Emerging Bioactive Agent Delivery-Based Regenerative Therapies for Lower Genitourinary Tissues
Abstract
:1. Introduction
2. Classification of Biomaterials Used in Delivery Systems
2.1. Inorganic Biomaterials
2.2. Organic Biomaterials
3. Delivery Strategies for Regenerative Therapy
3.1. Biomaterial Selection and Performance Optimization
3.2. Fabrication Strategies for Delivery Systems
3.3. Loading Strategies
4. Therapeutic Applications for Lower GU Injuries
4.1. Female Reproductive System
4.1.1. Uterus
4.1.2. Ovaries
4.1.3. Cervix and Vagina
4.2. Male Reproductive System
4.3. Urinary System
5. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Tullington, J.E.; Blecker, N. Lower Genitourinary Trauma; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Balzano, F.L.; Hudak, S.J. Military genitourinary injuries: Past, present, and future. Transl. Androl. Urol. 2018, 7, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Bayne, D.; Zaid, U.; Alwaal, A.; Harris, C.; McAninch, J.; Breyer, B. Lower genitourinary tract trauma. Trauma 2015, 18, 12–20. [Google Scholar] [CrossRef]
- Caneparo, C.; Sorroza-Martinez, L.; Chabaud, S.; Fradette, J.; Bolduc, S. Considerations for the clinical use of stem cells in genitourinary regenerative medicine. World J. Stem Cells 2021, 13, 1480–1512. [Google Scholar] [CrossRef] [PubMed]
- Atienza-Roca, P.; Cui, X.; Hooper, G.J.; Woodfield, T.B.F.; Lim, K.S. Growth factor delivery systems for tissue engineering and regenerative medicine. Adv. Exp. Med. Biol. 2018, 1078, 245–269. [Google Scholar] [CrossRef]
- Doostmohammadi, M.; Forootanfar, H.; Ramakrishna, S. Regenerative medicine and drug delivery: Progress via electrospun biomaterials. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 109, 110521. [Google Scholar] [CrossRef]
- Tang, X.; Sun, C. The roles of MicroRNAs in neural regenerative medicine. Exp. Neurol. 2020, 332, 113394. [Google Scholar] [CrossRef]
- Caballero Aguilar, L.M.; Silva, S.M.; Moulton, S.E. Growth factor delivery: Defining the next generation platforms for tissue engineering. J. Control. Release 2019, 306, 40–58. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Z.; Sandoval-Salaiza, D.A.; Afewerki, S.; Jimenez-Rodriguez, M.G.; Sanchez-Melgar, L.; Güemes-Aguilar, G.; Gonzalez-Sanchez, D.G.; Noble, O.; Lerma, C.; et al. Nanostructured non-newtonian drug delivery barrier prevents postoperative intrapericardial adhesions. ACS Appl. Mater. Interfaces 2021, 13, 29231–29246. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, X. Curcumin loading on alginate nano-micelle for anti-infection and colonic wound healing. J. Biomed. Nanotechnol. 2021, 17, 1160–1169. [Google Scholar] [CrossRef]
- Subbiah, R.; Guldberg, R.E. Materials science and design principles of growth factor delivery systems in tissue engineering and regenerative medicine. Adv. Healthc. Mater. 2019, 8, e1801000. [Google Scholar] [CrossRef]
- Hafeez, M.N.; d’Avanzo, N.; Russo, V.; Di Marzio, L.; Cilurzo, F.; Paolino, D.; Fresta, M.; Barboni, B.; Santos, H.A.; Celia, C. Tendon tissue repair in prospective of drug delivery, regenerative medicines, and innovative bioscaffolds. Stem Cells Int. 2021, 2021, 1488829. [Google Scholar] [CrossRef]
- Liu, R.; Poma, A. Advances in molecularly imprinted polymers as drug delivery systems. Molecules 2021, 26, 3589. [Google Scholar] [CrossRef]
- Harish, V.; Tewari, D.; Gaur, M.; Yadav, A.B.; Swaroop, S.; Bechelany, M.; Barhoum, A. Review on nanoparticles and nanostructured materials: Bioimaging, biosensing, drug delivery, tissue engineering, antimicrobial, and agro-food applications. Nanomaterials 2022, 12, 457. [Google Scholar] [CrossRef]
- Colucci, F.; Mancini, V.; Mattu, C.; Boffito, M. Designing multifunctional devices for regenerative pharmacology based on 3D scaffolds, drug-loaded nanoparticles, and thermosensitive hydrogels: A proof-of-concept study. Pharmaceutics 2021, 13, 464. [Google Scholar] [CrossRef]
- Mohseni, M.; Shokrollahi, P.; Barzin, J. Impact of supramolecular interactions on delivery of dexamethasone from a physical network of gelatin/ZnHAp composite scaffold. Int. J. Pharm. 2022, 615, 121520. [Google Scholar] [CrossRef]
- Li, X.; Wang, C.; Wang, L.; Huang, R.; Li, W.C.; Wang, X.; Wong, S.S.W.; Cai, Z.; Leung, K.C.; Jin, L. A glutathione-responsive silica-based nanosystem capped with in-situ polymerized cell-penetrating poly(disulfide)s for precisely modulating immuno-inflammatory responses. J. Colloid Interface Sci. 2022, 614, 322–336. [Google Scholar] [CrossRef]
- Wnorowska, U.; Fiedoruk, K.; Piktel, E.; Prasad, S.V.; Sulik, M.; Janion, M.; Daniluk, T.; Savage, P.B.; Bucki, R. Nanoantibiotics containing membrane-active human cathelicidin LL-37 or synthetic ceragenins attached to the surface of magnetic nanoparticles as novel and innovative therapeutic tools: Current status and potential future applications. J. Nanobiotechnol. 2020, 18, 3. [Google Scholar] [CrossRef]
- Kimicata, M.; Mahadik, B.; Fisher, J.P. Long-term sustained drug delivery via 3D printed masks for the development of a heparin-loaded interlayer in vascular tissue engineering applications. ACS Appl. Mater. Interfaces 2021, 13, 50812–50822. [Google Scholar] [CrossRef]
- Lin, J.; Wang, Z.; Huang, J.; Tang, S.; Saiding, Q.; Zhu, Q.; Cui, W. Microenvironment-protected exosome-hydrogel for facilitating endometrial regeneration, fertility restoration, and live birth of offspring. Small 2021, 17, e2007235. [Google Scholar] [CrossRef]
- Wang, L.; Cheng, W.; Zhu, J.; Li, W.; Li, D.; Yang, X.; Zhao, W.; Ren, M.; Ren, J.; Mo, X.; et al. Electrospun nanoyarn and exosomes of adipose-derived stem cells for urethral regeneration: Evaluations in vitro and in vivo. Colloids Surf. B Biointerfaces 2022, 209, 112218. [Google Scholar] [CrossRef]
- Liang, L.; Shen, Y.; Dong, Z.; Gu, X. Photoacoustic image-guided corpus cavernosum intratunical injection of adipose stem cell-derived exosomes loaded polydopamine thermosensitive hydrogel for erectile dysfunction treatment. Bioact. Mater. 2022, 9, 147–156. [Google Scholar] [CrossRef]
- Gimondi, S.; Guimarães, C.F.; Vieira, S.F.; Gonçalves, V.M.F.; Tiritan, M.E.; Reis, R.L.; Ferreira, H.; Neves, N.M. Microfluidic mixing system for precise PLGA-PEG nanoparticles size control. Nanomedicine 2022, 40, 102482. [Google Scholar] [CrossRef]
- Guo, G.; Gong, T.; Shen, H.; Wang, Q.; Jiang, F.; Tang, J.; Jiang, X.; Wang, J.; Zhang, X.; Bu, W. Self-amplification immunomodulatory strategy for tissue regeneration in diabetes based on cytokine-zifs system. Adv. Funct. Meter. 2021, 31, 2100795. [Google Scholar] [CrossRef]
- Wu, J.; Zhu, J.; Wu, Q.; An, Y.; Wang, K.; Xuan, T.; Zhang, J.; Song, W.; He, H.; Song, L.; et al. Mussel-inspired surface immobilization of heparin on magnetic nanoparticles for enhanced wound repair via sustained release of a growth factor and M2 macrophage polarization. ACS Appl. Mater. Interfaces 2021, 13, 2230–2244. [Google Scholar] [CrossRef]
- Babić Radić, M.M.; Filipović, V.V.; Vukomanović, M.; Nikodinović, R.J.; Tomić, S.L. Degradable 2-hydroxyethyl methacrylate/gelatin/alginate hydrogels infused by nanocolloidal graphene oxide as promising drug delivery and scaffolding biomaterials. Gels 2022, 8, 22. [Google Scholar] [CrossRef]
- Perteghella, S.; Crivelli, B.; Catenacci, L.; Sorrenti, M.; Bruni, G.; Necchi, V.; Vigani, B.; Sorlini, M.; Torre, M.L.; Chlapanidas, T. Stem cell-extracellular vesicles as drug delivery systems: New frontiers for silk/curcumin nanoparticles. Int. J. Pharm. 2017, 520, 86–97. [Google Scholar] [CrossRef]
- Gaurav, I.; Thakur, A.; Iyaswamy, A.; Wang, X.; Chen, X.; Yang, Z. Factors affecting extracellular vesicles based drug delivery systems. Molecules 2021, 26, 1544. [Google Scholar] [CrossRef]
- Skopinska-Wisniewska, J.; Flor, S.; Kozlowska, J. From supramolecular hydrogels to multifunctional carriers for biologically active substances. Int. J. Mol. Sci. 2021, 22, 7402. [Google Scholar] [CrossRef]
- Sun, Y.; Zheng, L.; Yang, Y.; Qian, X.; Fu, T.; Li, X.; Yang, Z.; Yan, H.; Cui, C.; Tan, W. Metal-organic framework nanocarriers for drug delivery in biomedical applications. Nanomicro. Lett. 2020, 12, 103. [Google Scholar] [CrossRef]
- Filippi, M.; Garello, F.; Yasa, O.; Kasamkattil, J.; Scherberich, A.; Katzschmann, R.K. Engineered magnetic nanocomposites to modulate cellular function. Small 2022, 18, e2104079. [Google Scholar] [CrossRef]
- Da, L.C.; Huang, Y.Z.; Xie, H.Q.; Zheng, B.H.; Huang, Y.C.; Du, S.R. Membranous extracellular matrix-based scaffolds for skin wound healing. Pharmaceutics 2021, 13, 1796. [Google Scholar] [CrossRef] [PubMed]
- Khosravi, A.; Hasani, A.; Rahimi, K.; Aliaghaei, A.; Pirani, M.; Azad, N.; Ramezani, F.; Tamimi, A.; Behnam, P.; Raoofi, A.; et al. Ameliorating effects of curcumin-loaded superparamagnetic iron oxide nanoparticles (SPIONs) on the mouse testis exposed to the transient hyperthermia: A molecular and stereological study. Acta Histochem. 2020, 122, 151632. [Google Scholar] [CrossRef] [PubMed]
- Sadahide, K.; Teishima, J.; Inoue, S.; Tamura, T.; Kamei, N.; Adachi, N.; Matsubara, A. Endoscopic repair of the urinary bladder with magnetically labeled mesenchymal stem cells: Preliminary report. Regen. Ther. 2019, 10, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Rastegari, E.; Hsiao, Y.J.; Lai, W.Y.; Lai, Y.H.; Yang, T.C.; Chen, S.J.; Huang, P.I.; Chiou, S.H.; Mou, C.Y.; Yueh, C. An update on mesoporous silica nanoparticle applications in nanomedicine. Pharmaceutics 2021, 13, 1067. [Google Scholar] [CrossRef]
- Hamam, F.; Nasr, A. Curcumin-loaded mesoporous silica particles as wound-healing agent: An in vivo study. Saudi J. Med. Med. Sci. 2020, 8, 17–24. [Google Scholar] [CrossRef]
- Wu, H.; Li, F.; Wang, S.; Lu, J.; Li, J.; Du, Y.; Sun, X.; Chen, X.; Gao, J.; Ling, D. Ceria nanocrystals decorated mesoporous silica nanoparticle based ROS-scavenging tissue adhesive for highly efficient regenerative wound healing. Biomaterials 2018, 151, 66–77. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, K.; Wang, J.; Zhao, R.; Zhang, Q.; Kong, X. Poly(amidoamine)-modified mesoporous silica nanoparticles as a mucoadhesive drug delivery system for potential bladder cancer therapy. Colloids Surf. B Biointerfaces 2020, 189, 110832. [Google Scholar] [CrossRef]
- Henry, N.; Clouet, J.; Le Bideau, J.; Le Visage, C.; Guicheux, J. Innovative strategies for intervertebral disc regenerative medicine: From cell therapies to multiscale delivery systems. Biotechnol. Adv. 2018, 36, 281–294. [Google Scholar] [CrossRef]
- Abbas, A.; Zhang, C.; Asad, M.; Waqas, A.; Khatoon, A.; Hussain, S.; Mir, S.H. Recent developments in artificial super-wettable surfaces based on bioinspired polymeric materials for biomedical applications. Polymers 2022, 14, 238. [Google Scholar] [CrossRef]
- Ciardulli, M.C.; Lovecchio, J.; Scala, P.; Lamparelli, E.P.; Dale, T.P.; Giudice, V.; Giordano, E.; Selleri, C.; Forsyth, N.R.; Maffulli, N.; et al. 3D biomimetic scaffold for growth factor controlled delivery: An in-vitro study of tenogenic events on wharton’s jelly mesenchymal stem cells. Pharmaceutics 2021, 13, 1448. [Google Scholar] [CrossRef]
- Sok, M.C.P.; Baker, N.; McClain, C.; Lim, H.S.; Turner, T.; Hymel, L.; Ogle, M.; Olingy, C.; Palacios, J.I.; Garcia, J.R.; et al. Dual delivery of IL-10 and AT-RvD1 from PEG hydrogels polarize immune cells towards pro-regenerative phenotypes. Biomaterials 2021, 268, 120475. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, D.; Hui, Q.; Bi, J.; Yu, B.; Huang, Z.; Hu, S.; Wang, Z.; Caranasos, T.; Rossi, J.; et al. Injection of ROS-responsive hydrogel loaded with basic fibroblast growth factor into the pericardial cavity for heart repair. Adv. Funct. Mater. 2021, 31, 2004377. [Google Scholar] [CrossRef]
- Yuan, Q.; Huang, J.; Xian, C.; Wu, J. Amino acid- and growth factor-based multifunctional nanocapsules for the modulation of the local microenvironment in tissue engineering. ACS Appl. Mater. Interfaces 2021, 13, 2165–2178. [Google Scholar] [CrossRef]
- Niu, G.; Jin, Z.; Zhang, C.; He, D.; Gao, X.; Zou, C.; Zhang, W.; Ding, J.; Das, B.C.; Severinov, K.; et al. An effective vaginal gel to deliver CRISPR/Cas9 system encapsulated in poly (beta-amino ester) nanoparticles for vaginal gene therapy. EBioMedicine 2020, 58, 102897. [Google Scholar] [CrossRef]
- Kandhasamy, S.; Liang, B.; Yang, D.P.; Zeng, Y. Antibacterial vitamin K3 carnosine peptide-laden silk fibroin electrospun fibers for improvement of skin wound healing in diabetic rats. ACS Appl. Bio. Mater. 2021, 4, 4769–4788. [Google Scholar] [CrossRef]
- Verma, A.; Tiwari, A.; Panda, P.K.; Saraf, S.; Jain, A.; Jain, S.K. Locust bean gum in drug delivery application. In Natural Polysaccharides in Drug Delivery and Biomedical Applications; Academic Press: Cambridge, MA, USA, 2019; pp. 203–222. [Google Scholar]
- Fey, C.; Betz, J.; Rosenbaum, C.; Kralisch, D.; Vielreicher, M.; Friedrich, O.; Metzger, M.; Zdzieblo, D. Bacterial nanocellulose as novel carrier for intestinal epithelial cells in drug delivery studies. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 109, 110613. [Google Scholar] [CrossRef]
- Troy, E.; Tilbury, M.A.; Power, A.M.; Wall, J.G. Nature-based biomaterials and their application in biomedicine. Polymers 2021, 13, 3321. [Google Scholar] [CrossRef]
- Liu, H.; Wang, C.; Li, C.; Qin, Y.; Wang, Z.; Yang, F.; Li, Z.; Wang, J. A functional chitosan-based hydrogel as a wound dressing and drug delivery system in the treatment of wound healing. RSC Adv. 2018, 8, 7533–7549. [Google Scholar] [CrossRef]
- Dubnika, A.; Egle, K.; Skrinda-Melne, M.; Skadins, I.; Rajadas, J.; Salma, I. Development of vancomycin delivery systems based on autologous 3D platelet-rich fibrin matrices for bone tissue engineering. Biomedicines 2021, 9, 814. [Google Scholar] [CrossRef]
- Koyyada, A.; Orsu, P. Natural gum polysaccharides as efficient tissue engineering and drug delivery biopolymers. J. Drug Deliv. Sci. Technol. 2021, 63, 102431. [Google Scholar] [CrossRef]
- Carvalho, T.; Guedes, G.; Sousa, F.L.; Freire, C.S.R.; Santos, H.A. Latest advances on bacterial cellulose-based materials for wound healing, delivery systems, and tissue engineering. Biotechnol. J. 2019, 14, e1900059. [Google Scholar] [CrossRef]
- Miao, T.; Wang, J.; Zeng, Y.; Liu, G.; Chen, X. Polysaccharide-based controlled release systems for therapeutics delivery and tissue engineering: From bench to bedside. Adv. Sci. 2018, 5, 1700513. [Google Scholar] [CrossRef]
- Palumbo, F.S.; Federico, S.; Pitarresi, G.; Fiorica, C.; Giammona, G. Gellan gum-based delivery systems of therapeutic agents and cells. Carbohydr. Polym. 2020, 229, 115430. [Google Scholar] [CrossRef] [PubMed]
- Xue, K.; Lv, S.; Zhu, C. Bringing naturally-occurring saturated fatty acids into biomedical research. J. Mater. Chem. B 2021, 9, 6973–6987. [Google Scholar] [CrossRef] [PubMed]
- Filipczak, N.; Yalamarty, S.S.K.; Li, X.; Khan, M.M.; Parveen, F.; Torchilin, V. Lipid-based drug delivery systems in regenerative medicine. Materials 2021, 14, 5371. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.; Wu, Y.W.; Blyth, C.; Lichtfuss, G.; Goubran, H.; Burnouf, T. Prospective therapeutic applications of platelet extracellular vesicles. Trends Biotechnol. 2021, 39, 598–612. [Google Scholar] [CrossRef] [PubMed]
- Serna, J.A.; Rueda-Gensini, L.; Céspedes-Valenzuela, D.N.; Cifuentes, J.; Cruz, J.C.; Muñoz-Camargo, C. Recent advances on stimuli-responsive hydrogels based on tissue-derived ecms and their components: Towards improving functionality for tissue engineering and controlled drug delivery. Polymers 2021, 13, 3263. [Google Scholar] [CrossRef]
- Khan, M.U.A.; Razaq, S.I.A.; Mehboob, H.; Rehman, S.; Al-Arjan, W.S.; Amin, R. Antibacterial and hemocompatible pH-responsive hydrogel for skin wound healing application: In vitro drug release. Polymers 2021, 13, 3703. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Montclare, J.K. Effect of divalent metal cations on the conformation, elastic behavior, and controlled release of a photocrosslinked protein engineered hydrogel. ACS Appl. Bio. Mater. 2021, 4, 3587–3597. [Google Scholar] [CrossRef]
- Kondiah, P.J.; Kondiah, P.P.D.; Choonara, Y.E.; Marimuthu, T.; Pillay, V. A 3D bioprinted pseudo-bone drug delivery scaffold for bone tissue engineering. Pharmaceutics 2020, 12, 166. [Google Scholar] [CrossRef]
- Hong, D.; Wu, J.; Xiao, X.; Li, X.; Xu, D.; Du, C. Antimicrobial peptides-loaded hydroxyapatite microsphere with different hierarchical structures for enhanced drug loading, sustained release and antibacterial activity. Front. Chem. 2021, 9, 747665. [Google Scholar] [CrossRef]
- Hong, Q.; Song, H.; Lan Chi, N.; Brindhadevi, K. Numerous nanoparticles as drug delivery system to control secondary immune response and promote spinal cord injury regeneration. Process. Biochem. 2022, 112, 145–153. [Google Scholar] [CrossRef]
- Bizeau, J.; Mertz, D. Design and applications of protein delivery systems in nanomedicine and tissue engineering. Adv. Colloid Interface Sci. 2021, 287, 102334. [Google Scholar] [CrossRef]
- Sun, Y.; Nan, D.; Jin, H.; Qu, X. Recent advances of injectable hydrogels for drug delivery and tissue engineering applications. Polymer Testing 2020, 81, 106283. [Google Scholar] [CrossRef]
- Baldwin, A.; Hartl, M.; Tschaikowsky, M.; Balzer, B.N.; Booth, B.W. Degradation and release of tannic acid from an injectable tissue regeneration bead matrix in vivo. J. Biomed. Mater. Res. B Appl. Biomater. 2022, 110, 1165–1177. [Google Scholar] [CrossRef]
- Dong, X.; Lu, X.; Kingston, K.; Brewer, E.; Juliar, B.A.; Kripfgans, O.D.; Fowlkes, J.B.; Franceschi, R.T.; Putnam, A.J.; Liu, Z.; et al. Controlled delivery of basic fibroblast growth factor (bFGF) using acoustic droplet vaporization stimulates endothelial network formation. Acta Biomater. 2019, 97, 409–419. [Google Scholar] [CrossRef]
- Huang, L.; Quesada, C.; Aliabouzar, M.; Fowlkes, J.B.; Franceschi, R.T.; Liu, Z.; Putnam, A.J.; Fabiilli, M.L. Spatially-directed angiogenesis using ultrasound-controlled release of basic fibroblast growth factor from acoustically-responsive scaffolds. Acta Biomater. 2021, 129, 73–83. [Google Scholar] [CrossRef]
- Yang, J.; Shen, M.; Wen, H.; Luo, Y.; Huang, R.; Rong, L.; Xie, J. Recent advance in delivery system and tissue engineering applications of chondroitin sulfate. Carbohydr Polym 2020, 230, 115650. [Google Scholar] [CrossRef]
- Maan, Z.; Masri, N.Z.; Willerth, S.M. Smart bioinks for the printing of human tissue models. Biomolecules 2022, 12, 141. [Google Scholar] [CrossRef]
- Fardous, J.; Yamamoto, E.; Omoso, Y.; Nagao, S.; Inoue, Y.; Yoshida, K.; Ikegami, Y.; Zhang, Y.; Shirakigawa, N.; Ono, F.; et al. Development of a gel-in-oil emulsion as a transdermal drug delivery system for successful delivery of growth factors. J. Biosci. Bioeng. 2021, 132, 95–101. [Google Scholar] [CrossRef]
- Alvear-Jiménez, A.Z.G.I.; Shen, Y.; Villaverde, G.; Lozano-Chamizo, L.; Guardia, P.; Tinoco, M.; Garcia-Pinel, B.; Prados, J.; Melguizo, C.; López-Romero, M.; et al. Electrospraying as a technique for the controlled synthesis of biocompatible PLGA@Ag2S and PLGA@Ag2S@SPION nanocarriers with drug release capability. Pharmaceutics 2022, 14, 214. [Google Scholar] [CrossRef]
- Mohan, T.; Ajdnik, U.; Nagaraj, C.; Lackner, F.; Dobaj Štiglic, A.; Palani, T.; Amornkitbamrung, L.; Gradišnik, L.; Maver, U.; Kargl, R.; et al. One-step fabrication of hollow spherical cellulose beads: Application in pH-responsive therapeutic delivery. ACS Appl. Mater. Interfaces 2022, 14, 3726–3739. [Google Scholar] [CrossRef]
- Xie, M.; Wu, D.; Li, G.; Yang, J.; Zhang, Y.S. Exosomes targeted towards applications in regenerative medicine. Nano Select 2021, 2, 880–908. [Google Scholar] [CrossRef]
- Feng, W.; Li, G.; Kang, X.; Wang, R.; Liu, F.; Zhao, D.; Li, H.; Bu, F.; Yu, Y.; Moriarty, T.F.; et al. Cascade-targeting poly(amino acid) nanoparticles eliminate intracellular bacteria via on-site antibiotic delivery. Adv. Mater. 2022, 34, e2109789. [Google Scholar] [CrossRef]
- Zhou, Y.L.; Yang, Q.Q.; Zhang, L.; Tang, J.B. Nanoparticle-coated sutures providing sustained growth factor delivery to improve the healing strength of injured tendons. Acta Biomater. 2021, 124, 301–314. [Google Scholar] [CrossRef]
- Fahimirad, S.; Abtahi, H.; Satei, P.; Ghaznavi-Rad, E.; Moslehi, M.; Ganji, A. Wound healing performance of PCL/chitosan based electrospun nanofiber electrosprayed with curcumin loaded chitosan nanoparticles. Carbohydr. Polym. 2021, 259, 117640. [Google Scholar] [CrossRef]
- Federico, S.; Pitarresi, G.; Palumbo, F.S.; Fiorica, C.; Catania, V.; Schillaci, D.; Giammona, G. An asymmetric electrospun membrane for the controlled release of ciprofloxacin and FGF-2: Evaluation of antimicrobial and chemoattractant properties. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 123, 112001. [Google Scholar] [CrossRef]
- Alizadehgiashi, M.; Nemr, C.R.; Chekini, M.; Ramos, D.P.; Mittal, N.; Ahmed, S.U.; Khuu, N.; Kelley, S.O.; Kumacheva, E. Multifunctional 3D-printed wound dressings. ACS Nano. 2021, 15, 12375–12387. [Google Scholar] [CrossRef]
- Sun, B.; Lian, M.; Han, Y.; Mo, X.; Jiang, W.; Qiao, Z.; Dai, K. A 3D-Bioprinted dual growth factor-releasing intervertebral disc scaffold induces nucleus pulposus and annulus fibrosus reconstruction. Bioact. Mater. 2021, 6, 179–190. [Google Scholar] [CrossRef]
- Chandrakala, V.; Aruna, V.; Angajala, G. Review on metal nanoparticles as nanocarriers: Current challenges and perspectives in drug delivery systems. Emergent. Mater. 2021, 1–23. [Google Scholar] [CrossRef]
- Li, Y.; Leng, Q.; Pang, X.; Shi, H.; Liu, Y.; Xiao, S.; Zhao, L.; Zhou, P.; Fu, S. Therapeutic effects of EGF-modified curcumin/chitosan nano-spray on wound healing. Regen. Biomater. 2021, 8, rbab009. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Zheng, X.; An, Y.; Wang, K.; Zhang, J.; He, H.; Wu, J. Zwitterionic hydrogel for sustained release of growth factors to enhance wound healing. Biomater. Sci. 2021, 9, 882–891. [Google Scholar] [CrossRef] [PubMed]
- Vijayan, A.; Nanditha, C.K.; Kumar, G.S.V. ECM-mimicking nanofibrous scaffold enriched with dual growth factor carrying nanoparticles for diabetic wound healing. Nanoscale Adv. 2021, 3, 3085. [Google Scholar] [CrossRef]
- Fallah-Darrehchi, M.; Zahedi, P.; Safarian, S.; Ghaffari-Bohlouli, P.; Aeinehvand, R. Conductive conduit based on electrospun poly (L-lactide-co-D, L-lactide) nanofibers containing 4-aminopyridine-loaded molecularly imprinted poly (methacrylic acid) nanoparticles used for peripheral nerve regeneration. Int. J. Biol. Macromol. 2021, 190, 499–507. [Google Scholar] [CrossRef]
- Rathinavel, S.; Indrakumar, J.; Korrapati, P.S.; Dharmalingam, S. Synthesis and fabrication of amine functionalized SBA-15 incorporated PVA/Curcumin nanofiber for skin wound healing application. Colloids Surf. A Physicochem. Eng. Asp. 2022, 637, 128185. [Google Scholar] [CrossRef]
- Bari, E.; Scocozza, F.; Perteghella, S.; Sorlini, M.; Auricchio, F.; Torre, M.L.; Conti, M. 3D bioprinted scaffolds containing mesenchymal stem/stromal lyosecretome: Next generation controlled release device for bone regenerative medicine. Pharmaceutics 2021, 13, 515. [Google Scholar] [CrossRef]
- Kamel, R.; El-Wakil, N.A.; Abdelkhalek, A.A.; Elkasabgy, N. Topical cellulose nanocrystals-stabilized nanoemulgel loaded with ciprofloxacin HCl with enhanced antibacterial activity and tissue regenerative properties. J. Drug Deliv. Sci. Technol. 2021, 64, 102553. [Google Scholar] [CrossRef]
- Shaw, G.S.; Samavedi, S. Potent particle-based vehicles for growth factor delivery from electrospun meshes: Fabrication and functionalization strategies for effective tissue regeneration. ACS Biomater. Sci. Eng. 2022, 8, 1–15. [Google Scholar] [CrossRef]
- Arriagada, F.; Gunther, G.; Morales, J. Nanoantioxidant-based silica particles as flavonoid carrier for drug delivery applications. Pharmaceutics 2020, 12, 302. [Google Scholar] [CrossRef]
- Fuhrmann, G.; Serio, A.; Mazo, M.; Nair, R.; Stevens, M.M. Active loading into extracellular vesicles significantly improves the cellular uptake and photodynamic effect of porphyrins. J. Control. Release 2015, 205, 35–44. [Google Scholar] [CrossRef]
- Li, P.C.; Chen, S.C.; Hsueh, Y.J.; Shen, Y.C.; Tsai, M.Y.; Hsu, L.W.; Yeh, C.K.; Chen, H.C.; Huang, C.C. Gelatin scaffold with multifunctional curcumin-loaded lipid-PLGA hybrid microparticles for regenerating corneal endothelium. Mater. Sci. Eng. C Mate.r Biol. Appl. 2021, 120, 111753. [Google Scholar] [CrossRef]
- Mohammadi, A.; Hosseinipour, M.; Abdolvand, H.; Najafabadi, S.A.A.; Sahraneshin Samani, F. Improvement in bioavailability of curcumin within the castor-oil based polyurethane nanocomposite through its conjugation on the surface of graphene oxide nanosheets. Polym. Adv. Technol. 2021, 33, 1126–1136. [Google Scholar] [CrossRef]
- Ren, T.; Chen, J.; Qi, P.; Xiao, P.; Wang, P. Goserelin/PLGA solid dispersion used to prepare long-acting microspheres with reduced initial release and reduced fluctuation of drug serum concentration in vivo. Int. J. Pharm. 2022, 615, 121474. [Google Scholar] [CrossRef]
- Kim, B.S.; Chun, S.Y.; Lee, J.K.; Lim, H.J.; Bae, J.S.; Chung, H.Y.; Atala, A.; Soker, S.; Yoo, J.J.; Kwon, T.G. Human amniotic fluid stem cell injection therapy for urethral sphincter regeneration in an animal model. BMC Med. 2012, 10, 94. [Google Scholar] [CrossRef]
- Wang, C.; Chen, C.; Guo, M.; Li, B.; Han, F.; Chen, W. Stretchable collagen-coated polyurethane-urea hydrogel seeded with bladder smooth muscle cells for urethral defect repair in a rabbit model. J. Mater. Sci. Mater. Med. 2019, 30, 135. [Google Scholar] [CrossRef]
- Salem, S.A.; Rashidbenam, Z.; Jasman, M.H.; Ho, C.C.K.; Sagap, I.; Singh, R.; Yusof, M.R.; Md Zainuddin, Z.; Haji Idrus, R.B.; Ng, M.H. Incorporation of Smooth Muscle Cells Derived from Human Adipose Stem Cells on Poly(Lactic-co-Glycolic Acid) Scaffold for the Reconstruction of Subtotally Resected Urinary Bladder in Athymic Rats. Tissue Eng. Regen. Med. 2020, 17, 553–563. [Google Scholar] [CrossRef]
- Sittadjody, S.; Criswell, T.; Jackson, J.D.; Atala, A.; Yoo, J.J. Regenerative medicine approaches in bioengineering female reproductive tissues. Reprod. Sci. 2021, 28, 1573–1595. [Google Scholar] [CrossRef]
- Cai, H.; Wu, B.; Li, Y.; Liu, Y.; Shi, L.; Gong, L.; Xia, Y.; Heng, B.C.; Wu, H.; Ouyang, H.; et al. Local delivery of silk-cellulose incorporated with stromal cell-derived factor-1alpha functionally improves the uterus repair. Tissue Eng. Part A 2019, 25, 1514–1526. [Google Scholar] [CrossRef]
- Li, X.; Lv, H.F.; Zhao, R.; Ying, M.F.; Samuriwo, A.T.; Zhao, Y.Z. Recent developments in bio-scaffold materials as delivery strategies for therapeutics for endometrium regeneration. Mater. Today Bio 2021, 11, 100101. [Google Scholar] [CrossRef]
- Chi, Y.; He, P.; Lei, L.; Lan, Y.; Hu, J.; Meng, Y.; Hu, L. Transdermal estrogen gel and oral aspirin combination therapy improves fertility prognosis via the promotion of endometrial receptivity in moderate to severe intrauterine adhesion. Mol. Med. Rep. 2018, 17, 6337–6344. [Google Scholar] [CrossRef]
- Jiang, P.; Tang, X.; Wang, H.; Dai, C.; Su, J.; Zhu, H.; Song, M.; Liu, J.; Nan, Z.; Ru, T.; et al. Collagen-binding basic fibroblast growth factor improves functional remodeling of scarred endometrium in uterine infertile women: A pilot study. Sci. China Life Sci. 2019, 62, 1617–1629. [Google Scholar] [CrossRef]
- Ma, J.; Zhan, H.; Li, W.; Zhang, L.; Yun, F.; Wu, R.; Lin, J.; Li, Y. Recent trends in therapeutic strategies for repairing endometrial tissue in intrauterine adhesion. Biomater. Res. 2021, 25, 40. [Google Scholar] [CrossRef]
- Chen, Y.; Fei, W.; Zhao, Y.; Wang, F.; Zheng, X.; Luan, X.; Zheng, C. Sustained delivery of 17beta-estradiol by human amniotic extracellular matrix (HAECM) scaffold integrated with PLGA microspheres for endometrium regeneration. Drug Deliv. 2020, 27, 1165–1175. [Google Scholar] [CrossRef]
- Yao, Q.; Zheng, Y.W.; Lan, Q.H.; Wang, L.F.; Huang, Z.W.; Chen, R.; Yang, Y.; Xu, H.L.; Kou, L.; Zhao, Y.Z. Aloe/poloxamer hydrogel as an injectable beta-estradiol delivery scaffold with multi-therapeutic effects to promote endometrial regeneration for intrauterine adhesion treatment. Eur. J. Pharm. Sci. 2020, 148, 105316. [Google Scholar] [CrossRef]
- Patel, S.K.; Valicherla, G.R.; Micklo, A.C.; Rohan, L.C. Drug delivery strategies for management of women’s health issues in the upper genital tract. Adv. Drug Deliv. Rev. 2021, 177, 113955. [Google Scholar] [CrossRef]
- Boroumand, S.; Hosseini, S.; Pashandi, Z.; Faridi-Majidi, R.; Salehi, M. Curcumin-loaded nanofibers for targeting endometriosis in the peritoneum of a mouse model. J. Mater. Sci. Mater. Med. 2019, 31, 8. [Google Scholar] [CrossRef]
- Xu, H.L.; Xu, J.; Zhang, S.S.; Zhu, Q.Y.; Jin, B.H.; ZhuGe, D.L.; Shen, B.X.; Wu, X.Q.; Xiao, J.; Zhao, Y.Z. Temperature-sensitive heparin-modified poloxamer hydrogel with affinity to KGF facilitate the morphologic and functional recovery of the injured rat uterus. Drug Deliv. 2017, 24, 867–881. [Google Scholar] [CrossRef]
- Lv, H.; Wu, B.; Song, J.; Wu, W.; Cai, W.; Xu, J. Hydrogel, a novel therapeutic and delivery strategy, in the treatment of intrauterine adhesions. J. Mater. Chem. B 2021, 9, 6536–6552. [Google Scholar] [CrossRef]
- Chang, C.W.; Seibel, A.J.; Avendano, A.; Cortes-Medina, M.G.; Song, J.W. Distinguishing specific CXCL12 isoforms on their angiogenesis and vascular permeability promoting properties. Adv. Healthc. Mater. 2020, 9, e1901399. [Google Scholar] [CrossRef]
- Wenbo, Q.; Lijian, X.; Shuangdan, Z.; Jiahua, Z.; Yanpeng, T.; Xuejun, Q.; Xianghua, H.; Jingkun, Z. Controlled releasing of SDF-1alpha in chitosan-heparin hydrogel for endometrium injury healing in rat model. Int. J. Biol. Macromol. 2020, 143, 163–172. [Google Scholar] [CrossRef]
- Cai, Y.; Wu, F.; Yu, Y.; Liu, Y.; Shao, C.; Gu, H.; Li, M.; Zhao, Y. Porous scaffolds from droplet microfluidics for prevention of intrauterine adhesion. Acta Biomater. 2019, 84, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Sun, H.; Lin, N.; Hou, X.; Wang, J.; Zhou, B.; Xu, P.; Xiao, Z.; Chen, B.; Dai, J.; et al. Regeneration of uterine horns in rats by collagen scaffolds loaded with collagen-binding human basic fibroblast growth factor. Biomaterials 2011, 32, 8172–8181. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Hu, S.; Yang, H.; Li, Z.; Huang, K.; Su, T.; Wang, S.; Cheng, K. Hyaluronic acid hydrogel integrated with mesenchymal stem cell-secretome to treat endometrial injury in a rat model of asherman’s syndrome. Adv. Healthc. Mater. 2019, 8, e1900411. [Google Scholar] [CrossRef] [PubMed]
- Sadri-Ardekani, H.; Atala, A. Regenerative medicine for the treatment of reproductive system disorders: Current and potential options. Adv. Drug Deliv. Rev. 2015, 82–83, 145–152. [Google Scholar] [CrossRef]
- Arjmand, B.; Alaei, S.; Heravani, N.F.; Alavi-Moghadam, S.; Payab, M.; Ebrahimpour, M.; Aghayan, H.R.; Goodarzi, P.; Larijani, B. Regenerative medicine perspectives in polycystic ovary syndrome. Adv. Exp. Med. Biol. 2021, 1341, 125–141. [Google Scholar] [CrossRef]
- Tanaka, A.; Nakamura, H.; Tabata, Y.; Fujimori, Y.; Kumasawa, K.; Kimura, T. Effect of sustained release of basic fibroblast growth factor using biodegradable gelatin hydrogels on frozen-thawed human ovarian tissue in a xenograft model. J. Obstet. Gynaecol. Res. 2018, 44, 1947–1955. [Google Scholar] [CrossRef]
- Shikanov, A.; Zhang, Z.; Xu, M.; Smith, R.M.; Rajan, A.; Woodruff, T.K.; Shea, L.D. Fibrin encapsulation and vascular endothelial growth factor delivery promotes ovarian graft survival in mice. Tissue Eng. Part A 2011, 17, 3095–3104. [Google Scholar] [CrossRef]
- Felder, S.; Masasa, H.; Orenbuch, A.; Levaot, N.; Shachar Goldenberg, M.; Cohen, S. Reconstruction of the ovary microenvironment utilizing macroporous scaffold with affinity-bound growth factors. Biomaterials 2019, 205, 11–22. [Google Scholar] [CrossRef]
- Ding, L.; Yan, G.; Wang, B.; Xu, L.; Gu, Y.; Ru, T.; Cui, X.; Lei, L.; Liu, J.; Sheng, X.; et al. Transplantation of UC-MSCs on collagen scaffold activates follicles in dormant ovaries of POF patients with long history of infertility. Sci. China Life Sci. 2018, 61, 1554–1565. [Google Scholar] [CrossRef]
- Yang, Y.; Lei, L.; Wang, S.; Sheng, X.; Yan, G.; Xu, L.; Liu, J.; Liu, M.; Zhen, X.; Ding, L.; et al. Transplantation of umbilical cord-derived mesenchymal stem cells on a collagen scaffold improves ovarian function in a premature ovarian failure model of mice. In Vitro Cell Dev. Biol. Anim. 2019, 55, 302–311. [Google Scholar] [CrossRef]
- Yoon, H.J.; Lee, Y.J.; Baek, S.; Chung, Y.S.; Kim, D.H.; Lee, J.H.; Shin, Y.C.; Shin, Y.M.; Ryu, C.; Kim, H.S.; et al. Hormone autocrination by vascularized hydrogel delivery of ovary spheroids to rescue ovarian dysfunctions. Sci. Adv. 2021, 7, eabe8873. [Google Scholar] [CrossRef]
- Sittadjody, S.; Enck, K.M.; Wells, A.; Yoo, J.J.; Atala, A.; Saul, J.M.; Opara, E.C. Encapsulation of mesenchymal stem cells in 3D ovarian cell constructs promotes stable and long-term hormone secretion with improved physiological outcomes in a syngeneic rat model. Ann. Biomed. Eng. 2020, 48, 1058–1070. [Google Scholar] [CrossRef]
- Green, L.J.; Zhou, H.; Padmanabhan, V.; Shikanov, A. Adipose-derived stem cells promote survival, growth, and maturation of early-stage murine follicles. Stem Cell Res. Ther. 2019, 10, 102. [Google Scholar] [CrossRef]
- Wang, X.; Wu, D.; Li, W.; Yang, L. Emerging biomaterials for reproductive medicine. Eng. Regen. 2021, 2, 230–245. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, Z.; Hua, C.; Ji, J.; Zhou, Z.; Fang, Y.; Weng, D.; Lu, L.; Pang, Y.; Sun, W. Design, modeling and 3D printing of a personalized cervix tissue implant with protein release function. Biomed. Mater. 2020, 15, 045005. [Google Scholar] [CrossRef]
- Caneparo, C.; Brownell, D.; Chabaud, S.; Bolduc, S. Genitourinary tissue engineering: Reconstruction and research models. Bioengineering 2021, 8, 99. [Google Scholar] [CrossRef]
- Boskey, E.R.; Telsch, K.M.; Whaley, K.J.; Moench, T.R.; Cone, R.A. Acid production by vaginal flora in vitro is consistent with the rate and extent of vaginal acidification. Infect. Immun. 1999, 67, 5170–5175. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, Y.; Chen, J.; Li, L.; Liu, X.; Zhang, L.; Ma, C.; Wang, Y.; Tian, W.; Song, X.; et al. Mesenchymal stem cell-based bioengineered constructs enhance vaginal repair in ovariectomized rhesus monkeys. Biomaterials 2021, 275, 120863. [Google Scholar] [CrossRef]
- Ghanbari, E.; Khazaei, M.; Ghahremani-Nasab, M.; Mehdizadeh, A.; Yousefi, M. Novel therapeutic approaches of tissue engineering in male infertility. Cell Tissue Res. 2020, 380, 31–42. [Google Scholar] [CrossRef]
- Eyni, H.; Ghorbani, S.; Nazari, H.; Hajialyani, M.; Razavi Bazaz, S.; Mohaqiq, M.; Ebrahimi Warkiani, M.; Sutherland, D.S. Advanced bioengineering of male germ stem cells to preserve fertility. J. Tissue Eng. 2021, 12, 20417314211060590. [Google Scholar] [CrossRef]
- Masuku, N.P.; Unuofin, J.O.; Lebelo, S.L. Advances in nanoparticle delivery system for erectile dysfunction: An updated review. Sex. Med. 2021, 9, 100420. [Google Scholar] [CrossRef]
- Afshar, A.; Aliaghaei, A.; Nazarian, H.; Abbaszadeh, H.A.; Naserzadeh, P.; Fathabadi, F.F.; Abdi, S.; Raee, P.; Aghajanpour, F.; Norouzian, M.; et al. Curcumin-loaded iron particle improvement of spermatogenesis in azoospermic mouse induced by long-term scrotal hyperthermia. Reprod. Sci. 2021, 28, 371–380. [Google Scholar] [CrossRef]
- Del Vento, F.; Vermeulen, M.; Ucakar, B.; Poels, J.; des Rieux, A.; Wyns, C. Significant benefits of nanoparticles containing a necrosis inhibitor on mice testicular tissue autografts outcomes. Int. J. Mol. Sci. 2019, 20, 5833. [Google Scholar] [CrossRef]
- Del Vento, F.; Poels, J.; Vermeulen, M.; Ucakar, B.; Giudice, M.G.; Kanbar, M.; des Rieux, A.; Wyns, C. Accelerated and improved vascular maturity after transplantation of testicular tissue in hydrogels supplemented with VEGF- and PDGF-loaded nanoparticles. Int. J. Mol. Sci. 2021, 22, 5779. [Google Scholar] [CrossRef]
- Ghorbani, S.; Eyni, H.; Khosrowpour, Z.; Salari Asl, L.; Shabani, R.; Nazari, H.; Mehdizadeh, M.; Ebrahimi Warkiani, M.; Amjadi, F. Spermatogenesis induction of spermatogonial stem cells using nanofibrous poly(l-lactic acid)/multi-walled carbon nanotube scaffolds and naringenin. Polym. Adv. Technol. 2019, 30, 3011–3025. [Google Scholar] [CrossRef]
- Wu, H.; Tang, W.H.; Zhao, L.M.; Liu, D.F.; Yang, Y.Z.; Zhang, H.T.; Zhang, Z.; Hong, K.; Lin, H.C.; Jiang, H. Nanotechnology-assisted adipose-derived stem cell (ADSC) therapy for erectile dysfunction of cavernous nerve injury: In vivo cell tracking, optimized injection dosage, and functional evaluation. Asian J. Androl. 2018, 20, 442–447. [Google Scholar] [CrossRef]
- Sharma, S.; Mandhani, A.; Bose, S.; Basu, B. Dynamically crosslinked polydimethylsiloxane-based polyurethanes with contact-killing antimicrobial properties as implantable alloplasts for urological reconstruction. Acta Biomater. 2021, 129, 122–137. [Google Scholar] [CrossRef] [PubMed]
- Pederzoli, F.; Joice, G.; Salonia, A.; Bivalacqua, T.J.; Sopko, N.A. Regenerative and engineered options for urethroplasty. Nat. Rev. Urol. 2019, 16, 453–464. [Google Scholar] [CrossRef]
- Ardeshirylajimi, A.; Ghaderian, S.M.; Omrani, M.D.; Moradi, S.L. Biomimetic scaffold containing PVDF nanofibers with sustained TGF-beta release in combination with AT-MSCs for bladder tissue engineering. Gene 2018, 676, 195–201. [Google Scholar] [CrossRef]
- Yan, H.; Zhong, L.; Jiang, Y.; Yang, J.; Deng, J.; Wei, S.; Opara, E.; Atala, A.; Mao, X.; Damaser, M.S.; et al. Controlled release of insulin-like growth factor 1 enhances urethral sphincter function and histological structure in the treatment of female stress urinary incontinence in a rat model. BJU Int. 2018, 121, 301–312. [Google Scholar] [CrossRef]
- Zhang, K.; Fang, X.; Zhu, J.; Yang, R.; Wang, Y.; Zhao, W.; Mo, X.; Fu, Q. Effective reconstruction of functional urethra promoted with ICG-001 delivery using core-shell collagen/poly(llactide-co-caprolactone) [P(LLA-CL)] nanoyarn-based scaffold: A study in dog model. Front. Bioeng. Biotechnol. 2020, 8, 774. [Google Scholar] [CrossRef] [PubMed]
- Vaddi, S.P.; Reddy, V.B.; Abraham, S.J. Buccal epithelium expanded and encapsulated in scaffold-hybrid approach to urethral stricture (BEES-HAUS) procedure: A novel cell therapy-based pilot study. Int. J. Urol. 2019, 26, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhou, S.; Yang, R.; Zou, Q.; Zhang, K.; Tian, Q.; Zhao, W.; Zong, L.; Fu, Q. Bioengineered bladder patches constructed from multilayered adipose-derived stem cell sheets for bladder regeneration. Acta Biomater. 2019, 85, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Duan, M.; Rahman, M.; Yang, M.; Zhao, W.; Zhou, S.; Gao, G.; Fu, Q. Use of bioactive extracellular matrix fragments as a urethral bulking agent to treat stress urinary incontinence. Acta Biomater. 2020, 117, 156–166. [Google Scholar] [CrossRef]
- Wang, B.; Lv, X.; Li, Z.; Zhang, M.; Yao, J.; Sheng, N.; Lu, M.; Wang, H.; Chen, S. Urethra-inspired biomimetic scaffold: A therapeutic strategy to promote angiogenesis for urethral regeneration in a rabbit model. Acta Biomater. 2020, 102, 247–258. [Google Scholar] [CrossRef]
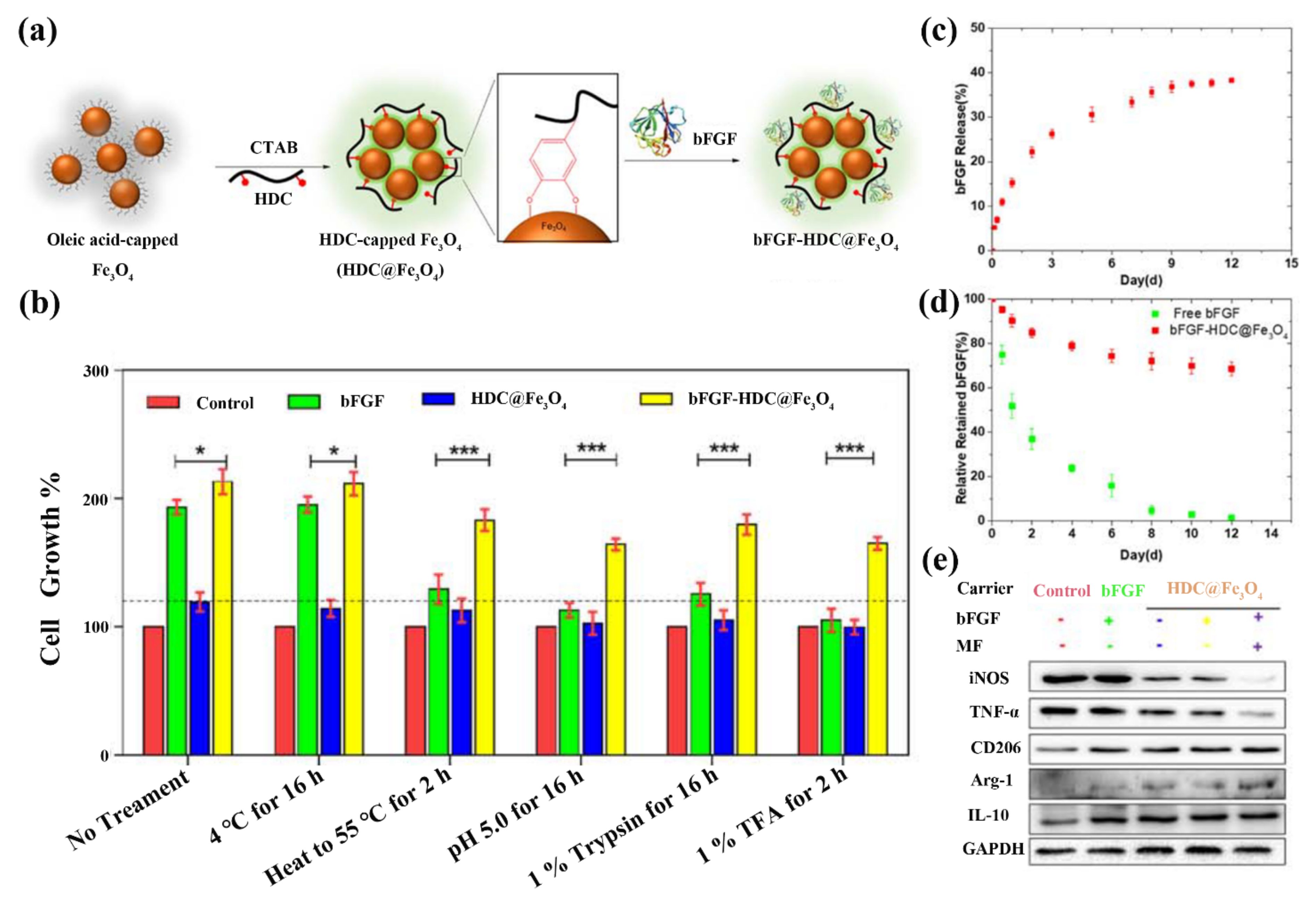
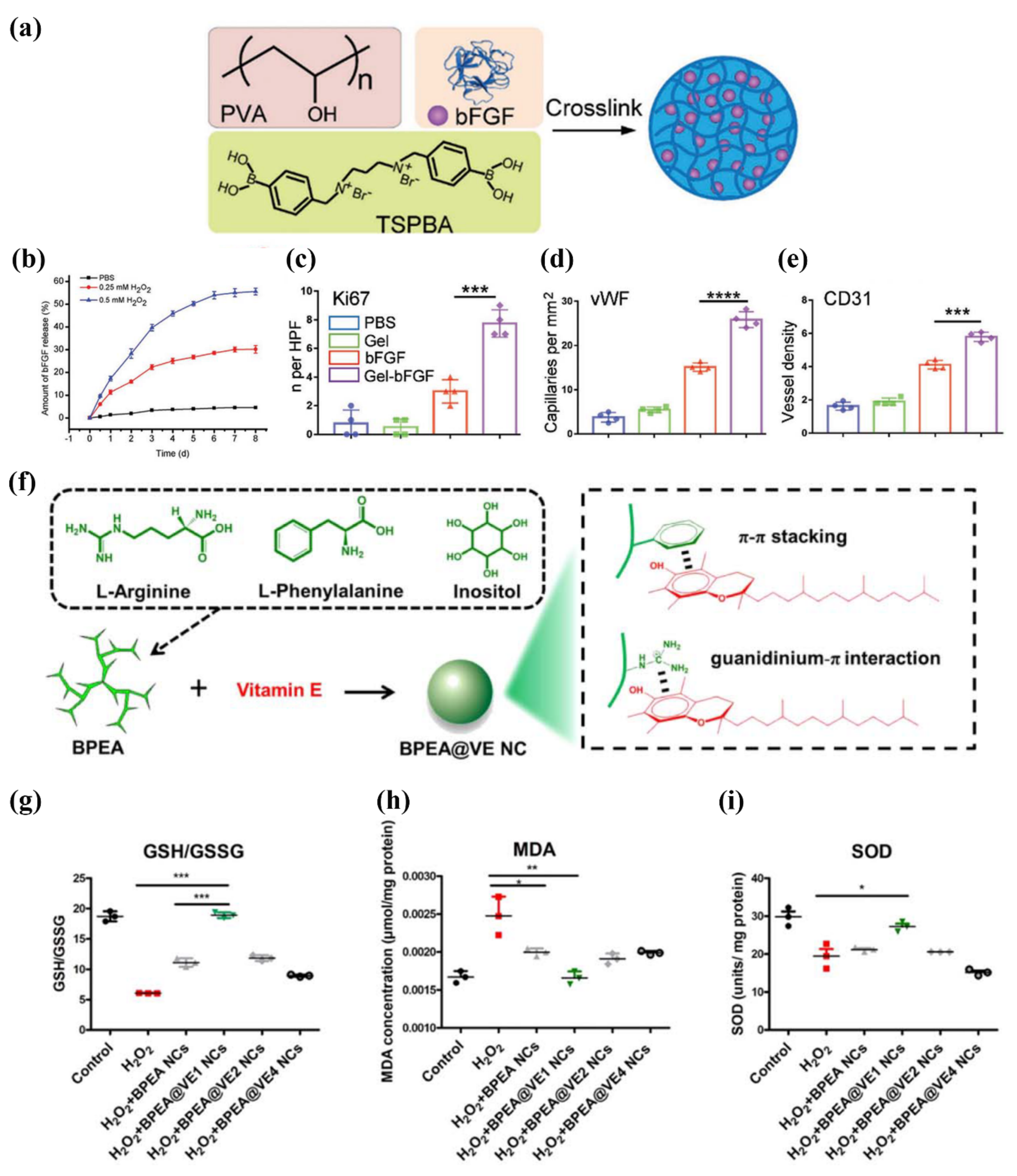

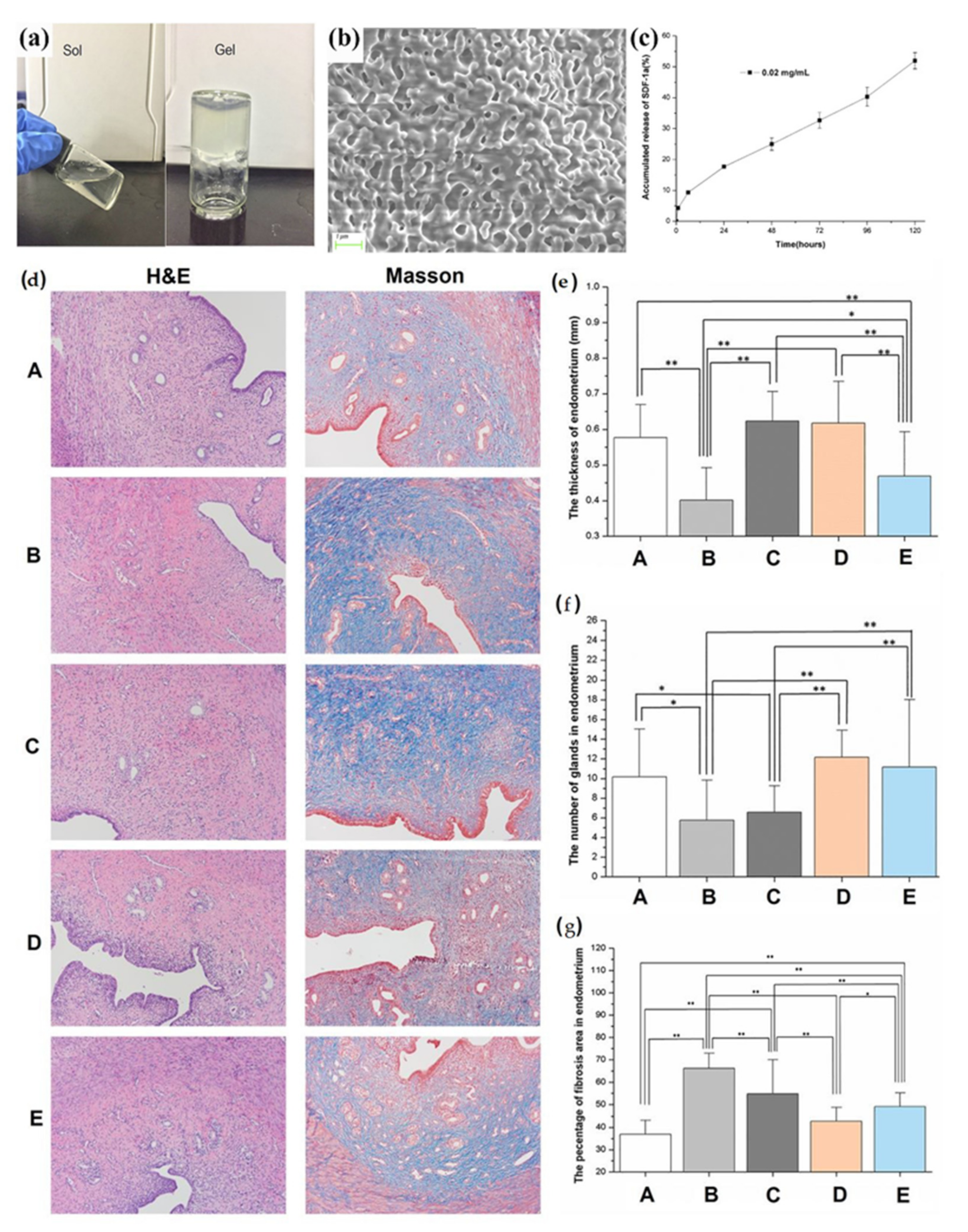
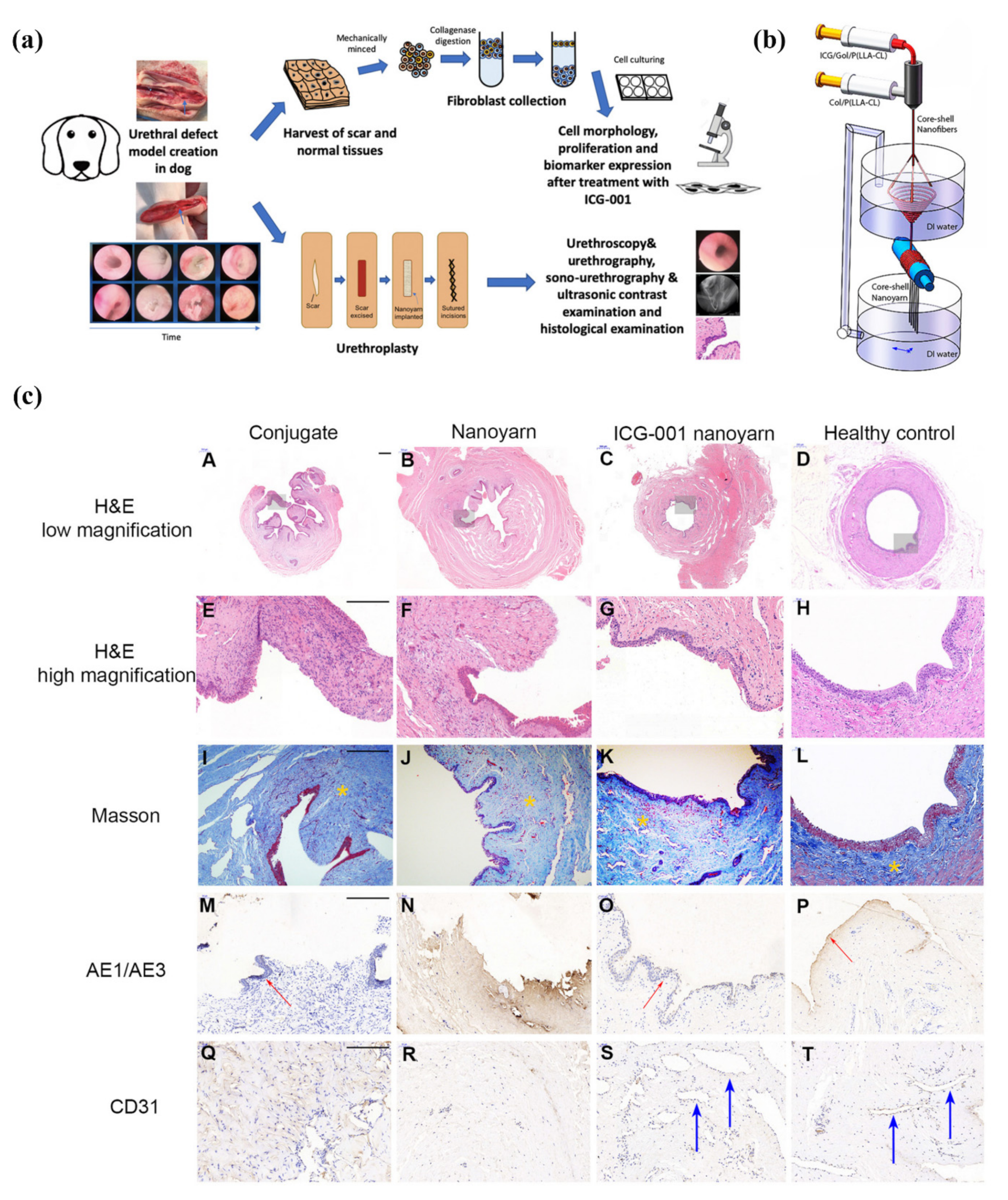
| Loading Strategy. | Bioactive Agent | Delivery System | Release Kinetics | Release Time (Days) | Ref. |
|---|---|---|---|---|---|
| Absorption | FGF-2 | Hydrogel | Sustained release | 3 | [84] |
| Absorption | Heparin | Hydrogel | Sustained release | 14 | [19] |
| Absorption | Curcumin | Hydrogel | Burst then sustained | 1.25 | [26] |
| Adsorption | bFGF and VEGF | Nanoparticle in electrospun nanofiber | Burst then sustained | 14 | [85] |
| Adsorption | 4-Aminopyridine | Electrospun nanofiber | Burst then sustained | 4 | [86] |
| Blending | Curcumin | Electrospun nanofiber | Controlled release | 3.5 | [87] |
| Blending | Ciprofloxacin and FGF-2 | Electrospun membrane | Sustained release | 7 | [79] |
| Blending | AgNPs, VEGF, BSA, and gentamycin | 3D-printed hydrogel | Burst then sustained | 3 | [80] |
| Blending | Secretome | 3D-printed macroporous scaffolds | Sustained release | 10 | [88] |
| Immobilization | CTGF and TGF-β3 | Nanoparticle in 3D-printed scaffold | Controlled release | 35 | [81] |
| Immobilization | bFGF | Nanoparticle | Sustained release | 12 | [25] |
| Immobilization | AT-RvD1, IL-10 | Hydrogel | Sustained release | 5 | [42] |
| Immobilization | bFGF | Hydrogel | Controlled release | 8 | [43] |
| Encapsulation | hGDF-5 | Nano-carrier in hydrogel | Controlled release | 18 | [41] |
| Encapsulation | bFGF | Emulsion in hydrogel | Ultrasound-controlled release | 8 | [68] |
| Encapsulation | Curcumin | Nanoparticle in nanofiber | Sustained release | 16 | [78] |
| Encapsulation | Curcumin | Hydrogel | Controlled release | 0.25 | [61] |
| Entrapment | Curcumin | Nanoparticle in extracellular vesicle | Burst then sustained | 4 | [27] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da, L.-C.; Sun, Y.; Lin, Y.-H.; Chen, S.-Z.; Chen, G.-X.; Zheng, B.-H.; Du, S.-R. Emerging Bioactive Agent Delivery-Based Regenerative Therapies for Lower Genitourinary Tissues. Pharmaceutics 2022, 14, 1718. https://doi.org/10.3390/pharmaceutics14081718
Da L-C, Sun Y, Lin Y-H, Chen S-Z, Chen G-X, Zheng B-H, Du S-R. Emerging Bioactive Agent Delivery-Based Regenerative Therapies for Lower Genitourinary Tissues. Pharmaceutics. 2022; 14(8):1718. https://doi.org/10.3390/pharmaceutics14081718
Chicago/Turabian StyleDa, Lin-Cui, Yan Sun, Yun-Hong Lin, Su-Zhu Chen, Gang-Xin Chen, Bei-Hong Zheng, and Sheng-Rong Du. 2022. "Emerging Bioactive Agent Delivery-Based Regenerative Therapies for Lower Genitourinary Tissues" Pharmaceutics 14, no. 8: 1718. https://doi.org/10.3390/pharmaceutics14081718
APA StyleDa, L.-C., Sun, Y., Lin, Y.-H., Chen, S.-Z., Chen, G.-X., Zheng, B.-H., & Du, S.-R. (2022). Emerging Bioactive Agent Delivery-Based Regenerative Therapies for Lower Genitourinary Tissues. Pharmaceutics, 14(8), 1718. https://doi.org/10.3390/pharmaceutics14081718






