Physics of Brain Cancer: Multiscale Alterations of Glioblastoma Cells under Extracellular Matrix Stiffening
Abstract
1. Introduction: Glioblastoma
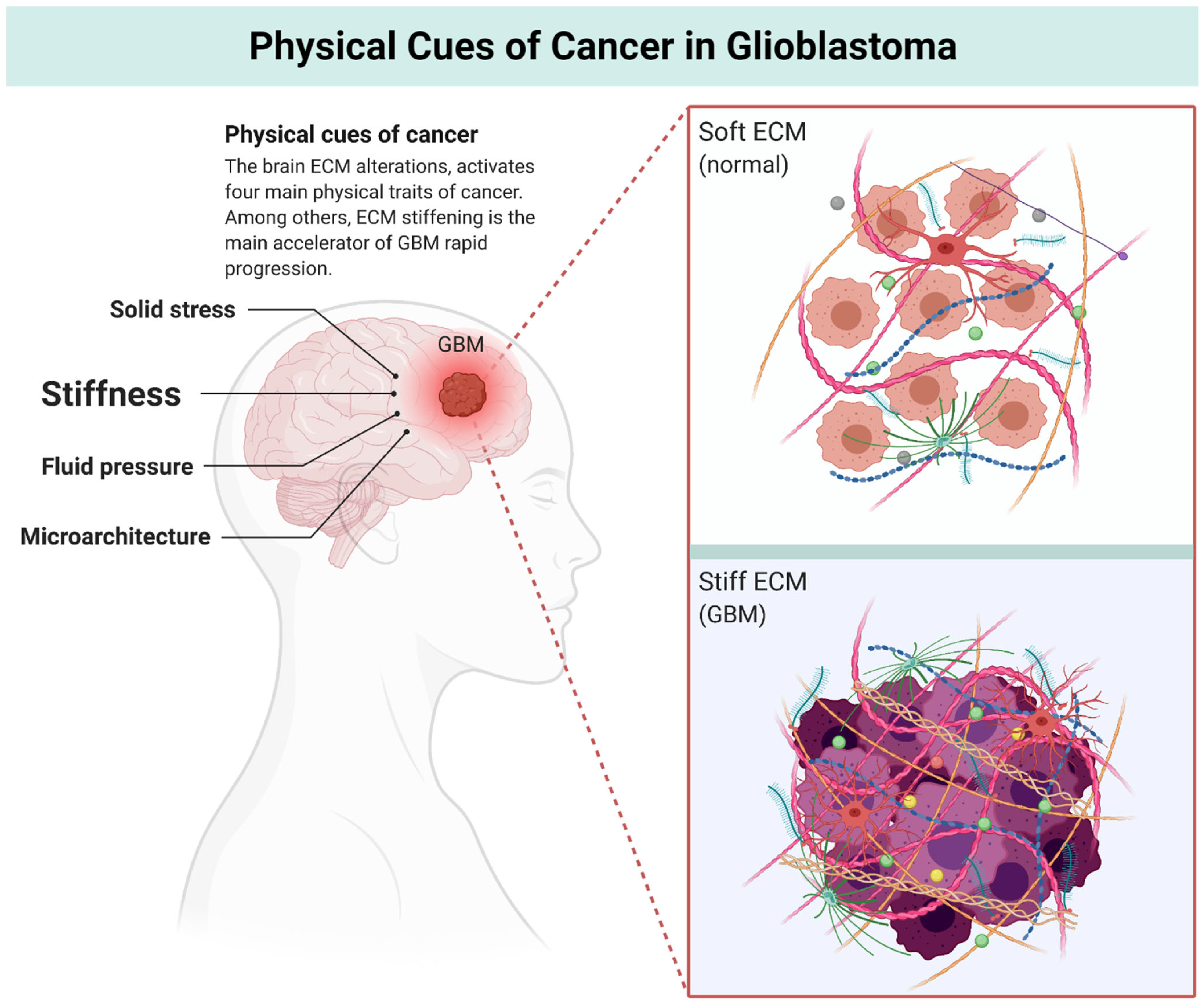
2. Physics of Cancer
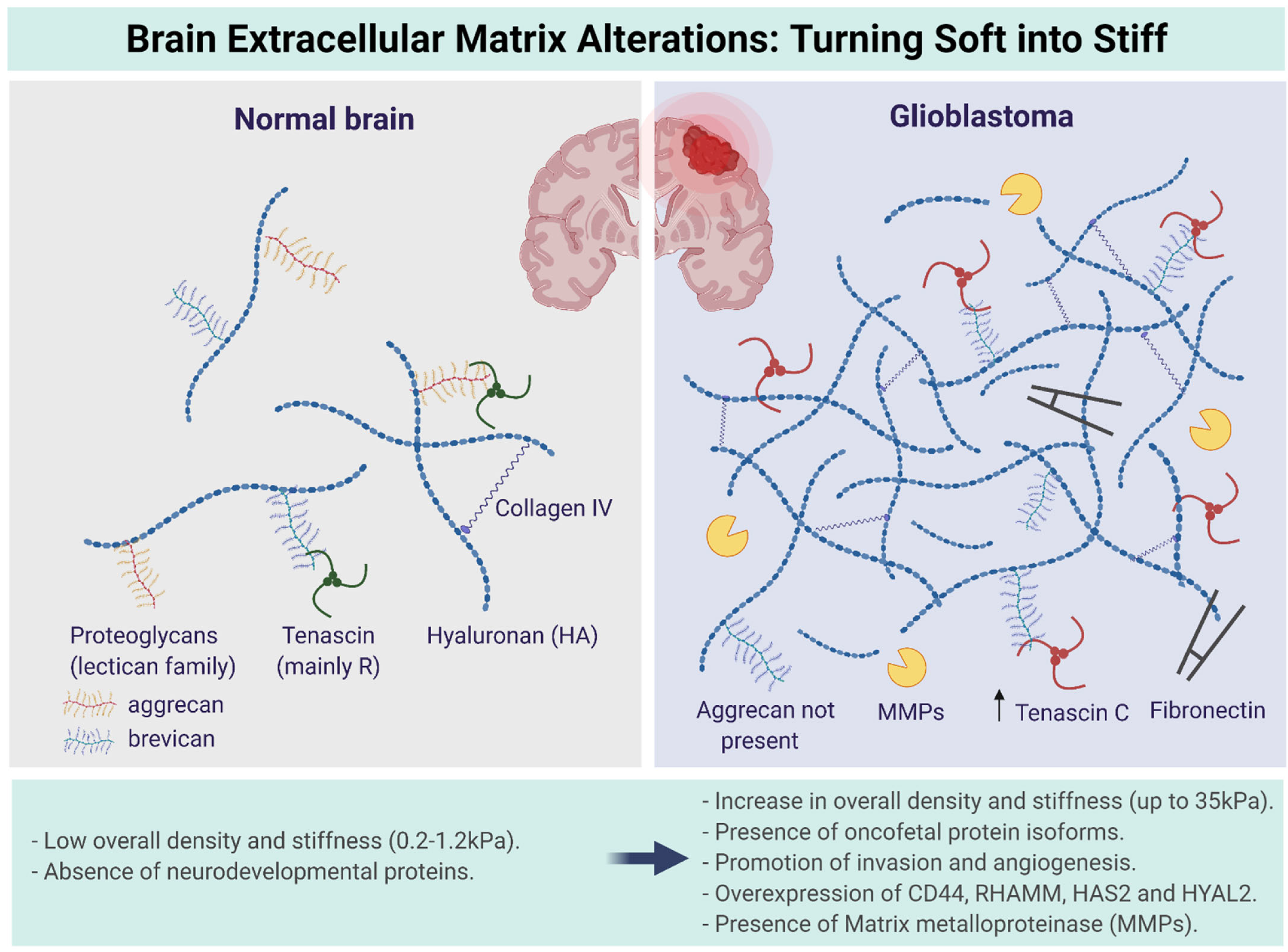
3. Multiscale Alterations of the Brain ECM during GBM Progression: Turning Soft into Stiff
- (i)
- GAGs are sugar molecules linked together by amino acids, which makes them repeating disaccharide units in which one is an amino sugar. Examples of GAGs include keratin sulfate, heparin sulfate, dermatin sulfate, and the most important one, hyaluronan or hyaluronic acid (HA) (non-sulfated). These sugars carry negative charges through a sulfate (SO3−) or carboxyl group (COO−). These negative charges make GAGs the most anionic molecules in mammalian cells [26]. GAGs are capable of high water retention due to their negative charges. The function of GAGs is mainly to regulate activity of secreted proteins and to immobilize secreted molecules close to where they are secreted to provide a reservoir of proteins for later use. They also play a role in tissue repair processes, including angiogenesis. Moreover, GAGs protect proteins from proteolytic degradation and alter or concentrate proteins for presentation to cell surface receptors [12,25,26].
- (ii)
- Proteoglycans are proteins that are heavily glycosylated. The basic proteoglycan unit consists of a core protein with one or more covalently attached GAG chains [27]. The chains are long, linear carbohydrate polymers that are negatively charged under physiological conditions due to the presence of sulfate and uronic acid groups. Proteoglycans are a major component of the ECM, i.e., the filler substance between the cells in an organism [28]. They form large complexes, both to other proteoglycans, such as hyaluronan, and to fibrous matrix proteins, such as collagen. They are also involved in binding cations such as sodium, potassium, and calcium, as well as water, but also regulate the movement of molecules through the matrix [25,28]. Evidence also shows they can affect the activity and stability of proteins and signaling molecules within the matrix [29]. The individual functions of proteoglycans can be attributed to either the protein core or the attached GAG chain. The most common type of proteoglycans within the brain ECM are aggrecan, brevican, glypican-1, versican, and tenascin-C [8,25].
- (iii)
- Glycoproteins provide structure and adhesive functions for the cells [12,25,30]. They have direct or indirect links with most of the intercellular and intracellular signaling pathways [10]. Collagen is one of the most important fibrous proteins, consisting of covalently intermolecular and intramolecular cross-linked helices. These helices are composed of hydroxyproline and hydroxylysine [31]. It is reported that collagen directly affects the ECM structure [8]. While fibrillar collagens are the most abundant proteins in the body and are highly expressed in the interstitial matrix of several organs, the normal adult brain contains very limited amounts of collagen, accounting for its soft consistency. The major form of collagen in the brain is collagen IV, which is present in the basement membrane surrounding the vascular endothelial cells. Collagen IV levels are upregulated in gliomas and localized to the basement membrane lining the vessel walls in astrocytomas of all grades, including GBM. Whether fibrillar collagens are present in gliomas is more contentious, and it has been reported that GBMs do not express intratumoral fibrillar collagen. The identity of the collagen producing cells is still unresolved but glioma cells can synthesize their own complement of ECM, including collagens I and IV [32]. During GBM progression, the collagen content of the brain does not increase rapidly and is reported to be tenfold lower compared to the GAGs. Therefore, collagen does not contribute to the ECM stiffening and generation of mechanical stress in GBM. However, aligned collagen fibers increase the presence of confined spaces within the brain and could fuel the migratory tendency of glioblastoma stem cells (GSCs) [33]. Elastin, another fibrous protein within the brain ECM, is a highly hydrophobic elastic protein secreted and organized in fibers and sheets and provides protection against tensile forces [34]. Fibronectin helps cells to attach to the matrix by first attaching to cells through its multiple binding domains via the RGD sequence [35,36].
- (iv)
- A wide variety of growth factors present within the ECM also continuously affect cell behavior by promoting cell adhesion, growth, proliferation, and differentiation during nervous system development. For instance, epidermal growth factor (EGF) is a polypeptide that acts as a signaling molecule in initiating mitosis and promotes rapid cell growth. Cytokine vascular endothelial growth factor (VEGF) controls brain angiogenesis and vascular network formation within the brain ECM. Fibroblast growth factors (FGF) regulate embryonic development, organogenesis, and tissue differentiation. Brain-derived growth factor (BDNF) serve as a survival factor during early ECM development [37].
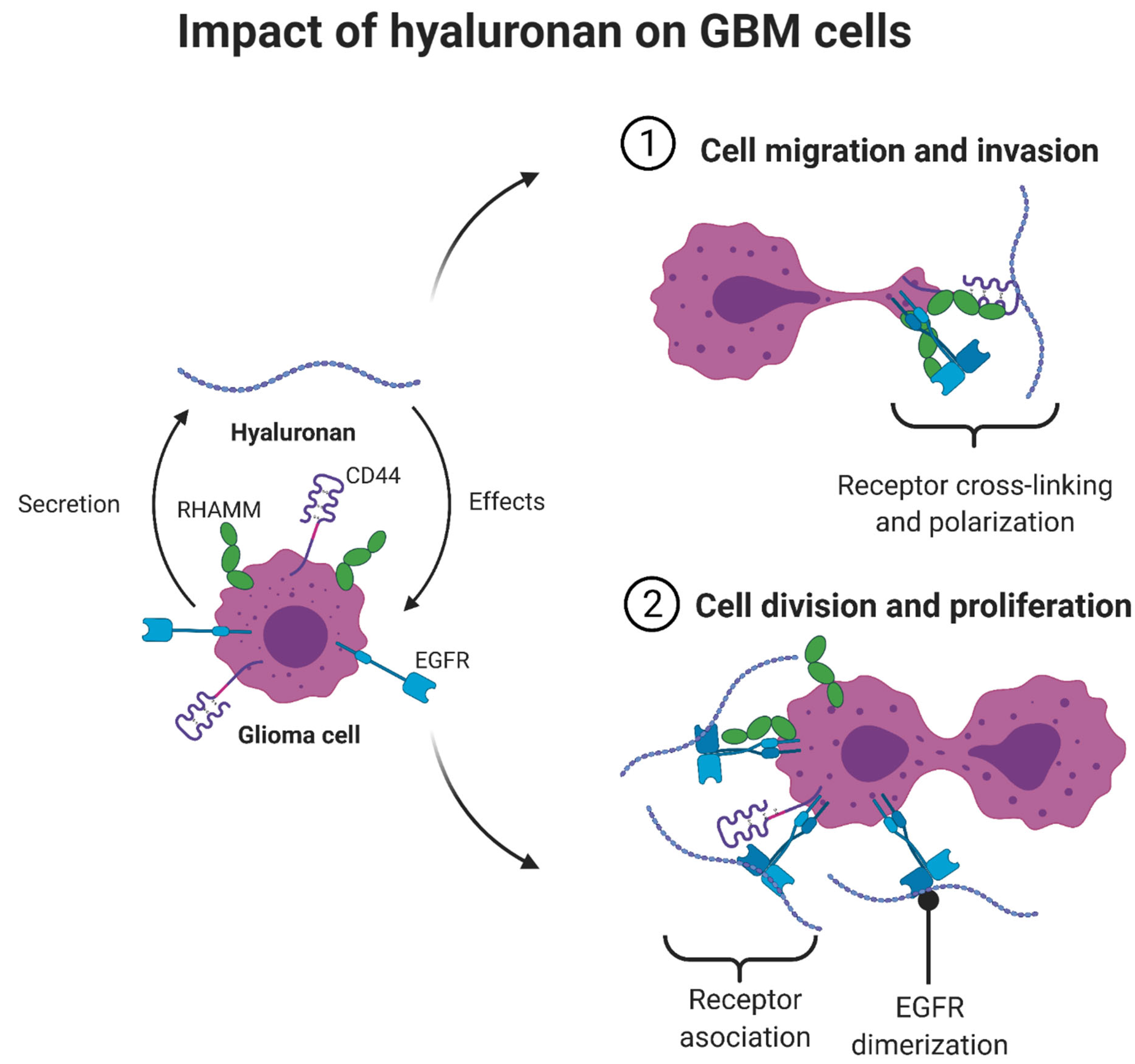
The Multifunctional Role of Hyaluronic Acid in ECM Alterations
4. Mechanics of the Brain and GBM Cells
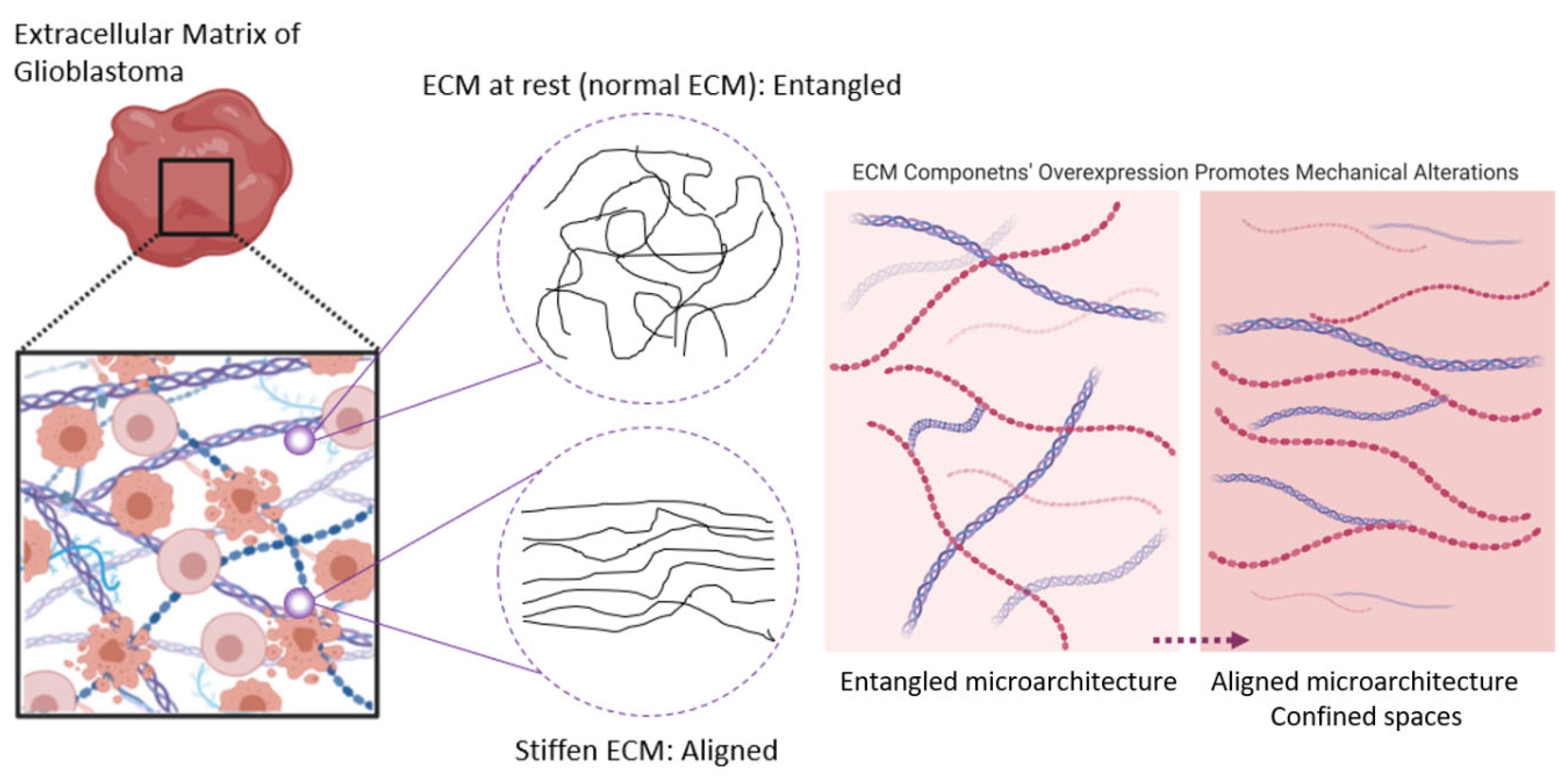
The Brain ECM under Tension: A Look into the Brain ECM Microarchitecture
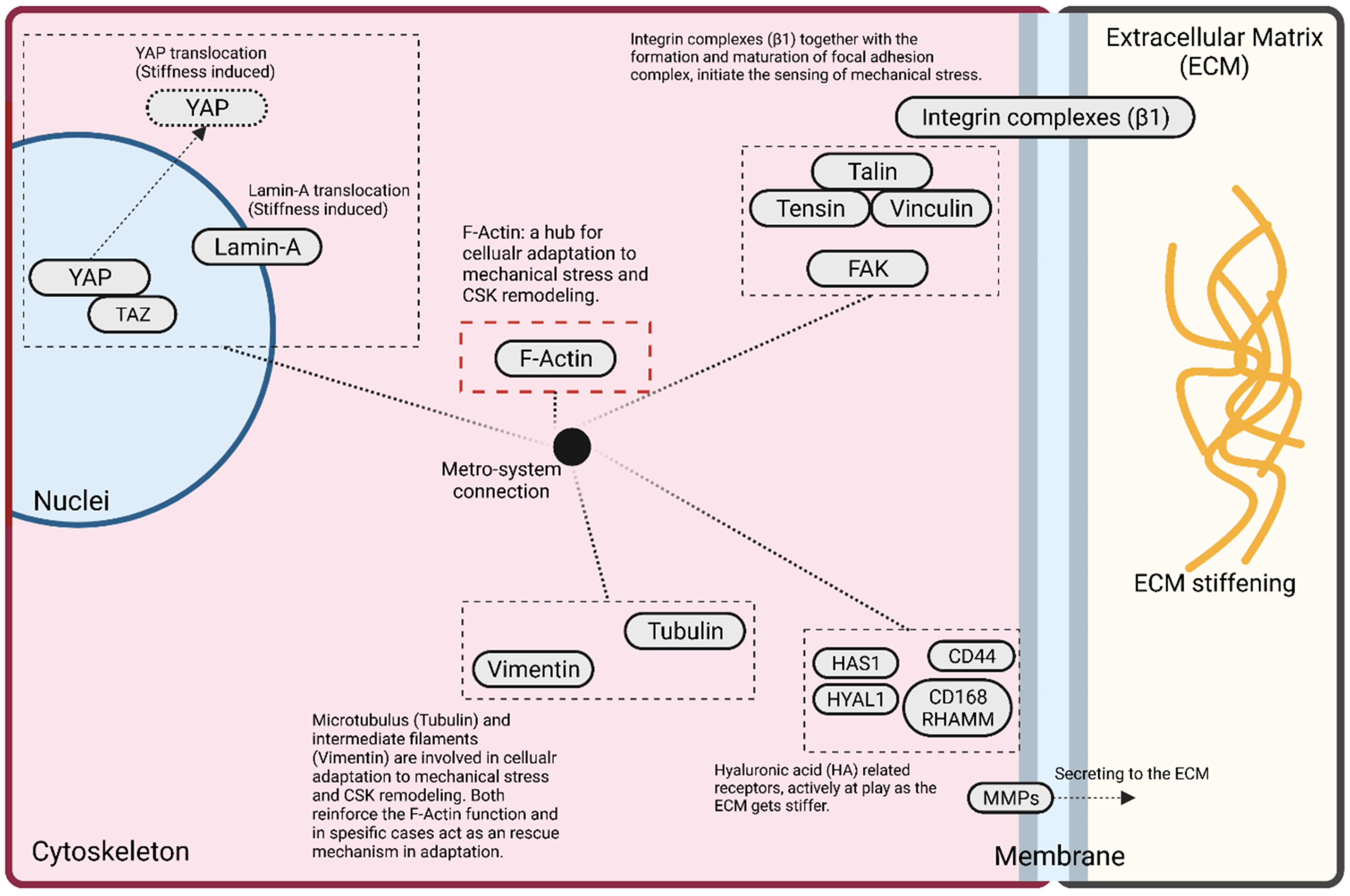
5. Mechanobiology of GBM Cells: Adaptive Cellular Signaling Pathways
5.1. F-Actin
5.2. Focal Adhesion
5.3. YAP
6. Current Bioengineered Strategies: In Vitro Tumor Microenvironment (TME) Models
7. Future Therapeutics: From Understanding to Tackling
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ohgaki, H.; Kleihues, P. The Definition of Primary and Secondary Glioblastoma. Clin. Cancer Res. 2013, 19, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Stathis, A. Treatment Overview. Handb. Lymphoma 2016, 20, 33–44. [Google Scholar] [CrossRef]
- Preusser, M.; De Ribaupierre, S.; Wöhrer, A.; Erridge, S.C.; Hegi, M.; Weller, M.; Stupp, R. Current Concepts and Management of Glioblastoma. Ann. Neurol. 2011, 70, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Bastiancich, C.; Danhier, P.; Préat, V.; Danhier, F. Anticancer Drug-Loaded Hydrogels as Drug Delivery Systems for the Local Treatment of Glioblastoma. J. Control. Release 2016, 243, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Khandwala, K.; Mubarak, F.; Minhas, K. The Many Faces of Glioblastoma: Pictorial Review of Atypical Imaging Features. Neuroradiol. J. 2020, 34, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Vollmann-Zwerenz, A.; Leidgens, V.; Feliciello, G.; Klein, C.A.; Hau, P. Tumor Cell Invasion in Glioblastoma. Int. J. Mol. Sci. 2020, 21, 1932. [Google Scholar] [CrossRef]
- Virga, J.; Szivos, L.; Hortobágyi, T.; Chalsaraei, M.K.; Zahuczky, G.; Steiner, L.; Tóth, J.; Reményi-Puskár, J.; Bognár, L.; Klekner, A. Extracellular Matrix Differences in Glioblastoma Patients with Different Prognoses. Oncol. Lett. 2019, 17, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Rejniak, K.A. Systems Biology of Tumor Microenvironment: Quantitative Modeling and Simulations; Springer: Berlin/Heidelberg, Germany, 2016; Volume 936, ISBN 978-3319420219. [Google Scholar]
- Neftel, C.; Laffy, J.; Filbin, M.G.; Hara, T.; Shore, M.E.; Rahme, G.J.; Richman, A.R.; Silverbush, D.; Shaw, M.L.; Hebert, C.M.; et al. An Integrative Model of Cellular States, Plasticity, and Genetics for Glioblastoma. Cell 2019, 178, 835–849.e21. [Google Scholar] [CrossRef]
- Mouw, J.K.; Ou, G.; Weaver, V.M. Extracellular Matrix Assembly: A Multiscale Deconstruction. Nat. Rev. Mol. Cell Biol. 2014, 15, 771–785. [Google Scholar] [CrossRef] [PubMed]
- Restall, I.; Bozek, D.A.; Chesnelong, C.; Weiss, S.; Luchman, H.A. Live-Cell Imaging Assays to Study Glioblastoma Brain Tumor Stem Cell Migration and Invasion. J. Vis. Exp. 2018, 2018, e58152. [Google Scholar] [CrossRef]
- Wolf, K.J.; Chen, J.; Coombes, J.D.; Aghi, M.K.; Kumar, S. Dissecting and Rebuilding the Glioblastoma Microenvironment with Engineered Materials. Nat. Rev. Mater. 2019, 4, 651–668. [Google Scholar] [CrossRef] [PubMed]
- Budday, S.; Ovaert, T.C.; Holzapfel, G.A.; Steinmann, P.; Kuhl, E. Fifty Shades of Brain: A Review on the Mechanical Testing and Modeling of Brain Tissue; Springer: Dordrecht, The Netherlands, 2019; ISBN 0123456789. [Google Scholar]
- Grundy, T.J.; De Leon, E.; Griffin, K.R.; Stringer, B.W.; Day, B.W.; Fabry, B.; Cooper-White, J.; O’Neill, G.M. Differential Response of Patient-Derived Primary Glioblastoma Cells to Environmental Stiffness. Sci. Rep. 2016, 6, 23353. [Google Scholar] [CrossRef] [PubMed]
- Nia, H.T.; Munn, L.L.; Jain, R.K. Mapping Physical Tumor Microenvironment and Drug Delivery. Clin. Cancer Res. 2019, 25, 2024–2026. [Google Scholar] [CrossRef] [PubMed]
- Palamà, I.E.; D’Amone, S.; Cortese, B. Microenvironmental Rigidity of 3D Scaffolds and Influence on Glioblastoma Cells: A Biomaterial Design Perspective. Front. Bioeng. Biotechnol. 2018, 6, 1–5. [Google Scholar] [CrossRef]
- Mair, D.B.; Ames, H.M.; Li, R. Mechanisms of Invasion and Motility of High-Grade Gliomas in the Brain. Mol. Biol. Cell 2018, 29, 2509–2515. [Google Scholar] [CrossRef]
- Wirtz, D.; Konstantopoulos, K.; Searson, P.C. The Physics of Cancer: The Role of Physical Interactions and Mechanical Forces in Metastasis. Nat. Rev. Cancer 2011, 11, 512–522. [Google Scholar] [CrossRef] [PubMed]
- Martino, F.; Perestrelo, A.R.; Vinarský, V.; Pagliari, S.; Forte, G. Cellular Mechanotransduction: From Tension to Function. Front. Physiol. 2018, 9, 1–21. [Google Scholar] [CrossRef]
- Nia, H.T.; Munn, L.L.; Jain, R.K. Physical Traits of Cancer. Science 2020, 370, eaaz0868. [Google Scholar] [CrossRef]
- Nagelkerke, A.; Bussink, J.; Rowan, A.E.; Span, P.N. The Mechanical Microenvironment in Cancer: How Physics Affects Tumours. Semin. Cancer Biol. 2015, 35, 62–70. [Google Scholar] [CrossRef]
- Pogoda, K.; Chin, L.; Georges, P.C.; Byfield, F.J.; Bucki, R.; Kim, R.; Weaver, M.; Wells, R.G.; Marcinkiewicz, C.; Janmey, P.A. Compression Stiffening of Brain and Its Effect on Mechanosensing by Glioma Cells. New J. Phys. 2014, 16, 1–17. [Google Scholar] [CrossRef]
- Zhang, Q.; Yu, Y.; Zhao, H. The Effect of Matrix Stiffness on Biomechanical Properties of Chondrocytes. Acta Biochim. Biophys. Sin. 2016, 48, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Hubmacher, D.; Apte, S.S. The Biology of the Extracellular Matrix: Novel Insights. Curr. Opin. Rheumatol. 2013, 25, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Quail, D.F.; Joyce, J.A. The Microenvironmental Landscape of Brain Tumors. Cancer Cell 2017, 31, 326–341. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, N.S.; Mancera, R.L. The Structure of Glycosaminoglycans and Their Interactions with Proteins. Chem. Biol. Drug Des. 2008, 72, 455–482. [Google Scholar] [CrossRef]
- Ruoslahti, E. Structural and Biology. Cell Differ. 1988, 4, 229–255. [Google Scholar]
- Iozzo, R.V. Matrix Proteoglycans: From Molecular Design to Cellular Function. Annu. Rev. Biochem. 1998, 67, 609–652. [Google Scholar] [CrossRef]
- Miyata, S.; Kitagawa, H. Chondroitin 6-Sulfation Regulates Perineuronal Net Formation by Controlling the Stability of Aggrecan. Neural Plast. 2016, 2016, 7–9. [Google Scholar] [CrossRef]
- Teodori, L.; Costa, A.; Marzio, R.; Perniconi, B.; Coletti, D.; Adamo, S.; Gupta, B.; Tarnok, A. Native Extracellular Matrix: A New Scaffolding Platform for Repair of Damaged Muscle. Front. Physiol. 2014, 5, 1–9. [Google Scholar] [CrossRef]
- Shoulders, M.D.; Raines, R.T. Collagen Structure and Stability Ann Rev Biochemistry. Annu. Rev. Biochem. 2009, 78, 929–958. [Google Scholar] [CrossRef]
- Payne, L.S.; Huang, P.H. The Pathobiology of Collagens in Glioma. Mol. Cancer Res. 2013, 11, 1129–1140. [Google Scholar] [CrossRef]
- Sood, D.; Chwalek, K.; Stuntz, E.; Pouli, D.; Du, C.; Tang-Schomer, M.; Georgakoudi, I.; Black, L.D.; Kaplan, D.L. Fetal Brain Extracellular Matrix Boosts Neuronal Network Formation in 3D Bioengineered Model of Cortical Brain Tissue. ACS Biomater. Sci. Eng. 2016, 2, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Debelle, L.; Tamburro, A.M. Elastin: Molecular Description and Function. Int. J. Biochem. Cell Biol. 1999, 31, 261–272. [Google Scholar] [CrossRef]
- Singh, P.; Carraher, C.; Schwarzbauer, J.E. Assembly of Fibronectin Extracellular Matrix. Annu. Rev. Cell Dev. Biol. 2010, 26, 397–419. [Google Scholar] [CrossRef] [PubMed]
- Johansson, S.; Svineng, G.; Wennerberg, K.; Armulik, A.; Lohikangas, L. Fibronectin-Integrin Interactions. Front. Biosci. 1997, 2, 126–146. [Google Scholar] [CrossRef]
- Brizzi, M.F.; Tarone, G.; Defilippi, P. Extracellular Matrix, Integrins, and Growth Factors as Tailors of the Stem Cell Niche. Curr. Opin. Cell Biol. 2012, 24, 645–651. [Google Scholar] [CrossRef]
- Belousov, A.; Titov, S.; Shved, N.; Garbuz, M.; Malykin, G.; Gulaia, V.; Kagansky, A.; Kumeiko, V. The Extracellular Matrix and Biocompatible Materials in Glioblastoma Treatment. Front. Bioeng. Biotechnol. 2019, 7, 341. [Google Scholar] [CrossRef]
- Heffernan, J.M.; Sirianni, R.W. Modeling Microenvironmental Regulation of Glioblastoma Stem Cells: A Biomaterials Perspective. Front. Mater. 2018, 5, 1–19. [Google Scholar] [CrossRef]
- Christofori, G.; Semb, H. The Role of the Cell-Adhesion Molecule E-Cadherin as a Tumour-Suppressor Gene. Trends Biochem. Sci. 1999, 24, 73–76. [Google Scholar] [CrossRef]
- Elangbam, C.S.; Qualls, C.W.; Dahlgren, R.R. Cell Adhesion Molecules—Update. Vet. Pathol. 1997, 34, 61–73. [Google Scholar] [CrossRef]
- Lukashev, M.E.; Werb, Z. ECM Signalling: Orchestrating Cell Behaviour and Misbehaviour. Trends Cell Biol. 1998, 8, 437–441. [Google Scholar] [CrossRef]
- Cha, J.; Kim, P. Biomimetic Strategies for the Glioblastoma Microenvironment. Front. Mater. 2017, 4, 1–8. [Google Scholar] [CrossRef]
- Chen, J.W.E.; Pedron, S.; Shyu, P.; Hu, Y.; Sarkaria, J.N.; Harley, B.A.C. Influence of Hyaluronic Acid Transitions in Tumor Microenvironment on Glioblastoma Malignancy and Invasive Behavior. Front. Mater. 2018, 5, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Riegler, J.; Labyed, Y.; Rosenzweig, S.; Javinal, V.; Castiglioni, A.; Dominguez, C.X.; Long, J.E.; Li, Q.; Sandoval, W.; Junttila, M.R.; et al. Tumor Elastography and Its Association with Collagen and the Tumor Microenvironment. Clin. Cancer Res. 2018, 24, 4455–4467. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.C.; Yang, C.H.; Cheng, L.H.; Chang, W.T.; Lin, Y.R.; Cheng, H.C. Fibronectin in Cancer: Friend or Foe. Cells 2019, 9, 27. [Google Scholar] [CrossRef]
- De Oliveira Rosario, L.V.; Da Rosa, B.G.; Goncalves, T.L.; Matias, D.I.L.; Freitas, C.; Ferrer, V.P. Glioblastoma Factors Increase the Migration of Human Brain Endothelial Cells in Vitro by Increasing MMP-9/CXCR4 Levels. Anticancer Res. 2020, 40, 2725–2737. [Google Scholar] [CrossRef]
- Barnes, J.M.; Przybyla, L.; Weaver, V.M. Tissue Mechanics Regulate Brain Development, Homeostasis and Disease. J. Cell Sci. 2017, 130, 71–82. [Google Scholar] [CrossRef]
- Stewart, D.C.; Rubiano, A.; Dyson, K.; Simmons, C.S. Mechanical Characterization of Human Brain Tumors from Patients and Comparison to Potential Surgical Phantoms. PLoS ONE 2017, 12, e177561. [Google Scholar] [CrossRef]
- Li, X.; Wang, J. Mechanical Tumor Microenvironment and Transduction: Cytoskeleton Mediates Cancer Cell Invasion and Metastasis. Int. J. Biol. Sci. 2020, 16, 2014–2018. [Google Scholar] [CrossRef]
- Duan, Q.; Zhang, H.; Zheng, J.; Zhang, L. Turning Cold into Hot: Firing up the Tumor Microenvironment. Trends Cancer 2020, 6, 605–618. [Google Scholar] [CrossRef]
- Necas, J.; Bartosikova, L.; Brauner, P.; Kolar, J. Hyaluronic Acid (Hyaluronan): A Review. Vet. Med. 2008, 53, 397–411. [Google Scholar] [CrossRef]
- Khaing, Z.Z.; Seidlits, S.K. Hyaluronic Acid and Neural Stem Cells: Implications for Biomaterial Design. J. Mater. Chem. B 2015, 3, 7850–7866. [Google Scholar] [CrossRef] [PubMed]
- Delpech, B.; Maingonnat, C.; Girard, N.; Chauzy, C.; Olivier, A.; Maunoury, R.; Tayot, J.; Creissard, P. Hyaluronan and Hyaluronectin in the Extracellular Matrix of Human Brain Tumour Stroma. Eur. J. Cancer 1993, 29, 1012–1017. [Google Scholar] [CrossRef]
- Zimmermann, D.R.; Dours-Zimmermann, M.T. Extracellular Matrix of the Central Nervous System: From Neglect to Challenge. Histochem. Cell Biol. 2008, 130, 635–653. [Google Scholar] [CrossRef]
- Cha, J.; Kang, S.G.; Kim, P. Strategies of Mesenchymal Invasion of Patient-Derived Brain Tumors: Microenvironmental Adaptation. Sci. Rep. 2016, 6, 24912. [Google Scholar] [CrossRef] [PubMed]
- Wolf, K.J.; Kumar, S. Hyaluronic Acid: Incorporating the Bio into the Material. ACS Biomater. Sci. Eng. 2019, 5, 3753–3765. [Google Scholar] [CrossRef] [PubMed]
- Bignami, A.; Hosley, M.; Dahl, D. Hyaluronic Acid and Hyaluronic Acid-Binding Proteins in Brain Extracellular Matrix. Anat. Embryol. 1993, 188, 419–433. [Google Scholar] [CrossRef]
- Pibuel, M.A.; Díaz, M.; Molinari, Y.; Poodts, D.; Silvestroff, L.; Lompardía, S.L.; Franco, P.; Hajos, S.E. 4-Methylumbelliferone as a Potent and Selective Antitumor Drug on a Glioblastoma Model. Glycobiology 2020, 31, 1–15. [Google Scholar] [CrossRef]
- Liu, M.; Tolg, C.; Turley, E. Dissecting the Dual Nature of Hyaluronan in the Tumor Microenvironment. Front. Immunol. 2019, 10, 1–9. [Google Scholar] [CrossRef]
- Kim, Y.; Kumar, S. CD44-Mediated Adhesion to Hyaluronic Acid Contributes to Mechanosensing and Invasive Motility. Mol. Cancer Res. 2014, 12, 1416–1429. [Google Scholar] [CrossRef]
- Chen, J.W.E.; Pedron, S.; Harley, B.A.C. The Combined Influence of Hydrogel Stiffness and Matrix-Bound Hyaluronic Acid Content on Glioblastoma Invasion. Macromol. Biosci. 2017, 17, 1–11. [Google Scholar] [CrossRef]
- Erickson, A.E.; Lan Levengood, S.K.; Sun, J.; Chang, F.C.; Zhang, M. Fabrication and Characterization of Chitosan–Hyaluronic Acid Scaffolds with Varying Stiffness for Glioblastoma Cell Culture. Adv. Healthc. Mater. 2018, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.; Stowers, R.; Nam, S.; Xia, Y.; Chaudhuri, O. Stress Relaxing Hyaluronic Acid-Collagen Hydrogels Promote Cell Spreading, Fiber Remodeling, and Focal Adhesion Formation in 3D Cell Culture. Biomaterials 2018, 154, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Zamboni, F.; Keays, M.; Hayes, S.; Albadarin, A.B.; Walker, G.M.; Kiely, P.A.; Collins, M.N. Enhanced Cell Viability in Hyaluronic Acid Coated Poly(Lactic-Co-Glycolic Acid) Porous Scaffolds within Microfluidic Channels. Int. J. Pharm. 2017, 532, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Urbanska, K.; Sokolowska, J.; Szmidt, M.; Sysa, P. Glioblastoma Multiforme—An Overview. Wspolczesna Onkol. 2014, 18, 307–312. [Google Scholar] [CrossRef]
- Sorribes, I.C.; Moore, M.N.J.; Byrne, H.M.; Jain, H.V. A Biomechanical Model of Tumor-Induced Intracranial Pressure and Edema in Brain Tissue. Biophys. J. 2019, 116, 1560–1574. [Google Scholar] [CrossRef]
- Reetz, K.; Abbas, Z.; Costa, A.S.; Gras, V.; Tiffin-Richards, F.; Mirzazade, S.; Holschbach, B.; Frank, R.D.; Vassiliadou, A.; Krüger, T.; et al. Increased Cerebral Water Content in Hemodialysis Patients. PLoS ONE 2015, 10, e122188. [Google Scholar] [CrossRef]
- Olson, J.E.; Mishler, L.; Dimlich, R.V.W. Brain Water Content, Brain Blood Volume, Blood Chemistry, and Pathology in a Model of Cerebral Edema. Ann. Emerg. Med. 1990, 19, 1113–1121. [Google Scholar] [CrossRef]
- Streitberger, K.J.; Lilaj, L.; Schrank, F.; Braun, J.; Hoffmann, K.T.; Reiss-Zimmermann, M.; Käs, J.A.; Sack, I. How Tissue Fluidity Influences Brain Tumor Progression. Proc. Natl. Acad. Sci. USA 2020, 117, 128–134. [Google Scholar] [CrossRef]
- Schiller, J.; Huster, D. New Methods to Study the Composition and Structure of the Extracellular Matrix in Natural and Bioengineered Tissues. Biomatter 2012, 2, 115–131. [Google Scholar] [CrossRef][Green Version]
- Venkatesan, R.; Lin, W.; Gurleyik, K.; He, Y.Y.; Paczynski, R.P.; Powers, W.J.; Hsu, C.Y. Absolute Measurements of Water Content Using Magnetic Resonance Imaging: Preliminary Findings in an in Vivo Focal Ischemic Rat Model. Magn. Reson. Med. 2000, 43, 146–150. [Google Scholar] [CrossRef]
- Ciasca, G.; Sassun, T.E.; Minelli, E.; Antonelli, M.; Papi, M.; Santoro, A.; Giangaspero, F.; Delfini, R.; De Spirito, M. Nano-Mechanical Signature of Brain Tumours. Nanoscale 2016, 8, 19629–19643. [Google Scholar] [CrossRef] [PubMed]
- Bunevicius, A.; Schregel, K.; Sinkus, R.; Golby, A.; Patz, S. REVIEW: MR Elastography of Brain Tumors. NeuroImage Clin. 2020, 25, 102109. [Google Scholar] [CrossRef]
- Fattahi, N.; Arani, A.; Perry, A.; Meyer, F.; Manduca, A.; Glaser, K.; Senjem, M.L.; Ehman, R.L.; Huston, J. MR Elastography Demonstrates Increased Brain Stiffness in Normal Pressure Hydrocephalus. Am. J. Neuroradiol. 2016, 37, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Maher, E.A.; Bachoo, R.M. Glioblastoma. 2014. Available online: https://www.sciencedirect.com/science/article/pii/B9780124105294000784?via%3Dihub (accessed on 1 May 2022).
- Bilston, L.E. Soft Tissue Rheology and Its Implications for Elastography: Challenges and Opportunities. NMR Biomed. 2018, 31, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, C.F.; Gasperini, L.; Marques, A.P.; Reis, R.L. The Stiffness of Living Tissues and Its Implications for Tissue Engineering. Nat. Rev. Mater. 2020, 5, 351–370. [Google Scholar] [CrossRef]
- Saleh, A.; Marhuenda, E.; Fabre, C.; Hassani, Z.; de Weille, J.; Boukhaddaoui, H.; Guelfi, S.; Maldonado, I.L.; Hugnot, J.P.; Duffau, H.; et al. A Novel 3D Nanofibre Scaffold Conserves the Plasticity of Glioblastoma Stem Cell Invasion by Regulating Galectin-3 and Integrin-Β1 Expression. Sci. Rep. 2019, 9, 14612. [Google Scholar] [CrossRef]
- Sharma, P.; Sheets, K.; Elankumaran, S.; Nain, A.S. The Mechanistic Influence of Aligned Nanofibers on Cell Shape, Migration and Blebbing Dynamics of Glioma Cells. Integr. Biol. 2013, 5, 1036–1044. [Google Scholar] [CrossRef]
- Beliveau, A.; Thomas, G.; Gong, J.; Wen, Q.; Jain, A. Aligned Nanotopography Promotes a Migratory State in Glioblastoma Multiforme Tumor Cells. Sci. Rep. 2016, 6, 26143. [Google Scholar] [CrossRef]
- Babu, P.K.V.; Radmacher, M. Mechanics of Brain Tissues Studied by Atomic Force Microscopy: A Perspective. Front. Neurosci. 2019, 13, 1–9. [Google Scholar] [CrossRef]
- Palombo, F.; Winlove, C.P.; Edginton, R.S.; Green, E.; Stone, N.; Caponi, S.; Madami, M.; Fioretto, D. Biomechanics of Fibrous Proteins of the Extracellular Matrix Studied by Brillouin Scattering. J. R. Soc. Interface 2014, 11, 20140739. [Google Scholar] [CrossRef]
- Nia, H.T.; Datta, M.; Seano, G.; Zhang, S.; Ho, W.W.; Roberge, S.; Huang, P.; Munn, L.L.; Jain, R.K. In Vivo Compression and Imaging in Mouse Brain to Measure the Effects of Solid Stress. Nat. Protoc. 2020, 15, 2321–2340. [Google Scholar] [CrossRef] [PubMed]
- Munn, L.L.; Nia, H.T. Mechanosensing Tensile Solid Stresses. Proc. Natl. Acad. Sci. USA 2019, 116, 21960–21962. [Google Scholar] [CrossRef] [PubMed]
- Seano, G.; Nia, H.T.; Emblem, K.E.; Datta, M.; Ren, J.; Krishnan, S.; Kloepper, J.; Pinho, M.C.; Ho, W.W.; Ghosh, M.; et al. Solid Stress in Brain Tumours Causes Neuronal Loss and Neurological Dysfunction and Can Be Reversed by Lithium. Nat. Biomed. Eng. 2019, 3, 230–245. [Google Scholar] [CrossRef] [PubMed]
- Simi, A.K.; Pang, M.; Nelson, C.M. Extracellular Matrix Stiffness Exists in a Feedback Loop That Drives Tumor Progression. Adv. Exp. Med. Biol. 2018, 1092, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Kamm, R.D.; Lee, R.T. Cell Mechanics and Mechanotransduction: Pathways, Probes, and Physiology. Am. J. Physiol.-Cell Physiol. 2004, 287, 1–11. [Google Scholar] [CrossRef]
- Interests, C.F. News and Views. Nature 1926, 118, 455–459. [Google Scholar] [CrossRef]
- Gkretsi, V.; Stylianopoulos, T. Cell Adhesion and Matrix Stiffness: Coordinating Cancer Cell Invasion and Metastasis. Front. Oncol. 2018, 8, 145. [Google Scholar] [CrossRef]
- Provenzano, P.P.; Keely, P.J. Mechanical Signaling through the Cytoskeleton Regulates Cell Proliferation by Coordinated Focal Adhesion and Rho GTPase Signaling. J. Cell Sci. 2011, 124, 1195–1205. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of YAP/TAZ in Mechanotransduction. Nature 2011, 474, 179–184. [Google Scholar] [CrossRef]
- Gill, B.J.; Gibbons, D.L.; Roudsari, L.C.; Saik, J.E.; Rizvi, Z.H.; Roybal, J.D.; Kurie, J.M.; West, J.L. A Synthetic Matrix with Independently Tunable Biochemistry and Mechanical Properties to Study Epithelial Morphogenesis and EMT in a Lung Adenocarcinoma Model. Cancer Res. 2012, 72, 6013–6023. [Google Scholar] [CrossRef]
- Wolfenson, H.; Yang, B.; Sheetz, M.P. Steps in Mechanotransduction Pathways That Control Cell Morphology. Annu. Rev. Physiol. 2019, 81, 585–605. [Google Scholar] [CrossRef] [PubMed]
- Jaalouk, D.E.; Lammerding, J. Mechanotransduction Gone Awry. Nat. Rev. Mol. Cell Biol. 2009, 10, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Farge, E. Mechanotransduction in Development, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2011; Volume 95, ISBN 9780123850652. [Google Scholar]
- Sun, M.; Chi, G.; Li, P.; Lv, S.; Xu, J.; Xu, Z.; Xia, Y.; Tan, Y.; Xu, J.; Li, L.; et al. Effects of Matrix Stiffness on the Morphology, Adhesion, Proliferation and Osteogenic Differentiation of Mesenchymal Stem Cells. Int. J. Med. Sci. 2018, 15, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, V. Cell–Extracellular Matrix Interaction in Glioma Growth. In Silico Model. J. Integr. Bioinform. 2020, 17, 27. [Google Scholar] [CrossRef]
- Dominguez, R.; Holmes, K.C. Actin Structure and Function. Annu. Rev. Biophys. 2011, 40, 169–186. [Google Scholar] [CrossRef]
- Pellegrin, S.; Mellor, H. Actin Stress Fibers. J. Cell Sci. 2007, 120, 3491–3499. [Google Scholar] [CrossRef]
- Seetharaman, S.; Etienne-Manneville, S. Cytoskeletal Crosstalk in Cell Migration. Trends Cell Biol. 2020, 30, 720–735. [Google Scholar] [CrossRef]
- Schwarz, U.S.; Gardel, M.L. United We Stand-Integrating the Actin Cytoskeleton and Cell-Matrix Adhesions in Cellular Mechanotransduction. J. Cell Sci. 2012, 125, 3051–3060. [Google Scholar] [CrossRef]
- Bhadriraju, K.; Hansen, L.K. Extracellular Matrix- and Cytoskeleton-Dependent Changes in Cell Shape and Stiffness. Exp. Cell Res. 2002, 278, 92–100. [Google Scholar] [CrossRef]
- Schlunck, G.; Han, H.; Wecker, T.; Kampik, D.; Meyer-ter-Vehn, T.; Grehn, F. Substrate Rigidity Modulates Cell-Matrix Interactions and Protein Expression in Human Trabecular Meshwork Cells. Investig. Ophthalmol. Vis. Sci. 2008, 49, 262–269. [Google Scholar] [CrossRef]
- Giardiello, F.M.; Hylind, L.M.; Trimbath, J.D.; Hamilton, S.R.; Romans, K.E.; Cruz-Correa, M.; Corretti, M.C.; Offerhaus, G.J.A.; Yang, V.W. Oral Contraceptives and Polyp Regression in Familial Adenomatous Polyposis. Gastroenterology 2005, 128, 1077–1080. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, M.A.; Modzelewska, K.; Kwong, L.; Keely, P.J. Focal Adhesion Regulation of Cell Behavior. Biochim. Biophys. Acta-Mol. Cell Res. 2004, 1692, 103–119. [Google Scholar] [CrossRef] [PubMed]
- Schlaepfer, D.D.; Hauck, C.R.; Sieg, D.J. Signaling through Focal Adhesion Kinase. Prog. Biophys. Mol. Biol. 1999, 71, 435–478. [Google Scholar] [CrossRef]
- Janiszewska, M.; Primi, M.C.; Izard, T. Cell Adhesion in Cancer: Beyond the Migration of Single Cells. J. Biol. Chem. 2020, 295, 2495–2505. [Google Scholar] [CrossRef]
- Alexandrova, A.Y.; Arnold, K.; Schaub, S.; Vasiliev, J.M.; Meister, J.J.; Bershadsky, A.D.; Verkhovsky, A.B. Comparative Dynamics of Retrograde Actin Flow and Focal Adhesions: Formation of Nascent Adhesions Triggers Transition from Fast to Slow Flow. PLoS ONE 2008, 3, e3234. [Google Scholar] [CrossRef]
- Kanoldt, V.; Kluger, C.; Barz, C.; Schweizer, A.L.; Ramanujam, D.; Windgasse, L.; Engelhardt, S.; Chrostek-Grashoff, A.; Grashoff, C. Metavinculin Modulates Force Transduction in Cell Adhesion Sites. Nat. Commun. 2020, 11, 1–10. [Google Scholar] [CrossRef]
- Umesh, V.; Rape, A.D.; Ulrich, T.A.; Kumar, S. Microenvironmental Stiffness Enhances Glioma Cell Proliferation by Stimulating Epidermal Growth Factor Receptor Signaling. PLoS ONE 2014, 9, e101771. [Google Scholar] [CrossRef]
- Abylkassov, R.; Xie, Y. Role of Yes-Associated Protein in Cancer: An Update (Review). Oncol. Lett. 2016, 12, 2277–2282. [Google Scholar] [CrossRef]
- Caliari, S.R.; Perepelyuk, M.; Cosgrove, B.D.; Tsai, S.J.; Lee, G.Y.; Mauck, R.L.; Wells, R.G.; Burdick, J.A. Stiffening Hydrogels for Investigating the Dynamics of Hepatic Stellate Cell Mechanotransduction during Myofibroblast Activation. Sci. Rep. 2016, 6, 21387. [Google Scholar] [CrossRef]
- Bandaru, P.; Cefaloni, G.; Vajhadin, F.; Lee, K.J.; Kim, H.J.; Cho, H.J.; Hartel, M.C.; Zhang, S.; Sun, W.; Goudie, M.J.; et al. Mechanical Cues Regulating Proangiogenic Potential of Human Mesenchymal Stem Cells through YAP-Mediated Mechanosensing. Small 2020, 16, 1–13. [Google Scholar] [CrossRef]
- Vallejo-Giraldo, C.; Genta, M.; Cauvi, O.; Goding, J.; Green, R. Hydrogels for 3D Neural Tissue Models: Understanding Cell-Material Interactions at a Molecular Level. Front. Bioeng. Biotechnol. 2020, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Koushki, N.; Ghagre, A.; Srivastava, L.K.; Sitaras, C.; Yoshie, H.; Molter, C.; Ehrlicher, A. Lamin A Redistribution Mediated by Nuclear Deformation Determines Dynamic Localization of YAP. BioRxiv 2020, 3, 1–32. [Google Scholar] [CrossRef]
- Belaadi, N.; Aureille, J.; Guilluy, C. Under Pressure: Mechanical Stress Management in the Nucleus. Cells 2016, 5, 27. [Google Scholar] [CrossRef] [PubMed]
- Rianna, C.; Radmacher, M.; Kumar, S. Direct Evidence That Tumor Cells Soften When Navigating Confined Spaces. Mol. Biol. Cell 2020, 31, 1726–1734. [Google Scholar] [CrossRef] [PubMed]
- Paul, C.D.; Mistriotis, P.; Konstantopoulos, K. Cancer Cell Motility: Lessons from Migration in Confined Spaces. Nat. Rev. Cancer 2017, 17, 131–140. [Google Scholar] [CrossRef]
- Shen, K.; Kenche, H.; Zhao, H.; Li, J.; Stone, J. The Role of Extracellular Matrix Stiffness in Regulating Cytoskeletal Remodeling via Vinculin in Synthetic Smooth Muscle Cells. Biochem. Biophys. Res. Commun. 2019, 508, 302–307. [Google Scholar] [CrossRef]
- Pegoraro, A.F.; Janmey, P.; Weitz, D.A. Mechanical Properties of the Cytoskeleton and Cells. Cold Spring Harb. Perspect. Biol. 2017, 9, a022038. [Google Scholar] [CrossRef]
- Harburger, D.S.; Calderwood, D.A. Integrin Signalling at a Glance. J. Cell Sci. 2009, 122, 1472. [Google Scholar] [CrossRef]
- Janmey, P.A. The Cytoskeleton and Cell Signaling: Component Localization and Mechanical Coupling. Physiol. Rev. 1998, 78, 763–781. [Google Scholar] [CrossRef]
- Koh, I.; Cha, J.; Park, J.; Choi, J.; Kang, S.G.; Kim, P. The Mode and Dynamics of Glioblastoma Cell Invasion into a Decellularized Tissue-Derived Extracellular Matrix-Based Three-Dimensional Tumor Model. Sci. Rep. 2018, 8, 4608. [Google Scholar] [CrossRef]
- Discher, D.E.; Janmey, P.; Wang, Y.L. Tissue Cells Feel and Respond to the Stiffness of Their Substrate. Science 2005, 310, 1139–1143. [Google Scholar] [CrossRef] [PubMed]
- Caló, E.; Khutoryanskiy, V.V. Biomedical Applications of Hydrogels: A Review of Patents and Commercial Products. Eur. Polym. J. 2015, 65, 252–267. [Google Scholar] [CrossRef]
- Raman, R.; Langer, R. Biohybrid Design Gets Personal: New Materials for Patient-Specific Therapy. Adv. Mater. 2019, 1901969, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Roman, N.; Chalmers, A.J. Patient-Specific 3D-Printed Glioblastomas. Nat. Biomed. Eng. 2019, 3, 498–499. [Google Scholar] [CrossRef] [PubMed]
- Pogoda, K.; Bucki, R.; Byfield, F.J.; Cruz, K.; Lee, T.; Marcinkiewicz, C.; Janmey, P.A. Soft Substrates Containing Hyaluronan Mimic the Effects of Increased Stiffness on Morphology, Motility, and Proliferation of Glioma Cells. Biomacromolecules 2017, 18, 3040–3051. [Google Scholar] [CrossRef] [PubMed]
- Karabiyik Acar, O.; Kayitmazer, A.B.; Torun Kose, G. Hyaluronic Acid/Chitosan Coacervate-Based Scaffolds. Biomacromolecules 2018, 19, 1198–1211. [Google Scholar] [CrossRef]
- Burdick, J.A.; Prestwich, G.D. Hyaluronic Acid Hydrogels for Biomedical Applications. Adv. Mater. 2011, 23, 41–56. [Google Scholar] [CrossRef]
- Khunmanee, S.; Jeong, Y.; Park, H. Crosslinking Method of Hyaluronic-Based Hydrogel for Biomedical Applications. J. Tissue Eng. 2017, 8, 2041731417726464. [Google Scholar] [CrossRef]
- Sung, K.E.; Su, G.; Pehlke, C.; Trier, S.M.; Eliceiri, K.W.; Keely, P.J.; Friedl, A.; Beebe, D.J. Control of 3-Dimensional Collagen Matrix Polymerization for Reproducible Human Mammary Fibroblast Cell Culture in Microfluidic Devices. Biomaterials 2009, 30, 4833–4841. [Google Scholar] [CrossRef]
- Rao, S.S.; Dejesus, J.; Short, A.R.; Otero, J.J.; Sarkar, A.; Winter, J.O. Glioblastoma Behaviors in Three-Dimensional Collagen-Hyaluronan Composite Hydrogels. ACS Appl. Mater. Interfaces 2013, 5, 9276–9284. [Google Scholar] [CrossRef]
- Ayuso, J.M.; Monge, R.; Martínez-González, A.; Virumbrales-Muñoz, M.; Llamazares, G.A.; Berganzo, J.; Hernández-Laín, A.; Santolaria, J.; Doblaré, M.; Hubert, C.; et al. Glioblastoma on a Microfluidic Chip: Generating Pseudopalisades and Enhancing Aggressiveness through Blood Vessel Obstruction Events. Neuro-Oncology 2017, 19, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Sakthivel, K.; Kumar, H.; Mohamed, M.G.A.; Talebjedi, B.; Shim, J.; Najjaran, H.; Hoorfar, M.; Kim, K. High Throughput Screening of Cell Mechanical Response Using a Stretchable 3D Cellular Microarray Platform. Small 2020, 16, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Mora-Boza, A.; Włodarczyk-Biegun, M.K.; Del Campo, A.; Vázquez-Lasa, B.; Román, J.S. Glycerylphytate as an Ionic Crosslinker for 3D Printing of Multi-Layered Scaffolds with Improved Shape Fidelity and Biological Features. Biomater. Sci. 2020, 8, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Wang, C.; Cui, Y.; Li, A.; Qiao, Y.; Qiu, D. Conjoined-Network Rendered Stiff and Tough Hydrogels from Biogenic Molecules. Sci. Adv. 2019, 5, eaau3442. [Google Scholar] [CrossRef]
- Jiang, T.; Munguia-Lopez, J.G.; Gu, K.; Bavoux, M.M.; Flores-Torres, S.; Kort-Mascort, J.; Grant, J.; Vijayakumar, S.; De Leon-Rodriguez, A.; Ehrlicher, A.J.; et al. Engineering Bioprintable Alginate/Gelatin Composite Hydrogels with Tunable Mechanical and Cell Adhesive Properties to Modulate Tumor Spheroid Growth Kinetics. Biofabrication 2020, 12, 015024. [Google Scholar] [CrossRef]
- Bahram, M.; Mohseni, N.; Moghtader, M. An Introduction to Hydrogels and Some Recent Applications. Emerg. Concepts Anal. Appl. Hydrogels 2016, 2, 16. [Google Scholar] [CrossRef]
- Liaw, C.Y.; Ji, S.; Guvendiren, M. Engineering 3D Hydrogels for Personalized In Vitro Human Tissue Models. Adv. Healthc. Mater. 2018, 7, 1–16. [Google Scholar] [CrossRef]
- Shin, Y.J.; Shafranek, R.T.; Tsui, J.H.; Walcott, J.; Nelson, A.; Kim, D.H. 3D Bioprinting of Mechanically Tuned Bioinks Derived from Cardiac Decellularized Extracellular Matrix. Acta Biomater. 2020, 119, 75–88. [Google Scholar] [CrossRef]
- Choudhury, D.; Tun, H.W.; Wang, T.; Naing, M.W. Organ-Derived Decellularized Extracellular Matrix: A Game Changer for Bioink Manufacturing? Trends Biotechnol. 2018, 36, 787–805. [Google Scholar] [CrossRef]
- Ferreira, L.P.; Gaspar, V.M.; Mano, J.F. Decellularized Extracellular Matrix for Bioengineering Physiomimetic 3D in Vitro Tumor Models. Trends Biotechnol. 2020, 38, 1397–1414. [Google Scholar] [CrossRef]
- Kabirian, F.; Mozafari, M. Decellularized ECM-Derived Bioinks: Prospects for the Future. Methods 2020, 171, 108–118. [Google Scholar] [CrossRef]
- Seo, Y.; Jeong, S.; Chung, J.J.; Kim, S.H.; Choi, N.; Jung, Y. Development of an Anisotropically Organized Brain DECM Hydrogel-Based 3D Neuronal Culture Platform for Recapitulating the Brain Microenvironment in Vivo. ACS Biomater. Sci. Eng. 2020, 6, 610–620. [Google Scholar] [CrossRef] [PubMed]
- Lam, D.; Enright, H.A.; Cadena, J.; Peters, S.K.G.; Sales, A.P.; Osburn, J.J.; Soscia, D.A.; Kulp, K.S.; Wheeler, E.K.; Fischer, N.O. Tissue-Specific Extracellular Matrix Accelerates the Formation of Neural Networks and Communities in a Neuron-Glia Co-Culture on a Multi-Electrode Array. Sci. Rep. 2019, 9, 4159. [Google Scholar] [CrossRef] [PubMed]
- Sackett, S.D.; Tremmel, D.M.; Ma, F.; Feeney, A.K.; Maguire, R.M.; Brown, M.E.; Zhou, Y.; Li, X.; O’Brien, C.; Li, L.; et al. Extracellular Matrix Scaffold and Hydrogel Derived from Decellularized and Delipidized Human Pancreas. Sci. Rep. 2018, 8, 10452. [Google Scholar] [CrossRef]
- Simsa, R.; Rothenbücher, T.; Gürbüz, H.; Ghosheh, N.; Emneus, J.; Jenndahl, L.; Kaplan, D.L.; Bergh, N.; Serrano, A.M.; Fogelstrand, P. Brain Organoid Formation on Decellularized Porcine Brain ECM Hydrogels. PLoS ONE 2021, 16, e245685. [Google Scholar] [CrossRef] [PubMed]
- Malagón-Romero, D.; Hernández, N.; Cardozo, C.; Godoy-Silva, R.D. Rheological Characterization of a Gel Produced Using Human Blood Plasma and Alginate Mixtures. J. Mech. Behav. Biomed. Mater. 2014, 34, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Tamayol, A.; Najafabadi, A.H.; Aliakbarian, B.; Arab-Tehrany, E.; Akbari, M.; Annabi, N.; Juncker, D.; Khademhosseini, A. Hydrogel Templates for Rapid Manufacturing of Bioactive Fibers and 3D Constructs. Adv. Healthc. Mater. 2015, 4, 2146–2153. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Starly, B.; Daly, A.C.; Burdick, J.A.; Groll, J.; Skeldon, G.; Shu, W.; Sakai, Y.; Shinohara, M.; Nishikawa, M.; et al. The Bioprinting Roadmap. Biofabrication 2020, 12, 022002. [Google Scholar] [CrossRef]
- Dobashi, T.; Yamamoto, T. Analysis of Heterogeneous Gelation Dynamics and Their Application to Blood Coagulation. Gels 2018, 4, 59. [Google Scholar] [CrossRef]
- Morgan, F.L.C.; Moroni, L.; Baker, M.B. Dynamic Bioinks to Advance Bioprinting. Adv. Healthc. Mater. 2020, 9, 1901798. [Google Scholar] [CrossRef]
- Khaddour, K.; Johanns, T.M.; Ansstas, G. The Landscape of Novel Therapeutics and Challenges in Glioblastoma Multiforme: Contemporary State and Future Directions. Pharmaceuticals 2020, 13, 389. [Google Scholar] [CrossRef] [PubMed]
- Hatoum, A.; Mohammed, R.; Zakieh, O. The Unique Invasiveness of Glioblastoma and Possible Drug Targets on Extracellular Matrix. Cancer Manag. Res. 2019, 11, 1843–1855. [Google Scholar] [CrossRef] [PubMed]
- Razinia, Z.; Castagnino, P.; Xu, T.; Vázquez-Salgado, A.; Puré, E.; Assoian, R.K. Stiffness-Dependent Motility and Proliferation Uncoupled by Deletion of CD44. Sci. Rep. 2017, 7, 16499. [Google Scholar] [CrossRef] [PubMed]
- Radel, C.; Rizzo, V. Integrin Mechanotransduction Stimulates Caveolin-1 Phosphorylation and Recruitment of Csk to Mediate Actin Reorganization. Am. J. Physiol.-Heart Circ. Physiol. 2005, 288, 936–945. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Material | Advantages | Disadvantages |
|---|---|---|
| Hyaluronic Acid (HA) [44,62,129,130,131,132] |
|
|
| Collagen [16,39,43,64,104,126,133,134,135] |
|
|
| Gelatin–Methacrylate (GelMa) [44,62,115,136,137,138,139,140,141] |
|
|
| Brain Decellularized Tissue (dECM) [124,127,142,143,144,145,146,147,148,149] |
|
|
| Human Blood Plasma [39,43,127,150,151,152,153,154] |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khoonkari, M.; Liang, D.; Kamperman, M.; Kruyt, F.A.E.; van Rijn, P. Physics of Brain Cancer: Multiscale Alterations of Glioblastoma Cells under Extracellular Matrix Stiffening. Pharmaceutics 2022, 14, 1031. https://doi.org/10.3390/pharmaceutics14051031
Khoonkari M, Liang D, Kamperman M, Kruyt FAE, van Rijn P. Physics of Brain Cancer: Multiscale Alterations of Glioblastoma Cells under Extracellular Matrix Stiffening. Pharmaceutics. 2022; 14(5):1031. https://doi.org/10.3390/pharmaceutics14051031
Chicago/Turabian StyleKhoonkari, Mohammad, Dong Liang, Marleen Kamperman, Frank A. E. Kruyt, and Patrick van Rijn. 2022. "Physics of Brain Cancer: Multiscale Alterations of Glioblastoma Cells under Extracellular Matrix Stiffening" Pharmaceutics 14, no. 5: 1031. https://doi.org/10.3390/pharmaceutics14051031
APA StyleKhoonkari, M., Liang, D., Kamperman, M., Kruyt, F. A. E., & van Rijn, P. (2022). Physics of Brain Cancer: Multiscale Alterations of Glioblastoma Cells under Extracellular Matrix Stiffening. Pharmaceutics, 14(5), 1031. https://doi.org/10.3390/pharmaceutics14051031








