. [Google Scholar] [CrossRef]
- Uddin, M.J.; Scoutaris, N.; Economidou, S.N.; Giraud, C.; Chowdhry, B.Z.; Donnelly, R.F.; Douroumis, D. 3D printed microneedles for anticancer therapy of skin tumours. Mater. Sci. Eng. C 2020, 107, 110248. [Google Scholar] [CrossRef]
- Ali, A.A.; McCrudden, C.M.; McCaffrey, J.; McBride, J.W.; Cole, G.; Dunne, N.J.; Robson, T.; Kissenpfennig, A.; Donnelly, R.F.; McCarthy, H.O. DNA vaccination for cervical cancer; a novel technology platform of RALA mediated gene delivery via polymeric microneedles. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 921–932. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Wang, J.; Hu, Q.; Hochu, G.M.; Xin, H.; Wang, C.; Gu, Z. Synergistic transcutaneous immunotherapy enhances antitumor immune responses through delivery of checkpoint inhibitors. ACS Nano 2016, 10, 8956–8963. [Google Scholar] [CrossRef] [PubMed]
- Quinn, H.L.; Larrañeta, E.; Donnelly, R.F. Dissolving microneedles: Safety considerations and future perspectives. Ther. Deliv. 2016, 7, 283–285. [Google Scholar] [CrossRef]
- Ngwa, W.; Boateng, F.; Kumar, R.; Irvine, D.J.; Formenti, S.; Ngoma, T.; Herskind, C.; Veldwijk, M.R.; Hildenbrand, G.L.; Hausmann, M.; et al. Smart radiation therapy biomaterials. Int. J. Radiat. Oncol.*Biol.*Phys. 2017, 97, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Hosu, O.; Mirel, S.; Săndulescu, R.; Cristea, C. Minireview: Smart tattoo, microneedle, point-of-care, and phone-based biosensors for medical screening, diagnosis, and monitoring. Anal. Lett. 2019, 52, 78–92. [Google Scholar] [CrossRef]
- Zhang, C.; Kim, J.P.; Creer, M.; Yang, J.; Liu, Z. A smartphone-based chloridometer for point-of-care diagnostics of cystic fibrosis. Biosens. Bioelectron. 2017, 97, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Khan, I.; Zhang, Q.; Liu, X.; Dostalek, J.; Liedberg, B.; Wang, Y. Lipopolysaccharides detection on a grating-coupled surface plasmon resonance smartphone biosensor. Biosens. Bioelectron. 2018, 99, 312–317. [Google Scholar] [CrossRef]

| Brand Name and Manufacturer | Types of Microneedles | Applications | References |
|---|---|---|---|
| DrugMAT and VatMAT; TheraJect Inc., Fremont, CA, USA | Dissolving microneedles |
| [22,23] |
| Nanoject®; Debiotech, Lausanne, Switzerland | Dissolvable peptide microneedle patch |
| [20] |
| Macroflux; Zosano Pharma Inc., Fremont, CA, USA | Metal microneedle patch |
| [24] |
| Onvax; Becton Dickinson, Franklin Lakes, NJ, USA | Microneedle array patch |
| [25] |
| MicroCor® PTH(1–34) Corium International Inc., Boston, MA, USA | Dissolving microneedles |
| [23,26] |
| Type of Microneedle | Advantages | Limitations |
|---|---|---|
| Solid |
|
|
| Coated |
|
|
| Dissolving |
|
|
| Hollow |
|
|
| Hydrogel forming |
|
|
| Materials | Fabrication Method | Types of Microneedles | Advantage | Disadvantage | References |
|---|---|---|---|---|---|
| Metal | Wet etching, lithography, metal injection molding, laser machining | Solid, Hollow | High mechanical strength, biocompatible | May induce allergic reactions, produce sharp waste | [28,31,32,33] |
| Bio-ceramic | Micro-molding | Solid | Resist towards chemical | Low tensile strength, may cause irritation if breaks inside the skin | [34,35,36] |
| Silicon | Deep reactive ion etching | Hollow | Microneedle of various shapes and sizes can be produced as it is flexible | Brittle, fabrication process is time-consuming, high cost | [19,37] |
| Sucrose, Hyaluronic acid | Micro-molding, droplet-born air-blowing | Dissolving | Biodegradable, low toxicity, able to stabilize protein molecules, good mechanical strength | Low mechanical strength | [38,39,40,41,42] |
| Polylactic acid (PLA), Poly-L-lacticacid (PLLA), Polyvinyl alcohol (PVA), Polyvinylpyrrolidone (PVP), Polycaprolactone (PCL) | Thermal micro-molding, micro-molding, centrifugal lithography | Solid, Coated, Dissolving | Biodegradable, inexpensive | Low mechanical strength | [32,35,38,43,44,45,46,47,48] |
| Stages in Cancer Therapy | Description | Barriers | References |
|---|---|---|---|
| Diagnosis | Medical history, local examination of tumor area and repeated analysis of bio-fluids (e.g., blood, urine and saliva) through lumbar punctures, bone marrow aspirations, biopsy and venepunctures |
| [74,75,76] |
| Surgery | Removal of tumor, usually performed as the only treatment prior to or after chemotherapy. |
| [77] |
| Chemotherapy |
| [76,78,79,80,81] | |
| Radiation | High-powered energy beams, such as protons, electrical energy or X-rays, target and destroy cancer cells |
| [73,75,82] |
| Photothermal | Employing plasmonic nanoparticles localized in tumors as exogenous energy absorbers that convert laser energy into heat, causing irreversible cellular damage and subsequent tumor destruction |
| [83] |
| Immunotherapy | First-line treatment that strengthens patient’s own immune system to naturally fight, defend and kill cancer cells Immune checkpoint blockade and cancer vaccines |
| [84,85] |
| Targeted |
| [77,86] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ganeson, K.; Alias, A.H.; Murugaiyah, V.; Amirul, A.-A.A.; Ramakrishna, S.; Vigneswari, S. Microneedles for Efficient and Precise Drug Delivery in Cancer Therapy. Pharmaceutics 2023, 15, 744. https://doi.org/10.3390/pharmaceutics15030744
Ganeson K, Alias AH, Murugaiyah V, Amirul A-AA, Ramakrishna S, Vigneswari S. Microneedles for Efficient and Precise Drug Delivery in Cancer Therapy. Pharmaceutics. 2023; 15(3):744. https://doi.org/10.3390/pharmaceutics15030744
Chicago/Turabian StyleGaneson, Keisheni, Ain Hafizah Alias, Vikneswaran Murugaiyah, Al-Ashraf Abdullah Amirul, Seeram Ramakrishna, and Sevakumaran Vigneswari. 2023. "Microneedles for Efficient and Precise Drug Delivery in Cancer Therapy" Pharmaceutics 15, no. 3: 744. https://doi.org/10.3390/pharmaceutics15030744
APA StyleGaneson, K., Alias, A. H., Murugaiyah, V., Amirul, A.-A. A., Ramakrishna, S., & Vigneswari, S. (2023). Microneedles for Efficient and Precise Drug Delivery in Cancer Therapy. Pharmaceutics, 15(3), 744. https://doi.org/10.3390/pharmaceutics15030744







