Dose Titration of Solid Dosage Forms via FDM 3D-Printed Mini-Tablets
Abstract
1. Introduction
- Single and multi-unit mini-tablets, i.e., 1, 5, 10, 15, 20 count(s), to test content uniformity at a low dose, i.e., 1 wt%, and to evaluate corresponding dissolution profiles to assess their similarity.
- Split tablets, i.e., full, half, and quarter sizes, to compare with multi-unit mini-tablets along with testing content uniformity and dissolution.
- Single unit mini-tablets formed using filaments of 10 and 20 wt% drug concentrations for testing feasibility of reducing the number of tablets to be administered.
2. Materials and Methods
2.1. Materials
2.2. Manufacturing of Filaments
2.3. FDM 3D Printing and Tablet Morphology
2.4. Thermo-Gravimetric Analysis (TGA)
2.5. Solid-State Characterization
2.6. Content Uniformity
2.7. Dissolution
3. Results
3.1. Mini-Tablet Printing and Tablet Morphology
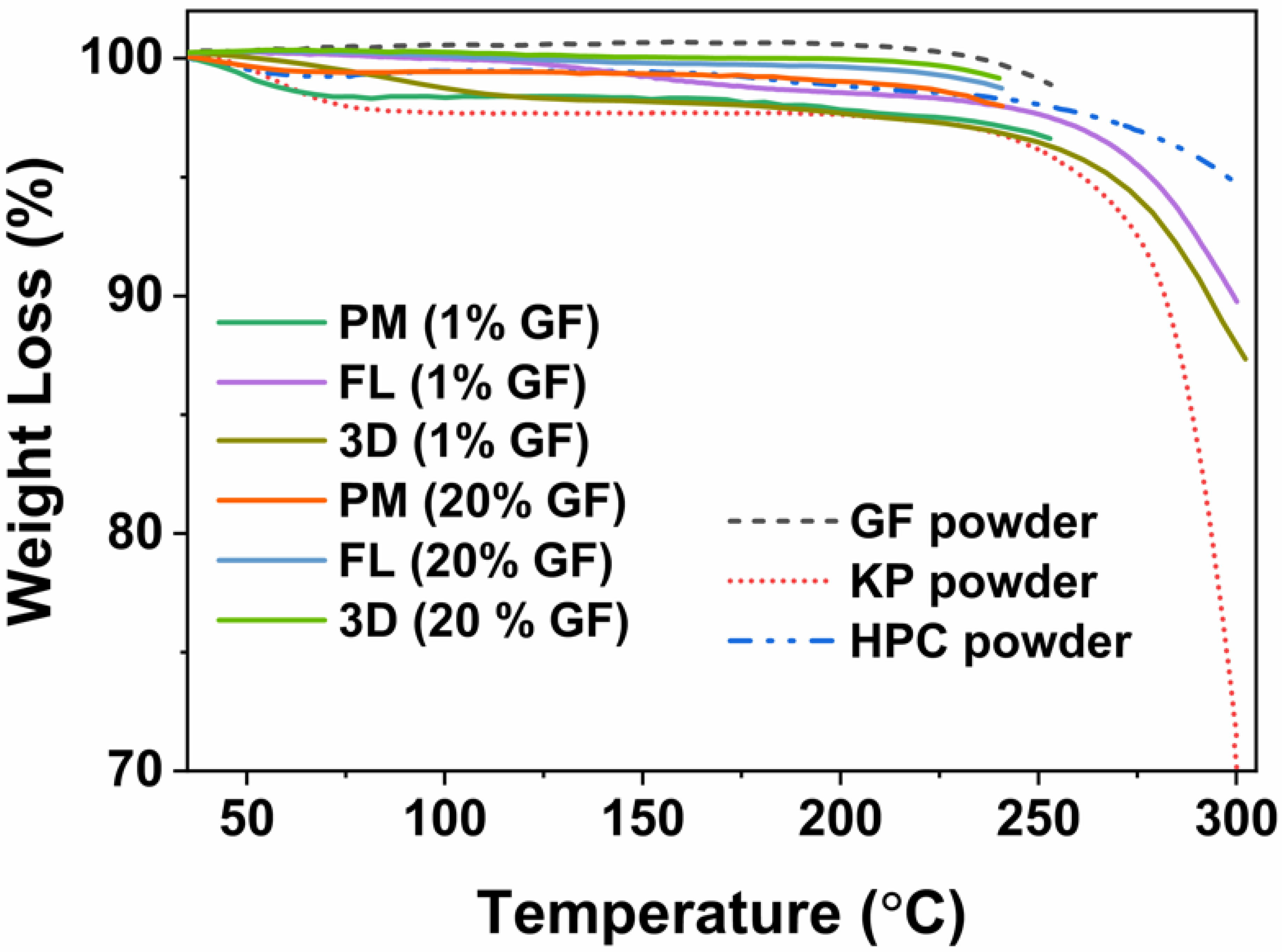
3.2. Thermo-Gravimetric Analysis (TGA)
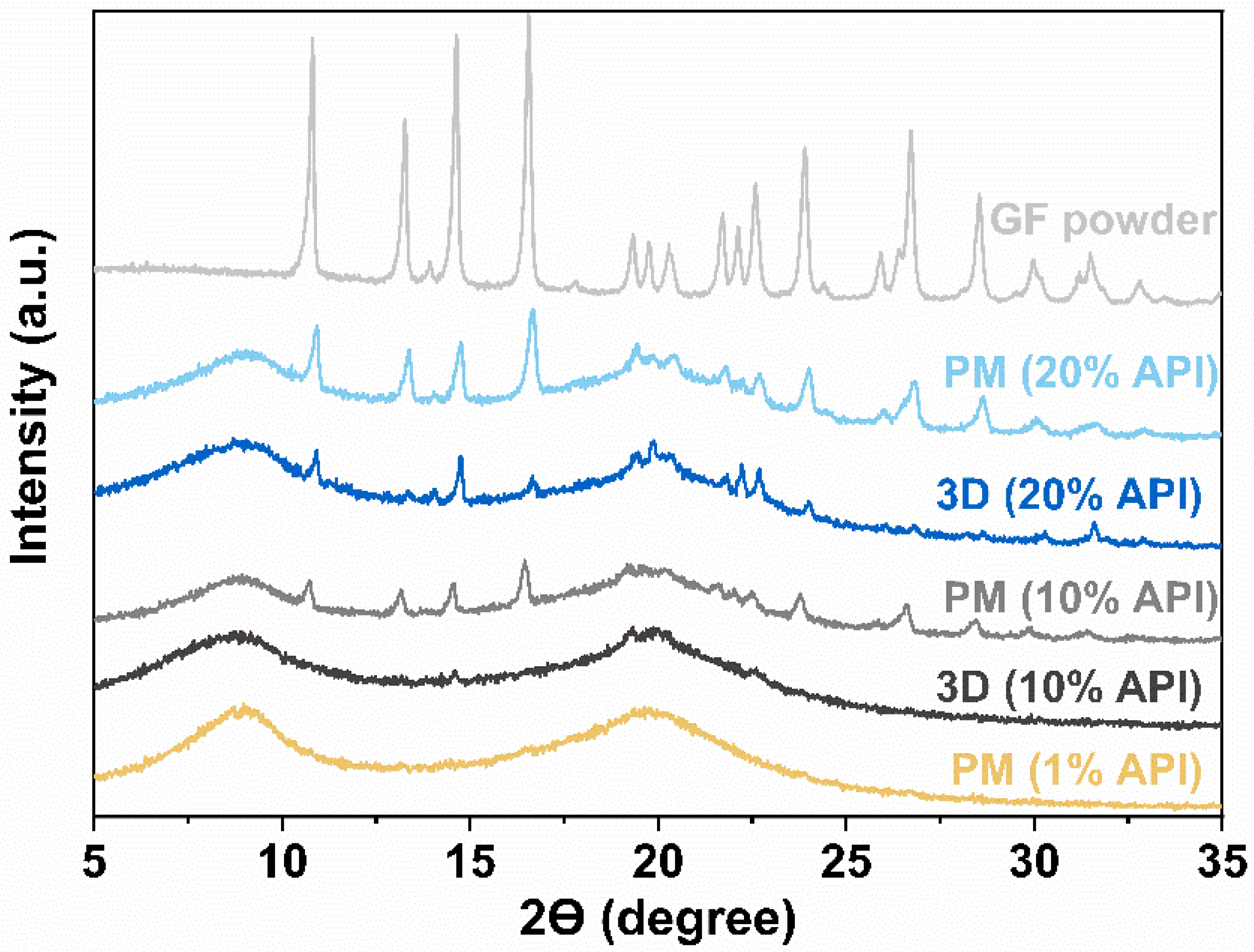
3.3. X-ray Diffraction (XRD)
3.4. Content Uniformity and Dose Titration
3.4.1. Content Uniformity
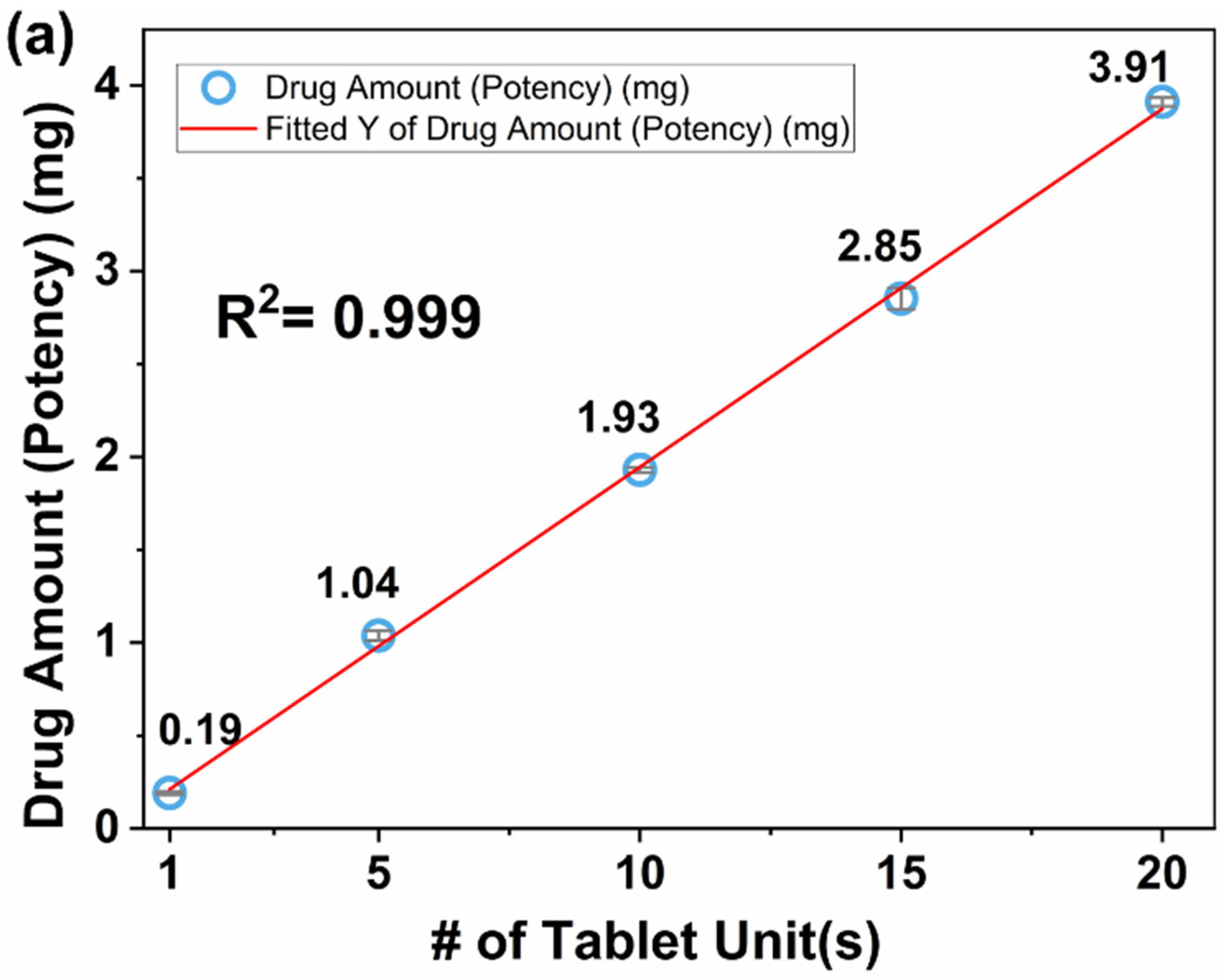
3.4.2. Dose Titration
3.5. Drug Dissolution
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ernest, T.B.; Craig, J.; Nunn, A.; Salunke, S.; Tuleu, C.; Breitkreutz, J.; Alex, R.; Hempenstall, J. Preparation of medicines for children–A hierarchy of classification. Int. J. Pharm. 2012, 435, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Strickley, R.G. Pediatric oral formulations: An updated review of commercially available pediatric oral formulations since 2007. J. Pharm. Sci. 2019, 108, 1335–1365. [Google Scholar] [CrossRef] [PubMed]
- Ranmal, S.R.; Cram, A.; Tuleu, C. Age-appropriate and acceptable paediatric dosage forms: Insights into end-user perceptions, preferences and practices from the Children’s Acceptability of Oral Formulations (CALF) Study. Int. J. Pharm. 2016, 514, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Salunke, S.; Tuleu, C. Formulating better medicines for children—Still too far to walk. Int. J. Pharm. 2016, 511, 1124–1126. [Google Scholar] [CrossRef] [PubMed]
- Richey, R.H.; Hughes, C.; Craig, J.V.; Shah, U.U.; Ford, J.L.; Barker, C.E.; Peak, M.; Nunn, A.J.; Turner, M.A. A systematic review of the use of dosage form manipulation to obtain required doses to inform use of manipulation in paediatric practice. Int. J. Pharm. 2017, 518, 155–166. [Google Scholar] [CrossRef]
- Breitkreutz, J.; Boos, J. Paediatric and geriatric drug delivery. Expert Opin. Drug Deliv. 2007, 4, 37–45. [Google Scholar] [CrossRef]
- McDevitt, J.T.; Gurst, A.H.; Chen, Y. Accuracy of tablet splitting. Pharmacother. J. Hum. Pharmacol. Drug Ther. 1998, 18, 193–197. [Google Scholar]
- Cohen, J.S. Tablet splitting: Imperfect perhaps, but better than excessive dosing. J. Am. Pharm. Assoc. 2002, 42, 160–162. [Google Scholar] [CrossRef]
- Teng, J.; Song, C.K.; Williams, R.L.; Polli, J.E. Lack of medication dose uniformity in commonly split tablets. J. Am. Pharm. Assoc. 2002, 42, 195–199. [Google Scholar] [CrossRef]
- Quinzler, R.; Gasse, C.; Schneider, A.; Kaufmann-Kolle, P.; Szecsenyi, J.; Haefeli, W. The frequency of inappropriate tablet splitting in primary care. Eur. J. Clin. Pharmacol. 2006, 62, 1065–1073. [Google Scholar] [CrossRef]
- Fawell, N.G.; Cookson, T.L.; Scranton, S.S. Relationship between tablet splitting and compliance, drug acquisition cost, and patient acceptance. Am. J. Health-Syst. Pharm. 1999, 56, 2542–2545. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.B.; Collier, J.S.; Sayeed, V.A.; Bryant, A.; Habib, M.J.; Khan, M.A. Tablet splitting of a narrow therapeutic index drug: A case with levothyroxine sodium. AAPS PharmSciTech 2010, 11, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
- van Riet-Nales, D.A.; Doeve, M.E.; Nicia, A.E.; Teerenstra, S.; Notenboom, K.; Hekster, Y.A.; van den Bemt, B.J. The accuracy, precision and sustainability of different techniques for tablet subdivision: Breaking by hand and the use of tablet splitters or a kitchen knife. Int. J. Pharm. 2014, 466, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Boggie, D.T.; DeLattre, M.L.; Schaefer, M.G.; Morreale, A.P.; Plowman, B.K. Accuracy of splitting unscored valdecoxib tablets. Am. J. Health-Syst. Pharm. 2004, 61, 1482–1483. [Google Scholar] [CrossRef] [PubMed]
- FDA. Best Practice of Tablet Splitting. Available online: https://www.fda.gov/drugs/ensuring-safe-use-medicine/best-practices-tablet-splitting (accessed on 26 October 2022).
- Collier, J.; Shah, R.; Bryant, A.; Habib, M.; Khan, M.; Faustino, P. Development and application of a validated HPLC method for the analysis of dissolution samples of levothyroxine sodium drug products. J. Pharm. Biomed. Anal. 2011, 54, 433–438. [Google Scholar] [CrossRef]
- Preis, M. Orally disintegrating films and mini-tablets—Innovative dosage forms of choice for pediatric use. AAPS PharmSciTech 2015, 16, 234–241. [Google Scholar] [CrossRef]
- Wening, K.; Breitkreutz, J. Oral drug delivery in personalized medicine: Unmet needs and novel approaches. Int. J. Pharm. 2011, 404, 1–9. [Google Scholar] [CrossRef]
- Brown, D.; Ford, J.; Nunn, A.; Rowe, P. An assessment of dose-uniformity of samples delivered from paediatric oral droppers. J. Clin. Pharm. Ther. 2004, 29, 521–529. [Google Scholar] [CrossRef]
- Rood, J.M.; Engels, M.J.; Ciarkowski, S.L.; Wagenknecht, L.D.; Dickinson, C.J.; Stevenson, J.G. Variability in compounding of oral liquids for pediatric patients: A patient safety concern. J. Am. Pharm. Assoc. 2014, 54, 383–389. [Google Scholar] [CrossRef]
- Mitra, B.; Thool, P.; Meruva, S.; Aycinena, J.A.; Li, J.; Patel, J.; Patel, K.; Agarwal, A.; Karki, S.; Bowen, W. Decoding the small size challenges of mini-tablets for enhanced dose flexibility and micro-dosing. Int. J. Pharm. 2020, 574, 118905. [Google Scholar] [CrossRef]
- Gaber, D.M.; Nafee, N.; Abdallah, O.Y. Mini-tablets versus pellets as promising multiparticulate modified release delivery systems for highly soluble drugs. Int. J. Pharm. 2015, 488, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Harris, D.; Hermans, E.; Klein, S.; Wagner-Hattler, L.; Walsh, J. Age-appropriate solid oral formulations for pediatric applications with a focus on multiparticulates and minitablets: Summary of September 2019 EuPFI workshop. Eur. J. Pharm. Biopharm. 2020, 153, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Munday, D. A comparison of the dissolution characteristics of theophylline from film coated granules and mini-tablets. Drug Dev. Ind. Pharm. 1994, 20, 2369–2379. [Google Scholar] [CrossRef]
- Tissen, C.; Woertz, K.; Breitkreutz, J.; Kleinebudde, P. Development of mini-tablets with 1 mm and 2 mm diameter. Int. J. Pharm. 2011, 416, 164–170. [Google Scholar] [CrossRef]
- Aleksovski, A.; Dreu, R.; Gašperlin, M.; Planinšek, O. Mini-tablets: A contemporary system for oral drug delivery in targeted patient groups. Expert Opin. Drug Deliv. 2015, 12, 65–84. [Google Scholar] [CrossRef] [PubMed]
- Mitra, B.; Chang, J.; Wu, S.-J.; Wolfe, C.N.; Ternik, R.L.; Gunter, T.Z.; Victor, M.C. Feasibility of mini-tablets as a flexible drug delivery tool. Int. J. Pharm. 2017, 525, 149–159. [Google Scholar] [CrossRef]
- Klingmann, V. Acceptability of mini-tablets in young children: Results from three prospective cross-over studies. AAPS PharmSciTech 2017, 18, 263–266. [Google Scholar] [CrossRef]
- Thomson, S.A.; Tuleu, C.; Wong, I.C.; Keady, S.; Pitt, K.G.; Sutcliffe, A.G. Minitablets: New modality to deliver medicines to preschool-aged children. Pediatrics 2009, 123, e235–e238. [Google Scholar] [CrossRef]
- Sujja-Areevath, J.; Munday, D.; Cox, P.; Khan, K. Relationship between swelling, erosion and drug release in hydrophillic natural gum mini-matrix formulations. Eur. J. Pharm. Sci. 1998, 6, 207–217. [Google Scholar] [CrossRef]
- Goh, H.P.; Heng, P.W.S.; Liew, C.V. Understanding die fill variation during mini-tablet production. Int. J. Pharm. 2017, 534, 279–286. [Google Scholar] [CrossRef]
- Palekar, S.; Nukala, P.K.; Mishra, S.M.; Kipping, T.; Patel, K. Application of 3D printing technology and quality by design approach for development of age-appropriate pediatric formulation of baclofen. Int. J. Pharm. 2019, 556, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Terán, M.M.; Flament, M. Development of multiparticulate orodispersible tablets for pediatric use. Int. J. Pharm. 2016, 2, 1143. [Google Scholar] [CrossRef]
- Lennartz, P.; Mielck, J. Minitabletting: Improving the compactability of paracetamol powder mixtures. Int. J. Pharm. 1998, 173, 75–85. [Google Scholar] [CrossRef]
- Klingmann, V.; Seitz, A.; Meissner, T.; Breitkreutz, J.; Moeltner, A.; Bosse, H.M. Acceptability of uncoated mini-tablets in neonates—A randomized controlled trial. J. Pediatr. 2015, 167, 893–896.e892. [Google Scholar] [CrossRef] [PubMed]
- Bayan, M.F.; Sbaih, H.M.; Saadh, M.J. Pharmaceutical mini-tablets overview. Indian J. Forensic Med. Toxicol. 2021, 15, 1291–1304. [Google Scholar]
- Priyanka, P.; Kumar, K.; Teotia, D. A comprehensive review on pharmaceutical mini tablets. J. Drug Deliv. Ther. 2018, 8, 382–390. [Google Scholar] [CrossRef]
- Zhao, J.; Yin, D.; Rowe, J.; Badawy, S.; Nikfar, F.; Pandey, P. Understanding the factors that control the quality of mini-tablet compression: Flow, particle size, and tooling dimension. J. Pharm. Sci. 2018, 107, 1204–1208. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Wang, C.; Liu, S.; Sun, C.C. Development of piroxicam mini-tablets enabled by spherical cocrystallization. Int. J. Pharm. 2020, 590, 119953. [Google Scholar] [CrossRef]
- Norman, J.; Madurawe, R.D.; Moore, C.M.; Khan, M.A.; Khairuzzaman, A. A new chapter in pharmaceutical manufacturing: 3D-printed drug products. Adv. Drug Deliv. Rev. 2017, 108, 39–50. [Google Scholar] [CrossRef]
- Abaci, A.; Gedeon, C.; Kuna, A.; Guvendiren, M. Additive manufacturing of oral tablets: Technologies, materials and printed tablets. Pharmaceutics 2021, 13, 156. [Google Scholar] [CrossRef]
- Awad, A.; Fina, F.; Trenfield, S.J.; Patel, P.; Goyanes, A.; Gaisford, S.; Basit, A.W. 3D printed pellets (miniprintlets): A novel, multi-drug, controlled release platform technology. Pharmaceutics 2019, 11, 148. [Google Scholar] [CrossRef] [PubMed]
- Junqueira, L.A.; Tabriz, A.G.; Raposo, F.J.; Carobini, L.R.; Vaz, U.P.; Brandão, M.A.F.; Douroumis, D.; Raposo, N.R.B. Coupling of fused deposition modeling and inkjet printing to produce drug loaded 3D printed tablets. Pharmaceutics 2022, 14, 159. [Google Scholar] [CrossRef] [PubMed]
- Ngo, T.D.; Kashani, A.; Imbalzano, G.; Nguyen, K.T.; Hui, D. Additive manufacturing (3D printing): A review of materials, methods, applications and challenges. Compos. Part B Eng. 2018, 143, 172–196. [Google Scholar] [CrossRef]
- Ragelle, H.; Rahimian, S.; Guzzi, E.A.; Westenskow, P.D.; Tibbitt, M.W.; Schwach, G.; Langer, R. Additive manufacturing in drug delivery: Innovative drug product design and opportunities for industrial application. Adv. Drug Deliv. Rev. 2021, 178, 113990. [Google Scholar] [CrossRef] [PubMed]
- Shpigel, T.; Uziel, A.; Lewitus, D.Y. SPHRINT–printing drug delivery microspheres from polymeric melts. Eur. J. Pharm. Biopharm. 2018, 127, 398–406. [Google Scholar] [CrossRef]
- Zhang, P.; Xu, P.; Chung, S.; Bandari, S.; Repka, M.A. Fabrication of bilayer tablets using hot melt extrusion-based dual-nozzle fused deposition modeling 3D printing. Int. J. Pharm. 2022, 624, 121972. [Google Scholar] [CrossRef]
- Korte, C.; Quodbach, J. Formulation development and process analysis of drug-loaded filaments manufactured via hot-melt extrusion for 3D-printing of medicines. Pharm. Dev. Technol. 2018, 23, 1117–1127. [Google Scholar] [CrossRef]
- Obeid, S.; Madžarević, M.; Ibrić, S. Tailoring amlodipine release from 3D printed tablets: Influence of infill patterns and wall thickness. Int. J. Pharm. 2021, 610, 121261. [Google Scholar] [CrossRef]
- Govender, R.; Abrahmsén-Alami, S.; Folestad, S.; Larsson, A. High Content Solid Dispersions for Dose Window Extension: A Basis for Design Flexibility in Fused Deposition Modelling. Pharm. Res. 2020, 37, 9. [Google Scholar] [CrossRef]
- Khalid, G.M.; Billa, N. Solid Dispersion Formulations by FDM 3D Printing—A Review. Pharmaceutics 2022, 14, 690. [Google Scholar] [CrossRef]
- Qian, H.; Chen, D.; Xu, X.; Li, R.; Yan, G.; Fan, T. FDM 3D-Printed Sustained-Release Gastric-Floating Verapamil Hydrochloride Formulations with Cylinder, Capsule and Hemisphere Shapes, and Low Infill Percentage. Pharmaceutics 2022, 14, 281. [Google Scholar] [CrossRef] [PubMed]
- Parulski, C.; Gresse, E.; Jennotte, O.; Felten, A.; Ziemons, E.; Lechanteur, A.; Evrard, B. Fused deposition modeling 3D printing of solid oral dosage forms containing amorphous solid dispersions: How to elucidate drug dissolution mechanisms through surface spectral analysis techniques? Int. J. Pharm. 2022, 626, 122157. [Google Scholar] [CrossRef] [PubMed]
- Melocchi, A.; Parietti, F.; Maroni, A.; Foppoli, A.; Gazzaniga, A.; Zema, L. Hot-melt extruded filaments based on pharmaceutical grade polymers for 3D printing by fused deposition modeling. Int. J. Pharm. 2016, 509, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Solanki, N.G.; Vasoya, J.M.; Shah, A.V.; Serajuddin, A.T. Development of 3D Printed Tablets by Fused Deposition Modeling Using Polyvinyl Alcohol as Polymeric Matrix for Rapid Drug Release. J. Pharm. Sci. 2020, 109, 1558–1572. [Google Scholar] [CrossRef]
- Wang, S.; Capoen, L.; D’hooge, D.R.; Cardon, L. Can the melt flow index be used to predict the success of fused deposition modelling of commercial poly (lactic acid) filaments into 3D printed materials? Plast. Rubber Compos. 2018, 47, 9–16. [Google Scholar] [CrossRef]
- Zhang, J.; Feng, X.; Patil, H.; Tiwari, R.V.; Repka, M.A. Coupling 3D printing with hot-melt extrusion to produce controlled-release tablets. Int. J. Pharm. 2017, 519, 186–197. [Google Scholar] [CrossRef]
- Maniruzzaman, M.; Boateng, J.S.; Snowden, M.J.; Douroumis, D. A review of hot-melt extrusion: Process technology to pharmaceutical products. Int. Sch. Res. Not. 2012, 2012, 436763. [Google Scholar] [CrossRef]
- Pawar, J.N.; Fule, R.A.; Maniruzzaman, M.; Amin, P.D. Solid crystal suspension of Efavirenz using hot melt extrusion: Exploring the role of crystalline polyols in improving solubility and dissolution rate. Mater. Sci. Eng. C 2017, 78, 1023–1034. [Google Scholar] [CrossRef]
- Sadia, M.; Isreb, A.; Abbadi, I.; Isreb, M.; Aziz, D.; Selo, A.; Timmins, P.; Alhnan, M.A. From ‘fixed dose combinations’ to ‘a dynamic dose combiner’: 3D printed bi-layer antihypertensive tablets. Eur. J. Pharm. Sci. 2018, 123, 484–494. [Google Scholar] [CrossRef]
- Scoutaris, N.; Ross, S.A.; Douroumis, D. 3D printed “Starmix” drug loaded dosage forms for paediatric applications. Pharm. Res. 2018, 35, 34. [Google Scholar] [CrossRef]
- Pietrzak, K.; Isreb, A.; Alhnan, M.A. A flexible-dose dispenser for immediate and extended release 3D printed tablets. Eur. J. Pharm. Biopharm. 2015, 96, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Okwuosa, T.C.; Stefaniak, D.; Arafat, B.; Isreb, A.; Wan, K.-W.; Alhnan, M.A. A lower temperature FDM 3D printing for the manufacture of patient-specific immediate release tablets. Pharm. Res. 2016, 33, 2704–2712. [Google Scholar] [CrossRef] [PubMed]
- Solanki, N.G.; Tahsin, M.; Shah, A.V.; Serajuddin, A.T. Formulation of 3D printed tablet for rapid drug release by fused deposition modeling: Screening polymers for drug release, drug-polymer miscibility and printability. J. Pharm. Sci. 2018, 107, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Goyanes, A.; Martinez, P.R.; Buanz, A.; Basit, A.W.; Gaisford, S. Effect of geometry on drug release from 3D printed tablets. Int. J. Pharm. 2015, 494, 657–663. [Google Scholar] [CrossRef]
- Arafat, B.; Qinna, N.; Cieszynska, M.; Forbes, R.T.; Alhnan, M.A. Tailored on demand anti-coagulant dosing: An in vitro and in vivo evaluation of 3D printed purpose-designed oral dosage forms. Eur. J. Pharm. Biopharm. 2018, 128, 282–289. [Google Scholar] [CrossRef]
- Rycerz, K.; Stepien, K.A.; Czapiewska, M.; Arafat, B.T.; Habashy, R.; Isreb, A.; Peak, M.; Alhnan, M.A. Embedded 3D printing of novel bespoke soft dosage form concept for pediatrics. Pharmaceutics 2019, 11, 630. [Google Scholar] [CrossRef]
- Sadia, M.; Sośnicka, A.; Arafat, B.; Isreb, A.; Ahmed, W.; Kelarakis, A.; Alhnan, M.A. Adaptation of pharmaceutical excipients to FDM 3D printing for the fabrication of patient-tailored immediate release tablets. Int. J. Pharm. 2016, 513, 659–668. [Google Scholar] [CrossRef]
- Skowyra, J.; Pietrzak, K.; Alhnan, M.A. Fabrication of extended-release patient-tailored prednisolone tablets via fused deposition modelling (FDM) 3D printing. Eur. J. Pharm. Sci. 2015, 68, 11–17. [Google Scholar] [CrossRef]
- Krause, J.; Müller, L.; Sarwinska, D.; Seidlitz, A.; Sznitowska, M.; Weitschies, W. 3D printing of mini tablets for pediatric use. Pharmaceuticals 2021, 14, 143. [Google Scholar] [CrossRef]
- Tagami, T.; Fukushige, K.; Ogawa, E.; Hayashi, N.; Ozeki, T. 3D printing factors important for the fabrication of polyvinylalcohol filament-based tablets. Biol. Pharm. Bull. 2017, 40, 357–364. [Google Scholar] [CrossRef]
- Öblom, H.; Zhang, J.; Pimparade, M.; Speer, I.; Preis, M.; Repka, M.; Sandler, N. 3D-printed isoniazid tablets for the treatment and prevention of tuberculosis—Personalized dosing and drug release. AAPS PharmSciTech 2019, 20, 52. [Google Scholar] [CrossRef] [PubMed]
- Borujeni, S.H.; Mirdamadian, S.Z.; Varshosaz, J.; Taheri, A. Three-dimensional (3D) printed tablets using ethyl cellulose and hydroxypropyl cellulose to achieve zero order sustained release profile. Cellulose 2020, 27, 1573–1589. [Google Scholar] [CrossRef]
- Chai, X.; Chai, H.; Wang, X.; Yang, J.; Li, J.; Zhao, Y.; Cai, W.; Tao, T.; Xiang, X. Fused deposition modeling (FDM) 3D printed tablets for intragastric floating delivery of domperidone. Sci. Rep. 2017, 7, 2829. [Google Scholar] [CrossRef] [PubMed]
- Buyukgoz, G.G.; Soffer, D.; Defendre, J.; Pizzano, G.M.; Davé, R.N. Exploring tablet design options for tailoring drug release and dose via fused deposition modeling (FDM) 3D printing. Int. J. Pharm. 2020, 591, 119987. [Google Scholar] [CrossRef] [PubMed]
- Kolter, K.; Karl, M.; Gryczke, A.; Ludwigshafen am Rhein, B. Hot-Melt Extrusion with BASF Pharma Polymers: Extrusion Compendium; BASF: Ludwigshafen, Germany, 2012. [Google Scholar]
- Rahman, M.; Coelho, A.; Tarabokija, J.; Ahmad, S.; Radgman, K.; Bilgili, E. Synergistic and Antagonistic Effects of Various Amphiphilic Polymer Combinations in Enhancing Griseofulvin Release from Ternary Amorphous Solid Dispersions. Eur. J. Pharm. Sci. 2020, 150, 105354. [Google Scholar] [CrossRef]
- Sarode, A.L.; Malekar, S.A.; Cote, C.; Worthen, D.R. Hydroxypropyl cellulose stabilizes amorphous solid dispersions of the poorly water soluble drug felodipine. Carbohydr. Polym. 2014, 112, 512–519. [Google Scholar] [CrossRef]
- Buyukgoz, G.G.; Kossor, C.G.; Davé, R.N. Enhanced Supersaturation via Fusion-Assisted Amorphization during FDM 3D Printing of Crystalline Poorly Soluble Drug Loaded Filaments. Pharmaceutics 2021, 13, 1857. [Google Scholar] [CrossRef]
- Patwardhan, K.; Asgarzadeh, F.; Dassinger, T.; Albers, J.; Repka, M.A. A quality by design approach to understand formulation and process variability in pharmaceutical melt extrusion processes. J. Pharm. Pharmacol. 2015, 67, 673–684. [Google Scholar] [CrossRef]
- FDA; Lostritto, R. Content Uniformity (CU) Testing for the 21st Century: CDER Perspective. In Proceedings of the AAPS Annual Meeting, Chicago, IL, USA, 14–18 October 2012; Available online: http://www.fda.gov/downloads/AboutFDA/CentersOffices/OfficeofMedicalProductsandTobacco/CDER/UCM341168.pdf (accessed on 28 August 2022).
- Bhakay, A.; Azad, M.; Bilgili, E.; Dave, R. Redispersible fast dissolving nanocomposite microparticles of poorly water-soluble drugs. Int. J. Pharm. 2014, 461, 367–379. [Google Scholar] [CrossRef]
- Li, M.; Ioannidis, N.; Gogos, C.; Bilgili, E. A comparative assessment of nanocomposites vs. amorphous solid dispersions prepared via nanoextrusion for drug dissolution enhancement. Eur. J. Pharm. Biopharm. 2017, 119, 68–80. [Google Scholar] [CrossRef]
- Hellberg, E.; Westberg, A.; Appelblad, P.; Mattsson, S. Evaluation of dissolution techniques for orally disintegrating mini-tablets. J. Drug Deliv. Sci. Technol. 2021, 61, 102191. [Google Scholar] [CrossRef]
- Klein, S.; Shah, V.P. A standardized mini paddle apparatus as an alternative to the standard paddle. AAPS PharmSciTech 2008, 9, 1179–1184. [Google Scholar] [CrossRef] [PubMed]
- Tidau, M.; Kwade, A.; Finke, J.H. Influence of High, Disperse API Load on Properties along the Fused-Layer Modeling Process Chain of Solid Dosage Forms. Pharmaceutics 2019, 11, 194. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Arevalo, F.; Coelho, A.; Bilgili, E. Hybrid nanocrystal–amorphous solid dispersions (HyNASDs) as alternative to ASDs for enhanced release of BCS Class II drugs. Eur. J. Pharm. Biopharm. 2019, 145, 12–26. [Google Scholar] [CrossRef]
- Siddiqui, A.; Rahman, Z.; Bykadi, S.; Khan, M.A. Chemometric methods for the quantification of crystalline tacrolimus in solid dispersion by powder X-ray diffractrometry. J. Pharm. Sci. 2014, 103, 2819–2828. [Google Scholar] [CrossRef]
- Ayyoubi, S.; Cerda, J.R.; Fernández-García, R.; Knief, P.; Lalatsa, A.; Healy, A.M.; Serrano, D.R. 3D printed spherical mini-tablets: Geometry versus composition effects in controlling dissolution from personalised solid dosage forms. Int. J. Pharm. 2021, 597, 120336. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, W.; Vo, A.Q.; Feng, X.; Ye, X.; Kim, D.W.; Repka, M.A. Hydroxypropyl methylcellulose-based controlled release dosage by melt extrusion and 3D printing: Structure and drug release correlation. Carbohydr. Polym. 2017, 177, 49–57. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, H.; Li, H.; Ou, Z.; Yang, G. 3D printed tablets with internal scaffold structure using ethyl cellulose to achieve sustained ibuprofen release. Eur. J. Pharm. Sci. 2018, 115, 11–18. [Google Scholar] [CrossRef]
- Gbureck, U.; Vorndran, E.; Müller, F.A.; Barralet, J.E. Low temperature direct 3D printed bioceramics and biocomposites as drug release matrices. J. Control. Release 2007, 122, 173–180. [Google Scholar] [CrossRef]
- Melocchi, A.; Uboldi, M.; Maroni, A.; Foppoli, A.; Palugan, L.; Zema, L.; Gazzaniga, A. 3D printing by fused deposition modeling of single-and multi-compartment hollow systems for oral delivery–A review. Int. J. Pharm. 2020, 579, 119155. [Google Scholar] [CrossRef]
- Fina, F.; Goyanes, A.; Rowland, M.; Gaisford, S.; Basit, A.W. 3D printing of tunable zero-order release printlets. Polymers 2020, 12, 1769. [Google Scholar] [CrossRef] [PubMed]
- Sadia, M.; Arafat, B.; Ahmed, W.; Forbes, R.T.; Alhnan, M.A. Channelled tablets: An innovative approach to accelerating drug release from 3D printed tablets. J. Control. Release 2018, 269, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Petry, I.; Löbmann, K.; Grohganz, H.; Rades, T.; Leopold, C.S. Solid state properties and drug release behavior of co-amorphous indomethacin-arginine tablets coated with Kollicoat® Protect. Eur. J. Pharm. Biopharm. 2017, 119, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Sarode, A.; Wang, P.; Cote, C.; Worthen, D.R. Low-viscosity hydroxypropylcellulose (HPC) grades SL and SSL: Versatile pharmaceutical polymers for dissolution enhancement, controlled release, and pharmaceutical processing. AAPS PharmSciTech 2013, 14, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Mendyk, A.; Pacławski, A.; Szlek, J.; Jachowicz, R. PhEq_bootstrap: Open-source software for the simulation of f2 distribution in cases of large variability in dissolution profiles. Dissolution Technol. 2013, 20, 13–17. [Google Scholar] [CrossRef]
- Paixão, P.; Gouveia, L.F.; Silva, N.; Morais, J.A. Evaluation of dissolution profile similarity–Comparison between the f2, the multivariate statistical distance and the f2 bootstrapping methods. Eur. J. Pharm. Biopharm. 2017, 112, 67–74. [Google Scholar] [CrossRef]



| Run | Formulation | HME Processing Temperature (°C) | HME Screw Speed (rpm) |
|---|---|---|---|
| F1 | 1 wt% GF + 84 wt% HPC + 15 wt% KP | 150 | 40 |
| F10 | 10 wt% GF + 75 wt% HPC + 15 wt% KP | 150 | 40 |
| F20 | 20 wt% GF + 65 wt% HPC + 15 wt% KP | 155 | 40 |
| Run | Theoretical Drug Concentration (wt%) | Radius * (mm) | Height * (mm) | Number of Unit(s) | |
|---|---|---|---|---|---|
| M1 | 1% (F1) | 1.5 | 2 | 1 | |
| M2 | 1.5 | 2 | 5 | ||
| M3 | 1.5 | 2 | 10 | ||
| M4 | 1.5 | 2 | 15 | ||
| M5 | 1.5 | 2 | 20 | ||
| M6 | 10% (F10) | 1.5 | 2 | 1 | |
| M7 | 20% (F20) | 1.5 | 2 | 1 | |
| F |  | 6.5 | 2.2 | 1 | |
| H |  | 1% (F1) | 6.5 | 2.2 | 1 |
| Q |  | 6.5 | 2.2 | 1 | |
| Run | DC 1 (wt%) | Number of Tablet Unit(s) | Tablet Mass (mg) | Drug Mass (mg) | RSD | LC% 2 | AV 3 |
|---|---|---|---|---|---|---|---|
| M1 | 1 | 1 | 19.18 ± 0.32 | 0.19 ± 0.01 | 4.57 | 99.67 ± 3.92 | 7.83 |
| M2 | 1 | 5 | 97.97 ± 1.95 | 1.04 ± 0.03 | 2.62 | 105.96 ± 0.84 | 0.44 |
| M3 | 1 | 10 | 191.53 ± 1.12 | 1.93 ± 0.01 | 0.77 | 100.79 ± 0.22 | 1.78 |
| M4 | 1 | 15 | 279.77 ± 3.84 | 2.85 ± 0.06 | 2.01 | 101.97 ± 0.66 | 2.39 |
| M5 | 1 | 20 | 379.13 ± 2.15 | 3.91 ± 0.02 | 0.60 | 103.17 ± 0.36 | 6.49 |
| F | 1 | 1 | 312.23 ± 11.11 | 2.93 ± 0.07 | 2.44 | 93.92 ± 1.04 | 6.67 |
| H | 1 | 0.5 | 201.97 ± 2.73 | 1.95 ± 0.02 | 0.97 | 96.73 ± 0.50 | 2.77 |
| Q | 1 | 0.25 | 104.63 ± 0.73 | 1.00 ± 0.00 | 0.33 | 95.92 ± 0.08 | 2.75 |
| M6 | 10 | 1 | 20.03 ± 0.39 | 1.95 ± 0.04 | 1.90 | 97.25 ± 0.52 | 2.30 |
| M7 | 20 | 1 | 19.60 ± 0.32 | 3.76 ± 0.06 | 1.61 | 96.00 ± 0.35 | 3.19 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gorkem Buyukgoz, G.; Kossor, C.G.; Ji, S.; Guvendiren, M.; Davé, R.N. Dose Titration of Solid Dosage Forms via FDM 3D-Printed Mini-Tablets. Pharmaceutics 2022, 14, 2305. https://doi.org/10.3390/pharmaceutics14112305
Gorkem Buyukgoz G, Kossor CG, Ji S, Guvendiren M, Davé RN. Dose Titration of Solid Dosage Forms via FDM 3D-Printed Mini-Tablets. Pharmaceutics. 2022; 14(11):2305. https://doi.org/10.3390/pharmaceutics14112305
Chicago/Turabian StyleGorkem Buyukgoz, Guluzar, Christopher G. Kossor, Shen Ji, Murat Guvendiren, and Rajesh N. Davé. 2022. "Dose Titration of Solid Dosage Forms via FDM 3D-Printed Mini-Tablets" Pharmaceutics 14, no. 11: 2305. https://doi.org/10.3390/pharmaceutics14112305
APA StyleGorkem Buyukgoz, G., Kossor, C. G., Ji, S., Guvendiren, M., & Davé, R. N. (2022). Dose Titration of Solid Dosage Forms via FDM 3D-Printed Mini-Tablets. Pharmaceutics, 14(11), 2305. https://doi.org/10.3390/pharmaceutics14112305









