Thermoresponsive Hydrogel Containing Viscum album Extract for Topic and Transdermal Use: Development, Stability and Cytotoxicity Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Harvesting and Identification
2.2. Viscum album Ethanolic Extract
2.3. Viscum album Ethanolic Dry Extract
2.3.1. Thin Layer Chromatography
2.3.2. Determination of VA_DE Total Flavonoid Content
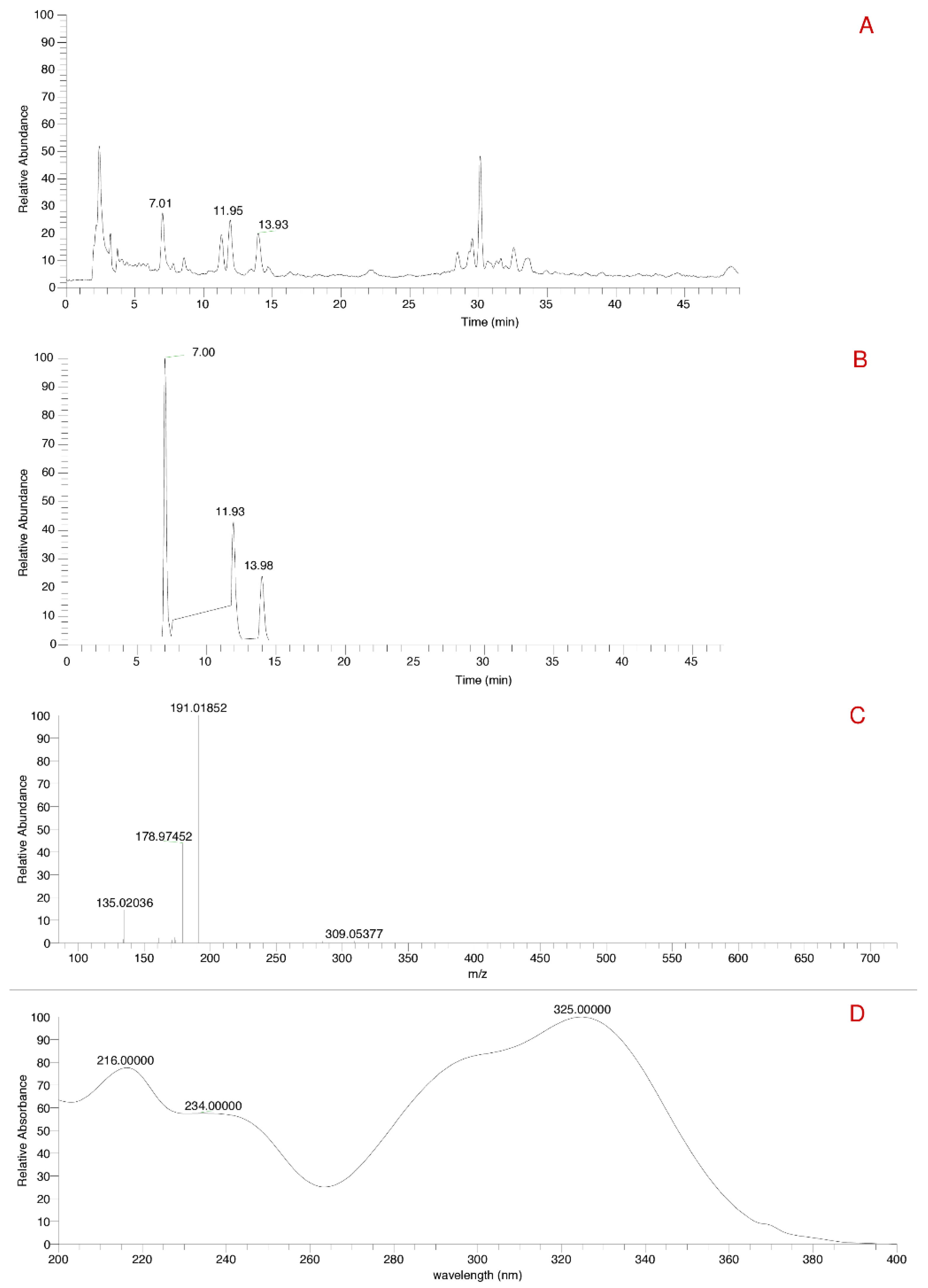
2.3.3. UHPLC-DAD-MS/MS
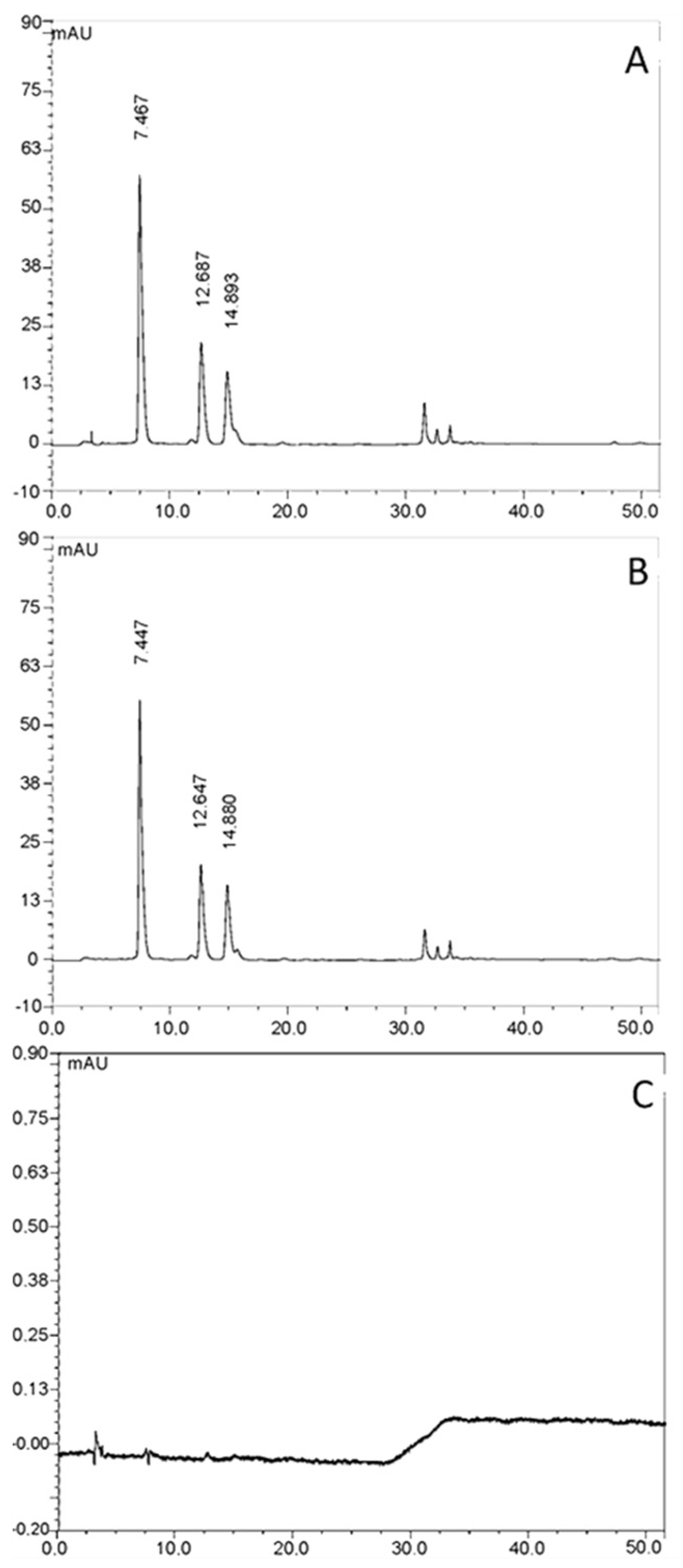
2.3.4. HPLC-UV-Vis Analysis
2.4. Viscum album Aqueous Extract
2.5. Viscum album Thermoresponsive Hydrogels
2.5.1. Rheology
2.5.2. Stability Studies
2.6. In Vitro Assays
2.6.1. Cell Lines and Culture Conditions
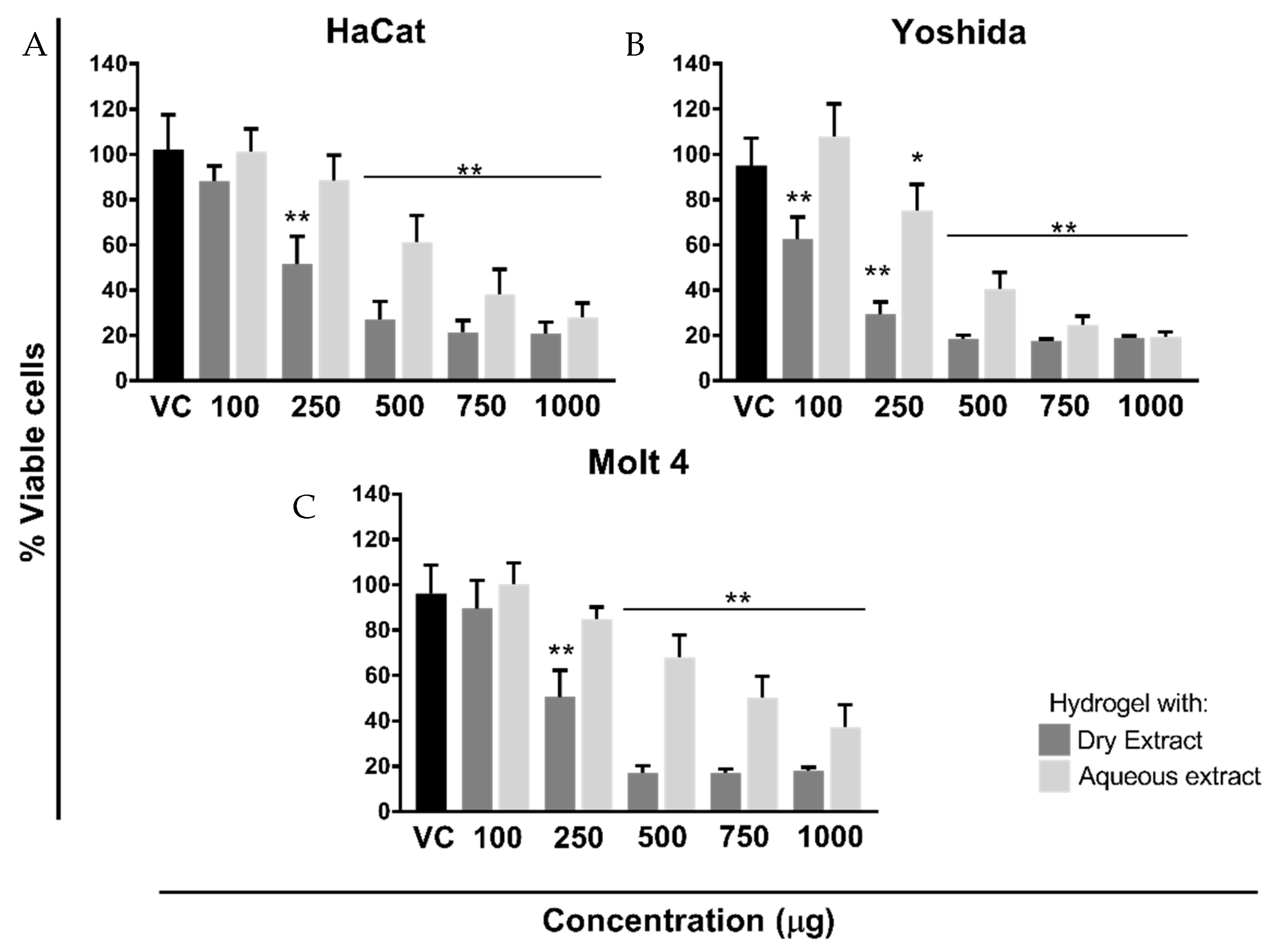
2.6.2. Cell Viability Assay
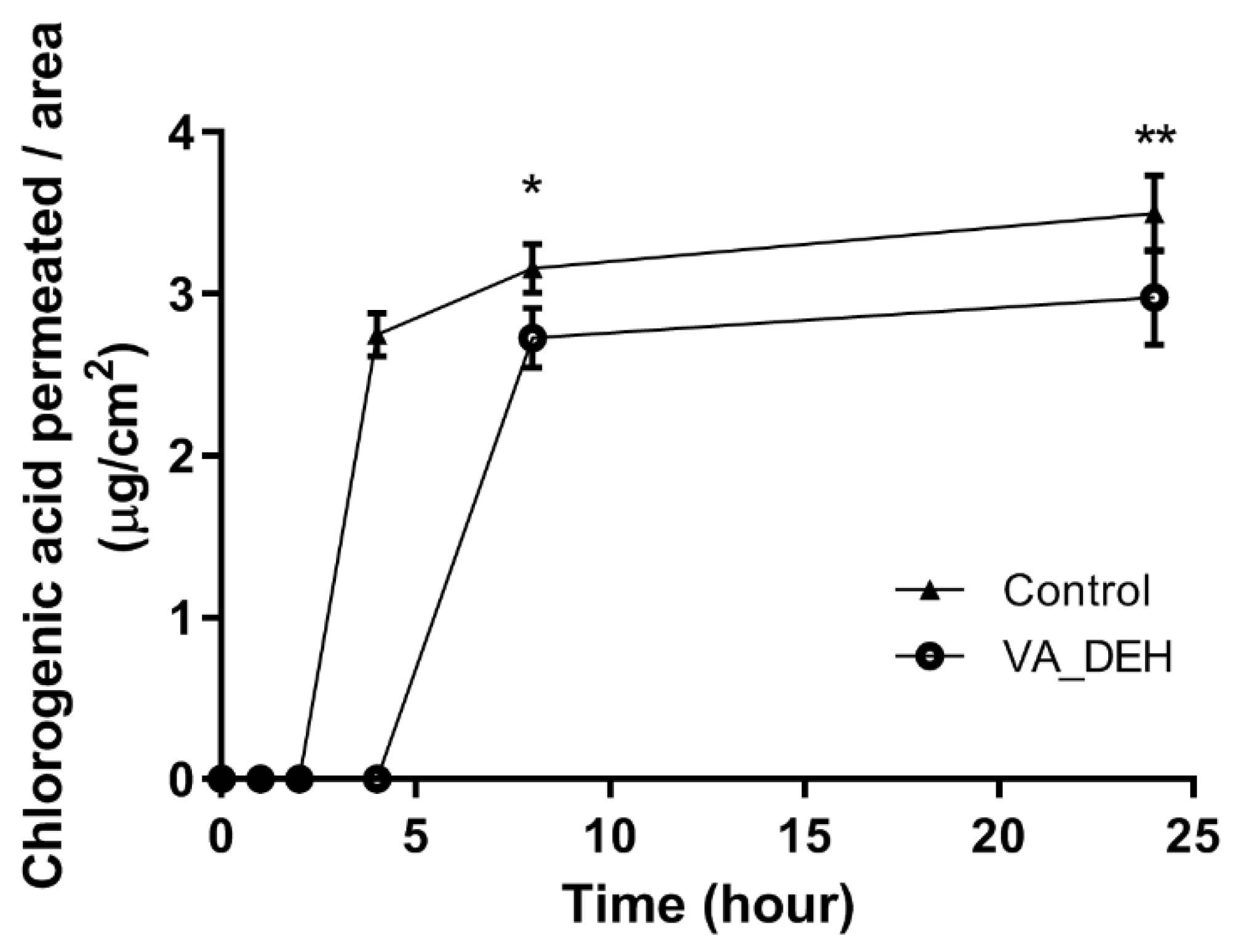
2.7. VA_DEH Ex Vivo Permeation Study
2.7.1. Skin Permeation Method
2.7.2. HPLC-DAD Assay
2.8. Statistical Analysis
3. Results and Discussion
3.1. Chemical Profile of Viscum album Dry Extract
3.1.1. Thin Layer Chromatography
3.1.2. Determination of Total Flavonoid Content
3.1.3. HPLC-DAD-MS/MS Analysis
3.1.4. HPLC-UV-Vis Analysis
3.2. Formulation Development
3.3. Rheology
3.4. Stability Studies
3.5. Cell Viability/Proliferation Assay
3.6. Permeation Studies
4. Conclusions
5. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Leite, M.L.; da Cunha, N.B.; Costa, F.F. Antimicrobial peptides, nanotechnology, and natural metabolites as novel approaches for cancer treatment. Pharmacol. Ther. 2018, 183, 160–176. [Google Scholar] [CrossRef]
- Deng, L.J.; Qi, M.; Li, N.; Lei, Y.H.; Zhang, D.M.; Chen, J.X. Natural products and their derivatives: Promising modulators of tumor immunotherapy. J. Leukoc. Biol. 2020, 108, 493–508. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [Green Version]
- Bar-Sela, G. White-Berry Mistletoe (Viscum album L.) as complementary treatment in cancer: Does it help? Eur. J. Integr. Med. 2011, 3, e55–e62. [Google Scholar] [CrossRef]
- Garcia-Garcia, J.D.; Anguiano-Cabello, J.C.; Arredondo-Valdes, R.; Candido Del Toro, C.A.; Martinez-Hernandez, J.L.; Segura-Ceniceros, E.P.; Govea-Salas, M.; Gonzalez-Chavez, M.L.; Ramos-Gonzalez, R.; Esparza-Gonzalez, S.C.; et al. Phytochemical Characterization of Phoradendron bollanum and Viscum album subs. austriacum as Mexican Mistletoe Plants with Antimicrobial Activity. Plants 2021, 10, 1299. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Wei, X.Y.; Qiu, Z.D.; Gong, L.; Chen, Z.Y.; Ma, Y.; Shen, Y.; Zhao, Y.J.; Wang, W.H.; Lai, C.J.; et al. Exploring the resources of the genus Viscum for potential therapeutic applications. J. Ethnopharmacol. 2021, 277, 114233. [Google Scholar] [CrossRef] [PubMed]
- Troger, W.; Galun, D.; Reif, M.; Schumann, A.; Stankovic, N.; Milicevic, M. Viscum album [L.] extract therapy in patients with locally advanced or metastatic pancreatic cancer: A randomised clinical trial on overall survival. Eur. J. Cancer 2013, 49, 3788–3797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ostermann, T.; Appelbaum, S.; Poier, D.; Boehm, K.; Raak, C.; Bussing, A. A Systematic Review and Meta-Analysis on the Survival of Cancer Patients Treated with a Fermented Viscum album L. Extract (Iscador): An Update of Findings. Complement. Med. Res. 2020, 27, 260–271. [Google Scholar] [CrossRef]
- Urech, K.; Baumgartner, S. Chemical Constituents of Viscum album L.: Implications for the Pharmaceutical Preparation of Mistletoe. In Mistletoe: From Mythology to Evidence-Based Medicine; Zänker, K.S., Kaveri, S.V., Eds.; Karger: Basel, Switzerland, 2015; Volume 4, pp. 11–23. [Google Scholar]
- Estko, M.; Baumgartner, S.; Urech, K.; Kunz, M.; Regueiro, U.; Heusser, P.; Weissenstein, U. Tumour cell derived effects on monocyte/macrophage polarization and function and modulatory potential of Viscum album lipophilic extract in vitro. BMC Complement. Altern. Med. 2015, 15, 130. [Google Scholar] [CrossRef] [Green Version]
- Timoshenko, A.V.; Cherenkevich, S.N.; Gabius, H.J. Viscum album agglutinin-induced aggregation of blood cells and the lectin effects on neutrophil function. Biomed. Pharmacother. 1995, 49, 153–158. [Google Scholar] [CrossRef]
- Oei, S.L.; Thronicke, A.; Schad, F. Mistletoe and Immunomodulation: Insights and Implications for Anticancer Therapies. Evid.-Based Complement. Altern. Med. 2019, 2019, 5893017. [Google Scholar] [CrossRef]
- Peñaloza, E.; Holandino, C.; Scherr, C.; Araujo, P.I.P.; Borges, R.M.; Urech, K.; Baumgartner, S.; Garrett, R. Comprehensive Metabolome Analysis of Fermented Aqueous Extracts of Viscum album L. by Liquid Chromatography-High Resolution Tandem Mass Spectrometry. Molecules 2020, 25, 4006. [Google Scholar] [CrossRef]
- Jager, T.; Holandino, C.; Melo, M.N.O.; Penaloza, E.M.C.; Oliveira, A.P.; Garrett, R.; Glauser, G.; Grazi, M.; Ramm, H.; Urech, K.; et al. Metabolomics by UHPLC-Q-TOF Reveals Host Tree-Dependent Phytochemical Variation in Viscum album L. Plants 2021, 10, 1726. [Google Scholar] [CrossRef] [PubMed]
- Holandino, C.; Melo, M.N.O.; Oliveira, A.P.; Batista, J.V.C.; Capella, M.A.M.; Garrett, R.; Grazi, M.; Ramm, H.; Torre, C.D.; Schaller, G.; et al. Phytochemical analysis and in vitro anti-proliferative activity of Viscum album ethanolic extracts. BMC Complement. Med. Ther. 2020, 20, 215. [Google Scholar] [CrossRef] [PubMed]
- Laffleur, F. Evaluation of chemical modified hydrogel formulation for topical suitability. Int. J. Biol. Macromol. 2017, 105, 1310–1314. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Mooney, D.J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 2016, 1, 16071. [Google Scholar] [CrossRef] [PubMed]
- Maudens, P.; Meyer, S.; Seemayer, C.A.; Jordan, O.; Allemann, E. Self-assembled thermoresponsive nanostructures of hyaluronic acid conjugates for osteoarthritis therapy. Nanoscale 2018, 10, 1845–1854. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.Y.; Hui, P.C.L.; Wat, E.; Ng, F.S.F.; Kan, C.W.; Lau, C.B.S.; Leung, P.C. Enhanced Transdermal Permeability via Constructing the Porous Structure of Poloxamer-Based Hydrogel. Polymers 2016, 8, 406. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Wat, E.; Hui, P.C.; Chan, B.; Ng, F.S.; Kan, C.W.; Wang, X.; Hu, H.; Wong, E.C.; Lau, C.B.; et al. Dual-functional transdermal drug delivery system with controllable drug loading based on thermosensitive poloxamer hydrogel for atopic dermatitis treatment. Sci. Rep. 2016, 6, 24112. [Google Scholar] [CrossRef] [Green Version]
- Devi, D.R.; Sandhya, P.; Vedha Hari, B.N. Poloxamer: A Novel Functional Molecule for Drug Delivery and Gene Therapy. J. Pharm. Sci. Res. 2013, 5, 159–165. [Google Scholar]
- Vishnubhakthula, S.; Elupula, R.; Duran-Lara, E.F. Recent Advances in Hydrogel-Based Drug Delivery for Melanoma Cancer Therapy: A Mini Review. J. Drug Deliv. 2017, 2017, 7275985. [Google Scholar] [CrossRef] [PubMed]
- Sepantafar, M.; Maheronnaghsh, R.; Mohammadi, H.; Radmanesh, F.; Hasani-Sadrabadi, M.M.; Ebrahimi, M.; Baharvand, H. Engineered Hydrogels in Cancer Therapy and Diagnosis. Trends Biotechnol. 2017, 35, 1074–1087. [Google Scholar] [CrossRef] [PubMed]
- Bodratti, A.M.; Alexandridis, P. Formulation of Poloxamers for Drug Delivery. J. Funct. Biomater. 2018, 9, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fakhari, A.; Corcoran, M.; Schwarz, A. Thermogelling properties of purified poloxamer 407. Heliyon 2017, 3, e00390. [Google Scholar] [CrossRef]
- Patel, H.R.; Patel, R.P.; Patel, M.M. Poloxamers: A pharmaceutical excipients with therapeutic behaviors. Int. J. PharmTech Res. 2009, 1, 299–303. [Google Scholar]
- Climate-Data.org. Available online: https://en.climate-data.org/europe/switzerland/basel-city/basel-437/ (accessed on 12 November 2021).
- ANVISA. Brazilian Homeopathic Pharmacopoeia; National Health Surveillance Agency: Brasília, Brasil, 2011.
- ANSM. Mistletoe from the Apple Tree; French National Agency for Medicines and Health Products Safety: Paris, France, 2010.
- Rolim, A.; Maciel, C.P.M.; Kaneko, T.M.; Consiglieri, V.O.; Salgado-Santos, I.M.N.; Velasco, M.V.R. Validation Assay for Total Flavonoids, as Rutin Equivalents, from Trichilia catigua Adr. Juss (Meliaceae) and Ptychopetalum olacoides Bentham (Olacaceae) Commercial Extract. J. AOAC Int. 2005, 88, 1015–1019. [Google Scholar] [CrossRef] [Green Version]
- Melo, M.N.O.; Oliveira, A.P.; Wiecikowski, A.F.; Carvalho, R.S.; Castro, J.L.; de Oliveira, F.A.G.; Pereira, H.M.G.; da Veiga, V.F.; Capella, M.M.A.; Rocha, L.; et al. Phenolic compounds from Viscum album tinctures enhanced antitumor activity in melanoma murine cancer cells. Saudi Pharm. J. 2018, 26, 311–322. [Google Scholar] [CrossRef]
- Schaller, G.; Urech, K.; Giannattasio, M. Cytotoxicity of Different Viscotoxins and Extracts from the European Subspecies of Viscum album L. Phytother. Res. 1996, 10, 473–477. [Google Scholar] [CrossRef]
- ANVISA. Brazilian Pharmacopeia, 6th ed.; National Health Surveillance Agency: Brasília, Brazil, 2019.
- Pinto, I.C.; Cerqueira-Coutinho, C.S.; Santos, E.P.; Carmo, F.A.; Ricci-Junior, E. Development and characterization of repellent formulations based on nanostructured hydrogels. Drug Dev. Ind. Pharm. 2017, 43, 67–73. [Google Scholar] [CrossRef]
- Flaten, G.E.; Palac, Z.; Engesland, A.; Filipovic-Grcic, J.; Vanic, Z.; Skalko-Basnet, N. In vitro skin models as a tool in optimization of drug formulation. Eur. J. Pharm. Sci. 2015, 75, 10–24. [Google Scholar] [CrossRef] [Green Version]
- Todo, H. Transdermal Permeation of Drugs in Various Animal Species. Pharmaceutics 2017, 9, 33. [Google Scholar] [CrossRef] [Green Version]
- Santis, A.K.; de Freitas, Z.M.; Ricci-Junior, E.; de Brito-Gitirana, L.; Fonseca, L.B.; Santos, E.P. Nifedipine in semi-solid formulations for topical use in peripheral vascular disease: Preparation, characterization, and permeation assay. Drug Dev. Ind. Pharm. 2013, 39, 1098–1106. [Google Scholar] [CrossRef] [PubMed]
- Luczkiewicz, M.; Cisowski, W.; Kaiser, P.; Ochocka, R.A.P. Comparative Analysis of Phenolic Acids in Mistletoe Plants from Various Hosts. Acta Pol. Pharm.-Drug Res. 2001, 58, 373–379. [Google Scholar]
- Pietrzak, W.; Nowak, R.; Olech, M. Effect of extraction method on phenolic content and antioxidant activity of mistletoe extracts from Viscum album subsp. abietis. Chem. Pap. 2014, 68. [Google Scholar] [CrossRef]
- Pietrzak, W.; Nowak, R.; Gawlik-Dziki, U.; Lemieszek, M.K.; Rzeski, W. LC-ESI-MS/MS Identification of Biologically Active Phenolic Compounds in Mistletoe Berry Extracts from Different Host Trees. Molecules 2017, 22, 624. [Google Scholar] [CrossRef] [Green Version]
- Tahirovic, A.; Basic, N. Determination of phenolic content and antioxidant properties of methanolic extracts from Viscum album ssp. album Beck. Bull. Chem. Technol. Bosnia Herzegovina. 2017, 49, 25–30. [Google Scholar]
- Wagner, H.; Jordan, E. An immunologically active arabinogalactan from Viscum album ‘berries’. Phytochemistry 1988, 27, 2511–2517. [Google Scholar] [CrossRef]
- Kang, S.N. Ethanol Extracts from Mistletoe (Viscum album L.) Act as Natural Antioxidants and Antimicrobial Agents in Uncooked Pork Patties during Refrigerated Storage. Asian-Australas J. Anim. Sci. 2016, 29, 109–118. [Google Scholar] [CrossRef]
- Mocan, A.; Schafberg, M.; Crișan, G.; Rohn, S. Determination of lignans and phenolic components of Schisandra chinensis (Turcz.) Baill. using HPLC-ESI-ToF-MS and HPLC-online TEAC: Contribution of individual components to overall antioxidant activity and comparison with traditional antioxidant assays. J. Funct. Foods 2016, 24, 579–594. [Google Scholar] [CrossRef]
- Meinhart, A.D.; Damin, F.M.; Caldeirao, L.; da Silveira, T.F.F.; Filho, J.T.; Godoy, H.T. Chlorogenic acid isomer contents in 100 plants commercialized in Brazil. Food Res. Int. 2017, 99, 522–530. [Google Scholar] [CrossRef]
- Campa, C.; Rakotomalal, J.J.R.; de Kochko, A.; Hamon, S. Chlorogenic acids: Diversity in green beans of wild coffee species. Adv. Plant Physiol. 2008, 10, 421–437. [Google Scholar]
- Popova, O.I. Phenolic compounds of Viscum album. Chem. Nat. Compd. 1991, 27, 123. [Google Scholar] [CrossRef]
- Diez-Sales, O.; Garrigues, T.M.; Herraez, J.V.; Belda, R.; Martin-Villodre, A.; Herraez, M. In vitro percutaneous penetration of acyclovir from solvent systems and Carbopol 971-P hydrogels: Influence of propylene glycol. J. Pharm. Sci. 2005, 94, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- Paudel, K.S.; Milewski, M.; Swadley, C.L.; Brogden, N.K.; Ghosh, P.; Stinchcomb, A.L. Challenges and opportunities in dermal/transdermal delivery. Ther. Deliv. 2010, 1, 109–131. [Google Scholar] [CrossRef] [Green Version]
- Censi, R.; Martena, V.; Hoti, E.; Malaj, L.; Di Martino, P. Permeation and skin retention of quercetin from microemulsions containing Transcutol(R) P. Drug Dev. Ind. Pharm. 2012, 38, 1128–1133. [Google Scholar] [CrossRef] [PubMed]
- Qwist, P.K.; Sander, C.; Okkels, F.; Jessen, V.; Baldursdottir, S.; Rantanen, J. On-line rheological characterization of semi-solid formulations. Eur. J. Pharm. Sci. 2019, 128, 36–42. [Google Scholar] [CrossRef]
- Wagner, J.R.; Mount, E.M.; Giles, H.F. Polymer Rheology. In Extrusion; Elsevier: New York, NY, USA, 2014; pp. 233–240. [Google Scholar]
- Zignani, M.; Tabatabay, C.; Gurny, R. Topical semi-solid drug delivery: Kinetics and tolerance of ophthalmic hydrogels. Adv. Drug Deliv. Rev. 1995, 16, 51–60. [Google Scholar] [CrossRef]
- Yan, C.; Pochan, D.J. Rheological properties of peptide-based hydrogels for biomedical and other applications. Chem. Soc. Rev. 2010, 39, 3528–3540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barradas, T.N.; Senna, J.P.; Cardoso, S.A.; de Holanda, E.S.K.G.; Elias Mansur, C.R. Formulation characterization and in vitro drug release of hydrogel-thickened nanoemulsions for topical delivery of 8-methoxypsoralen. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 92, 245–253. [Google Scholar] [CrossRef]
- Douglas, J.F. Weak and Strong Gels and the Emergence of the Amorphous Solid State. Gels 2018, 4, 19. [Google Scholar] [CrossRef] [Green Version]
- Hao, Z.-Q.; Chen, Z.-J.; Chang, M.-C.; Meng, J.-L.; Liu, J.-Y.; Feng, C.-P. Rheological properties and gel characteristics of polysaccharides from fruit-bodies of Sparassis crispa. Int. J. Food Prop. 2018, 21, 2283–2295. [Google Scholar] [CrossRef] [Green Version]
- Cho, C.-W.; Shin, S.-C.; Oh, I.-J. Thermorheologic Properties of Aqueous Solutions and Gels of Poloxamer 407. Drug Dev. Ind. Pharm. 2008, 23, 1227–1232. [Google Scholar] [CrossRef]
- Dewan, M.; Sarkar, G.; Bhowmik, M.; Das, B.; Chattoapadhyay, A.K.; Rana, D.; Chattopadhyay, D. Effect of gellan gum on the thermogelation property and drug release profile of Poloxamer 407 based ophthalmic formulation. Int. J. Biol. Macromol. 2017, 102, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Yeo, W.H.; Ramasamy, T.; Kim, D.W.; Cho, H.J.; Kim, Y.I.; Cho, K.H.; Yong, C.S.; Kim, J.O.; Choi, H.G. Docetaxel-loaded thermosensitive liquid suppository: Optimization of rheological properties. Arch. Pharm. Res. 2013, 36, 1480–1486. [Google Scholar] [CrossRef]
- Gioffredi, E.; Boffito, M.; Calzone, S.; Giannitelli, S.M.; Rainer, A.; Trombetta, M.; Mozetic, P.; Chiono, V. Pluronic F127 Hydrogel Characterization and Biofabrication in Cellularized Constructs for Tissue Engineering Applications. Procedia CIRP 2016, 49, 125–132. [Google Scholar] [CrossRef] [Green Version]
- Duarte, I.; Silveira, J.; Hafner, M.F.S.; Toyota, R.; Pedroso, D.M.M. Sensitive skin: Review of an ascending concept. An. Bras. Dermatol. 2017, 92, 521–525. [Google Scholar] [CrossRef]
- Daudt, R.M.; Back, P.I.; Cardozo, N.S.; Marczak, L.D.; Kulkamp-Guerreiro, I.C. Pinhao starch and coat extract as new natural cosmetic ingredients: Topical formulation stability and sensory analysis. Carbohydr. Polym. 2015, 134, 573–580. [Google Scholar] [CrossRef] [Green Version]
- Ban, E.; Park, M.; Jeong, S.; Kwon, T.; Kim, E.H.; Jung, K.; Kim, A. Poloxamer-Based Thermoreversible Gel for Topical Delivery of Emodin: Influence of P407 and P188 on Solubility of Emodin and Its Application in Cellular Activity Screening. Molecules 2017, 22, 246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desai, S.D.; Blanchard, J. Evaluation of pluronic F127-based sustained-release ocular delivery systems for pilocarpine using the albino rabbit eye model. J. Pharm. Sci. 1998, 87, 1190–1195. [Google Scholar] [CrossRef]
- Dao, H.; Lakhani, P.; Police, A.; Kallakunta, V.; Ajjarapu, S.S.; Wu, K.W.; Ponkshe, P.; Repka, M.A.; Narasimha Murthy, S. Microbial Stability of Pharmaceutical and Cosmetic Products. AAPS PharmSciTech 2018, 19, 60–78. [Google Scholar] [CrossRef]
- De Spiegeleer, B.; Wattyn, E.; Slegers, G.; Van der Meeren, P.; Vlaminck, K.; Van Vooren, L. The importance of the cosolvent propylene glycol on the antimicrobial preservative efficacy of a pharmaceutical formulation by DOE-ruggedness testing. Pharm. Dev. Technol. 2006, 11, 275–284. [Google Scholar] [CrossRef]
- Crinnion, W.J. Toxic Effects of the Easily Avoidable Phthalates and Parabens. Altern. Med. Rev. 2010, 15, 190–196. [Google Scholar]
- Kizhedath, A.; Wilkinson, S.; Glassey, J. Assessment of hepatotoxicity and dermal toxicity of butyl paraben and methyl paraben using HepG2 and HDFn in vitro models. Toxicology In Vitro 2019, 55, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Soni, M.G.; Carabin, I.G.; Burdock, G.A. Safety assessment of esters of p-hydroxybenzoic acid (parabens). Food Chem. Toxicol. 2005, 43, 985–1015. [Google Scholar] [CrossRef] [PubMed]
- Al Jitan, S.; Alkhoori, S.A.; Yousef, L.F. Phenolic Acids from Plants: Extraction and Application to Human Health. Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2018; pp. 389–417. [Google Scholar]
- El Gharras, H. Polyphenols: Food sources, properties and applications—A review. Int. J. Food Sci. Technol. 2009, 44, 2512–2518. [Google Scholar] [CrossRef]
- Hussain, M.A.; Khan, M.Q.; Hussain, N. Antimicrobial Screening of Viscum album L. Extracts. Int. Chem. Biol. Environ. Eng. 2011, 6, 203–208. [Google Scholar]
- Choi, S.; Kim, S.; Han, D.; Hwang, Y.; Lee, K.; Kim, D.; Cho, K.; Park, S.; Kim, H.; Kim, S.; et al. Comparative Assessment of Biological Activities of Mistletoes for Cosmetic Applications: Viscum album Var. Coloratum (Kom.) Ohwi and Loranthus Tanakae Franch. & Sav. J. Cosmet. Sci. 2019, 70, 235–245. [Google Scholar] [PubMed]
- Michaels, A.S.; Chandrasekaran, S.K.; Shaw, J.E. Drug permeation through human skin: Theory andinvitro experimental measurement. AIChE J. 1975, 21, 985–996. [Google Scholar] [CrossRef]
- Shin, S.-C.; Cho, C.-W.; Oh, I.-J. Effects of non-ionic surfactants as permeation enhancers towards piroxicam from the poloxamer gel through rat skins. Int. J. Pharm. 2001, 222, 199–203. [Google Scholar] [CrossRef]
- Escobar-Chavez, J.J.; Quintanar-Guerrero, D.; Ganem-Quintanar, A. In vivo skin permeation of sodium naproxen formulated in pluronic F-127 gels: Effect of Azone and Transcutol. Drug Dev. Ind. Pharm. 2005, 31, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Ganem-Quintanar, A.; Lafforgue, C.; Falson-Rieg, F.; Buri, P. Evaluation of the transepidermal permeation of diethylene glycol monoethyl ether and skin water loss. Int. J. Pharm. 1997, 147, 165–171. [Google Scholar] [CrossRef]


| Formulation | Poloxamer 407 | Propylene Glycol | Transcutol® | Viscum album | Water | |
|---|---|---|---|---|---|---|
| Aqueous Extract (AE) | Dry Extract (DE) | |||||
| VA_DEH | 20% (w/w) | 5% (w/w) | 5% (w/w) | - | 5% (w/w) a | q.s. c |
| VA_AEH | 20% (w/w) | 5% (w/w) | 5% (w/w) | 5% (w/w) b | - | q.s. c |
| Formulation | Recovery % | |
|---|---|---|
| After 10 s | After 60 s | |
| VA_AEH | 54.9% | 56.1% |
| VA_DEH | 52.3% | 55.3% |
| Formulation | Damping Factor (tan δ) a | G′ = G″ (γ b, τcr c e G′) |
|---|---|---|
| VA_AEH | 8.47 e−2 | γ = 6.79% τcr = 2.46 e2 Pa G′= 2.56 e3 Pa |
| VA_DEH | 6.23 e−2 | γ = 6.75% τcr = 2.72 e2 Pa G′= 2.85 e3 Pa |
| Storage (Days) | pH | Viscosity (Pa.s) | ||
|---|---|---|---|---|
| VA_DEH | VA_AEH | VA_DEH | VA_AEH | |
| 7 | 5.48 ± 0.03 | 4.52 ± 0.02 | 372.50 ± 7.78 | 331.00 ± 2.83 |
| 15 | 5.49 ± 0.05 | 4.51 ± 0.03 | 368.00 ± 9.89 | 322.00 ± 5.66 |
| 30 | 5.46 ± 0.04 | 4.66 ± 0.01 | 355.50 ± 0.71 | 328.50 ± 3.53 |
| 60 | 5.24 ± 0.02 | 4.32 ± 0.00 | 381.50 ± 10.61 | 309.50 ± 2.12 |
| 90 | 5.32 ± 0.03 | 4.55 ± 0.02 | 370.50 ± 2.12 | 300.00 ± 5.66 |
| 180 | 5.29 ± 0.03 | 4.44 ± 0.01 | 316.00 ± 4.24 | 307.00 ± 2.83 |
| Viscum album ssp. abietis | ||||
|---|---|---|---|---|
| Hydrogel (mg) | Dry Extract (µg) | Chlorogenic Acid (µg) * | Aqueous Extract (µg) | Viscotoxin (µg) * |
| 20 | 1000 | 18.88 | 1000 | 421.00 |
| 15 | 750 | 14.10 | 750 | 315.75 |
| 10 | 500 | 9.40 | 500 | 210.50 |
| 5 | 250 | 4.70 | 250 | 105.25 |
| 2 | 100 | 1.88 | 100 | 42.10 |
| IC50 (mg/mL) | |||
|---|---|---|---|
| Formulation | HaCat | Yoshida | Molt-4 |
| VA_DEH | 5.813 | 2.786 | 5.265 |
| VA_AEH | 12.40 | 8.856 | 15.04 |
| Time (h) | Amount of Chlorogenic Acid Permeated (µg/cm2) ± SD | |
|---|---|---|
| Control | VA_DEH | |
| 4 | 2.75 ± 0.13 | 0 |
| 8 | 3.16 ± 0.15 | 2.73 ± 0.19 |
| 24 | 3.50 ± 0.23 | 2.98 ± 0.30 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batista, J.V.D.C.; Matos, A.P.S.; Oliveria, A.P.; Ricci Júnior, E.; Freitas, Z.M.; Oliveira, C.A.; Toma, H.K.; Capella, M.A.M.; Rocha, L.M.; Weissenstein, U.; et al. Thermoresponsive Hydrogel Containing Viscum album Extract for Topic and Transdermal Use: Development, Stability and Cytotoxicity Activity. Pharmaceutics 2022, 14, 37. https://doi.org/10.3390/pharmaceutics14010037
Batista JVDC, Matos APS, Oliveria AP, Ricci Júnior E, Freitas ZM, Oliveira CA, Toma HK, Capella MAM, Rocha LM, Weissenstein U, et al. Thermoresponsive Hydrogel Containing Viscum album Extract for Topic and Transdermal Use: Development, Stability and Cytotoxicity Activity. Pharmaceutics. 2022; 14(1):37. https://doi.org/10.3390/pharmaceutics14010037
Chicago/Turabian StyleBatista, João V. D. C., Ana Paula S. Matos, Adriana P. Oliveria, Eduardo Ricci Júnior, Zaida M. Freitas, Catarina A. Oliveira, Helena K. Toma, Marcia A. M. Capella, Leandro M. Rocha, Ulrike Weissenstein, and et al. 2022. "Thermoresponsive Hydrogel Containing Viscum album Extract for Topic and Transdermal Use: Development, Stability and Cytotoxicity Activity" Pharmaceutics 14, no. 1: 37. https://doi.org/10.3390/pharmaceutics14010037
APA StyleBatista, J. V. D. C., Matos, A. P. S., Oliveria, A. P., Ricci Júnior, E., Freitas, Z. M., Oliveira, C. A., Toma, H. K., Capella, M. A. M., Rocha, L. M., Weissenstein, U., Baumgartner, S., & Holandino, C. (2022). Thermoresponsive Hydrogel Containing Viscum album Extract for Topic and Transdermal Use: Development, Stability and Cytotoxicity Activity. Pharmaceutics, 14(1), 37. https://doi.org/10.3390/pharmaceutics14010037






