Nanoparticles for Lymph Node-Directed Delivery
Abstract
1. Introduction
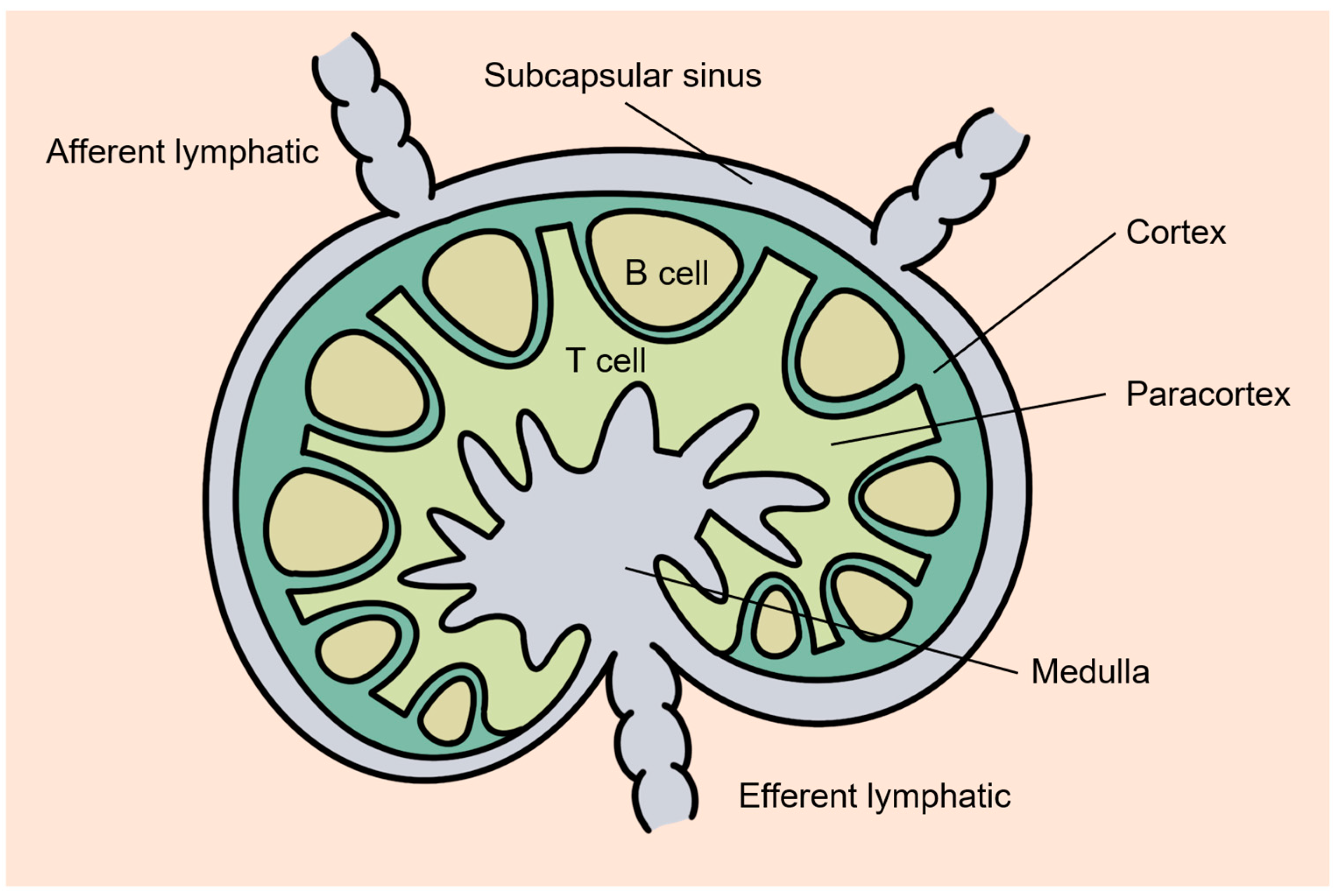
2. Principles of Lymphatic Delivery
3. Nanovehicles for Lymphatic Delivery
3.1. Environment-Responsive Materials
3.2. Tailored Nanoparticles
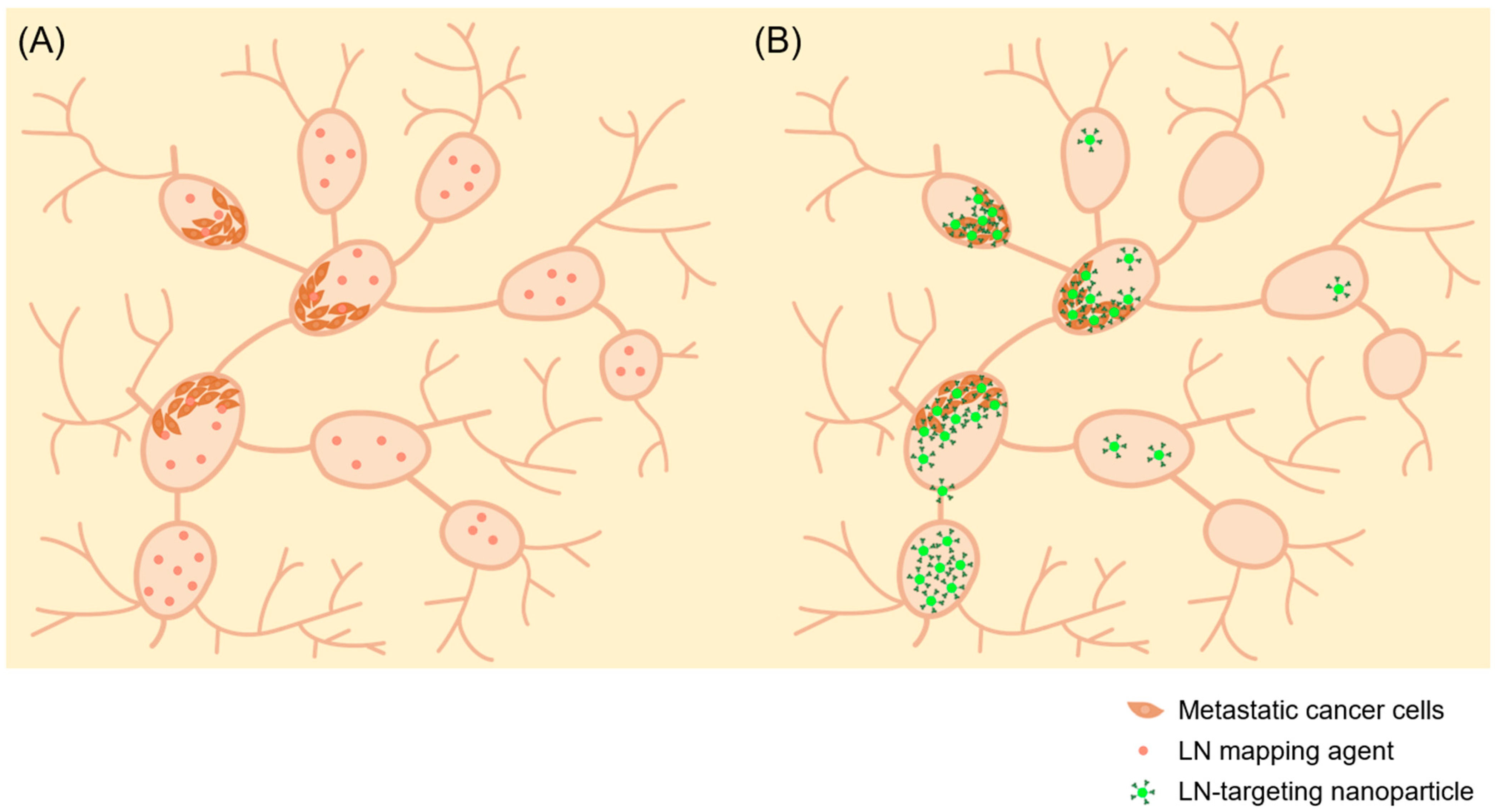
4. Anti-Metastatic Efficacy of Lymph Node Directed Delivery of Nanoparticles
4.1. In Vivo Antimetastatic Efficacy in Experimental Animal Models
4.2. Clinical Study
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Schudel, A.; Francis, D.M.; Thomas, S.N. Material design for lymph node drug delivery. Nat. Rev. Mater. 2019, 4, 415–428. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.E.; Turley, S.J. Stromal infrastructure of the lymph node and coordination of immunity. Trends Immunol. 2015, 36, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Achen, M.G.; Stacker, S.A. Molecular control of lymphatic metastasis. Ann. N. Y. Acad. Sci. 2008, 1131, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Mariani, G.; Moresco, L.; Viale, G.; Villa, G.; Bagnasco, M.; Canavese, G.; Buscombe, J.; Strauss, H.W.; Paganelli, G. Radioguided sentinel lymph node biopsy in breast cancer surgery. J. Nucl. Med. 2001, 42, 1198–1215. [Google Scholar]
- Li, P.; Sun, D. Advanced diagnostic imaging of sentinel lymph node in early stage breast cancer. J. Clin. Ultrasound 2022, 50, 415–421. [Google Scholar] [CrossRef]
- Polom, K.; Murawa, D.; Rho, Y.S.; Nowaczyk, P.; Hünerbein, M.; Murawa, P. Current trends and emerging future of indocyanine green usage in surgery and oncology: A literature review. Cancer 2011, 117, 4812–4822. [Google Scholar] [CrossRef]
- Miao, Y.; Gu, C.; Zhu, Y.; Yu, B.; Shen, Y.; Cong, H. Recent Progress in Fluorescence Imaging of the Near-Infrared II Window. ChemBioChem 2018, 19, 2522–2541. [Google Scholar] [CrossRef]
- Pieper, C.C.; Hur, S.; Sommer, C.-M.; Nadolski, G.; Maleux, G.; Kim, J.; Itkin, M. Back to the Future: Lipiodol in Lymphography—From Diagnostics to Theranostics. Investig. Radiol. 2019, 54, 600–615. [Google Scholar] [CrossRef]
- van Leeuwen, F.W.B.; Winter, A.; van Der Poel, H.G.; Eiber, M.; Suardi, N.; Graefen, M.; Wawroschek, F.; Maurer, T. Technologies for image-guided surgery for managing lymphatic metastases in prostate cancer. Nat. Rev. Urol. 2019, 16, 159–171. [Google Scholar] [CrossRef]
- Sakorafas, G.H.; Peros, G.; Cataliotti, L.; Vlastos, G. Lymphedema following axillary lymph node dissection for breast cancer. Surg. Oncol. 2006, 15, 153–165. [Google Scholar] [CrossRef]
- Porter, C.J.H.; Trevaskis, N.L. Targeting immune cells within lymph nodes. Nat. Nanotechnol. 2020, 15, 423–425. [Google Scholar] [CrossRef]
- Jeong, S.H.; Jang, J.H.; Cho, H.Y.; Lee, Y.B. Soft- and hard-lipid nanoparticles: A novel approach to lymphatic drug delivery. Arch. Pharm. Res. 2018, 41, 797–814. [Google Scholar] [CrossRef]
- Wang, Z.; Xia, H.; Chen, B.; Wang, Y.; Yin, Q.; Yan, Y.; Yang, Y.; Tang, M.; Liu, J.; Zhao, R.; et al. pH-Amplified CRET Nanoparticles for In Vivo Imaging of Tumor Metastatic Lymph Nodes. Angew. Chem. Int. Ed. 2021, 60, 14512–14520. [Google Scholar] [CrossRef]
- Bennett, Z.T.; Feng, Q.; Bishop, J.A.; Huang, G.; Sumer, B.D.; Gao, J. Detection of Lymph Node Metastases by Ultra-pH-Sensitive Polymeric Nanoparticles. Theranostics 2020, 10, 3340–3350. [Google Scholar] [CrossRef]
- McCright, J.; Skeen, C.; Yarmovsky, J.; Maisel, K. Nanoparticles with dense poly(ethylene glycol) coatings with near neutral charge are maximally transported across lymphatics and to the lymph nodes. Acta Biomater. 2022, 145, 146–158. [Google Scholar] [CrossRef]
- Schudel, A.; Chapman, A.P.; Yau, M.-K.; Higginson, C.J.; Francis, D.M.; Manspeaker, M.P.; Avecilla, A.R.C.; Rohner, N.A.; Finn, M.G.; Thomas, S.N. Programmable multistage drug delivery to lymph nodes. Nat. Nanotechnol. 2020, 15, 491–499. [Google Scholar] [CrossRef]
- Kang, T.; Zhu, Q.; Wei, D.; Feng, J.; Yao, J.; Jiang, T.; Song, Q.; Wei, X.; Chen, H.; Gao, X.; et al. Nanoparticles Coated with Neutrophil Membranes Can Effectively Treat Cancer Metastasis. ACS Nano 2017, 11, 1397–1411. [Google Scholar] [CrossRef]
- Gong, H.; Chao, Y.; Xiang, J.; Han, X.; Song, G.; Feng, L.; Liu, J.; Yang, G.; Chen, Q.; Liu, Z. Hyaluronidase To Enhance Nanoparticle-Based Photodynamic Tumor Therapy. Nano Lett. 2016, 16, 2512–2521. [Google Scholar] [CrossRef]
- Tsujimoto, H.; Morimoto, Y.; Takahata, R.; Nomura, S.; Yoshida, K.; Hiraki, S.; Horiguchi, H.; Miyazaki, H.; Ono, S.; Saito, D.; et al. Theranostic Photosensitive Nanoparticles for Lymph Node Metastasis of Gastric Cancer. Ann. Surg. Oncol. 2015, 22, S923–S928. [Google Scholar] [CrossRef]
- Cabral, H.; Makino, J.; Matsumoto, Y.; Mi, P.; Wu, H.; Nomoto, T.; Toh, K.; Yamada, N.; Higuchi, Y.; Konishi, S.; et al. Systemic Targeting of Lymph Node Metastasis through the Blood Vascular System by Using Size-Controlled Nanocarriers. ACS Nano 2015, 9, 4957–4967. [Google Scholar] [CrossRef]
- Jeanbart, L.; Ballester, M.; de Titta, A.; Corthésy, P.; Romero, P.; Hubbell, J.A.; Swartz, M.A. Enhancing efficacy of anticancer vaccines by targeted delivery to tumor-draining lymph nodes. Cancer Immunol. Res. 2014, 2, 436–447. [Google Scholar] [CrossRef] [PubMed]
- Khullar, O.V.; Griset, A.P.; Gibbs-Strauss, S.L.; Chirieac, L.R.; Zubris, K.A.V.; Frangioni, J.V.; Grinstaff, M.W.; Colson, Y.L. Nanoparticle Migration and Delivery of Paclitaxel to Regional Lymph Nodes in a Large Animal Model. J. Am. Coll. Surg. 2012, 214, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.; Yu, X.; Jin, C.; Yang, F.; Fu, D.; Long, J.; Xu, J.; Zhan, C.; Lu, W. LyP-1-conjugated nanoparticles for targeting drug delivery to lymphatic metastatic tumors. Int. J. Pharm. 2010, 385, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Yu, X.; Wei, J.; Zeng, F.; Li, Y.; Yang, X.; Luo, Q.; Zhang, Z. Metastatic status of sentinel lymph nodes in breast cancer determined with photoacoustic microscopy via dual-targeting nanoparticles. Light Sci. Appl. 2020, 9, 164. [Google Scholar] [CrossRef]
- Mei, L.; Rao, J.; Liu, Y.; Li, M.; Zhang, Z.; He, Q. Effective treatment of the primary tumor and lymph node metastasis by polymeric micelles with variable particle sizes. J. Control. Release 2018, 292, 67–77. [Google Scholar] [CrossRef]
- Kim, K.S.; Lee, S.; Na, K.; Bae, Y.H. Ovalbumin and Poly(i:c) Encapsulated Dendritic Cell-Targeted Nanoparticles for Immune Activation in the Small Intestinal Lymphatic System. Adv. Healthc. Mater. 2022, 11, 2200909. [Google Scholar] [CrossRef]
- Liu, H.; Moynihan, K.D.; Zheng, Y.; Szeto, G.L.; Li, A.V.; Huang, B.; Van Egeren, D.S.; Park, C.; Irvine, D.J. Structure-based programming of lymph-node targeting in molecular vaccines. Nature 2014, 507, 519–522. [Google Scholar] [CrossRef]
- Cai, X.; Liu, X.; Liao, L.-D.; Bandla, A.; Ling, J.M.; Liu, Y.-H.; Thakor, N.; Bazan, G.C.; Liu, B. Encapsulated Conjugated Oligomer Nanoparticles for Real-Time Photoacoustic Sentinel Lymph Node Imaging and Targeted Photothermal Therapy. Small 2016, 12, 4873–4880. [Google Scholar] [CrossRef]
- Muhanna, N.; MacDonald, T.D.; Chan, H.; Jin, C.S.; Burgess, L.; Cui, L.; Chen, J.; Irish, J.C.; Zheng, G. Multimodal Nanoparticle for Primary Tumor Delineation and Lymphatic Metastasis Mapping in a Head-and-Neck Cancer Rabbit Model. Adv. Healthc. Mater. 2015, 4, 2164–2169. [Google Scholar] [CrossRef]
- Kato, S.; Mori, S.; Kodama, T. A Novel Treatment Method for Lymph Node Metastasis Using a Lymphatic Drug Delivery System with Nano/Microbubbles and Ultrasound. J. Cancer 2015, 6, 1282–1294. [Google Scholar] [CrossRef]
- Tseng, Y.-C.; Xu, Z.; Guley, K.; Yuan, H.; Huang, L. Lipid–calcium phosphate nanoparticles for delivery to the lymphatic system and SPECT/CT imaging of lymph node metastases. Biomaterials 2014, 35, 4688–4698. [Google Scholar] [CrossRef]
- Shi, H.; Yan, R.; Wu, L.; Sun, Y.; Liu, S.; Zhou, Z.; He, J.; Ye, D. Tumor-targeting CuS nanoparticles for multimodal imaging and guided photothermal therapy of lymph node metastasis. Acta Biomater. 2018, 72, 256–265. [Google Scholar] [CrossRef]
- Uthaman, S.; Kim, H.S.; Revuri, V.; Min, J.-J.; Lee, Y.-K.; Huh, K.M.; Park, I.-K. Green synthesis of bioactive polysaccharide-capped gold nanoparticles for lymph node CT imaging. Carbohydr. Polym. 2018, 181, 27–33. [Google Scholar] [CrossRef]
- Kang, S.; Ahn, S.; Lee, J.; Kim, J.Y.; Choi, M.; Gujrati, V.; Kim, H.; Kim, J.; Shin, E.C.; Jon, S. Effects of gold nanoparticle-based vaccine size on lymph node delivery and cytotoxic T-lymphocyte responses. J. Control. Release 2017, 256, 56–67. [Google Scholar] [CrossRef]
- Vu-Quang, H.; Yoo, M.-K.; Jeong, H.-J.; Lee, H.-J.; Muthiah, M.; Rhee, J.H.; Lee, J.-H.; Cho, C.-S.; Jeong, Y.Y.; Park, I.-K. Targeted delivery of mannan-coated superparamagnetic iron oxide nanoparticles to antigen-presenting cells for magnetic resonance-based diagnosis of metastatic lymph nodes in vivo. Acta Biomater. 2011, 7, 3935–3945. [Google Scholar] [CrossRef]
- Oghabian, M.A.; Gharehaghaji, N.; Amirmohseni, S.; Khoei, S.; Guiti, M. Detection sensitivity of lymph nodes of various sizes using USPIO nanoparticles in magnetic resonance imaging. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 496–499. [Google Scholar] [CrossRef]
- An, M.; Li, M.; Xi, J.; Liu, H. Silica Nanoparticle as a Lymph Node Targeting Platform for Vaccine Delivery. ACS Appl. Mater. Interfaces 2017, 9, 23466–23475. [Google Scholar] [CrossRef]
- Qiao, R.; Liu, C.; Liu, M.; Hu, H.; Liu, C.; Hou, Y.; Wu, K.; Lin, Y.; Liang, J.; Gao, M. Ultrasensitive in Vivo Detection of Primary Gastric Tumor and Lymphatic Metastasis Using Upconversion Nanoparticles. ACS Nano 2015, 9, 2120–2129. [Google Scholar] [CrossRef]
- Huo, D.; He, J.; Li, H.; Huang, A.J.; Zhao, H.Y.; Ding, Y.; Zhou, Z.Y.; Hu, Y. X-ray CT guided fault-free photothermal ablation of metastatic lymph nodes with ultrafine HER-2 targeting W18O49 nanoparticles. Biomaterials 2014, 35, 9155–9166. [Google Scholar] [CrossRef]
- Yang, F.; Jin, C.; Yang, D.; Jiang, Y.; Li, J.; Di, Y.; Hu, J.; Wang, C.; Ni, Q.; Fu, D. Magnetic functionalised carbon nanotubes as drug vehicles for cancer lymph node metastasis treatment. Eur. J. Cancer 2011, 47, 1873–1882. [Google Scholar] [CrossRef]
- Zhou, K.; Wang, Y.; Huang, X.; Luby-Phelps, K.; Sumer, B.D.; Gao, J. Tunable, Ultrasensitive pH-Responsive Nanoparticles Targeting Specific Endocytic Organelles in Living Cells. Angew. Chem. Int. Ed. 2011, 50, 6109–6114. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-H.; Nguyen, D.-T.; Kim, D.-D. Recent studies on modulating hyaluronic acid-based hydrogels for controlled drug delivery. J. Pharm. Investig. 2022, 52, 397–413. [Google Scholar] [CrossRef]
- Tan, T.; Hu, H.; Wang, H.; Li, J.; Wang, Z.; Wang, J.; Wang, S.; Zhang, Z.; Li, Y. Bioinspired lipoproteins-mediated photothermia remodels tumor stroma to improve cancer cell accessibility of second nanoparticles. Nat. Commun. 2019, 10, 3322. [Google Scholar] [CrossRef] [PubMed]
- Tsopelas, C.; Sutton, R. Why Certain Dyes Are Useful for Localizing the Sentinel Lymph Node. J. Nucl. Med. 2002, 43, 1377. [Google Scholar] [PubMed]
- Markuszewski, M.; Buszewska-Forajta, M.; Artymowicz, M.; Połom, W.; Roslan, M.; Markuszewski, M. Binding indocyanine green to human serum albumin potentially enhances the detection of sentinel lymph nodes. An initial step for facilitating the detection of first-station nodes in penile and other urological cancers. Arch. Med. Sci. 2022, 18, 719–725. [Google Scholar] [CrossRef]
- Pavlista, D.; Eliska, O. Analysis of direct oil contrast lymphography of upper limb lymphatics traversing the axilla—A lesson from the past—Contribution to the concept of axillary reverse mapping. Eur. J. Surg. Oncol. 2012, 38, 390–394. [Google Scholar] [CrossRef]
- Karakatsanis, A.; Daskalakis, K.; Stålberg, P.; Olofsson, H.; Andersson, Y.; Eriksson, S.; Bergkvist, L.; Wärnberg, F. Superparamagnetic iron oxide nanoparticles as the sole method for sentinel node biopsy detection in patients with breast cancer. Br. J. Surg. 2017, 104, 1675–1685. [Google Scholar] [CrossRef]
- Winter, A.; Woenkhaus, J.; Wawroschek, F. A novel method for intraoperative sentinel lymph node detection in prostate cancer patients using superparamagnetic iron oxide nanoparticles and a handheld magnetometer: The initial clinical experience. Ann. Surg. Oncol. 2014, 21, 4390–4396. [Google Scholar] [CrossRef]
- Winter, A.; Kowald, T.; Paulo, T.S.; Goos, P.; Engels, S.; Gerullis, H.; Schiffmann, J.; Chavan, A.; Wawroschek, F. Magnetic resonance sentinel lymph node imaging and magnetometer-guided intraoperative detection in prostate cancer using superparamagnetic iron oxide nanoparticles. Int. J. Nanomed. 2018, 13, 6689–6698. [Google Scholar] [CrossRef]
- Gu, J.; Wang, J.; Nie, X.; Wang, W.; Shang, J. Potential role for carbon nanoparticles identification and preservation in situ of parathyroid glands during total thyroidectomy and central compartment node dissection. Int. J. Clin. Exp. Med. 2015, 8, 9640. [Google Scholar]
- Wu, G.; Cai, L.; Hu, J.; Zhao, R.; Ge, J.; Zhao, Y.; Shi, J.; Wang, Z. Role of carbon nanoparticles in patients with thyroid carcinoma undergoing total thyroidectomy plus bilateral central neck dissection. Zhonghua Yi Xue Za Zhi 2015, 95, 912–916. [Google Scholar]
- Xue, S.; Ren, P.; Wang, P.; Chen, G. Short and Long-Term Potential Role of Carbon Nanoparticles in Total Thyroidectomy with Central Lymph Node Dissection. Sci. Rep. 2018, 8, 11936. [Google Scholar] [CrossRef]
- Tenchov, R.; Bird, R.; Curtze, A.E.; Zhou, Q. Lipid Nanoparticles—From Liposomes to mRNA Vaccine Delivery, a Landscape of Research Diversity and Advancement. ACS Nano 2021, 15, 16982–17015. [Google Scholar] [CrossRef]
- Angelova, A.; Angelov, B. Dual and multi-drug delivery nanoparticles towards neuronal survival and synaptic repair. Neural Regen. Res. 2017, 12, 886–889. [Google Scholar] [CrossRef]
| Category | Materials | Diameter | Surface | Cargo | Probe | Route | Anima Model | End Point | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Polymer | PC7A-luminol-pyropheophorbide-a | 33.2 ± 5.9 nm (pH7.4) | Luminol/pyropheophorbide-a | S.C. | Balb/c mice bearing 4T1-GFP or CT26-GFP tumor | In vivo imaging | [13] | ||
| Methacrylate | 23–28 nm | ICG | I.V. | Orthotopic Balb/cj mice-bearing 4T1.2 tumor | In vivo imaging | [14] | |||
| Polystyrene | 100 nm or 40 nm | Polyethylene glycol (PEG) | Red fluorescent dye | I.D. | C57Bl/6J mice | In vivo imaging | [15] | ||
| Poly(propylene sulphide) | 27 ± 0.6 nm | CpG DNA | Rhodamine | I.D. | C57BL/6J mice bearing EL4-GFP tumor | In vivo efficacy | [16] | ||
| poly(lactic-co-glycolic acid) | 94.8 ± 7.6 nm | Neutrophil membrane | Carfilzomib | Coumarin-6, DiR dyes | I.V. | Balb/c nude mice bearing 4T1 tumor | In vivo efficacy | [17] | |
| poly (maleic anhydride-alt-1-octadecene) | about 33 nm | PEG | Chlorin e6 | Chlorin e6 | I.V. | Balb/c mice bearing 4T1 tumor | In vivo efficacy | [18] | |
| Poly (sarcosine)–poly (L-lactic acid) | 40–50 nm | ICG | I.V. | Balb/c nude mice bearing MKN45 tumor | In vivo efficacy | [19] | |||
| Poly(ethylene glycol)-b-poly(l-glutamic acid) | 30 ± 2 nm or 73 ± 1 nm | Dichloro(1,2-diammino cyclohexane) platinum(II) | Alexa 647, Alexa 555 dyes | I.V. | C57BL/6J mice bearing B16F10-Luc tumor | In vivo efficacy | [20] | ||
| Pluronic-stabilized polyphenylene sulfide | 30 nm | CpG DNA, ovalbumin, Trp-2 | S.C. | C57BL/6 mice bearing EG.7-OVA tumor or EL4 tumor | In vivo efficacy | [21] | |||
| 1,4-O-methacryloyl hydroquinone, coumarin comonomer | 50, 100 nm | Paclitaxel | IR-813 | I.D. | Yorkshire pigs | In vivo imaging | [22] | ||
| Poly(lactic-co-glycolic acid) | 80~90nm | LyP-1, PEG | Coumarin | S.C. | Balb/c nude mice bearing BxPC-3 tumor | In vivo imaging | [23] | ||
| Lipid | 1,2-Dimyristoyl-sn-glycero-3-phosphocholine, 1,2-dimyristoyl-sn-glycero-3-phosphoethanolamine and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-2000] | ~43 nm | Hyaluronic acid/HDL-mimicking peptide | DiR-BOA dye | S.C. | Albino-C57BL/6 mice-bearing 4T1-tfRF tumor | In vivo imaging | [24] | |
| Azide/alkyne-functionalized lipid | ~25 nm | Paclitaxel | Alexa 647 dye | I.V. | Balb/c mice bearing 4T1 tumor | In vivo efficacy | [25] | ||
| 1,2-dioleoyl-3-trimethylammonium-propane and 1,2-dioleoyl-sn-glycerol-3-phosphocholine | 157 ± 3 nm | Chondroitin sulfate-g-mannose and chondroitin sulfate-g-glycocholic acid | Ovalbumin/poly I:C | Fluorescence-labelled PEG-diacyl lipid | P.O. | C57BL/6 mice bearing B16F10 tumor & balb/c mice bearing Ct26 tumor | In vivo efficacy | [26] | |
| Endogenous albumin | - | PEG-diacyl lipid-conjugated antigen peptide | CpG DNA | S.C. | C57BL/6 mice bearing TC-1 tumor or B16F10 tumor | In vivo efficacy | [27] | ||
| 1,2-distearoyl- sn -glycero-3-phosphoethanolamine-N-[maleimide(polyethylene glycol)-2000] | ≈45 nm | Cyclic arginine-glycine-aspartic acid | N4 oligomer | S.C. | Wistar rats | In vivo imaging | [28] | ||
| Pyropheophorbide–lipid | 120 ± 5 nm | Copper 64 | I.V. | New Zealand white rabbits bearing VX2 tumor | In vivo imaging | [29] | |||
| 1,2-distearoyl-snglycero-3-phosphatidyl choline, 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-methoxy-polyethyleneglycol | ~100 nm | Perfluoropropane | I.N | MXH10/Mo/lpr mice | In vivo efficacy | [30] | |||
| Cholesterol, 1,2-dioleoyl-sn-glycero-3-phosphocholine, dioleoylphosphatydic acid,1,2-Dioleoyl-3-trimethylammonium-propane chloride salt, 1,2-distearoryl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethyleneglycol-2000), and 1-oleoyl-2-[12-[(7-nitro-2-1,3-benzoxadiazol-4-yl)amino] dodecanoyl]-sn-glycero-3-phosphocholine | ~25 nm | Indium-111 | I.V. | Balb/c mice bearing 4T1-luc2-GFP tumor | In vivo imaging | [31] | |||
| Metal | Copper sulfide nanoparticles | ~21 nm | cRGD | Cy 5.5 dye | I.T. | Balc/c mice bearing MKN45 tumor | In vivo imaging | [32] | |
| Mannan-capped gold nanoparticles | 9.18 ± 0.71 nm | Mannan | S.C. | C57BL/6 mice | In vivo imaging | [33] | |||
| Gold nanoparticles | 10, 22, 33 nm | Ovalbumin | S.C. | C57BL/6 mice bearing EG.7-OVA tumor | In vivo efficacy | [34] | |||
| Superparamagnetic iron oxide | 46.2 ± 1.9 nm | Mannan | FITC dye | I.V. | SPF/VAF outbred rats, Balb/c mice | In vivo imaging | [35] | ||
| Iron oxide nanoparticles | 20 nm | Dextran | S.C. | Wistar normal rats | In vivo imaging | [36] | |||
| Inorganic | Silica nanoparticles | 77 nm | CpG DNA, ovalbumin | S.C. | C57BL/6 mice bearing EG.7-OVA tumor | In vivo efficacy | [37] | ||
| NaGdF4:Yb,Er@NaGdF4 | 18.6 ± 0.9 nm | MGb2 antibody | I.V. | BALB/c nude mice bearing SGC7901-Luc tumor | In vivo imaging | [38] | |||
| W18O49 | 4.5 nm | HER-2 antibody | S.C. | Balb/c nude mice bearing MM435/Luc tumor | In vivo efficacy | [39] | |||
| Carbon | Multi-walled carbon nanotubes | 40~60 nm | Polyacrylic acid, Fe3O4 nanoparticles | Gemcitabine | S.C. | Balb/c nude mice bearing BxPC-3 tumor or SW1990 tumor | In vivo efficacy | [40] |
| Tracer | Detection Method | Trade Name |
|---|---|---|
| Iodine | X-ray | Lipiodol Ultra-Fluid |
| Technetium-99m | SPECT | Technecoll |
| Iron oxide | MR imaging | MagTrace, COMBIDEX |
| Perflutren | US imaging | Optison |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.; Kang, S.; Park, H.; Sun, J.G.; Kim, E.C.; Shim, G. Nanoparticles for Lymph Node-Directed Delivery. Pharmaceutics 2023, 15, 565. https://doi.org/10.3390/pharmaceutics15020565
Lee J, Kang S, Park H, Sun JG, Kim EC, Shim G. Nanoparticles for Lymph Node-Directed Delivery. Pharmaceutics. 2023; 15(2):565. https://doi.org/10.3390/pharmaceutics15020565
Chicago/Turabian StyleLee, Jaeseong, Sungtaek Kang, Hyeseon Park, Jeong Gil Sun, Eun Chae Kim, and Gayong Shim. 2023. "Nanoparticles for Lymph Node-Directed Delivery" Pharmaceutics 15, no. 2: 565. https://doi.org/10.3390/pharmaceutics15020565
APA StyleLee, J., Kang, S., Park, H., Sun, J. G., Kim, E. C., & Shim, G. (2023). Nanoparticles for Lymph Node-Directed Delivery. Pharmaceutics, 15(2), 565. https://doi.org/10.3390/pharmaceutics15020565







