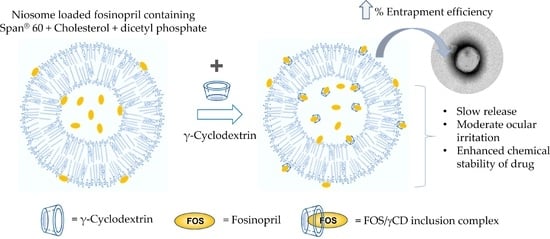
Physicochemical and Stability Evaluation of Topical Niosomal Encapsulating Fosinopril/γ-Cyclodextrin Complex for Ocular Delivery
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Niosomal Formulations Containing FOS
2.3. Physicochemical and Chemical Characterizations
2.3.1. Osmolality, pH and Viscosity Determination
2.3.2. Particle Size, Size Distribution, and Zeta Potential
2.3.3. Determining Drug Content and Entrapment Efficiency (EE)
2.3.4. Transmission Electron Microscopy (TEM) Analysis
2.4. In Vitro Release Study
2.5. Ex Vivo Permeation Study
2.6. Cell Viability and Short Time Exposure (STE) Test
2.7. Physical and Chemical Stability Studies
2.8. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical and Chemical Characterizations of Niosomal Formulations Containing FOS
3.1.1. Osmolality, pH and Viscosity
3.1.2. Particle Size, Size Distribution and Zeta Potential
3.1.3. %EE of FOS-Loaded Niosomal Formulations
3.1.4. TEM Analysis
3.2. In Vitro Release Study
3.3. Ex Vivo Permeation Study
3.4. Cell Viability and STE Test
3.5. Physical and Chemical Stability Studies of FOS
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sugrue, M.F. The pharmacology of antiglaucoma drugs. Pharmacol. Ther. 1989, 43, 91–138. [Google Scholar] [CrossRef]
- Alward, W.L.M. Medical management of glaucoma. N. Engl. J. Med. 1998, 339, 1298–1307. [Google Scholar] [CrossRef] [PubMed]
- Quigley, H.A.; Broman, A.T. The number of people with glaucoma worldwide in 2010 and 2020. Br. J. Ophthalmol. 2006, 90, 262–267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tham, Y.C.; Li, X.; Wong, T.Y.; Quigley, H.A.; Aung, T.; Cheng, C.Y. Global prevalence of glaucoma and projections of glaucoma burden through 2040: A systematic review and meta-analysis. Ophthalmology 2014, 121, 2081–2090. [Google Scholar] [CrossRef]
- Kwon, Y.H.; Fingert, J.H.; Kuehn, M.H.; Alward, W.L. Primary open-angle glaucoma. N. Engl. J. Med. 2009, 360, 1113–1124. [Google Scholar] [CrossRef] [Green Version]
- Costagliola, C.; Di Benedetto, R.; De Caprio, L.; Verde, R.; Mastropasqua, L. Effect of oral captopril (SQ 14225) on intraocular pressure in man. Eur. J. Ophthalmol. 1995, 5, 19–25. [Google Scholar] [CrossRef]
- Mehta, A.; Iyer, L.; Parmar, S.; Shah, G.; Goyal, R. Oculohypotensive effect of perindopril in acute and chronic models of glaucoma in rabbits. Can. J. Physiol. Pharmacol. 2010, 88, 595–600. [Google Scholar] [CrossRef]
- Loftsson, T.; Thorisdóttir, S.; Fridriksdóttir, H.; Stefánsson, E. Enalaprilat and enalapril maleate eyedrops lower intraocular pressure in rabbits. Acta Ophthalmol. 2010, 88, 337–341. [Google Scholar] [CrossRef]
- Lotti, V.J.; Pawlowski, N. Prostaglandins mediate the ocular hypotensive action of the angiotensin converting enzyme inhibitor MK-422 (enalaprilat) in African green monkeys. J. Ocul. Pharmacol. Ther. 1990, 6, 1–7. [Google Scholar] [CrossRef]
- Rachmani, R.; Lidar, M.; Levy, Z.; Ravid, M. Effect of enalapril on the incidence of retinopathy in normotensive patients with type 2 diabetes. Eur. J. Intern. Med. 2000, 11, 48–50. [Google Scholar] [CrossRef]
- Manschot, S.M.; Gispen, W.H.; Kappelle, L.J.; Biessels, G.J. Nerve conduction velocity and evoked potential latencies in streptozotocin-diabetic rats: Effects of treatment with an angiotensin converting enzyme inhibitor. Diabetes/Metab. Res. Rev. 2003, 19, 469–477. [Google Scholar] [CrossRef]
- Choudhary, R.; Kapoor, M.S.; Singh, A.; Bodakhe, S.H. Therapeutic targets of renin-angiotensin system in ocular disorders. J. Curr. Ophthalmol. 2017, 29, 7–16. [Google Scholar] [CrossRef] [Green Version]
- Narayanam, M.; Singh, S. Characterization of stress degradation products of fosinopril by using LC-MS/TOF, MSn and on-line H/D exchange. J. Pharm. Biomed. Anal. 2014, 92, 135–143. [Google Scholar] [CrossRef]
- Hnin, H.M.; Stefánsson, E.; Loftsson, T.; Rungrotmongkol, T.; Jansook, P. Angiotensin converting enzyme inhibitors/cyclodextrin inclusion complexes: Solution and solid-state characterizations and their thermal stability. J. Incl. Phenom. Macrocycl. Chem. 2022, 102, 347–358. [Google Scholar] [CrossRef]
- Boyd, B.J. Past and future evolution in colloidal drug delivery systems. Expert Opin. Drug Deliv. 2008, 5, 69–85. [Google Scholar] [CrossRef]
- Moghassemi, S.; Hadjizadeh, A. Nano-niosomes as nanoscale drug delivery systems: An illustrated review. J. Control. Release 2014, 185, 22–36. [Google Scholar] [CrossRef]
- Uchegbu, I.F.; Vyas, S.P. Non-ionic surfactant based vesicles (niosomes) in drug delivery. Int. J. Pharm. 1998, 172, 33–70. [Google Scholar] [CrossRef]
- Carafa, M.; Santucci, E.; Alhaique, F.; Coviello, T.; Murtas, E.; Riccieri, F.M.; Lucania, G.; Torrisi, M.R. Preparation and properties of new unilamellar non-ionic/ionic surfactant vesicles. Int. J. Pharm. 1998, 160, 51–59. [Google Scholar] [CrossRef]
- Vyas, S.; Mysore, N.; Jaitely, V.; Venkatesan, N. Discoidal niosome based controlled ocular delivery of timolol maleate. Die Pharm. 1998, 53, 466–469. [Google Scholar]
- Abdelbary, G.; El-gendy, N. Niosome-encapsulated gentamicin for ophthalmic controlled delivery. AAPS PharmSciTech 2008, 9, 740–747. [Google Scholar] [CrossRef]
- Saettone, M.; Perini, G.; Carafa, M.; Santucci, E.; Alhaique, F. Non-ionic surfactant vesicles as ophthalmic carriers for cyclopentolate. A preliminary evaluation. STP Pharma Sci. 1996, 6, 94–98. [Google Scholar]
- Guinedi, A.S.; Mortada, N.D.; Mansour, S.; Hathout, R.M. Preparation and evaluation of reverse-phase evaporation and multilamellar niosomes as ophthalmic carriers of acetazolamide. Int. J. Pharm. 2005, 306, 71–82. [Google Scholar] [CrossRef]
- Abdelkader, H.; Wu, Z.; Al-Kassas, R.; Alany, R.G. Niosomes and discomes for ocular delivery of naltrexone hydrochloride: Morphological, rheological, spreading properties and photo-protective effects. Int. J. Pharm. 2012, 433, 142–148. [Google Scholar] [CrossRef]
- Kaur, I.P.; Garg, A.; Singla, A.K.; Aggarwal, D. Vesicular systems in ocular drug delivery: An overview. Int. J. Pharm. 2004, 269, 1–14. [Google Scholar] [CrossRef]
- Jain, S.; Jain, V.; Mahajan, S. Lipid based vesicular drug delivery systems. Adv. Pharm. 2014, 2014, 574673. [Google Scholar] [CrossRef] [Green Version]
- Kaur, I.P.; Smitha, R. Penetration enhancers and ocular bioadhesives: Two new avenues for ophthalmic drug delivery. Drug Dev. Ind. Pharm. 2002, 28, 353–369. [Google Scholar] [CrossRef]
- Manconi, M.; Manca, M.L.; Valenti, D.; Escribano, E.; Hillaireau, H.; Fadda, A.M.; Fattal, E. Chitosan and hyaluronan coated liposomes for pulmonary administration of curcumin. Int. J. Pharm. 2017, 525, 203–210. [Google Scholar] [CrossRef]
- Asasutjarit, R.; Managit, C.; Phanaksri, T.; Treesuppharat, W.; Fuongfuchat, A. Formulation development and in vitro evaluation of transferrin-conjugated liposomes as a carrier of ganciclovir targeting the retina. Int. J. Pharm. 2020, 577, 119084. [Google Scholar] [CrossRef]
- Asasutjarit, R.; Theerachayanan, T.; Kewsuwan, P.; Veeranondha, S.; Fuongfuchat, A.; Ritthidej, G.C. Gamma sterilization of diclofenac sodium loaded- N-trimethyl chitosan nanoparticles for ophthalmic use. Carbohydr. Polym. 2017, 157, 603–612. [Google Scholar] [CrossRef]
- Takahashi, Y.; Koike, M.; Honda, H.; Ito, Y.; Sakaguchi, H.; Suzuki, H.; Nishiyama, N. Development of the short time exposure (STE) test: An in vitro eye irritation test using SIRC cells. Toxicol. Vitr. 2008, 22, 760–770. [Google Scholar] [CrossRef]
- ICH. Stability Testing of New Drug Substances and Products (Q1AR2); European Medicines Agency: Amsterdam, The Netherlands, 2003. [Google Scholar]
- Mathis, G.A. Clinical Ophthalmic Pharmacology and Therapeutics: Ocular Drug Delivery. Veterinary Ophthalmology; Lippincott Williams & Wilkins: Orlando, FL, USA, 1999; pp. 291–297. [Google Scholar]
- Yasin, M.N.; Hussain, S.; Malik, F.; Hameed, A.; Sultan, T.; Qureshi, F.; Riaz, H.; Perveen, G.; Wajid, A. Preparation and characterization of chloramphenicol niosomes and comparison with chloramphenicol eye drops (0.5% w/v) in experimental conjunctivitis in albino rabbits. Pak. J. Pharm. Sci. 2012, 25, 117–121. [Google Scholar] [PubMed]
- Frisch, D.; Eyring, H.; Kincaid, J.F. Pressure and temperature effects on the viscosity of liquids. J. Appl. Phys. 1940, 11, 75–80. [Google Scholar] [CrossRef]
- Kramer, I.; Haber, M.; Duis, A. Formulation requirements for the ophthalmic use of antiseptics. Dev. Ophthalmol. 2002, 33, 85–116. [Google Scholar]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M.R. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suwakul, W.; Ongpipattanakul, B.; Vardhanabhuti, N. Preparation and characterization of propylthiouracil niosomes. J. Liposome Res. 2006, 16, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, T.; Sternberg, B.; Florence, A.T. Preparation and properties of vesicles (niosomes) of sorbitan monoesters (Span 20, 40, 60 and 80) and a sorbitan triester (Span 85). Int. J. Pharm. 1994, 105, 1–6. [Google Scholar] [CrossRef]
- Ruckmani, K.; Jayakar, B.; Ghosal, S.K. Nonionic surfactant vesicles (niosomes) of cytarabine hydrochloride for effective treatment of leukemias: Encapsulation, storage, and In vitro release. Drug Dev. Ind. Pharm. 2000, 26, 217–222. [Google Scholar] [CrossRef]
- Khazaeli, P.; Pardakhty, A.; Shoorabi, H. Caffeine-loaded niosomes: Characterization and in vitro release studies. Drug Deliv. 2007, 14, 447–452. [Google Scholar] [CrossRef]
- Manconi, M.; Sinico, C.; Valenti, D.; Loy, G.; Fadda, A.M. Niosomes as carriers for tretinoin. I. Preparation and properties. Int. J. Pharm. 2002, 234, 237–248. [Google Scholar] [CrossRef]
- Tabbakhian, M.; Tavakoli, N.; Jaafari, M.R.; Daneshamouz, S. Enhancement of follicular delivery of finasteride by liposomes and niosomes: 1. In vitro permeation and in vivo deposition studies using hamster flank and ear models. Int. J. Pharm. 2006, 323, 1–10. [Google Scholar] [CrossRef]
- Hasan, A.A. Design and in vitro characterization of small unilamellar niosomes as ophthalmic carrier of dorzolamide hydrochloride. Pharm. Dev. Tech. 2014, 19, 748–754. [Google Scholar] [CrossRef]
- Abdelkader, H.; Ismail, S.; Kamal, A.; Alany, R.G. Design and evaluation of controlled-release niosomes and discomes for naltrexone hydrochloride ocular delivery. J. Pharm. Sci. 2011, 100, 1833–1846. [Google Scholar] [CrossRef]
- Valente, A.J.; Söderman, O. The formation of host–guest complexes between surfactants and cyclodextrins. Adv. Colloid Interface Sci. 2014, 205, 156–176. [Google Scholar] [CrossRef]
- Tsianou, M.; Fajalia, A.I. Cyclodextrins and surfactants in aqueous solution above the critical micelle concentration: Where are the cyclodextrins located? Langmuir 2014, 30, 13754–13764. [Google Scholar] [CrossRef]
- Machado, N.D.; Silva, O.F.; de Rossi, R.H.; Fernández, M.A. Cyclodextrin modified niosomes to encapsulate hydrophilic compounds. RSC Adv. 2018, 8, 29909–29916. [Google Scholar] [CrossRef] [Green Version]
- Zubairu, Y.; Negi, L.M.; Iqbal, Z.; Talegaonkar, S. Design and development of novel bioadhesive niosomal formulation for the transcorneal delivery of anti-infective agent: In-vitro and ex-vivo investigations. Asian J. Pharm. Sci. 2015, 10, 322–330. [Google Scholar] [CrossRef] [Green Version]
- Junyaprasert, V.B.; Teeranachaideekul, V.; Supaperm, T. Effect of charged and non-ionic membrane additives on physicochemical properties and stability of niosomes. AAPS PharmSciTech 2008, 9, 851. [Google Scholar] [CrossRef]
- Silva, O.F.; Correa, N.M.; Silber, J.J.; de Rossi, R.H.; Fernández, M.A. Supramolecular assemblies obtained by mixing different cyclodextrins and AOT or BHDC reverse micelles. Langmuir 2014, 30, 3354–3362. [Google Scholar] [CrossRef]
- Zhou, C.; Cheng, X.; Zhao, Q.; Yan, Y.; Wang, J.; Huang, J. Self-assembly of nonionic surfactant tween 20@2β-CD inclusion complexes in dilute solution. Langmuir 2013, 29, 13175–13182. [Google Scholar] [CrossRef]
- Essa, E.A. Effect of formulation and processing variables on the particle size of sorbitan monopalmitate niosomes. Asian J. Pharm. 2010, 4, 227–233. [Google Scholar] [CrossRef]
- Balakrishnan, P.; Shanmugam, S.; Lee, W.S.; Lee, W.M.; Kim, J.O.; Oh, D.H.; Kim, D.-D.; Kim, J.S.; Yoo, B.K.; Choi, H.-G.; et al. Formulation and in vitro assessment of minoxidil niosomes for enhanced skin delivery. Int. J. Pharm. 2009, 377, 1–8. [Google Scholar] [CrossRef]
- Jaehnig, F.; Harlos, K.; Vogel, H.; Eibl, H. Electrostatic interactions at charged lipid membranes. Electrostatically induced tilt. Biochemistry 1979, 18, 1459–1468. [Google Scholar] [CrossRef]
- Chi, L.; Wu, D.; Li, Z.; Zhang, M.; Liu, H.; Wang, C.; Gui, S.; Geng, M.; Li, H.; Zhang, J. Modified release and improved stability of unstable BCS II drug by using cyclodextrin complex as carrier to remotely load drug into niosomes. Mol. Pharm. 2016, 13, 113–124. [Google Scholar] [CrossRef]
- Marianecci, C.; Rinaldi, F.; Esposito, S.; Di Marzio, L.; Carafa, M. Niosomes encapsulating ibuprofen-cyclodextrin complexes: Preparation and characterization. Curr. Drug Targets 2013, 14, 1070–1078. [Google Scholar] [CrossRef]
- Paul, B.K.; Ghosh, N.; Mondal, R.; Mukherjee, S. Contrasting effects of salt and temperature on niosome-bound norharmane: Direct evidence for positive heat capacity change in the niosome: β-cyclodextrin interaction. J. Phys. Chem. B 2016, 120, 4091–4101. [Google Scholar] [CrossRef]
- Sheena, I.; Singh, U.; Aithal, K.; Udupa, N. Pilocarpine β-cyclodextrin complexation and niosomal entrapment. Pharm. Sci. 1997, 3, 383–386. [Google Scholar]
- Oommen, E.; Shenoy, B.D.; Udupa, N.; Kamath, R.; Devi, P.U. Antitumour efficacy of cyclodextrin-complexed and niosome-encapsulated plumbagin in mice bearing melanoma B16F1. Pharm. Pharmacol. Commun. 1999, 5, 281–285. [Google Scholar] [CrossRef]
- D’souza, S.; Ray, J.; Pandey, S.; Udupa, N. Absorption of ciprofloxacin and norfloxacin when administered as niosome-encapsulated inclusion complexes. J. Pharm. Pharmacol. 1997, 49, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Oommen, E.; Tiwari, S.B.; Udupa, N.; Kamath, R.; Devi, P.U. Niosome entrapped β-cyclodextrin methotrexate complex as a drug delivery system. Indian J. Pharmacol. 1999, 31, 279–284. [Google Scholar]
- Hao, Y.-M.; Li, K. Entrapment and release difference resulting from hydrogen bonding interactions in niosome. Int. J. Pharm. 2011, 403, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Wax, M.B.; Camras, C.B.; Fiscella, R.G.; Girkin, C.; Singh, K.; Weinreb, R.N. Emerging perspectives in glaucoma: Optimizing 24-hour control of intraocular pressure. Am. J. Ophthalmol. 2002, 133, S1–S10. [Google Scholar] [CrossRef]
- Barar, J.; Javadzadeh, A.R.; Omidi, Y. Ocular novel drug delivery: Impacts of membranes and barriers. Expert Opin. Drug Deliv. 2008, 5, 567–581. [Google Scholar] [CrossRef]
- Loftsson, T.; Sigurđsson, H.H.; Konráđsdóttir, F.; Gísladóttir, S.; Jansook, P.; Stefánsson, E. Topical drug delivery to the posterior segment of the eye: Anatomical and physiological considerations. Die Pharm. 2008, 63, 171–179. [Google Scholar] [CrossRef]
- Hämäläinen, K.M.; Kananen, K.; Auriola, S.; Kontturi, K.; Urtti, A. Characterization of paracellular and aqueous penetration routes in cornea, conjunctiva, and sclera. Investig. Ophthalmol. Vis. Sci. 1997, 38, 627–634. [Google Scholar]
- Loch, C.; Zakelj, S.; Kristl, A.; Nagel, S.; Guthoff, R.; Weitschies, W.; Seidlitz, A. Determination of permeability coefficients of ophthalmic drugs through different layers of porcine, rabbit and bovine eyes. Eur. J. Pharm. Sci. 2012, 47, 131–138. [Google Scholar] [CrossRef]
- Ahmed, I.; Patton, T.F. Importance of the noncorneal absorption route in topical ophthalmic drug delivery. Investig. Ophthalmol. Vis. Sci. 1985, 26, 584–587. [Google Scholar]
- Gharib, R.; Greige-Gerges, H.; Fourmentin, S.; Charcosset, C.; Auezova, L. Liposomes incorporating cyclodextrin–drug inclusion complexes: Current state of knowledge. Carbohydr. Polym. 2015, 129, 175–186. [Google Scholar] [CrossRef]
- Bates, T.R.; Nightingale, C.H.; Dixon, E. Kinetics of hydrolysis of polyoxyethylene (20) sorbitan fatty acid ester surfactants. J. Pharm. Pharmacol. 1973, 25, 470–477. [Google Scholar] [CrossRef]
- Kopermsub, P.; Mayen, V.; Warin, C. Potential use of niosomes for encapsulation of nisin and EDTA and their antibacterial activity enhancement. Food Res. Int. 2011, 44, 605–612. [Google Scholar] [CrossRef]
- Khan, M.I.; Madni, A.; Peltonen, L. Development and in-vitro characterization of sorbitan monolaurate and poloxamer 184 based niosomes for oral delivery of diacerein. Eur. J. Pharm. Sci. 2016, 95, 88–95. [Google Scholar] [CrossRef]


 ) Sp-DCP and (
) Sp-DCP and (  ) Sp-DCP+γCD, and, (
) Sp-DCP+γCD, and, (  ) blank Sp-DCP and (
) blank Sp-DCP and (  ) blank Sp-DCP+γCD, at various concentrations in the SIRC cells (n = 4, mean ± SD).
) blank Sp-DCP+γCD, at various concentrations in the SIRC cells (n = 4, mean ± SD).
 ) Sp-DCP and (
) Sp-DCP and (  ) Sp-DCP+γCD, and, (
) Sp-DCP+γCD, and, (  ) blank Sp-DCP and (
) blank Sp-DCP and (  ) blank Sp-DCP+γCD, at various concentrations in the SIRC cells (n = 4, mean ± SD).
) blank Sp-DCP+γCD, at various concentrations in the SIRC cells (n = 4, mean ± SD).

| Formulation a | Span® 60-Niosome | Brij® 76-Niosome | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sp-SC24 | Sp-DCP | Sp-STA | Sp-SC24+γCD | Sp-DCP+γCD | Sp-STA+γCD | Br-SC24 | Br-DCP | Br-STA | Br-SC24+γCD | Br-DCP+γCD | Br-STA+γCD | |
| Ingredients in organic phase (μM) b | ||||||||||||
| Span® 60 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | - | - | - | - | - | - |
| Brij® 76 | - | - | - | - | - | - | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 |
| Cholesterol | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 | 47.5 |
| SC24 | 5 | - | - | 5 | - | - | 5 | - | - | 5 | - | - |
| DCP | - | 5 | - | - | 5 | - | - | 5 | - | - | 5 | - |
| STA | - | - | 5 | - | - | 5 | - | - | 5 | - | - | 5 |
| Ingredients in aqueous phase (% w/v) c | ||||||||||||
| FOS | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| γCD | - | - | - | 5 | 5 | 5 | - | - | - | 5 | 5 | 5 |
| Formulation | pH | Osmolality (mOsm/kg) | Viscosity (mPa.s) | |
|---|---|---|---|---|
| 25 ± 1 °C | 34 ± 1 °C | |||
| Span® 60-Niosome | ||||
| Sp-SC24 | 7.02 ± 0.05 | 358 ± 5 | 1.48 ± 0.01 | 1.18 ± 0.01 |
| Sp-DCP | 6.73 ± 0.04 | 364 ± 6 | 1.81 ± 0.02 | 1.30 ± 0.01 |
| Sp-STA | 7.26 ± 0.03 | 366 ± 8 | 1.38 ± 0.02 | 1.12 ± 0.01 |
| Sp-SC24+γCD | 6.83 ± 0.03 | 372 ± 5 | 1.76 ± 0.02 | 1.50 ± 0.01 |
| Sp-DCP+γCD | 6.70 ± 0.03 | 374 ± 6 | 1.98 ± 0.02 | 1.72 ± 0.02 |
| Sp-STA+γCD | 6.75 ± 0.01 | 382 ± 5 | 1.75 ± 0.01 | 1.52 ± 0.01 |
| Brij® 76-Niosome | ||||
| Br-SC24 | 6.91 ± 0.01 | 346 ± 6 | 1.43 ± 0.01 | 1.21 ± 0.01 |
| Br-DCP | 6.95 ± 0.01 | 354 ± 8 | 1.64 ± 0.02 | 1.34 ± 0.01 |
| Br-STA | 7.22 ± 0.03 | 359 ± 10 | 1.41 ± 0.01 | 1.15 ± 0.01 |
| Br-SC24+γCD | 6.87 ± 0.02 | 364 ± 8 | 1.68 ± 0.02 | 1.34 ± 0.02 |
| Br-DCP+γCD | 6.78 ± 0.08 | 378 ± 3 | 1.86 ± 0.01 | 1.56 ± 0.01 |
| Br-STA+γCD | 6.86 ± 0.05 | 379 ± 9 | 1.65 ± 0.02 | 1.38 ± 0.01 |
| Formulation | Z-Average (d.nm) | Size Distribution (PDI) | Zeta Potential (mV) | %EE |
|---|---|---|---|---|
| Sp-SC24 | 245.1 ± 5.02 | 0.46 ± 0.03 | −32.70 ± 1.64 | 21.34 ± 0.42 |
| Sp-DCP | 262.4 ± 5.00 | 0.45 ± 0.01 | −37.70 ± 1.15 | 28.68 ± 0.77 |
| Sp-STA | 250.4 ± 6.31 | 0.35 ± 0.03 | −15.43 ± 1.46 | 9.20 ± 0.30 |
| Sp-SC24+γCD | 198.0 ± 4.50 | 0.52 ± 0.01 | −20.27 ± 0.67 | 25.99 ± 0.78 |
| Sp-DCP+γCD | 246.8 ± 3.71 | 0.42 ± 0.01 | −27.17 ± 1.63 | 34.43 ± 0.80 |
| Sp-STA+γCD | 229.1 ± 5.16 | 0.36 ± 0.06 | −13.40 ± 1.91 | 11.30 ± 0.85 |
| Br-SC24 | 257.2 ± 4.29 | 0.32 ± 0.01 | −24.30 ± 2.01 | 10.70 ± 0.27 |
| Br-DCP | 212.0 ± 0.72 | 0.36 ± 0.03 | −34.97 ± 0.35 | 12.94 ± 0.57 |
| Br-STA | 214.8 ± 4.01 | 0.37 ± 0.02 | −7.41 ± 0.40 | 7.73 ± 0.97 |
| Br-SC24+γCD | 246.0 ± 0.96 | 0.11 ± 0.02 | −21.20 ± 1.04 | 12.58 ± 0.85 |
| Br-DCP+γCD | 200.0 ± 1.87 | 0.32 ± 0.01 | −23.73 ± 1.97 | 14.02 ± 0.10 |
| Br-STA+γCD | 211.6 ± 1.52 | 0.34 ± 0.05 | −6.94 ± 0.43 | 8.09 ± 0.80 |
| Formulation | Cornea | Sclera | ||
|---|---|---|---|---|
| Flux ± S.D. (μgh−1 cm−2) | Papp ± S.D. (×10−6 cms−1) | Flux ± S.D. (μgh−1 cm−2) | Papp ± S.D. (×10−6 cms−1) | |
| Sp-DCP | 31.086 ± 6.32 | 0.920 ± 0.18 | 40.066 ± 40.35 | 1.155 ± 0.11 |
| Sp-DCP+γCD | 22.843 ± 7.95 | 0.635 ± 0.21 | 33.092 ± 2.38 | 0.927 ± 0.08 |
| FOS/γCD complex | 62.794 ± 6.23 a | 1.870 ± 0.18 a | 86.762 ± 5.25 a | 2.583 ± 0.16 a |
| Concentration of the Test Samples | Test Samples | %CV of the SIRC Cells | Criteria for Scoring | Obtained Scores |
|---|---|---|---|---|
| (I) 5% | (1) Blank Sp-DCP | 67 ± 5 | If CV >70%: scored 0 | 1 |
| (2) Blank Sp-DCP + γCD | 63 ± 3 | If CV ≤ 70%: scored 1 | 1 | |
| (3) Sp-DCP | 52 ± 4 | 1 | ||
| (4) Sp-DCP + γCD | 47 ± 4 | 1 | ||
| (II) 0.05% | (1) Blank Sp-DCP | 87 ± 6 | If CV >70%: scored 1If CV ≤ 70%: scored 2 | 1 |
| (2) Blank Sp-DCP + γCD | 85 ± 4 | 1 | ||
| (3) Sp-DCP | 83 ± 4 | 1 | ||
| (4) Sp-DCP + γCD | 81 ± 2 | 1 | ||
| Total score (I and II) | (1) Blank Sp-DCP | 2 | ||
| (2) Blank Sp-DCP+γCD | 2 | |||
| (3) Sp-DCP | 2 | |||
| (4) Sp-DCP+γCD | 2 | |||
| Time (Month) | Formulations | ||
|---|---|---|---|
| Sp-DCP | Sp-DCP+γCD | FOS/γCD Complex | |
| 5 ± 3 °C | |||
| 1 Month | 97.95 ± 0.70 | 98.44 ± 0.64 | 81.09 ± 0.92 |
| 3 Months | 93.72 ± 0.73 | 95.21 ± 0.39 | 73.84 ± 0.68 |
| 6 Months | 88.33 ± 0.54 | 92.75 ± 0.83 | 51.10 ± 1.18 |
| 30 ± 2 °C (65 ± 5% RH) | |||
| 1 Month | 93.32 ± 0.53 | 95.13 ± 0.86 | 28.72 ± 0.30 |
| 3 Months | 83.40 ± 0.78 | 87.37 ± 0.57 | 20.94 ± 0.73 |
| 6 Months | 17.17 ± 0.59 | 23.67 ± 0.57 | 8.49 ± 0.70 |
| 40 ± 2 °C (75 ± 5% RH) | |||
| 1 Month | 46.09 ± 0.88 | 56.34 ± 0.82 | 19.95 ± 0.60 |
| 3 Months | 27.88 ± 0.71 | 36.70 ± 1.08 | 12.26 ± 0.36 |
| 6 Months | 7.75 ± 0.83 | 10.68 ± 1.06 | 3.59 ± 0.70 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hnin, H.M.; Stefánsson, E.; Loftsson, T.; Asasutjarit, R.; Charnvanich, D.; Jansook, P. Physicochemical and Stability Evaluation of Topical Niosomal Encapsulating Fosinopril/γ-Cyclodextrin Complex for Ocular Delivery. Pharmaceutics 2022, 14, 1147. https://doi.org/10.3390/pharmaceutics14061147
Hnin HM, Stefánsson E, Loftsson T, Asasutjarit R, Charnvanich D, Jansook P. Physicochemical and Stability Evaluation of Topical Niosomal Encapsulating Fosinopril/γ-Cyclodextrin Complex for Ocular Delivery. Pharmaceutics. 2022; 14(6):1147. https://doi.org/10.3390/pharmaceutics14061147
Chicago/Turabian StyleHnin, Hay Marn, Einar Stefánsson, Thorsteinn Loftsson, Rathapon Asasutjarit, Dusadee Charnvanich, and Phatsawee Jansook. 2022. "Physicochemical and Stability Evaluation of Topical Niosomal Encapsulating Fosinopril/γ-Cyclodextrin Complex for Ocular Delivery" Pharmaceutics 14, no. 6: 1147. https://doi.org/10.3390/pharmaceutics14061147
APA StyleHnin, H. M., Stefánsson, E., Loftsson, T., Asasutjarit, R., Charnvanich, D., & Jansook, P. (2022). Physicochemical and Stability Evaluation of Topical Niosomal Encapsulating Fosinopril/γ-Cyclodextrin Complex for Ocular Delivery. Pharmaceutics, 14(6), 1147. https://doi.org/10.3390/pharmaceutics14061147