Photochemical Restoration of Light Sensitivity in the Degenerated Canine Retina
Abstract
1. Introduction
2. Material and Methods
2.1. Animals
2.2. Intravitreal Injections with and without Prior Vitrectomy
2.3. MEA Recording
2.4. qRT-PCR
2.5. Histology and Immunohistochemistry
3. Results
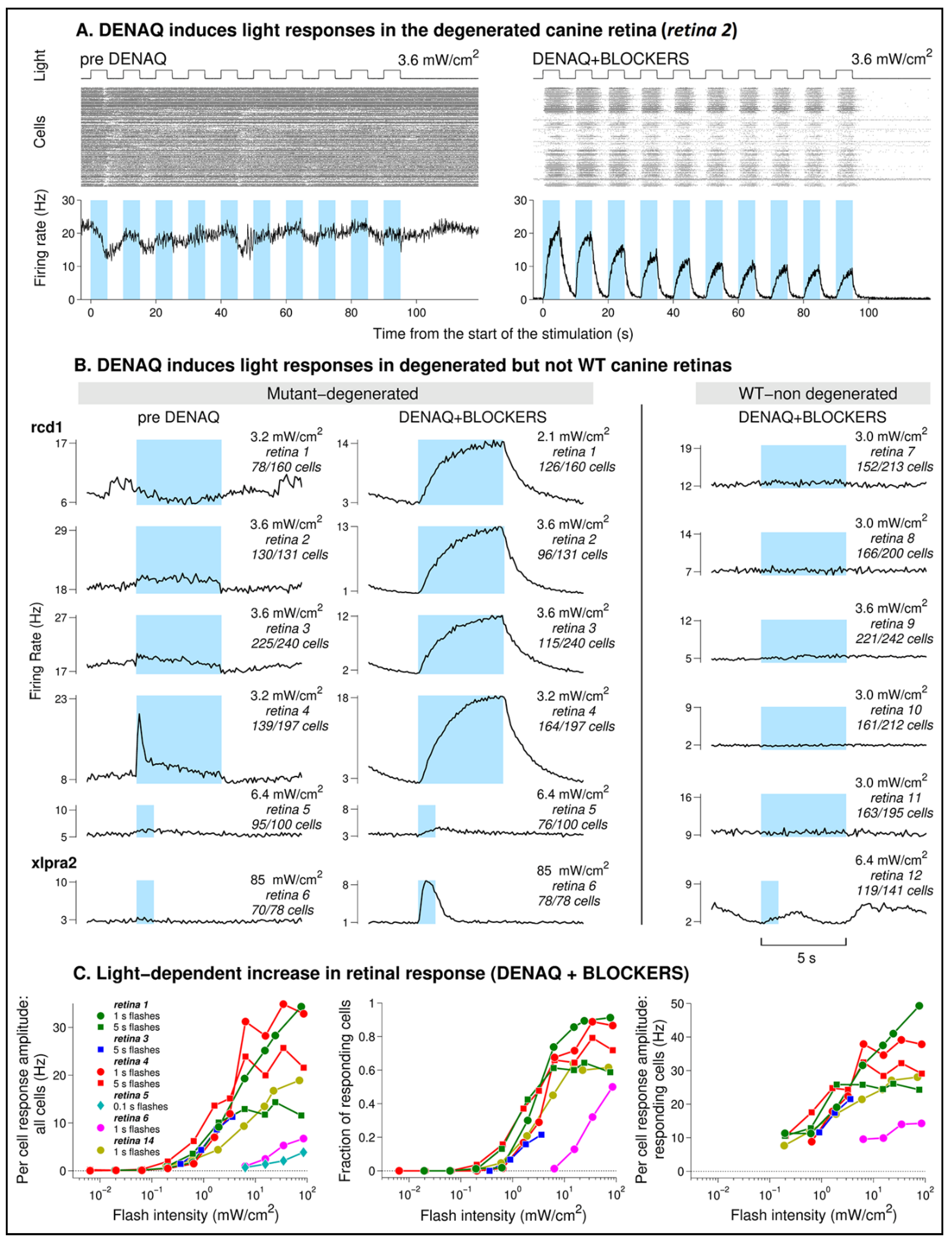
3.1. MEA Analysis following Ex Vivo Application of DENAQ on Degenerated and WT Retinal Explants
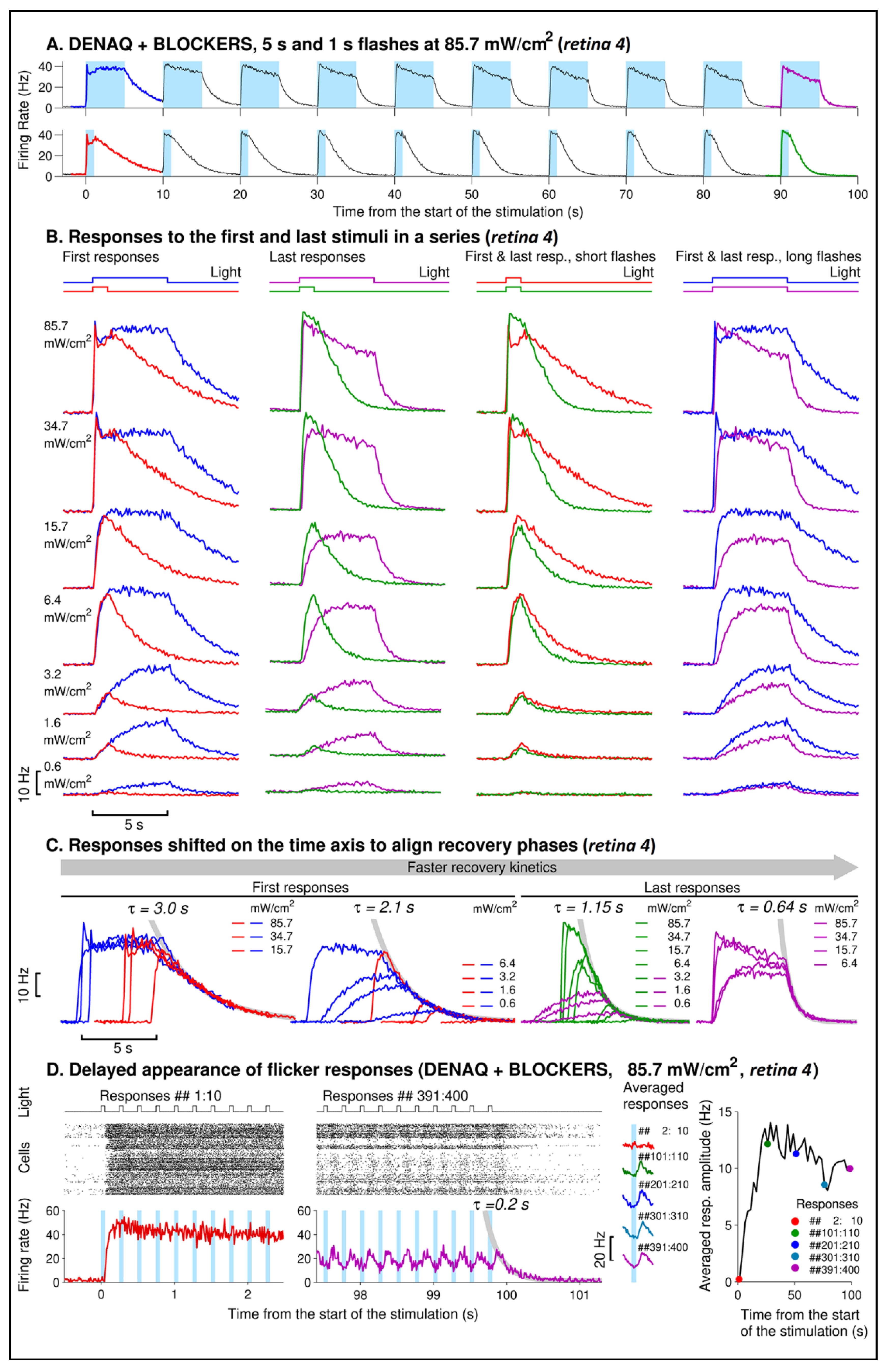
3.2. Kinetics of DENAQ-Driven RGC Firing in Degenerated Retinas
3.3. MEA Analysis of Cone versus DENAQ-Driven RGC Firing
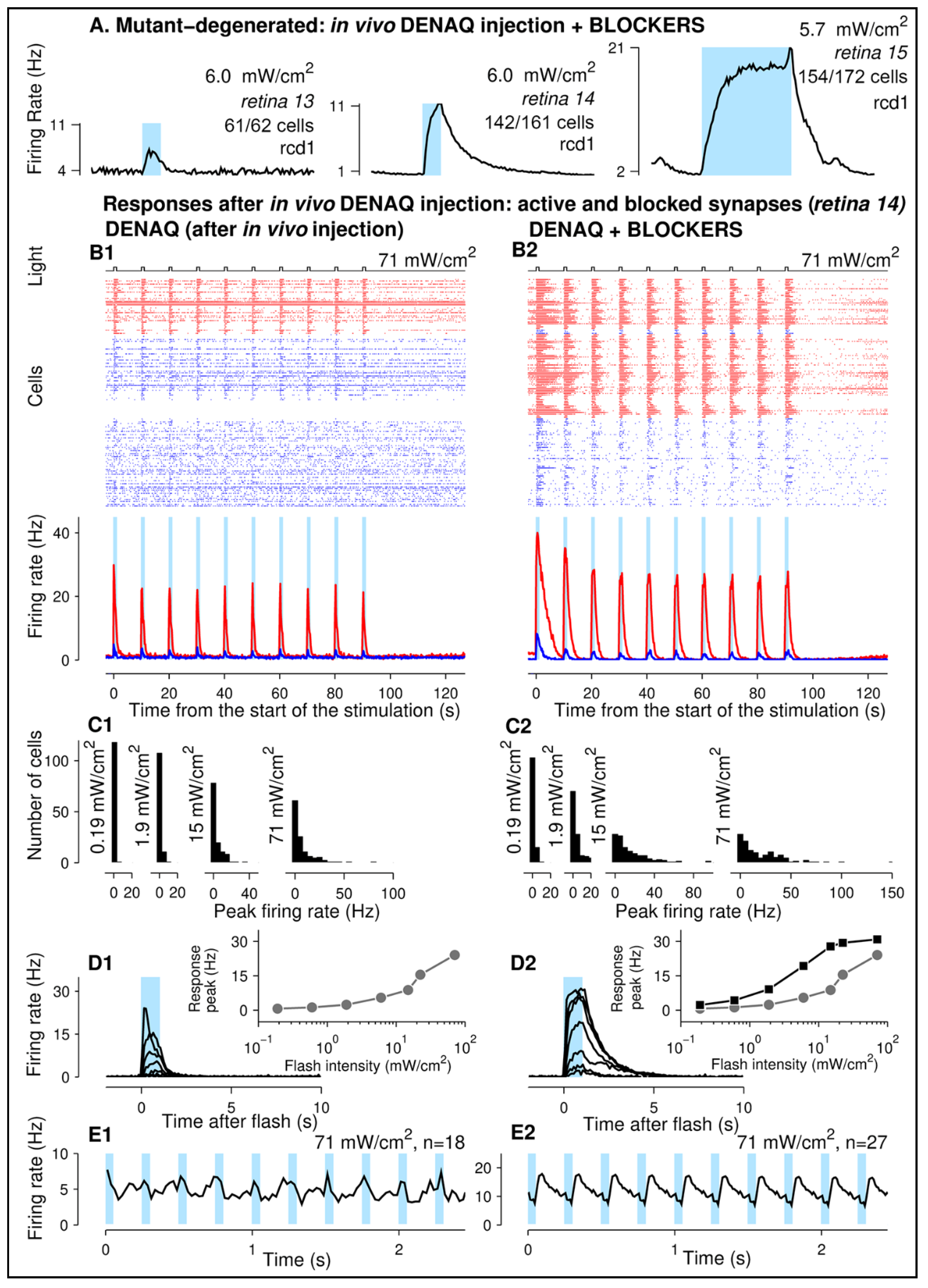
3.4. DENAQ Confers Light Sensitivity to Mutant Canine Retinas following Intravitreal Injection
3.5. P2X Receptors and HCN Channels Are Upregulated in Degenerated Canine Retinas
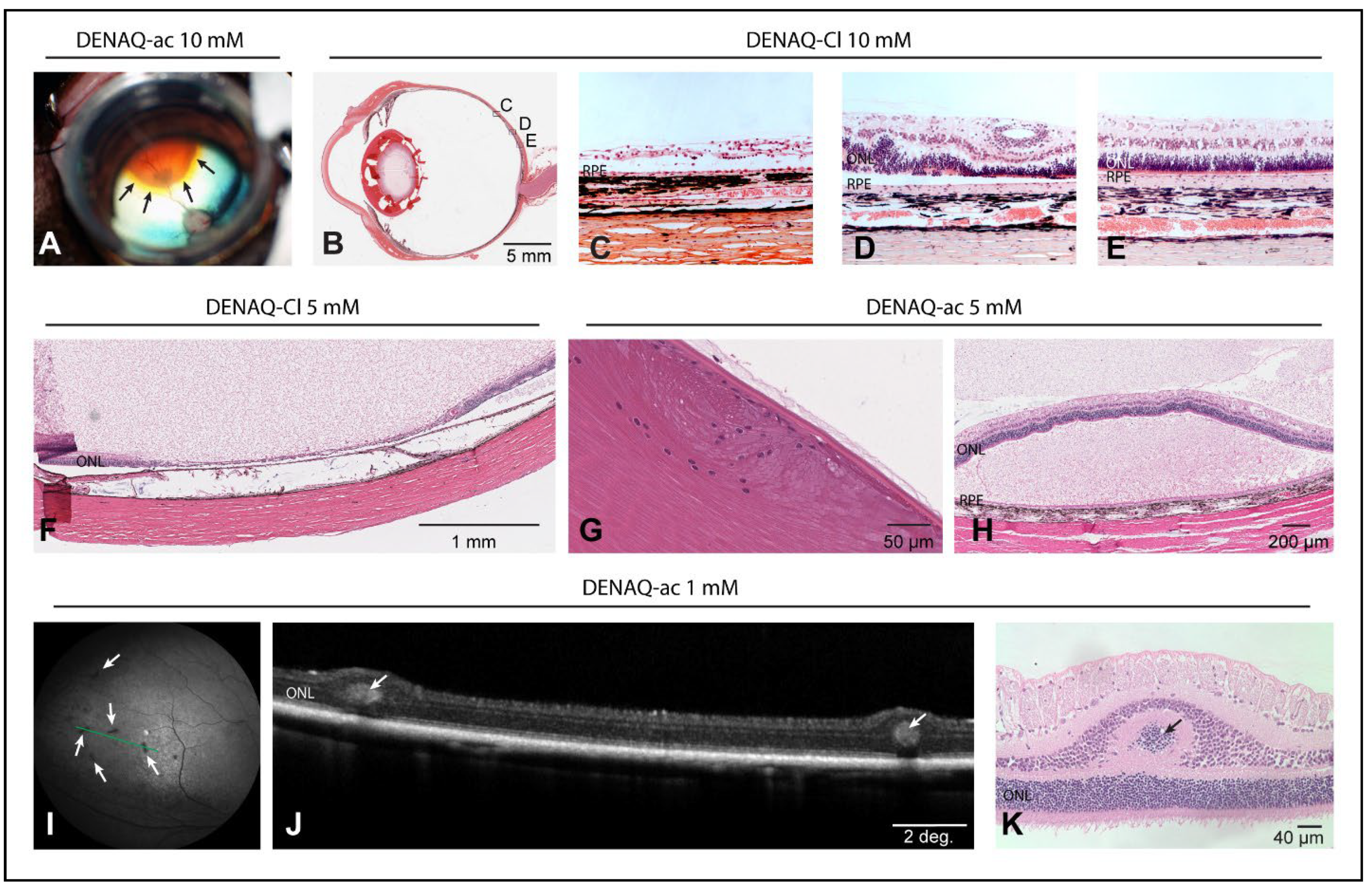
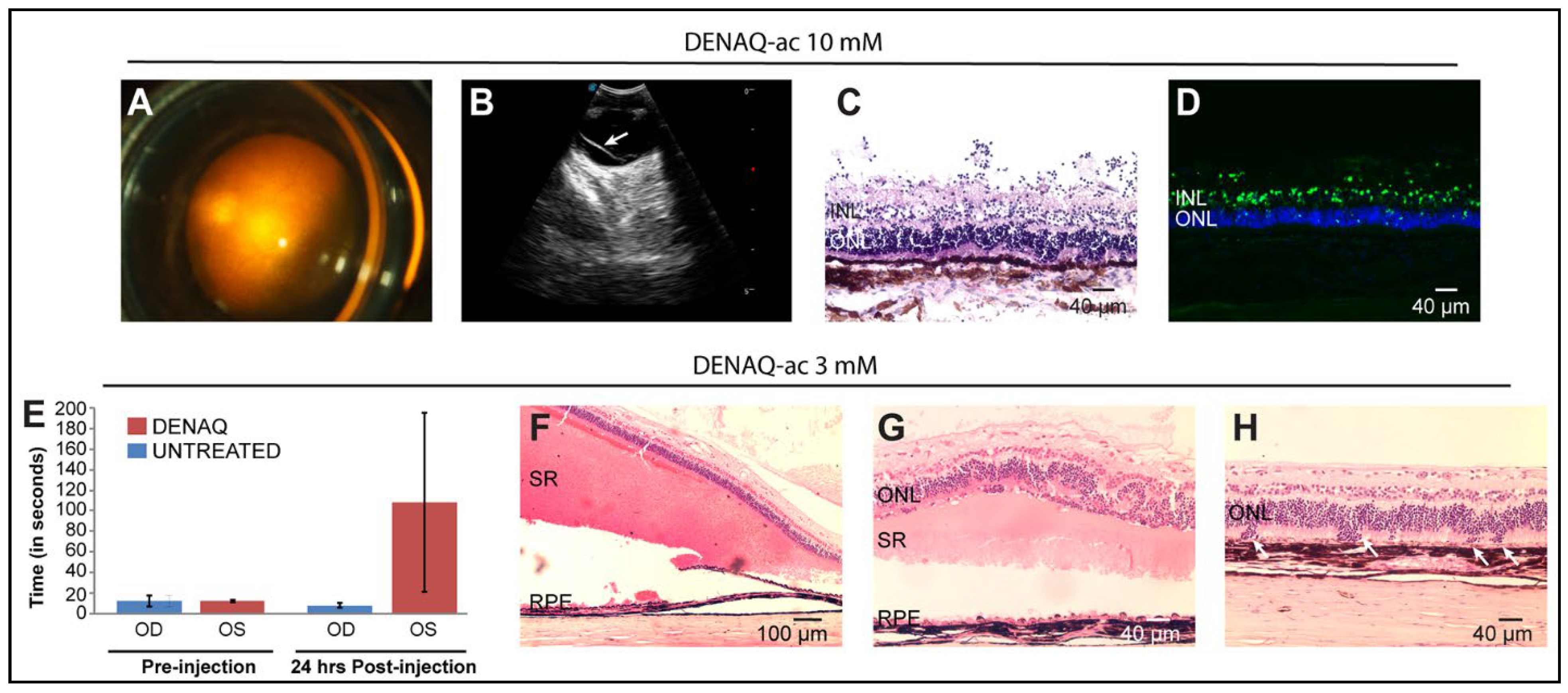
3.6. Dose-Dependent Focal Retinal Toxicity following Intravitreal Injection of DENAQ
3.7. Delivery of DENAQ in Vitrectomized Eyes Expands the Ocular Tolerance Range
4. Discussion
4.1. DENAQ Drives Light Responses in the Degenerated Canine Retinas
4.2. Repeated Light Stimuli Slow Activation and Accelerate Recovery of the DENAQ-Driven Responses
4.3. DENAQ can Override Responses of the Surviving Cones in the Degenerated Retinas
4.4. Switching off Retinal Synaptic Transmission Enhances DENAQ-Driven Response
4.5. Efficiency of DENAQ Treatment
4.6. DENAQ-Associated Ocular Toxicity
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Lamb, T.D.; Pugh, E.N., Jr. Phototransduction, dark adaptation, and rhodopsin regeneration the proctor lecture. Invest. Ophthalmol. Vis. Sci. 2006, 47, 5137–5152. [Google Scholar] [CrossRef] [PubMed]
- Ku, C.A.; Pennesi, M.E. The new landscape of retinal gene therapy. Am. J. Med. Genet. C Semin. Med. Genet. 2020, 184, 846–859. [Google Scholar] [CrossRef] [PubMed]
- Van Gelder, R.N.; Chiang, M.F.; Dyer, M.A.; Greenwell, T.N.; Levin, L.A.; Wong, R.O.; Svendsen, C.N. Regenerative and restorative medicine for eye disease. Nat. Med. 2022, 28, 1149–1156. [Google Scholar] [CrossRef] [PubMed]
- Bloch, E.; Luo, Y.; da Cruz, L. Advances in retinal prosthesis systems. Ther. Adv. Ophthalmol. 2019, 11, 2515841418817501. [Google Scholar] [CrossRef]
- Gasparini, S.J.; Llonch, S.; Borsch, O.; Ader, M. Transplantation of photoreceptors into the degenerative retina: Current state and future perspectives. Prog. Retin. Eye Res. 2019, 69, 30445193. [Google Scholar] [CrossRef]
- De Silva, S.R.; Moore, A.T. Optogenetic approaches to therapy for inherited retinal degenerations. J. Physiol. 2022, 600, 4623. [Google Scholar] [CrossRef]
- Tochitsky, I.; Kienzler, M.A.; Isacoff, E.; Kramer, R.H. Restoring Vision to the Blind with Chemical Photoswitches. Chem. Rev. 2018, 118, 10748–10773. [Google Scholar] [CrossRef]
- Polosukhina, A.; Litt, J.; Tochitsky, I.; Nemargut, J.; Sychev, Y.; De Kouchkovsky, I.; Huang, T.; Borges, K.; Trauner, D.; Van Gelder, R.N.; et al. Photochemical restoration of visual responses in blind mice. Neuron 2012, 75, 271–282. [Google Scholar] [CrossRef]
- Tochitsky, I.; Polosukhina, A.; Degtyar, V.E.; Gallerani, N.; Smith, C.M.; Friedman, A.; Van Gelder, R.N.; Trauner, D.; Kaufer, D.; Kramer, R.H. Restoring Visual Function to Blind Mice with a Photoswitch that Exploits Electrophysiological Remodeling of Retinal Ganglion Cells. Neuron 2014, 81, 800–813. [Google Scholar] [CrossRef]
- Laprell, L.; Tochitsky, I.; Kaur, K.; Manookin, M.B.; Stein, M.; Barber, D.M.; Schon, C.; Michalakis, S.; Biel, M.; Kramer, R.H.; et al. Photopharmacological control of bipolar cells restores visual function in blind mice. J. Clin. Invest. 2017, 127, 2598–2611. [Google Scholar] [CrossRef]
- Tochitsky, I.; Trautman, J.; Gallerani, N.; Malis, J.G.; Kramer, R.H. Restoring visual function to the blind retina with a potent, safe and long-lasting photoswitch. Sci. Rep. 2017, 7, 45487. [Google Scholar] [CrossRef]
- Mourot, A.; Kienzler, M.A.; Banghart, M.R.; Fehrentz, T.; Huber, F.M.E.; Stein, M.; Kramer, R.H.; Trauner, D. Tuning photochromic ion channel blockers. ACS Chem. Neurosci. 2011, 2, 536–543. [Google Scholar] [CrossRef]
- Tochitsky, I.; Helft, Z.; Meseguer, V.; Fletcher, R.B.; Vessey, K.A.; Telias, M.; Denlinger, B.; Malis, J.; Fletcher, E.L.; Kramer, R.H. How Azobenzene Photoswitches Restore Visual Responses to the Blind Retina. Neuron 2016, 92, 100–113. [Google Scholar] [CrossRef]
- Suber, M.L.; Pittler, S.J.; Qin, N.; Wright, G.C.; Holcombe, V.; Lee, R.H.; Craft, C.M.; Lolley, R.N.; Baehr, W.; Hurwitz, R.L. Irish setter dogs affected with rod/cone dysplasia contain a nonsense mutation in the rod cGMP phosphodiesterase beta-subunit gene. Proc. Natl. Acad. Sci. USA 1993, 90, 3968–3972. [Google Scholar] [CrossRef]
- Goldstein, O.; Mezey, J.G.; Schweitzer, P.A.; Boyko, A.R.; Gao, C.; Bustamante, C.D.; Jordan, J.A.; Aguirre, G.D.; Acland, G.M. IQCB1 and PDE6B mutations cause similar early onset retinal degenerations in two closely related terrier dog breeds. Invest. Ophthalmol. Vis. Sci. 2013, 54, 7005–7019. [Google Scholar] [CrossRef]
- Zhang, Q.; Acland, G.M.; Wu, W.X.; Johnson, J.L.; Pearce-Kelling, S.E.; Tulloch, B.; Vervoort, R.; Wright, A.F.; Aguirre, G.D. Different RPGR exon ORF15 mutations in Canids provide insights into photoreceptor cell degeneration. Hum. Mol. Genet. 2002, 11, 993–1003. [Google Scholar] [CrossRef]
- Aguirre, G.D.; Rubin, L.F. Rod-cone dysplasia (progressive retinal atrophy) in Irish setters. J. Am. Vet. Med. Assoc. 1975, 166, 157–164. [Google Scholar]
- Beltran, W.A.; Hammond, P.; Acland, G.M.; Aguirre, G.D. A frameshift mutation in RPGR exon ORF15 causes photoreceptor degeneration and inner retina remodeling in a model of X-linked retinitis pigmentosa. Invest. Ophthalmol. Vis. Sci. 2006, 47, 1669–1681. [Google Scholar] [CrossRef]
- Gaub, B.M.; Berry, M.H.; Holt, A.E.; Reiner, A.; Kienzler, M.A.; Dolgova, N.; Nikonov, S.; Aguirre, G.D.; Beltran, W.A.; Flannery, J.G.; et al. Restoration of visual function by expression of a light-gated mammalian ion channel in retinal ganglion cells or ON-bipolar cells. Proc. Natl. Acad. Sci. USA 2014, 111, E5574–E5583. [Google Scholar] [CrossRef]
- Sudharsan, R.; Beiting, D.P.; Aguirre, G.D.; Beltran, W.A. Involvement of Innate Immune System in Late Stages of Inherited Photoreceptor Degeneration. Sci. Rep. 2017, 7, 17897. [Google Scholar] [CrossRef]
- Beltran, W.A.; Cideciyan, A.V.; Guziewicz, K.E.; Iwabe, S.; Swider, M.; Scott, E.M.; Savina, S.V.; Ruthel, G.; Stefano, F.; Zhang, L.; et al. Canine retina has a primate fovea-like bouquet of cone photoreceptors which is affected by inherited macular degenerations. PLoS ONE 2014, 9, e90390. [Google Scholar] [CrossRef] [PubMed]
- Franke, H.; Klimke, K.; Brinckmann, U.; Grosche, J.; Francke, M.; Sperlagh, B.; Reichenbach, A.; Liebert, U.G.; Illes, P. P2X(7) receptor-mRNA and -protein in the mouse retina; changes during retinal degeneration in BALBCrds mice. Neurochem. Int. 2005, 47, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Mure, L.S.; Hatori, M.; Zhu, Q.; Demas, J.; Kim, I.M.; Nayak, S.K.; Panda, S. Melanopsin-Encoded Response Properties of Intrinsically Photosensitive Retinal Ganglion Cells. Neuron 2016, 90, 1016–1027. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.Y. A retinal ganglion cell that can signal irradiance continuously for 10 hours. J. Neurosci. 2012, 32, 11478–11485. [Google Scholar] [CrossRef] [PubMed]
- Mure, L.S.; Vinberg, F.; Hanneken, A.; Panda, S. Functional diversity of human intrinsically photosensitive retinal ganglion cells. Science 2019, 366, 1251–1255. [Google Scholar] [CrossRef]
- Telias, M.; Denlinger, B.; Helft, Z.; Thornton, C.; Beckwith-Cohen, B.; Kramer, R.H. Retinoic Acid Induces Hyperactivity, and Blocking Its Receptor Unmasks Light Responses and Augments Vision in Retinal Degeneration. Neuron 2019, 102, 574–586.e5. [Google Scholar] [CrossRef]
- Harkat, M.; Peverini, L.; Cerdan, A.H.; Dunning, K.; Beudez, J.; Martz, A.; Calimet, N.; Specht, A.; Cecchini, M.; Chataigneau, T.; et al. On the permeation of large organic cations through the pore of ATP-gated P2X receptors. Proc. Natl. Acad. Sci. USA 2017, 114, E3786–E3795. [Google Scholar] [CrossRef]
- Della Santina, L.; Piano, I.; Cangiano, L.; Caputo, A.; Ludwig, A.; Cervetto, L.; Gargini, C. Processing of retinal signals in normal and HCN deficient mice. PLoS ONE 2012, 7, e29812. [Google Scholar] [CrossRef]
- Lancaster, B.; Zucker, R.S. Photolytic manipulation of Ca2+ and the time course of slow, Ca(2+)-activated K+ current in rat hippocampal neurones. J. Physiol. 1994, 475, 229–239. [Google Scholar] [CrossRef]
- Sahel, J.A.; Boulanger-Scemama, E.; Pagot, C.; Arleo, A.; Galluppi, F.; Martel, J.N.; Esposti, S.D.; Delaux, A.; de Saint Aubert, J.B.; de Montleau, C.; et al. Partial recovery of visual function in a blind patient after optogenetic therapy. Nat. Med. 2021, 27, 1223–1229. [Google Scholar] [CrossRef]
- García-Ayuso, D.; Di Pierdomenico, J.; Vidal-Sanz, M.; Villegas-Pérez, M.P. Retinal Ganglion Cell Death as a Late Remodeling Effect of Photoreceptor Degeneration. Int. J. Mol. Sci. 2019, 20, 4649. [Google Scholar] [CrossRef]
- Pfeiffer, R.L.; Marc, R.E.; Jones, B.W. Persistent remodeling and neurodegeneration in late-stage retinal degeneration. Prog. Retin. Eye Res. 2020, 74, 100771. [Google Scholar] [CrossRef]
- Santos, A.; Humayun, M.S.; de Juan, E., Jr.; Greenburg, R.J.; Marsh, M.J.; Klock, I.B.; Milam, A.H. Preservation of the inner retina in retinitis pigmentosa. A morphometric analysis. Arch. Ophthalmol. 1997, 115, 511–515. [Google Scholar] [CrossRef]
- Ramkumar, H.L.; Nguyen, B.; Bartsch, D.U.; Saunders, L.J.; Muftuoglu, I.K.; You, Q.; Freeman, W.R. Reduced ganglion cell volume on optical coherence tomography in patients with geographic atrophy. Retina 2018, 38, 2159–2167. [Google Scholar] [CrossRef]
- Bringmann, A.; Pannicke, T.; Grosche, J.; Francke, M.; Wiedemann, P.; Skatchkov, S.N.; Osborne, N.N.; Reichenbach, A. Muller cells in the healthy and diseased retina. Prog. Retin. Eye Res. 2006, 25, 397–424. [Google Scholar] [CrossRef]
- Pannicke, T.; Fischer, W.; Biedermann, B.; Schädlich, H.; Grosche, J.; Faude, F.; Wiedemann, P.; Allgaier, C.; Illes, P.; Burnstock, G.; et al. P2X7 receptors in Müller glial cells from the human retina. J. Neurosci. 2000, 20, 5965–5972. [Google Scholar] [CrossRef]
- Reichenbach, A.; Bringmann, A. New functions of Muller cells. Glia 2013, 61, 651–678. [Google Scholar] [CrossRef]
- Cao, K.J.; Lyons, E.F.; Smith, B.E.; Denlinger, B.L.; Ma, H.; Shirian, J.D.; Kramer, R.H. Cyclodextrin-Assisted Delivery of Azobenzene Photoswitches for Uniform and Long-Term Restoration of Light Responses in Degenerated Retinas of Blind Mice. Adv. Ther. 2021, 4, 2100127. [Google Scholar] [CrossRef]

| DENAQ Concentration | Visual Impairment | Ocular Discomfort | Anterior Uveitis | Glaucoma | Posterior Subcapsular Cataract | Posterior Uveitis | Retinal Detachment | Retinal Atrophy | Disrupted Retinal Lamination | Retinal Folds |
|---|---|---|---|---|---|---|---|---|---|---|
| 10 mM | 2/4 | 2/4 | 4/4 | 1/4 | 4/4 | 2/4 | 1/4 | 2/4 | 2/4 | 0/4 |
| 5 mM | 6/6 * | 0/6 | 6/6 | 0/6 | 5/6 | 5/6 | 5/6 | 3/6 | 2/6 | 0/6 |
| 1 mM | 4/4 | 0/4 | 4/4 | 0/4 | 2/4 | 0/4 | 1/4 ** | 0/4 | 0/4 | 2/4 |
| 0.3 mM | 0/3 | 1/3 | 2/3 | 0/3 | 2/3 | 1/3 | 0/3 | 0/3 | 0/3 | 0/3 |
| 0.1 mM | 0/3 | 1/3 | 2/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 |
| Vehicle *** | 0/2 | 0/2 | 2/2 | 0/2 | 0/2 | 0/2 | 0/2 | 0/2 | 0/2 | 0/2 |
| DENAQ Concentration | Visual Impairment | Ocular Discomfort | Anterior Uveitis | Vitreal Hemorrhage | Retinal Detachment | Retinal Atrophy | Disrupted Retinal Lamination |
|---|---|---|---|---|---|---|---|
| 10 mM | 1/3 | 3/3 | 3/3 | 0/3 | 3/3 | 1/2 | 1/2 |
| 3 mM | 2/3 * | 2/3 | 3/3 | 1/3 | 1/3 | 0/2 | 2/2 |
| 1 mM | 0/3 | 1/3 | 2/3 | 0/3 | 0/3 | 0/3 | 0/3 |
| Vehicle ** | 0/1 | 0/1 | 0/1 | 0/1 | 0/1 | 0/1 | 0/1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nikonov, S.; Dolgova, N.; Sudharsan, R.; Tochitsky, I.; Iwabe, S.; Guzman, J.-M.; Van Gelder, R.N.; Kramer, R.H.; Aguirre, G.D.; Beltran, W.A. Photochemical Restoration of Light Sensitivity in the Degenerated Canine Retina. Pharmaceutics 2022, 14, 2711. https://doi.org/10.3390/pharmaceutics14122711
Nikonov S, Dolgova N, Sudharsan R, Tochitsky I, Iwabe S, Guzman J-M, Van Gelder RN, Kramer RH, Aguirre GD, Beltran WA. Photochemical Restoration of Light Sensitivity in the Degenerated Canine Retina. Pharmaceutics. 2022; 14(12):2711. https://doi.org/10.3390/pharmaceutics14122711
Chicago/Turabian StyleNikonov, Sergei, Natalia Dolgova, Raghavi Sudharsan, Ivan Tochitsky, Simone Iwabe, Jose-Manuel Guzman, Russell N. Van Gelder, Richard H. Kramer, Gustavo D. Aguirre, and William A. Beltran. 2022. "Photochemical Restoration of Light Sensitivity in the Degenerated Canine Retina" Pharmaceutics 14, no. 12: 2711. https://doi.org/10.3390/pharmaceutics14122711
APA StyleNikonov, S., Dolgova, N., Sudharsan, R., Tochitsky, I., Iwabe, S., Guzman, J.-M., Van Gelder, R. N., Kramer, R. H., Aguirre, G. D., & Beltran, W. A. (2022). Photochemical Restoration of Light Sensitivity in the Degenerated Canine Retina. Pharmaceutics, 14(12), 2711. https://doi.org/10.3390/pharmaceutics14122711






