The Effects of Various Food Products on Bisphosphonate’s Availability
Abstract
1. Introduction
- most food products will reduce the absorption of bisphosphonate;
- eating a meal in a short period after/before taking bisphosphonates will slow down the gastric emptying kinetics of API (the active pharmaceutical ingredient). It may extend the residence time of the API in the stomach and result in its longer prevalence in the absorption window in the proximal duodenum. Interestingly, if the food does not bind bisphosphonates, the increase in the systemic absorption becomes very likely.
- (1)
- evaluation of the sorption capacity of a series of digested food samples in a simulated intestinal fluid environment;
- (2)
- evaluation of the drug availability after oral administration with a given food environment in simulated conditions;
- (3)
- evaluation of drug sorption using a simulating needle device.
2. Materials and Methods
2.1. Materials
2.2. Simulated Body Fluid
2.2.1. Simulated Gastric Fluid (SGF)
2.2.2. Simulated Intestinal Fluid (SIF)
2.3. Risedronate Sorption
2.3.1. Sorption Capacity of Digested Food Samples
2.3.2. Pharmaceutical Availability after Oral Administration with the Food Environment
2.3.3. Drug Sorption Using a Simulating Needle Device
2.4. HPLC Analysis
2.5. Data Evaluation
3. Results and Discussion
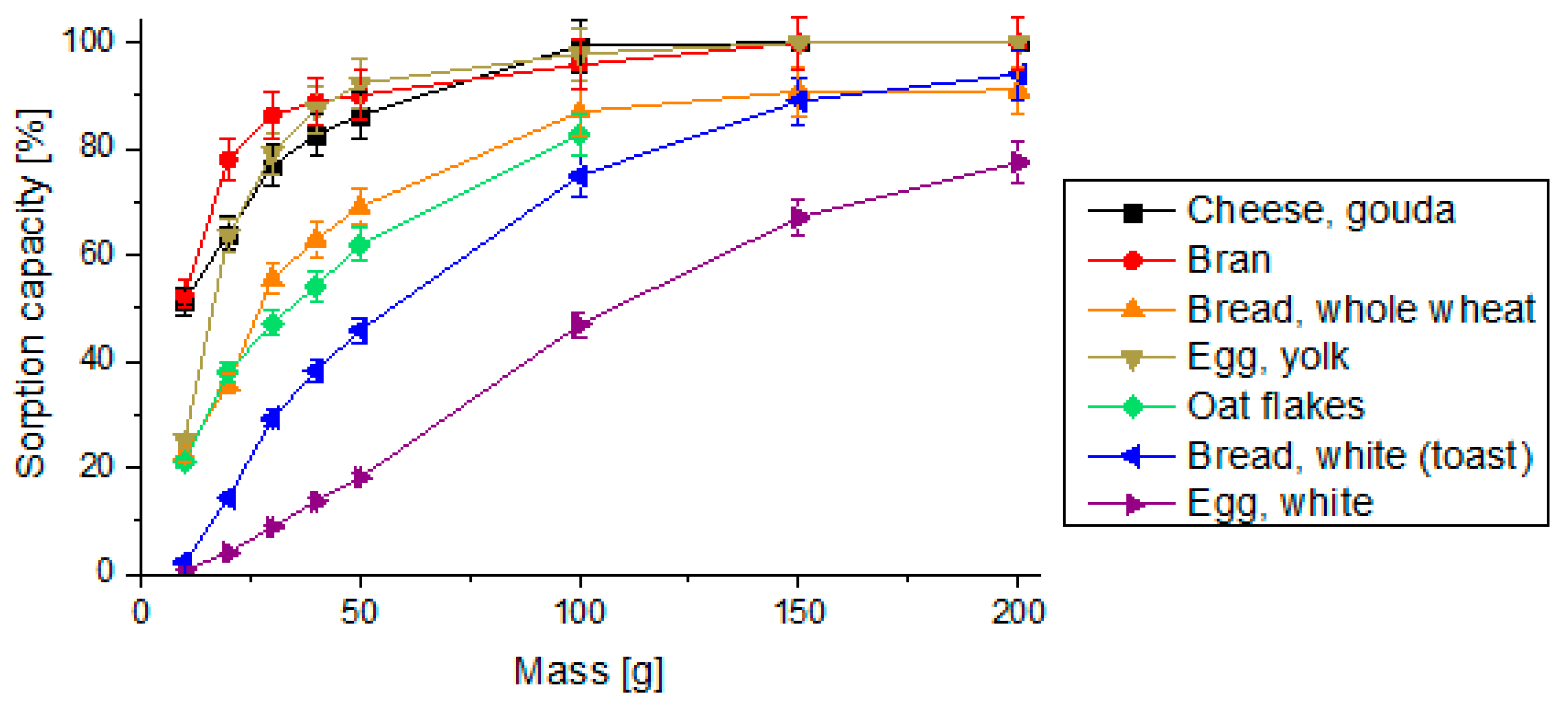
3.1. Sorption Capacity of Digested Food Samples
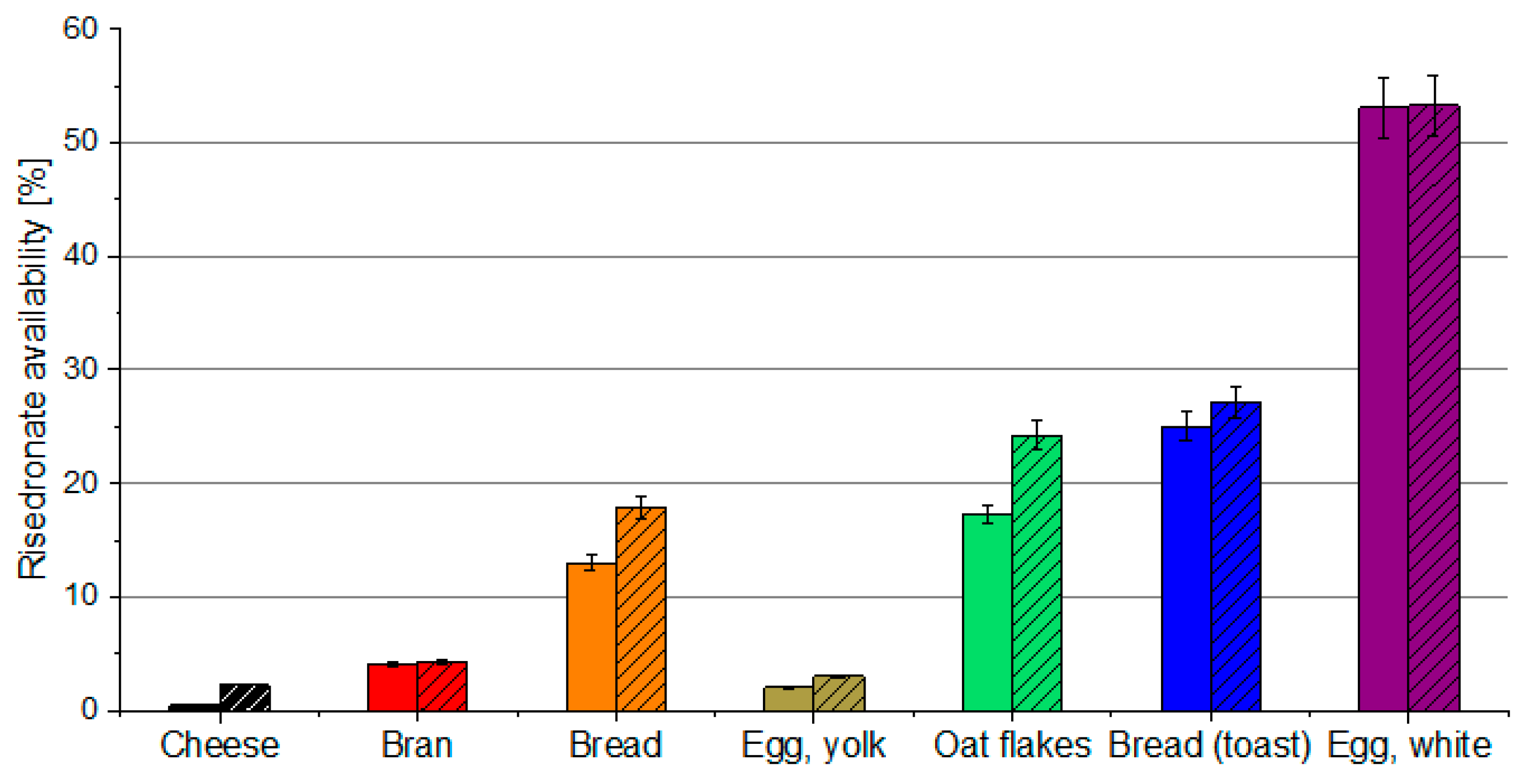
3.2. Drug Availability after Oral Administration with Food Environment
3.3. Drug Sorption Using Simulating Needle Device
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Elisabeth, V.; Castagner, G.; Leroux, J. Biomedical applications of bisphosphonates. J. Control. Release 2013, 167, 175–188. [Google Scholar]
- Bartl, R.; Frisch, B.; Tresckow, B.; Bartl, C. Bisphosphonates in Medical Practice: Actions-Side Effects-Indications-Strategies; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Stappaerts, J.; Wuyts, B.; Tack, J.; Annaert, P.; Augustijns, P. Human and simulated intestinal fluids as solvent systems to explore food effects on intestinal solubility and permeability. Eur. J. Pharm. Sci. 2014, 63, 78–186. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration (FDA). Guidance for Industry: Food-Effect Bioavailability and Fed Bioequivalence Studies; Rockville, M.D., Ed.; US Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research: Rockville, MD, USA, 2002.
- Ogura, Y.; Gonsho, A.; Cyong, J.; Orimo, H. Clinical trial of risedronate in Japanese volunteers: A study on the effects of timing of dosing on absorption. J. Bone Miner. Metab. 2004, 22, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, D.Y.; Heise, M.A.; Pallone, K.A.; Clay, M.E.; Nesbitt, J.D.; Russell, D.A.; Melson, C.W. The effect of dosing regimen on the pharmacokinetics of risedronate. J. Clin. Pharmacol. 1999, 48, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, L.; Charles, P.; Bekker, P.J.; DiGennaro, J.; Johnston, C.C., Jr. Risedronate increases bone mass in an early postmenopausal population: Two years of treatment plus one year of follow-up. J. Clin. Endocrinol. Metab. 1998, 83, 396–402. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lin, J.H. Bisphosphonates: A Review of Their Pharmacokinetic Properties. Bone 1996, 18, 75–85. [Google Scholar] [CrossRef]
- Pazianas, M.; Abrahamsen, B.; Ferrari, S.; Russel, R.G.G. Eliminating the need for fasting with oral administration of bisphosphonates. Ther. Clin. Risk Manag. 2013, 9, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Schlemmer, U.; Frølich, W.; Prieto, R.M.; Grases, F. Phytate in foods and significance for humans: Food sources, intake, processing, bioavailability, protective role and analysis. Mol. Nutr. Food Res. 2009, 53, 330–375. [Google Scholar] [CrossRef]
- FoodData Central-USDA. Available online: https://fdc.nal.usda.gov/index.html (accessed on 2 February 2020).
- Pietrzyńska, M.; Voelkel, A. Optimization of the in-needle extraction device for the direct flow of the liquid sample through the sorbent layer. Talanta 2014, 129, 392–397. [Google Scholar] [CrossRef]
- Pietrzyńska, M.; Czerwiński, M.; Voelkel, A. Poly(vinyl alcohol)/hydroxyapatite monolithic in-needle extraction (MINE) device: Preparation and examination of drug affinity. Eur. J. Pharm. Sci. 2017, 105, 195–202. [Google Scholar] [CrossRef]
- Seo, Y.H.; Chung, Y.H.; Lim, C.; Jeong, J.H. UPLC-UV Method for Determination of Risedronate in Human Urine. J. Chromatogr. Sci. 2014, 52, 713–718. [Google Scholar] [CrossRef]
- Zacharis, C.K.; Tzanavaras, P.D. Determination of bisphosphonate active pharmaceutical ingredients in pharmaceuticals and biological material: A review of analytical methods. J. Pharm. Biomed. Anal. 2008, 48, 483–496. [Google Scholar] [CrossRef]
- Zielińska, M.; Voelkel, A. Risedronate extraction from artificial urine with using monolithic polymer-based anion exchangers. J. Iran. Chem. Soc. 2019, 16, 93–100. [Google Scholar] [CrossRef]



| No. | Product | Ca2+ (mg/100 g) | Mg2+ (mg/100 g) | Fiber (mg/100 g) |
|---|---|---|---|---|
| 1 | Cheese, Gouda | 700 | 29 | 0 |
| 2 | Bran | 389 | 362 | 29.3 |
| 3 | Bread, whole wheat | 161 | 75 | 6 |
| 4 | Egg, yolk | 129 | 5 | 0 |
| 5 | Oat flakes | 80 | 143 | 8 |
| 6 | Bread, white (toast) | 47 | 59 | 4.7 |
| 7 | Egg, white | 7 | 11 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zielińska, M.; Garbacz, G.; Sczodrok, J.; Voelkel, A. The Effects of Various Food Products on Bisphosphonate’s Availability. Pharmaceutics 2022, 14, 717. https://doi.org/10.3390/pharmaceutics14040717
Zielińska M, Garbacz G, Sczodrok J, Voelkel A. The Effects of Various Food Products on Bisphosphonate’s Availability. Pharmaceutics. 2022; 14(4):717. https://doi.org/10.3390/pharmaceutics14040717
Chicago/Turabian StyleZielińska, Monika, Grzegorz Garbacz, Jaroslaw Sczodrok, and Adam Voelkel. 2022. "The Effects of Various Food Products on Bisphosphonate’s Availability" Pharmaceutics 14, no. 4: 717. https://doi.org/10.3390/pharmaceutics14040717
APA StyleZielińska, M., Garbacz, G., Sczodrok, J., & Voelkel, A. (2022). The Effects of Various Food Products on Bisphosphonate’s Availability. Pharmaceutics, 14(4), 717. https://doi.org/10.3390/pharmaceutics14040717







