Structure of Lacticaseicin 30 and Its Engineered Variants Revealed an Interplay between the N-Terminal and C-Terminal Regions in the Activity against Gram-Negative Bacteria
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains, Plasmids and Culture Conditions
2.2. Construction of Lacticaseicin 30 Variant Peptides Carrying N-Terminal Part (N-Ter Lacticaseicin 30), or the Central and C-Terminal Parts (C-Ter Lacticaseicin 30) and Their Expression in E. coli Cells
2.3. Construction of Lacticaseicin 30 Variant Plasmids for Expression in E. coli Cells
2.4. Expression and Purification of Lacticaseicin 30 and Its Variants
2.5. Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF MS)
2.6. Reverse Phase-High Performance Liquid Chromatography (RP-HPLC) Coupled to Electrospray Ionization-Mass Spectrometry (ESI-MS)
2.7. Analysis of the Ser-Lacticaseicin 30 Amino Acid Sequence by Peptide Fingerprinting
2.8. Alphafold2 Structure Prediction of Lacticaseicin 30 and Its Truncated Variants
2.9. Circular Dichroism Spectroscopy
2.10. Antimicrobial Activity
3. Results
3.1. Heterologous Expression and Characterization of Lacticaseicin 30
3.2. Conformational Analysis of Lacticaseicin 30
3.3. Design of Lacticaseicin 30 Variants
3.4. The N-Terminal Region Is Sufficient to Exert Anti-Gram-Negative Activity
3.5. Amino Acids Critical for the Anti-Gram-Negative Activity of Lacticaseicin 30
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Michael, C.A.; Dominey-Howes, D.; Labbate, M. The antimicrobial resistance crisis: Causes, consequences, and management. Front. Public Health 2014, 2, 145. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, V.K. Off-Label Abuse of Antibiotics by Bacteria. Gut. Microbes 2014, 5, 3–4. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations; Review on Antimicrobial Resistance: London, UK, 2016. [Google Scholar]
- Zalewska-Piątek, B.; Piątek, R. Bacteriophages as Potential Tools for Use in Antimicrobial Therapy and Vaccine Development. Pharmaceuticals 2021, 14, 331. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.K.; Abedon, S.T.; Loc-Carrillo, C. Phage Cocktails and the Future of Phage Therapy. Future Microbiol. 2013, 8, 769–783. [Google Scholar] [CrossRef]
- Kadouri, D.E.; To, K.; Shanks, R.M.Q.; Doi, Y. Predatory Bacteria: A Potential Ally against Multidrug-Resistant Gram-Negative Pathogens. PLoS ONE 2013, 8, e63397. [Google Scholar] [CrossRef]
- Schneitz, C. Competitive Exclusion in Poultry—30 Years of Research. Food Control 2005, 16, 657–667. [Google Scholar] [CrossRef]
- Cotter, P.D.; Ross, R.P.; Hill, C. Bacteriocins—A Viable Alternative to Antibiotics? Nat. Rev. Microbiol. 2013, 11, 95–105. [Google Scholar] [CrossRef]
- Ołdak, A.; Zielińska, D. Bacteriocins from Lactic Acid Bacteria as an Alternative to Antibiotics. Postepy Hig. Med. Dosw. Online 2017, 71, 328–338. [Google Scholar] [CrossRef]
- Soltani, S.; Hammami, R.; Cotter, P.D.; Rebuffat, S.; Said, L.B.; Gaudreau, H.; Bédard, F.; Biron, E.; Drider, D.; Fliss, I. Bacteriocins as a New Generation of Antimicrobials: Toxicity Aspects and Regulations. FEMS Microbiol. Rev. 2021, 45, fuaa039. [Google Scholar] [CrossRef]
- Allen, H.K. Alternatives to Antibiotics: Why and How; NAM Perspectives: Washington, DC, USA, 2017. [Google Scholar]
- Prokaryotic Antimicrobial Peptides: From Genes to Applications; Drider, D., Rebuffat, S., Eds.; Springer: New York, NY, USA, 2011; ISBN 978-1-4419-7691-8. [Google Scholar]
- Flaherty, R.A.; Freed, S.D.; Lee, S.W. The Wide World of Ribosomally Encoded Bacterial Peptides. PLoS Pathog. 2014, 10, e1004221. [Google Scholar] [CrossRef]
- Salazar, F.; Ortiz, A.; Sansinenea, E. Characterisation of Two Novel Bacteriocin-like Substances Produced by Bacillus amyloliquefaciens ELI149 with Broad-Spectrum Antimicrobial Activity. J. Glob Antimicrob. Resist. 2017, 11, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-C.; Lin, C.-H.; Sung, C.T.; Fang, J.-Y. Antibacterial Activities of Bacteriocins: Application in Foods and Pharmaceuticals. Front. Microbiol. 2014, 5, 241. [Google Scholar] [CrossRef]
- Dobson, A.; Cotter, P.D.; Ross, R.P.; Hill, C. Bacteriocin Production: A Probiotic Trait? Appl. Environ. Microbiol. 2012, 78, 1–6. [Google Scholar] [CrossRef]
- Chikindas, M.L.; Weeks, R.; Drider, D.; Chistyakov, V.A.; Dicks, L.M. Functions and Emerging Applications of Bacteriocins. Curr. Opin. Biotechnol. 2018, 49, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Belguesmia, Y.; Madi, A.; Sperandio, D.; Merieau, A.; Feuilloley, M.; Prévost, H.; Drider, D.; Connil, N. Growing Insights into the Safety of Bacteriocins: The Case of Enterocin S37. Res. Microbiol. 2011, 162, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Eveno, M.; Savard, P.; Belguesmia, Y.; Bazinet, L.; Gancel, F.; Drider, D.; Fliss, I. Compatibility, Cytotoxicity, and Gastrointestinal Tenacity of Bacteriocin-Producing Bacteria Selected for a Consortium Probiotic Formulation to Be Used in Livestock Feed. Probiotics Antimicrob. Proteins 2021, 13, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Simons, A.; Alhanout, K.; Duval, R.E. Bacteriocins, Antimicrobial Peptides from Bacterial Origin: Overview of Their Biology and Their Impact against Multidrug-Resistant Bacteria. Microorganisms 2020, 8, 639. [Google Scholar] [CrossRef]
- Pérez-Ramos, A.; Madi-Moussa, D.; Coucheney, F.; Drider, D. Current Knowledge of the Mode of Action and Immunity Mechanisms of LAB-Bacteriocins. Microorganisms 2021, 9, 2107. [Google Scholar] [CrossRef]
- Alvarez-Sieiro, P.; Montalbán-López, M.; Mu, D.; Kuipers, O.P. Bacteriocins of Lactic Acid Bacteria: Extending the Family. Appl. Microbiol. Biotechnol. 2016, 100, 2939–2951. [Google Scholar] [CrossRef]
- Cotter, P.D.; Hill, C.; Ross, R.P. Bacteriocins: Developing Innate Immunity for Food. Nat. Rev. Microbiol. 2005, 3, 777–788. [Google Scholar] [CrossRef]
- Drider, D.; Bendali, F.; Naghmouchi, K.; Chikindas, M.L. Bacteriocins: Not Only Antibacterial Agents. Probiotics Antimicrob. Proteins 2016, 8, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Messaoudi, S.; Kergourlay, G.; Dalgalarrondo, M.; Choiset, Y.; Ferchichi, M.; Prévost, H.; Pilet, M.-F.; Chobert, J.-M.; Manai, M.; Dousset, X. Purification and Characterization of a New Bacteriocin Active against Campylobacter Produced by Lactobacillus salivarius SMXD51. Food Microbiol. 2012, 32, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Stern, N.J.; Svetoch, E.A.; Eruslanov, B.V.; Perelygin, V.V.; Mitsevich, E.V.; Mitsevich, I.P.; Seal, B.S. Isolation of a Lactobacillus salivarius Strain and Purification of Its Bacteriocin, Which Is Inhibitory to Campylobacter jejuni in the Chicken Gastrointestinal System. Antimicrob. Agents Chemother 2006, 50, 3111–3116. [Google Scholar] [CrossRef] [PubMed]
- Todorov, S.D.; Dicks, L.M.T. Lactobacillus plantarum Isolated from Molasses Produces Bacteriocins Active against Gram-Negative Bacteria. Enzym. Microb. Technol. 2005, 36, 318–326. [Google Scholar] [CrossRef]
- Naghmouchi, K.; Drider, D.; Fliss, I. Action of Divergicin M35, a Class IIa Bacteriocin, on Liposomes and Listeria. J. Appl. Microbiol. 2007, 102, 1508–1517. [Google Scholar] [CrossRef] [PubMed]
- Belguesmia, Y.; Bendjeddou, K.; Kempf, I.; Boukherroub, R.; Drider, D. Heterologous Biosynthesis of Five New Class II Bacteriocins from Lactobacillus paracasei CNCM I-5369 with Antagonistic Activity against Pathogenic Escherichia coli Strains. Front. Microbiol. 2020, 11, 1198. [Google Scholar] [CrossRef] [PubMed]
- Madi-Moussa, D.; Coucheney, F.; Drider, D. Expression of Five Class II Bacteriocins with Activity against Escherichia coli in Lacticaseibacillus paracasei CNCM I-5369, and in a Heterologous Host. Biotechnol. Rep. 2021, 30, e00632. [Google Scholar] [CrossRef] [PubMed]
- Sezonov, G.; Joseleau-Petit, D.; D’Ari, R. Escherichia coli physiology in Luria-Bertani broth. J. Bacteriol. 2007, 189, 8746–8749. [Google Scholar] [CrossRef]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory, 3rd ed.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2001. [Google Scholar]
- Schägger, H. Tricine–SDS-PAGE. 1. Nat. Protoc. 2006, 1, 16–22. [Google Scholar] [CrossRef]
- Wessel, D.; Flügge, U.I. A Method for the Quantitative Recovery of Protein in Dilute Solution in the Presence of Detergents and Lipids. Anal. Biochem. 1984, 138, 141–143. [Google Scholar] [CrossRef]
- Marty, M.T.; Baldwin, A.J.; Marklund, E.G.; Hochberg, G.K.A.; Benesch, J.L.P.; Robinson, C.V. Bayesian Deconvolution of Mass and Ion Mobility Spectra: From Binary Interactions to Polydisperse Ensembles. Anal. Chem. 2015, 87, 4370–4376. [Google Scholar] [CrossRef] [PubMed]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly Accurate Protein Structure Prediction with AlphaFold. 7873. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Hornak, V.; Abel, R.; Okur, A.; Strockbine, B.; Roitberg, A.; Simmerling, C. Comparison of Multiple Amber Force Fields and Development of Improved Protein Backbone Parameters. Proteins Struct. Funct. Bioinform. 2006, 65, 712–725. [Google Scholar] [CrossRef]
- Eastman, P.; Swails, J.; Chodera, J.D.; McGibbon, R.T.; Zhao, Y.; Beauchamp, K.A.; Wang, L.-P.; Simmonett, A.C.; Harrigan, M.P.; Stern, C.D.; et al. OpenMM 7: Rapid Development of High-Performance Algorithms for Molecular Dynamics. PLoS Comput. Biol. 2017, 13, e1005659. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Meng, E.C.; Couch, G.S.; Croll, T.I.; Morris, J.H.; Ferrin, T.E. UCSF ChimeraX: Structure Visualization for Researchers, Educators, and Developers. Protein Sci. 2021, 30, 70–82. [Google Scholar] [CrossRef]
- Green, A.A. The Preparation of Acetate and Phosphate Buffer Solutions of Known PH and Ionic Strength. J. Am. Chem. Soc. 1933, 55, 2331–2336. [Google Scholar] [CrossRef]
- Micsonai, A.; Wien, F.; Bulyáki, É.; Kun, J.; Moussong, É.; Lee, Y.-H.; Goto, Y.; Réfrégiers, M.; Kardos, J. BeStSel: A Web Server for Accurate Protein Secondary Structure Prediction and Fold Recognition from the Circular Dichroism Spectra. Nucleic Acids Res. 2018, 46, W315–W322. [Google Scholar] [CrossRef]
- Roy, A.; Kucukural, A.; Zhang, Y. I-TASSER: A Unified Platform for Automated Protein Structure and Function Prediction. Nat. Protoc. 2010, 5, 725–738. [Google Scholar] [CrossRef]
- Yang, F.; Hou, C.; Zeng, X.; Qiao, S. The Use of Lactic Acid Bacteria as a Probiotic in Swine Diets. Pathogens 2015, 4, 34–45. [Google Scholar] [CrossRef]
- Daba, H.; Pandian, S.; Gosselin, J.F.; Simard, R.E.; Huang, J.; Lacroix, C. Detection and activity of a bacteriocin produced by Leuconostoc mesenteroides. Appl. Environ. Microbiol. 1991, 57, 3450–3455. [Google Scholar] [CrossRef]
- Batdorj, B.; Dalgalarrondo, M.; Choiset, Y.; Pedroche, J.; Métro, F.; Prévost, H.; Chobert, J.-M.; Haertlé, T. Purification and Characterization of Two Bacteriocins Produced by Lactic Acid Bacteria Isolated from Mongolian Airag. J. Appl. Microbiol. 2006, 101, 837–848. [Google Scholar] [CrossRef] [PubMed]
- Goulet, A.; Cambillau, C. Present Impact of AlphaFold2 Revolution on Structural Biology, and an Illustration with the Structure Prediction of the Bacteriophage J-1 Host Adhesion Device. Front. Mol. Biosci. 2022, 9, 907452. [Google Scholar] [CrossRef] [PubMed]
- Tulumello, D.V.; Deber, C.M. SDS Micelles as a Membrane-Mimetic Environment for Transmembrane Segments. Biochemistry 2009, 48, 12096–12103. [Google Scholar] [CrossRef] [PubMed]
- Pommer, A.J.; Wallis, R.; Moore, G.R.; James, R.; Kleanthous, C. Enzymological Characterization of the Nuclease Domain from the Bacterial Toxin Colicin E9 from Escherichia coli. Biochem. J. 1998, 334, 387–392. [Google Scholar] [CrossRef]
- Meade, E.; Slattery, M.A.; Garvey, M. Bacteriocins, Potent Antimicrobial Peptides and the Fight against Multi Drug Resistant Species: Resistance Is Futile? Antibiotics 2020, 9, 32. [Google Scholar] [CrossRef]
- da Silva Sabo, S.; Vitolo, M.; González, J.M.D.; Oliveira, R.P. de S. Overview of Lactobacillus plantarum as a Promising Bacteriocin Producer among Lactic Acid Bacteria. Food Res. Int. 2014, 64, 527–536. [Google Scholar] [CrossRef]
- Li, J.; Koh, J.-J.; Liu, S.; Lakshminarayanan, R.; Verma, C.S.; Beuerman, R.W. Membrane Active Antimicrobial Peptides: Translating Mechanistic Insights to Design. Front. Neurosci. 2017, 11, 73. [Google Scholar] [CrossRef]
- Epand, R.M.; Vogel, H.J. Diversity of Antimicrobial Peptides and Their Mechanisms of Action. Biochim. Biophys Acta 1999, 1462, 11–28. [Google Scholar] [CrossRef]
- Harris, F.; Dennison, S.; Phoenix, D.A. Anionic antimicrobial peptides from eukaryotic organisms. Curr. Protein Pept. Sci. 2009, 10, 585–606. [Google Scholar] [CrossRef]
- Clark, S.; Jowitt, T.A.; Harris, L.K.; Knight, C.G.; Dobson, C.B. The Lexicon of Antimicrobial Peptides: A Complete Set of Arginine and Tryptophan Sequences. Commun. Biol. 2021, 4, 605. [Google Scholar] [CrossRef]
- van Kraaij, C.; Breukink, E.; Noordermeer, M.A.; Demel, R.A.; Siezen, R.J.; Kuipers, O.P.; de Kruijff, B. Pore Formation by Nisin Involves Translocation of Its C-Terminal Part across the Membrane. Biochemistry 1998, 37, 16033–16040. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, L.; Fimland, G.; Nissen-Meyer, J. The C-Terminal Domain of Pediocin-like Antimicrobial Peptides (Class IIa Bacteriocins) Is Involved in Specific Recognition of the C-Terminal Part of Cognate Immunity Proteins and in Determining the Antimicrobial Spectrum. J. Biol. Chem. 2005, 280, 9243–9250. [Google Scholar] [CrossRef] [PubMed]
- Rihakova, J.; Petit, V.W.; Demnerova, K.; Prévost, H.; Rebuffat, S.; Drider, D. Insights into Structure-Activity Relationships in the C-Terminal Region of Divercin V41, a Class IIa Bacteriocin with High-Level Antilisterial Activity. Appl. Environ. Microbiol. 2009, 75, 1811–1819. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.D.T.; Sothiselvam, S.; Lu, T.K.; Fuente-Nunez, C. Peptide Design Principles for Antimicrobial Applications. J. Mol. Biol. 2019, 431, 3547–3567. [Google Scholar] [CrossRef]
- Mattiuzzo, M.; Bandiera, A.; Gennaro, R.; Benincasa, M.; Pacor, S.; Antcheva, N.; Scocchi, M. Role of the Escherichia coli SbmA in the Antimicrobial Activity of Proline-Rich Peptides. Mol. Microbiol. 2007, 66, 151–163. [Google Scholar] [CrossRef]
- Seefeldt, A.C.; Nguyen, F.; Antunes, S.; Pérébaskine, N.; Graf, M.; Arenz, S.; Inampudi, K.K.; Douat, C.; Guichard, G.; Wilson, D.N.; et al. The Proline-Rich Antimicrobial Peptide Onc112 Inhibits Translation by Blocking and Destabilizing the Initiation Complex. Nat. Struct Mol. Biol. 2015, 22, 470–475. [Google Scholar] [CrossRef]
- Welch, N.G.; Li, W.; Hossain, M.A.; Separovic, F.; O’Brien-Simpson, N.M.; Wade, J.D. (Re)Defining the Proline-Rich Antimicrobial Peptide Family and the Identification of Putative New Members. Front. Chem. 2020, 8, 607769. [Google Scholar] [CrossRef]
- Kwon, Y.M.; Kim, H.J.; Kim, Y.I.; Kang, Y.J.; Lee, I.H.; Jin, B.R.; Han, Y.S.; Cheon, H.M.; Ha, N.G.; Seo, S.J. Comparative Analysis of Two Attacin Genes from Hyphantria Cunea. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2008, 151, 213–220. [Google Scholar] [CrossRef]
- Lee, J.H.; Cho, K.S.; Lee, J.; Yoo, J.; Lee, J.; Chung, J. Diptericin-like Protein: An Immune Response Gene Regulated by the Anti-Bacterial Gene Induction Pathway in Drosophila. Gene 2001, 271, 233–238. [Google Scholar] [CrossRef]


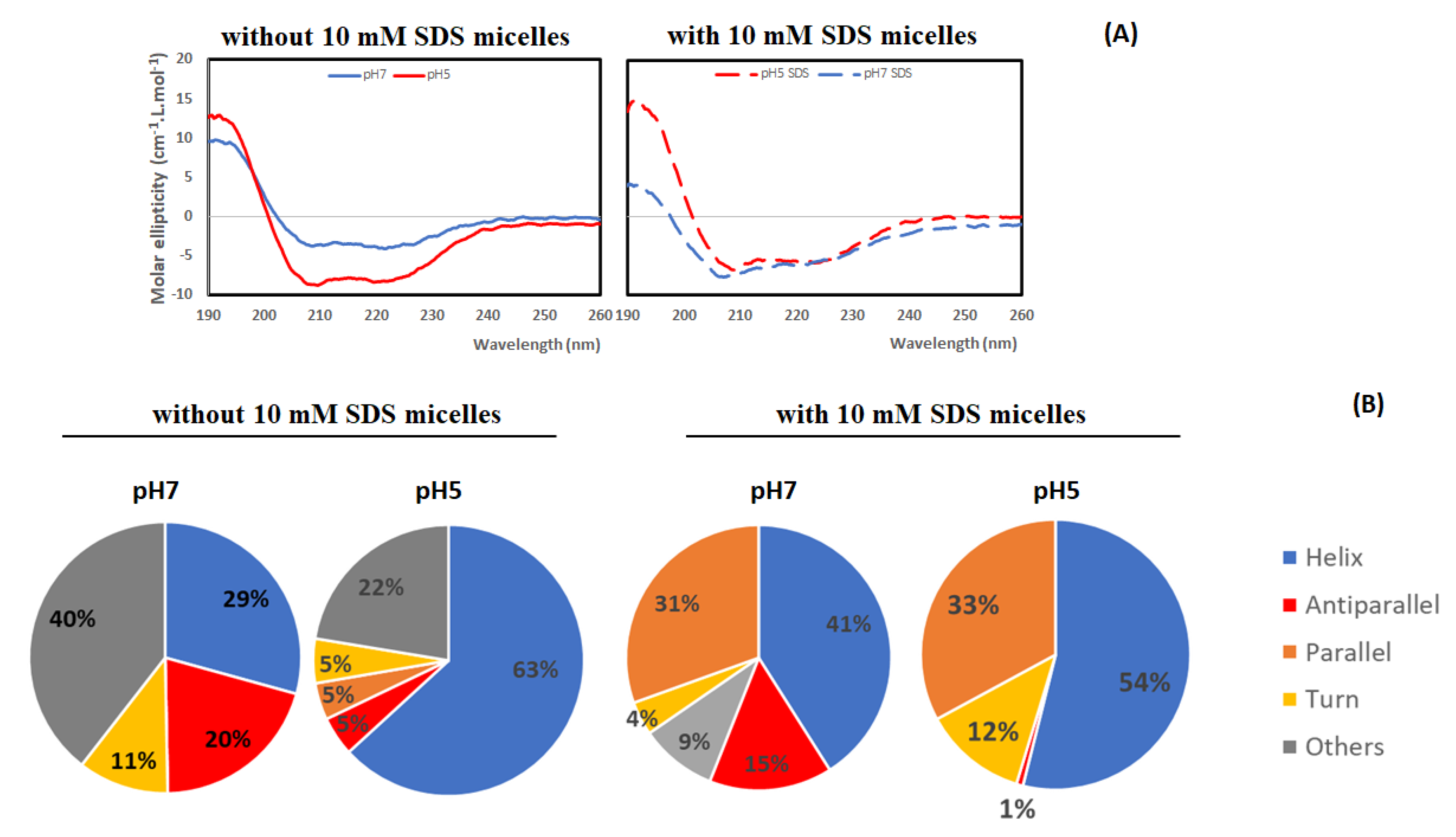
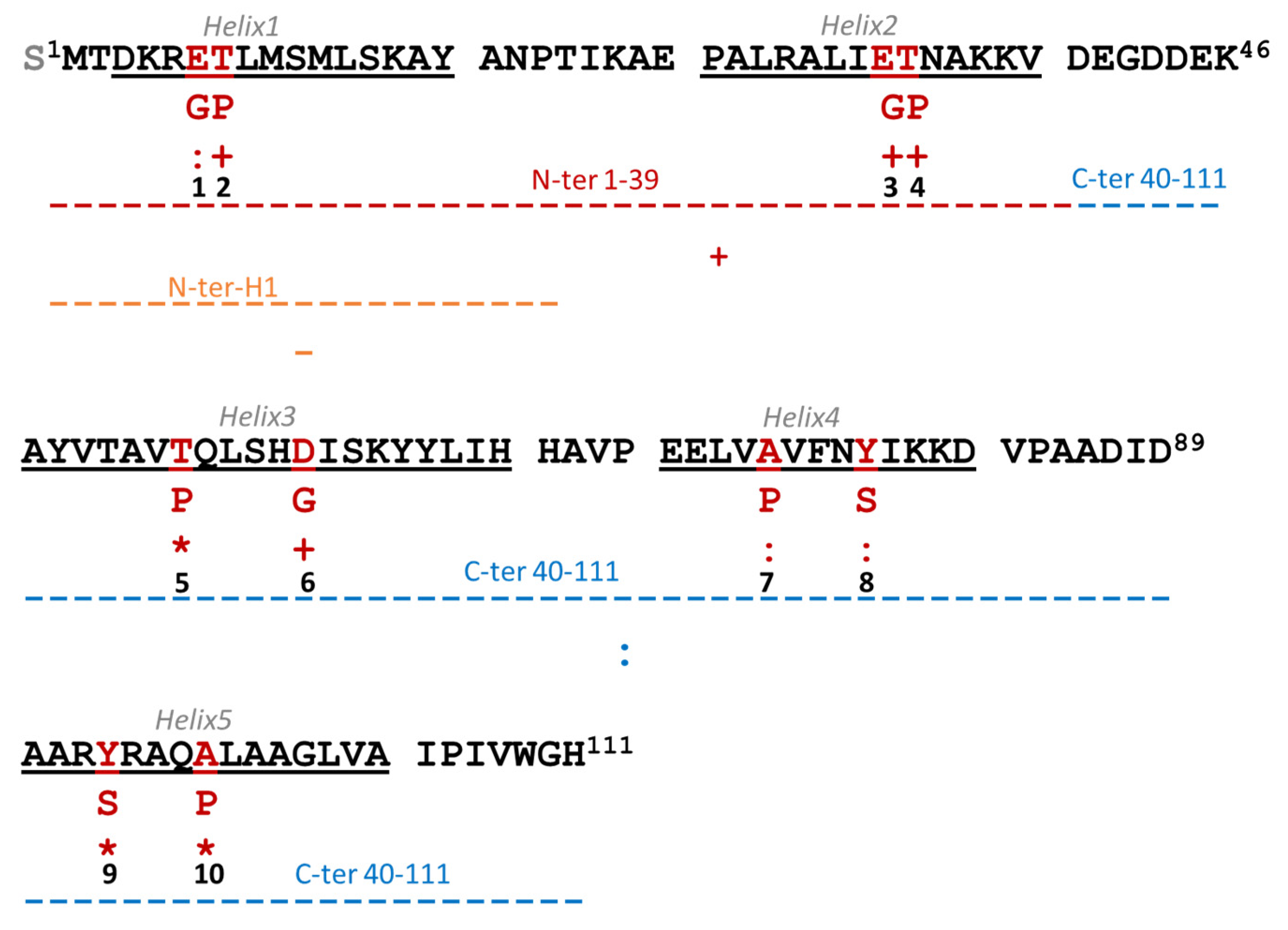
| Peptides | Molecular Mas (Da) | Escherichia coli ATCC 8739 | Salmonella enterica Serotype Newport 6962 | Proteus vulgaris ATCC 3342 | Pseudomonas aeruginosa ATCC 27853 | Listeria innocua CIP 80.11 |
|---|---|---|---|---|---|---|
| Lacticaseicin 30 | 12,252.09 | 40 | 80 | 40 | 160 | 100 |
| N-ter Lacticaseicin 30 | 4236.03 | 40 | 80 | 40 | 320 | 100 |
| C-ter Lacticaseicin 30 | 8034.08 | 160 | 160 | 160 | 320 | 100 |
| N-ter-H1 Lacticaseicin 30 | 1905.27 | - | - | - | - | 100 |
| Peptides | Molecular Mass (Da) | Putative Helix | Escherichia coli ATCC 8739 | Salmonella Newport ATCC 6962 | Proteus vulgaris ATCC 33420 | Pseudomonas aeruginosa ATCC 27853 |
|---|---|---|---|---|---|---|
| Lacticaseicin 30 | 12,252.09 | - | 40 | 80 | 40 | 100 |
| E6G | 12,180.03 | H1 | 100 | 160 | 100 | 400 |
| T7P | 12,248.10 | H1 | 60 | 160 | 60 | 200 |
| E32G | 12,180.03 | H2 | 40 | 80 | 40 | 100 |
| T33P | 12,248.10 | H2 | 40 | 80 | 40 | 100 |
| T52P | 12,248.10 | H3 | 70 | 140 | 70 | 200 |
| D57G | 12,194.06 | H3 | 40 | 80 | 40 | 100 |
| A74P | 12,278.13 | H4 | 100 | 160 | 100 | 200 |
| Y78S | 12,176.00 | H4 | 100 | 200 | 100 | 400 |
| Y93S | 12,176.00 | H5 | 70 | 140 | 70 | 200 |
| A97P | 12,278.13 | H5 | 60 | 100 | 60 | 200 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Madi-Moussa, D.; Deracinois, B.; Teiar, R.; Li, Y.; Mihasan, M.; Flahaut, C.; Rebuffat, S.; Coucheney, F.; Drider, D. Structure of Lacticaseicin 30 and Its Engineered Variants Revealed an Interplay between the N-Terminal and C-Terminal Regions in the Activity against Gram-Negative Bacteria. Pharmaceutics 2022, 14, 1921. https://doi.org/10.3390/pharmaceutics14091921
Madi-Moussa D, Deracinois B, Teiar R, Li Y, Mihasan M, Flahaut C, Rebuffat S, Coucheney F, Drider D. Structure of Lacticaseicin 30 and Its Engineered Variants Revealed an Interplay between the N-Terminal and C-Terminal Regions in the Activity against Gram-Negative Bacteria. Pharmaceutics. 2022; 14(9):1921. https://doi.org/10.3390/pharmaceutics14091921
Chicago/Turabian StyleMadi-Moussa, Désiré, Barbara Deracinois, Radja Teiar, Yanyan Li, Marius Mihasan, Christophe Flahaut, Sylvie Rebuffat, Françoise Coucheney, and Djamel Drider. 2022. "Structure of Lacticaseicin 30 and Its Engineered Variants Revealed an Interplay between the N-Terminal and C-Terminal Regions in the Activity against Gram-Negative Bacteria" Pharmaceutics 14, no. 9: 1921. https://doi.org/10.3390/pharmaceutics14091921
APA StyleMadi-Moussa, D., Deracinois, B., Teiar, R., Li, Y., Mihasan, M., Flahaut, C., Rebuffat, S., Coucheney, F., & Drider, D. (2022). Structure of Lacticaseicin 30 and Its Engineered Variants Revealed an Interplay between the N-Terminal and C-Terminal Regions in the Activity against Gram-Negative Bacteria. Pharmaceutics, 14(9), 1921. https://doi.org/10.3390/pharmaceutics14091921










