Comprehensive Assessment of Somatostatin Receptors in Various Neoplasms: A Systematic Review
Abstract
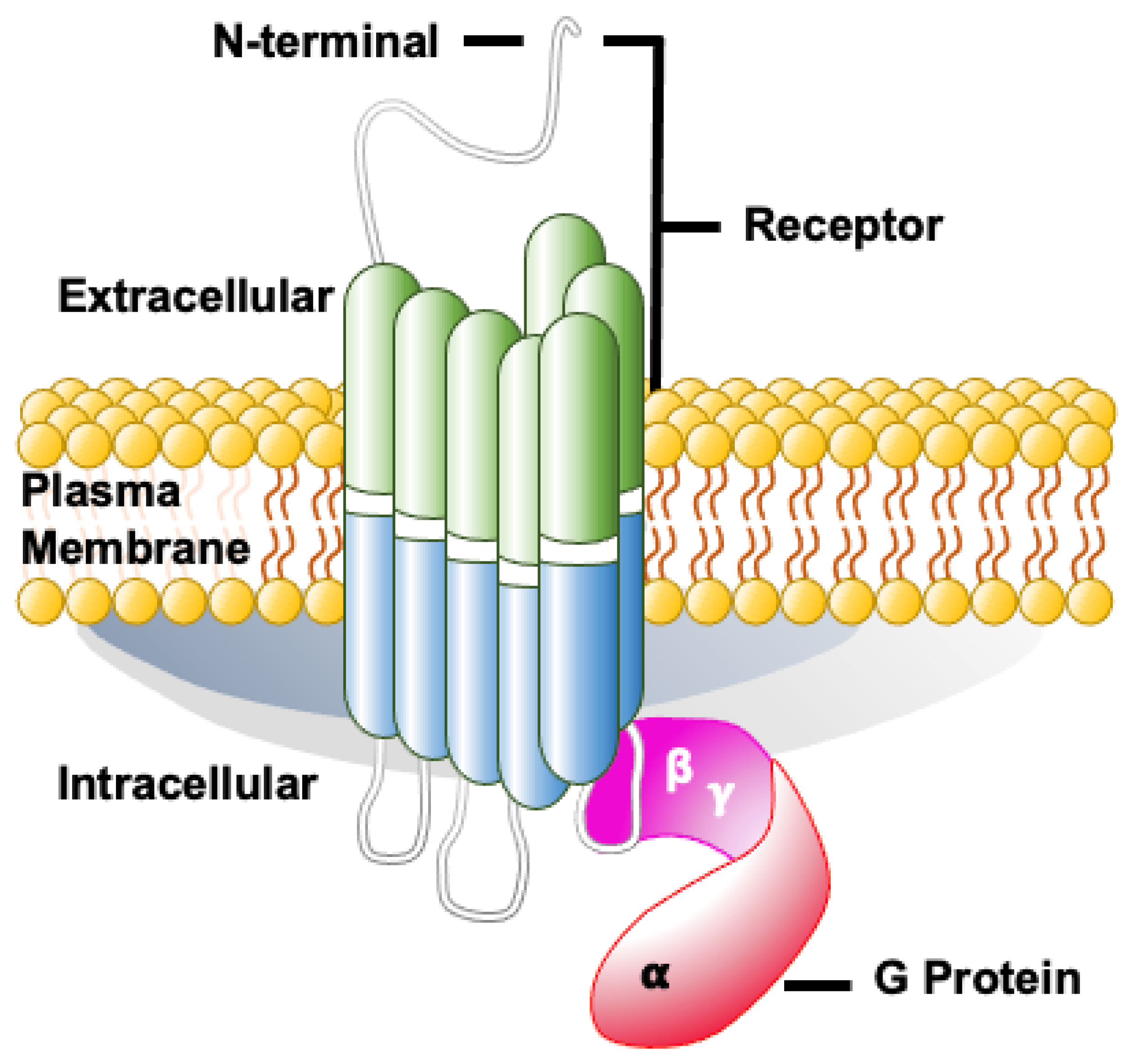
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Lymphoma
3.2. Endometrial Neoplasm
3.3. Prostate Neoplasm
3.4. Gastrointestinal Cancers
3.5. Thyroid Carcinoma
3.6. Thymoma
3.7. Merkel Cell Carcinoma (MCC)
3.8. Thoracic Neoplasms
3.9. Meningioma
3.10. Pheochromocytoma and Paraganglioma
3.11. Head and Neck Squamous Cell Carcinoma
3.11.1. Laryngeal Squamous Cell Carcinoma
3.11.2. Nasopharyngeal Carcinoma
3.12. Breast Cancer
3.13. Ovarian Tumors
3.14. Malignant and Uveal Melanoma
Uveal Melanoma
3.15. Neuroblastoma
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Anzola, L.K.; Glaudemans, A.; Dierckx, R.; Martinez, F.A.; Moreno, S.; Signore, A. Somatostatin receptor imaging by SPECT and PET in patients with chronic inflammatory disorders: A systematic review. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2496–2513. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tang, K.; Zhang, Q.; Li, H.; Wen, Z.; Zhang, H.; Zhang, H. Somatostatin receptor-based molecular imaging and therapy for neuroendocrine tumors. Biomed. Res. Int. 2013, 2013, 102819. [Google Scholar] [CrossRef] [PubMed]
- Bronstein-Sitton, N. Somatostatin and the Somatostatin Receptors: Versatile Regulators of Biological Activity. Pathways 2006, 6. Available online: https://www.alomone.com/article/somatostatin-somatostatin-receptors-versatile-regulators-biological-activity (accessed on 20 November 2020).
- Murphy, A.; Weerakkody, Y. Octreotide Scintigraphy. Radiopedia 2010. Available online: https://radiopaedia.org/articles/octreotide-scintigraphy?lang=us (accessed on 20 November 2020).
- UCLA Health. DOTATATE PET/CT Represents Enhanced Molecular Imaging for Neuroendocrine Tumors. Available online: https://www.uclahealth.org/workfiles/clinical_updates/pharmacology-nuclear/14v1-11_DONTATE.pdf (accessed on 20 November 2020).
- National Cancer Institutes. FDA Approves New Treatment for Certain Neuroendocrine Tumors. Available online: https://www.cancer.gov/news-events/cancer-currents-blog/2018/lutathera-fda-gastrointestinal-nets (accessed on 20 November 2020).
- Dalm, V.A.; Hofland, L.J.; Mooy, C.M.; Waaijers, M.A.; van Koetsveld, P.M.; Langerak, A.W.; Staal, F.T.; van der Lely, A.J.; Lamberts, S.W.; van Hagen, M.P. Somatostatin receptors in malignant lymphomas: Targets for radiotherapy? J. Nucl. Med. 2004, 45, 8–16. [Google Scholar]
- Ferone, D.; Semino, C.; Boschetti, M.; Cascini, G.L.; Minuto, F.; Lastoria, S. Initial staging of lymphoma with octreotide and other receptor imaging agents. Semin. Nucl. Med. 2005, 35, 176–185. [Google Scholar] [CrossRef][Green Version]
- Raderer, M.; Traub, T.; Formanek, M.; Virgolini, I.; Osterreicher, C.; Fiebiger, W.; Penz, M.; Jager, U.; Pont, J.; Chott, A.; et al. Somatostatin-receptor scintigraphy for staging and follow-up of patients with extraintestinal marginal zone B-cell lymphoma of the mucosa associated lymphoid tissue (MALT)-type. Br. J. Cancer 2001, 85, 1462–1466. [Google Scholar] [CrossRef]
- Stollberg, S.; Kammerer, D.; Neubauer, E.; Schulz, S.; Simonitsch-Klupp, I.; Kiesewetter, B.; Raderer, M.; Lupp, A. Differential somatostatin and CXCR4 chemokine receptor expression in MALT-type lymphoma of gastric and extragastric origin. J. Cancer Res. Clin. Oncol. 2016, 142, 2239–2247. [Google Scholar] [CrossRef]
- Ruuska, T.; Ramirez Escalante, Y.; Vaittinen, S.; Gardberg, M.; Kiviniemi, A.; Marjamaki, P.; Kemppainen, J.; Jyrkkio, S.; Minn, H. Somatostatin receptor expression in lymphomas: A source of false diagnosis of neuroendocrine tumor at (68)Ga-DOTANOC PET/CT imaging. Acta Oncol. 2018, 57, 283–289. [Google Scholar] [CrossRef]
- Lugtenburg, P.J.; Lowenberg, B.; Valkema, R.; Oei, H.Y.; Lamberts, S.W.; Eijkemans, M.J.; van Putten, W.L.; Krenning, E.P. Somatostatin receptor scintigraphy in the initial staging of low-grade non-Hodgkin’s lymphomas. J. Nucl. Med. 2001, 42, 222–229. [Google Scholar]
- Zhao, Y.; Peng, L.; Li, X.; Zhang, Y. Expression of somatostatin and its receptor 1-5 in endometriotic tissues and cells. Exp. Ther. Med. 2018, 16, 3777–3784. [Google Scholar] [CrossRef]
- Werner, C.; Dirsch, O.; Dahmen, U.; Grimm, M.O.; Schulz, S.; Lupp, A. Evaluation of Somatostatin and CXCR4 Receptor Expression in a Large Set of Prostate Cancer Samples Using Tissue Microarrays and Well-Characterized Monoclonal Antibodies. Transl. Oncol. 2020, 13, 100801. [Google Scholar] [CrossRef] [PubMed]
- Savelli, G.; Muni, A.; Falchi, R.; Zaniboni, A.; Barbieri, R.; Valmadre, G.; Minari, C.; Casi, C.; Rossini, P. Somatostatin receptors over-expression in castration resistant prostate cancer detected by PET/CT: Preliminary report of in six patients. Ann. Trans. Med. 2015, 3, 145. [Google Scholar] [CrossRef]
- Hennigs, J.K.; Muller, J.; Adam, M.; Spin, J.M.; Riedel, E.; Graefen, M.; Bokemeyer, C.; Sauter, G.; Huland, H.; Schlomm, T.; et al. Loss of somatostatin receptor subtype 2 in prostate cancer is linked to an aggressive cancer phenotype, high tumor cell proliferation and predicts early metastatic and biochemical relapse. PLoS ONE 2014, 9, e100469. [Google Scholar] [CrossRef]
- Halmos, G.; Schally, A.V.; Sun, B.; Davis, R.; Bostwick, D.G.; Plonowski, A. High expression of somatostatin receptors and messenger ribonucleic acid for its receptor subtypes in organ-confined and locally advanced human prostate cancers. J. Clin. Endocrinol. Metab. 2000, 85, 2564–2571. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Romiti, A.; Di Rocco, R.; Milione, M.; Ruco, L.; Ziparo, V.; Zullo, A.; Duranti, E.; Sarcina, I.; Barucca, V.; D’Antonio, C.; et al. Somatostatin receptor subtype 2 A (SSTR2A) and HER2 expression in gastric adenocarcinoma. Anticancer Res. 2012, 32, 115–119. [Google Scholar] [PubMed]
- Hu, C.; Yi, C.; Hao, Z.; Cao, S.; Li, H.; Shao, X.; Zhang, J.; Qiao, T.; Fan, D. The effect of somatostatin and SSTR3 on proliferation and apoptosis of gastric cancer cells. Cancer Biol. Ther. 2004, 3, 726–730. [Google Scholar] [CrossRef][Green Version]
- Chen, W.; Ding, R.; Tang, J.; Li, H.; Chen, C.; Zhang, Y.; Zhang, Q.; Zhu, X. Knocking Out SST Gene of BGC823 Gastric Cancer Cell by CRISPR/Cas9 Enhances Migration, Invasion and Expression of SEMA5A and KLF2. Cancer Manag. Res. 2020, 12, 1313–1321. [Google Scholar] [CrossRef]
- Casini Raggi, C.; Calabro, A.; Renzi, D.; Briganti, V.; Cianchi, F.; Messerini, L.; Valanzano, R.; Cameron Smith, M.; Cortesini, C.; Tonelli, F.; et al. Quantitative evaluation of somatostatin receptor subtype 2 expression in sporadic colorectal tumor and in the corresponding normal mucosa. Clin. Cancer Res. 2002, 8, 419–427. [Google Scholar]
- Zhao, J.; Liang, Q.; Cheung, K.F.; Kang, W.; Dong, Y.; Lung, R.W.; Tong, J.H.; To, K.F.; Sung, J.J.; Yu, J. Somatostatin receptor 1, a novel EBV-associated CpG hypermethylated gene, contributes to the pathogenesis of EBV-associated gastric cancer. Br. J. Cancer 2013, 108, 2557–2564. [Google Scholar] [CrossRef]
- Ain, K.B.; Taylor, K.D.; Tofiq, S.; Venkataraman, G. Somatostatin receptor subtype expression in human thyroid and thyroid carcinoma cell lines. J. Clin. Endocrinol. Metab. 1997, 82, 1857–1862. [Google Scholar] [CrossRef]
- Ferone, D.; van Hagen, M.P.; Kwekkeboom, D.J.; van Koetsveld, P.M.; Mooy, D.M.; Lichtenauer-Kaligis, E.; Schonbrunn, A.; Colao, A.; Lamberts, S.W.; Hofland, L.J. Somatostatin receptor subtypes in human thymoma and inhibition of cell proliferation by octreotide in vitro. J. Clin. Endocrinol. Metab. 2000, 85, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, G.; Lastoria, S.; Colao, A.; Vergara, E.; Varrella, P.; Biondi, E.; Selleri, C.; Catalano, L.; Lombardi, G.; Bianco, A.R.; et al. Successful treatment of a patient with a thymoma and pure red-cell aplasia with octreotide and prednisone. N. Engl. J. Med. 1997, 336, 263–265. [Google Scholar] [CrossRef] [PubMed]
- Guida, M.; D’Alo, A.; Mangia, A.; Di Pinto, F.; Sonnessa, M.; Albano, A.; Sciacovelli, A.; Asabella, A.N.; Fucci, L. Somatostatin Receptors in Merkel-Cell Carcinoma: A Therapeutic Opportunity Using Somatostatin Analog Alone or in Association With Checkpoint Inhibitors Immunotherapy. A Case Report. Front. Oncol. 2020, 10, 1073. [Google Scholar] [CrossRef] [PubMed]
- Gardair, C.; Samimi, M.; Touze, A.; Coursaget, P.; Lorette, G.; Caille, A.; Wierzbicka, E.; Croue, A.; Avenel-Audran, M.; Aubin, F.; et al. Somatostatin Receptors 2A and 5 Are Expressed in Merkel Cell Carcinoma with No Association with Disease Severity. Neuroendocrinology 2015, 101, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Akaike, T.; Qazi, J.; Anderson, A.; Behnia, F.S.; Shinohara, M.M.; Akaike, G.; Hippe, D.S.; Thomas, H.; Takagishi, S.R.; Lachance, K.; et al. High somatostatin receptor expression and efficacy of somatostatin analogues in patients with metastatic Merkel cell carcinoma. Br. J. Dermatol. 2021, 184, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Papotti, M. Quantitation of somatostatin receptor type 2 in neuroendocrine (merkel cell) carcinoma of the skin by competitive RT-PCR. Endocr. Pathol. 1999, 10, 37–46. [Google Scholar] [CrossRef]
- Anderson, A. Therapeutic targeting of somatostatin receptors in patients with metastatic Merkel cell carcinoma: A retrospective case series. J. Clin. Oncol. 2015, 33, e20031. [Google Scholar] [CrossRef]
- Callison, J.C., Jr.; Walker, R.C.; Massion, P.P. Somatostatin Receptors in Lung Cancer: From Function to Molecular Imaging and Therapeutics. J. Lung. Cancer 2011, 10, 69–76. [Google Scholar] [CrossRef][Green Version]
- Dimitrakopoulou-Strauss, A.; Georgoulias, V.; Eisenhut, M.; Herth, F.; Koukouraki, S.; Macke, H.R.; Haberkorn, U.; Strauss, L.G. Quantitative assessment of SSTR2 expression in patients with non-small cell lung cancer using(68)Ga-DOTATOC PET and comparison with (18)F-FDG PET. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 823–830. [Google Scholar] [CrossRef]
- Muscarella, L.A.; D’Alessandro, V.; la Torre, A.; Copetti, M.; De Cata, A.; Parrella, P.; Sperandeo, M.; Pellegrini, F.; Frusciante, V.; Maiello, E.; et al. Gene expression of somatostatin receptor subtypes SSTR2a, SSTR3 and SSTR5 in peripheral blood of neuroendocrine lung cancer affected patients. Cell. Oncol. 2011, 34, 435–441. [Google Scholar] [CrossRef]
- Lapa, C.; Hanscheid, H.; Wild, V.; Pelzer, T.; Schirbel, A.; Werner, R.A.; Droll, S.; Herrmann, K.; Buck, A.K.; Luckerath, K. Somatostatin receptor expression in small cell lung cancer as a prognostic marker and a target for peptide receptor radionuclide therapy. Oncotarget 2016, 7, 20033–20040. [Google Scholar] [CrossRef] [PubMed]
- Righi, L.; Volante, M.; Tavaglione, V.; Bille, A.; Daniele, L.; Angusti, T.; Inzani, F.; Pelosi, G.; Rindi, G.; Papotti, M. Somatostatin receptor tissue distribution in lung neuroendocrine tumours: A clinicopathologic and immunohistochemical study of 218 ‘clinically aggressive’ cases. Ann. Oncol. 2010, 21, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Liu, S.V.; Subramaniam, D.S.; Torres, T.; Loda, M.; Esposito, G.; Giaccone, G. Phase I study of the (177)Lu-DOTA(0)-Tyr(3)-Octreotate (lutathera) in combination with nivolumab in patients with neuroendocrine tumors of the lung. J. Immunother. Cancer 2020, 8, e000980. [Google Scholar] [CrossRef] [PubMed]
- Schulz, S.; Pauli, S.U.; Schulz, S.; Handel, M.; Dietzmann, K.; Firsching, R.; Hollt, V. Immunohistochemical determination of five somatostatin receptors in meningioma reveals frequent overexpression of somatostatin receptor subtype sst2A. Clin. Cancer Res. 2000, 6, 1865–1874. [Google Scholar]
- Wu, W.; Zhou, Y.; Wang, Y.; Liu, L.; Lou, J.; Deng, Y.; Zhao, P.; Shao, A. Clinical Significance of Somatostatin Receptor (SSTR) 2 in Meningioma. Front. Oncol. 2020, 10, 1633. [Google Scholar] [CrossRef]
- Leijon, H.; Remes, S.; Hagstrom, J.; Louhimo, J.; Maenpaa, H.; Schalin-Jantti, C.; Miettinen, M.; Haglund, C.; Arola, J. Variable somatostatin receptor subtype expression in 151 primary pheochromocytomas and paragangliomas. Hum. Pathol. 2019, 86, 66–75. [Google Scholar] [CrossRef]
- Nassim, P.; Alsafi, A.; Vergine, M.; Ramachandran, R.; Martin, N.; Palazzo, F.; Pinato, D.; Sharma, R.; Meeran, K.; Dina, R.; et al. Expression of Somatostatin Receptors in Phaeochromocytoma and Paragangliomas. Endocr. Abstr. 2012, 28, 140. [Google Scholar]
- Kaemmerer, D.; Sanger, J.; Arsenic, R.; D’Haese, J.G.; Neumann, J.; Schmitt-Graeff, A.; Wirtz, R.M.; Schulz, S.; Lupp, A. Evaluation of somatostatin, CXCR4 chemokine and endothelin A receptor expression in a large set of paragangliomas. Oncotarget 2017, 8, 89958–89969. [Google Scholar] [CrossRef]
- Misawa, K.; Misawa, Y.; Kondo, H.; Mochizuki, D.; Imai, A.; Fukushima, H.; Uehara, T.; Kanazawa, T.; Mineta, H. Aberrant methylation inactivates somatostatin and somatostatin receptor type 1 in head and neck squamous cell carcinoma. PLoS ONE 2015, 10, e0118588. [Google Scholar] [CrossRef]
- Schartinger, V.H.; Falkeis, C.; Laimer, K.; Sprinzl, G.M.; Riechelmann, H.; Rasse, M.; Virgolini, I.; Dudás, J. Neuroendocrine differentiation in head and neck squamous cell carcinoma. J. Laryngol. Otol. 2012, 126, 1261–1270. [Google Scholar] [CrossRef]
- Shen, Z.; Chen, X.; Li, Q.; Zhou, C.; Li, J.; Ye, H.; Duan, S. SSTR2 promoter hypermethylation is associated with the risk and progression of laryngeal squamous cell carcinoma in males. Diagn. Pathol. 2016, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Loh, K.S.; Waser, B.; Tan, L.K.; Ruan, R.S.; Stauffer, E.; Reubi, J.C. Somatostatin receptors in nasopharyngeal carcinoma. Virchows Arch. 2002, 441, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Schartinger, V.H.; Dudas, J.; Url, C.; Reinold, S.; Virgolini, I.J.; Kroiss, A.; Riechelmann, H.; Uprimny, C. 68Ga-DOTA0-Tyr3-octreotide positron emission tomography in nasopharyngeal carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Kumar, U.; Grigorakis, S.I.; Watt, H.L.; Sasi, R.; Snell, L.; Watson, P.; Chaudhari, S. Somatostatin receptors in primary human breast cancer: Quantitative analysis of mRNA for subtypes 1–5 and correlation with receptor protein expression and tumor pathology. Breast Cancer Res. Treat. 2005, 92, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Tan, H.; Zhao, Y.; Zhou, Y.; Cao, L. Expression and selective activation of somatostatin receptor subtypes induces cell cycle arrest in cancer cells. Oncol. Lett. 2019, 17, 1723–1731. [Google Scholar] [CrossRef]
- Reubi, J.C.; Horisberger, U.; Klijn, J.G.; Foekens, J.A. Somatostatin receptors in differentiated ovarian tumors. Am. J. Pathol. 1991, 138, 1267–1272. [Google Scholar]
- Hall, G.H.; Turnbull, L.W.; Richmond, I.; Helboe, L.; Atkin, S.L. Localisation of somatostatin and somatostatin receptors in benign and malignant ovarian tumours. Br. J. Cancer 2002, 87, 86–90. [Google Scholar] [CrossRef][Green Version]
- Schulz, S.; Schmitt, J.; Quednow, C.; Rooessner, A.; Weise, W. Immunohistochemical detection of somatostatin receptors in human ovarian tumors. Gynecol. Oncol. 2002, 84, 235–240. [Google Scholar] [CrossRef]
- Lum, S.S.; Fletcher, W.S.; O’Dorisio, M.S.; Nance, R.W.; Pommier, R.F.; Caprara, M. Distribution and functional significance of somatostatin receptors in malignant melanoma. World J. Surg. 2001, 25, 407–412. [Google Scholar] [CrossRef]
- Harda, K.; Szabo, Z.; Szabo, E.; Olah, G.; Fodor, K.; Szasz, C.; Mehes, G.; Schally, A.V.; Halmos, G. Somatostatin Receptors as Molecular Targets in Human Uveal Melanoma. Molecules 2018, 23, 1535. [Google Scholar] [CrossRef]
- Valsecchi, M.E.; Coronel, M.; Intenzo, C.M.; Kim, S.M.; Witkiewicz, A.K.; Sato, T. Somatostatin receptor scintigraphy in patients with metastatic uveal melanoma. Melanoma Res. 2013, 23, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Alexander, N.; Marrano, P.; Thorner, P.; Naranjo, A.; Van Ryn, C.; Martinez, D.; Batra, V.; Zhang, L.; Irwin, M.S.; Baruchel, S. Prevalence and Clinical Correlations of Somatostatin Receptor-2 (SSTR2) Expression in Neuroblastoma. J. Pediatr. Hematol. Oncol. 2019, 41, 222–227. [Google Scholar] [CrossRef] [PubMed]

| Cancers | SSTR5 | SSTR4 | SSTR3 | SSTR2 | SSTR1 | |
|---|---|---|---|---|---|---|
| MALT-type lymphoma (Stollberg et al., 55, protein) [10] | Gastric and extragastric origin | 50% | 18% | 35% | 27% | 2% |
| Gastric lymphoma | Higher SSTR3, SSTR4, and SSTR5 expression than extragastric tumors. | |||||
| Extragastric tumor | SSTR5 negativity: inversely associated with patient outcome | |||||
| Endometrial cancer (Zhao et al., mRNA) [13] | Ectopic endometrium (15) | 96.7% | 50% | 53.3% | 70% | 43.3% |
| Normal endometrium (15) | 64.3% | 28.6% | 21.4% | 7.1% | 7.1% | |
| Prostate cancer | Werner et al. (276, protein) [14] | 10.5% | 0.7% | 9.1% | 0% | |
| Halmos et al. (80, mRNA) [17] | 64% | 14% | 86% | |||
| Gastric cancer | Romiti et al. (51, protein) [18] | SSTR2A 74.5% | ||||
| Hu et al. (80, mRNA and protein) [19] | 62.5% (normal gastric mucosa). | |||||
| 25% (gastric cancer) | ||||||
| Merkel cell cancer | Gardair et al. (98, protein) [27] | 44.9% | SSTR2A 59.2% | |||
| Papotti et al. (10, mRNA) [29] | 90% | |||||
| Thyroid carcinoma and thymoma (Ain et al., mRNA) [23] | Normal thyroid (5) | 60 | 0 | 100 | 60 | 20 |
| Follicular Adenoma (2) | Present | None | Dominant | Present | Dominant | |
| Papillary CA (5) | Faint | Faint | Faint | Faint | Faint | |
| Anaplastic CA (2) | Variable | Very few | Variable | Faint | Variable | |
| Normal Thymus (3) | Strongly positive | Strongly positive | Strongly positive | |||
| Thymoma (1) (loss of somatostatin production) | Strongly positive | |||||
| Lung cancer (Kim et al., 9, uptake) [36] | 71.4% | |||||
| Meningioma (Schulz et al., 40, protein) [37] | 75% | |||||
| Pheochromocytoma and paraganglioma | Leijon et al. (151, uptake) [39] | 0% | 0% | 78% | 74% | 0% |
| Parvizi et al. (77, uptake) [40] | 95% | |||||
| Kaemmerer et al. (55, uptake) [41] | 47% | 14% | 35% | SST2A 89% | 35% | |
| Head and neck cancer | Misawa et al. (36, methylation) [42] | 64% | ||||
| Schartinger et al. (78, mRNA) [43] | 82% | Rare | Rare | 54% | 69% | |
| Laryngeal cancer (Shen et al., 87, methylation) [44] | Significant association and independent prognostic factor | |||||
| Nasopharyngeal cancer (Loh et al., 12, protein) [45] | 75% | |||||
| Breast cancer | Zou et al. (160, protein) [48] | 44.4% | 71.3% | 41.9% | 34.4% | 90% |
| Kumar et al. (98, mRNA) [47] | 68% | 68% | 89% | 79% | 84% | |
| Ovarian cancer | Hall et al. (63, protein) [50] | 71% | 29% | 77% | 76% | |
| Schulz et al. (47, protein) [51] | 21% | 17% | 42% | 28% | 19% | |
| Malignant melanoma (Lum et al., 17, mRNA) [52] | 9% | 57% | 61% | 83% | 96% | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Priyadarshini, S.; Allison, D.B.; Chauhan, A. Comprehensive Assessment of Somatostatin Receptors in Various Neoplasms: A Systematic Review. Pharmaceutics 2022, 14, 1394. https://doi.org/10.3390/pharmaceutics14071394
Priyadarshini S, Allison DB, Chauhan A. Comprehensive Assessment of Somatostatin Receptors in Various Neoplasms: A Systematic Review. Pharmaceutics. 2022; 14(7):1394. https://doi.org/10.3390/pharmaceutics14071394
Chicago/Turabian StylePriyadarshini, Shista, Derek B. Allison, and Aman Chauhan. 2022. "Comprehensive Assessment of Somatostatin Receptors in Various Neoplasms: A Systematic Review" Pharmaceutics 14, no. 7: 1394. https://doi.org/10.3390/pharmaceutics14071394
APA StylePriyadarshini, S., Allison, D. B., & Chauhan, A. (2022). Comprehensive Assessment of Somatostatin Receptors in Various Neoplasms: A Systematic Review. Pharmaceutics, 14(7), 1394. https://doi.org/10.3390/pharmaceutics14071394






