Optimization of Sentinel Lymph Node Imaging Methodology Using Anionic Liposome and Hyaluronidase
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Liposome Preparation
2.3. Liposomes Uptake by Macrophages Suspended from LN
2.4. Cell Culture
2.5. Animal Model
2.6. SLN Imaging
2.7. DoE Approach
2.8. Statistical Analysis
3. Results
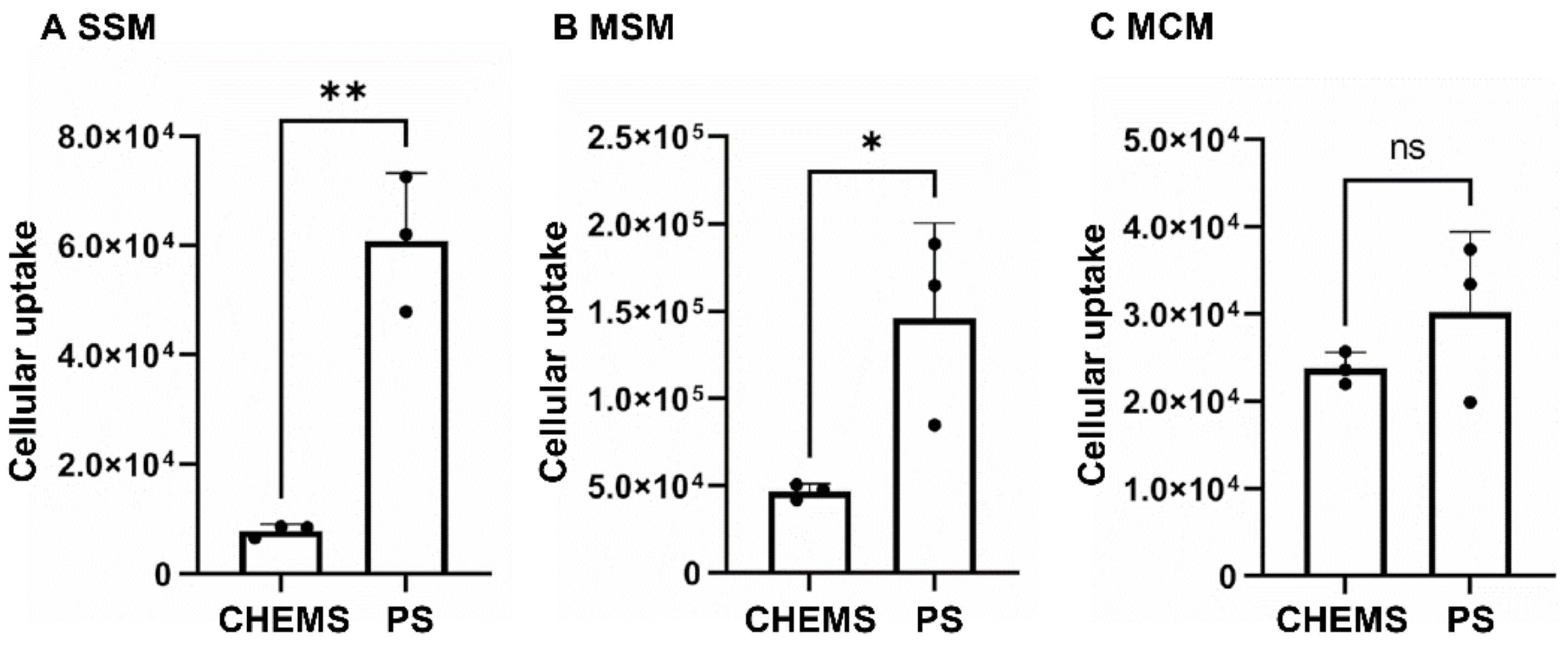
3.1. Affinity of PS-Containing Liposomes to LN-Residential Macrophages
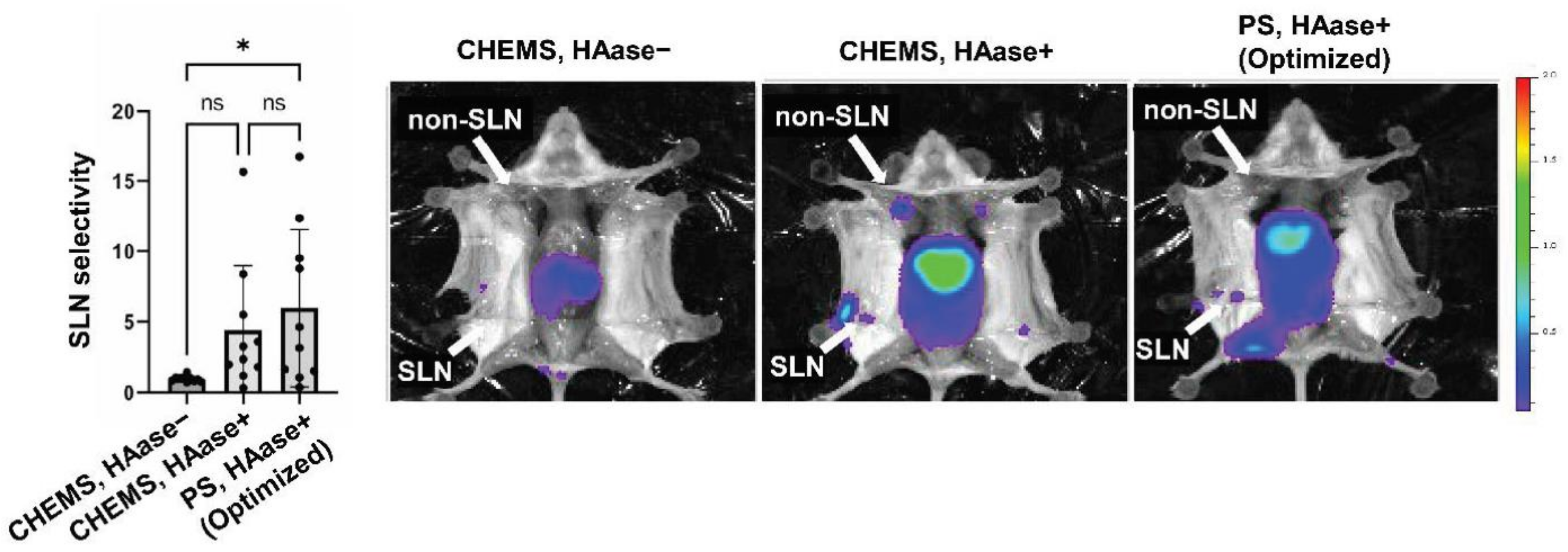
3.2. Optimization of SLN Imaging Using Anionic Liposomes
3.3. Verification Study to the Result of DoE
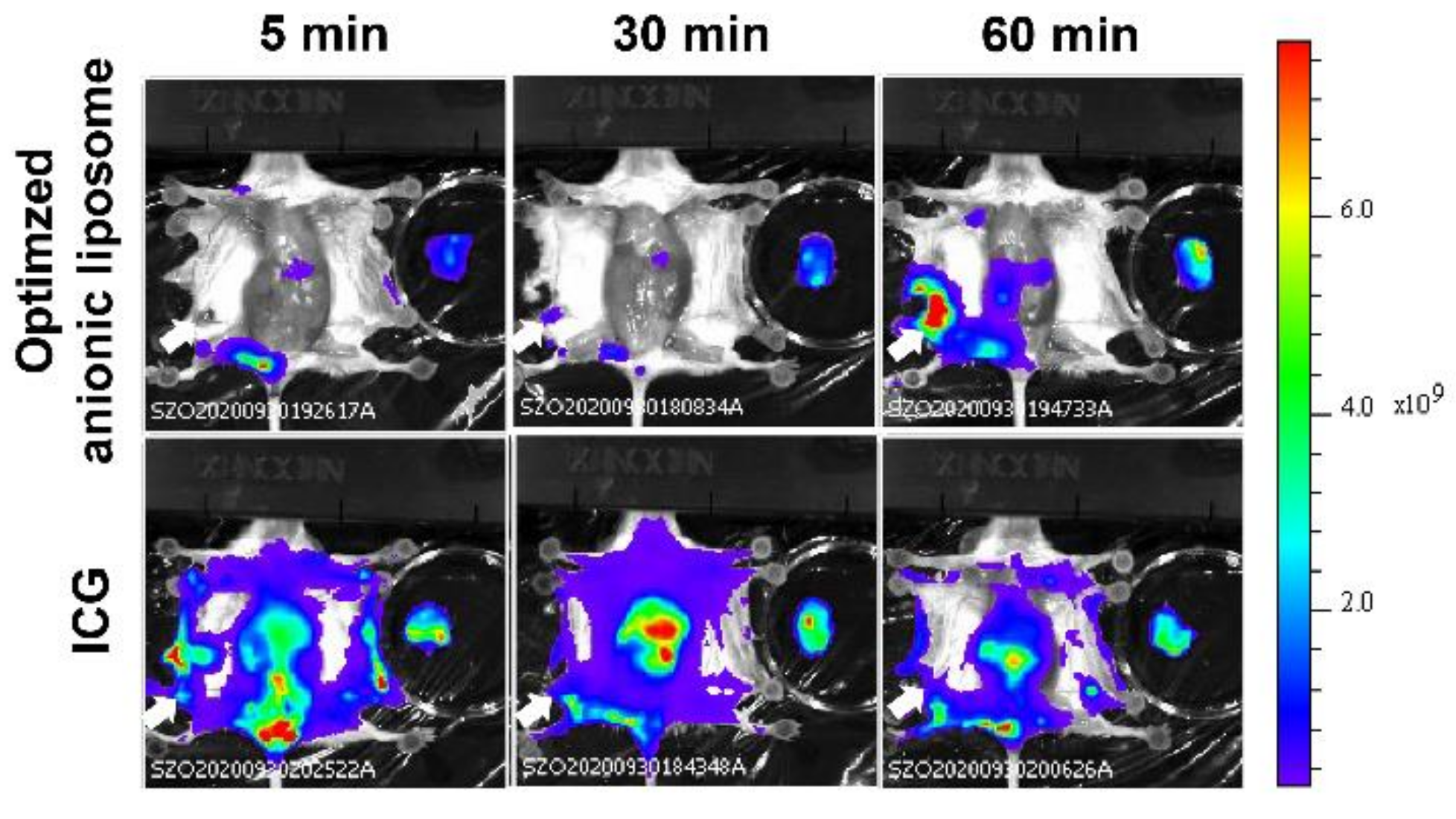
3.4. Comparative Study of Optimized Methodology with ICG
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hsueh, E.C.; Hansen, N.; Giuliano, A.E. Intraoperative lymphatic mapping and sentinel lymph node dissection in breast cancer. CA Cancer J. Clin. 2000, 50, 279–291. [Google Scholar] [CrossRef][Green Version]
- Giuliano, A.E.; Kirgan, D.M.; Guenther, J.M.; Morton, D.L. Lymphatic mapping and sentinel lymphadenectomy for breast cancer. Ann. Surg. 1994, 220, 391–398, discussion 398-401. [Google Scholar] [CrossRef]
- Blaheta, H.J.; Schittek, B.; Breuninger, H.; Garbe, C. Detection of micrometastasis in sentinel lymph nodes of patients with primary cutaneous melanoma. Recent Results Cancer Res. 2001, 158, 137–146. [Google Scholar] [CrossRef]
- Doepker, M.P.; Zager, J.S. Sentinel lymph node mapping in melanoma in the twenty-first century. Surg. Oncol Clin. N. Am. 2015, 24, 249–260. [Google Scholar] [CrossRef]
- Reintgen, M.; Kerivan, L.; Reintgen, E.; Swaninathan, S.; Reintgen, D. Breast Lymphatic Mapping and Sentinel Lymph Node Biopsy: State of the Art: 2015. Clin. Breast Cancer 2016, 16, 155–165. [Google Scholar] [CrossRef]
- Giuliano, A.E.; Jones, R.C.; Brennan, M.; Statman, R. Sentinel lymphadenectomy in breast cancer. J. Clin. Oncol. 1997, 15, 2345–2350. [Google Scholar] [CrossRef]
- Giuliano, A.E.; Dale, P.S.; Turner, R.R.; Morton, D.L.; Evans, S.W.; Krasne, D.L. Improved axillary staging of breast cancer with sentinel lymphadenectomy. Ann. Surg. 1995, 222, 394–399, discussion 399–401. [Google Scholar] [CrossRef]
- Matsuura, Y.; Ichinose, J.; Nakao, M.; Okumura, S.; Mun, M. Recent fluorescence imaging technology applications of indocyanine green in general thoracic surgery. Surg. Today 2020, 50, 1332–1342. [Google Scholar] [CrossRef] [PubMed]
- Goyal, A. New Technologies for Sentinel Lymph Node Detection. Breast Care 2018, 13, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Sugie, T.; Sawada, T.; Tagaya, N.; Kinoshita, T.; Yamagami, K.; Suwa, H.; Ikeda, T.; Yoshimura, K.; Niimi, M.; Shimizu, A.; et al. Comparison of the indocyanine green fluorescence and blue dye methods in detection of sentinel lymph nodes in early-stage breast cancer. Ann. Surg. Oncol. 2013, 20, 2213–2218. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Cai, X.; Kim, B.; Stacy, A.J.; Wang, L.V.; Lanza, G.M. Rapid synthesis of near infrared polymeric micelles for real-time sentinel lymph node imaging. Adv. Healthc. Mater. 2012, 1, 582–589. [Google Scholar] [CrossRef]
- Lee, S.B.; Yoon, G.; Lee, S.W.; Jeong, S.Y.; Ahn, B.C.; Lim, D.K.; Lee, J.; Jeon, Y.H. Combined Positron Emission Tomography and Cerenkov Luminescence Imaging of Sentinel Lymph Nodes Using PEGylated Radionuclide-Embedded Gold Nanoparticles. Small 2016, 12, 4894–4901. [Google Scholar] [CrossRef]
- Winter, A.; Engels, S.; Goos, P.; Suykers, M.C.; Gudenkauf, S.; Henke, R.P.; Wawroschek, F. Accuracy of Magnetometer-Guided Sentinel Lymphadenectomy after Intraprostatic Injection of Superparamagnetic Iron Oxide Nanoparticles in Prostate Cancer: The SentiMag Pro II Study. Cancers 2019, 12, 32. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, Y.; Akita, S.; Akita, H.; Miura, N.; Gomi, M.; Manabe, I.; Kubota, Y.; Mitsukawa, N. Development of a mouse model for the visual and quantitative assessment of lymphatic trafficking and function by in vivo imaging. Sci. Rep. 2018, 8, 5921. [Google Scholar] [CrossRef] [PubMed]
- Gomi, M.; Sakurai, Y.; Okada, T.; Miura, N.; Tanaka, H.; Akita, H. Development of Sentinel LN Imaging with a Combination of HAase Based on a Comprehensive Analysis of the Intra-lymphatic Kinetics of LPs. Mol. Ther. 2021, 29, 225–235. [Google Scholar] [CrossRef]
- Terada, T.; Kulkarni, J.A.; Huynh, A.; Chen, S.; van der Meel, R.; Tam, Y.Y.C.; Cullis, P.R. Characterization of Lipid Nanoparticles Containing Ionizable Cationic Lipids Using Design-of-Experiments Approach. Langmuir 2021, 37, 1120–1128. [Google Scholar] [CrossRef]
- Liu, X.; Bahloul, B.; Lai Kuen, R.; Andrieux, K.; Roques, C.; Scherman, D. Cationic lipid nanoparticle production by microfluidization for siRNA delivery. Int. J. Pharm. 2021, 605, 120772. [Google Scholar] [CrossRef]
- Deshkar, S.S.; Bhalerao, S.G.; Jadhav, M.S.; Shirolkar, S.V. Formulation and Optimization of Topical Solid Lipid Nanoparticles based Gel of Dapsone Using Design of Experiment. Pharm. Nanotechnol. 2018, 6, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Chao, Y.; Xiang, J.; Han, X.; Song, G.; Feng, L.; Liu, J.; Yang, G.; Chen, Q.; Liu, Z. Hyaluronidase To Enhance Nanoparticle-Based Photodynamic Tumor Therapy. Nano Lett. 2016, 16, 2512–2521. [Google Scholar] [CrossRef]
- Eikenes, L.; Tari, M.; Tufto, I.; Bruland, O.S.; de Lange Davies, C. Hyaluronidase induces a transcapillary pressure gradient and improves the distribution and uptake of liposomal doxorubicin (Caelyx) in human osteosarcoma xenografts. Br. J. Cancer 2005, 93, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Kacker, R.N.; Lagergren, E.S.; Filliben, J.J. Taguchi’s Orthogonal Arrays Are Classical Designs of Experiments. J. Res. Natl. Inst. Stand. Technol 1991, 96, 577–591. [Google Scholar] [CrossRef]
- Miyanishi, M.; Tada, K.; Koike, M.; Uchiyama, Y.; Kitamura, T.; Nagata, S. Identification of Tim4 as a phosphatidylserine receptor. Nature 2007, 450, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Tietjen, G.T.; Gong, Z.; Chen, C.H.; Vargas, E.; Crooks, J.E.; Cao, K.D.; Heffern, C.T.; Henderson, J.M.; Meron, M.; Lin, B.; et al. Molecular mechanism for differential recognition of membrane phosphatidylserine by the immune regulatory receptor Tim4. Proc. Natl. Acad. Sci. USA 2014, 111, E1463-1472. [Google Scholar] [CrossRef]
- Phan, T.G.; Green, J.A.; Gray, E.E.; Xu, Y.; Cyster, J.G. Immune complex relay by subcapsular sinus macrophages and noncognate B cells drives antibody affinity maturation. Nat. Immunol. 2009, 10, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Manzanet, R.; Sanjuan, M.A.; Wu, H.Y.; Quintana, F.J.; Xiao, S.; Anderson, A.C.; Weiner, H.L.; Green, D.R.; Kuchroo, V.K. T and B cell hyperactivity and autoimmunity associated with niche-specific defects in apoptotic body clearance in TIM-4-deficient mice. Proc. Natl. Acad. Sci. USA 2010, 107, 8706–8711. [Google Scholar] [CrossRef]
- Matsumoto, A.; Takahashi, Y.; Nishikawa, M.; Sano, K.; Morishita, M.; Charoenviriyakul, C.; Saji, H.; Takakura, Y. Role of Phosphatidylserine-Derived Negative Surface Charges in the Recognition and Uptake of Intravenously Injected B16BL6-Derived Exosomes by Macrophages. J. Pharm. Sci. 2017, 106, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Pucci, F.; Garris, C.; Lai, C.P.; Newton, A.; Pfirschke, C.; Engblom, C.; Alvarez, D.; Sprachman, M.; Evavold, C.; Magnuson, A.; et al. SCS macrophages suppress melanoma by restricting tumor-derived vesicle-B cell interactions. Science 2016, 352, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Bordet, E.; Fretaud, M.; Crisci, E.; Bouguyon, E.; Rault, S.; Pezant, J.; Pleau, A.; Renson, P.; Giuffra, E.; Larcher, T.; et al. Macrophage-B Cell Interactions in the Inverted Porcine Lymph Node and Their Response to Porcine Reproductive and Respiratory Syndrome Virus. Front. Immunol. 2019, 10, 953. [Google Scholar] [CrossRef]
- Zeng, F.; Chen, Z.; Chen, R.; Shufesky, W.J.; Bandyopadhyay, M.; Camirand, G.; Oberbarnscheidt, M.H.; Sullivan, M.L.G.; Baty, C.J.; Yang, M.Q.; et al. Graft-derived extracellular vesicles transported across subcapsular sinus macrophages elicit B cell alloimmunity after transplantation. Sci. Transl. Med. 2021, 13, 585, eabb0122. [Google Scholar] [CrossRef]
- Doherty, G.J.; Tempero, M.; Corrie, P.G. HALO-109-301: A Phase III trial of PEGPH20 (with gemcitabine and nab-paclitaxel) in hyaluronic acid-high stage IV pancreatic cancer. Future Oncol. 2018, 14, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Trevaskis, N.L.; Kaminskas, L.M.; Porter, C.J. From sewer to saviour—targeting the lymphatic system to promote drug exposure and activity. Nat. Rev. Drug Discov. 2015, 14, 781–803. [Google Scholar] [CrossRef] [PubMed]
- Palmeri, D.; van Zante, A.; Huang, C.C.; Hemmerich, S.; Rosen, S.D. Vascular endothelial junction-associated molecule, a novel member of the immunoglobulin superfamily, is localized to intercellular boundaries of endothelial cells. J. Biol. Chem. 2000, 275, 19139–19145. [Google Scholar] [CrossRef] [PubMed]
- Jalkanen, S.; Salmi, M. Lymphatic endothelial cells of the lymph node. Nat. Rev. Immunol. 2020, 20, 566–578. [Google Scholar] [CrossRef] [PubMed]


| Run | Phospholipid X1 | Cholesterol X2 | PEG Molar Ratio X3 | Diameter of Filter X4 | HAase X5 |
|---|---|---|---|---|---|
| 1 | PC | Chol | 1% | 50 | + |
| 2 | PC | Chol | 1% | 100 | − |
| 3 | PC | Chol | 5% | 50 | − |
| 4 | PC | Chol | 5% | 100 | + |
| 5 | PC | CHEMS | 1% | 50 | − |
| 6 | PC | CHEMS | 1% | 100 | + |
| 7 | PC | CHEMS | 5% | 50 | + |
| 8 | PC | CHEMS | 5% | 100 | − |
| 9 | PS | Chol | 1% | 50 | − |
| 10 | PS | Chol | 1% | 100 | + |
| 11 | PS | Chol | 5% | 50 | + |
| 12 | PS | Chol | 5% | 100 | − |
| 13 | PS | CHEMS | 1% | 50 | + |
| 14 | PS | CHEMS | 1% | 100 | − |
| 15 | PS | CHEMS | 5% | 50 | − |
| 16 | PS | CHEMS | 5% | 100 | + |
| Run | Z-Average (nm) | Zeta Potential (mV) | PDI | SLN Selectivity |
|---|---|---|---|---|
| 1 | 84 | 0.5 | 0.036 | 1.221 |
| 2 | 119 | −0.2 | 0.072 | 0.873 |
| 3 | 81 | 0.1 | 0.056 | 1.015 |
| 4 | 116 | 0.2 | 0.023 | 4.811 |
| 5 | 83 | −28.9 | 0.082 | 1.298 |
| 6 | 134 | −29.7 | 0.041 | 0.977 |
| 7 | 76 | −12.9 | 0.074 | 1.534 |
| 8 | 138 | −11.2 | 0.074 | 0.858 |
| 9 | 81 | −31.2 | 0.077 | 1.183 |
| 10 | 108 | −31.5 | 0.085 | 4.730 |
| 11 | 84 | −13.7 | 0.075 | 6.707 |
| 12 | 115 | −14.8 | 0.060 | 0.964 |
| 13 | 79 | −31.8 | 0.071 | 4.108 |
| 14 | 101 | −32.6 | 0.107 | 1.444 |
| 15 | 76 | −14.7 | 0.081 | 2.536 |
| 16 | 118 | −12.9 | 0.059 | 3.502 |
| Factor | F | p-Value | Factor | F | p-Value |
|---|---|---|---|---|---|
| X1 | 48.2 | 0.002 | X1 × 4 | 12.2 | 0.025 |
| X2 | 8.38 | 0.044 | X1 × 5 | 21.6 | 0.010 |
| X3 | 11.3 | 0.028 | X2 × 3 | 7.26 | 0.054 |
| X4 | 0.634 | 0.470 | X2 × 4 | 4.74 | 0.095 |
| X5 | 92.3 | <0.001 | X2 × 5 | 27.2 | 0.007 |
| X3 × 5 | 7.44 | 0.053 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakurai, Y.; Suzuoki, M.; Gomi, M.; Tanaka, H.; Akita, H. Optimization of Sentinel Lymph Node Imaging Methodology Using Anionic Liposome and Hyaluronidase. Pharmaceutics 2021, 13, 1462. https://doi.org/10.3390/pharmaceutics13091462
Sakurai Y, Suzuoki M, Gomi M, Tanaka H, Akita H. Optimization of Sentinel Lymph Node Imaging Methodology Using Anionic Liposome and Hyaluronidase. Pharmaceutics. 2021; 13(9):1462. https://doi.org/10.3390/pharmaceutics13091462
Chicago/Turabian StyleSakurai, Yu, Miho Suzuoki, Masaki Gomi, Hiroki Tanaka, and Hidetaka Akita. 2021. "Optimization of Sentinel Lymph Node Imaging Methodology Using Anionic Liposome and Hyaluronidase" Pharmaceutics 13, no. 9: 1462. https://doi.org/10.3390/pharmaceutics13091462
APA StyleSakurai, Y., Suzuoki, M., Gomi, M., Tanaka, H., & Akita, H. (2021). Optimization of Sentinel Lymph Node Imaging Methodology Using Anionic Liposome and Hyaluronidase. Pharmaceutics, 13(9), 1462. https://doi.org/10.3390/pharmaceutics13091462






