In Vitro Studies of Graphene for Management of Dental Caries and Periodontal Disease: A Concise Review
Abstract
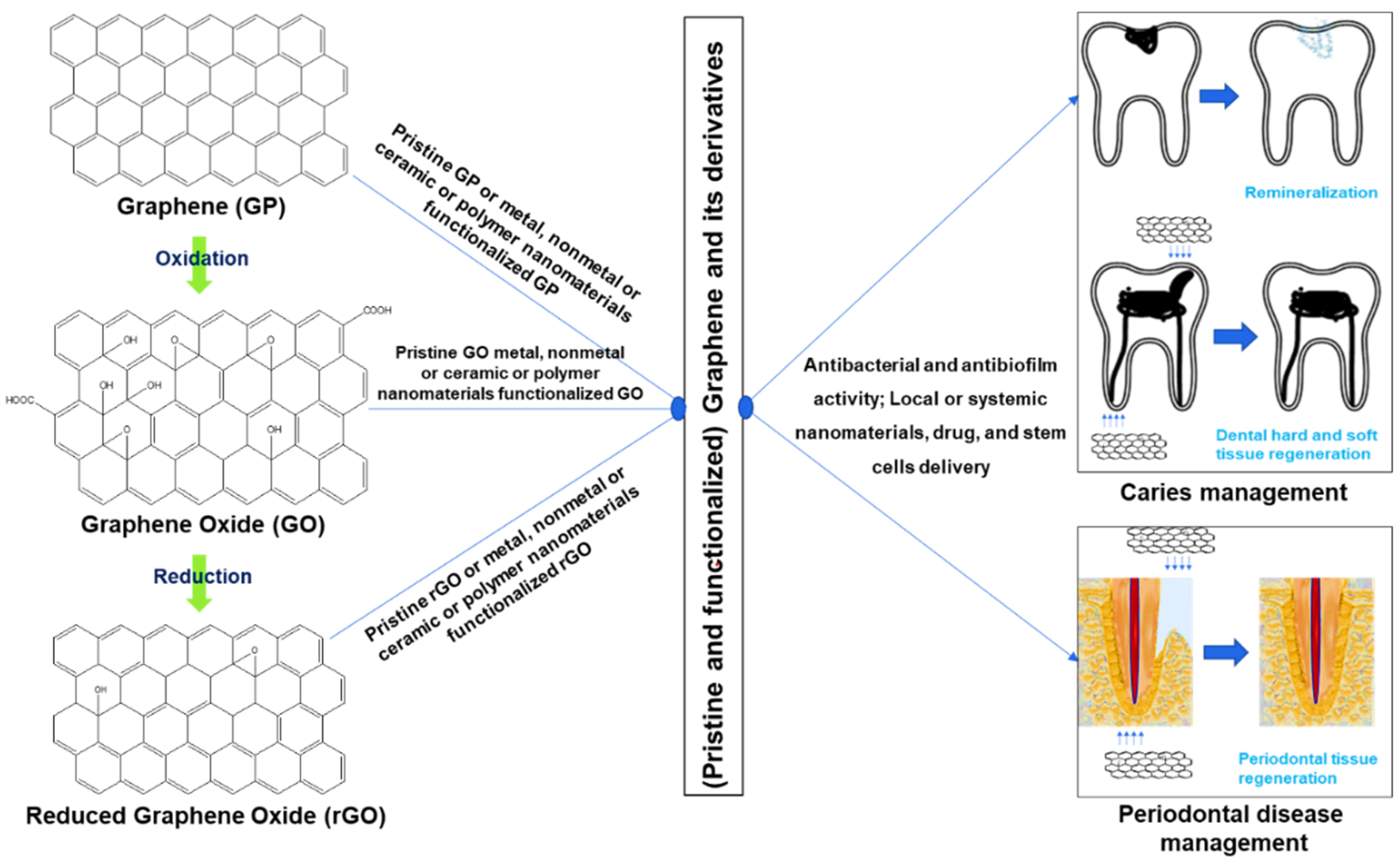
1. Introduction
2. Materials and Methods
2.1. Graphene Derivatives Used for Caries and Periodontal Disease Management
2.2. Literature Survey
3. Result and Discussion
3.1. Graphene Derivatives in Caries Management
3.1.1. Application against Cariogenic Pathogens
3.1.2. Application for Tooth Remineralization
3.2. Graphene Derivatives in Periodontal Disease Management
3.2.1. Application against Periodontal Pathogens
3.2.2. Application for Periodontal Tissue Regeneration
3.2.3. Application for Periodontal Bone Regeneration
| Graphene and Its Derivatives | Properties [Reference(s)] |
|---|---|
| Graphene | |
| Graphene | Inhibits oral fungi biofilm [107] Inhibits oral periodontal pathogenic bacteria [98] Increases bone regeneration [122,135,137,140] |
| Graphene-hydroxyapatite | Increases bone regeneration [136] |
| Graphene-titanium | Inhibits periodontal pathogenic bacteria [97,99] |
| Graphene Oxide | |
| Graphene Oxide | Inhibits periodontal pathogenic biofilms [60,103,104,105] Increases bone regeneration [104] |
| Increases periodontal tissue regeneration [113,121] | |
| Graphene Oxide-polyetheretherketone | Increases bone regeneration [145] |
| Graphene Oxide-chitosan | Inhibits periodontal pathogenic biofilms [101,102] |
| Graphene Oxide-hydroxyapatite | Increases bone regeneration [132] |
| Graphene Oxide-silk fibroin | Increases bone regeneration [116,125] |
| Graphene Oxide-titanium | Increases bone regeneration [120] |
| Graphene Oxide-chitosan-hydroxyapatite | Increases bone regeneration [138] |
| Graphene Oxide-lysozyme-titanium | Inhibits oral bacteria [100] Increases bone regeneration [100] |
| Graphene Oxide-minocycline hydrochloride-titanium | Inhibits oral bacteria [106] Increases bone regeneration [106] |
| Graphene Oxide-dexamethasone-titanium | Increases bone regeneration [126] |
| Graphene Oxide-bone morphogenetic protein 2-titanium | Increases bone regeneration [143] |
| Reduced Graphene Oxide | |
| Reduced Graphene Oxide | Increases bone regeneration [146,148] |
| Reduced Graphene Oxide-chitosan | Increases bone regeneration [123] |
| Reduced Graphene Oxide-hydroxyapatite | Increases bone regeneration [127] |
| Reduced Graphene Oxide-titanium | Increases bone regeneration [147] |
| Reduced Graphene Oxide-dexamethasone-titanium | Increases bone regeneration [126] |
| Graphene OxideQuantum Dots | |
| Graphene Oxide Quantum Dots | Living cell labeling [128] |
| Graphene Oxide Quantum Dots-curcumin | Inhibits periodontal pathogenic bacteria [109] |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Allen, M.J.; Tung, V.C.; Kaner, R.B. Honeycomb carbon: A review of graphene. Chem. Rev. 2010, 110, 132–145. [Google Scholar] [PubMed]
- Gurunathan, S.; Kim, J.-H. Synthesis, toxicity, biocompatibility, and biomedical applications of graphene and graphene-related materials. Int. J. Nanomed. 2016, 11, 1927. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-Y.; An, S.S.A.; Hulme, J. Current applications of graphene oxide in nanomedicine. Int. J. Nanomed. 2015, 10, 9. [Google Scholar]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286. [Google Scholar] [PubMed]
- Geim, A.K. Graphene: Status and prospects. Science 2009, 324, 1530–1534. [Google Scholar]
- De Bellis, G.; Tamburrano, A.; Dinescu, A.; Santarelli, M.L.; Sarto, M.S. Electromagnetic properties of composites containing graphite nanoplatelets at radio frequency. Carbon 2011, 49, 4291–4300. [Google Scholar] [CrossRef]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and graphene oxide: Synthesis, properties, and applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef]
- Huang, X.; Yin, Z.; Wu, S.; Qi, X.; He, Q.; Zhang, Q.; Yan, Q.; Boey, F.; Zhang, H. Graphene-based materials: Synthesis, characterization, properties, and applications. Small 2011, 7, 1876–1902. [Google Scholar]
- Bykkam, S.; Narsingam, S.; Ahmadipour, M.; Dayakar, T.; Rao, K.V.; Chakra, C.S.; Kalakotla, S. Few layered graphene sheet decorated by ZnO nanoparticles for anti-bacterial application. Superlattices Microstruct. 2015, 83, 776–784. [Google Scholar] [CrossRef]
- Liu, L.; Bai, H.; Liu, J.; Sun, D.D. Multifunctional graphene oxide-TiO2-Ag nanocomposites for high performance water disinfection and decontamination under solar irradiation. J. Hazard. Mater. 2013, 261, 214–223. [Google Scholar] [CrossRef]
- Qu, J.-C.; Ren, C.-L.; Dong, Y.-L.; Chang, Y.-P.; Zhou, M.; Chen, X.-G. Facile synthesis of multifunctional graphene oxide/AgNPs-Fe3O4 nanocomposite: A highly integrated catalysts. Chem. Eng. J. 2012, 211, 412–420. [Google Scholar] [CrossRef]
- Ji, H.; Sun, H.; Qu, X. Antibacterial applications of graphene-based nanomaterials: Recent achievements and challenges. Adv. Drug Deliv. Rev. 2016, 105, 176–189. [Google Scholar] [CrossRef]
- Nizami, M.Z.I.; Nishina, Y.; Yamamoto, T.; Shinoda-Ito, Y.; Takashiba, S. Functionalized graphene oxide shields tooth dentin from decalcification. J. Dent. Res. 2020, 99, 182–188. [Google Scholar] [CrossRef]
- Safari, N.; Golafshan, N.; Kharaziha, M.; Reza Toroghinejad, M.; Utomo, L.; Malda, J.; Castilho, M. Stable and Antibacterial Magnesium–Graphene Nanocomposite-Based Implants for Bone Repair. ACS Biomater. Sci. Eng. 2020, 6, 6253–6262. [Google Scholar] [CrossRef]
- Jia, L.-C.; Sun, W.-J.; Zhou, C.-G.; Yan, D.-X.; Zhang, Q.-C.; Li, Z.-M. Integrated strength and toughness in graphene/calcium alginate films for highly efficient electromagnetic interference shielding. J. Mater. Chem. C 2018, 6, 9166–9174. [Google Scholar] [CrossRef]
- Jiang, H. Chemical preparation of graphene-based nanomaterials and their applications in chemical and biological sensors. Small 2011, 7, 2413–2427. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Dong, S. Graphene nanosheet: Synthesis, molecular engineering, thin film, hybrids, and energy and analytical applications. Chem. Soc. Rev. 2011, 40, 2644–2672. [Google Scholar] [CrossRef]
- Liao, C.; Li, Y.; Tjong, S.C. Graphene Nanomaterials: Synthesis, Biocompatibility, and Cytotoxicity. Int. J. Mol. Sci. 2018, 19, 3564. [Google Scholar] [CrossRef]
- Kumar, P.; Huo, P.; Zhang, R.; Liu, B. Antibacterial Properties of Graphene-Based Nanomaterials. Nanomaterials 2019, 9, 737. [Google Scholar] [CrossRef]
- Nizami, M.Z.I.; Takashiba, S.; Nishina, Y. Graphene oxide: A new direction in dentistry. Appl. Mater. Today 2020, 19, 100576. [Google Scholar] [CrossRef]
- Shang, L.; Qi, Y.; Lu, H.; Pei, H.; Li, Y.; Qu, L.; Wu, Z.; Zhang, W. Graphene and graphene oxide for tissue engineering and regeneration. In Theranostic Bionanomaterials; Elsevier: Amsterdam, The Netherlands, 2019; pp. 165–185. [Google Scholar]
- Nyvad, B.; Crielaard, W.; Mira, A.; Takahashi, N.; Beighton, D. Dental caries from a molecular microbiological perspective. Caries Res. 2013, 47, 89–102. [Google Scholar] [CrossRef]
- Eke, P.I.; Dye, B.A.; Wei, L.; Thornton-Evans, G.O.; Genco, R.J. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J. Dent. Res. 2012, 91, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Wade, W.G. The oral microbiome in health and disease. Pharm. Res. 2013, 69, 137–143. [Google Scholar] [CrossRef]
- Loesche, W.J. Role of Streptococcus mutans in human dental decay. Microbiol. Rev. 1986, 50, 353–380. [Google Scholar] [CrossRef] [PubMed]
- Binder Gallimidi, A.; Fischman, S.; Revach, B.; Bulvik, R.; Maliutina, A.; Rubinstein, A.M.; Nussbaum, G.; Elkin, M. Periodontal pathogens Porphyromonas gingivalis and Fusobacterium nucleatum promote tumor progression in an oral-specific chemical carcinogenesis model. Oncotarget 2015, 6, 22613–22623. [Google Scholar] [CrossRef] [PubMed]
- Marcenes, W.; Kassebaum, N.J.; Bernabé, E.; Flaxman, A.; Naghavi, M.; Lopez, A.; Murray, C.J. Global burden of oral conditions in 1990–2010: A systematic analysis. J. Dent. Res. 2013, 92, 592–597. [Google Scholar] [CrossRef]
- Nazir, M.; Al-Ansari, A.; Al-Khalifa, K.; Alhareky, M.; Gaffar, B.; Almas, K. Global Prevalence of Periodontal Disease and Lack of Its Surveillance. Sci. World J. 2020, 2020, 2146160. [Google Scholar] [CrossRef]
- Sader, F.; Denis, J.-F.; Roy, S. Tissue regeneration in dentistry: Can salamanders provide insight? Oral Dis. 2018, 24, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Ripamonti, U. Redefining the induction of periodontal tissue regeneration in primates by the osteogenic proteins of the transforming growth factor-β supergene family. J. Periodontal. Res. 2016, 51, 699–715. [Google Scholar] [CrossRef] [PubMed]
- Balic, A. Biology Explaining Tooth Repair and Regeneration: A Mini-Review. Gerontology 2018, 64, 382–388. [Google Scholar] [CrossRef]
- Amrollahi, P.; Shah, B.; Seifi, A.; Tayebi, L. Recent advancements in regenerative dentistry: A review. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 69, 1383–1390. [Google Scholar] [CrossRef] [PubMed]
- Abou Neel, E.A.; Chrzanowski, W.; Salih, V.M.; Kim, H.W.; Knowles, J.C. Tissue engineering in dentistry. J. Dent. 2014, 42, 915–928. [Google Scholar] [CrossRef]
- Magne, T.M.; de Oliveira Vieira, T.; Alencar, L.M.R.; Junior, F.F.M.; Gemini-Piperni, S.; Carneiro, S.V.; Fechine, L.; Freire, R.M.; Golokhvast, K.; Metrangolo, P.; et al. Graphene and its derivatives: Understanding the main chemical and medicinal chemistry roles for biomedical applications. J. Nanostructure Chem. 2021, 12, 693–727. [Google Scholar] [CrossRef]
- Kuila, T.; Bose, S.; Mishra, A.K.; Khanra, P.; Kim, N.H.; Lee, J.H. Chemical functionalization of graphene and its applications. Prog. Mater. Sci. 2012, 57, 1061–1105. [Google Scholar] [CrossRef]
- Banerjee, A.N. Graphene and its derivatives as biomedical materials: Future prospects and challenges. Interface Focus 2018, 8, 20170056. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, M.; Kang, F. Graphene derivatives: Graphane, fluorographene, graphene oxide, graphyne and graphdiyne. J. Mater. Chem. A 2014, 2, 13193–13206. [Google Scholar] [CrossRef]
- Al Jahdaly, B.A.; Elsadek, M.F.; Ahmed, B.M.; Farahat, M.F.; Taher, M.M.; Khalil, A.M. Outstanding Graphene Quantum Dots from Carbon Source for Biomedical and Corrosion Inhibition Applications: A Review. Sustainability 2021, 13, 2127. [Google Scholar] [CrossRef]
- Sahu, D.; Sutar, H.; Senapati, P.; Murmu, R.; Roy, D. Graphene, Graphene-Derivatives and Composites: Fundamentals, Synthesis Approaches to Applications. J. Compos. Sci. 2021, 5, 181. [Google Scholar] [CrossRef]
- Dimiev, A.M.; Eigler, S. Graphene oxide: Fundamentals and applications; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Dreyer, D.R.; Park, S.; Bielawski, C.W.; Ruoff, R.S. The chemistry of graphene oxide. Chem. Soc. Rev. 2010, 39, 228–240. [Google Scholar] [CrossRef]
- Jeong, H.Y.; Kim, J.Y.; Kim, J.W.; Hwang, J.O.; Kim, J.-E.; Lee, J.Y.; Yoon, T.H.; Cho, B.J.; Kim, S.O.; Ruoff, R.S. Graphene oxide thin films for flexible nonvolatile memory applications. Nano Lett. 2010, 10, 4381–4386. [Google Scholar] [CrossRef]
- Kavitha, C. A review on reduced Graphene oxide hybrid nano composites and their prominent applications. Mater. Today: Proc. 2022, 49, 811–816. [Google Scholar] [CrossRef]
- Razaq, A.; Bibi, F.; Zheng, X.; Papadakis, R.; Jafri, S.H.M.; Li, H. Review on Graphene-, Graphene Oxide-, Reduced Graphene Oxide-Based Flexible Composites: From Fabrication to Applications. Materials 2022, 15, 1012. [Google Scholar] [CrossRef] [PubMed]
- Priyadarsini, S.; Mohanty, S.; Mukherjee, S.; Basu, S.; Mishra, M. Graphene and graphene oxide as nanomaterials for medicine and biology application. J. Nanostruct. Chem. 2018, 8, 123–137. [Google Scholar] [CrossRef]
- Rout, D.R.; Jena, H.M. Removal of malachite green dye from aqueous solution using reduced graphene oxide as an adsorbent. Mater. Today Proc. 2021, 47, 1173–1182. [Google Scholar] [CrossRef]
- Li, L.; Wu, G.; Yang, G.; Peng, J.; Zhao, J.; Zhu, J.-J. Focusing on luminescent graphene quantum dots: Current status and future perspectives. Nanoscale 2013, 5, 4015–4039. [Google Scholar] [CrossRef]
- Ritter, K.A.; Lyding, J.W. The influence of edge structure on the electronic properties of graphene quantum dots and nanoribbons. Nat. Mater. 2009, 8, 235–242. [Google Scholar] [CrossRef]
- Chen, F.; Gao, W.; Qiu, X.; Zhang, H.; Liu, L.; Liao, P.; Fu, W.; Luo, Y. Graphene quantum dots in biomedical applications: Recent advances and future challenges. Front. Lab. Med. 2017, 1, 192–199. [Google Scholar] [CrossRef]
- Wong, A.; Subar, P.E.; Young, D.A. Dental Caries: An Update on Dental Trends and Therapy. Adv. Pediatr. 2017, 64, 307–330. [Google Scholar] [CrossRef]
- Pitts, N.B.; Zero, D.T.; Marsh, P.D.; Ekstrand, K.; Weintraub, J.A.; Ramos-Gomez, F.; Tagami, J.; Twetman, S.; Tsakos, G.; Ismail, A. Dental caries. Nat. Rev. Dis. Primers 2017, 3, 17030. [Google Scholar] [CrossRef]
- Nizami, M.Z.I.; Xu, V.W.; Yin, I.X.; Yu, O.Y.; Chu, C.H. Metal and Metal Oxide Nanoparticles in Caries Prevention: A Review. Nanomaterials 2021, 11, 3446. [Google Scholar] [CrossRef]
- Arifa, M.K.; Ephraim, R.; Rajamani, T. Recent advances in dental hard tissue remineralization: A review of literature. Int. J. Clin. Pediatric Dent. 2019, 12, 139. [Google Scholar] [CrossRef]
- Neel, E.; Aljabo, A.; Strange, A.; Ibrahim, S.; Coathup, M.; Young, A.; Mudera, V. Demineralization–remineralization dynamics in teeth and bone. Int. J. Nanomed. 2016, 11, 4743. [Google Scholar] [CrossRef] [PubMed]
- Bader, J.D.; Shugars, D.A.; Bonito, A.J. A systematic review of selected caries prevention and management methods. Community Dent. Oral Epidemiol. 2001, 29, 399–411. [Google Scholar] [CrossRef]
- Hu, W.; Peng, C.; Luo, W.; Lv, M.; Li, X.; Li, D.; Huang, Q.; Fan, C. Graphene-based antibacterial paper. ACS Nano 2010, 4, 4317–4323. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhao, Q.; Peng, J.; Yang, X.; Yu, D.; Zhao, W. Antibacterial and mechanical properties of reduced graphene-silver nanoparticle nanocomposite modified glass ionomer cements. J. Dent. 2020, 96, 103332. [Google Scholar] [CrossRef] [PubMed]
- Akram, Z.; Aati, S.; Clode, P.; Saunders, M.; Ngo, H.; Fawzy, A.S. Formulation of nano-graphene doped with nano silver modified dentin bonding agents with enhanced interfacial stability and antibiofilm properties. Dent. Mater. 2022, 38, 347–362. [Google Scholar] [CrossRef] [PubMed]
- Kulshrestha, S.; Khan, S.; Meena, R.; Singh, B.R.; Khan, A.U. A graphene/zinc oxide nanocomposite film protects dental implant surfaces against cariogenic Streptococcus mutans. Biofouling 2014, 30, 1281–1294. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Zhu, X.; Qi, Z.; Wang, C.; Mao, X.; Zhu, C.; He, Z.; Li, M.; Tang, Z. Killing dental pathogens using antibacterial graphene oxide. ACS Appl. Mater. Interfaces 2015, 7, 5605–5611. [Google Scholar] [CrossRef]
- Zhao, M.; Shan, T.; Wu, Q.; Gu, L. The Antibacterial Effect of Graphene Oxide on Streptococcus mutans. J. Nanosci. Nanotechnol. 2020, 20, 2095–2103. [Google Scholar] [CrossRef]
- Mao, M.; Zhang, W.; Huang, Z.; Huang, J.; Wang, J.; Li, W.; Gu, S. Graphene Oxide-Copper Nanocomposites Suppress Cariogenic Streptococcus mutans Biofilm Formation. Int. J. Nanomed. 2021, 16, 7727–7739. [Google Scholar] [CrossRef]
- Rago, I.; Bregnocchi, A.; Zanni, E.; D’Aloia, A.; De Angelis, F.; Bossu, M.; De Bellis, G.; Polimeni, A.; Uccelletti, D.; Sarto, M. Antimicrobial activity of graphene nanoplatelets against Streptococcus mutans. In Proceedings of the 2015 IEEE 15th International Conference on Nanotechnology (IEEE-NANO), Rome, Italy, 27–30 July 2015; pp. 9–12. [Google Scholar]
- Nasim, I.; Kumar, S.R.; Vishnupriya, V.; Jabin, Z. Cytotoxicity and anti-microbial analysis of silver and graphene oxide bio nanoparticles. Bioinformation 2020, 16, 831. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Yang, X.; Chen, Y.; Fu, Y.; Yu, D.; Zhao, W. Effect of graphene oxide-silver nanocomposites on Streptococcus mutans proliferation and biofilm formation. Chin. J. Stomatol. Res. (Electron. Ed.) 2018, 12, 83. [Google Scholar]
- Bregnocchi, A.; Zanni, E.; Uccelletti, D.; Marra, F.; Cavallini, D.; De Angelis, F.; De Bellis, G.; Bossù, M.; Ierardo, G.; Polimeni, A. Graphene-based dental adhesive with anti-biofilm activity. J. Nanobiotech. 2017, 15, 89. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.-Y.; Zhu, G.-Y.; Yu, C.-H.; Chen, G.-Y.; Zhang, C.-L.; Zeng, X.; Chen, Q.-M.; Peng, Q. Functionalized graphene oxide nanosheets with unique three-in-one properties for efficient and tunable antibacterial applications. Nano Res. 2021, 14, 185–190. [Google Scholar] [CrossRef]
- Joshi, S.; Siddiqui, R.; Sharma, P.; Kumar, R.; Verma, G.; Saini, A. Green synthesis of peptide functionalized reduced graphene oxide (rGO) nano bioconjugate with enhanced antibacterial activity. Sci. Rep. 2020, 10, 9441. [Google Scholar] [CrossRef] [PubMed]
- Jang, W.; Kim, H.-S.; Alam, K.; Ji, M.-K.; Cho, H.-S.; Lim, H.-P. Direct-deposited graphene oxide on dental implants for antimicrobial activities and osteogenesis. Int. J. Nanomed. 2021, 16, 5745. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-H.; Chen, G.-Y.; Xia, M.-Y.; Xie, Y.; Chi, Y.-Q.; He, Z.-Y.; Zhang, C.-L.; Zhang, T.; Chen, Q.-M.; Peng, Q. Understanding the sheet size-antibacterial activity relationship of graphene oxide and the nano-bio interaction-based physical mechanisms. Colloids Surf. B Biointerfaces 2020, 191, 111009. [Google Scholar] [CrossRef]
- Javanbakht, T.; Hadian, H.; Wilkinson, K. Comparative Study of Physicochemical Properties and Antibiofilm Activity of Graphene Oxide Nanoribbons. 2020. Available online: https://essuir.sumdu.edu.ua/handle/123456789/77165 (accessed on 18 September 2022).
- Gamal, R.; Gomaa, Y.F.; Said, A.M. Incorporating nano graphene oxide to poly-methyl methacrylate; antibacterial effect and thermal expansion. J. Mod. Res. 2019, 1, 19–23. [Google Scholar] [CrossRef]
- Sun, L.; Yan, Z.; Duan, Y.; Zhang, J.; Liu, B. Improvement of the mechanical, tribological and antibacterial properties of glass ionomer cements by fluorinated graphene. Dent. Mater. 2018, 34, e115–e127. [Google Scholar] [CrossRef]
- Di Carlo, S.; De Angelis, F.; Brauner, E.; Pranno, N.; Tassi, G.; Senatore, M.; Bossù, M. Flexural strength and elastic modulus evaluation of structures made by conventional PMMA and PMMA reinforced with graphene. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 5201–5208. [Google Scholar]
- Gholibegloo, E.; Karbasi, A.; Pourhajibagher, M.; Chiniforush, N.; Ramazani, A.; Akbari, T.; Bahador, A.; Khoobi, M. Carnosine-graphene oxide conjugates decorated with hydroxyapatite as promising nanocarrier for ICG loading with enhanced antibacterial effects in photodynamic therapy against Streptococcus mutans. J. Photochem. Photobiol. B Biol. 2018, 181, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Liu, Y.; Zhang, H.; Lei, L. Nano-graphene oxide with antisense vicR RNA reduced exopolysaccharide synthesis and biofilm aggregation for Streptococcus mutans. Dent. Mater. J. 2020, 39, 2019–2039. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A.M.; Goncalves, I.C.; Magalhaes, F.D. Graphene-based materials biocompatibility: A review. Colloids Surf. B Biointerfaces 2013, 111, 188–202. [Google Scholar] [CrossRef] [PubMed]
- Nam, H.-J.; Kim, Y.-M.; Kwon, Y.H.; Kim, I.-R.; Park, B.-S.; Son, W.-S.; Lee, S.-M.; Kim, Y.-I. Enamel surface remineralization effect by fluorinated graphite and bioactive glass-containing orthodontic bonding resin. Materials 2019, 12, 1308. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Bai, Y.; Su, J.; Ma, W.; Jia, R.l. Graphene oxide/fluorhydroxyapatite composites with enhanced chemical stability, mechanical, and biological properties for dental applications. Int. J. Appl. Ceram. Technol. 2017, 14, 1088–1100. [Google Scholar] [CrossRef]
- Lee, S.-M.; Yoo, K.-H.; Yoon, S.-Y.; Kim, I.-R.; Park, B.-S.; Son, W.-S.; Ko, C.-C.; Son, S.-A.; Kim, Y.-I. Enamel anti-demineralization effect of orthodontic adhesive containing bioactive glass and graphene oxide: An in-vitro study. Materials 2018, 11, 1728. [Google Scholar] [CrossRef]
- Velo, M.M.d.A.C.; de Lima Nascimento, T.R.; Obeid, A.T.; Castellano, L.C.; Costa, R.M.; Brondino, N.C.M.; Fonseca, M.G.; Silikas, N.; Mondelli, R.F.L. Enhancing the mechanical properties and providing bioactive potential for graphene oxide/montmorillonite hybrid dental resin composites. Sci. Rep. 2022, 12, 10259. [Google Scholar] [CrossRef] [PubMed]
- Nahorny, S.; Zanin, H.; Christino, V.A.; Marciano, F.R.; Lobo, A.O.; Soares, L.E.S. Multi-walled carbon nanotubes/graphene oxide hybrid and nanohydroxyapatite composite: A novel coating to prevent dentin erosion. Mater. Sci. Eng. C 2017, 79, 199–208. [Google Scholar] [CrossRef]
- Wu, R.; Zhao, Q.; Lu, S.; Fu, Y.; Yu, D.; Zhao, W. Inhibitory effect of reduced graphene oxide-silver nanocomposite on progression of artificial enamel caries. J. Appl. Oral Sci. 2018, 27. [Google Scholar] [CrossRef]
- Núñez, J.D.; Benito, A.M.; Gonzalez, R.; Aragón, J.; Arenal, R.; Maser, W.K. Integration and bioactivity of hydroxyapatite grown on carbon nanotubes and graphene oxide. Carbon 2014, 79, 590–604. [Google Scholar] [CrossRef]
- Son, S.-A.; Kim, D.-H.; Yoo, K.-H.; Yoon, S.-Y.; Kim, Y.-I. Mesoporous bioactive glass combined with graphene oxide quantum dot as a new material for a new treatment option for dentin hypersensitivity. Nanomaterials 2020, 10, 621. [Google Scholar] [CrossRef] [PubMed]
- Zanni, E.; Chandraiahgari, C.R.; De Bellis, G.; Montereali, M.R.; Armiento, G.; Ballirano, P.; Polimeni, A.; Sarto, M.S.; Uccelletti, D. Zinc oxide nanorods-decorated graphene nanoplatelets: A promising antimicrobial agent against the cariogenic bacterium Streptococcus mutans. Nanomaterials 2016, 6, 179. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Jo, J.-K.; Kim, D.-A.; Patel, K.D.; Kim, H.-W.; Lee, H.-H. Nano-graphene oxide incorporated into PMMA resin to prevent microbial adhesion. Dent. Mater. 2018, 34, e63–e72. [Google Scholar] [CrossRef]
- Yin, D.; Li, Y.; Lin, H.; Guo, B.; Du, Y.; Li, X.; Jia, H.; Zhao, X.; Tang, J.; Zhang, L. Functional graphene oxide as a plasmid-based Stat3 siRNA carrier inhibits mouse malignant melanoma growth in vivo. Nanotechnology 2013, 24, 105102. [Google Scholar] [CrossRef] [PubMed]
- Lei, L.; Stipp, R.; Chen, T.; Wu, S.; Hu, T.; Duncan, M. Activity of Streptococcus mutans VicR is modulated by antisense RNA. J. Dent. Res. 2018, 97, 1477–1484. [Google Scholar] [CrossRef] [PubMed]
- Dou, C.; Ding, N.; Luo, F.; Hou, T.; Cao, Z.; Bai, Y.; Liu, C.; Xu, J.; Dong, S. Graphene-based microRNA transfection blocks preosteoclast fusion to increase bone formation and vascularization. Adv. Sci. 2018, 5, 1700578. [Google Scholar] [CrossRef]
- Roth, G. Global Burden of Disease Collaborative Network. Global Burden of Disease Study 2017 (GBD 2017) Results. Seattle, United States: Institute for Health Metrics and Evaluation (IHME), 2018. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Nair, S.; Faizuddin, M.; Dharmapalan, J. Role of autoimmune responses in periodontal disease. Autoimmune Dis. 2014, 2014, 596824. [Google Scholar] [CrossRef]
- Usui, M.; Onizuka, S.; Sato, T.; Kokabu, S.; Ariyoshi, W.; Nakashima, K. Mechanism of alveolar bone destruction in periodontitis—Periodontal bacteria and inflammation. Jpn. Dent. Sci. Rev. 2021, 57, 201–208. [Google Scholar] [CrossRef]
- Carcuac, O.; Berglundh, T. Composition of human peri-implantitis and periodontitis lesions. J. Dent. Res. 2014, 93, 1083–1088. [Google Scholar] [CrossRef]
- Radaelli, K.; Alberti, A.; Corbella, S.; Francetti, L. The Impact of Peri-Implantitis on Systemic Diseases and Conditions: A Review of the Literature. Int. J. Dent. 2021, 2021, 5536566. [Google Scholar] [CrossRef]
- Rafiei, M.; Kiani, F.; Sayehmiri, F.; Sayehmiri, K.; Sheikhi, A.; Zamanian Azodi, M. Study of Porphyromonas gingivalis in periodontal diseases: A systematic review and meta-analysis. Med. J. Islam. Repub. Iran 2017, 31, 62. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Qiao, S.; Zhang, X.; Li, Y.; Zhang, Y.; Wei, S.; Shi, J.; Lai, H. Graphene-reinforced titanium enhances soft tissue seal. Front. Bioeng. Biotechnol. 2021, 9, 665305. [Google Scholar] [CrossRef] [PubMed]
- Bacali, C.; Carpa, R.; Buduru, S.; Moldovan, M.L.; Baldea, I.; Constantin, A.; Moldovan, M.; Prodan, D.; Dascalu Rusu, L.M.; Lucaciu, O.; et al. Association of Graphene Silver Polymethyl Methacrylate (PMMA) with Photodynamic Therapy for Inactivation of Halitosis Responsible Bacteria in Denture Wearers. Nanomaterials 2021, 11, 1643. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhao, W.; Zhao, C.; Zhang, W.; Yan, Z. Graphene coated Ti-6Al-4V exhibits antibacterial and antifungal properties against oral pathogens. J. Prosthodont. 2022. [Google Scholar] [CrossRef]
- Li, M.; Li, H.; Pan, Q.; Gao, C.; Wang, Y.; Yang, S.; Zan, X.; Guan, Y. Graphene Oxide and Lysozyme Ultrathin Films with Strong Antibacterial and Enhanced Osteogenesis. Langmuir 2019, 35, 6752–6761. [Google Scholar] [CrossRef]
- Yang, S.; Yu, W.; Zhang, J.; Han, X.; Wang, J.; Sun, D.; Shi, R.; Zhou, Y.; Zhang, H.; Zhao, J. The antibacterial property of zinc oxide/graphene oxide modified porous polyetheretherketone against S. sanguinis, F. nucleatum and P. gingivalis. Biomed. Mater. 2022, 17, 025013. [Google Scholar] [CrossRef]
- Guo, C.; Lu, R.; Wang, X.; Chen, S. Graphene oxide-modified polyetheretherketone with excellent antibacterial properties and biocompatibility for implant abutment. Macromol. Res. 2021, 29, 351–359. [Google Scholar] [CrossRef]
- Pourhajibagher, M.; Etemad-Moghadam, S.; Alaeddini, M.; Mousavi, M.; Bahador, A. DNA-aptamer-nanographene oxide as a targeted bio-theragnostic system in antimicrobial photodynamic therapy against Porphyromonas gingivalis. Sci. Rep. 2022, 12, 1–18. [Google Scholar] [CrossRef]
- Qin, W.; Wang, C.; Jiang, C.; Sun, J.; Yu, C.; Jiao, T. Graphene oxide enables the reosteogenesis of previously contaminated titanium in vitro. J. Dent. Res. 2020, 99, 922–929. [Google Scholar] [CrossRef]
- Gao, Y.; Kang, K.; Luo, B.; Sun, X.; Lan, F.; He, J.; Wu, Y. Graphene oxide and mineralized collagen-functionalized dental implant abutment with effective soft tissue seal and romotely repeatable photodisinfection. Regen. Biomater. 2022, 9, rbac024. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Qiu, J.; Liu, X. Minocycline hydrochloride-loaded graphene oxide films on implant abutments for peri-implantitis treatment in beagle dogs. J. Periodontol. 2020, 91, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Agarwalla, S.V.; Ellepola, K.; Silikas, N.; Neto, A.C.; Seneviratne, C.J.; Rosa, V. Persistent inhibition of Candida albicans biofilm and hyphae growth on titanium by graphene nanocoating. Dent. Mater. 2021, 37, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.; Shi, H.; Qi, Y.; Li, J.; Jing, A.; Liu, Q.; Feng, W.; Li, G.; Gao, S. Specific anti-biofilm activity of carbon quantum dots by destroying P. gingivalis biofilm related genes. Int. J. Nanomed. 2020, 15, 5473. [Google Scholar] [CrossRef]
- Pourhajibagher, M.; Parker, S.; Chiniforush, N.; Bahador, A. Photoexcitation triggering via semiconductor graphene quantum dots by photochemical doping with curcumin versus perio-pathogens mixed biofilms. Photodiagnosis Photodyn. Ther. 2019, 28, 125–131. [Google Scholar] [CrossRef]
- Hägi, T.T.; Laugisch, O.; Ivanovic, A.; Sculean, A. Regenerative periodontal therapy. Quintessence Int. 2014, 45, 185–192. [Google Scholar] [CrossRef]
- Karring, T. Regenerative periodontal therapy. J. Int. Acad. Periodontol. 2000, 2, 101–109. [Google Scholar]
- Kao, D.W.; Fiorellini, J.P. Regenerative periodontal therapy. Front. Oral Biol. 2012, 15, 149–159. [Google Scholar] [CrossRef]
- Kawamoto, K.; Miyaji, H.; Nishida, E.; Miyata, S.; Kato, A.; Tateyama, A.; Furihata, T.; Shitomi, K.; Iwanaga, T.; Sugaya, T. Characterization and evaluation of graphene oxide scaffold for periodontal wound healing of class II furcation defects in dog. Int. J. Nanomed. 2018, 13, 2365. [Google Scholar] [CrossRef]
- Nishida, E.; Miyaji, H.; Kato, A.; Takita, H.; Iwanaga, T.; Momose, T.; Ogawa, K.; Murakami, S.; Sugaya, T.; Kawanami, M. Graphene oxide scaffold accelerates cellular proliferative response and alveolar bone healing of tooth extraction socket. Int. J. Nanomed. 2016, 11, 2265–2277. [Google Scholar] [CrossRef]
- Jiao, D.; Zheng, A.; Liu, Y.; Zhang, X.; Wang, X.; Wu, J.; She, W.; Lv, K.; Cao, L.; Jiang, X. Bidirectional differentiation of BMSCs induced by a biomimetic procallus based on a gelatin-reduced graphene oxide reinforced hydrogel for rapid bone regeneration. Bioact. Mater. 2021, 6, 2011–2028. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Lozano, F.; García-Bernal, D.; Aznar-Cervantes, S.; Ros-Roca, M.; Algueró, M.; Atucha, N.; Lozano-García, A.; Moraleda, J.; Cenis, J. Effects of composite films of silk fibroin and graphene oxide on the proliferation, cell viability and mesenchymal phenotype of periodontal ligament stem cells. J. Mater. Sci. Mater. Med. 2014, 25, 2731–2741. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Liu, S.; Li, J.; Guo, K.; Yuan, Q.; Zhong, A.; Yang, J.; Wang, J.; Sun, J.; Wang, Z. Collagen functionalized with graphene oxide enhanced biomimetic mineralization and in situ bone defect repair. ACS Appl. Mater. Interfaces 2018, 10, 44080–44091. [Google Scholar] [CrossRef] [PubMed]
- Choe, G.; Oh, S.; Seok, J.M.; Park, S.A.; Lee, J.Y. Graphene oxide/alginate composites as novel bioinks for three-dimensional mesenchymal stem cell printing and bone regeneration applications. Nanoscale 2019, 11, 23275–23285. [Google Scholar] [CrossRef] [PubMed]
- Dinescu, S.; Ionita, M.; Ignat, S.R.; Costache, M.; Hermenean, A. Graphene Oxide Enhances Chitosan-Based 3D Scaffold Properties for Bone Tissue Engineering. Int. J. Mol. Sci. 2019, 20, 5077. [Google Scholar] [CrossRef]
- Zhou, Q.; Yang, P.; Li, X.; Liu, H.; Ge, S. Bioactivity of periodontal ligament stem cells on sodium titanate coated with graphene oxide. Sci. Rep. 2016, 6, 19343. [Google Scholar] [CrossRef]
- Park, J.; Park, S.; Kim, J.E.; Jang, K.J.; Seonwoo, H.; Chung, J.H. Enhanced Osteogenic Differentiation of Periodontal Ligament Stem Cells Using a Graphene Oxide-Coated Poly(ε-caprolactone) Scaffold. Polymers 2021, 13, 797. [Google Scholar] [CrossRef]
- Xie, H.; Cao, T.; Gomes, J.V.; Neto, A.H.C.; Rosa, V. Two and three-dimensional graphene substrates to magnify osteogenic differentiation of periodontal ligament stem cells. Carbon 2015, 93, 266–275. [Google Scholar] [CrossRef]
- Kim, J.; Kim, Y.-R.; Kim, Y.; Lim, K.T.; Seonwoo, H.; Park, S.; Cho, S.-P.; Hong, B.H.; Choung, P.-H.; Chung, T.D. Graphene-incorporated chitosan substrata for adhesion and differentiation of human mesenchymal stem cells. J. Mater. Chem. B 2013, 1, 933–938. [Google Scholar] [CrossRef]
- Jin, L.; Lee, J.H.; Jin, O.S.; Shin, Y.C.; Kim, M.J.; Hong, S.W.; Lee, M.H.; Park, J.-C.; Han, D.-W. Stimulated osteogenic differentiation of human mesenchymal stem cells by reduced graphene oxide. J. Nanosci. Nanotechnol. 2015, 15, 7966–7970. [Google Scholar] [CrossRef]
- Vera-Sánchez, M.; Aznar-Cervantes, S.; Jover, E.; Garcia-Bernal, D.; Onate-Sanchez, R.E.; Hernández-Romero, D.; Moraleda, J.M.; Collado-González, M.; Rodríguez-Lozano, F.J.; Cenis, J.L. Silk-fibroin and graphene oxide composites promote human periodontal ligament stem cell spontaneous differentiation into osteo/cementoblast-like cells. Stem Cells Dev. 2016, 25, 1742–1754. [Google Scholar] [CrossRef] [PubMed]
- Ren, N.; Li, J.; Qiu, J.; Yan, M.; Liu, H.; Ji, D.; Huang, J.; Yu, J.; Liu, H. Growth and accelerated differentiation of mesenchymal stem cells on graphene-oxide-coated titanate with dexamethasone on surface of titanium implants. Dent. Mater. 2017, 33, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Shin, Y.C.; Jin, O.S.; Kang, S.H.; Hwang, Y.-S.; Park, J.-C.; Hong, S.W.; Han, D.-W. Reduced graphene oxide-coated hydroxyapatite composites stimulate spontaneous osteogenic differentiation of human mesenchymal stem cells. Nanoscale 2015, 7, 11642–11651. [Google Scholar] [CrossRef]
- Yao, M.; Ran, Q.-C.; Long, S.-R.; Miao, L.-Y. Fluorescent labeling application of graphene oxide quantum dots in living human periodontal ligament stem cells. J. Med. Postgrad. 2020, 12, 587–591. [Google Scholar]
- Stegeman, C.; Boyd, L. Prevention strategies for oral components of systemic conditions. In Prevention in Clinical Oral Health Care; Elsevier: Amsterdam, The Netherlands, 2008; pp. 244–263. [Google Scholar]
- Arner, J.W.; Santrock, R.D. A historical review of common bone graft materials in foot and ankle surgery. Foot Ankle Spec 2014, 7, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Papaspyridakos, P.; Chen, C.J.; Singh, M.; Weber, H.P.; Gallucci, G.O. Success criteria in implant dentistry: A systematic review. J. Dent. Res. 2012, 91, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Nizami, M.Z.I.; Campéon, B.D.L.; Nishina, Y. Electrodeposition of hydroxyapatite and graphene oxide improves the bioactivity of medical grade stainless steel. Mater. Today Sustain. 2022, 19, 100193. [Google Scholar] [CrossRef]
- Park, C.; Park, S.; Lee, D.; Choi, K.S.; Lim, H.-P.; Kim, J. Graphene as an enabling strategy for dental implant and tissue regeneration. Tissue Eng. Regen. Med. 2017, 14, 481–493. [Google Scholar] [CrossRef]
- Shin, Y.C.; Song, S.-J.; Jeong, S.J.; Kim, B.; Kwon, I.K.; Hong, S.W.; Oh, J.-W.; Han, D.-W. Graphene-based nanocomposites as promising options for hard tissue regeneration. Cut.-Edge Enabling Technol. Regen. Med. 2018, 1078, 103–117. [Google Scholar]
- Gu, M.; Lv, L.; Du, F.; Niu, T.; Chen, T.; Xia, D.; Wang, S.; Zhao, X.; Liu, J.; Liu, Y. Effects of thermal treatment on the adhesion strength and osteoinductive activity of single-layer graphene sheets on titanium substrates. Sci. Rep. 2018, 8, 8141. [Google Scholar] [CrossRef]
- Fan, Z.; Wang, J.; Wang, Z.; Ran, H.; Li, Y.; Niu, L.; Gong, P.; Liu, B.; Yang, S. One-pot synthesis of graphene/hydroxyapatite nanorod composite for tissue engineering. Carbon 2014, 66, 407–416. [Google Scholar] [CrossRef]
- Xie, H.; Chua, M.; Islam, I.; Bentini, R.; Cao, T.; Viana-Gomes, J.C.; Neto, A.H.C.; Rosa, V. CVD-grown monolayer graphene induces osteogenic but not odontoblastic differentiation of dental pulp stem cells. Dent. Mater. 2017, 33, e13–e21. [Google Scholar] [CrossRef] [PubMed]
- Suo, L.; Jiang, N.; Wang, Y.; Wang, P.; Chen, J.; Pei, X.; Wang, J.; Wan, Q. The enhancement of osseointegration using a graphene oxide/chitosan/hydroxyapatite composite coating on titanium fabricated by electrophoretic deposition. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 635–645. [Google Scholar] [CrossRef]
- Jung, H.S.; Lee, T.; Kwon, I.K.; Kim, H.S.; Hahn, S.K.; Lee, C.S. Surface modification of multipass caliber-rolled Ti alloy with dexamethasone-loaded graphene for dental applications. ACS Appl. Mater. Interfaces 2015, 7, 9598–9607. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Sun, J.; Zou, D.; Song, J.; Yang, S. Graphene-Modified Titanium Surface Enhances Local Growth Factor Adsorption and Promotes Osteogenic Differentiation of Bone Marrow Stromal Cells. Front. Bioeng. Biotechnol. 2021, 8, 621788. [Google Scholar] [CrossRef]
- Li, Q.; Wang, Z. Involvement of FAK/P38 Signaling Pathways in Mediating the Enhanced Osteogenesis Induced by Nano-Graphene Oxide Modification on Titanium Implant Surface. Int. J. Nanomed. 2020, 15, 4659–4676. [Google Scholar] [CrossRef]
- Di Carlo, R.; Di Crescenzo, A.; Pilato, S.; Ventrella, A.; Piattelli, A.; Recinella, L.; Chiavaroli, A.; Giordani, S.; Baldrighi, M.; Camisasca, A. Osteoblastic differentiation on graphene oxide-functionalized titanium surfaces: An in vitro study. Nanomaterials 2020, 10, 654. [Google Scholar] [CrossRef]
- La, W.G.; Park, S.; Yoon, H.H.; Jeong, G.J.; Lee, T.J.; Bhang, S.H.; Han, J.Y.; Char, K.; Kim, B.S. Delivery of a therapeutic protein for bone regeneration from a substrate coated with graphene oxide. Small 2013, 9, 4051–4060. [Google Scholar] [CrossRef]
- La, W.-G.; Jin, M.; Park, S.; Yoon, H.-H.; Jeong, G.-J.; Bhang, S.H.; Park, H.; Char, K.; Kim, B.-S. Delivery of bone morphogenetic protein-2 and substance P using graphene oxide for bone regeneration. Int. J. Nanomed. 2014, 9, 107. [Google Scholar]
- Amiryaghoubi, N.; Pesyan, N.N.; Fathi, M.; Omidi, Y. Injectable thermosensitive hybrid hydrogel containing graphene oxide and chitosan as dental pulp stem cells scaffold for bone tissue engineering. Int. J. Biol. Macromol. 2020, 162, 1338–1357. [Google Scholar] [CrossRef]
- Norahan, M.H.; Amroon, M.; Ghahremanzadeh, R.; Rabiee, N.; Baheiraei, N. Reduced graphene oxide: Osteogenic potential for bone tissue engineering. IET Nanobiotech. 2019, 13, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.C.; Bae, J.-H.; Lee, J.H.; Raja, I.S.; Kang, M.S.; Kim, B.; Hong, S.W.; Huh, J.-B.; Han, D.-W. Enhanced osseointegration of dental implants with reduced graphene oxide coating. Biomater. Res. 2022, 26, 11. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.T.; Seonwoo, H.; Choi, K.S.; Jin, H.; Jang, K.J.; Kim, J.; Kim, J.W.; Kim, S.Y.; Choung, P.H.; Chung, J.H. Pulsed-electromagnetic-field-assisted reduced graphene oxide substrates for multidifferentiation of human mesenchymal stem cells. Adv. Healthc. Mater. 2016, 5, 2069–2079. [Google Scholar] [CrossRef] [PubMed]
| Graphene and Its Derivatives | Properties [Reference(s)] |
|---|---|
| Graphene | |
| Graphene | Inhibits cariogenic biofilm [63,66] |
| Graphene-silver nanoparticles | Inhibits cariogenic biofilm [58] |
| Graphene-zinc nanoparticles | Inhibits cariogenic biofilm [59] |
| Graphene-zinc oxide nanoparticles | Inhibits cariogenic biofilm [86] |
| Graphene-fluorine | Inhibits cariogenic biofilm [73,78] Promotes enamel and dentin mineralization [78] |
| Graphene Oxide | |
| Graphene oxide | Inhibits cariogenic bacteria [13,60,61,67,69,70] and fungi [87] Inhibits cariogenic biofilm [69,76,88,89] Promotes enamel and dentin mineralization [79,81,82,84] |
| Graphene oxide-silver nanoparticles | Inhibits cariogenic bacteria [13,57,64,65] |
| Graphene oxide-bioactive glass | Inhibits cariogenic bacteria [80] Promotes enamel and dentin mineralization [80] |
| Graphene oxide-silver-calcium fluoride | Inhibits cariogenic bacteria [13] |
| Graphene oxide-carnosine-hydroxyapatite | Inhibits cariogenic bacteria [75] |
| Graphene oxide-copper | Inhibits cariogenic biofilm [62] |
| Graphene oxide-polyethyleneimine | Promotes enamel and dentin mineralization [90] |
| Graphene oxide-poly-methyl methacrylate | Inhibits cariogenic bacteria [72,74] |
| Graphene oxide-nanoribbon | Inhibits cariogenic biofilm [71] |
| Reduced Graphene Oxide | |
| Reduced graphene oxide | Inhibits cariogenic bacteria [68] |
| Reduced graphene oxide-silver nanoparticles | Inhibits cariogenic biofilm [64] Promotes enamel and dentin mineralization [83] |
| Graphene Oxide Quantum Dots | |
| Graphene oxide quantum dots-bioactive glass | Promotes enamel and dentin mineralization [85] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nizami, M.Z.I.; Yin, I.X.; Lung, C.Y.K.; Niu, J.Y.; Mei, M.L.; Chu, C.H. In Vitro Studies of Graphene for Management of Dental Caries and Periodontal Disease: A Concise Review. Pharmaceutics 2022, 14, 1997. https://doi.org/10.3390/pharmaceutics14101997
Nizami MZI, Yin IX, Lung CYK, Niu JY, Mei ML, Chu CH. In Vitro Studies of Graphene for Management of Dental Caries and Periodontal Disease: A Concise Review. Pharmaceutics. 2022; 14(10):1997. https://doi.org/10.3390/pharmaceutics14101997
Chicago/Turabian StyleNizami, Mohammed Zahedul Islam, Iris Xiaoxue Yin, Christie Ying Kei Lung, John Yun Niu, May Lei Mei, and Chun Hung Chu. 2022. "In Vitro Studies of Graphene for Management of Dental Caries and Periodontal Disease: A Concise Review" Pharmaceutics 14, no. 10: 1997. https://doi.org/10.3390/pharmaceutics14101997
APA StyleNizami, M. Z. I., Yin, I. X., Lung, C. Y. K., Niu, J. Y., Mei, M. L., & Chu, C. H. (2022). In Vitro Studies of Graphene for Management of Dental Caries and Periodontal Disease: A Concise Review. Pharmaceutics, 14(10), 1997. https://doi.org/10.3390/pharmaceutics14101997









