Nanostructured Drug Delivery Systems for Targeting 5-α-Reductase Inhibitors to the Hair Follicle
Abstract
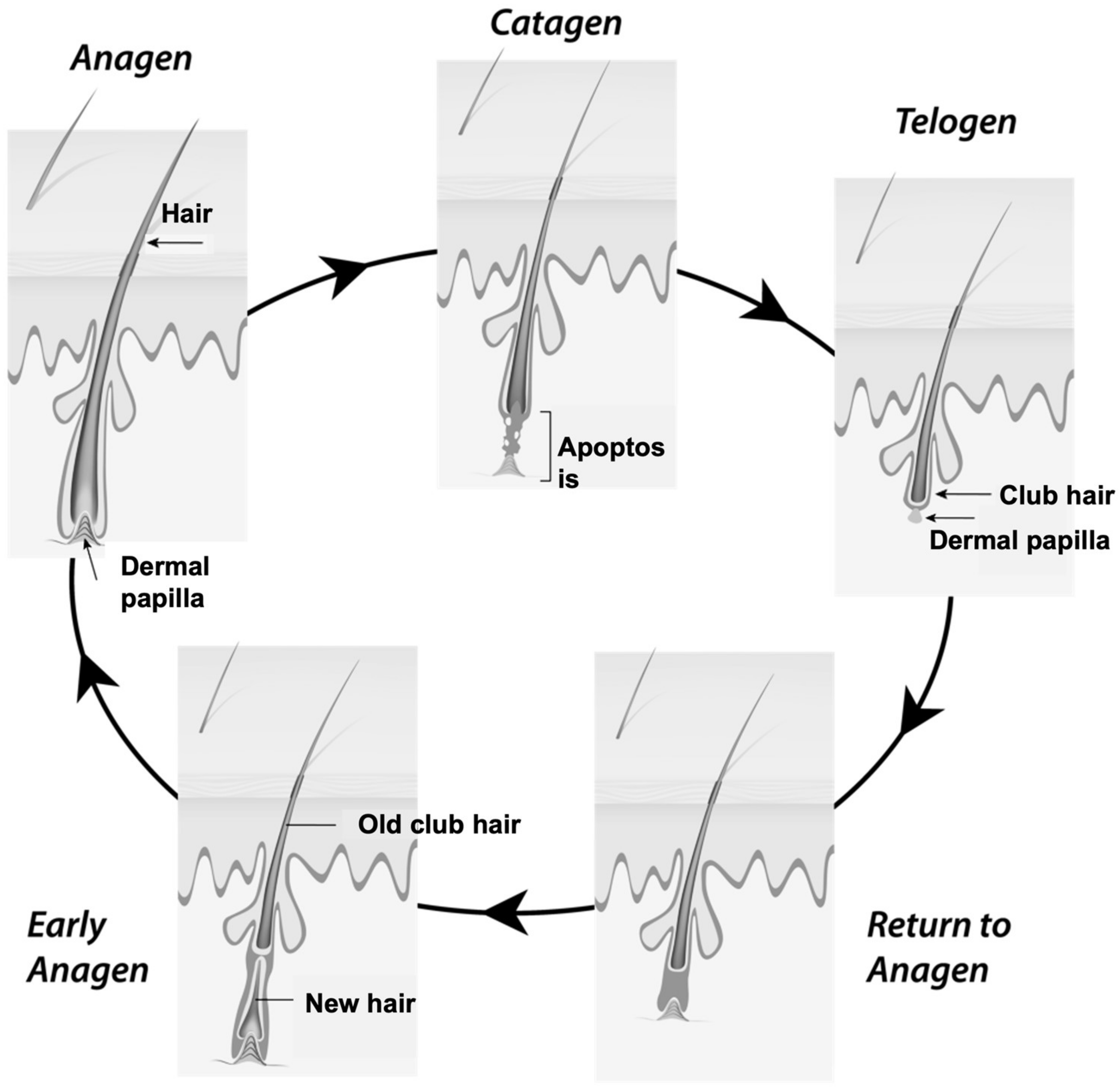
:1. Introduction
2. Current Treatments
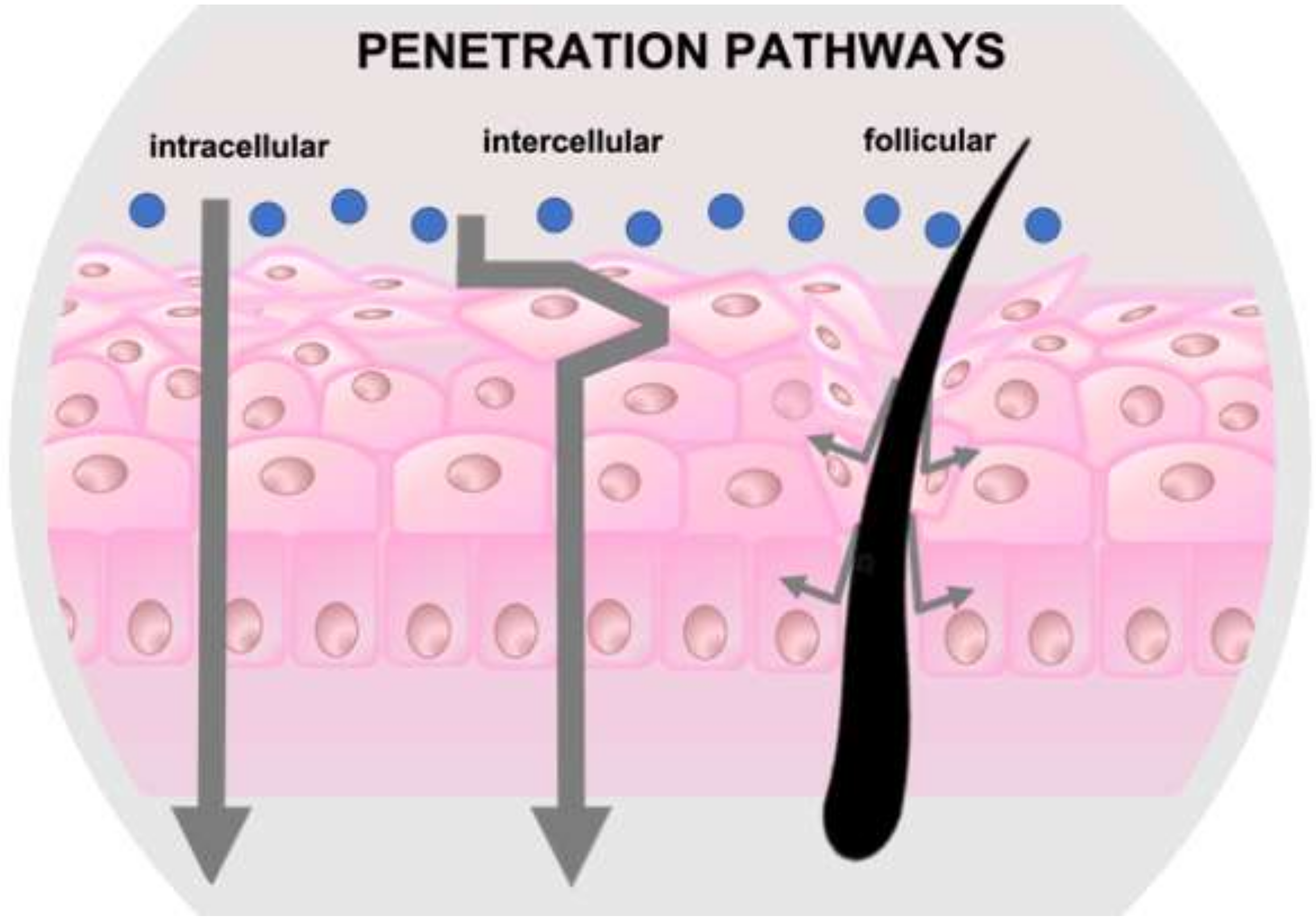
3. Drug Delivery to the Hair Follicle
- The infundibulum, which participates in the increase in surface area and interrupts the epidermal barrier down to the lower portion of the follicles, constituting an efficient reservoir for the permeation of substances;
- The sebaceous gland, which is related to the follicle through the sebaceous duct; and
- The area of the hair bulb, where the stem cells of the germinative matrix are found and appear as an ideal target for the therapy of skin diseases. The cells in the hair matrix may represent potential target sites for hair growth control [17].
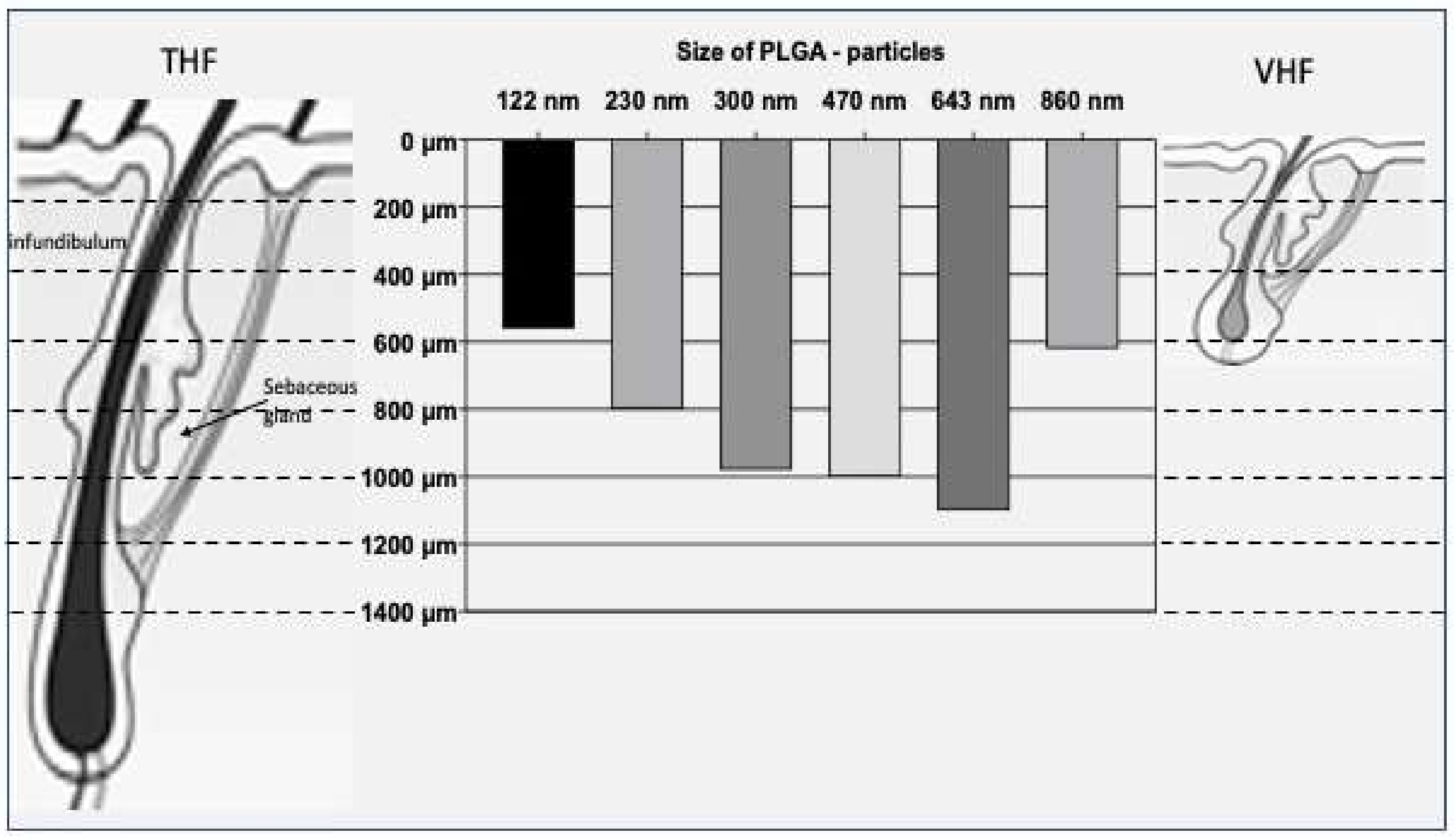
3.1. Influence of the Particle Size
3.2. Influence of Excipients and External Stimuli
4. Topical Formulations Containing 5-α-Reductase Inhibitors: State of the Art
5. Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Stough, D.; Stenn, K.; Haber, R.; Parsley, W.M.; Vogel, J.E.; Whiting, D.A.; Washenik, K. Psychological effect, pathophysiology, and management of androgenetic alopecia in men. Mayo Clin. Proc. 2005, 80, 1316–1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monti, D.; Tampucci, S.; Burgalassi, S.; Chetoni, P.; Lenzi, C.; Pirone, A.; Mailland, F. Topical formulations containing finasteride. Part I: In vitro permeation/penetration study and in vivo pharmacokinetics in hairless rat. J. Pharm. Sci. 2014, 103, 2307–2314. [Google Scholar] [CrossRef] [PubMed]
- Motofei, I.G.; Rowland, D.L.; Baconi, D.L.; Tampa, M.; Sârbu, M.I.; Păunică, S.; Constantin, V.D.; Bălălău, C.; Păunică, I.; Georgescu, S.R. Androgenetic alopecia; drug safety and therapeutic strategies. Expert Opin. Drug Saf. 2018, 17, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Ellis, J.A.; Sinclair, R.; Harrap, S.B. Androgenetic alopecia: Pathogenesis and potential for therapy. Expert Rev. Mol. Med. 2002, 4, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Redler, S.; Messenger, A.G.; Betz, R.C. Genetics and other factors in the aetiology of female pattern hair loss. Exp. Dermatol. 2017, 26, 510–517. [Google Scholar] [CrossRef]
- Starace, M.; Orlando, G.; Alessandrini, A.; Piraccini, B.M. Female Androgenetic Alopecia: An Update on Diagnosis and Manage-ment. Am. J. Clin. Dermatol. 2020, 21, 69–84. [Google Scholar] [CrossRef]
- Nyholt, D.R.; Gillespie, N.A.; Heath, A.C.; Martin, N.G. Genetic basis of male pattern baldness. J. Invest. Dermatol. 2003, 121, 1561–1564. [Google Scholar]
- Mercuri, S.R.; Paolino, G.; Di Nicola, M.R.; Vollono, L. Investigating the Safety and Efficacy of Platelet-Rich Plasma (PRP) Treat-ment for Female Androgenetic Alopecia: Review of the Literature. Medicina 2021, 57, 311. [Google Scholar] [CrossRef]
- Bienová, M.; Kucerová, R.; Fiurásková, M.; Hajdúch, M.; Koláŕ, Z. Androgenetic alopecia and current methods of treatment. Acta Dermatovenerol. Alp. Pannonica. Adriat. 2005, 14, 5–8. [Google Scholar]
- Ustuner, E.T. Cause of androgenic alopecia: Crux of the matter. Plast. Reconstr. Surg. Glob. Open 2013, 1, e64. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J.; Jing, J.; Wu, X.; Lv, Z. Serum Levels of Androgen-Associated Hormones Are Correlated with Curative Effect in Androgenic Alopecia in Young Men. Med. Sci. Monit. 2018, 24, 7770–7777. [Google Scholar] [CrossRef] [PubMed]
- Pantelireis, N.; Higgins, C.A. A bald statement—Current approaches to manipulate miniaturisation focus only on promoting hair growth. Exp. Dermatol. 2018, 27, 959–965. [Google Scholar] [CrossRef] [PubMed]
- York, K.; Meah, N.; Bhoyrul, B.; Sinclair, R. A review of the treatment of male pattern hair loss. Expert Opin. Pharmacother. 2020, 21, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Ashique, S.; Sandhu, N.K.; Haque, S.N.; Koley, K. A Systemic Review on Topical Marketed Formulations, Natural Products, and Oral Supplements to Prevent Androgenic Alopecia: A Review. Nat. Prod. Bioprospect. 2020, 10, 345–365. [Google Scholar] [CrossRef]
- Piraccini, B.M.; Blume-Peytavi, U.; Scarci, F.; Jansat, J.M.; Falqués, M.; Otero, R.; Tamarit, M.L.; Galván, J.; Tebbs, V.; Massana, E. Topical Finasteride Study Group. Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: A phase III, randomized, controlled clinical trial. J. Eur. Acad. Dermatol. Venereol. 2021, 36, 286–294. [Google Scholar] [CrossRef]
- Fang, C.L.; Aljuffali, I.A.; Li, Y.C.; Fang, J.Y. Delivery and targeting of nanoparticles into hair follicles. Ther. Deliv. 2014, 5, 991–1006. [Google Scholar] [CrossRef]
- Patzelt, A.; Richter, H.; Knorr, F.; Schäfer, U.; Lehr, C.M.; Dähne, L.; Sterry, W.; Lademann, J. Selective follicular targeting by modification of the particle sizes. J. Control. Release 2011, 150, 45–48. [Google Scholar] [CrossRef]
- Blume-Peytavi, U.; Vogt, A. Human hair follicle: Reservoir function and selective targeting. Br. J. Dermatol. 2011, 165 (Suppl. 2), 13–17. [Google Scholar] [CrossRef]
- Vogt, A.; Mandt, N.; Lademann, J.; Schaefer, H.; Blume-Peytavi, U. Follicular targeting—A promising tool in selective dermatotherapy. J. Investig. Dermatol. Symp. Proc. 2005, 10, 252–255. [Google Scholar] [CrossRef] [Green Version]
- Kwack, M.H.; Sung, Y.K.; Chung, E.J.; Im, S.U.; Ahn, J.S.; Kim, M.K.; Kim, J.C. Dihydrotestosterone-inducible dickkopf 1 from balding dermal papilla cells causes apoptosis in follicular keratinocytes. J. Invest. Dermatol. 2008, 128, 262–269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costa, C.; Cavaco-Paulo, A.; Matamá, T. Mapping hair follicle-targeted delivery by particle systems: What has science accom-plished so far? Int. J. Pharm. 2021, 610, 121273. [Google Scholar] [CrossRef] [PubMed]
- Lademann, J.; Patzelt, A.; Richter, H.; Antoniou, C.; Sterry, W.; Knorr, F. Determination of the cuticula thickness of human and porcine hairs and their potential influence on the penetration of nanoparticles into the hair follicles. J. Biomed. Opt. 2009, 14, 021014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ossadnik, M.; Richter, H.; Teichmann, A.; Koch, S.; Schafer, U.; Wepf, R.; Sterry, W.; Lademann, J. Investigation of differences in follicular penetration of particle- and nonparticle-containing emulsions by laser scanning microscopy. Laser Phys. 2006, 16, 747–750. [Google Scholar] [CrossRef]
- Roque, L.V.; Dias, I.S.; Cruz, N.; Rebelo, A.; Roberto, A.; Rijo, P.; Reis, C.P. Design of Finasteride-Loaded Nanoparticles for Potential Treatment of Alopecia. Skin Pharmacol. Physiol. 2017, 30, 197–204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Angelo, T.; El-Sayed, N.; Jurisic, M.; Koenneke, A.; Gelfuso, G.M.; Cunha-Filho, M.; Taveira, S.F.; Lemor, R.; Schneider, M.; Gratieri, T. Effect of physical stimuli on hair follicle deposition of clobetasol-loaded Lipid Nanocarriers. Sci. Rep. 2020, 10, 176. [Google Scholar] [CrossRef] [Green Version]
- Aljuffali, I.A.; Sung, C.T.; Shen, F.M.; Huang, C.T.; Fang, J.Y. Squarticles as a lipid nanocarrier for delivering diphencyprone and minoxidil to hair follicles and human dermal papilla cells. AAPS J. 2014, 16, 140–150. [Google Scholar] [CrossRef] [Green Version]
- Lademann, J.; Knorr, F.; Richter, H.; Jung, S.; Meinke, M.C.; Rühl, E.; Alexiev, U.; Calderon, M.; Patzelt, A. Hair follicles as a target structure for nanoparticles. J. Innov. Opt. Health Sci. 2014, 8, 1530004. [Google Scholar] [CrossRef] [Green Version]
- Toll, R.; Jacobi, U.; Richter, H.; Lademann, J.; Schaefer, H.; Blume-Peytavi, U. Penetration profile of microspheres in follicular targeting of terminal hair follicles. J. Investig. Dermatol. 2004, 123, 168–176. [Google Scholar] [CrossRef] [Green Version]
- Rancan, F.; Papakostas, D.; Hadam, S.; Hackbarth, S.; Delair, T.; Primard, C.; Verrier, B.; Sterry, W.; Blume-Peytavi, U.; Vogt, A. Investigation of polylactic acid (PLA) nanoparticles as drug delivery systems for local dermatotherapy. Pharm. Res. 2009, 26, 2027–2036. [Google Scholar] [CrossRef]
- Schneider, M.; Stracke, F.; Hansen, S.; Schaefer, U.F. Nanoparticles and their interactions with the dermal barrier. Dermatoendo-crinology 2009, 1, 197–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, H.; Chang, X.; Du, D.; Liu, W.; Liu, J.; Weng, T.; Yang, Y.; Xu, H.; Yang, X. Podophyllotoxin-loaded solid lipid nanoparticles for epidermal targeting. J. Control. Release 2006, 110, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.; Kang, H.S.; Park, W.S.; Han, S.H.; Kim, J.; Chang, I.S. Transdermal delivery of mixnoxidil with block copolymer nanoparticles. J. Control. Release 2004, 97, 477–484. [Google Scholar] [CrossRef]
- Alvarez-Román, R.; Naik, A.; Kalia, Y.N.; Guy, R.H.; Fessi, H. Skin penetration and distribution of polymeric nanoparticles. J. Control. Release 2004, 99, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Vogt, A.; Combadiere, B.; Hadam, S.; Stieler, K.M.; Lademann, J.; Schaefer, H.; Autran, B.; Sterry, W.; Blume-Peytavi, U. 40 nm, but not 750 or 1500 nm, nanoparticles enter epidermal CD1a+ cells after transcutaneous application on human skin. J. Invest. Dermatol. 2006, 126, 1316–1322. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Price, G.J.; Guy, R.H. Disposition of nanoparticles and an associated lipophilic permeant following topical application to the skin. Mol. Pharm. 2009, 6, 1441–1448. [Google Scholar] [CrossRef] [Green Version]
- Lekki, J.; Stachura, Z.; Dabros, W.; Stachura, J.; Menzel, F.; Reinert, T.; Butz, T.; Jan, L.U.P.; Gontier, E.; Ynsa, M.D.; et al. On the follicular pathway of percutaneous uptake of nanoparticles: Ion microscopy and autoradiography studies. Nucl. Instrum. Meth. Phys. Res. B 2007, 260, 174–177. [Google Scholar] [CrossRef]
- Mahe, B.; Vogt, A.; Liard, C.; Duffy, D.; Abadie, V.; Bonduelle, O.; Boissonnas, A.; Sterry, W.; Verrier, B.; Blume-Peytavi, U.; et al. Nanoparticle-based targeting of vaccine compounds to skin antigen-presenting cells by hair follicles and their transport in mice. J. Invest. Dermatol. 2009, 129, 1156–1164. [Google Scholar] [CrossRef] [Green Version]
- Schaefer, H.; Lademann, J. The role of follicular penetration. A differential view. Skin Pharmacol. Appl. Skin Physiol. 2001, 14 (Suppl. 1), 23–27. [Google Scholar] [CrossRef]
- Fresta, M.; Mancuso, A.; Cristiano, M.C.; Urbanek, K.; Cilurzo, F.; Cosco, D.; Iannone, M.; Paolino, D. Targeting of the Pilosebaceous Follicle by Liquid Crystal Nanocarriers: In Vitro and In Vivo Effects of the Entrapped Minoxidil. Pharmaceutics 2020, 12, 1127. [Google Scholar] [CrossRef]
- Pelikh, O.; Keck, C.M. Hair Follicle Targeting and Dermal Drug Delivery with Curcumin Drug Nanocrystals—Essential Influ-ence of Excipients. Nanomaterials 2020, 10, 2323. [Google Scholar] [CrossRef]
- Matos, B.N.; Reis, T.A.; Gratieri, T.; Gelfuso, G.M. Chitosan nanoparticles for targeting and sustaining minoxidil sulphate delivery to hair follicles. Int. J. Biol. Macromol. 2015, 75, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.A.D.; Taveira, S.F.; Lima, E.M.; Marreto, R.N. In vitro skin penetration of clobetasol from lipid nanoparticles: Drug extraction and quantitation in different skin layers. Braz. J. Pharm. Sci. 2012, 48, 811–817. [Google Scholar] [CrossRef] [Green Version]
- Lademann, J.; Richter, H.; Teichmann, A.; Otberg, N.; Blume-Peytavi, U.; Luengo, J.; Weiss, B.; Schaefer, U.F.; Lehr, C.M.; Wepf, R.; et al. Nanoparticles—An efficient carrier for drug delivery into the hair follicles. Eur. J. Pharm. Biopharm. 2007, 66, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Pelikh, O.; Eckert, R.W.; Pinnapireddy, S.R.; Keck, C.M. Hair follicle targeting with curcumin nanocrystals: Influence of the for-mulation properties on the penetration efficacy. J. Control. Release 2021, 329, 598–613. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.N.; Schulte, H.L.; Duarte, N.; Lima, E.M.; Sá-Barreto, L.L.; Gratieri, T.; Gelfuso, G.M.; Cunha-Filho, M.S.S. Solid effervescent formulations as new approach for topical minoxidil delivery. Eur. J. Pharm. Sci. 2017, 96, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Patzelt, A.; Lademann, J. Drug delivery to hair follicles. Expert Opin. Drug Deliv. 2013, 10, 787–797. [Google Scholar] [CrossRef]
- Ahmed, T.A.; Al-Abd, A.M. Effect of finasteride particle size reduction on its pharmacokinetic, tissue distribution and cellular permeation. Drug Deliv. 2018, 25, 555–563. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.H.; Yeom, D.W.; Song, Y.S.; Cho, H.R.; Choi, Y.S.; Kang, M.J.; Choi, Y.W. Improved oral absorption of dutasteride via Soluplus®-based supersaturable self-emulsifying drug delivery system (S-SEDDS). Int. J. Pharm. 2015, 478, 341–347. [Google Scholar] [CrossRef]
- Tampucci, S.; Burgalassi, S.; Chetoni, P.; Lenzi, C.; Pirone, A.; Mailland, F.; Caserini, M.; Monti, D. Topical formulations containing finasteride. Part II: Determination of finasteride penetration into hair follicles using the differential stripping technique. J. Pharm. Sci. 2014, 103, 2323–2329. [Google Scholar] [CrossRef]
- Bhatia, S. Nanoparticles Types, Classification, Characterization, Fabrication Methods and Drug Delivery Applications. In Natural Polymer Drug Delivery Systems; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Terreni, E.; Chetoni, P.; Tampucci, S.; Burgalassi, S.; Al-Kinani, A.A.; Alany, R.G.; Monti, D. Assembling Surfactants-Mucoadhesive Polymer Nanomicelles (ASMP-Nano) for Ocular Delivery of Cyclosporine-A. Pharmaceutics 2020, 12, 253. [Google Scholar] [CrossRef] [Green Version]
- Fang, J.Y.; Hwang, T.L.; Huang, Y.L. Liposomes as Vehicles for Enhancing Drug Delivery Via Skin Routes. Curr. Nanosci. 2006, 2, 55–70. [Google Scholar] [CrossRef]
- Carita, A.C.; Eloy, J.O.; Chorilli, M.; Lee, R.J.; Leonardi, G.R. Recent Advances and Perspectives in Liposomes for Cutaneous Drug Delivery. Curr. Med. Chem. 2018, 25, 606–635. [Google Scholar] [CrossRef] [PubMed]
- Noor, N.M.; Abdul-Aziz, A.; Sheikh, K.; Somavarapu, S.; Taylor, K.M.G. In Vitro Performance of Dutasteride-Nanostructured Lipid Carriers Coated with Lauric Acid-Chitosan Oligomer for Dermal Delivery. Pharmaceutics 2020, 12, 994. [Google Scholar] [CrossRef] [PubMed]
- Pervaiz, F.; Saleem, M.; Ashames, A.; Rehmani, S.; Qaiser, R.; Noreen, S.; Murtaza, G. Development and ex-vivo skin permeation studies of finasteride–poly(lactic acid-co-glycolic acid) and minoxidil–chitosan nanoparticulate systems. J. Bioact. Compat. Polym. 2020, 35, 77–91. [Google Scholar] [CrossRef]
- Patzelt, A.; Lademann, J. Recent advances in follicular drug delivery of nanoparticles. Expert Opin. Drug Deliv. 2020, 17, 49–60. [Google Scholar] [CrossRef]
- Krishnan, V.; Mitragotri, S. Nanoparticles for topical drug delivery: Potential for skin cancer treatment. Adv. Drug Deliv. Rev. 2020, 153, 87–108. [Google Scholar] [CrossRef]
- Tabbakhian, M.; Tavakoli, N.; Jaafari, M.R.; Daneshamouz, S. Enhancement of follicular delivery of finasteride by liposomes and niosomes 1. In vitro permeation and in vivo deposition studies using hamster flank and ear models. Int. J. Pharm. 2006, 323, 1–10. [Google Scholar] [CrossRef]
- Rao, Y.F.; Zheng, F.Y.; Zhang, X.G.; Gao, J.Q.; Liang, W.Q. In vitro percutaneous permeation and skin accumulation of finasteride using vesicular ethosomal carriers. AAPS Pharmscitech 2008, 9, 860–865. [Google Scholar] [CrossRef] [Green Version]
- Noor, N.M.; Sheikh, K.; Somavarapu, S.; Taylor, K.M.G. Preparation and characterization of dutasteride-loaded nanostructured lipid carriers coated with stearic acid-chitosan oligomer for topical delivery. Eur. J. Pharm. Biopharm. 2017, 117, 372–384. [Google Scholar] [CrossRef]
- Afiune, L.A.F.; Ushirobira, C.Y.; Barbosa, D.P.P.; de Souza, P.E.N.; Leles, M.I.G.; Cunha-Filho, M.; Gelfuso, G.M.; Soler, M.A.G.; Gratieri, T. Novel iron oxide nanocarriers loading finasteride or dutasteride: Enhanced skin penetration for topical treatment of alopecia. Int. J. Pharm. 2020, 587, 119709. [Google Scholar] [CrossRef] [PubMed]
- Wilczewska, A.Z.; Niemirowicz, K.; Markiewicz, K.H.; Car, H. Nanoparticles as drug delivery systems. Pharmacol Rep. 2012, 64, 1020–1037. [Google Scholar] [CrossRef]
- Kumar, P.; Gulbake, A.; Jain, S.K. Liposomes a vesicular nanocarrier: Potential advancements in cancer chemotherapy. Crit. Rev. Ther. Drug Carrier Syst. 2012, 29, 355–419. [Google Scholar] [CrossRef] [PubMed]
- Shoukath, A.M.; Ateeq, A.; Moghis, A.; Saifuddin, S.; Ahmad, I. Lipid Based Pharmaceutical Preparations for Oral and TopiCal Application; Their Compositions, Methods, and Uses. Thereof. Patent WO 2,010,036,947 A2, 27 September 2008. [Google Scholar]
- Hasson, V.; Castiglioni, M. Topical Formulations for Treating Dermatological Disorders Including Male Pattern. Baldness. Patent WO 2,019,084,503 A1, 2 May 2019. [Google Scholar]
- Nam, K. Nanoliposome-Microbubble Assembly Encapsulating Hair Loss Treatment Drug and Composition Comprising Same for Alleviation or Treatment of Hair. Loss. Patent WO 2,019,245,142 A1, 26 December 2019. [Google Scholar]
- Kumar, R.; Singh, B.; Bakshi, G.; Katare, O.P. Development of liposomal systems of finasteride for topical applications: Design, characterization, and in vitro evaluation. Pharm. Dev. Technol. 2007, 12, 591–601. [Google Scholar] [CrossRef]
- Mir-Palomo, S.; Nácher, A.; Vila-Busó, M.A.O.; Caddeo, C.; Manca, M.L.; Saurí, A.R.; Escribano-Ferrer, E.; Manconi, M.; Díez-Sales, O. Co-loading of finasteride and baicalin in phospholipid vesicles tailored for the treatment of hair disorders. Nanoscale 2020, 12, 16143–16152. [Google Scholar] [CrossRef] [PubMed]
- Caon, T.; Porto, L.C.; Granada, A.; Tagliari, M.P.; Silva, M.A.; Simões, C.M.; Borsali, R.; Soldi, V. Chitosan-decorated polystyrene-b-poly(acrylic acid) polymersomes as novel carriers for topical delivery of finasteride. Eur. J. Pharm. Sci. 2014, 52, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Ushirobira, C.Y.; Afiune, L.A.F.; Pereira, M.N.; Cunha-Filho, M.; Gelfuso, G.M.; Gratieri, T. Dutasteride nanocapsules for hair follicle targeting: Effect of chitosan-coating and physical stimulus. Int. J. Biol. Macromol. 2020, 151, 56–61. [Google Scholar] [CrossRef]
- Irfan, M.M.; Shah, S.U.; Khan, I.U.; Munir, M.U.; Khan, N.R.; Shah, K.U.; Rehman, S.U.; Sohaib, M.; Basit, H.M.; Mahmood, S. Physicochemical Characterization of Finasteride Nanosystem for Enhanced Topical Delivery. Int. J. Nanomed. 2021, 16, 1207–1220. [Google Scholar] [CrossRef]
- El-Tamimy, M. Pharmaceutical. Formulation. Patent WO 2,017,096,427 A1, 15 June 2017. [Google Scholar]
- Mailland, F.; Mura, E. Film-Forming Liquid Formulations for Drug Release to Hair and. Scalp. Patent WO 2,010,015,556 A1, 11 February 2010. [Google Scholar]
- Ahn, B.K.; Ko, K.S.; Park, S.H.; Ki, M.H.; Lim, J.L. Pharmaceutical Composition Comprising 5alpha-Reductase. Inhibitor. Patent WO 2,016,013,829 A1, 28 January 2016. [Google Scholar]
- Venkatraman, S.; Seah, X.Y.; Lim, S.G.; Thng, T.G.; Boey, Y.C.F. An Injectable Composition for Localized Controlled and Sustained Delivery. System. Patent WO 2,018,070,940 A1, 19 April 2018. [Google Scholar]
| Pharmaceutical Form/Route | Therapeutic Indications | Regulatory Authority |
|---|---|---|
| Minoxidil | ||
| Solution, 2%; topical | AGA in male and female | AIFA, FDA |
| Solution 5%; topical | AGA in male | AIFA, FDA |
| Solution 20 mg/mL; topical | AGA in male and female | AIFA, FDA |
| Solution 50 mg/mL; topical | AGA in male | AIFA, FDA |
| Aerosol, foam, 5%; topical | AGA in male and female | AIFA, FDA |
| Finasteride | ||
| Tablets 1 mg; oral | AGA in male | AIFA, FDA |
| Sprayable solution 2.275 mg/mL; topical | AGA in male | AIFA |
| Formulation | Particle Size | Target Region | Reference |
|---|---|---|---|
| Hydrogel suspension of PLGA particles containing fluoresceinamine |
|
| [17] |
| Aqueous suspension of silica particles containing fluoresceinamine |
|
| [7] |
| Colloidal suspension of polymeric nanoparticles with finasteride in water and Pluronic® F68 | 316.5 nm | hair follicle | [24] |
| Nanostructured lipid carriers containing clobetasol | 173.80 nm | hair follicle (with deepest depth obtained after 12 h) | [25] |
| Aqueous suspension of fluorescein labeled nanoparticles | 320 nm | 1500 μm depth | [27] |
| Fluoresbrite Yellow Green rigid Carboxylate Microspheres |
|
| [28] |
| Suspension of polylactic acid (PLA) nanoparticles with fluorescent dye | 228 nm and 365 nm | penetration in hair follicles and release of loaded dyes into the surrounding tissues. | [29] |
| Polystyrene and iron-based particles | 20 nm | penetrate hair follicles | [30] |
| Podophyllotoxin-loaded solid lipid nanoparticles stabilized by poloxamer 188 (P-SLN) and soybean lecithin (T-SLN) | 73.4 nm (P-SLN, negatively charged) and 123.1 nm (T-SLN) |
| [31] |
| Aqueous nanoparticle suspension based on poly(ε-caprolactone)-block-poly(ethylene glycol) containing minoxidil | 40 nm (S-NP) and 130-nm (L-NP) | hair follicles is the main pathway of this minoxidil-loaded nanoparticles | [32] |
| Polystyrene nanoparticles containing fluorescein 5-isothiocyanate | 20 nm and 200 nm | preferential accumulation of FITC nanoparticles in the follicular openings | [33] |
| Nanoparticles suspension in PBS pH 7.4 with fluorescent probes | 40 nm 750 or 1500 nm |
| [34] |
| Polymeric nanoparticles | <50 nm | retention in skin furrows and around follicles | [35] |
| Titanium oxide nanoparticles | 20 nm |
| [36] |
| Polystyrene nanoparticles | 40 and 200 nm |
| [37] |
| Polymeric microparticles | < 3 μm 3 and 10 μm >10 μm |
| [38] |
| Type and Concentration of Excipient | Effect on Passive Dermal Diffusion | Effect on Hair Follicle Targeting |
|---|---|---|
| Glycerol 2% | ↑ | ↔ |
| Glycerol 5% * | ↑↑↑↑ | ↓ |
| Urea 5% | ↑↑ | ↔ |
| Urea 10% | ↑ | ↓ |
| Propylene glycol 5% ** | ↑↑↑↑ | ↑↑↑ |
| EtOH 2% *** | ↓ | ↑↑↑ |
| Olive oil 2% | ↔ | ↑ |
| Formulation | Active Principle | Lipid Component | Particle Size and Surface Charge | Reference |
|---|---|---|---|---|
| Liposomes | Finasteride or minoxidil | cholesterol, cholesterol derivatives, sterols, PC ¹, PE ², PI ³, SPH ⁴, phosphatidic acid, mono-, di- and triglycerides derivatives galactolipids, mannolipids. | 30–1000 nm n.a. * | [64] |
| Liposomes dispersed in aqueous gel (siliconic derivative a with occlusive effect) | Finasteride or dutasteride | PC ¹, PE ², PI ³, SPH ⁴, phosphatidic acid or sterols (cholesterol) | n.a. * | [65] |
| Liposomes associated with microbubbles | Finasteride, dutasteride or minoxidil | PC ¹, cholesterol and cationic phospholipids (e.g., DPPE ⁵) | n.a. * | [66] |
| Liposomes and Niosomes | Finasteride | DMPC 6, dicetyl phosphate, cholesterol, Brij 72 (polyoxyethylene 2 stearyl ether), Brij 76 (polyoxyethylene 10 stearyl ether), Brij 97 (polyoxyethylene 10 oleyl ether), Span 40 (sorbitan monopalmitate) | 1.9–4.4 μm negatively charged | [58] |
| Multilamellar liposomes | Finasteride | PC ¹, cholesterol, dicetyl phosphate |
15.4–24.1 μm neutral and negatively charged | [67] |
| Liposomes, hyalurosomes, glycerosomes and glycerol-hyalurosomes | Finasteride and baicalin | Lipoid® S75, soybean lecithin |
65–110 nm negatively charged | [68] |
| Formulation | Active Principle | Composition | Strategy | Reference |
|---|---|---|---|---|
| Chitosan-based | Finasteride, dutasteride, others | Chitosan, chitosan derivatives, volatile solvent (i.e., ethanol) | Film-forming solution for topical application allowing long lasting scalp contact and continuous drug release | [73] |
| Lipidic solution | Finasteride o dutasteride in association with corticosteroids and/or FANS | Sorbitan esters with unsaturated fatty acids, phospholipids and retinyl palmitate, tocopherol acetate, cholesterol | Liquid crystal formation after formulation exposure to aqueous fluid after injection or topical application allowing a sustained drug release | [74] |
| Polymeric non aqueous solution | Finasteride, dutasteride, minoxidil | biocompatible and biodegradable polymers/water miscible organic solvent | Subcutaneous injection allow in situ gel depot formation and controlled and substained drug release | [75] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tampucci, S.; Paganini, V.; Burgalassi, S.; Chetoni, P.; Monti, D. Nanostructured Drug Delivery Systems for Targeting 5-α-Reductase Inhibitors to the Hair Follicle. Pharmaceutics 2022, 14, 286. https://doi.org/10.3390/pharmaceutics14020286
Tampucci S, Paganini V, Burgalassi S, Chetoni P, Monti D. Nanostructured Drug Delivery Systems for Targeting 5-α-Reductase Inhibitors to the Hair Follicle. Pharmaceutics. 2022; 14(2):286. https://doi.org/10.3390/pharmaceutics14020286
Chicago/Turabian StyleTampucci, Silvia, Valentina Paganini, Susi Burgalassi, Patrizia Chetoni, and Daniela Monti. 2022. "Nanostructured Drug Delivery Systems for Targeting 5-α-Reductase Inhibitors to the Hair Follicle" Pharmaceutics 14, no. 2: 286. https://doi.org/10.3390/pharmaceutics14020286
APA StyleTampucci, S., Paganini, V., Burgalassi, S., Chetoni, P., & Monti, D. (2022). Nanostructured Drug Delivery Systems for Targeting 5-α-Reductase Inhibitors to the Hair Follicle. Pharmaceutics, 14(2), 286. https://doi.org/10.3390/pharmaceutics14020286










