Protein-Based Adjuvants for Vaccines as Immunomodulators of the Innate and Adaptive Immune Response: Current Knowledge, Challenges, and Future Opportunities
Abstract
1. Introduction
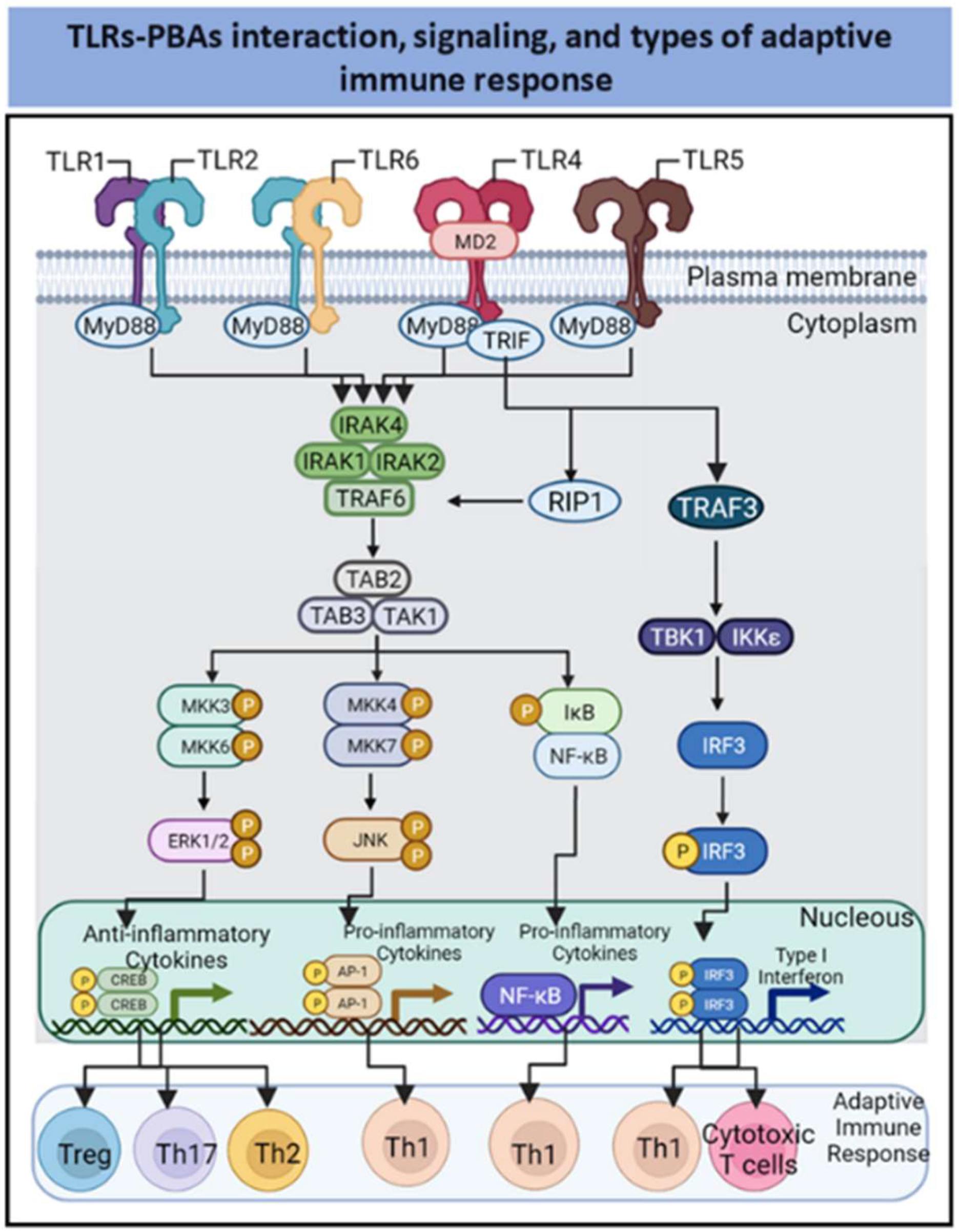
2. PBAs as Agonists of Innate Immune Receptors
2.1. TLR-2-Dependent Activation by PBAs
| Specie | PBA | Innate Immune Response | Adaptive Immune Response | Reference |
|---|---|---|---|---|
| Fusobacterium | FomA Porin | Proinflammatory cytokines and costimulatory cytokines | OVA-Specific antibodies in a preclinical model | [36,37] |
| Shigella flexneri | Major outer membrane protein (MOMP) | Proinflammatory profile | ND | [38] |
| Shigella dysenteriae | Porin | Activation of mitogen-activated protein kinase (MAPK) and nuclear factor B (NF-B). Up-regulation of CD80; MHC class II; and CD40. B-cell activation. | Th1 immune response | [41,42,43] |
| Vibrio cholerae | OmpU | M1 polarization and NF-κB activation | ND | [44] |
| Neisseria meningitidis | PorB | Pro-inflammatory cytokines, activation of APCs | Increases of Follicular Dendritic cells; Th1 immune response; Antigen-Cross presentation | [45,46] |
| Salmonella typhi | OmpC and OmpF | Pro-inflammatory cytokines, activation of APCs | Increase in IgG antibody titers | [47] |
| Mycobacterium tuberculosis | Early secreted antigenic target protein 6 (ESAT-6) | Proinflammatory cytokines | Th17 immune response with a role in protection against M. tuberculosis infection | [48] |
| Brucella abortus | rBCSP31 | Proinflammatory cytokines; priming of CD4+ T-cells | Th1 type immune response protects against B. abortus Infection. | [49] |
| Streptococcus pneumoniae | DnaJ-ΔA146Ply | production of IL-12 in BM-DCs | Th1 Immune Response against Streptococcus pneumoniae | [50] |
| Streptococcus pneumoniae | Endopeptidase O (PepO) | Proinflammatory cytokines | ND | [51] |
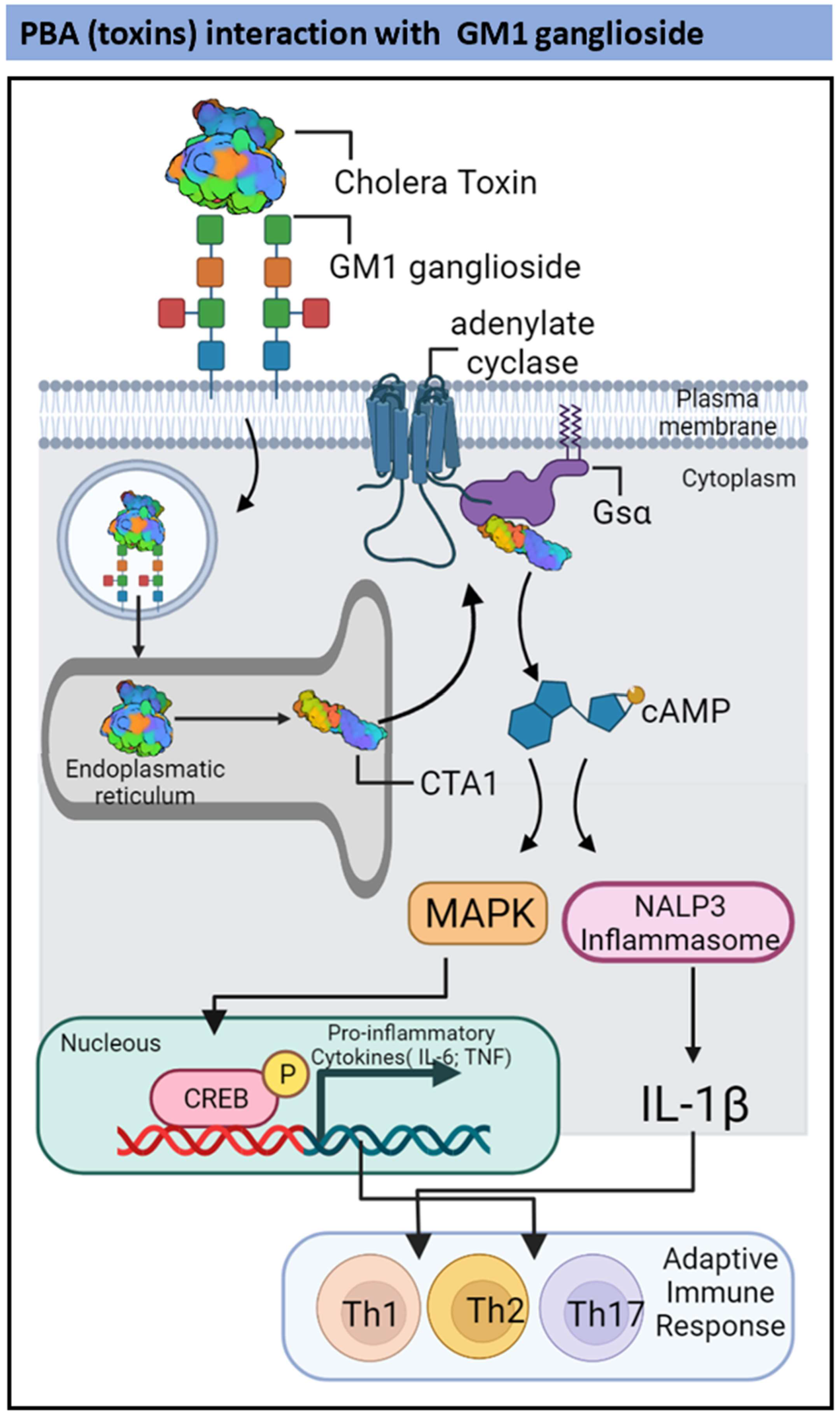
2.2. TLR2-and Ganglioside-Dependent Activation by PBAs
| Specie | PBA | Innate Immune Response | Adaptive Immune Response | Reference |
|---|---|---|---|---|
| Vibrio cholerae | Cholera toxin | Pro-inflammatory cytokines in DCs and CD4 T-cells. | Induction of IL-17-producing CD4+ Th17 cells | [67,68] |
| Escherichia coli | B-pentamers of the type II LTs LT-IIa and LT-IIb | Pro-inflammatory cytokines in humans THP-1 cells and NF-κB | ND | [69,70,71,72] |
2.3. TLR4-Dependent Activation by PBAs
| Specie | PBA | Innate Immune Response | Adaptive Immune Response | Reference |
|---|---|---|---|---|
| Brucella abortus | Omp16 | Induction of proinflammatory cytokines, and APC activation | Th1 immune response against B. abortus infection | [89] |
| Brucella spp. | BLS | Pro-inflammatory cytokines | ND | [90] |
| Mycobacterium tuberculosis | GrpE; RpfE; Rv0652; HBHA | Induction of proinflammatory cytokines, and DCs activation | Th1 immune response | [91,92,93,94] |
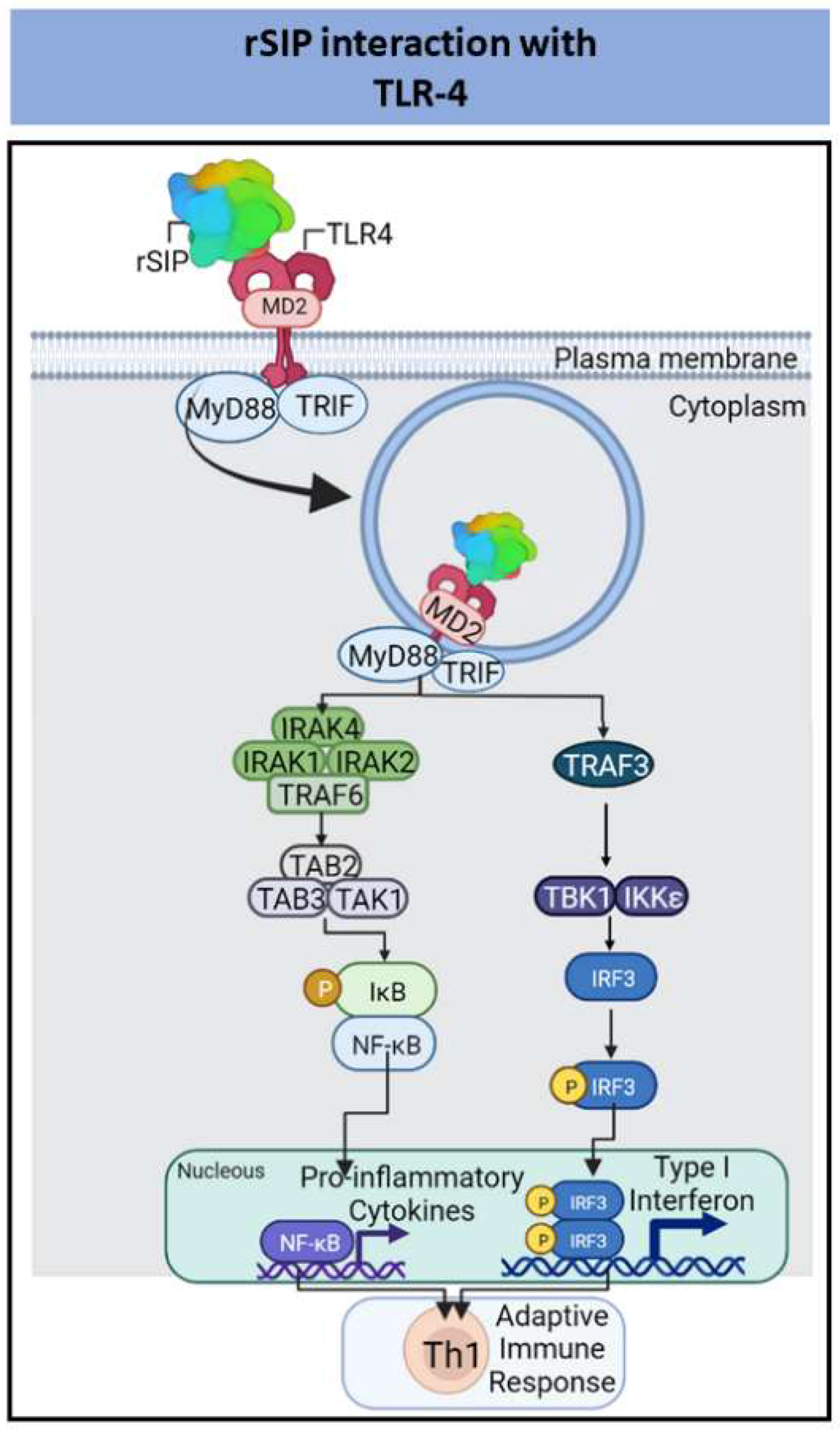
| Streptococcus Group B (GBS) | Recombinant Surface Immunogenic Protein (rSIP) | Pro-inflammatory cytokines | Increase of humoral immune response and protection against GBS | [79,82,83,85] |
| Streptococcus pneumoniae | Pneumolisin | (NF-κB) activation and secretion TNF-α; IL-6) | ND | [49,87] |
| Streptococcus pneumoniae | DnaJ | MAPKs, NF-B and PI3K-Akt activation | Th1 and Th17 activation | [86,88] |
| Mycobacterium avium subsp | MAP CobT | MAP kinases and NF-kB activation | Th1 immune response | [95] |
| Human | HSP70L1 | NF-κB and MAPKs activation. Secretion of proinflammatory cytokines | Th1 immune response | [78] |
| Human | high mobility group box 1 proteins (HMGB1) | (NF-κB) activation and secretion of proinflammatory cytokines (TNF-α; IL-6; IL-1β) | ND | [96,97] |
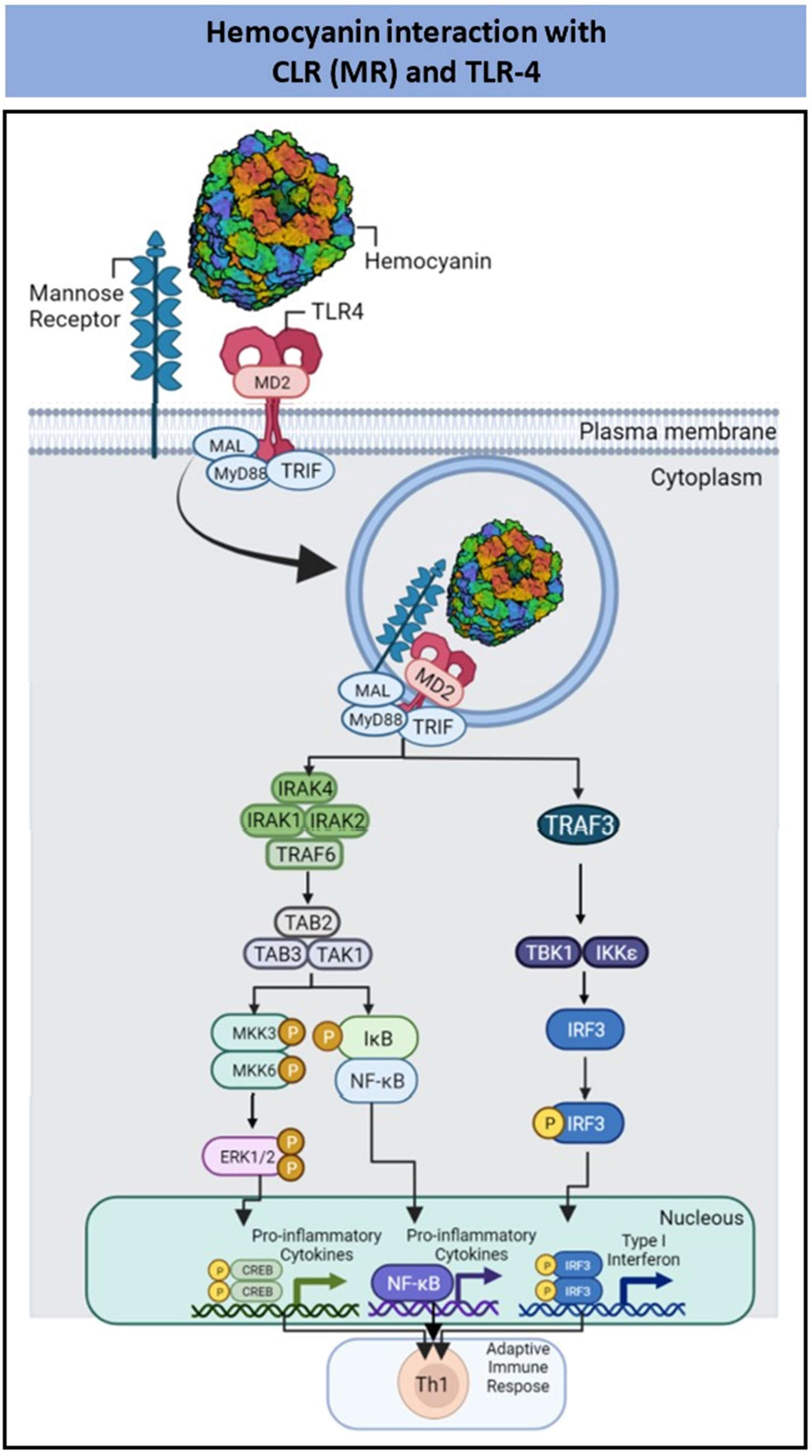
| Mollusk Hemocyanins | FLH; CCH; KLH | Pro-inflammatory cytokines and ERK1/2 phosphorylation | Th1 immune response | [98,99,100,101,102,103,104,105] |
| Plant Lectins | Mistletoe lectin I; Soybean agglutinin; Mistletoe lectin; Jacalin | NF-κB activation | Increase humoral immune response and Th2 immune response | [106,107,108,109,110,111] |
2.4. TLR5-Dependent Activation by PBAs
2.5. Interaction of PBAs with C-Type Lectin Receptors
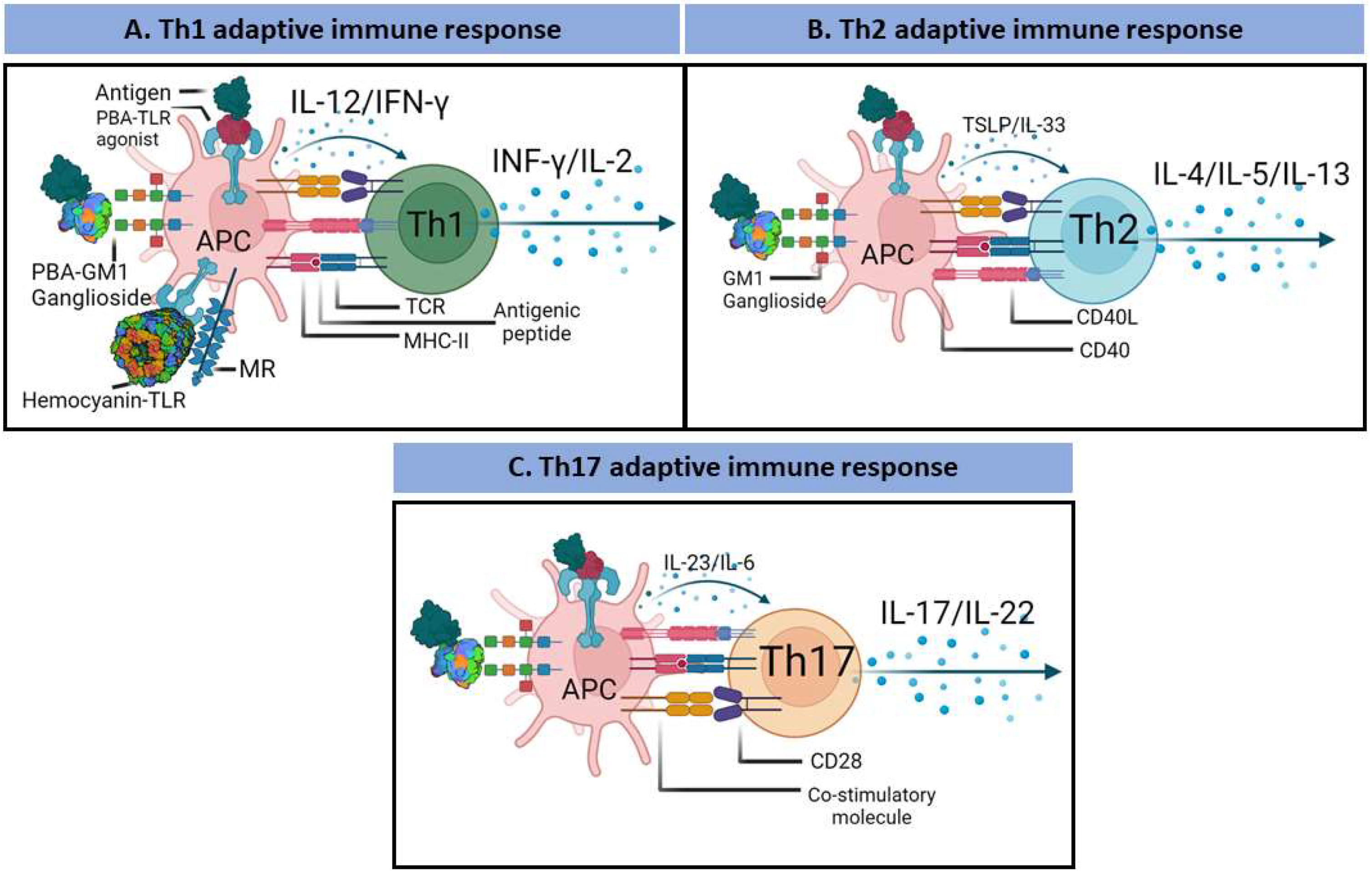
3. PBAs and the Adaptive Immune Response
4. Phase Clinical Studies for Evaluating PBAs as Immunomodulators
4.1. Flagellin
4.2. Toxoid Adjuvants
4.3. Hemocyanin and Antitumor Vaccines
5. Challenges to the Development of Recombinant PBAs
5.1. Recombinant Bacterial PBAs
5.2. Recombinant Hemocyanin
6. Pharmaceutical and Relevant Regulatory Considerations in the Assessment of PBA Safety
6.1. Vaccine Administration System
6.2. Safety of Adjuvants: Regulatory Framework
7. Advantages/Disadvantages of PBAs Compared to Conventional Adjuvants
7.1. Natural Biocompatible and Biodegradable Polymers
7.2. Minimal Reactogenicity and Toxicity
7.3. Binding to Innate Immune Receptors
7.4. Natural and Synthetic Vaccine-Adjuvant Sources
8. Opportunities for PBA Application in Vaccines
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Iwasaki, A.; Omer, S.B. Why and How Vaccines Work. Cell 2020, 183, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Wilson-Welder, J.H.; Torres, M.P.; Kipper, M.J.; Mallapragada, S.K.; Wannemuehler, M.J.; Narasimhan, B. Vaccine Adjuvants: Current Challenges and Future Approaches. J. Pharm. Sci. 2009, 98, 1278–1316. [Google Scholar] [CrossRef] [PubMed]
- Awate, S.; Babiuk, L.A.B.; Mutwiri, G. Mechanisms of Action of Adjuvants. Front. Immunol. 2013, 4, 114. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, F.T.; Di Pasquale, A.; Yarzabal, J.P.; Garçon, N. Safety Assessment of Adjuvanted Vaccines: Methodological Considerations. Hum. Vaccines Immunother. 2015, 11, 1814–1824. [Google Scholar] [CrossRef]
- Carter, D.; Duthie, M.S.; Reed, S.G. Adjuvants. In Vaccination Strategies Against Highly Variable Pathogens; Hangartner, L., Burton, D.R., Eds.; Current Topics in Microbiology and Immunology; Springer International Publishing: Cham, Switzerland, 2018; Volume 428, pp. 103–127. ISBN 978-3-030-58003-2. [Google Scholar]
- Pulendran, B.; Arunachalam, P.S.; O’Hagan, D.T. Emerging Concepts in the Science of Vaccine Adjuvants. Nat. Rev. Drug Discov. 2021, 20, 454–475. [Google Scholar] [CrossRef]
- Hem, S.L.; HogenEsch, H. Relationship between Physical and Chemical Properties of Aluminum-Containing Adjuvants and Immunopotentiation. Expert Rev. Vaccines 2007, 6, 685–698. [Google Scholar] [CrossRef]
- Alving, C.R.; Beck, Z.; Matyas, G.R.; Rao, M. Liposomal Adjuvants for Human Vaccines. Expert Opin. Drug Deliv. 2016, 13, 807–816. [Google Scholar] [CrossRef]
- Sun, B.; Yu, S.; Zhao, D.; Guo, S.; Wang, X.; Zhao, K. Polysaccharides as Vaccine Adjuvants. Vaccine 2018, 36, 5226–5234. [Google Scholar] [CrossRef]
- Bode, C.; Zhao, G.; Steinhagen, F.; Kinjo, T.; Klinman, D.M. CpG DNA as a Vaccine Adjuvant. Expert Rev. Vaccines 2011, 10, 499–511. [Google Scholar] [CrossRef]
- Kumar, S.; Sunagar, R.; Gosselin, E. Bacterial Protein Toll-Like-Receptor Agonists: A Novel Perspective on Vaccine Adjuvants. Front. Immunol. 2019, 10, 1144. [Google Scholar] [CrossRef]
- Hotez, P.J.; Corry, D.B.; Strych, U.; Bottazzi, M.E. COVID-19 Vaccines: Neutralizing Antibodies and the Alum Advantage. Nat. Rev. Immunol. 2020, 20, 399–400. [Google Scholar] [CrossRef]
- Turner, M.D.; Nedjai, B.; Hurst, T.; Pennington, D.J. Cytokines and Chemokines: At the Crossroads of Cell Signalling and Inflammatory Disease. Biochim. Biophys. Acta 2014, 1843, 2563–2582. [Google Scholar] [CrossRef]
- Mir, M.A.; AlBaradie, R.S.; Alharbi, A.R. Regulation of Immune System by Costimulatory Molecules; Nova Science Publishers, Inc.: New York, NY, USA, 2013; ISBN 978-3-659-39067-8. [Google Scholar]
- Hill, A.; Beitelshees, M.; Pfeifer, B.A. Vaccine Delivery and Immune Response Basics. In Vaccine Delivery Technology; Pfeifer, B.A., Hill, A., Eds.; Methods in Molecular Biology; Springer: Berlin/Heidelberg, Germany, 2021; Volume 2183. [Google Scholar] [CrossRef]
- Sánchez-Ramón, S.; Conejero, L.; Netea, M.G.; Sancho, D.; Palomares, Ó.; Subiza, J.L. Trained Immunity-Based Vaccines: A New Paradigm for the Development of Broad-Spectrum Anti-Infectious Formulations. Front. Immunol. 2018, 9, 2936. [Google Scholar] [CrossRef]
- Uematsu, S.; Akira, S. Toll-like Receptors (TLRs) and Innate Immunity. In Handbook of Experimental Pharmacology; Bauer, S., Hartmann, G., Akira, S., Eds.; Springer: Berlin, Germany, 2008; ISBN 978-3-540-72166-6. [Google Scholar]
- Blum, J.S.; Wearsch, P.A.; Cresswell, P. Pathways of Antigen Processing. Annu. Rev. Immunol. 2013, 31, 443–473. [Google Scholar] [CrossRef]
- Nutt, S.L.; Chopin, M. Transcriptional Networks Driving Dendritic Cell Differentiation and Function. Immunity 2020, 52, 942–956. [Google Scholar] [CrossRef]
- Cyster, J.G.; Allen, C.D.C. B Cell Responses: Cell Interaction Dynamics and Decisions. Cell 2019, 177, 524–540. [Google Scholar] [CrossRef]
- Chaplin, D.D. Overview of the immune response. J. Allergy Clin. Immunol. 2010, 125, S3–S23. [Google Scholar] [CrossRef]
- Janeway, C.A. Pillars Article: Approaching the Asymptote? Evolution and Revolution in Immunology. Cold Spring Harb. Symp. Quant. Biol. 1989, 54, 1–13, Erratum in J. Immunol. 1989, 191, 4475–4487. [Google Scholar] [CrossRef]
- Matzinger, P. Tolerance, Danger, and the Extended Family. Annu. Rev. Immunol. 1994, 12, 991–1045. [Google Scholar] [CrossRef]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen Recognition and Innate Immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef]
- Bell, J.K.; Mullen, G.E.D.; Leifer, C.A.; Mazzoni, A.; Davies, D.R.; Segal, D.M. Leucine-Rich Repeats and Pathogen Recognition in Toll-like Receptors. Trends Immunol. 2003, 24, 528–533. [Google Scholar] [CrossRef]
- O’Neill, L.A.J.; Bowie, A.G. The Family of Five: TIR-Domain-Containing Adaptors in Toll-like Receptor Signalling. Nat. Rev. Immunol. 2007, 7, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Honda, K.; Takaoka, A.; Taniguchi, T. Type I Inteferon Gene Induction by the Interferon Regulatory Factor Family of Transcription Factors. Immunity 2006, 25, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Clabbers, M.T.B.; Holmes, S.; Muusse, T.W.; Vajjhala, P.R.; Thygesen, S.J.; Malde, A.K.; Hunter, D.J.B.; Croll, T.I.; Flueckiger, L.; Nanson, J.D.; et al. MyD88 TIR Domain Higher-Order Assembly Interactions Revealed by Microcrystal Electron Diffraction and Serial Femtosecond Crystallography. Nat. Commun. 2021, 12, 2578. [Google Scholar] [CrossRef]
- Steinhagen, F.; Kinjo, T.; Bode, C.; Klinman, D.M. TLR-Based Immune Adjuvants. Vaccine 2011, 29, 3341–3355. [Google Scholar] [CrossRef]
- Kaur, A.; Kaushik, D.; Piplani, S.; Mehta, S.K.; Petrovsky, N.; Salunke, D.B. TLR2 Agonistic Small Molecules: Detailed Structure–Activity Relationship, Applications, and Future Prospects. J. Med. Chem. 2021, 64, 233–278. [Google Scholar] [CrossRef]
- Hancock, R.E. Role of Porins in Outer Membrane Permeability. J. Bacteriol. 1987, 169, 929–933. [Google Scholar] [CrossRef]
- Schirmer, T. General and Specific Porins from Bacterial Outer Membranes. J. Struct. Biol. 1998, 121, 101–109. [Google Scholar] [CrossRef]
- Simonet, V.; Malléa, M.; Fourel, D.; Bolla, J.-M.; Pages, J.-M. Crucial Domains Are Conserved in Enterobacteriaceae Porins. FEMS Microbiol. Lett. 1996, 136, 91–97. [Google Scholar] [CrossRef]
- Nikaido, H. Molecular Basis of Bacterial Outer Membrane Permeability Revisited. Microbiol. Mol. Biol. Rev. 2003, 67, 593–656. [Google Scholar] [CrossRef]
- Faller, M.; Niederweis, M.; Schulz, G.E. The Structure of a Mycobacterial Outer-Membrane Channel. Science 2004, 303, 1189–1192. [Google Scholar] [CrossRef]
- Bolstad, A.I.; Jensen, H.B.; Bakken, V. Taxonomy, Biology, and Periodontal Aspects of Fusobacterium nucleatum. Clin. Microbiol. Rev. 1996, 9, 55–71. [Google Scholar] [CrossRef]
- Toussi, D.N.; Liu, X.; Massari, P. The FomA Porin from Fusobacterium nucleatum Is a Toll-Like Receptor 2 Agonist with Immune Adjuvant Activity. Clin. Vaccine Immunol. 2012, 19, 1093–1101. [Google Scholar] [CrossRef]
- Pore, D.; Mahata, N.; Pal, A.; Chakrabarti, M.K. 34kDa MOMP of Shigella flexneri Promotes TLR2 Mediated Macrophage Activation with the Engagement of NF-κB and p38 MAP Kinase Signaling. Mol. Immunol. 2010, 47, 1739–1746. [Google Scholar] [CrossRef]
- Massari, P.; Toussi, D.N.; Tifrea, D.F.; de la Maza, L.M. Toll-Like Receptor 2-Dependent Activity of Native Major Outer Membrane Protein Proteosomes of Chlamydia trachomatis. Infect. Immun. 2013, 81, 303–310. [Google Scholar] [CrossRef]
- Duan, T.; Du, Y.; Xing, C.; Wang, H.Y.; Wang, R.-F. Toll-Like Receptor Signaling and Its Role in Cell-Mediated Immunity. Front. Immunol. 2022, 13, 812774. [Google Scholar] [CrossRef]
- Biswas, A.; Banerjee, P.; Biswas, T. Porin of Shigella dysenteriae Directly Promotes Toll-like Receptor 2-Mediated CD4+ T Cell Survival and Effector Function. Mol. Immunol. 2009, 46, 3076–3085. [Google Scholar] [CrossRef]
- Biswas, A.; Banerjee, P.; Mukherjee, G.; Biswas, T. Porin of Shigella dysenteriae Activates Mouse Peritoneal Macrophage Through Toll-like Receptors 2 and 6 to Induce Polarized Type I Response. Mol. Immunol. 2007, 44, 812–820. [Google Scholar] [CrossRef]
- Ray, A.; Biswas, T. Porin of Shigella dysenteriae Enhances Toll-like Receptors 2 and 6 of Mouse Peritoneal B-2 Cells and Induces the Expression of Immunoglobulin M, Immunoglobulin G2a and Immunoglobulin A. Immunology 2005, 114, 94–100. [Google Scholar] [CrossRef]
- Khan, J.; Sharma, P.K.; Mukhopadhaya, A. Vibrio cholerae Porin OmpU Mediates M1-Polarization of Macrophages/Monocytes via TLR1/TLR2 Activation. Immunobiology 2015, 220, 1199–1209. [Google Scholar] [CrossRef]
- Massari, P.; Visintin, A.; Gunawardana, J.; Halmen, K.A.; King, C.A.; Golenbock, D.T.; Wetzler, L.M. Meningococcal Porin PorB Binds to TLR2 and Requires TLR1 for Signaling. J. Immunol. 2006, 176, 2373–2380. [Google Scholar] [CrossRef]
- Lisk, C.; Yuen, R.; Kuniholm, J.; Antos, D.; Reiser, M.L.; Wetzler, L.M. Toll-Like Receptor Ligand Based Adjuvant, PorB, Increases Antigen Deposition on Germinal Center Follicular Dendritic Cells While Enhancing the Follicular Dendritic Cells Network. Front. Immunol. 2020, 11, 1254. [Google Scholar] [CrossRef]
- Pérez-Toledo, M.; Pacheco, P.N.V.; Pastelin-Palacios, R.; Gil-Cruz, C.; Perez-Shibayama, C.; Moreno-Eutimio, M.A.; Becker, I.; Pérez-Tapia, S.M.; Arriaga-Pizano, L.; Cunningham, A.F.; et al. Salmonella Typhi Porins OmpC and OmpF Are Potent Adjuvants for T-Dependent and T-Independent Antigens. Front. Immunol. 2017, 8, 230. [Google Scholar] [CrossRef]
- Chatterjee, S.; Dwivedi, V.P.; Singh, Y.; Siddiqui, I.; Sharma, P.; Van Kaer, L.; Chattopadhyay, D.; Das, G. Early Secreted Antigen ESAT-6 of Mycobacterium tuberculosis Promotes Protective T Helper 17 Cell Responses in a Toll-Like Receptor-2-Dependent Manner. PLoS Pathog. 2011, 7, e1002378. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-Y.; Liu, Y.; Gao, X.-X.; Gao, X.; Cai, H. TLR2 and TLR4 Signaling Pathways Are Required for Recombinant Brucella abortus BCSP31-Induced Cytokine Production, Functional Upregulation of Mouse Macrophages, and the Th1 Immune Response In Vivo and In Vitro. Cell. Mol. Immunol. 2014, 11, 477–494. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yuan, T.; Yuan, J.; Su, Y.; Sun, X.; Wu, J.; Zhang, H.; Min, X.; Zhang, X.; Yin, Y. Expression of Toll-Like Receptor 2 by Dendritic Cells Is Essential for the DnaJ-ΔA146Ply-Mediated Th1 Immune Response against Streptococcus pneumoniae. Infect. Immun. 2018, 86, e00651-17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Kang, L.; Yao, H.; He, Y.; Wang, X.; Xu, W.; Song, Z.; Yin, Y.; Zhang, X. Streptococcus pneumoniae Endopeptidase O (PepO) Elicits a Strong Innate Immune Response in Mice via TLR2 and TLR4 Signaling Pathways. Front. Cell. Infect. Microbiol. 2016, 6, 23. [Google Scholar] [CrossRef]
- Gilbert, R.J.C. Pore-Forming Toxins. Cell. Mol. Life Sci. 2002, 59, 832–844. [Google Scholar] [CrossRef]
- Dunne, A.; Mielke, L.A.; Allen, A.C.; Sutton, C.E.; Higgs, R.; Cunningham, C.C.; Higgins, S.C.; Mills, K.H.G. A Novel TLR2 Agonist from Bordetella pertussis is a Potent Adjuvant that Promotes Protective Immunity with an Acellular Pertussis Vaccine. Mucosal Immunol. 2015, 8, 607–617. [Google Scholar] [CrossRef]
- Punturieri, A.; Copper, P.; Polak, T.; Christensen, P.J.; Curtis, J.L. Conserved Nontypeable Haemophilus influenzae-Derived TLR2-Binding Lipopeptides Synergize with IFN-β to Increase Cytokine Production by Resident Murine and Human Alveolar Macrophages. J. Immunol. 2006, 177, 673–680. [Google Scholar] [CrossRef]
- Jin, M.S.; Kim, S.E.; Heo, J.Y.; Lee, M.E.; Kim, H.M.; Paik, S.-G.; Lee, H.; Lee, J.-O. Crystal Structure of the TLR1-TLR2 Heterodimer Induced by Binding of a Tri-Acylated Lipopeptide. Cell 2007, 130, 1071–1082. [Google Scholar] [CrossRef]
- Dallas, W.S.; Falkow, S. Amino Acid Sequence Homology between Cholera Toxin and Escherichia coli Heat-Labile Toxin. Nature 1980, 288, 499–501. [Google Scholar] [CrossRef]
- Sixma, T.K.; Kalk, K.H.; van Zanten, B.A.M.; Dauter, Z.; Kingma, J.; Witholt, B.; Hol, W.G.J. Refined Structure of Escherichia coli Heat-Labile Enterotoxin, a Close Relative of Cholera Toxin. J. Mol. Biol. 1993, 230, 890–918. [Google Scholar] [CrossRef]
- Zhang, R.-G.; Scott, D.L.; Westbrook, M.L.; Nance, S.; Spangler, B.; Shipley, G.G.; Westbrook, E.M. The Three-Dimensional Crystal Structure of Cholera Toxin. J. Mol. Biol. 1995, 251, 563–573. [Google Scholar] [CrossRef]
- Tsai, B.; Rapoport, T.A. Unfolded Cholera Toxin Is Transferred to the ER Membrane and Released from Protein Disulfide Isomerase upon Oxidation by Ero1. J. Cell Biol. 2002, 159, 207–216. [Google Scholar] [CrossRef]
- Sánchez, J.; Holmgren, J. Cholera Toxin Structure, Gene Regulation and Pathophysiological and Immunological Aspects. Cell. Mol. Life Sci. 2008, 65, 1347–1360. [Google Scholar] [CrossRef]
- Craft, J.J.W.; Shen, T.-W.; Brier, L.M.; Briggs, J.M. Biophysical Characteristics of Cholera Toxin and Escherichia coli Heat-Labile Enterotoxin Structure and Chemistry Lead to Differential Toxicity. J. Phys. Chem. B 2015, 119, 1048–1061. [Google Scholar] [CrossRef]
- Lavelle, E.C.; Ward, R.W. Mucosal Vaccines—Fortifying the Frontiers. Nat. Rev. Immunol. 2022, 22, 236–250. [Google Scholar] [CrossRef]
- Anosova, N.G.; Chabot, S.; Shreedhar, V.; Borawski, J.A.; Dickinson, B.L.; Neutra, M.R. Cholera toxin, E. coli Heat-Labile Toxin, and Non-Toxic Derivatives Induce Dendritic Cell Migration into the Follicle-Associated Epithelium of Peyer’s Patches. Mucosal Immunol. 2008, 1, 59–67. [Google Scholar] [CrossRef]
- Pizza, M.; Giuliani, M.M.; Fontana, M.R.; Monaci, E.; Douce, G.; Dougan, G.; Mills, K.H.G.; Rappuoli, R.; del Giudice, G. Mucosal Vaccines: Non Toxic Derivatives of LT and CT as Mucosal Adjuvants. Vaccine 2001, 19, 2534–2541. [Google Scholar] [CrossRef]
- Lycke, N. Recent Progress in Mucosal Vaccine Development: Potential and Limitations. Nat. Rev. Immunol. 2012, 12, 592–605. [Google Scholar] [CrossRef]
- Moore, P.; He, K.; Tsai, B. Establishment of an In Vitro Transport Assay That Reveals Mechanistic Differences in Cytosolic Events Controlling Cholera Toxin and T-Cell Receptor α Retro-Translocation. PLoS ONE 2013, 8, e75801. [Google Scholar] [CrossRef]
- Kim, M.-S.; Yi, E.-J.; Kim, Y.-I.; Kim, S.H.; Jung, Y.-S.; Kim, S.-R.; Iwawaki, T.; Ko, H.-J.; Chang, S.-Y. ERdj5 in Innate Immune Cells Is a Crucial Factor for the Mucosal Adjuvanticity of Cholera Toxin. Front. Immunol. 2019, 10, 1249. [Google Scholar] [CrossRef]
- Datta, S.K.; Sabet, M.; Nguyen, K.P.L.; Valdez, P.A.; Gonzalez-Navajas, J.M.; Islam, S.; Mihajlov, I.; Fierer, J.; Insel, P.A.; Webster, N.J.; et al. Mucosal Adjuvant Activity of Cholera Toxin Requires Th17 Cells and Protects against Inhalation Anthrax. Proc. Natl. Acad. Sci. USA 2010, 107, 10638–10643. [Google Scholar] [CrossRef]
- Connell, T.D. Cholera Toxin, LT-I, LT-IIa and LT-IIb: The Critical Role of Ganglioside Binding in Immunomodulation by Type I and Type II Heat-Labile Enterotoxins. Expert Rev. Vaccines 2007, 6, 821–834. [Google Scholar] [CrossRef]
- Liang, S.; Wang, M.; Triantafilou, K.; Triantafilou, M.; Nawar, H.F.; Russell, M.W.; Connell, T.D.; Hajishengallis, G. The A Subunit of Type IIb Enterotoxin (LT-IIb) Suppresses the Proinflammatory Potential of the B Subunit and Its Ability to Recruit and Interact with TLR2. J. Immunol. 2007, 178, 4811–4819. [Google Scholar] [CrossRef]
- Liang, S.; Hosur, K.B.; Nawar, H.F.; Russell, M.W.; Connell, T.D.; Hajishengallis, G. In Vivo and In Vitro Adjuvant Activities of the B Subunit of Type IIb Heat-Labile Enterotoxin (LT-IIb-B5) from Escherichia coli. Vaccine 2009, 27, 4302–4308. [Google Scholar] [CrossRef]
- Liang, S.; Wang, M.; Tapping, R.I.; Stepensky, V.; Nawar, H.F.; Triantafilou, M.; Triantafilou, K.; Connell, T.D.; Hajishengallis, G. Ganglioside GD1a Is an Essential Coreceptor for Toll-like Receptor 2 Signaling in Response to the B Subunit of Type IIb Enterotoxin. J. Biol. Chem. 2007, 282, 7532–7542. [Google Scholar] [CrossRef]
- Wang, Y.-Q.; Bazin-Lee, H.; Evans, J.T.; Casella, C.R.; Mitchell, T.C. MPL Adjuvant Contains Competitive Antagonists of Human TLR4. Front. Immunol. 2020, 11, 577823. [Google Scholar] [CrossRef] [PubMed]
- Yatim, N.; Cullen, S.; Albert, M.L. Dying Cells Actively Regulate Adaptive Immune Responses. Nat. Rev. Immunol. 2017, 17, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.H.; Woodruff, M.C.; Grigoryan, L.; Maier, B.; Lee, S.H.; Mandal, P.; Cortese, M.; Natrajan, M.S.; Ravindran, R.; Ma, H.; et al. Squalene Emulsion-Based Vaccine Adjuvants Stimulate CD8 T Cell, but Not Antibody Responses, Through a RIPK3-Dependent Pathway. eLife 2020, 9, e52687. [Google Scholar] [CrossRef] [PubMed]
- Marichal, T.; Ohata, K.; Bedoret, D.; Mesnil, C.; Sabatel, C.; Kobiyama, K.; Lekeux, P.; Coban, C.; Akira, S.; Ishii, K.J.; et al. DNA Released from Dying Host Cells Mediates Aluminum Adjuvant Activity. Nat. Med. 2011, 17, 996–1002. [Google Scholar] [CrossRef] [PubMed]
- Rovere-Querini, P.; Capobianco, A.; Scaffidi, P.; Valentinis, B.; Catalanotti, F.; Giazzon, M.; Dumitriu, I.E.; Müller-Knapp, S.; Iannacone, M.; Traversari, C.; et al. HMGB1 is an Endogenous Immune Adjuvant Released by Necrotic Cells. EMBO Rep. 2004, 5, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.; Wu, Y.; Huang, X.; Wang, W.; Ang, B.; Cao, X.; Wan, T. Toll-like Receptor 4 (TLR4) Is Essential for Hsp70-like Protein 1 (HSP70L1) to Activate Dendritic Cells and Induce Th1 Response. J. Biol. Chem. 2011, 286, 30393–30400. [Google Scholar] [CrossRef]
- Diaz-Dinamarca, D.A.; Manzo, R.A.; Soto, D.A.; Avendaño-Valenzuela, M.J.; Bastias, D.N.; Soto, P.I.; Escobar, D.F.; Vasquez-Saez, V.; Carrión, F.; Pizarro-Ortega, M.S.; et al. Surface Immunogenic Protein of Streptococcus Group B Is an Agonist of Toll-Like Receptors 2 and 4 and a Potential Immune Adjuvant. Vaccines 2020, 8, 29. [Google Scholar] [CrossRef]
- Brodeur, B.R.; Boyer, M.; Charlebois, I.; Hamel, J.; Couture, F.; Rioux, C.R.; Martin, D. Identification of Group B Streptococcal Sip Protein, Which Elicits Cross-Protective Immunity. Infect. Immun. 2000, 68, 5610–5618. [Google Scholar] [CrossRef]
- Rioux, S.; Martin, D.; Ackermann, H.-W.; Dumont, J.; Hamel, J.; Brodeur, B.R. Localization of Surface Immunogenic Protein on Group B Streptococcus. Infect. Immun. 2001, 69, 5162–5165. [Google Scholar] [CrossRef]
- Soto, J.A.; Diaz-Dinamarca, D.A.; Soto, D.A.; Barrientos, M.J.; Carrión, F.; Kalergis, A.M.; Vasquez, A.E. Cellular Immune Response Induced by Surface Immunogenic Protein with AbISCO-100 Adjuvant Vaccination Decreases Group B Streptococcus Vaginal Colonization. Mol. Immunol. 2019, 111, 198–204. [Google Scholar] [CrossRef]
- Diaz-Dinamarca, D.A.; Hernandez, C.; Escobar, D.F.; Soto, D.A.; Muñoz, G.A.; Badilla, J.F.; Manzo, R.A.; Carrión, F.; Kalergis, A.M.; Vasquez, A.E. Mucosal Vaccination with Lactococcus lactis—Secreting Surface Immunological Protein Induces Humoral and Cellular Immune Protection against Group B Streptococcus in a Murine Model. Vaccines 2020, 8, 146. [Google Scholar] [CrossRef]
- Díaz-Dinamarca, D.A.; Jerias, J.I.; Soto, D.A.; Soto, J.A.; Díaz, N.V.; Leyton, Y.Y.; Villegas, R.A.; Kalergis, A.M.; Vásquez, A.E. The Optimisation of the Expression of Recombinant Surface Immunogenic Protein of Group B Streptococcus in Escherichia coli by Response Surface Methodology Improves Humoral Immunity. Mol. Biotechnol. 2018, 60, 215–225. [Google Scholar] [CrossRef]
- Diaz-Dinamarca, D.A.; Soto, D.A.; Leyton, Y.Y.; Altamirano-Lagos, M.J.; Avendaño, M.J.; Kalergis, A.M.; Vasquez, A.E. Oral Vaccine Based on a Surface Immunogenic Protein Mixed with Alum Promotes a Decrease in Streptococcus agalactiae Vaginal Colonization in a Mouse Model. Mol. Immunol. 2018, 103, 63–70. [Google Scholar] [CrossRef]
- Wu, Y.; Cui, J.; Zhang, X.; Gao, S.; Ma, F.; Yao, H.; Sun, X.; He, Y.; Yin, Y.; Xu, W. Pneumococcal DnaJ Modulates Dendritic Cell-Mediated Th1 and Th17 Immune Responses Through Toll-like Receptor 4 Signaling Pathway. Immunobiology 2017, 222, 384–393. [Google Scholar] [CrossRef]
- Malley, R.; Henneke, P.; Morse, S.C.; Cieslewicz, M.J.; Lipsitch, M.; Thompson, C.M.; Kurt-Jones, E.; Paton, J.C.; Wessels, M.R.; Golenbock, D.T. Recognition of Pneumolysin by Toll-like Receptor 4 Confers Resistance to Pneumococcal Infection. Proc. Natl. Acad. Sci. USA 2003, 100, 1966–1971. [Google Scholar] [CrossRef]
- Su, Y.; Li, D.; Xing, Y.; Wang, H.; Wang, J.; Yuan, J.; Wang, X.; Cui, F.; Yin, Y.; Zhang, X. Subcutaneous Immunization with Fusion Protein DnaJ-ΔA146Ply without Additional Adjuvants Induces Both Humoral and Cellular Immunity against Pneumococcal Infection Partially Depending on TLR4. Front. Immunol. 2017, 8, 686. [Google Scholar] [CrossRef]
- Pasquevich, K.A.; García Samartino, C.; Coria, L.M.; Estein, S.M.; Zwerdling, A.; Ibañez, A.E.; Barrionuevo, P.; de Oliveira, F.S.; Carvalho, N.B.; Borkowski, J.; et al. The Protein Moiety of Brucella abortus Outer Membrane Protein 16 Is a New Bacterial Pathogen-Associated Molecular Pattern That Activates Dendritic Cells In Vivo, Induces a Th1 Immune Response, and Is a Promising Self-Adjuvanting Vaccine against Systemic and Oral Acquired Brucellosis. J. Immunol. 2010, 184, 5200–5212. [Google Scholar] [CrossRef]
- Berguer, P.M.; Mundiñano, J.; Piazzon, I.; Goldbaum, F.A. A Polymeric Bacterial Protein Activates Dendritic Cells via TLR4. J. Immunol. 2006, 176, 2366–2372. [Google Scholar] [CrossRef]
- Choi, H.-G.; Kim, W.S.; Back, Y.W.; Kim, H.; Kwon, K.W.; Kim, J.-S.; Shin, S.J.; Kim, H.-J. Mycobacterium tuberculosis RpfE Promotes Simultaneous Th1- and Th17-Type T-Cell Immunity via TLR4-Dependent Maturation of Dendritic Cells: Cellular Immune Response. Eur. J. Immunol. 2015, 45, 1957–1971. [Google Scholar] [CrossRef]
- Jung, I.D.; Jeong, S.K.; Lee, C.-M.; Noh, K.T.; Heo, D.R.; Shin, Y.K.; Yun, C.-H.; Koh, W.-J.; Akira, S.; Whang, J.; et al. Enhanced Efficacy of Therapeutic Cancer Vaccines Produced by Co-Treatment with Mycobacterium tuberculosis Heparin-Binding Hemagglutinin, a Novel TLR4 Agonist. Cancer Res. 2011, 71, 2858–2870. [Google Scholar] [CrossRef]
- Lee, S.J.; Shin, S.J.; Lee, M.H.; Lee, M.-G.; Kang, T.H.; Park, W.S.; Soh, B.Y.; Park, J.H.; Shin, Y.K.; Kim, H.W.; et al. A Potential Protein Adjuvant Derived from Mycobacterium tuberculosis Rv0652 Enhances Dendritic Cells-Based Tumor Immunotherapy. PLoS ONE 2014, 9, e104351. [Google Scholar] [CrossRef]
- Kim, W.S.; Jung, I.D.; Kim, J.-S.; Kim, H.M.; Kwon, K.W.; Park, Y.-M.; Shin, S.J. Mycobacterium tuberculosis GrpE, A Heat-Shock Stress Responsive Chaperone, Promotes Th1-Biased T Cell Immune Response via TLR4-Mediated Activation of Dendritic Cells. Front. Cell. Infect. Microbiol. 2018, 8, 95. [Google Scholar] [CrossRef]
- Byun, E.-H.; Kim, W.S.; Kim, J.-S.; Won, C.-J.; Choi, H.-G.; Kim, H.-J.; Cho, S.-N.; Lee, K.; Zhang, T.; Hur, G.M.; et al. Mycobacterium paratuberculosis CobT Activates Dendritic Cells via Engagement of Toll-like Receptor 4 Resulting in Th1 Cell Expansion. J. Biol. Chem. 2012, 287, 38609–38624. [Google Scholar] [CrossRef]
- He, M.; Bianchi, M.E.; Coleman, T.R.; Tracey, K.J.; Al-Abed, Y. Exploring the Biological Functional Mechanism of the HMGB1/TLR4/MD-2 Complex by Surface Plasmon Resonance. Mol. Med. 2018, 24, 21. [Google Scholar] [CrossRef]
- Yang, H.; Tracey, K.J. Targeting HMGB1 in inflammation. Biochim. Biophys. Acta 2010, 1799, 149–156. [Google Scholar] [CrossRef]
- Salazar, M.L.; Jimènez, J.M.; Villar, J.; Rivera, M.; Báez, M.; Manubens, A.; Becker, M.I. N-Glycosylation of Mollusk hemocyanins Contributes to Their Structural Stability and Immunomodulatory Properties in Mammals. J. Biol. Chem. 2019, 294, 19546–19564. [Google Scholar] [CrossRef]
- Jiménez, J.M.; Salazar, M.L.; Arancibia, S.; Villar, J.; Salazar, F.; Brown, G.D.; Lavelle, E.C.; Martínez-Pomares, L.; Ortiz-Quintero, J.; Lavandero, S.; et al. TLR4, but Neither Dectin-1 nor Dectin-2, Participates in the Mollusk Hemocyanin-Induced Proinflammatory Effects in Antigen-Presenting Cells from Mammals. Front. Immunol. 2019, 10, 1136. [Google Scholar] [CrossRef]
- Becker, M.I.; Arancibia, S.; Salazar, F.; del Campo, M.; de Ioannes, A. Mollusk Hemocyanins as Natural Immunostimulants in Biomedical Applications. In Immune Response Activation; Duc, G.H.T., Ed.; InTech: Rijeka, Croatia, 2014; ISBN 978-953-51-1374. [Google Scholar]
- Arancibia, S.; Espinoza, C.; Salazar, F.; del Campo, M.; Tampe, R.; Zhong, T.-Y.; de Ioannes, P.; Moltedo, B.; Ferreira, J.; Lavelle, E.; et al. A Novel Immunomodulatory Hemocyanin from the Limpet Fissurella latimarginata Promotes Potent Anti-Tumor Activity in Melanoma. PLoS ONE 2014, 9, e87240. [Google Scholar] [CrossRef]
- De Ioannes, P.; Moltedo, B.; Oliva, H.; Pacheco, R.; Faunes, F.; De Ioannes, A.E.; Becker, M.I. Hemocyanin of the Molluscan Concholepas concholepas Exhibits an Unusual Heterodecameric Array of Subunits. J. Biol. Chem. 2004, 279, 26134–26142. [Google Scholar] [CrossRef] [PubMed]
- Becker, M.I.; Fuentes, A.; Del Campo, M.; Manubens, A.; Nova, E.; Oliva, H.; Faunes, F.; Valenzuela, M.A.; Campos-Vallette, M.; Aliaga, A.; et al. Immunodominant Role of CCHA Subunit of Concholepas Hemocyanin is Associated with Unique Biochemical Properties. Int. Immunopharmacol. 2009, 9, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Villar, J.; Salazar, M.L.; Jiménez, J.M.; del Campo, M.; Manubens, A.; Gleisner, M.A.; Ávalos, I.; Salazar-Onfray, F.; Salazar, F.; Mitchell, D.A.; et al. C-type Lectin Receptors MR and DC-SIGN Are Involved in Recognition of Hemocyanins, Shaping Their Immunostimulatory Effects on Human Dendritic Cells. Eur. J. Immunol. 2021, 51, 1715–1731. [Google Scholar] [CrossRef] [PubMed]
- Slovin, S.F.; Ragupathi, G.; Fernandez, C.; Jefferson, M.P.; Diani, M.; Wilton, A.S.; Powell, S.; Spassova, M.; Reis, C.; Clausen, H.; et al. A Bivalent Conjugate Vaccine in the Treatment of Biochemically Relapsed Prostate Cancer: A Study of Glycosylated MUC-2-KLH and Globo H-KLH Conjugate Vaccines Given with the New Semi-Synthetic Saponin Immunological Adjuvant GPI-0100 OR QS-21. Vaccine 2005, 23, 3114–3122. [Google Scholar] [CrossRef]
- Unitt, J.; Hornigold, D. Plant Lectins are Novel Toll-like Receptor Agonists. Biochem. Pharmacol. 2011, 81, 1324–1328. [Google Scholar] [CrossRef]
- Reyna-Margarita, H.-R.; Irais, C.-M.; Mario-Alberto, R.-G.; Agustina, R.-M.; Luis-Benjamín, S.-G.; David, P.-E. Plant Phenolics and Lectins as Vaccine Adjuvants. Curr. Pharm. Biotechnol. 2019, 20, 1236–1243. [Google Scholar] [CrossRef]
- Schötterl, S.; Miemietz, J.T.; Ilina, E.I.; Wirsik, N.M.; Ehrlich, I.; Gall, A.; Huber, S.M.; Lentzen, H.; Mittelbronn, M.; Naumann, U. Mistletoe-Based Drugs Work in Synergy with Radio-Chemotherapy in the Treatment of Glioma In Vitro and In Vivo in Glioblastoma Bearing Mice. Evid. Based Complement. Altern. Med. 2019, 2019, 1376140. [Google Scholar] [CrossRef]
- Lavelle, E.C.; Grant, G.; Pusztai, A.; Pfüller, U.; Leavy, O.; McNeela, E.; Mills, K.H.G.; O’Hagan, D.T. Mistletoe Lectins Enhance Immune Responses to Intranasally Co-Administered Herpes Simplex Virus Glycoprotein D2. Immunology 2002, 107, 268–274. [Google Scholar] [CrossRef]
- Lavelle, E.; Grant, G.; Pfüller, U.; O’Hagan, D. Immunological Implications of the Use of Plant Lectins for Drug and Vaccine Targeting to the Gastrointestinal Tract. J. Drug Target. 2004, 12, 89–95. [Google Scholar] [CrossRef]
- Sander, V.A.; Corigliano, M.G.; Clemente, M. Promising Plant-Derived Adjuvants in the Development of Coccidial Vaccines. Front. Vet. Sci. 2019, 6, 20. [Google Scholar] [CrossRef]
- Velkova, L.; Dimitrov, I.; Schwarz, H.; Stevanovic, S.; Voelter, W.; Salvato, B.; Dolashka-Angelova, P. Structure of Hemocyanin from garden snail Helix lucorum. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2010, 157, 16–25. [Google Scholar] [CrossRef]
- Dolashka, P.; Genov, N.; Parvanova, K.; Voelter, W.; Geiger, M.; Stoeva, S. Rapana thomasiana grosse (gastropoda) Haemocyanin: Spectroscopic Studies of the Structure in Solution and the Conformational Stability of the Native Protein and Its Structural Subunits. Biochem. J. 1996, 315, 139–144. [Google Scholar] [CrossRef]
- Harris, J.R.; Scheffler, D.; Gebauer, W.; Lehnert, R.; Markl, J. Haliotis tuberculata hemocyanin (HtH): Analysis of Oligomeric Stability of HtH1 and HtH2, and Comparison with Keyhole Limpet Hemocyanin KLH1 and KLH2. Micron 2000, 31, 613–622. [Google Scholar] [CrossRef]
- Harris, J.R.; Markl, J. Keyhole Limpet Hemocyanin (KLH): A Biomedical Review. Micron 1999, 30, 597–623. [Google Scholar] [CrossRef]
- Gatsogiannis, C.; Markl, J. Keyhole Limpet Hemocyanin: 9-Å CryoEM Structure and Molecular Model of the KLH1 Didecamer Reveal the Interfaces and Intricate Topology of the 160 Functional Units. J. Mol. Biol. 2009, 385, 963–983. [Google Scholar] [CrossRef]
- Kato, S.; Matsui, T.; Tanaka, Y. Molluscan Hemocyanins. In Vertebrate and Invertebrate Respiratory Proteins, Lipoproteins and other Body Fluid Proteins; Hoeger, U., Harris, J.R., Eds.; Subcellular Biochemistry; Springer International Publishing: Cham, Switzerland, 2020; Volume 94, pp. 195–218. ISBN 978-3-030-41768-0. [Google Scholar]
- Markl, J. Evolution of Molluscan Hemocyanin Structures. Biochim. Biophys. Acta Proteins Proteom. 2013, 1834, 1840–1852. [Google Scholar] [CrossRef]
- Kato, S.; Matsui, T.; Gatsogiannis, C.; Tanaka, Y. Molluscan Hemocyanin: Structure, Evolution, and Physiology. Biophys. Rev. 2018, 10, 191–202. [Google Scholar] [CrossRef]
- Geyer, H.; Wuhrer, M.; Resemann, A.; Geyer, R. Identification and Characterization of Keyhole Limpet Hemocyanin N-Glycans Mediating Cross-Reactivity with Schistosoma mansoni. J. Biol. Chem. 2005, 280, 40731–40748. [Google Scholar] [CrossRef] [PubMed]
- Velkova, L.; Dolashka, P.; van Beeumen, J.; Devreese, B. N-Glycan Structures of β-HlH Subunit of Helix lucorum hemocyanin. Carbohydr. Res. 2017, 449, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gielens, C.; Idakieva, K.; van den Bergh, V.; Siddiqui, N.I.; Parvanova, K.; Compernolle, F. Mass Spectral Evidence for N-Glycans with Branching on Fucose in a Molluscan Hemocyanin. Biochem. Biophys. Res. Commun. 2005, 331, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, N.I.; Idakieva, K.; Demarsin, B.; Doumanova, L.; Compernolle, F.; Gielens, C. Involvement of Glycan Chains in the Antigenicity of Rapana thomasiana Hemocyanin. Biochem. Biophys. Res. Commun. 2007, 361, 705–711. [Google Scholar] [CrossRef]
- Albuquerque, D.A.; A Martins, G.; Campos-Neto, A.; Silva, J.S. The Adjuvant Effect of Jacalin on the Mouse Humoral Immune Response to Trinitrophenyl and Trypanosoma cruzi. Immunol. Lett. 1999, 68, 375–381. [Google Scholar] [CrossRef]
- Yoon, T.J.; Yoo, Y.C.; Kang, T.B.; Her, E.; Kim, S.-H.; Kim, K.; Azuma, I.; Kim, J.B. Cellular and Humoral Adjuvant Activity of Lectins Isolated from Korean Mistletoe (Viscum album colaratum). Int. Immunopharmacol. 2001, 1, 881–889. [Google Scholar] [CrossRef]
- Ricci-Azevedo, R.; Roque-Barreira, M.-C.; Gay, N.J. Targeting and Recognition of Toll-Like Receptors by Plant and Pathogen Lectins. Front. Immunol. 2017, 8, 1820. [Google Scholar] [CrossRef]
- Cardoso, M.R.D.; Mota, C.M.; Ribeiro, D.P.; Santiago, F.M.; Carvalho, J.V.; Araujo, E.C.B.; Silva, N.M.; Mineo, T.W.P.; Roque-Barreira, M.C.; Mineo, J.R.; et al. ArtinM, a d-mannose-Binding Lectin from Artocarpus integrifolia, Plays a Potent Adjuvant and Immunostimulatory Role in Immunization against Neospora caninum. Vaccine 2011, 29, 9183–9193. [Google Scholar] [CrossRef]
- Andersen-Nissen, E.; Smith, K.D.; Strobe, K.L.; Barrett, S.L.R.; Cookson, B.T.; Logan, S.M.; Aderem, A. Evasion of Toll-like receptor 5 by flagellated bacteria. Proc. Natl. Acad. Sci. USA 2005, 102, 9247–9252. [Google Scholar] [CrossRef]
- Hayashi, F.; Smith, K.D.; Ozinsky, A.; Hawn, T.R.; Yi, E.C.; Goodlett, D.R.; Eng, J.K.; Akira, S.; Underhill, D.; Aderem, A. The Innate Immune Response to Bacterial Flagellin Is Mediated by Toll-like Receptor 5. Nature 2001, 410, 1099–1103. [Google Scholar] [CrossRef]
- Imada, K. Bacterial Flagellar Axial Structure and Its Construction. Biophys. Rev. 2018, 10, 559–570. [Google Scholar] [CrossRef]
- Mimori-Kiyosue, Y.; Yamashita, I.; Fujiyoshi, Y.; Yamaguchi, S.; Namba, K. Role of the Outermost Subdomain of Salmonella flagellin in the Filament Structure Revealed by Electron Cryomicroscopy. J. Mol. Biol. 1997, 284, 521–530. [Google Scholar] [CrossRef]
- Samatey, F.A.; Imada, K.; Nagashima, S.; Vonderviszt, F.; Kumasaka, T.; Yamamoto, M.; Namba, K. Structure of the Bacterial Flagellar Protofilament and Implications for a Switch for Supercoiling. Nature 2001, 410, 331–337. [Google Scholar] [CrossRef]
- Murthy, K.G.; Deb, A.; Goonesekera, S.; Szabó, C.; Salzman, A.L. Identification of Conserved Domains in Salmonella muenchen Flagellin That Are Essential for Its Ability to Activate TLR5 and to Induce an Inflammatory Response in Vitro. J. Biol. Chem. 2004, 279, 5667–5675. [Google Scholar] [CrossRef]
- Donnelly, M.A.; Steiner, T.S. Two Nonadjacent Regions in Enteroaggregative Escherichia coli Flagellin Are Required for Activation of Toll-like Receptor 5. J. Biol. Chem. 2002, 277, 40456–40461. [Google Scholar] [CrossRef]
- Eaves-Pyles, T.D.; Wong, H.R.; Odoms, K.; Pyles, R.B. Salmonella Flagellin-Dependent Proinflammatory Responses Are Localized to the Conserved Amino and Carboxyl Regions of the Protein. J. Immunol. 2001, 167, 7009–7016. [Google Scholar] [CrossRef]
- Cui, D.; Zhang, J.; Zuo, Y.; Huo, S.; Zhang, Y.; Wang, L.; Li, X.; Zhong, F. Recombinant Chicken Interleukin-7 as a Potent Adjuvant Increases the Immunogenicity and Protection of Inactivated Infectious Bursal Disease Vaccine. Vet. Res. 2018, 49, 10. [Google Scholar] [CrossRef]
- Rady, H.F.; Dai, G.; Huang, W.; Shellito, J.E.; Ramsay, A.J. Flagellin Encoded in Gene-Based Vector Vaccines Is a Route-Dependent Immune Adjuvant. PLoS ONE 2016, 11, e0148701. [Google Scholar] [CrossRef]
- Ciacci-Woolwine, F.; Blomfield, I.C.; Richardson, S.H.; Mizel, S.B. Salmonella Flagellin Induces Tumor Necrosis Factor Alpha in a Human Promonocytic Cell Line. Infect. Immun. 1998, 66, 1127–1134. [Google Scholar] [CrossRef]
- Wyant, T.L.; Tanner, M.K.; Sztein, M.B. Salmonella typhi Flagella Are Potent Inducers of Proinflammatory Cytokine Secretion by Human Monocytes. Infect. Immun. 1999, 67, 3619–3624. [Google Scholar] [CrossRef]
- McDermott, P.F.; Ciacci-Woolwine, F.; Snipes, J.A.; Mizel, S.B. High-Affinity Interaction between Gram-Negative Flagellin and a Cell Surface Polypeptide Results in Human Monocyte Activation. Infect. Immun. 2000, 68, 5525–5529. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Levy, O.; Dowling, D.J. The TLR5 Agonist Flagellin Shapes Phenotypical and Functional Activation of Lung Mucosal Antigen Presenting Cells in Neonatal Mice. Front. Immunol. 2020, 11, 171. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.-I.; Kurnasov, O.; Natarajan, V.; Hong, M.; Gudkov, A.V.; Osterman, A.L.; Wilson, I.A. Structural Basis of TLR5-Flagellin Recognition and Signaling. Science 2012, 335, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Ben-Yedidia, T.; Tarrab-Hazdai, R.; Schechtman, D.; Arnon, R. Intranasal Administration of Synthetic Recombinant Peptide-Based Vaccine Protects Mice from Infection by Schistosoma mansoni. Infect. Immun. 1999, 67, 4360–4366. [Google Scholar] [CrossRef] [PubMed]
- Delaney, K.N.; Phipps, J.P.; Johnson, J.B.; Mizel, S.B. A Recombinant Flagellin-Poxvirus Fusion Protein Vaccine Elicits Complement-Dependent Protection Against Respiratory Challenge with Vaccinia Virus in Mice. Viral Immunol. 2010, 23, 201–210. [Google Scholar] [CrossRef]
- Bargieri, D.Y.; Leite, J.A.; Lopes, S.C.P.; Sbrogio-Almeida, M.E.; Braga, C.J.M.; Ferreira, L.C.S.; Soares, I.S.; Costa, F.T.M.; Rodrigues, M.M. Immunogenic Properties of a Recombinant Fusion Protein Containing the C-Terminal 19kDa of Plasmodium falciparum Merozoite Surface Protein-1 and the Innate Immunity Agonist FliC Flagellin of Salmonella Typhimurium. Vaccine 2010, 28, 2818–2826. [Google Scholar] [CrossRef]
- Kaba, S.A.; Karch, C.P.; Seth, L.; Ferlez, K.M.B.; Storme, C.K.; Pesavento, D.M.; Laughlin, P.Y.; Bergmann-Leitner, E.S.; Burkhard, P.; Lanar, D.E. Self-Assembling Protein Nanoparticles with Built-In Flagellin Domains Increases Protective Efficacy of a Plasmodium falciparum Based Vaccine. Vaccine 2018, 36, 906–914. [Google Scholar] [CrossRef]
- Ajamian, L.; Melnychuk, L.; Jean-Pierre, P.; Zaharatos, G.J. DNA Vaccine-Encoded Flagellin Can Be Used as an Adjuvant Scaffold to Augment HIV-1 Gp41 Membrane Proximal External Region Immunogenicity. Viruses 2018, 10, 100. [Google Scholar] [CrossRef]
- Lee, S.E.; Koh, Y.I.; Kim, M.-K.; Kim, Y.R.; Kim, S.Y.; Nam, J.H.; Choi, Y.D.; Bae, S.J.; Ko, Y.J.; Ryu, H.-J.; et al. Inhibition of Airway Allergic Disease by Co-Administration of Flagellin with Allergen. J. Clin. Immunol. 2008, 28, 157–165. [Google Scholar] [CrossRef]
- Kim, E.H.; Kim, J.H.; Samivel, R.; Bae, J.-S.; Chung, Y.-J.; Chung, P.-S.; Lee, S.E.; Mo, J.-H. Intralymphatic Treatment of Flagellin-Ovalbumin Mixture Reduced Allergic Inflammation in Murine Model of Allergic Rhinitis. Allergy 2016, 71, 629–639. [Google Scholar] [CrossRef]
- Treanor, J.J.; Taylor, D.N.; Tussey, L.; Hay, C.; Nolan, C.; Fitzgerald, T.; Liu, G.; Kavita, U.; Song, L.; Dark, I.; et al. Safety and Immunogenicity of a Recombinant Hemagglutinin Influenza–Flagellin Fusion Vaccine (VAX125) in Healthy Young Adults. Vaccine 2010, 28, 8268–8274. [Google Scholar] [CrossRef]
- Taylor, D.N.; Treanor, J.J.; Sheldon, E.; Johnson, C.; Umlauf, S.; Song, L.; Kavita, U.; Liu, G.; Tussey, L.; Ozer, K.; et al. Development of VAX128, a Recombinant Hemagglutinin (HA) Influenza-Flagellin Fusion Vaccine with Improved Safety and Immune Response. Vaccine 2012, 30, 5761–5769. [Google Scholar] [CrossRef]
- Gauthier, L.; Babych, M.; Segura, M.; Bourgault, S.; Archambault, D. Identification of a Novel TLR5 Agonist Derived from the P97 Protein of Mycoplasma hyopneumoniae. Immunobiology 2020, 225, 151962. [Google Scholar] [CrossRef]
- Lu, Y.; Swartz, J.R. Functional Properties of Flagellin as a Stimulator of Innate Immunity. Sci. Rep. 2016, 6, 18379. [Google Scholar] [CrossRef]
- Melin, N.; Sánchez-Taltavull, D.; Fahrner, R.; Keogh, A.; Dosch, M.; Büchi, I.; Zimmer, Y.; Medová, M.; Beldi, G.; Aebersold, D.M.; et al. Synergistic Effect of the TLR5 Agonist CBLB502 and Its Downstream Effector IL-22 against Liver Injury. Cell Death Dis. 2021, 12, 366. [Google Scholar] [CrossRef]
- Ding, X.; Bian, G.; Leigh, N.D.; Qiu, J.; McCarthy, P.L.; Liu, H.; Aygun-Sunar, S.; Burdelya, L.G.; Gudkov, A.V.; Cao, X. A TLR5 Agonist Enhances CD8+ T Cell-Mediated Graft-versus-Tumor Effect without Exacerbating Graft-versus-Host Disease. J. Immunol. 2012, 189, 4719–4727. [Google Scholar] [CrossRef]
- Cummings, R.D.; McEver, R.P. C-Type Lectins. Essentials of Glycobiology, 2nd ed.; Cold Spring Harbor: New York, NY, USA, 2009; ISBN 978-0-87969-770-9. [Google Scholar]
- Hoving, J.C.; Wilson, G.J.; Brown, G.D. Signaling C-Type Lectin Receptors, Microbial Recognition and Immunity. Cell. Microbiol. 2014, 16, 185–194. [Google Scholar] [CrossRef]
- Engering, A.; Geijtenbeek, T.B.H.; van Vliet, S.J.; Wijers, M.; van Liempt, E.; Demaurex, N.; Lanzavecchia, A.; Fransen, J.; Figdor, C.G.; Piguet, V.; et al. The Dendritic Cell-Specific Adhesion Receptor DC-SIGN Internalizes Antigen for Presentation to T Cells. J. Immunol. 2002, 168, 2118–2126. [Google Scholar] [CrossRef] [PubMed]
- Court, N.; Vasseur, V.; Vacher, R.; Frémond, C.; Shebzukhov, Y.; Yeremeev, V.V.; Maillet, I.; Nedospasov, S.A.; Gordon, S.; Fallon, P.G.; et al. Partial Redundancy of the Pattern Recognition Receptors, Scavenger Receptors, and C-Type Lectins for the Long-Term Control of Mycobacterium tuberculosis Infection. J. Immunol. 2010, 184, 7057–7070. [Google Scholar] [CrossRef]
- Heitmann, L.; Schoenen, H.; Ehlers, S.; Lang, R.; Hölscher, C. Mincle Is Not Essential for Controlling Mycobacterium tuberculosis Infection. Immunobiology 2013, 218, 506–516. [Google Scholar] [CrossRef]
- Gesheva, V.; Idakieva, K.; Kerekov, N.; Nikolova, K.; Mihaylova, N.; Doumanova, L.; Tchorbanov, A. Marine Gastropod Hemocyanins as Adjuvants of Non-Conjugated Bacterial and Viral Proteins. Fish Shellfish Immunol. 2011, 30, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Gesheva, V.; Chausheva, S.; Stefanova, N.; Mihaylova, N.; Doumanova, L.; Idakieva, K.; Tchorbanov, A. Helix pomatia Hemocyanin—A Novel Bio-Adjuvant for Viral and Bacterial Antigens. Int. Immunopharmacol. 2015, 26, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Román, J.J.M.; del Campo, M.; Villar, J.; Paolini, F.; Curzio, G.; Venuti, A.; Jara, L.; Ferreira, J.; Murgas, P.; Lladser, A.; et al. Immunotherapeutic Potential of Mollusk Hemocyanins in Combination with Human Vaccine Adjuvants in Murine Models of Oral Cancer. J. Immunol. Res. 2019, 2019, 7076942. [Google Scholar] [CrossRef]
- Means, T.K.; Hayashi, F.; Smith, K.D.; Aderem, A.; Luster, A.D. The Toll-Like Receptor 5 Stimulus Bacterial Flagellin Induces Maturation and Chemokine Production in Human Dendritic Cells. J. Immunol. 2003, 170, 5165–5175. [Google Scholar] [CrossRef] [PubMed]
- Roth, G.A.; Picece, V.C.T.M.; Ou, B.S.; Luo, W.; Pulendran, B.; Appel, E.A. Designing Spatial and Temporal Control of Vaccine Responses. Nat. Rev. Mater. 2021, 7, 174–195. [Google Scholar] [CrossRef]
- Pollard, A.J.; Bijker, E.M. A Guide to Vaccinology: From Basic Principles to New Developments. Nat. Rev. Immunol. 2021, 21, 83–100. [Google Scholar] [CrossRef]
- Deng, J.; Yu, X.-Q.; Wang, P.-H. Inflammasome Activation and Th17 Responses. Mol. Immunol. 2019, 107, 142–164. [Google Scholar] [CrossRef]
- Conroy, H.; Marshall, N.A.; Mills, K.H.G. TLR Ligand Suppression or Enhancement of Treg Cells? A Double-Edged Sword in Immunity to Tumours. Oncogene 2008, 27, 168–180. [Google Scholar] [CrossRef]
- MacLeod, M.K.L.; David, A.; McKee, A.S.; Crawford, F.; Kappler, J.W.; Marrack, P. Memory CD4 T Cells That Express CXCR5 Provide Accelerated Help to B Cells. J. Immunol. 2011, 186, 2889–2896. [Google Scholar] [CrossRef]
- Crotty, S. T Follicular Helper Cell Biology: A Decade of Discovery and Diseases. Immunity 2019, 50, 1132–1148. [Google Scholar] [CrossRef]
- Mesin, L.; Ersching, J.; Victora, G.D. Germinal Center B Cell Dynamics. Immunity 2016, 45, 471–482. [Google Scholar] [CrossRef]
- Sanchez-Trincado, J.L.; Gomez-Perosanz, M.; Reche, P.A. Fundamentals and Methods for T- and B-Cell Epitope Prediction. J. Immunol. Res. 2017, 2017, 2680160. [Google Scholar] [CrossRef]
- Nithichanon, A.; Rinchai, D.; Gori, A.; Lassaux, P.; Peri, C.; Conchillio-Solé, O.; Ferrer-Navarro, M.; Gourlay, L.J.; Nardini, M.; Vila, J.; et al. Sequence- and Structure-Based Immunoreactive Epitope Discovery for Burkholderia pseudomallei Flagellin. PLoS Negl. Trop. Dis. 2015, 9, e0003917. [Google Scholar] [CrossRef]
- Saylor, K.; Gillam, F.; Lohneis, T.; Zhang, C. Designs of Antigen Structure and Composition for Improved Protein-Based Vaccine Efficacy. Front. Immunol. 2020, 11, 283. [Google Scholar] [CrossRef]
- Home—ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/home (accessed on 14 June 2022).
- Burdelya, L.G.; Krivokrysenko, V.I.; Tallant, T.C.; Strom, E.; Gleiberman, A.S.; Gupta, D.; Kurnasov, O.V.; Fort, F.L.; Osterman, A.L.; DiDonato, J.A.; et al. An Agonist of Toll-Like Receptor 5 Has Radioprotective Activity in Mouse and Primate Models. Science 2008, 320, 226–230. [Google Scholar] [CrossRef]
- Hossain, M.S.; Ramachandiran, S.; Gewirtz, A.T.; Waller, E.K. Recombinant TLR5 Agonist CBLB502 Promotes NK Cell-Mediated Anti-CMV Immunity in Mice. PLoS ONE 2014, 9, e96165. [Google Scholar] [CrossRef]
- Tussey, L.; Strout, C.; Davis, M.; Johnson, C.; Lucksinger, G.; Umlauf, S.; Song, L.; Liu, G.; Abraham, K.; White, C.J. Phase 1 Safety and Immunogenicity Study of a Quadrivalent Seasonal Flu Vaccine Comprising Recombinant Hemagglutinin-Flagellin Fusion Proteins. Open Forum Infect. Dis. 2016, 3, ofw015. [Google Scholar] [CrossRef]
- Frey, S.E.; Lottenbach, K.; Graham, I.; Anderson, E.; Bajwa, K.; May, R.C.; Mizel, S.B.; Graff, A.; Belshe, R.B. A Phase I Safety and Immunogenicity Dose Escalation Trial of Plague Vaccine, Flagellin/F1/V, in Healthy Adult Volunteers (DMID 08-0066). Vaccine 2017, 35, 6759–6765. [Google Scholar] [CrossRef]
- Rivalland, G.; Loveland, B.; Mitchell, P. Update on Mucin-1 Immunotherapy in Cancer: A Clinical Perspective. Expert Opin. Biol. Ther. 2015, 15, 1773–1787. [Google Scholar] [CrossRef]
- Mizel, S.B.; Graff, A.H.; Sriranganathan, N.; Ervin, S.; Lees, C.J.; Lively, M.O.; Hantgan, R.R.; Thomas, M.J.; Wood, J.; Bell, B. Flagellin-F1-V Fusion Protein Is an Effective Plague Vaccine in Mice and Two Species of Nonhuman Primates. Clin. Vaccine Immunol. 2009, 16, 21–28. [Google Scholar] [CrossRef]
- Deng, L.; Cho, K.J.; Fiers, W.; Saelens, X. M2e-Based Universal Influenza A Vaccines. Vaccines 2015, 3, 105–136. [Google Scholar] [CrossRef]
- Talbot, H.K.; Rock, M.T.; Johnson, C.; Tussey, L.; Kavita, U.; Shanker, A.; Shaw, A.R.; Taylor, D.N. Immunopotentiation of Trivalent Influenza Vaccine When Given with VAX102, a Recombinant Influenza M2e Vaccine Fused to the TLR5 Ligand Flagellin. PLoS ONE 2010, 5, e14442. [Google Scholar] [CrossRef]
- Turley, C.B.; Rupp, R.E.; Johnson, C.; Taylor, D.N.; Wolfson, J.; Tussey, L.; Kavita, U.; Stanberry, L.; Shaw, A. Safety and Immunogenicity of a Recombinant M2e–Flagellin Influenza Vaccine (STF2.4xM2e) in Healthy Adults. Vaccine 2011, 29, 5145–5152. [Google Scholar] [CrossRef]
- Taylor, D.N.; Treanor, J.J.; Strout, C.; Johnson, C.; Fitzgerald, T.; Kavita, U.; Ozer, K.; Tussey, L.; Shaw, A. Induction of a Potent Immune Response in the Elderly Using the TLR-5 Agonist, Flagellin, with a Recombinant Hemagglutinin Influenza–Flagellin Fusion Vaccine (VAX125, STF2.HA1 SI). Vaccine 2011, 29, 4897–4902. [Google Scholar] [CrossRef]
- Holmgren, J. An Update on Cholera Immunity and Current and Future Cholera Vaccines. Trop. Med. Infect. Dis. 2021, 6, 64. [Google Scholar] [CrossRef]
- Stanford, M.; Whittall, T.; Bergmeier, L.A.; Lindblad, M.; Lundin, S.; Shinnick, T.; Mizushima, Y.; Holmgren, J.; Lehner, T. Oral Tolerization with Peptide 336–351 Linked to Cholera Toxin B Subunit in Preventing Relapses of Uveitis in Behcet’s Disease. Clin. Exp. Immunol. 2004, 137, 201–208. [Google Scholar] [CrossRef]
- Pan, S.-C.; Hsu, W.-T.; Lee, W.-S.; Wang, N.-C.; Chen, T.-J.; Liu, M.-C.; Pai, H.-C.; Hsu, Y.-S.; Chang, M.; Hsieh, S.-M. A Double-Blind, Randomized Controlled Trial to Evaluate the Safety and Immunogenicity of an Intranasally Administered Trivalent Inactivated Influenza Vaccine with the Adjuvant LTh(αK): A Phase II Study. Vaccine 2020, 38, 1048–1056. [Google Scholar] [CrossRef]
- Challa, S.; Szczepanek, S.M.; Rood, D.; Barrette, R.W.; Silbart, L.K. Bacterial Toxin Fusion Proteins Elicit Mucosal Immunity against a Foot-and-Mouth Disease Virus Antigen When Administered Intranasally to Guinea Pigs. Adv. Virol. 2011, 2011, 713769. [Google Scholar] [CrossRef] [PubMed]
- Savelkoul, H.F.J.; Ferro, V.A.; Strioga, M.M.; Schijns, V.E.J.C. Choice and Design of Adjuvants for Parenteral and Mucosal Vaccines. Vaccines 2015, 3, 148–171. [Google Scholar] [CrossRef] [PubMed]
- Qadri, F.; Akhtar, M.; Bhuiyan, T.R.; Chowdhury, M.I.; Ahmed, T.; Rafique, T.A.; Khan, A.; Rahman, S.I.A.; Khanam, F.; Lundgren, A.; et al. Safety and Immunogenicity of the Oral, Inactivated, Enterotoxigenic Escherichia coli Vaccine ETVAX in Bangladeshi Children and Infants: A Double-Blind, Randomised, Placebo-Controlled Phase 1/2 Trial. Lancet Infect. Dis. 2020, 20, 208–219. [Google Scholar] [CrossRef]
- Curtis, J.E.; Hersh, E.M.; Harris, J.E.; McBRIDE, C.; Freireich, E.J. The Human Primary Immune Response to Keyhole Limpet Haemocyanin: Interrelationships of Delayed Hypersensitivity, Antibody Response and In Vitro Blast Transformation. Clin. Exp. Immpunol. 1970, 6, 473–491. [Google Scholar] [CrossRef]
- Weigle, W.O. Immunochemical Properties of Hemocyanin. Immunochemistry 1964, 1, 295–302. [Google Scholar] [CrossRef]
- Swanson, M.A.; Schwartz, R.S. Immunosuppressive Therapy: The Relation between Clinical Response and Immunologic Competence. N. Engl. J. Med. 1967, 277, 163–170. [Google Scholar] [CrossRef]
- Del Campo, M.; Arancibia, S.; Nova, E.; Salazar, F.; González, A.; Moltedo, B.; de Ionannes, P.; Ferreira, J.; Manubens, A.; Becker, M.I. Hemocyanins as Immunostimulants. Rev. Med. Chile 2010, 139, 236–246. [Google Scholar]
- Swaminathan, A.; Lucas, R.; Dear, K.; McMichael, A.J. Keyhole Limpet Haemocyanin—A Model Antigen for Human Immunotoxicological Studies: KLH as Neoantigen in Human Immunotoxicological Studies. Br. J. Clin. Pharmacol. 2014, 78, 1135–1142. [Google Scholar] [CrossRef]
- Olsson, C.A.; Chute, R.; Rao, C.N. Immunologic Reduction of Bladder Cancer Recurrence Rate. J. Urol. 1974, 111, 173–176. [Google Scholar] [CrossRef]
- Jurincic, C.D.; Engelmann, U.; Gasch, J.; Klippel, K.F. Immunotherapy in Bladder Cancer with Keyhole-Limpet Hemocyanin: A Randomized Study. J. Urol. 1988, 139, 723–726. [Google Scholar] [CrossRef]
- Results for KLH at ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/results?term=KLH (accessed on 14 June 2022).
- Gilewski, T.A.; Ragupathi, G.; Dickler, M.; Powell, S.; Bhuta, S.; Panageas, K.; Koganty, R.R.; Chin-Eng, J.; Hudis, C.; Norton, L.; et al. Immunization of High-Risk Breast Cancer Patients with Clustered sTn-KLH Conjugate plus the Immunologic Adjuvant QS-21. Clin. Cancer Res. 2007, 13, 2977–2985. [Google Scholar] [CrossRef]
- Miles, D.; Roché, H.; Martin, M.; Perren, T.J.; Cameron, D.A.; Glaspy, J.; Dodwell, D.; Parker, J.; Mayordomo, J.; Tres, A.; et al. Phase III Multicenter Clinical Trial of the Sialyl-TN (STn)-Keyhole Limpet Hemocyanin (KLH) Vaccine for Metastatic Breast Cancer. Oncologist 2011, 16, 1092–1100. [Google Scholar] [CrossRef]
- Ragupathi, G.; Meyers, M.; Adluri, S.; Howard, L.; Musselli, C.; Livingston, P.O. Induction of Antibodies against GD3 Ganglioside in Melanoma Patients by Vaccination with GD3-Lactone-KLH Conjugate Plus Immunological Adjuvant QS-21. Int. J. Cancer 2000, 85, 659–666. [Google Scholar] [CrossRef]
- Eggermont, A.M.M.; Suciu, S.; Rutkowski, P.; Marsden, J.; Santinami, M.; Corrie, P.; Aamdal, S.; Ascierto, P.A.; Patel, P.M.; Kruit, W.H.; et al. Adjuvant Ganglioside GM2-KLH/QS-21 Vaccination Versus Observation After Resection of Primary Tumor > 1.5 mm in Patients with Stage II Melanoma: Results of the EORTC 18961 Randomized Phase III Trial. J. Clin. Oncol. 2013, 31, 3831–3837. [Google Scholar] [CrossRef]
- Krug, L.M.; Ragupathi, G.; Ng, K.K.; Hood, C.; Jennings, H.J.; Guo, Z.; Kris, M.G.; Miller, V.; Pizzo, B.; Tyson, L.; et al. Vaccination of Small Cell Lung Cancer Patients with Polysialic Acid or N-Propionylated Polysialic Acid Conjugated to Keyhole Limpet Hemocyanin. Clin. Cancer Res. 2004, 10, 916–923. [Google Scholar] [CrossRef]
- Krug, L.M.; Ragupathi, G.; Hood, C.; George, C.; Hong, F.; Shen, R.; Abrey, L.; Jennings, H.J.; Kris, M.G.; Livingston, P.O. Immunization with N-Propionyl Polysialic Acid–KLH Conjugate in Patients with Small Cell Lung Cancer Is Safe and Induces IgM Antibodies Reactive with SCLC Cells and Bactericidal against Group B Meningococci. Cancer Immunol. Immunother. 2012, 61, 9–18. [Google Scholar] [CrossRef]
- Tchorbanov, A.; Idakieva, K.; Mihaylova, N.; Doumanova, L. Modulation of the Immune Response Using Rapana thomasiana Hemocyanin. Int. Immunopharmacol. 2008, 8, 1033–1038. [Google Scholar] [CrossRef]
- Markl, J.; Lieb, B.; Gebauer, W.; Altenhein, B.; Meissner, U.; Harris, J.R. Marine Tumor Vaccine Carriers: Structure of the Molluscan Hemocyanins KLH and HtH. J. Cancer Res. Clin. Oncol. 2001, 127, R3–R9. [Google Scholar] [CrossRef]
- Keller, H.; Lieb, B.; Altenhein, B.; Gebauer, D.; Richter, S.; Stricker, S.; Markl, J. Abalone (Haliotis tuberculata) Hemocyanin Type 1 (HtH1). Organization of the ≈ 400 kDa Subunit, and Amino Acid Sequence of Its Functional Units f, g and h. Eur. J. Biochem. 1999, 264, 27–38. [Google Scholar] [CrossRef]
- Reyes, D.; Salazar, L.; Espinoza, E.; Pereda, C.; Castellón, E.; Valdevenito, R.; Huidobro, C.; Becker, M.I.; Lladser, A.; López, M.N.; et al. Tumour Cell Lysate-Loaded Dendritic Cell Vaccine Induces Biochemical and Memory Immune Response in Castration-Resistant Prostate Cancer Patients. Br. J. Cancer 2013, 109, 1488–1497. [Google Scholar] [CrossRef]
- Röllig, C.; Schmidt, C.; Bornhäuser, M.; Ehninger, G.; Schmitz, M.; Auffermann-Gretzinger, S. Induction of Cellular Immune Responses in Patients with Stage-I Multiple Myeloma After Vaccination with Autologous Idiotype-pulsed Dendritic Cells. J. Immunother. 2011, 34, 100–106. [Google Scholar] [CrossRef]
- Langlais, C.; Korn, B. Recombinant Protein Expression in Bacteria. In Encyclopedic Reference of Genomics and Proteomics in Molecular Medicine; Springer: Berlin/Heidelberg, Germany, 2006; pp. 1609–1616. ISBN 978-3-540-44244-8. [Google Scholar]
- Gupta, S.K.; Shukla, P. Glycosylation Control Technologies for Recombinant Therapeutic Proteins. Appl. Microbiol. Biotechnol. 2018, 102, 10457–10468. [Google Scholar] [CrossRef]
- Hannig, G.; Makrides, S.C. Strategies for Optimizing Heterologous Protein Expression in Escherichia coli. Trends Biotechnol. 1998, 16, 54–60. [Google Scholar] [CrossRef]
- Mierendorf, R.C.; Morris, B.B.; Hammer, B.; Novy, R.E. Expression and Purification of Recombinant Proteins Using the PET System. Methods Mol. Med. 1998, 13, 257–292. [Google Scholar] [CrossRef] [PubMed]
- Studier, F.W.; Moffatt, B.A. Use of Bacteriophage T7 RNA Polymerase to Direct Selective High-Level Expression of Cloned Genes. J. Mol. Biol. 1986, 189, 113–130. [Google Scholar] [CrossRef]
- Boisguérin, V. Recombinant Expression of Molluscan Hemocyanin (KLH) Substructures in a Prokaryotic System: E. Coli. Ph.D. Dissertation, Johannes Gutenberg-Universität, Mainz, Germany, 2006. [Google Scholar]
- Mizel, S.B.; Bates, J.T. Flagellin as an Adjuvant: Cellular Mechanisms and Potential. J. Immunol. 2010, 185, 5677–5682. [Google Scholar] [CrossRef]
- Biedma, M.E.; Cayet, D.; Tabareau, J.; Rossi, A.H.; Ivičak-Kocjan, K.; Moreno, G.; Errea, A.; Soulard, D.; Parisi, G.; Jerala, R.; et al. Recombinant Flagellins with Deletions in Domains D1, D2, and D3: Characterization as Novel Immunoadjuvants. Vaccine 2019, 37, 652–663. [Google Scholar] [CrossRef]
- Verma, S.K. Overexpression, Purification, and Immunogenicity of Recombinant Porin Proteins of Salmonella enterica Serovar Typhi (S. typhi). J. Microbiol. Biotechnol. 2009, 19, 1034–1040. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, L.-A.S.; Kerr, A.R.; Douce, G.R.; Paterson, G.K.; Dilts, D.A.; Liu, D.-F.; Mitchell, T.J. Construction and Immunological Characterization of a Novel Nontoxic Protective Pneumolysin Mutant for Use in Future Pneumococcal Vaccines. Infect. Immun. 2006, 74, 586–593. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, H.; Zhang, S.; Zeng, L.; Xu, X.; Wu, K.; Wang, W.; Yin, N.; Song, Z.; Zhang, X.; et al. Mucosal Immunization with Recombinant Fusion Protein DnaJ-ΔA146Ply Enhances Cross-Protective Immunity against Streptococcus pneumoniae Infection in Mice via Interleukin 17A. Infect. Immun. 2014, 82, 1666–1675. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, W.; Wang, S. Effect of Vaccine Administration Modality on Immunogenicity and Efficacy. Expert Rev. Vaccines 2015, 14, 1509–1523. [Google Scholar] [CrossRef]
- Lavelle, E.C.; O’Hagan, D.T. Delivery Systems and Adjuvants for Oral Vaccines. Expert Opin. Drug Deliv. 2006, 3, 747–762. [Google Scholar] [CrossRef]
- Rosenbaum, P.; Tchitchek, N.; Joly, C.; Pozo, A.R.; Stimmer, L.; Langlois, S.; Hocini, H.; Gosse, L.; Pejoski, D.; Cosma, A.; et al. Vaccine Inoculation Route Modulates Early Immunity and Consequently Antigen-Specific Immune Response. Front. Immunol. 2021, 12, 645210. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines on the Nonclinical Evaluation of Vaccine Adjuvants and Adjuvanted Vaccines; Annex 2, TRS No. 987; WHO: Geneva, Switzerland, 2014; pp. 987, 59–100. [Google Scholar]
- Gruber, M.F.; Marshall, V.B. Regulation and Testing of Vaccines. In Plotkin’s Vaccines; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1547–1565.e2. [Google Scholar] [CrossRef]
- Reiser, M.L.; Mosaheb, M.M.; Lisk, C.; Platt, A.; Wetzler, L.M. The TLR2 Binding Neisserial Porin PorB Enhances Antigen Presenting Cell Trafficking and Cross-Presentation. Sci. Rep. 2017, 7, 736. [Google Scholar] [CrossRef]
- Wetzler, L.M. Innate Immune Function of the Neisserial Porins and the Relationship to Vaccine Adjuvant Activity. Future Microbiol. 2010, 5, 749–758. [Google Scholar] [CrossRef]
- Anderson, J.M. Biocompatibility. In Polymer Science: A Comprehensive Reference; Elsevier: Amsterdam, The Netherlands, 2012; pp. 363–383. ISBN 978-0-08-087862-1. [Google Scholar]
- Gupta, R.K.; Relyveld, E.H.; Lindblad, E.B.; Bizzini, B.; Ben-Efraim, S.; Gupta, C.K. Adjuvants—A Balance between Toxicity and Adjuvanticity. Vaccine 1993, 11, 293–306. [Google Scholar] [CrossRef]
- Balaji, A.B.; Pakalapati, H.; Khalid, M.; Walvekar, R.; Siddiqui, H. Natural and Synthetic Biocompatible and Biodegradable Polymers. In Biodegradable and Biocompatible Polymer Composites; Elsevier: Amsterdam, The Netherlands, 2018; pp. 3–32. ISBN 978-0-08-100970-3. [Google Scholar]
- Poulain-Godefroy, O.; Vendeville, C.; Locht, C.; Riveau, G. Bordetella pertussis Filamentous Hemagglutinin Delivered by Mucosal Routes Enhances Immunoglobulin Levels in Serum and Mucosal Fluids. FEMS Microbiol. Immunol. 2008, 54, 129–136. [Google Scholar] [CrossRef]
- Pasquevich, K.A.; Ibañez, A.E.; Coria, L.M.; Samartino, C.G.; Estein, S.M.; Zwerdling, A.; Barrionuevo, P.; Oliveira, F.S.; Seither, C.; Warzecha, H.; et al. An Oral Vaccine Based on U-Omp19 Induces Protection against B. Abortus Mucosal Challenge by Inducing an Adaptive IL-17 Immune Response in Mice. PLoS ONE 2011, 6, e16203. [Google Scholar] [CrossRef]
- Batista-Duharte, A.; Martínez, D.T.; Carlos, I.Z. Efficacy and Safety of Immunological Adjuvants. Where Is the Cut-Off? Biomed. Pharmacother. 2018, 105, 616–624. [Google Scholar] [CrossRef]
- Petrovsky, N. Comparative Safety of Vaccine Adjuvants: A Summary of Current Evidence and Future Needs. Drug Saf. 2015, 38, 1059–1074. [Google Scholar] [CrossRef]
- Wang, Z.-B.; Xu, J. Better Adjuvants for Better Vaccines: Progress in Adjuvant Delivery Systems, Modifications, and Adjuvant–Antigen Codelivery. Vaccines 2020, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Vasserman, Y.; Pitcovski, J. Genetic Detoxification and Adjuvant-Activity Retention of Escherichia coli Enterotoxin LT. Avian Pathol. 2006, 35, 134–140. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sasaki, E.; Asanuma, H.; Momose, H.; Furuhata, K.; Mizukami, T.; Hamaguchi, I. Immunogenicity and Toxicity of Different Adjuvants Can Be Characterized by Profiling Lung Biomarker Genes After Nasal Immunization. Front. Immunol. 2020, 11, 2171. [Google Scholar] [CrossRef]
- Savar, N.S.; Jahanian-Najafabadi, A.; Mahdavi, M.; Shokrgozar, M.A.; Jafari, A.; Bouzari, S. In Silico and In Vivo Studies of Truncated Forms of Flagellin (FliC) of Enteroaggregative Escherichia coli Fused to FimH from Uropathogenic Escherichia coli as a Vaccine Candidate against Urinary Tract Infections. J. Biotechnol. 2014, 175, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Guy, B. The Perfect Mix: Recent Progress in Adjuvant Research. Nat. Rev. Microbiol. 2007, 5, 396–397. [Google Scholar] [CrossRef] [PubMed]
- Huleatt, J.W.; Jacobs, A.R.; Tang, J.; Desai, P.; Kopp, E.B.; Huang, Y.; Song, L.; Nakaar, V.; Powell, T. Vaccination with Recombinant Fusion Proteins Incorporating Toll-like Receptor Ligands Induces Rapid Cellular and Humoral Immunity. Vaccine 2007, 25, 763–775. [Google Scholar] [CrossRef]
- Oliveira, L.V.N.; Wang, R.; Specht, C.A.; Levitz, S.M. Vaccines for Human Fungal Diseases: Close but Still a Long Way to Go. NPJ Vaccines 2021, 6, 33. [Google Scholar] [CrossRef]
- Krupka, M.; Zachova, K.; Cahlikova, R.; Vrbkova, J.; Novak, Z.; Sebela, M.; Weigl, E.; Raska, M. Endotoxin-Minimized HIV-1 P24 Fused to Murine Hsp70 Activates Dendritic Cells, Facilitates Endocytosis and P24-Specific Th1 Response in Mice. Immunol. Lett. 2015, 166, 36–44. [Google Scholar] [CrossRef]
- Jiang, J.; Xie, D.; Zhang, W.; Xiao, G.; Wen, J. Fusion of Hsp70 to Mage-A1 Enhances the Potency of Vaccine-Specific Immune Responses. J. Transl. Med. 2013, 11, 300. [Google Scholar] [CrossRef]
- Qian, X.; Lu, Y.; Liu, Q.; Chen, K.; Zhao, Q.; Song, J. Prophylactic, Therapeutic and Anti-Metastatic Effects of an HPV-16mE6Δ/ME7/TBhsp70Δ Fusion Protein Vaccine in an Animal Model. Immunol. Lett. 2006, 102, 191–201. [Google Scholar] [CrossRef]
- Wortham, C.; Grinberg, L.; Kaslow, D.C.; Briles, D.E.; McDaniel, L.S.; Lees, A.; Flora, M.; Snapper, C.M.; Mond, J.J. Enhanced Protective Antibody Responses to PspA after Intranasal or Subcutaneous Injections of PspA Genetically Fused to Granulocyte-Macrophage Colony-Stimulating Factor or Interleukin-2. Infect. Immun. 1998, 66, 1513–1520. [Google Scholar] [CrossRef]
- McCluskie, M.J.; Thorn, J.; Mehelic, P.R.; Kolhe, P.; Bhattacharya, K.; Finneman, J.I.; Stead, D.R.; Piatchek, M.B.; Zhang, N.; Chikh, G.; et al. Molecular Attributes of Conjugate Antigen Influence Function of Antibodies Induced by Anti-Nicotine Vaccine in Mice and Non-Human Primates. Int. Immunopharmacol. 2015, 25, 518–527. [Google Scholar] [CrossRef]
- Danishefsky, S.J.; Shue, Y.-K.; Chang, M.N.; Wong, C.-H. Development of Globo-H Cancer Vaccine. Acc. Chem. Res. 2015, 48, 643–652. [Google Scholar] [CrossRef]
- Adotevi, O.; Vingert, B.; Freyburger, L.; Shrikant, P.; Lone, Y.C.; Quintin-Colonna, F.; Haicheur, N.; Amessou, M.; Herbelin, A.; Langlade-Demoyen, P.; et al. B Subunit of Shiga Toxin-Based Vaccines Synergize with α-Galactosylceramide to Break Tolerance against Self Antigen and Elicit Antiviral Immunity. J. Immunol. 2007, 179, 3371–3379. [Google Scholar] [CrossRef]
- Hrafnkelsdottir, K.; Valgeirsson, J.; Bjarnadottir, S.; Olafsdottir, S.; Olafsdottir, K.; Hedinsdottir, S.T.; Magnusdottir, E.V.; Gizurarson, S. Immunization Prevents DDT Buildup in Mouse Tissues. Int. Immunopharmacol. 2007, 7, 1179–1184. [Google Scholar] [CrossRef]
- McCluskie, M.J.; Pryde, D.C.; Gervais, D.P.; Stead, D.R.; Zhang, N.; Benoit, M.; Robertson, K.; Kim, I.-J.; Tharmanathan, T.; Merson, J.R.; et al. Enhancing Immunogenicity of a 3′aminomethylnicotine-DT-Conjugate Anti-Nicotine Vaccine with CpG Adjuvant in Mice and Non-Human Primates. Int. Immunopharmacol. 2013, 16, 50–56. [Google Scholar] [CrossRef]
- Thorn, J.M.; Bhattacharya, K.; Crutcher, R.; Sperry, J.; Isele, C.; Kelly, B.; Yates, L.; Zobel, J.; Zhang, N.; Davis, H.L.; et al. The Effect of Physicochemical Modification on the Function of Antibodies Induced by Anti-Nicotine Vaccine in Mice. Vaccines 2017, 5, 11. [Google Scholar] [CrossRef]
- Xu, Z.; Rivera-Hernandez, T.; Chatterjee, O.; Walker, M.J.; Moyle, P.M. Semisynthetic, Self-Adjuvanting Vaccine Development: Efficient, Site-Specific Sortase A-Mediated Conjugation of Toll-like Receptor 2 Ligand FSL-1 to Recombinant Protein Antigens under Native Conditions and Application to a Model Group A Streptococcal Vaccine. J. Control. Release 2020, 317, 96–108. [Google Scholar] [CrossRef]
- Thérien, A.; Bédard, M.; Carignan, D.; Rioux, G.; Gauthier-Landry, L.; Laliberté-Gagné, M.È.; Bolduc, M.; Savard, P.; Leclerc, D. A Versatile Papaya Mosaic Virus (PapMV) Vaccine Platform Based on Sortase-Mediated Antigen Coupling. J. Nanobiotechnol. 2017, 15, 54. [Google Scholar] [CrossRef]
- Laliberté-Gagné, M.È.; Bolduc, M.; Thérien, A.; Garneau, C.; Casault, P.; Savard, P.; Estaquier, J.; Leclerc, D. Increased Immunogenicity of Full-Length Protein Antigens through Sortase-Mediated Coupling on the PapMV Vaccine Platform. Vaccines 2019, 7, 49. [Google Scholar] [CrossRef]
- Sun, B.; Ji, Z.; Liao, Y.-P.; Wang, M.; Wang, X.; Dong, J.; Chang, C.H.; Li, R.; Zhang, H.; Nel, A.E.; et al. Engineering an Effective Immune Adjuvant by Designed Control of Shape and Crystallinity of Aluminum Oxyhydroxide Nanoparticles. ACS Nano 2013, 7, 10834–10849. [Google Scholar] [CrossRef]
- Li, X.; Aldayel, A.M.; Cui, Z. Aluminum Hydroxide Nanoparticles Show a Stronger Vaccine Adjuvant Activity than Traditional Aluminum Hydroxide Microparticles. J. Control. Release 2014, 173, 148–157. [Google Scholar] [CrossRef]
- Jiang, H.; Wang, Q.; Li, L.; Zeng, Q.; Li, H.; Gong, T.; Zhang, Z.; Sun, X. Turning the Old Adjuvant from Gel to Nanoparticles to Amplify CD8+ T Cell Responses. Adv. Sci. 2018, 5, 1700426. [Google Scholar] [CrossRef]
- Shaw, A.C.; Goldstein, D.R.; Montgomery, R.R. Age-Dependent Dysregulation of Innate Immunity. Nat. Rev. Immunol. 2013, 13, 875–887. [Google Scholar] [CrossRef]
- Weng, N.-P. Aging of the Immune System: How Much Can the Adaptive Immune System Adapt? Immunity 2006, 24, 495–499. [Google Scholar] [CrossRef]
- McElhaney, J.E. Influenza Vaccine Responses in Older Adults. Ageing Res. Rev. 2011, 10, 379–388. [Google Scholar] [CrossRef]
- Jefferson, T.; Rivetti, D.; Rivetti, A.; Rudin, M.; Di Pietrantonj, C.; Demicheli, V. Efficacy and Effectiveness of Influenza Vaccines in Elderly People: A Systematic Review. Lancet 2005, 366, 1165–1174. [Google Scholar] [CrossRef]
- Panda, A.; Qian, F.; Mohanty, S.; van Duin, D.; Newman, F.K.; Zhang, L.; Chen, S.; Towle, V.; Belshe, R.B.; Fikrig, E.; et al. Age-Associated Decrease in TLR Function in Primary Human Dendritic Cells Predicts Influenza Vaccine Response. J. Immunol. 2010, 184, 2518–2527. [Google Scholar] [CrossRef] [PubMed]
- Qian, F.; Wang, X.; Zhang, L.; Chen, S.; Piecychna, M.; Allore, H.; Bockenstedt, L.; Malawista, S.; Bucala, R.; Shaw, A.C.; et al. Age-Associated Elevation in TLR5 Leads to Increased Inflammatory Responses in the Elderly. Aging Cell 2012, 11, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Leng, J.; Stout-Delgado, H.W.; Kavita, U.; Jacobs, A.; Tang, J.; Du, W.; Tussey, L.; Goldstein, D.R. Efficacy of a Vaccine That Links Viral Epitopes to Flagellin in Protecting Aged Mice from Influenza Viral Infection. Vaccine 2011, 29, 8147–8155. [Google Scholar] [CrossRef]
- Salazar, F.; Bignell, E.; Brown, G.D.; Cook, P.C.; Warris, A. Pathogenesis of Respiratory Viral and Fungal Coinfections. Clin. Microbiol. Rev. 2022, 35, e00094-21. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L. Mucosal Adjuvants: Opportunities and Challenges: An Overview of Novel Adjuvants Designed for Improving Vaccine Efficacy. Hum. Vaccines Immunother. 2016, 12, 2456–2458. [Google Scholar] [CrossRef] [PubMed]
- Bonam, S.R.; Partidos, C.D.; Halmuthur, S.K.M.; Muller, S. An Overview of Novel Adjuvants Designed for Improving Vaccine Efficacy. Trends Pharmacol. Sci. 2017, 38, 771–793. [Google Scholar] [CrossRef] [PubMed]
| Specie | PBA | Innate Immune Response | Adaptive Immune Response | Reference |
|---|---|---|---|---|
| Mycoplasma hyopneumoniae | P97 protein | IL-8 secretion | Th1/Th2 immune response | [152] |
| Salmonella strains | Flagellin | NF-kB and MAPKs activation. Induction of proinflammatory mediators resulting in the upregulated expression of cytokines, such as TNF-α, IL-6, IL-8, and proinflammatory free radical synthesizing enzymes, such as the inducible nitric oxide synthase | Efficient immune response in elderly subjects immunized with recombinant hemagglutinin influenza–flagellin fusion vaccine (VAX125) | [128,137,141,153] |
| Pharmacologically optimized derivate from Salmonella flagellin | Recombinant protein-based on Flagellin, Entolimod (CBLB502) | NF-κB-, AP-1- and STAT3-driven immunomodulatory signaling pathway. Induction of CXCL9 and -10 | NK cell-dependent activation of dendritic cells followed by stimulation of a CD8+ T-cell | [151,154,155] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díaz-Dinamarca, D.A.; Salazar, M.L.; Castillo, B.N.; Manubens, A.; Vasquez, A.E.; Salazar, F.; Becker, M.I. Protein-Based Adjuvants for Vaccines as Immunomodulators of the Innate and Adaptive Immune Response: Current Knowledge, Challenges, and Future Opportunities. Pharmaceutics 2022, 14, 1671. https://doi.org/10.3390/pharmaceutics14081671
Díaz-Dinamarca DA, Salazar ML, Castillo BN, Manubens A, Vasquez AE, Salazar F, Becker MI. Protein-Based Adjuvants for Vaccines as Immunomodulators of the Innate and Adaptive Immune Response: Current Knowledge, Challenges, and Future Opportunities. Pharmaceutics. 2022; 14(8):1671. https://doi.org/10.3390/pharmaceutics14081671
Chicago/Turabian StyleDíaz-Dinamarca, Diego A., Michelle L. Salazar, Byron N. Castillo, Augusto Manubens, Abel E. Vasquez, Fabián Salazar, and María Inés Becker. 2022. "Protein-Based Adjuvants for Vaccines as Immunomodulators of the Innate and Adaptive Immune Response: Current Knowledge, Challenges, and Future Opportunities" Pharmaceutics 14, no. 8: 1671. https://doi.org/10.3390/pharmaceutics14081671
APA StyleDíaz-Dinamarca, D. A., Salazar, M. L., Castillo, B. N., Manubens, A., Vasquez, A. E., Salazar, F., & Becker, M. I. (2022). Protein-Based Adjuvants for Vaccines as Immunomodulators of the Innate and Adaptive Immune Response: Current Knowledge, Challenges, and Future Opportunities. Pharmaceutics, 14(8), 1671. https://doi.org/10.3390/pharmaceutics14081671








