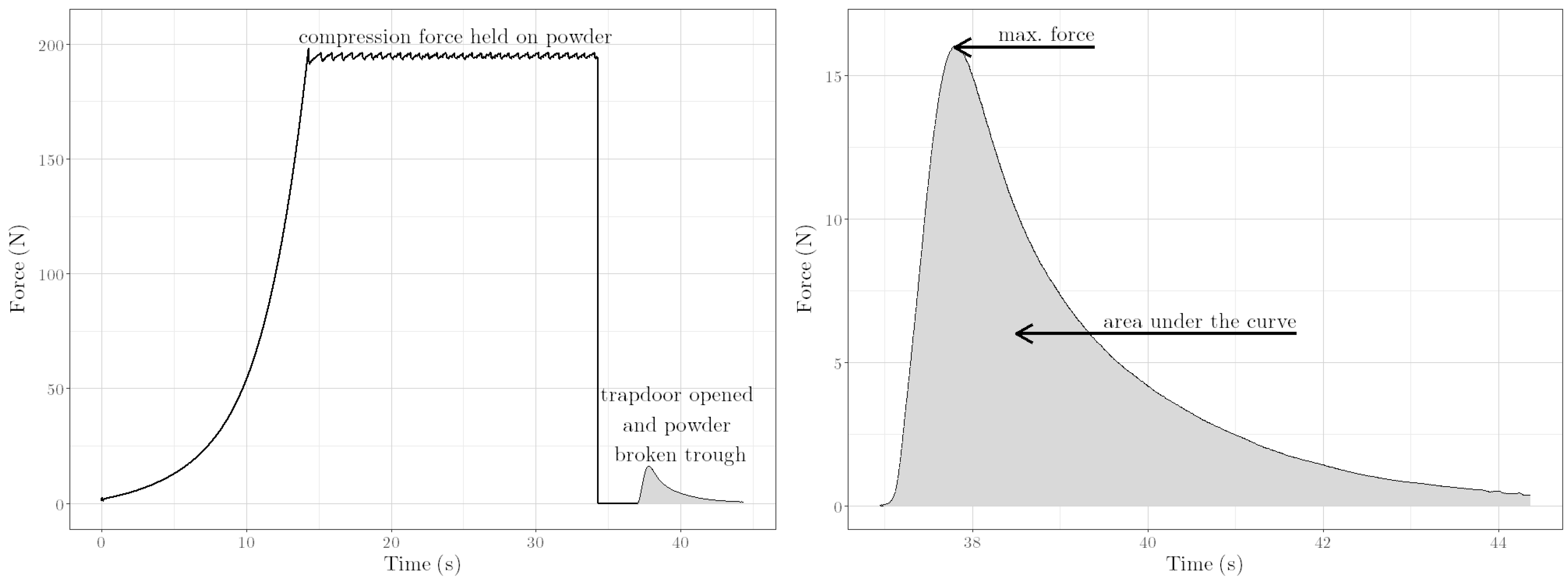
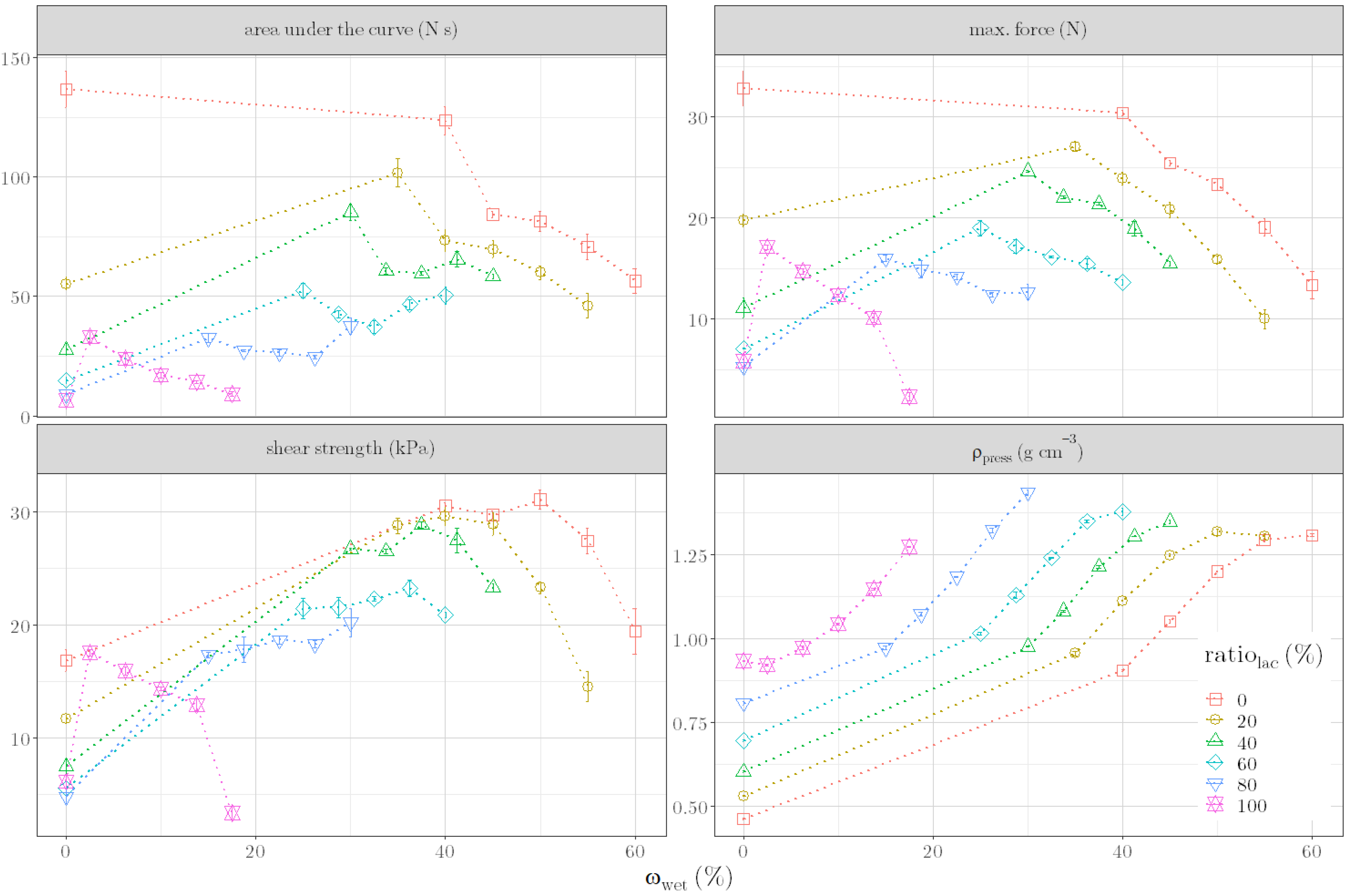
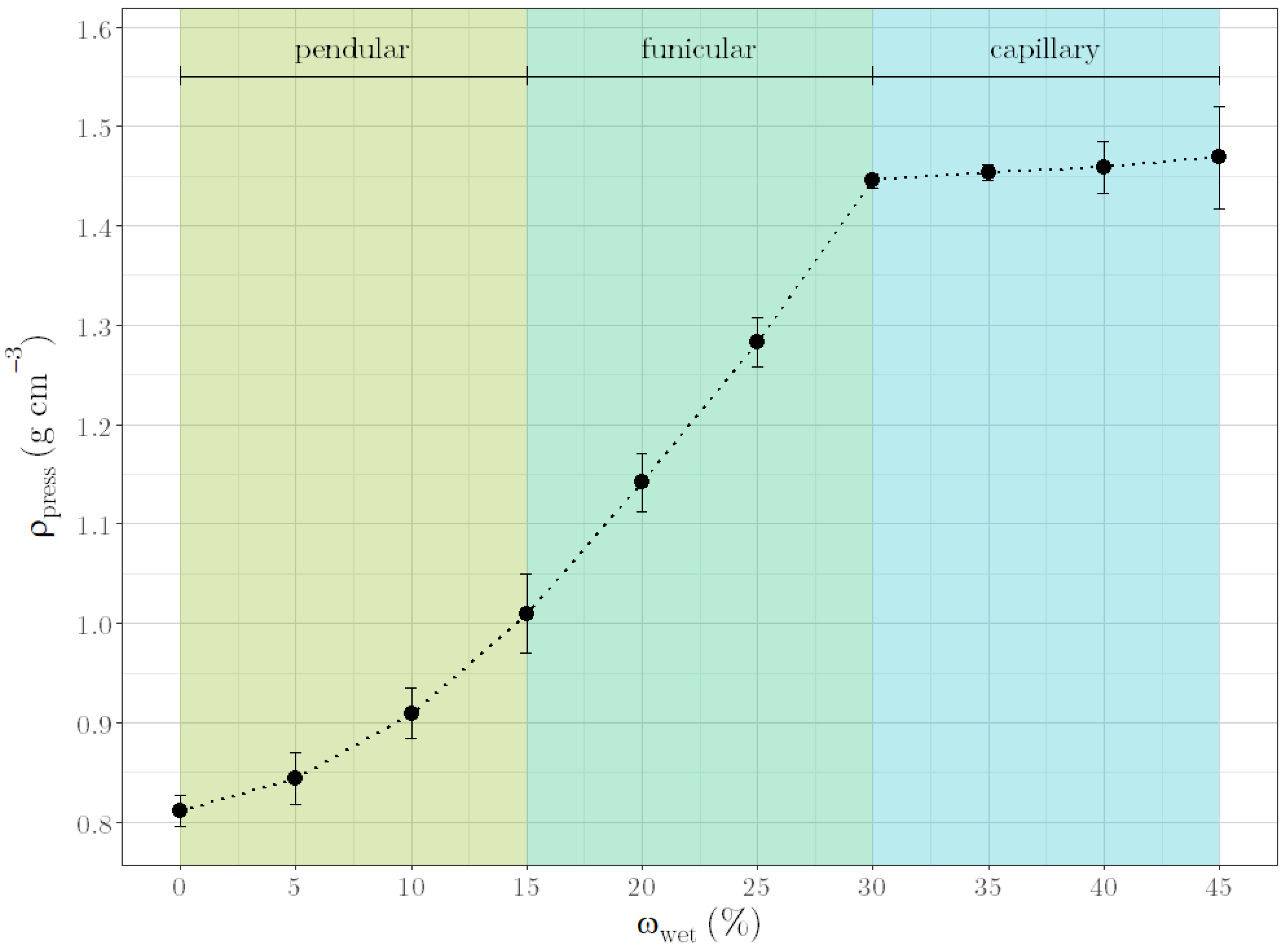
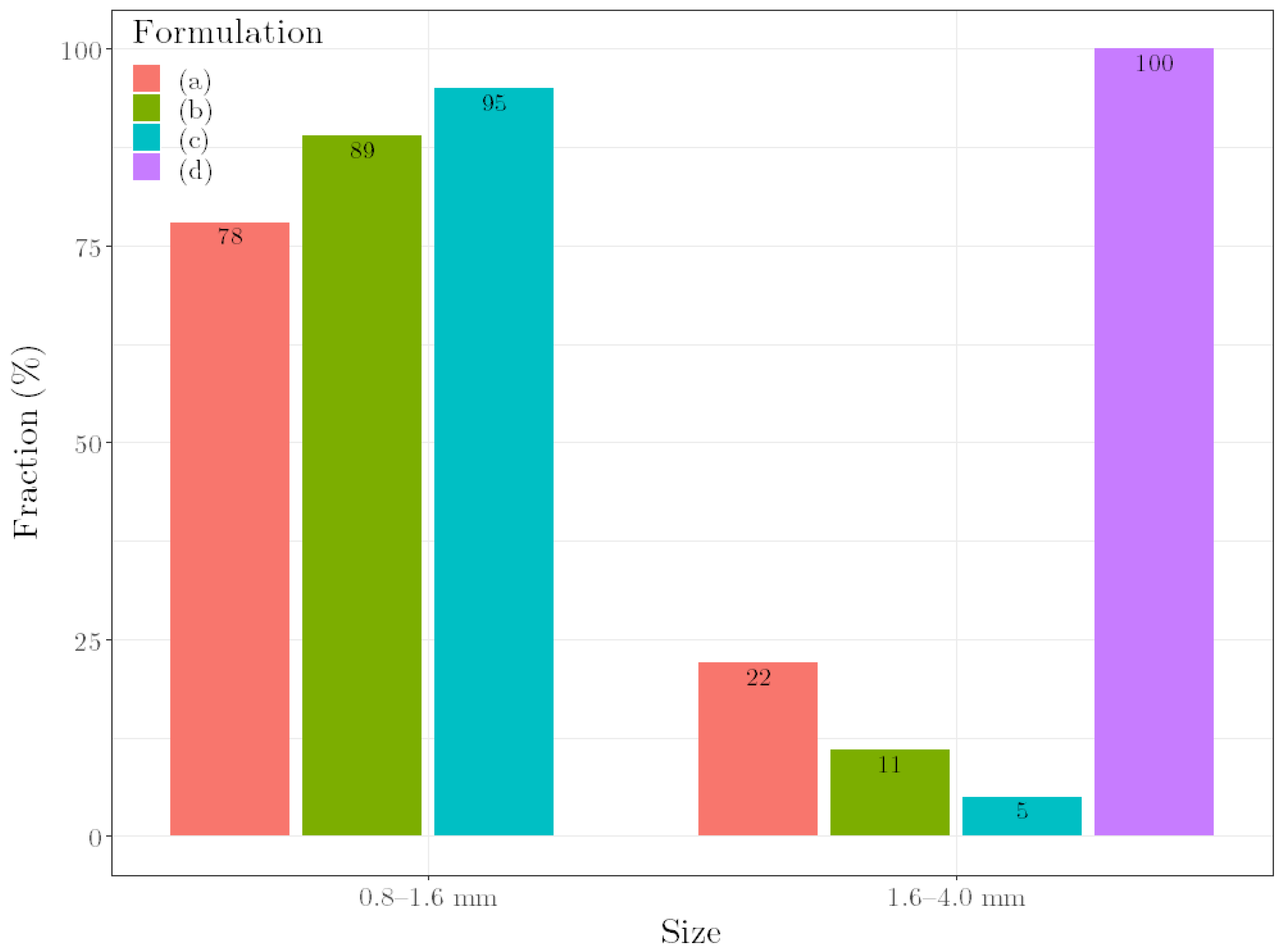
1. Introduction
In the pharmaceutical industry granulation is used as a process for size enlargement of small particles. For the granulation of pharmaceutical solids, wet granulation is often applied. This process starts with mixing dry powder, followed by wetting and nucleation once the liquid is added. The nuclei form the basis for the following granule growth and consolidation. If too much liquid is added, granules become a wet mass or slurry due to oversaturation [
1,
2]. It can thus be concluded that the moisture content is a key factor for the quality of granules [
3]. The amount of liquid determines the degree of saturation in the granule structure. The saturation degree is defined by the percentage of intragranular voids filled with liquid and can be described by three states of granules. With increasing liquid loading, the structure changes from pendular, over funicular to capillary state as the saturation reaches 100% [
2]. Beyond full saturation, powder particles become suspended in a continuous liquid phase [
4].
A critical unit operation is the determination of liquid requirement as there is no intrinsic endpoint. In fact, the optimal endpoint depends on the desired properties of the granules [
5], on process conditions and the applied equipment [
1], and on the physical properties of the dry raw materials, such as solubility, surface area, particle shape and size distribution. These properties influence the packing pattern of the powder mixture and, thus, the degree of densification and, consequently, the saturation level of the mixture associated with the liquid requirement [
2,
6].
Therefore, many different approaches exist for determining the required moisture content for granules. An objective method is estimating the required liquid amount by applying a mathematical calculation. For this purpose, Leuenberger et al. [
7] developed the following equation.
In Equation (1), is the required water content on a dry basis, is the porosity of the tapped powder, is the density of the liquid, is the true density of the solid. corresponds to a mean complete formation of liquid bridges in the pile and is the equilibrium moisture content of the product at 100% air humidity. Limitations for the use of this equation are, on the one hand, porosity to be estimated on the basis of the tapped density. On the other hand, for , the packing of the particles to be either cubic or rhombohedral has to be estimated, as well as the degree of filling pore spaces. Furthermore, this calculation only provides the optimal moisture content related to the saturation level, but it does not consider the mechanical properties of the granules.
These properties are even more important if granules are further processed [
8], for example, during tabletting or extrusion. In a multi-step manufacturing process, such as pellet production, granulation is followed by extrusion and spheronization. The liquid requirement of the initial granule mass thus directly correlates with the pellet quality [
3]. The so-called hand pressure test is, for this reason, typically applied during formulation development. In this test, the granules are compressed by hand, and if the mass does not crumble, the amount of liquid is suitable for further processing [
9,
10,
11,
12]. The main disadvantage of this method is the subjectivity and the required experience needed to reproducibly perform this test. A scientifically sound upgrade of this method is the torque rheological characterization performed with a mixing torque rheometer [
11,
13,
14,
15]. The changes in torque thereby display the resistance of the granule mass relative to mixing. Plotting changes in torque against the binder ratio allows identifying different wetting phases during granulation. Moreover, the binder ratio yielding the maximum change in torque was identified as optimal [
13,
14,
15]. This optimal value has also been shown to be relevant for extrusion–spheronization [
11]. Indirectly, the liquid requirement can also be evaluated by physical properties of the granules, such as bulk and tapped density and the resulting Carr’s compressibility index [
16,
17,
18]. The disadvantage is that the samples must first be dried before standard analytical methods; for example, sieving for particle sizing can be used.
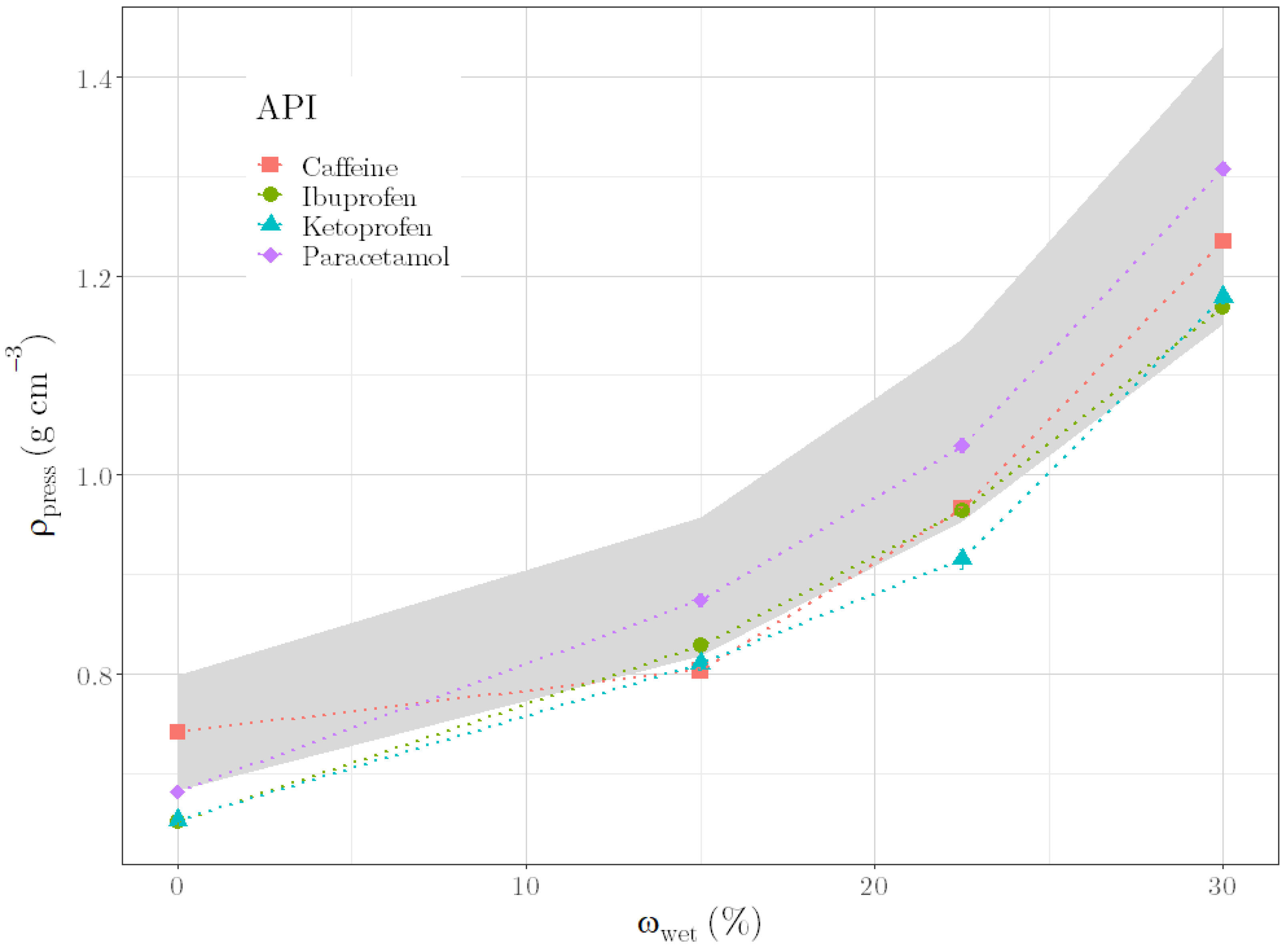
In our work, we aimed to identify an alternative parameter that can predict the quality of the pellets already during granulation by means of the liquid requirement of the formulation. Based on a placebo formulation, a suitable alternative quality parameter, connected with the formulation’s composition and with the moisture content, should be identified. The applicability of this parameter should be verified by predicting liquid requirements for further formulation compositions. In addition, we wanted to carry out extrusion and spheronization based on predicted moisture contents and to discuss the quality of resulting pellets. Since formulations containing active pharmaceutical ingredients (APIs) are more commonly produced in the industry than placebo formulations, initial experiments should be conducted to assess whether the identified parameter could also be applied for API formulations.
4. Conclusions
To evaluate the quality of the granules during granulation, the ρpress of the granule mass was identified as an alternative parameter. This parameter showed a relation with formulation compositions as well as with moisture content. A regression model, based on this relation, was successfully used to predict liquid requirements for unknown formulation compositions. In addition, uniform pellets were produced by carrying out the multi-step manufacturing process of granulation, extrusion and spheronization with the predicted liquid requirements for different formulations. Since these results could verify the suitability of ρpress as an alternative quality parameter, the applicability of the parameter to other placebo formulations was demonstrated. Moreover, first experiments with API formulations suggested the applicability to industrially relevant formulations. In conclusion, the results indicate that once a ρpress is defined for an optimal formulation, changes in material batches and even material types could be compensated with the right amount of water. However, one remaining question is which material properties, in addition to the tapped density and the brittleness, influence ρpress or correlate with it. If defined material properties could be determined, ρpress would not have to be defined for an optimal formulation but could be determined generally for corresponding material properties. One remaining disadvantage of the presented method is that the optimal ρpress for ideal further processing depends on the process parameters of the further processing itself (such as the extruder die). Therefore, no generally valid statement can be made about an optimal ρpress; this must be individually adapted to the multi-step manufacturing process’ parameters used.