Hydroxycinnamic Acids and Derivatives Formulations for Skin Damages and Disorders: A Review
Abstract
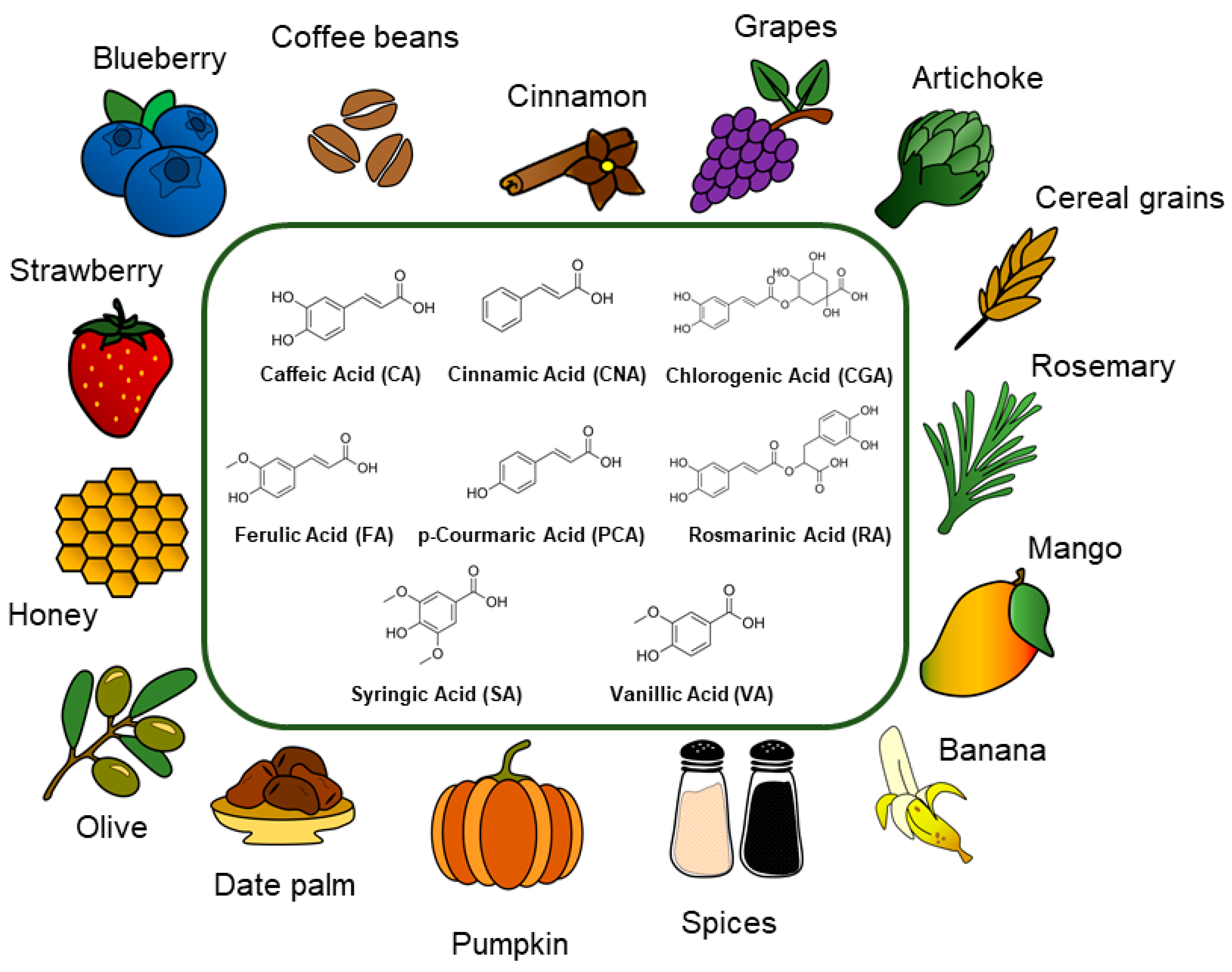
:1. Introduction
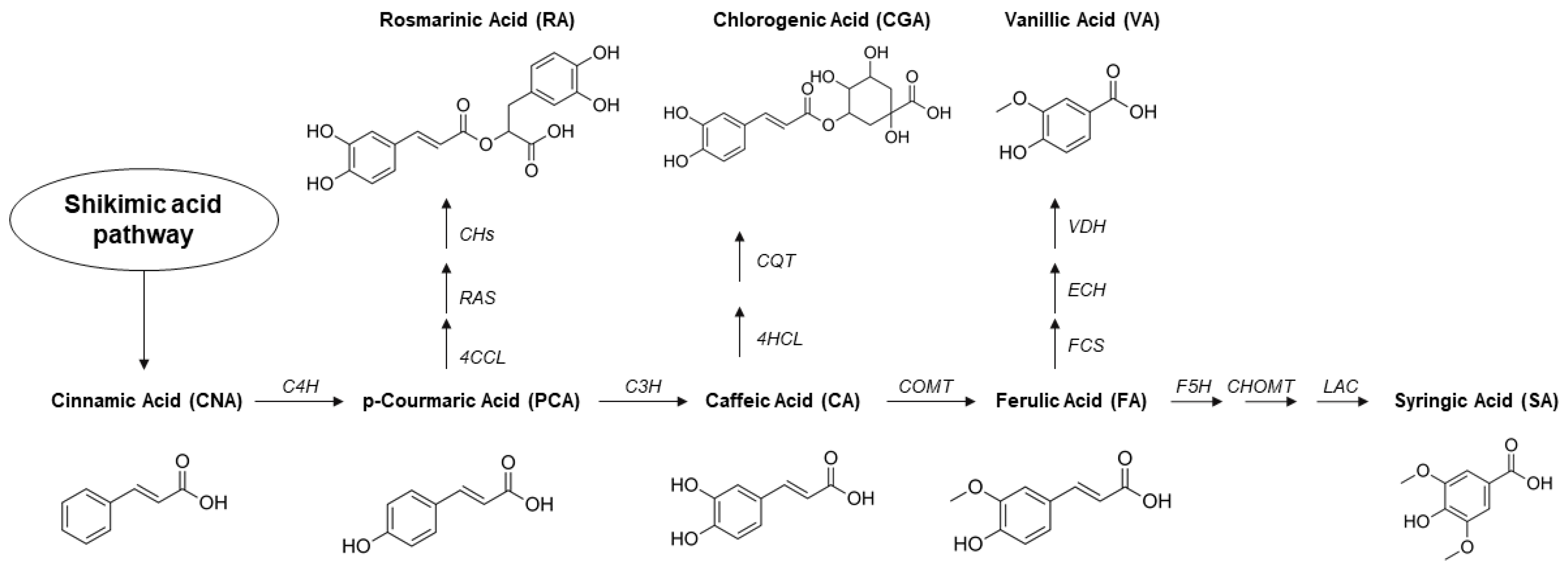
2. Hydroxycinnamic Acids and Derivatives: General Uses and Current Applications
2.1. Cinnamic Acid
2.2. Ferulic Acid
2.3. p-Coumaric Acid
2.4. Caffeic Acid
2.5. Syringic Acid
2.6. Vanillic Acid
2.7. Rosmarinic Acid
2.8. Chlorogenic Acid
3. Hydroxycinnamic Acids and Derivatives: Activities on Skin Disorders
3.1. Wound Healing
3.2. Burns and UV-Induced Damages
3.3. Dermatitis and Inflammation
3.4. Psoriasis
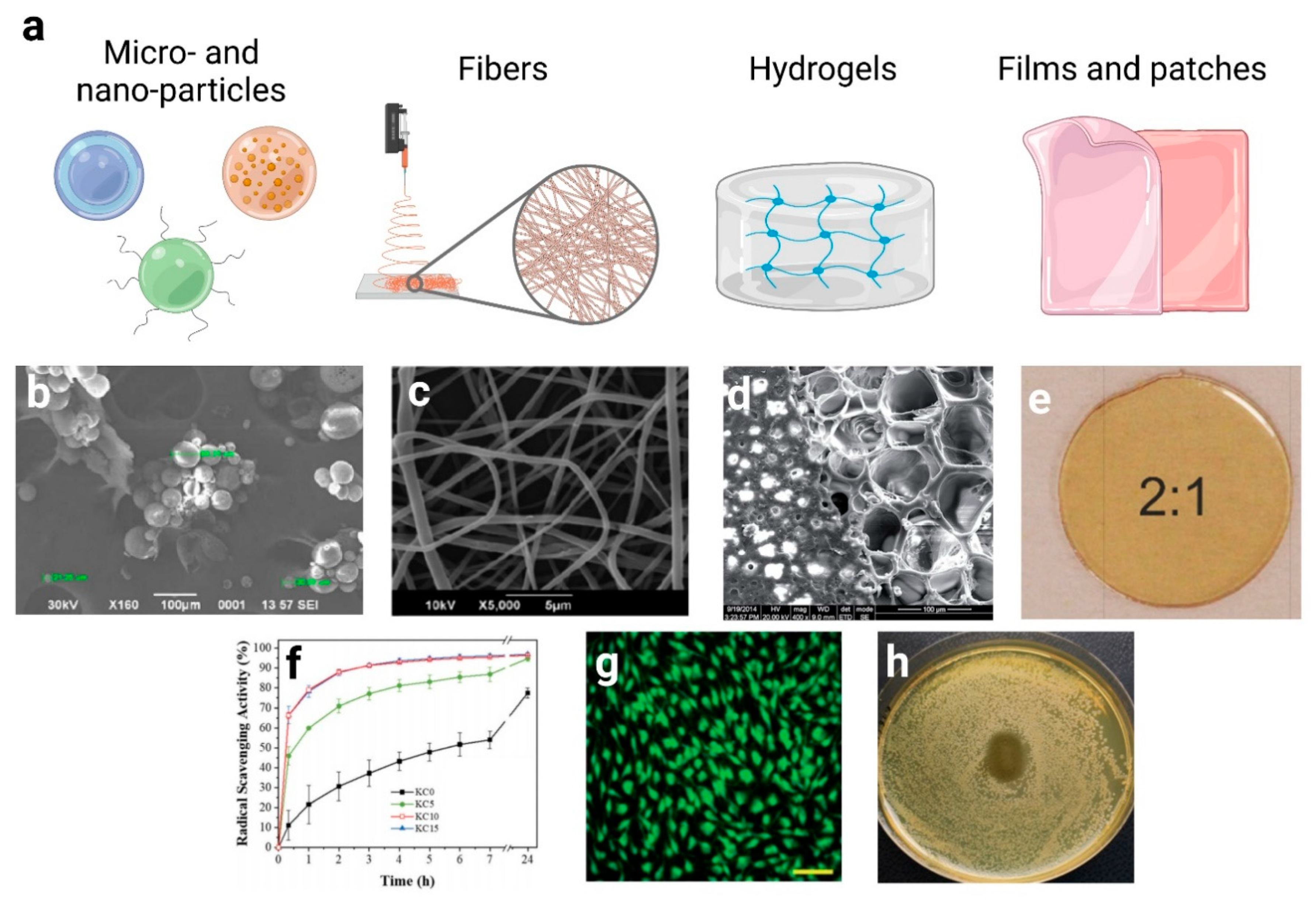
4. Hydroxycinnamic Acids and Derivatives: Advanced Formulations
4.1. Micro and Nano-Particles
4.2. Fibers
4.3. Hydrogels and Scaffolds
4.4. Films and Creams
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| AA | Arachidonic acid |
| AD | Atopic dermatitis |
| AP-1 | Activator Protein-1 |
| CA | Caffeic Acid |
| CAPE | Caffeic Acid Phenethyl Ester |
| CH | Chitosan |
| CinAc | Cinnamic Acid |
| CGA | Chlorogenic Acid |
| COX-2 | cyclooxygenase-2 |
| DMSO | Dimethyl Sulfoxide |
| DNCB | 2,4-Dinitrochlorobenzene |
| DNFB | 1-Fluoro-2,4-dinitrobenzene |
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| ECM | Extracellular Matrix |
| EGFR | Epidermal Growth Factor Receptor |
| FA | Ferulic Acid |
| GPX | Glutathione Peroxidase |
| GRAS | generally recognize As Safety |
| GSH | glutathione |
| HaCaT | Human Dermal Keratinocyte |
| HDFa | Human Dermal Fibroblast adult |
| ICAM-1 | Intracellular Adhesion Molecule-1 |
| IFN-γ | Interferon-gamma |
| IgE | Immunoglobulin E |
| IL-10 | Interleukin-10 |
| IL-12 | Interleukin-12 |
| IL-13 | Interleukin-13 |
| IL-17 | Interleukin-17 |
| IL-17A | Interleukin-17 A |
| IL-17RA | Interleukin-17 receptor A |
| IL-1β | Interleukin-1β |
| IL-2 | Interleukin-2 |
| IL-23 | Interleukin-23 |
| IL-31 | Interleukin-31 |
| IL-4 | Interleukin-4 |
| IL-5 | Interleukin-5 |
| IL-6 | Interleukin-6 |
| IL-8 | Interleukin-8 |
| IMQ | Imiquimod |
| IRAK1 | Interleukin-1 Receptor Associated Kinase 1 |
| IRAK4 | Interleukin-1 Receptor Associated Kinase 4 |
| JAK | Janus Kinase |
| LPS | Lipopolysaccharide |
| MAPK | Mitogen-activated protein kinase |
| MDCK | Madin-Darby Canine Kidney |
| MESL | oil-in-water-type Microemulsion |
| MMP-1 | Matrix Metalloproteinase-1 |
| MMP-3 | Matrix Metalloproteinase-3 |
| MMP-9 | Matrix Metalloproteinase-9 |
| MyD88 | Myeloid Differentiation Factor 88 |
| NADH | Nicotinamide adenine dinucleotide hydride |
| NFE2 | Nuclear Factor Erythroid 2 |
| NF-κB | Nuclear Factor kappa-light-chain-enhancer of activated B cells |
| NHDF-neo | normal human dermal fibroblast-neonatal |
| NLC | Nanostructured Lipid Carrier |
| NLRP3 | Nucleotide-binding oligomerization domain |
| NO | Nitric Oxide |
| NRF2 | nuclear factor erythroid 2–related factor 2 |
| PCA | p-coumaric acid |
| PCL | Poly ε-caprolactone |
| PGE2 | Prostaglandin E2 |
| pHEMA | poly(2-hydroxyethyl methacrylate) |
| p-IκB-α | nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor alpha |
| PIK3/AKT | Phosphoinositide 3-kinases; alfa serine/threonine-protein kinases |
| PLA | Poly Lactid Acid |
| PLGA | Poly Lactic Co-Glycolic Acid |
| PPARγ | Peroxisome proliferator-activated receptor γ |
| PTP- κ | Protein tyrosine phosphatases-kappa |
| PVP | Polyvinylpyrrolidone |
| RA | rosmarinic acid |
| ROS | reactive oxygen species |
| SA | syringic acid |
| SOD | Superoxide dismutase |
| STAT | signal transducers and activators of transcription |
| Th2 | T helper type 2 cells |
| Th17 | T helper type 17 cells |
| TLR-3 | Tool-Like Receptor-3 |
| TLR-4 | Tool-Like Receptor-4 |
| TNFα | Tumor Necrosis Factor α |
| TPA | 12-O-tetradecanoylphorbol-13-acetate |
| VA | vanillic acid |
| γ-PGA | Poly-γ-Glutamic Acid |
References
- Suarato, G.; Bertorelli, R.; Athanassiou, A. Borrowing from Nature: Biopolymers and biocomposites as smart wound care materials. Front. Bioeng. Biotechnol. 2018, 6, 137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raghav, A.; Khan, Z.A.; Labala, R.K.; Ahmad, J.; Noor, S.; Mishra, B.K. Financial burden of diabetic foot ulcers to world: A progressive topic to discuss always. Ther. Adv. Endocrinol. Metab. 2018, 9, 29–31. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, S.F.; Skov, L.; Dodge, R.; Hedegaard, M.S.; Kjellberg, J. Socioeconomic costs and health inequalities from psoriasis: A cohort study. Dermatology 2019, 235, 372–379. [Google Scholar] [CrossRef]
- Drucker, A.M.; Wang, A.R.; Li, W.-Q.; Sevetson, E.; Block, J.K.; Qureshi, A.A. The burden of atopic dermatitis: Summary of a report for the National Eczema Association. J. Investig. Dermatol. 2017, 137, 26–30. [Google Scholar] [CrossRef] [Green Version]
- Chandra, A.; Ray, A.; Senapati, S.; Chatterjee, R. Genetic and epigenetic basis of psoriasis pathogenesis. Mol. Immunol. 2015, 64, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Frykberg, R.G.; Banks, J. Challenges in the treatment of chronic wounds. Adv. Wound Care 2015, 4, 560–582. [Google Scholar] [CrossRef] [Green Version]
- Kaddoura, I.; Abu-Sittah, G.; Ibrahim, A.; Karamanoukian, R.; Papazian, N. Burn injury: Review of pathophysiology and therapeutic modalities in major burns. Ann. Burn. Fire Disasters 2017, 30, 95. [Google Scholar]
- Hall, T.J.; Villapún, V.M.; Addison, O.; Webber, M.A.; Lowther, M.; Louth, S.E.; Mountcastle, S.E.; Brunet, M.Y.; Cox, S.C. A call for action to the biomaterial community to tackle antimicrobial resistance. Biomater. Sci. 2020, 8, 4951–4974. [Google Scholar] [CrossRef]
- Ejtahed, H.-S.; Hasani-Ranjbar, S.; Siadat, S.D.; Larijani, B. The most important challenges ahead of microbiome pattern in the post era of the COVID-19 pandemic. J. Diabetes Metab. Disord. 2020, 19, 2031–2033. [Google Scholar] [CrossRef]
- Proksch, E. pH in nature, humans and skin. J. Dermatol. 2018, 45, 1044–1052. [Google Scholar] [CrossRef]
- Da Silva, N.T.; Quintana, H.T.; Bortolin, J.A.; Ribeiro, D.A.; de Oliveira, F. Burn injury induces skeletal muscle degeneration, inflammatory host response, and oxidative stress in wistar rats. J. Burn. Care Res. 2015, 36, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Dunnill, C.; Patton, T.; Brennan, J.; Barrett, J.; Dryden, M.; Cooke, J.; Leaper, D.; Georgopoulos, N.T. Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int. Wound J. 2017, 14, 89–96. [Google Scholar] [CrossRef]
- Todke, P.; Shah, V.H. Psoriasis: Implication to disease and therapeutic strategies, with an emphasis on drug delivery approaches. Int. J. Dermatol. 2018, 57, 1387–1402. [Google Scholar] [CrossRef]
- Hon, K.L.; Leung, A.K.; Barankin, B. Barrier repair therapy in atopic dermatitis: An overview. Am. J. Clin. Dermatol. 2013, 14, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Nobili, S.; Lippi, D.; Witort, E.; Donnini, M.; Bausi, L.; Mini, E.; Capaccioli, S. Natural compounds for cancer treatment and prevention. Pharmacol. Res. 2009, 59, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Tundis, R.; Loizzo, M.; Bonesi, M.; Menichini, F. Potential role of natural compounds against skin aging. Curr. Med. Chem. 2015, 22, 1515–1538. [Google Scholar] [CrossRef]
- Marrelli, M.; Menichini, G.; Provenzano, E.; Conforti, F. Applications of natural compounds in the photodynamic therapy of skin cancer. Curr. Med. Chem. 2014, 21, 1371–1390. [Google Scholar] [CrossRef]
- Sychrová, A.; Koláriková, I.; Žemlička, M.; Šmejkal, K. Natural compounds with dual antimicrobial and anti-inflammatory effects. Phytochem. Rev. 2020, 19, 1471–1502. [Google Scholar] [CrossRef]
- Yong, H.; Liu, Y.; Yun, D.; Zong, S.; Jin, C.; Liu, J. Chitosan films functionalized with different hydroxycinnamic acids: Preparation, characterization and application for pork preservation. Foods 2021, 10, 536. [Google Scholar] [CrossRef] [PubMed]
- Taofiq, O.; González-Paramás, A.M.; Barreiro, M.F.; Ferreira, I.C. Hydroxycinnamic acids and their derivatives: Cosmeceutical significance, challenges and future perspectives, a review. Molecules 2017, 22, 281. [Google Scholar] [CrossRef]
- Ou, S.; Kwok, K.C. Ferulic acid: Pharmaceutical functions, preparation and applications in foods. J. Sci. Food Agric. 2004, 84, 1261–1269. [Google Scholar] [CrossRef]
- Adefegha, S.A. Functional foods and nutraceuticals as dietary intervention in chronic diseases; novel perspectives for health promotion and disease prevention. J. Diet. Suppl. 2018, 15, 977–1009. [Google Scholar] [CrossRef]
- Wu, D.; Zhou, J.; Creyer, M.N.; Yim, W.; Chen, Z.; Messersmith, P.B.; Jokerst, J.V. Phenolic-enabled nanotechnology: Versatile particle engineering for biomedicine. Chem. Soc. Rev. 2021, 50, 4432–4483. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Jiang, H.; Xu, C.; Gu, L. A review: Using nanoparticles to enhance absorption and bioavailability of phenolic phytochemicals. Food Hydrocoll. 2015, 43, 153–164. [Google Scholar] [CrossRef]
- Peña-Torres, E.F.; González-Ríos, H.; Avendaño-Reyes, L.; Valenzuela-Grijalva, N.V.; Pinelli-Saavedra, A.; Muhlia-Almazán, A.; Peña-Ramos, E.A. Hydroxycinnamic acids in animal production: Pharmacokinetics, pharmacodynamics and growth promoting effects. Review. Rev. Mex. Cienc. Pecu. 2019, 10, 391–415. [Google Scholar] [CrossRef]
- El-Seedi, H.R.; El-Said, A.M.; Khalifa, S.A.; Goransson, U.; Bohlin, L.; Borg-Karlson, A.-K.; Verpoorte, R. Biosynthesis, natural sources, dietary intake, pharmacokinetic properties, and biological activities of hydroxycinnamic acids. J. Agric. Food Chem. 2012, 60, 10877–10895. [Google Scholar] [CrossRef] [PubMed]
- Coman, V.; Vodnar, D.C. Hydroxycinnamic acids and human health: Recent advances. J. Sci. Food Agric. 2020, 100, 483–499. [Google Scholar] [CrossRef]
- Adisakwattana, S. Cinnamic acid and its derivatives: Mechanisms for prevention and management of diabetes and its complications. Nutrients 2017, 9, 163. [Google Scholar] [CrossRef] [Green Version]
- Neelam; Khatkar, A.; Sharma, K.K. Phenylpropanoids and its derivatives: Biological activities and its role in food, pharmaceutical and cosmetic industries. Crit. Rev. Food Sci. Nutr. 2020, 60, 2655–2675. [Google Scholar] [CrossRef]
- Gunia-Krzyżak, A.; Słoczyńska, K.; Popiół, J.; Koczurkiewicz, P.; Marona, H.; Pękala, E. Cinnamic acid derivatives in cosmetics: Current use and future prospects. Int. J. Cosmet. Sci. 2018, 40, 356–366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, N.; Rao, A.S.; Nandal, A.; Kumar, S.; Yadav, S.S.; Ganaie, S.A.; Narasimhan, B. Phytochemical and pharmacological review of Cinnamomum verum J. Presl-a versatile spice used in food and nutrition. Food Chem. 2021, 338, 127773. [Google Scholar] [CrossRef] [PubMed]
- Forero-Doria, O.; Araya-Maturana, R.; Barrientos-Retamal, A.; Morales-Quintana, L.; Guzmán, L. N-alkylimidazolium salts functionalized with p-coumaric and cinnamic acid: A study of their antimicrobial and antibiofilm effects. Molecules 2019, 24, 3484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruwizhi, N.; Aderibigbe, B.A. Cinnamic Acid Derivatives and Their Biological Efficacy. Int. J. Mol. Sci. 2020, 21, 5712. [Google Scholar] [CrossRef]
- Pontiki, E.; Hadjipavlou-Litina, D. Multi-target cinnamic acids for oxidative stress and inflammation: Design, synthesis, biological evaluation and modeling studies. Molecules 2019, 24, 12. [Google Scholar] [CrossRef] [Green Version]
- Adisakwattana, S.; Moonsan, P.; Yibchok-Anun, S. Insulin-releasing properties of a series of cinnamic acid derivatives in vitro and in vivo. J. Agric. Food Chem. 2008, 56, 7838–7844. [Google Scholar] [CrossRef]
- Hafizur, R.M.; Hameed, A.; Shukrana, M.; Raza, S.A.; Chishti, S.; Kabir, N.; Siddiqui, R.A. Cinnamic acid exerts anti-diabetic activity by improving glucose tolerance in vivo and by stimulating insulin secretion in vitro. Phytomedicine 2015, 22, 297–300. [Google Scholar] [CrossRef]
- Zhu, R.; Liu, H.; Liu, C.; Wang, L.; Ma, R.; Chen, B.; Li, L.; Niu, J.; Fu, M.; Zhang, D. Cinnamaldehyde in diabetes: A review of pharmacology, pharmacokinetics and safety. Pharmacol. Res. 2017, 122, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.H.; Jo, Y.O.; Cho, C.-W.; Son, D.; Park, S.; Rho, J.; Choi, S.Y. Inhibitory effects of cinnamic acid on melanin biosynthesis in skin. Biol. Pharm. Bull. 2008, 31, 946–948. [Google Scholar] [CrossRef] [Green Version]
- Mathew, S.; Abraham, T.E. Ferulic acid: An antioxidant found naturally in plant cell walls and feruloyl esterases involved in its release and their applications. Crit. Rev. Biotechnol. 2004, 24, 59–83. [Google Scholar] [CrossRef]
- Faulds, C.B.; Williamson, G. The role of hydroxycinnamates in the plant cell wall. J. Sci. Food Agric. 1999, 79, 393–395. [Google Scholar] [CrossRef]
- Contardi, M.; Kossyvaki, D.; Picone, P.; Summa, M.; Guo, X.; Heredia-Guerrero, J.A.; Giacomazza, D.; Carzino, R.; Goldoni, L.; Scoponi, G. Electrospun Polyvinylpyrrolidone (PVP) hydrogels containing hydroxycinnamic acid derivatives as potential wound dressings. Chem. Eng. J. 2021, 409, 128144. [Google Scholar] [CrossRef]
- Sharma, S.; Jaiswal, A.K.; Duffy, B.; Jaiswal, S. Ferulic acid incorporated active films based on poly (lactide)/poly (butylene adipate-co-terephthalate) blend for food packaging. Food Packag. Shelf Life 2020, 24, 100491. [Google Scholar] [CrossRef]
- Ou, S.; Wang, Y.; Tang, S.; Huang, C.; Jackson, M.G. Role of ferulic acid in preparing edible films from soy protein isolate. J. Food Eng. 2005, 70, 205–210. [Google Scholar] [CrossRef]
- Patzke, H.; Schieber, A. Growth-inhibitory activity of phenolic compounds applied in an emulsifiable concentrate-ferulic acid as a natural pesticide against Botrytis cinerea. Food Res. Int. 2018, 113, 18–23. [Google Scholar] [CrossRef]
- Han, H.S.; Seol, H.; Kang, D.H.; Ahmed, M.S.; You, J.-M.; Jeon, S. Electrochemical oxidation and determination of dopamine in the presence of AA using ferulic acid functionalized electrochemically reduced graphene. Sens. Actuators B Chem. 2014, 204, 289–296. [Google Scholar] [CrossRef]
- Picone, P.; Bondi, M.L.; Picone, P.; Bondi, M.L.; Montana, G.; Bruno, A.; Pitarresi, G.; Giammona, G.; Di Carlo, M. Ferulic acid inhibits oxidative stress and cell death induced by Ab oligomers: Improved delivery by solid lipid nanoparticles. Free Radic. Res. 2009, 43, 1133–1145. [Google Scholar] [CrossRef]
- Hassanzadeh, P.; Arbabi, E.; Atyabi, F.; Dinarvand, R. Ferulic acid exhibits antiepileptogenic effect and prevents oxidative stress and cognitive impairment in the kindling model of epilepsy. Life Sci. 2017, 179, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Nankar, R.; Prabhakar, P.; Doble, M. Hybrid drug combination: Combination of ferulic acid and metformin as anti-diabetic therapy. Phytomedicine 2017, 37, 10–13. [Google Scholar] [CrossRef]
- Wang, S.; Gao, Z.; Chen, X.; Lian, X.; Zhu, H.; Zheng, J.; Sun, L. The anticoagulant ability of ferulic acid and its applications for improving the blood compatibility of silk fibroin. Biomed. Mater. 2008, 3, 044106. [Google Scholar] [CrossRef]
- Zhang, E.; Shen, F. Blood compatibility of a ferulic acid (FA)-eluting PHBHHx system for biodegradable magnesium stent application. Mater. Sci. Eng. C 2015, 52, 37–45. [Google Scholar] [CrossRef]
- Mancuso, C.; Santangelo, R. Ferulic acid: Pharmacological and toxicological aspects. Food Chem. Toxicol. 2014, 65, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; You, X.; Guan, S.; Huang, J.; Wang, L.; Zhang, J.; Wu, J. Poly (ferulic acid) with an anticancer effect as a drug nanocarrier for enhanced colon cancer therapy. Adv. Funct. Mater. 2019, 29, 1808646. [Google Scholar] [CrossRef]
- Fetoni, A.R.; Mancuso, C.; Eramo, S.L.M.; Ralli, M.; Piacentini, R.; Barone, E.; Paludetti, G.; Troiani, D. In vivo protective effect of ferulic acid against noise-induced hearing loss in the guinea-pig. Neuroscience 2010, 169, 1575–1588. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-Y.; Woung, L.-C.; Yen, J.-C.; Tseng, P.-C.; Chiou, S.-H.; Sung, Y.-J.; Liu, K.-T.; Cheng, Y.-H. Thermosensitive chitosan-based hydrogels for sustained release of ferulic acid on corneal wound healing. Carbohydr. Polym. 2016, 135, 308–315. [Google Scholar] [CrossRef]
- Grimaudo, M.A.; Concheiro, A.; Alvarez-Lorenzo, C. Crosslinked Hyaluronan Electrospun Nanofibers for Ferulic Acid Ocular Delivery. Pharmaceutics 2020, 12, 274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Contardi, M.; Montano, S.; Liguori, G.; Heredia-Guerrero, J.A.; Galli, P.; Athanassiou, A.; Bayer, I.S. treatment of coral Wounds by combining an Antiseptic Bilayer film and an injectable Antioxidant Biopolymer. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Ziyatdinova, G.; Kozlova, E.; Budnikov, H. Selective electrochemical sensor based on the electropolymerized p-coumaric acid for the direct determination of l-cysteine. Electrochim. Acta 2018, 270, 369–377. [Google Scholar] [CrossRef]
- Esakkimuthu, S.; Krishnamurthy, V.; Wang, S.; Hu, X.; Swaminathan, K.; Abomohra, A.E.-F. Application of p-coumaric acid for extraordinary lipid production in Tetradesmus obliquus: A sustainable approach towards enhanced biodiesel production. Renew. Energy 2020, 157, 368–376. [Google Scholar] [CrossRef]
- Rodriguez, A.; Kildegaard, K.R.; Li, M.; Borodina, I.; Nielsen, J. Establishment of a yeast platform strain for production of p-coumaric acid through metabolic engineering of aromatic amino acid biosynthesis. Metab. Eng. 2015, 31, 181–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boo, Y.C. p-Coumaric acid as an active ingredient in cosmetics: A review focusing on its antimelanogenic effects. Antioxidants 2019, 8, 275. [Google Scholar] [CrossRef] [Green Version]
- Song, K.; An, S.M.; Kim, M.; Koh, J.-S.; Boo, Y.C. Comparison of the antimelanogenic effects of p-coumaric acid and its methyl ester and their skin permeabilities. J. Dermatol. Sci. 2011, 63, 17–22. [Google Scholar] [CrossRef]
- Guglielmi, F.; Luceri, C.; Giovannelli, L.; Dolara, P.; Lodovici, M. Effect of 4-coumaric and 3, 4-dihydroxybenzoic acid on oxidative DNA damage in rat colonic mucosa. Br. J. Nutr. 2003, 89, 581–587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boeing, T.; Costa, P.; Venzon, L.; Meurer, M.; Mariano, L.N.B.; França, T.C.S.; Gouveia, L.; de Bassi, A.C.; Steimbach, V.; de Souza, P. Gastric healing effect of p-coumaric acid isolated from Baccharis dracunculifolia DC on animal model. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2021, 394, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.-T.; Ching, L.-C.; Yen, G.-C. Inducing gene expression of cardiac antioxidant enzymes by dietary phenolic acids in rats. J. Nutr. Biochem. 2009, 20, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Moneim, A.; Abd El-Twab, S.M.; Yousef, A.I.; Reheim, E.S.A.; Ashour, M.B. Modulation of hyperglycemia and dyslipidemia in experimental type 2 diabetes by gallic acid and p-coumaric acid: The role of adipocytokines and PPARγ. Biomed. Pharmacother. 2018, 105, 1091–1097. [Google Scholar] [CrossRef]
- Lopes, S.P.; Yepes, L.M.; Pérez-Castillo, Y.; Robledo, S.M.; de Sousa, D.P. Alkyl and Aryl Derivatives Based on p-Coumaric Acid Modification and Inhibitory Action against Leishmania braziliensis and Plasmodium falciparum. Molecules 2020, 25, 3178. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhou, J.; Wang, L.; Li, B.; Guo, J.; Guan, X.; Han, Q.; Zhang, H. Caffeic acid reduces cutaneous tumor necrosis factor alpha (TNF-α), IL-6 and IL-1β levels and ameliorates skin edema in acute and chronic model of cutaneous inflammation in mice. Biol. Pharm. Bull. 2014, 37, 347–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magnani, C.; Isaac, V.L.B.; Correa, M.A.; Salgado, H.R.N. Caffeic acid: A review of its potential use in medications and cosmetics. Analytical Methods 2014, 6, 3203–3210. [Google Scholar] [CrossRef]
- Działo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The potential of plant phenolics in prevention and therapy of skin disorders. Int. J.Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef] [Green Version]
- Choi, H.G.; Tran, P.T.; Lee, J.-H.; Min, B.S.; Kim, J.A. Anti-inflammatory activity of caffeic acid derivatives isolated from the roots of Salvia miltiorrhiza Bunge. Arch. Pharmacal Res. 2018, 41, 64–70. [Google Scholar] [CrossRef]
- Dimitris, D.; Ekaterina-Michaela, T.; Christina, K.; Ioannis, S.; Ioanna, S.K.; Aggeliki, L.; Sophia, H.; Michael, R.; Helen, S. Melissa officinalis ssp. altissima extracts: A therapeutic approach targeting psoriasis in mice. J. Ethnopharmacol. 2020, 246, 112208. [Google Scholar] [CrossRef] [PubMed]
- Sundarrajan, S.; Arumugam, M. A systems pharmacology perspective to decipher the mechanism of action of Parangichakkai chooranam, a Siddha formulation for the treatment of psoriasis. Biomed. Pharmacother. 2017, 88, 74–86. [Google Scholar] [CrossRef]
- Dudonné, S.; Poupard, P.; Coutiere, P.; Woillez, M.; Richard, T.; Mérillon, J.-M.; Vitrac, X. Phenolic composition and antioxidant properties of poplar bud (Populus nigra) extract: Individual antioxidant contribution of phenolics and transcriptional effect on skin aging. J. Agric. Food Chem. 2011, 59, 4527–4536. [Google Scholar] [CrossRef] [PubMed]
- Im, K.; Lee, J.Y.; Byeon, H.; Hwang, K.W.; Kang, W.; Whang, W.K.; Min, H. In Vitro antioxidative and anti-inflammatory activities of the ethanol extract of eggplant (Solanum melongena) stalks in macrophage RAW 264.7 cells. Food Agric. Immunol. 2016, 27, 758–771. [Google Scholar] [CrossRef] [Green Version]
- Heidari, M.; Bahramsoltani, R.; Abdolghaffari, A.H.; Rahimi, R.; Esfandyari, M.; Baeeri, M.; Hassanzadeh, G.; Abdollahi, M.; Farzaei, M.H. Efficacy of topical application of standardized extract of Tragopogon graminifolius in the healing process of experimental burn wounds. J. Tradit. Complement. Med. 2019, 9, 54–59. [Google Scholar] [CrossRef]
- Juneja, K.; Mishra, R.; Chauhan, S.; Gupta, S.; Roy, P.; Sircar, D. Metabolite profiling and wound-healing activity of Boerhavia diffusa leaf extracts using in vitro and in vivo models. J. Tradit. Complement. Med. 2020, 10, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.H.; Lee, S.H.; Cha, P.H.; Kim, M.Y.; Min, D.S.; Choi, K.Y. Polygonum aviculare L. and its active compounds, quercitrin hydrate, caffeic acid, and rutin, activate the Wnt/β-catenin pathway and induce cutaneous wound healing. Phytother. Res. 2016, 30, 848–854. [Google Scholar] [CrossRef]
- Song, Y.; Zeng, R.; Hu, L.; Maffucci, K.G.; Ren, X.; Qu, Y. In vivo wound healing and in vitro antioxidant activities of Bletilla striata phenolic extracts. Biomed. Pharmacother. 2017, 93, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, X.; Bai, R.; Zhang, N.; Kan, J.; Jin, C. Synthesis, characterization, and antioxidant activity of caffeic-acid-grafted corn starch. Starch Stärke 2018, 70, 1700141. [Google Scholar] [CrossRef]
- Alevizou, E.I.; Voutsas, E.C. Solubilities of p-coumaric and caffeic acid in ionic liquids and organic solvents. J. Chem. Thermodyn. 2013, 62, 69–78. [Google Scholar] [CrossRef]
- Rodrigues, J.; Araújo, R.; Prather, K.L.; Kluskens, L.; Rodrigues, L. Heterologous production of caffeic acid from tyrosine in Escherichia coli. Enzym. Microb. Technol. 2015, 71, 36–44. [Google Scholar] [CrossRef] [Green Version]
- Yu, S.-H.; Hsieh, H.-Y.; Pang, J.-C.; Tang, D.-W.; Shih, C.-M.; Tsai, M.-L.; Tsai, Y.-C.; Mi, F.-L. Active films from water-soluble chitosan/cellulose composites incorporating releasable caffeic acid for inhibition of lipid oxidation in fish oil emulsions. Food Hydrocoll. 2013, 32, 9–19. [Google Scholar] [CrossRef]
- Lee, P.; Ward, K.; Tschulik, K.; Chapman, G.; Compton, R. Electrochemical detection of glutathione using a poly (caffeic acid) nanocarbon composite modified electrode. Electroanalysis 2014, 26, 366–373. [Google Scholar] [CrossRef]
- Damasceno, S.S.; Santos, N.A.; Santos, I.M.; Souza, A.L.; Souza, A.G.; Queiroz, N. Caffeic and ferulic acids: An investigation of the effect of antioxidants on the stability of soybean biodiesel during storage. Fuel 2013, 107, 641–646. [Google Scholar] [CrossRef]
- Utsunomiya, H.; Ichinose, M.; Ikeda, K.; Uozaki, M.; Morishita, J.; Kuwahara, T.; Koyama, A.H.; Yamasaki, H. Inhibition by caffeic acid of the influenza A virus multiplication in vitro. Int. J. Mol. Med. 2014, 34, 1020–1024. [Google Scholar] [CrossRef] [Green Version]
- Srinivasulu, C.; Ramgopal, M.; Ramanjaneyulu, G.; Anuradha, C.; Kumar, C.S. Syringic acid (SA)‒a review of its occurrence, biosynthesis, pharmacological and industrial importance. Biomed. Pharmacother. 2018, 108, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, J.; Wu, F.; Zhou, X. Changes in rhizosphere microbial communities in potted cucumber seedlings treated with syringic acid. PLoS ONE 2018, 13, e0200007. [Google Scholar] [CrossRef] [PubMed]
- Attanasio, A.; Diano, N.; Grano, V.; Sicuranza, S.; Rossi, S.; Bencivenga, U.; Fraconte, L.; Martino, S.D.; Canciglia, P.; Mita, D.G. Nonisothermal bioreactors in the treatment of vegetation waters from olive oil: Laccase versus syringic acid as bioremediation model. Biotechnol. Prog. 2005, 21, 806–815. [Google Scholar] [CrossRef]
- Gimeno, O.; Fernandez, L.A.; Carbajo, M.; Beltran, F.; Rivas, J. Photocatalytic ozonation of phenolic wastewaters: Syringic acid, tyrosol and gallic acid. J. Environ. Sci. Health Part A 2007, 43, 61–69. [Google Scholar] [CrossRef]
- Muthukumaran, J.; Srinivasan, S.; Venkatesan, R.S.; Ramachandran, V.; Muruganathan, U. Syringic acid, a novel natural phenolic acid, normalizes hyperglycemia with special reference to glycoprotein components in experimental diabetic rats. J. Acute Dis. 2013, 2, 304–309. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Zhang, L.; Wang, X.; Wu, W.; Qin, R. Effect of Syringic acid on antioxidant biomarkers and associated inflammatory markers in mice model of asthma. Drug Dev. Res. 2019, 80, 253–261. [Google Scholar] [CrossRef]
- Srinivasan, S.; Muthukumaran, J.; Muruganathan, U.; Venkatesan, R.S.; Jalaludeen, A.M. Antihyperglycemic effect of syringic acid on attenuating the key enzymes of carbohydrate metabolism in experimental diabetic rats. Biomed. Prev. Nutr. 2014, 4, 595–602. [Google Scholar] [CrossRef]
- Karamac, M.; Kosiñska, A.; Pegg, R.B. Comparison of radical-scavenging activities for selected phenolic acids. Pol. J. Food Nutr. Sci. 2005, 14, 165–170. [Google Scholar]
- Abaza, M.-S.; Al-Attiyah, R.A.; Bhardwaj, R.; Abbadi, G.; Koyippally, M.; Afzal, M. Syringic acid from Tamarix aucheriana possesses antimitogenic and chemo-sensitizing activities in human colorectal cancer cells. Pharm. Biol. 2013, 51, 1110–1124. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Liu, R.; Niu, W. Phytochemical and antiproliferative activity of proso millet. PLoS ONE 2014, 9, e104058. [Google Scholar] [CrossRef] [Green Version]
- Cheemanapalli, S.; Anuradha, M.C.; Madhusudhana, P.; Mahesh, M.; Raghavendra, B.P.; Kumar, S.C. Exploring the binding affinity of novel syringic acid analogues and critical determinants of selectivity as potent proteasome inhibitors. Anti Cancer Agents Med. Chem. 2016, 16, 1496–1510. [Google Scholar] [CrossRef]
- Ghitescu, R.-E.; Popa, A.-M.; Popa, V.I.; Rossi, R.M.; Fortunato, G. Encapsulation of polyphenols into pHEMA e-spun fibers and determination of their antioxidant activities. Int. J. Pharm. 2015, 494, 278–287. [Google Scholar] [CrossRef]
- Vishnu, K.; Chatterjee, N.S.; Ajeeshkumar, K.; Lekshmi, R.; Tejpal, C.; Mathew, S.; Ravishankar, C. Microencapsulation of sardine oil: Application of vanillic acid grafted chitosan as a bio-functional wall material. Carbohydr. Polym. 2017, 174, 540–548. [Google Scholar] [CrossRef]
- Asha, K.; Ajeeshkumar, K.; Chatterjee, N.S.; Anandan, R.; Mathew, S. Microencapsulation of β-carotene with vanillic acid grafted chitosan improves stability and glutathione content in rats. Asclepius 2018, 1, 1–7. [Google Scholar]
- Da Silva, L.; Silva, F.; Kubota, L.; Lopes, C.; Lima, P.; Costa, E.; Júnior, W.P.; Goulart, M. Amperometric sensor based on carbon nanotubes and electropolymerized vanillic acid for simultaneous determination of ascorbic acid, dopamine, and uric acid. J. Solid State Electrochem. 2016, 20, 2389–2393. [Google Scholar] [CrossRef]
- Chou, T.H.; Ding, H.Y.; Hung, W.J.; Liang, C.H. Antioxidative characteristics and inhibition of α-melanocyte-stimulating hormone-stimulated melanogenesis of vanillin and vanillic acid from Origanum vulgare. Exp. Dermatol. 2010, 19, 742–750. [Google Scholar] [CrossRef]
- Kumar, S.; Prahalathan, P.; Raja, B. Antihypertensive and antioxidant potential of vanillic acid, a phenolic compound in L-NAME-induced hypertensive rats: A dose-dependence study. Redox Rep. 2011, 16, 208–215. [Google Scholar] [CrossRef]
- Kumar, S.; Prahalathan, P.; Raja, B. Vanillic acid: A potential inhibitor of cardiac and aortic wall remodeling in l-NAME induced hypertension through upregulation of endothelial nitric oxide synthase. Environ. Toxicol. Pharmacol. 2014, 38, 643–652. [Google Scholar] [CrossRef]
- Delaquis, P.; Stanich, K.; Toivonen, P. Effect of pH on the inhibition of Listeria spp. by vanillin and vanillic acid. J. Food Prot. 2005, 68, 1472–1476. [Google Scholar] [CrossRef] [PubMed]
- Prince, P.S.M.; Rajakumar, S.; Dhanasekar, K. Protective effects of vanillic acid on electrocardiogram, lipid peroxidation, antioxidants, proinflammatory markers and histopathology in isoproterenol induced cardiotoxic rats. Eur. J. Pharmacol. 2011, 668, 233–240. [Google Scholar] [CrossRef]
- Velli, S.K.; Sundaram, J.; Murugan, M.; Balaraman, G.; Thiruvengadam, D. Protective effect of vanillic acid against benzo (a) pyrene induced lung cancer in Swiss albino mice. J. Biochem. Mol. Toxicol. 2019, 33, e22382. [Google Scholar] [CrossRef]
- Ji, G.; Sun, R.; Hu, H.; Xu, F.; Yu, X.; Veeraraghavan, V.P.; Mohan, S.K.; Chi, X. Vannilic acid ameliorates hyperglycemia-induced oxidative stress and inflammation in streptozotocin-induced diabetic rats. J. King Saud Univ. Sci. 2020, 32, 2905–2911. [Google Scholar] [CrossRef]
- Ghasemian, M.; Owlia, S.; Owlia, M.B. Review of Anti-Inflammatory Herbal Medicines. Adv. Pharmacol. Sci. 2016, 2016, 9130979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ge, L.; Zhu, M.; Li, X.; Xu, Y.; Ma, X.; Shi, R.; Li, D.; Mu, C. Development of active rosmarinic acid-gelatin biodegradable films with antioxidant and long-term antibacterial activities. Food Hydrocoll. 2018, 83, 308–316. [Google Scholar] [CrossRef]
- Marchev, A.S.; Vasileva, L.V.; Amirova, K.M.; Savova, M.S.; Koycheva, I.K.; Balcheva-Sivenova, Z.P.; Vasileva, S.M.; Georgiev, M.I. Rosmarinic acid-From bench to valuable applications in food industry. Trends Food Sci. Technol. 2021. [Google Scholar] [CrossRef]
- Vatankhah, E.; Hamedi, S.; Ramezani, O. Surfactant-assisted incorporation of rosmarinic acid into electrosprayed poly (lactic-co-glycolic acid) microparticles with potential for cosmetic and pharmaceutical applications. Polym. Test. 2020, 81, 106180. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, T.H.; Kang, K.C.; Pyo, H.B.; Jeong, H.H. Microencapsulation of rosmarinic acid using polycaprolactone and various surfactants. Int. J. Cosmet. Sci. 2010, 32, 185–191. [Google Scholar] [CrossRef]
- Amoah, S.K.; Sandjo, L.P.; Kratz, J.M.; Biavatti, M.W. Rosmarinic Acid--Pharmaceutical and Clinical Aspects. Planta. Med. 2016, 82, 388–406. [Google Scholar] [CrossRef] [Green Version]
- Nadeem, M.; Imran, M.; Aslam Gondal, T.; Imran, A.; Shahbaz, M.; Muhammad Amir, R.; Wasim Sajid, M.; Batool Qaisrani, T.; Atif, M.; Hussain, G.; et al. Therapeutic Potential of Rosmarinic Acid: A Comprehensive Review. Appl. Sci. 2019, 9, 3139. [Google Scholar] [CrossRef] [Green Version]
- Jiang, W.-L.; Chen, X.-G.; Qu, G.-W.; Yue, X.-D.; Zhu, H.-B.; Tian, J.-W.; Fu, F.-H. Rosmarinic acid protects against experimental sepsis by inhibiting proinflammatory factor release and ameliorating hemodynamics. Shock 2009, 32, 608–613. [Google Scholar] [CrossRef]
- Scheckel, K.A.; Degner, S.C.; Romagnolo, D.F. Rosmarinic acid antagonizes activator protein-1-dependent activation of cyclooxygenase-2 expression in human cancer and nonmalignant cell lines. J. Nutr. 2008, 138, 2098–2105. [Google Scholar] [CrossRef]
- Clifford, M.N. Chlorogenic acids and other cinnamates–nature, occurrence and dietary burden. J. Sci. Food Agric. 1999, 79, 362–372. [Google Scholar] [CrossRef]
- Fazel Nabavi, S.; Tejada, S.; Setzer, N.W.; Gortzi, O.; Sureda, A.; Braidy, N.; Daglia, M.; Manayi, A.; Mohammad Nabavi, S. Chlorogenic acid and mental diseases: From chemistry to medicine. Curr. Neuropharmacol. 2017, 15, 471–479. [Google Scholar] [CrossRef] [Green Version]
- Fu, S.; Wu, C.; Wu, T.; Yu, H.; Yang, S.; Hu, Y. Preparation and characterisation of chlorogenic acid-gelatin: A type of biologically active film for coating preservation. Food Chem. 2017, 221, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, H.; Niu, N.; Chen, Z.; Li, S.; Liu, S.-X.; Li, J. Fluorescent Poly (vinyl alcohol) Films Containing Chlorogenic Acid Carbon Nanodots for Food Monitoring. ACS Appl. Nano Mater. 2020, 3, 7611–7620. [Google Scholar] [CrossRef]
- Salimi, A.; Hallaj, R.; Ghadermazi, M. Modification of carbon ceramic electrode prepared with sol–gel technique by a thin film of chlorogenic acid: Application to amperometric detection of NADH. Talanta 2005, 65, 888–894. [Google Scholar] [CrossRef]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef]
- Yun, N.; Kang, J.-W.; Lee, S.-M. Protective effects of chlorogenic acid against ischemia/reperfusion injury in rat liver: Molecular evidence of its antioxidant and anti-inflammatory properties. J. Nutr. Biochem. 2012, 23, 1249–1255. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, A.; Yamamoto, N.; Jokura, H.; Yamamoto, M.; Fujii, A.; Tokimitsu, I.; Saito, I. Chlorogenic acid attenuates hypertension and improves endothelial function in spontaneously hypertensive rats. J. Hypertens. 2006, 24, 1065–1073. [Google Scholar] [CrossRef]
- Lu, H.; Tian, Z.; Cui, Y.; Liu, Z.; Ma, X. Chlorogenic acid: A comprehensive review of the dietary sources, processing effects, bioavailability, beneficial properties, mechanisms of action, and future directions. Compr. Rev. Food Sci. Food Saf. 2020, 19, 3130–3158. [Google Scholar] [CrossRef]
- Shin, H.S.; Satsu, H.; Bae, M.-J.; Zhao, Z.; Ogiwara, H.; Totsuka, M.; Shimizu, M. Anti-inflammatory effect of chlorogenic acid on the IL-8 production in Caco-2 cells and the dextran sulphate sodium-induced colitis symptoms in C57BL/6 mice. Food Chem. 2015, 168, 167–175. [Google Scholar] [CrossRef]
- Feng, R.; Lu, Y.; Bowman, L.L.; Qian, Y.; Castranova, V.; Ding, M. Inhibition of activator protein-1, NF-κB, and MAPKs and induction of phase 2 detoxifying enzyme activity by chlorogenic acid. J. Biol. Chem. 2005, 280, 27888–27895. [Google Scholar] [CrossRef] [Green Version]
- Han, G.; Ceilley, R. Chronic wound healing: A review of current management and treatments. Adv. Ther. 2017, 34, 599–610. [Google Scholar] [CrossRef] [Green Version]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef]
- Gethin, G. Understanding the inflammatory process in wound healing. Br. J. Community Nurs. 2012, 17, S17–S22. [Google Scholar] [CrossRef]
- Blakytny, R.; Jude, E. The molecular biology of chronic wounds and delayed healing in diabetes. Diabet. Med. 2006, 23, 594–608. [Google Scholar] [CrossRef]
- Rosique, R.G.; Rosique, M.J.; Farina Junior, J.A. Curbing inflammation in skin wound healing: A review. Int. J. Inflamm. 2015, 2015, 316235. [Google Scholar] [CrossRef] [Green Version]
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin wound healing process and new emerging technologies for skin wound care and regeneration. Pharmaceutics 2020, 12, 735. [Google Scholar] [CrossRef]
- Ghaisas, M.M.; Kshirsagar, S.B.; Sahane, R.S. Evaluation of wound healing activity of ferulic acid in diabetic rats. Int. Wound J. 2014, 11, 523–532. [Google Scholar] [CrossRef]
- Ren, J.; Yang, M.; Xu, F.; Chen, J.; Ma, S. Acceleration of wound healing activity with syringic acid in streptozotocin induced diabetic rats. Life Sci. 2019, 233, 116728. [Google Scholar] [CrossRef]
- Koganov, M.M.; Dueva, O.V.; Tsorin, B.L. Activities of plant-derived phenols in a fibroblast cell culture model. J. Nat. Prod. 1999, 62, 481–483. [Google Scholar] [CrossRef]
- Song, H.S.; Park, T.W.; Sohn, U.D.; Shin, Y.K.; Choi, B.C.; Kim, C.J.; Sim, S.S. The effect of caffeic acid on wound healing in skin-incised mice. Korean J. Physiol. Pharmacol. 2008, 12, 343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexandru, V.; Gaspar, A.; Savin, S.; Toma, A.; Tatia, R.; Gille, E. Phenolic content, antioxidant activity and effect on collagen synthesis of a traditional wound healing polyherbal formula. Studia Univ. Vasile Goldis Arad. Ser. Stiintele Vietii 2015, 25, 41. [Google Scholar]
- Bagdas, D.; Etoz, B.C.; Gul, Z.; Ziyanok, S.; Inan, S.; Turacozen, O.; Gul, N.Y.; Topal, A.; Cinkilic, N.; Tas, S. In vivo systemic chlorogenic acid therapy under diabetic conditions: Wound healing effects and cytotoxicity/genotoxicity profile. Food Chem. Toxicol. 2015, 81, 54–61. [Google Scholar] [CrossRef]
- Bagdas, D.; Gul, N.Y.; Topal, A.; Tas, S.; Ozyigit, M.O.; Cinkilic, N.; Gul, Z.; Etoz, B.C.; Ziyanok, S.; Inan, S. Pharmacologic overview of systemic chlorogenic acid therapy on experimental wound healing. Naunyn Schmiedeberg’s Arch. Pharmacol. 2014, 387, 1101–1116. [Google Scholar] [CrossRef] [PubMed]
- Davis, F.M.; denDekker, A.; Joshi, A.D.; Wolf, S.J.; Audu, C.; Melvin, W.J.; Mangum, K.; Riordan, M.O.; Kunkel, S.L.; Gallagher, K.A. Palmitate-TLR4 signaling regulates the histone demethylase, JMJD3, in macrophages and impairs diabetic wound healing. Eur. J. Immunol. 2020, 50, 1929–1940. [Google Scholar] [CrossRef]
- Jere, S.W.; Abrahamse, H.; Houreld, N.N. The JAK/STAT signaling pathway and photobiomodulation in chronic wound healing. Cytokine Growth Factor Rev. 2017, 38, 73–79. [Google Scholar] [CrossRef]
- Vinaik, R.; Abdullahi, A.; Barayan, D.; Jeschke, M.G. NLRP3 inflammasome activity is required for wound healing after burns. Transl. Res. 2020, 217, 47–60. [Google Scholar] [CrossRef]
- Hiebert, P.; Werner, S. Regulation of Wound Healing by the NRF2 Transcription Factor—More Than Cytoprotection. Int. J. Mol. Sci. 2019, 20, 3856. [Google Scholar] [CrossRef] [Green Version]
- Thuraisingam, T.; Xu, Y.Z.; Eadie, K.; Heravi, M.; Guiot, M.-C.; Greemberg, R.; Gaestel, M.; Radzioch, D. MAPKAPK-2 signaling is critical for cutaneous wound healing. J. Investig. Dermatol. 2010, 130, 278–286. [Google Scholar] [CrossRef] [Green Version]
- Kępa, M.; Miklasińska-Majdanik, M.; Wojtyczka, R.D.; Idzik, D.; Korzeniowski, K.; Smoleń-Dzirba, J.; Wąsik, T.J. Antimicrobial potential of caffeic acid against Staphylococcus aureus clinical strains. BioMed. Res. Int. 2018, 2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gheibi, N.; Divan Khosroshahi, N.; Habibi, M. Effect of Caffeic Acid and Low-Power Laser Light Co-Exposure on Viability of Pseudomonas aeruginosa. J. Biotechnol. Health Sci. 2015. [Google Scholar] [CrossRef] [Green Version]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial activity and mode of action of ferulic and gallic acids against pathogenic bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef]
- Ignatova, M.; Manolova, N.; Rashkov, I.; Markova, N. Antibacterial and antioxidant electrospun materials from poly (3-hydroxybutyrate) and polyvinylpyrrolidone containing caffeic acid phenethyl ester–“in” and “on” strategies for enhanced solubility. Int. J. Pharm. 2018, 545, 342–356. [Google Scholar] [CrossRef]
- Pinho, E.; Soares, G.; Henriques, M. Evaluation of antibacterial activity of caffeic acid encapsulated by β-cyclodextrins. J. Microencapsul. 2015, 32, 804–810. [Google Scholar] [CrossRef] [Green Version]
- Lou, Z.; Wang, H.; Rao, S.; Sun, J.; Ma, C.; Li, J. p-Coumaric acid kills bacteria through dual damage mechanisms. Food Control 2012, 25, 550–554. [Google Scholar] [CrossRef]
- Monte, J.; Abreu, A.C.; Borges, A.; Simões, L.C.; Simões, M. Antimicrobial activity of selected phytochemicals against Escherichia coli and Staphylococcus aureus and their biofilms. Pathogens 2014, 3, 473–498. [Google Scholar] [CrossRef] [Green Version]
- Kabir, F.; Katayama, S.; Tanji, N.; Nakamura, S. Antimicrobial effects of chlorogenic acid and related compounds. J. Korean Soc. Appl. Biol. Chem. 2014, 57, 359–365. [Google Scholar] [CrossRef]
- Kim, J.-H.; Yu, D.; Eom, S.-H.; Kim, S.-H.; Oh, J.; Jung, W.K.; Kim, Y.-M. Synergistic antibacterial effects of chitosan-caffeic acid conjugate against antibiotic-resistant acne-related bacteria. Mar. Drugs 2017, 15, 167. [Google Scholar] [CrossRef]
- Kfoury, M.; Landy, D.; Auezova, L.; Greige-Gerges, H.; Fourmentin, S. Effect of cyclodextrin complexation on phenylpropanoids’ solubility and antioxidant activity. Beilstein J. Org. Chem. 2014, 10, 2322–2331. [Google Scholar] [CrossRef] [Green Version]
- Shiozawa, R.; Inoue, Y.; Murata, I.; Kanamoto, I. Effect of antioxidant activity of caffeic acid with cyclodextrins using ground mixture method. Asian J. Pharm. Sci. 2018, 13, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Luís, Â.; Silva, F.; Sousa, S.; Duarte, A.P.; Domingues, F. Antistaphylococcal and biofilm inhibitory activities of gallic, caffeic, and chlorogenic acids. Biofouling 2014, 30, 69–79. [Google Scholar] [CrossRef]
- Evers, L.H.; Bhavsar, D.; Mailänder, P. The biology of burn injury. Exp. Dermatol. 2010, 19, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Hseu, Y.-C.; Korivi, M.; Lin, F.-Y.; Li, M.-L.; Lin, R.-W.; Wu, J.-J.; Yang, H.-L. Trans-cinnamic acid attenuates UVA-induced photoaging through inhibition of AP-1 activation and induction of Nrf2-mediated antioxidant genes in human skin fibroblasts. J. Dermatol. Sci. 2018, 90, 123–134. [Google Scholar] [CrossRef] [Green Version]
- Fernando, P.M.D.J.; Piao, M.J.; Kang, K.A.; Ryu, Y.S.; Hewage, S.R.K.M.; Chae, S.W.; Hyun, J.W. Rosmarinic acid attenuates cell damage against UVB radiation-induced oxidative stress via enhancing antioxidant effects in human HaCaT cells. Biomol. Ther. 2016, 24, 75. [Google Scholar] [CrossRef] [Green Version]
- Vostálová, J.; Zdařilová, A.; Svobodová, A. Prunella vulgaris extract and rosmarinic acid prevent UVB-induced DNA damage and oxidative stress in HaCaT keratinocytes. Arch. Dermatol. Res. 2010, 302, 171–181. [Google Scholar] [CrossRef]
- Lembo, S.; Balato, A.; Di Caprio, R.; Cirillo, T.; Giannini, V.; Gasparri, F.; Monfrecola, G. The modulatory effect of ellagic acid and rosmarinic acid on ultraviolet-B-induced cytokine/chemokine gene expression in skin keratinocyte (HaCaT) cells. BioMed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef]
- Hahn, H.J.; Kim, K.B.; An, I.S.; Ahn, K.J.; Han, H.J. Protective effects of rosmarinic acid against hydrogen peroxide-induced cellular senescence and the inflammatory response in normal human dermal fibroblasts. Mol. Med. Rep. 2017, 16, 9763–9769. [Google Scholar] [CrossRef] [Green Version]
- Psotova, J.; Svobodova, A.; Kolarova, H.; Walterova, D. Photoprotective properties of Prunella vulgaris and rosmarinic acid on human keratinocytes. J. Photochem. Photobiol. B Biol. 2006, 84, 167–174. [Google Scholar] [CrossRef]
- Sánchez-Campillo, M.; Gabaldon, J.; Castillo, J.; Benavente-García, O.; Del Baño, M.; Alcaraz, M.; Vicente, V.; Alvarez, N.; Lozano, J. Rosmarinic acid, a photo-protective agent against UV and other ionizing radiations. Food Chem. Toxicol. 2009, 47, 386–392. [Google Scholar] [CrossRef] [PubMed]
- Abraham, S.K.; Sarma, L.; Kesavan, P. Protective effects of chlorogenic acid, curcumin and β-carotene against γ-radiation-induced in vivo chromosomal damage. Mutat. Res. Lett. 1993, 303, 109–112. [Google Scholar] [CrossRef]
- Her, Y.; Lee, T.-K.; Kim, J.D.; Kim, B.; Sim, H.; Lee, J.-C.; Ahn, J.H.; Park, J.H.; Lee, J.-W.; Hong, J. Topical Application of Aronia melanocarpa Extract Rich in Chlorogenic Acid and Rutin Reduces UVB-Induced Skin Damage via Attenuating Collagen Disruption in Mice. Molecules 2020, 25, 4577. [Google Scholar] [CrossRef]
- Ha, S.J.; Lee, J.; Kim, H.; Song, K.-M.; Lee, N.H.; Kim, Y.E.; Lee, H.; Kim, Y.H.; Jung, S.K. Preventive effect of Rhus javanica extract on UVB-induced skin inflammation and photoaging. J. Funct. Foods 2016, 27, 589–599. [Google Scholar] [CrossRef]
- Kwon, K.-R.; Alam, M.B.; Park, J.-H.; Kim, T.-H.; Lee, S.-H. Attenuation of UVB-induced photo-aging by polyphenolic-rich Spatholobus suberectus stem extract via modulation of MAPK/AP-1/MMPs signaling in human keratinocytes. Nutrients 2019, 11, 1341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeon, J.; Sung, J.; Lee, H.; Kim, Y.; Jeong, H.S.; Lee, J. Protective activity of caffeic acid and sinapic acid against UVB-induced photoaging in human fibroblasts. J. Food Biochem. 2019, 43, e12701. [Google Scholar] [CrossRef]
- Staniforth, V.; Huang, W.-C.; Aravindaram, K.; Yang, N.-S. Ferulic acid, a phenolic phytochemical, inhibits UVB-induced matrix metalloproteinases in mouse skin via posttranslational mechanisms. J. Nutr. Biochem. 2012, 23, 443–451. [Google Scholar] [CrossRef]
- Peres, D.D.A.; Sarruf, F.D.; de Oliveira, C.A.; Velasco, M.V.R.; Baby, A.R. Ferulic acid photoprotective properties in association with UV filters: Multifunctional sunscreen with improved SPF and UVA-PF. J. Photochem. Photobiol. B Biol. 2018, 185, 46–49. [Google Scholar] [CrossRef]
- Ha, S.J.; Lee, J.; Park, J.; Kim, Y.H.; Lee, N.H.; Kim, Y.E.; Song, K.-M.; Chang, P.-S.; Jeong, C.-H.; Jung, S.K. Syringic acid prevents skin carcinogenesis via regulation of NoX and EGFR signaling. Biochem. Pharmacol. 2018, 154, 435–445. [Google Scholar] [CrossRef]
- Choi, E.-J.; Ryu, Y.B.; Tang, Y.; Kim, B.R.; Lee, W.S.; Debnath, T.; Fan, M.; Lee, H.-S.; Kim, E.-K. Effect of cinnamamides on atopic dermatitis through regulation of IL-4 in CD4+ cells. J. Enzym. Inhib. Med. Chem. 2019, 34, 613–619. [Google Scholar] [CrossRef] [Green Version]
- Ong, P.Y.; Leung, D.Y. Bacterial and viral infections in atopic dermatitis: A comprehensive review. Clin. Rev. Allergy Immunol. 2016, 51, 329–337. [Google Scholar] [CrossRef]
- Zhou, Z.; Shi, T.; Hou, J.; Li, M. Ferulic acid alleviates atopic dermatitis-like symptoms in mice via its potent anti-inflammatory effect. Immunopharmacol. Immunotoxicol. 2020, 42, 156–164. [Google Scholar] [CrossRef]
- Lim, K.-M.; Bae, S.; Koo, J.E.; Kim, E.-S.; Bae, O.-N.; Lee, J.Y. Suppression of skin inflammation in keratinocytes and acute/chronic disease models by caffeic acid phenethyl ester. Arch. Dermatol. Res. 2015, 307, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Jang, A.-H.; Kim, T.-H.; Kim, G.-D.; Kim, J.E.; Kim, H.J.; Kim, S.S.; Jin, Y.-H.; Park, Y.S.; Park, C.-S. Rosmarinic acid attenuates 2, 4-dinitrofluorobenzene-induced atopic dermatitis in NC/Nga mice. Int. Immunopharmacol. 2011, 11, 1271–1277. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Jung, E.; Koh, J.; Kim, Y.S.; Park, D. Effect of rosmarinic acid on atopic dermatitis. J. Dermatol. 2008, 35, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Parveen, A.; Kang, M.C.; Subedi, L.; Lee, J.H.; Park, S.Y.; Jin, M.R.; Yoon, H.; Son, Y.K.; Kim, S.Y. Pyrus ussuriensis Maxim. leaves extract ameliorates DNCB-induced atopic dermatitis-like symptoms in NC/Nga mice. Phytomedicine 2018, 48, 76–83. [Google Scholar] [CrossRef]
- Yang, W.S.; Jeong, D.; Yi, Y.-S.; Park, J.G.; Seo, H.; Moh, S.H.; Hong, S.; Cho, J.Y. IRAK1/4-targeted anti-inflammatory action of caffeic acid. Med. Inflamm. 2013, 2013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Búfalo, M.C.; Ferreira, I.; Costa, G.; Francisco, V.; Liberal, J.; Cruz, M.T.; Lopes, M.C.; Batista, M.T.; Sforcin, J.M. Propolis and its constituent caffeic acid suppress LPS-stimulated pro-inflammatory response by blocking NF-κB and MAPK activation in macrophages. J. Ethnopharmacol. 2013, 149, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.Q.; Khan, R.; Qamar, W.; Lateef, A.; Ali, F.; Tahir, M.; Sultana, S. Caffeic acid attenuates 12-O-tetradecanoyl-phorbol-13-acetate (TPA)-induced NF-κB and COX-2 expression in mouse skin: Abrogation of oxidative stress, inflammatory responses and proinflammatory cytokine production. Food Chem. Toxicol. 2012, 50, 175–183. [Google Scholar] [CrossRef]
- Kim, M.-C.; Kim, S.-J.; Kim, D.-S.; Jeon, Y.-D.; Park, S.J.; Lee, H.S.; Um, J.-Y.; Hong, S.-H. Vanillic acid inhibits inflammatory mediators by suppressing NF-κB in lipopolysaccharide-stimulated mouse peritoneal macrophages. Immunopharmacol. Immunotoxicol. 2011, 33, 525–532. [Google Scholar] [CrossRef]
- Calixto-Campos, C.; Carvalho, T.T.; Hohmann, M.S.; Pinho-Ribeiro, F.A.; Fattori, V.; Manchope, M.F.; Zarpelon, A.C.; Baracat, M.M.; Georgetti, S.R.; Casagrande, R. Vanillic acid inhibits inflammatory pain by inhibiting neutrophil recruitment, oxidative stress, cytokine production, and NFκB activation in mice. J. Nat. Prod. 2015, 78, 1799–1808. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, S.F.; Gabr, N.M.; Zaki, J.T.; El Awdan, S.A.; Mina, S.A. The anti-inflammatory, anti-ulcer activities and phytochemical investigation of Cucumis melo L. cv. Ismailawi fruits. Nat. Prod. Res. 2020, 1–5. [Google Scholar] [CrossRef]
- Rocha, J.; Eduardo-Figueira, M.; Barateiro, A.; Fernandes, A.; Brites, D.; Bronze, R.; Duarte, C.M.; Serra, A.T.; Pinto, R.; Freitas, M. Anti-inflammatory effect of rosmarinic acid and an extract of Rosmarinus officinalis in rat models of local and systemic inflammation. Basic Clin. Pharmacol. Toxicol. 2015, 116, 398–413. [Google Scholar] [CrossRef]
- Usha, T.; Middha, S.K.; Bhattacharya, M.; Lokesh, P.; Goyal, A.K. Rosmarinic acid, a new polyphenol from Baccaurea ramiflora Lour. leaf: A probable compound for its anti-inflammatory activity. Antioxidants 2014, 3, 830–842. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, M.D.; Almeida, M.C.; Lopes, N.P.; De Souza, G.E.P. Evaluation of the anti-inflammatory, analgesic and antipyretic activities of the natural polyphenol chlorogenic acid. Biol. Pharm. Bull. 2006, 29, 2236–2240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ha, H.; Lee, H.; Seo, C.S.; Lim, H.-S.; Lee, J.K.; Lee, M.-Y.; Shin, H. Artemisia capillaris inhibits atopic dermatitis-like skin lesions in Dermatophagoides farinae-sensitized Nc/Nga mice. BMC Complement. Altern. Med. 2014, 14, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Rapalli, V.K.; Waghule, T.; Gorantla, S.; Dubey, S.K.; Saha, R.N.; Singhvi, G. Psoriasis: Pathological mechanisms, current pharmacological therapies, and emerging drug delivery systems. Drug Discov. Today 2020. [Google Scholar] [CrossRef] [PubMed]
- Saleem, S.; Iqubal, M.K.; Garg, S.; Ali, J.; Baboota, S. Trends in nanotechnology-based delivery systems for dermal targeting of drugs: An enticing approach to offset psoriasis. Expert Opin. Drug Deliv. 2020, 17, 817–838. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.-W.; Jiang, R.-H.; Kim, K.-D.; Lee, J.-H.; Kim, C.-D.; Yin, W.-T.; Lee, J.-H. Rosmarinic acid inhibits poly (I: C)-induced inflammatory reaction of epidermal keratinocytes. Life Sci. 2016, 155, 189–194. [Google Scholar] [CrossRef]
- Zhang, M.; Li, N.; Cai, R.; Gu, J.; Xie, F.; Wei, H.; Lu, C.; Wu, D. Rosmarinic acid protects mice from imiquimod induced psoriasis-like skin lesions by inhibiting the IL-23/Th17 axis via regulating Jak2/Stat3 signaling pathway. Phytother. Res. 2021. [Google Scholar] [CrossRef]
- Koycheva, I.K.; Vasileva, L.V.; Amirova, K.M.; Marchev, A.S.; Balcheva-Sivenova, Z.P.; Georgiev, M.I. Biotechnologically Produced Lavandula angustifolia Mill. Extract Rich in Rosmarinic Acid Resolves Psoriasis-Related Inflammation Through Janus Kinase/Signal Transducer and Activator of Transcription Signaling. Front. Pharmacol. 2021, 12, 940. [Google Scholar] [CrossRef] [PubMed]
- Lo, H.-Y.; Li, C.-C.; Cheng, H.-M.; Liu, I.-C.; Ho, T.-Y.; Hsiang, C.-Y. Ferulic acid altered IL-17A/IL-17RA interaction and protected against imiquimod-induced psoriasis-like skin injury in mice. Food Chem. Toxicol. 2019, 129, 365–375. [Google Scholar] [CrossRef]
- Cheng, H.-M.; Chen, F.-Y.; Li, C.-C.; Lo, H.-Y.; Liao, Y.-F.; Ho, T.-Y.; Hsiang, C.-Y. Oral administration of vanillin improves imiquimod-induced psoriatic skin inflammation in mice. J. Agric. Food Chem. 2017, 65, 10233–10242. [Google Scholar] [CrossRef]
- Lee, S.Y.; Nam, S.; Hong, I.K.; Kim, H.; Yang, H.; Cho, H.J. Antiproliferation of keratinocytes and alleviation of psoriasis by the ethanol extract of Artemisia capillaris. Phytother. Res. 2018, 32, 923–932. [Google Scholar] [CrossRef]
- Zhang, Y.-T.; Xu, Y.-M.; Zhang, S.-J.; Zhao, J.-H.; Wang, Z.; Xu, D.-Q.; Feng, N.-P. In vivo microdialysis for the evaluation of transfersomes as a novel transdermal delivery vehicle for cinnamic acid. Drug Dev. Ind. Pharm. 2014, 40, 301–307. [Google Scholar] [CrossRef]
- Aitipamula, S.; Das, S. Cocrystal formulations: A case study of topical formulations consisting of ferulic acid cocrystals. Eur. J. Pharm. Biopharm. 2020, 149, 95–104. [Google Scholar] [CrossRef]
- Suzuki, T.; Sasai, A.; Tsujimoto, H.; Yasunaga, T.; Ogawa, N.; Yamamoto, H. Promoting effect of type 17 collagen production by chlorogenic acid using PLGA nanoparticles in the human epidermal keratinocyte cell. J. Drug Deliv. Sci. Technol. 2020, 58, 101624. [Google Scholar] [CrossRef]
- Ammar, N.M.; Hassan, H.A.; Mohammed, M.A.; Serag, A.; Abd El-Alim, S.H.; Elmotasem, H.; El Raey, M.; El Gendy, A.N.; Sobeh, M.; Abdel-Hamid, A.-H.Z. Metabolomic profiling to reveal the therapeutic potency of Posidonia oceanica nanoparticles in diabetic rats. RSC Adv. 2021, 11, 8398–8410. [Google Scholar] [CrossRef]
- Chhabra, P.; Chauhan, G.; Kumar, A. Augmented healing of full thickness chronic excision wound by rosmarinic acid loaded chitosan encapsulated graphene nanopockets. Drug Dev. Ind. Pharm. 2020, 46, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Alberti, T.; Coelho, D.; Voytena, A.; Iacovski, R.; Mazzarino, L.; Maraschin, M.; Veleirinho, B. Effect of propolis nanoparticles on early-stage wound healing in a diabetic noncontractile wound model. Nanotechnol. Adv. Mater. Sci. 2019, 2, 1–10. [Google Scholar]
- Sguizzato, M.; Mariani, P.; Ferrara, F.; Drechsler, M.; Hallan, S.S.; Huang, N.; Simelière, F.; Khunti, N.; Cortesi, R.; Marchetti, N. Nanoparticulate Gels for Cutaneous Administration of Caffeic Acid. Nanomaterials 2020, 10, 961. [Google Scholar] [CrossRef]
- Carbone, C.; Caddeo, C.; Grimaudo, M.A.; Manno, D.E.; Serra, A.; Musumeci, T. Ferulic Acid-NLC with Lavandula Essential Oil: A Possible Strategy for Wound-Healing? Nanomaterials 2020, 10, 898. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Luna, A.; Talero, E.; Ávila-Román, J.; Romero, A.M.F.; Rabasco, A.M.; Motilva, V.; González-Rodríguez, M.L. Preparation and In Vivo Evaluation of Rosmarinic Acid-Loaded Transethosomes After Percutaneous Application on a Psoriasis Animal Model. AAPS Pharm.Sci. Tech. 2021, 22, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Srivastava, N.; Yadav, K.S.; Sinha, P.; Yadav, N.P. Preparation, optimization, characterization and bioevaluation of rosmarinic acid loaded phytovesicles for anti-inflammatory activity. J. Drug Deliv. Sci. Technol. 2020, 59, 101888. [Google Scholar] [CrossRef]
- Memic, A.; Abudula, T.; Mohammed, H.S.; Joshi Navare, K.; Colombani, T.; Bencherif, S.A. Latest progress in electrospun nanofibers for wound healing applications. ACS Appl. Bio Mater. 2019, 2, 952–969. [Google Scholar] [CrossRef]
- Suarato, G.; Contardi, M.; Perotto, G.; Jose’A, H.-G.; Fiorentini, F.; Ceseracciu, L.; Pignatelli, C.; Debellis, D.; Bertorelli, R.; Athanassiou, A. From fabric to tissue: Recovered wool keratin/polyvinylpyrrolidone biocomposite fibers as artificial scaffold platform. Mater. Sci. Eng. C 2020, 116, 111151. [Google Scholar] [CrossRef]
- Rogina, A. Electrospinning process: Versatile preparation method for biodegradable and natural polymers and biocomposite systems applied in tissue engineering and drug delivery. Appl. Surf. Sci. 2014, 296, 221–230. [Google Scholar] [CrossRef]
- Bombin, A.D.J.; Dunne, N.; McCarthy, H.O. Electrospinning of natural polymers for the production of nanofibres for wound healing applications. Mater. Sci. Eng. C 2020, 110994. [Google Scholar] [CrossRef] [PubMed]
- Kossyvaki, D.; Suarato, G.; Summa, M.; Gennari, A.; Francini, N.; Gounaki, I.; Venieri, D.; Tirelli, N.; Bertorelli, R.; Athanassiou, A. Keratin–cinnamon essential oil biocomposite fibrous patches for skin burn care. Mater. Adv. 2020, 1, 1805–1816. [Google Scholar] [CrossRef]
- Ignatova, M.G.; Manolova, N.E.; Rashkov, I.B.; Markova, N.D.; Toshkova, R.A.; Georgieva, A.K.; Nikolova, E.B. Poly (3-hydroxybutyrate)/caffeic acid electrospun fibrous materials coated with polyelectrolyte complex and their antibacterial activity and in vitro antitumor effect against HeLa cells. Mater. Sci. Eng. C 2016, 65, 379–392. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, V.; Alam, M.; Ahmad, N.; Balakrishnan, S.B.; Ganesan, V.; Shanmugasundaram, E.; Rajagopal, B.; Thambusamy, S. Electrospun poly (vinyl alcohol) nanofibers incorporating caffeic acid/cyclodextrins through the supramolecular assembly for antibacterial activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 249, 119308. [Google Scholar] [CrossRef]
- Oh, G.-W.; Ko, S.-C.; Je, J.-Y.; Kim, Y.-M.; Oh, J.; Jung, W.-K. Fabrication, characterization and determination of biological activities of poly (ε-caprolactone)/chitosan-caffeic acid composite fibrous mat for wound dressing application. Int. J. Biol. Macromol. 2016, 93, 1549–1558. [Google Scholar] [CrossRef]
- Chuysinuan, P.; Pavasant, P.; Supaphol, P. Preparation and characterization of caffeic acid-grafted electrospun poly (l-lactic acid) fiber mats for biomedical applications. ACS Appl. Mater. Interfaces 2012, 4, 3031–3040. [Google Scholar] [CrossRef] [PubMed]
- Adomavičiūtė, E.; Stanys, S.; Žilius, M.; Juškaitė, V.; Pavilonis, A.; Briedis, V. Formation and biopharmaceutical characterization of electrospun PVP mats with propolis and silver nanoparticles for fast releasing wound dressing. BioMed. Res. Int. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Adomavičiūtė, E.; Pupkevičiūtė, S.; Juškaitė, V.; Žilius, M.; Stanys, S.; Pavilonis, A.; Briedis, V. Formation and investigation of electrospun PLA materials with propolis extracts and silver nanoparticles for biomedical applications. J. Nanomater. 2017, 2017. [Google Scholar] [CrossRef] [Green Version]
- Vatankhah, E. Rosmarinic acid-loaded electrospun nanofibers: In vitro release kinetic study and bioactivity assessment. Eng. Life Sci. 2018, 18, 732–742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandova-Herrera, I.; Ledezma-Pérez, A.; De-León, A.; Alvaro-Canche, C.; Torres-Lubian, R.; Romero-García, J. Fabrication and Sustained Release of Chlorogenic Acid from Poly (vinyl alcohol)/Poly (γ-glutamic Acid) Blends Electrospun Mats. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Balaji, A.; Vellayappan, M.; John, A.; Subramanian, A.; Jaganathan, S.; Supriyanto, E.; Razak, S. An insight on electrospun-nanofibers-inspired modern drug delivery system in the treatment of deadly cancers. RSC Adv. 2015, 5, 57984–58004. [Google Scholar] [CrossRef]
- Poornima, B.; Korrapati, P.S. Fabrication of chitosan-polycaprolactone composite nanofibrous scaffold for simultaneous delivery of ferulic acid and resveratrol. Carbohydr. Polym. 2017, 157, 1741–1749. [Google Scholar] [CrossRef]
- Zhao, Z.; Vizetto-Duarte, C.; Moay, Z.K.; Setyawati, M.I.; Rakshit, M.; Kathawala, M.H.; Ng, K.W. Composite hydrogels in three-dimensional in vitro models. Front. Bioeng. Biotechnol. 2020, 8, 611. [Google Scholar] [CrossRef]
- Tomatsu, I.; Peng, K.; Kros, A. Photoresponsive hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2011, 63, 1257–1266. [Google Scholar] [CrossRef]
- Debele, T.A.; Su, W.-P. Polysaccharide and protein-based functional wound dressing materials and applications. Int. J. Polym. Mater. Polym. Biomater. 2020, 1–22. [Google Scholar] [CrossRef]
- Das, S.; Wong, A.B. Stabilization of ferulic acid in topical gel formulation via nanoencapsulation and pH optimization. Sci. Rep. 2020, 10, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, S.; Murali, R.; Arathanaikotti, D.; Gopinath, A.; Senthilkumar, C.; Kesavan, S.; Madhan, B. Ferulic acid loaded microspheres reinforced in 3D hybrid scaffold for antimicrobial wound dressing. Int. J. Biol. Macromol. 2021, 177, 463–473. [Google Scholar] [CrossRef]
- Raja, S.T.K.; Thiruselvi, T.; Aravindhan, R.; Mandal, A.B.; Gnanamani, A. In vitro and in vivo assessments of a 3-(3, 4-dihydroxyphenyl)-2-propenoic acid bioconjugated gelatin-based injectable hydrogel for biomedical applications. J. Mater. Chem. B 2015, 3, 1230–1244. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.R.; Subbarayan, R.; Dinesh, M.G.; Arumugam, G.; Raja, S.T.K. Differentiation of human gingival mesenchymal stem cells into neuronal lineages in 3D bioconjugated injectable protein hydrogel construct for the management of neuronal disorder. Exp. Mol. Med. 2016, 48, e209. [Google Scholar] [CrossRef] [Green Version]
- Budhiraja, A.; Dhingra, G. Development and characterization of a novel antiacne niosomal gel of rosmarinic acid. Drug Deliv. 2015, 22, 723–730. [Google Scholar] [CrossRef] [Green Version]
- Azadmanesh, F.; Pourmadadi, M.; Zavar Reza, J.; Yazdian, F.; Omidi, M.; Haghirosadat, B.F. Synthesis of a novel nanocomposite containing chitosan as a three-dimensional printed wound dressing technique: Emphasis on gene expression. Biotechnol. Prog. 2021, e3132. [Google Scholar] [CrossRef]
- Kanitkar, A.; Smoak, M.; Chen, C.; Aita, G.; Scherr, T.; Madsen, L.; Hayes, D. Synthesis of novel polyesters for potential applications in skin tissue engineering. J. Chem. Technol. Biotechnol. 2016, 91, 733–741. [Google Scholar] [CrossRef]
- Moreira, J.; Vale, A.C.; Alves, N.M. Spin-coated freestanding films for biomedical applications. J. Mater. Chem. B 2021, 9, 3778–3799. [Google Scholar] [CrossRef]
- Kathe, K.; Kathpalia, H. Film forming systems for topical and transdermal drug delivery. Asian J. Pharm. Sci. 2017, 12, 487–497. [Google Scholar] [CrossRef]
- Contardi, M.; Russo, D.; Suarato, G.; Heredia-Guerrero, J.A.; Ceseracciu, L.; Penna, I.; Margaroli, N.; Summa, M.; Spanò, R.; Tassistro, G. Polyvinylpyrrolidone/hyaluronic acid-based bilayer constructs for sequential delivery of cutaneous antiseptic and antibiotic. Chem. Eng. J. 2019, 358, 912–923. [Google Scholar] [CrossRef]
- Fiorentini, F.; Suarato, G.; Grisoli, P.; Zych, A.; Bertorelli, R.; Athanassiou, A. Plant-based biocomposite films as potential antibacterial patches for skin wound healing. Eur. Polym. J. 2021, 150, 110414. [Google Scholar] [CrossRef]
- Liakos, I.; Rizzello, L.; Scurr, D.J.; Pompa, P.P.; Bayer, I.S.; Athanassiou, A. All-natural composite wound dressing films of essential oils encapsulated in sodium alginate with antimicrobial properties. Int. J. Pharm. 2014, 463, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Neto, R.J.G.; Genevro, G.M.; de Almeida Paulo, L.; Lopes, P.S.; de Moraes, M.A.; Beppu, M.M. Characterization and in vitro evaluation of chitosan/konjac glucomannan bilayer film as a wound dressing. Carbohydr. Polym. 2019, 212, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Contardi, M.; Heredia-Guerrero, J.A.; Perotto, G.; Valentini, P.; Pompa, P.P.; Spanò, R.; Goldoni, L.; Bertorelli, R.; Athanassiou, A.; Bayer, I.S. Transparent ciprofloxacin-povidone antibiotic films and nanofiber mats as potential skin and wound care dressings. Eur. J. Pharm. Sci. 2017, 104, 133–144. [Google Scholar] [CrossRef]
- Bouthillette, M.; Beccati, D.; Akthakul, A.; Ramadurai, N.; Nashat, A.; Langer, R.; Anderson, R.R.; Sakamoto, F.H. A crosslinked polymer skin barrier film for moderate to severe atopic dermatitis: A pilot study in adults. J. Am. Acad. Dermatol 2020, 82, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.-W.; Kim, M.-H.; Sohn, S.-Y.; Kim, K.-T.; Park, J.-H.; Lee, S.-Y.; Lee, J.-Y.; Kim, D.-D. Curcumin-loaded lipid-hybridized cellulose nanofiber film ameliorates imiquimod-induced psoriasis-like dermatitis in mice. Biomaterials 2018, 182, 245–258. [Google Scholar] [CrossRef] [PubMed]
- Contardi, M.; Heredia-Guerrero, J.A.; Guzman-Puyol, S.; Summa, M.; BENITEZ, J.J.; Goldoni, L.; Caputo, G.; Cusimano, G.; Picone, P.; Di Carlo, M. Combining Dietary Phenolic Antioxidants with Polyvinylpyrrolidone: Transparent Biopolymer Films based on p-Coumaric Acid for Controlled Release. J. Mater. Chem. B 2019, 7, 1384–1396. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.I.; Nguyen, T.T.; Peng, Z.; Chang, M. Targeting MMP-9 in diabetic foot ulcers. Pharmaceuticals 2019, 12, 79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Contardi, M.; Alfaro-Pulido, A.; Picone, P.; Guzman-Puyol, S.; Goldoni, L.; Benítez, J.J.; Heredia, A.; Barthel, M.J.; Ceseracciu, L.; Cusimano, G. Low molecular weight ε-caprolactone-p-coumaric acid copolymers as potential biomaterials for skin regeneration applications. PLoS ONE 2019, 14, e0214956. [Google Scholar] [CrossRef]
- Žilius, M.; Ramanauskienė, K.; Briedis, V. Release of propolis phenolic acids from semisolid formulations and their penetration into the human skin in vitro. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seo, Y.; Kim, S.; Boo, Y.; Baek, J.; Lee, S.; Koh, J. Effects of p-coumaric acid on erythema and pigmentation of human skin exposed to ultraviolet radiation. Clin. Exp. Dermatol. Clin. Dermatol. 2011, 36, 260–266. [Google Scholar] [CrossRef]
- Küba, M.; Türkoğlu, A.; Oğuz, A.; Tuncer, M.; Kaya, Ş.; Başol, Ö.; Bilge, H.; Tatlı, F. Comparison of local rosmarinic acid and topical dexpanthenol applications on wound healing in a rat experimental wound model. Folia Morphol. 2020. [Google Scholar] [CrossRef]
- Kitagawa, S.; Yoshii, K.; Morita, S.-Y.; Teraoka, R. Efficient topical delivery of chlorogenic acid by an oil-in-water microemulsion to protect skin against UV-induced damage. Chem. Pharm. Bull. 2011, 59, 793–796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qi, W.; Zhao, T.; Yang, W.-W.; Wang, G.-H.; Yu, H.; Zhao, H.-X.; Yang, C.; Sun, L.-X. Comparative pharmacokinetics of chlorogenic acid after oral administration in rats. J. Pharm. Anal. 2011, 1, 270–274. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharyya, S.; Majhi, S.; Saha, B.P.; Mukherjee, P.K. Chlorogenic acid–phospholipid complex improve protection against UVA induced oxidative stress. J. Photochem. Photobiol. B Biol. 2014, 130, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Yutani, R.; Kikuchi, T.; Teraoka, R.; Kitagawa, S. Efficient delivery and distribution in skin of chlorogenic acid and resveratrol induced by microemulsion using sucrose laurate. Chem. Pharm. Bull. 2014, 62, 274–280. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.-C.; Liou, S.-S.; Tzeng, T.-F.; Lee, S.-L.; Liu, I.-M. Effect of topical application of chlorogenic acid on excision wound healing in rats. Planta Med. 2013, 79, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Waghule, T.; Singhvi, G.; Dubey, S.K.; Pandey, M.M.; Gupta, G.; Singh, M.; Dua, K. Microneedles: A smart approach and increasing potential for transdermal drug delivery system. Biomed. Pharmacother. 2019, 109, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Zhang, Q.; Yang, T.; Liu, Y.; Liu, R. 3D printing of multi-scalable structures via high penetration near-infrared photopolymerization. Nat. Commun. 2020, 11, 3462. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.-y.; Gu, Z.-x.; You, X.; Liu, G.; Tan, Y.; Zhang, H. Screening of edible mushrooms for release of ferulic acid from wheat bran by fermentation. Enzym. Microb. Technol. 2010, 46, 125–128. [Google Scholar] [CrossRef]
- Antinori, M.E.; Contardi, M.; Suarato, G.; Armirotti, A.; Bertorelli, R.; Mancini, G.; Debellis, D.; Athanassiou, A. Advanced mycelium materials as potential self-growing biomedical scaffolds. Sci. Rep. 2021, 11, 12630. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, K.B.; Zo, S.M.; Han, S.S. Novel biomimetic chitin-glucan polysaccharide nano/microfibrous fungal-scaffolds for tissue engineering applications. Int. J. Biol. Macromol. 2020, 149, 724–731. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Contardi, M.; Lenzuni, M.; Fiorentini, F.; Summa, M.; Bertorelli, R.; Suarato, G.; Athanassiou, A. Hydroxycinnamic Acids and Derivatives Formulations for Skin Damages and Disorders: A Review. Pharmaceutics 2021, 13, 999. https://doi.org/10.3390/pharmaceutics13070999
Contardi M, Lenzuni M, Fiorentini F, Summa M, Bertorelli R, Suarato G, Athanassiou A. Hydroxycinnamic Acids and Derivatives Formulations for Skin Damages and Disorders: A Review. Pharmaceutics. 2021; 13(7):999. https://doi.org/10.3390/pharmaceutics13070999
Chicago/Turabian StyleContardi, Marco, Martina Lenzuni, Fabrizio Fiorentini, Maria Summa, Rosalia Bertorelli, Giulia Suarato, and Athanassia Athanassiou. 2021. "Hydroxycinnamic Acids and Derivatives Formulations for Skin Damages and Disorders: A Review" Pharmaceutics 13, no. 7: 999. https://doi.org/10.3390/pharmaceutics13070999
APA StyleContardi, M., Lenzuni, M., Fiorentini, F., Summa, M., Bertorelli, R., Suarato, G., & Athanassiou, A. (2021). Hydroxycinnamic Acids and Derivatives Formulations for Skin Damages and Disorders: A Review. Pharmaceutics, 13(7), 999. https://doi.org/10.3390/pharmaceutics13070999








