Cylindrical Microparticles Composed of Mesoporous Silica Nanoparticles for the Targeted Delivery of a Small Molecule and a Macromolecular Drug to the Lungs: Exemplified with Curcumin and siRNA
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Rhodamine B Functionalization of mSNP for Visualization
2.3. BET Measurement to Evaluate Pore Volume and Loading with Curcumin
- p = partial vapor pressure of the adsorbed gas in equilibrium with the surface at 77.4 K (boiling point of liquid nitrogen) (Pa);
- p0 = saturation pressure of the adsorbed gas (Pa);
- Va = volume of adsorbed gas under standard conditions (273.15 K and 1.013 × 105 Pa) (mL);
- Vm = volume of the adsorbed gas, to enable monolayer formation on the sample surface (mL);
- C = dimensionless constant related to the adsorption enthalpy of the adsorbing gas on the powder sample.
2.4. mSNP Pore Loading with Curcumin
2.5. Determination of Colloidal Properties
2.6. Preparation of Cylindrical Microparticles Composed of mSNPs
2.7. Aerodynamic Characteristics Analysed by Next Generation Impactor
2.8. Differentiation of Human Monocytes (THP-1) into Macrophage-Like Cells
2.9. Human Alveolar Basal Epithelial Cell Line A549
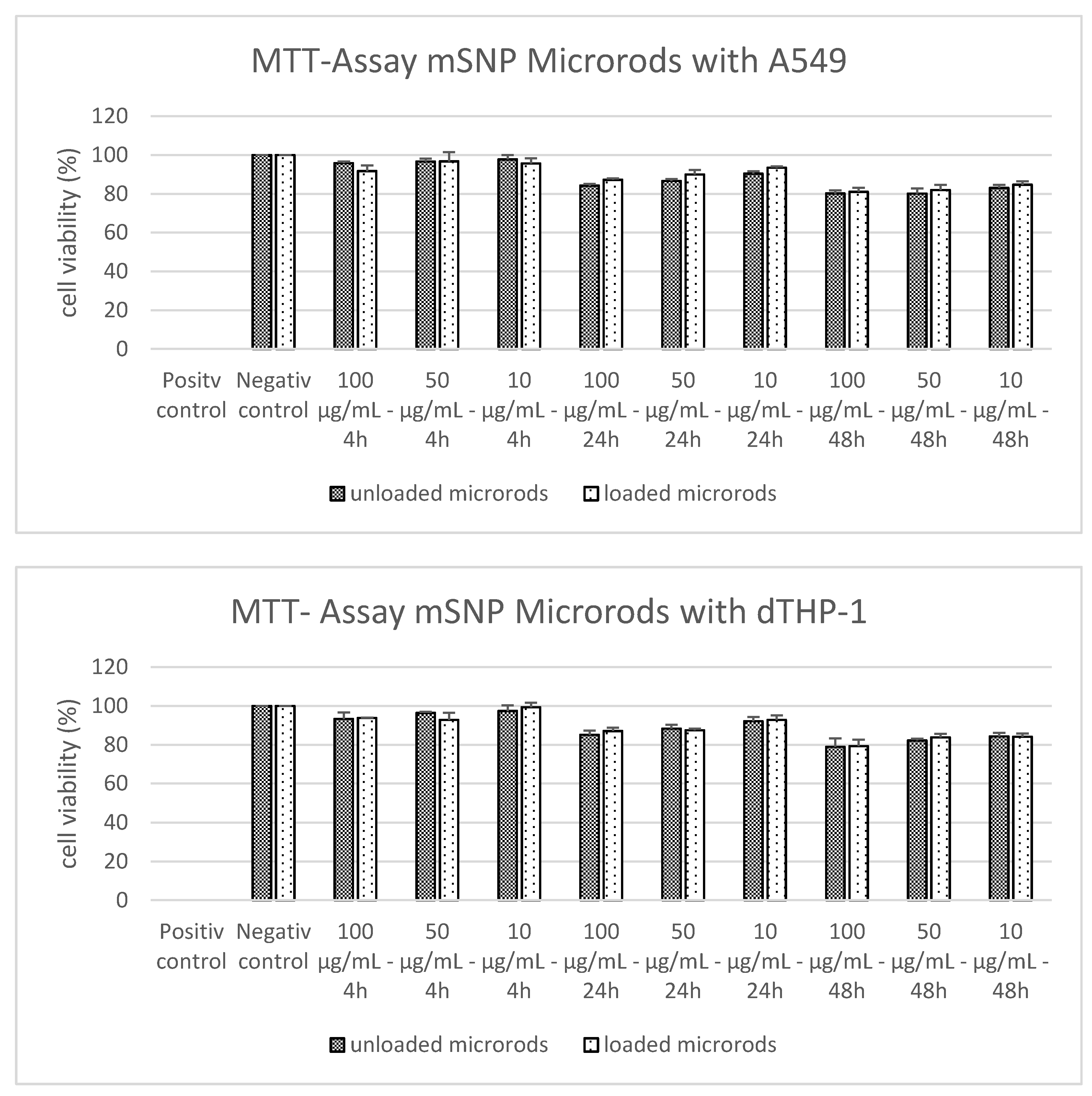
2.10. Determination of Cell Viability by MTT Assay
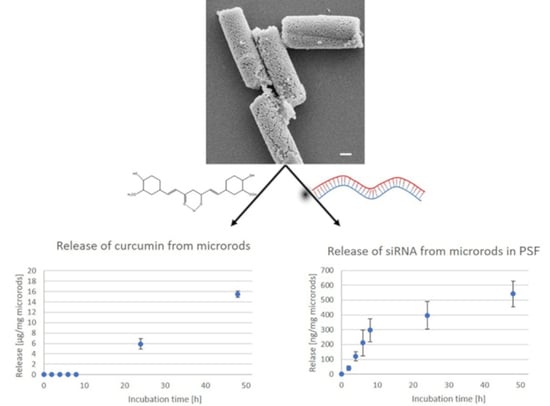
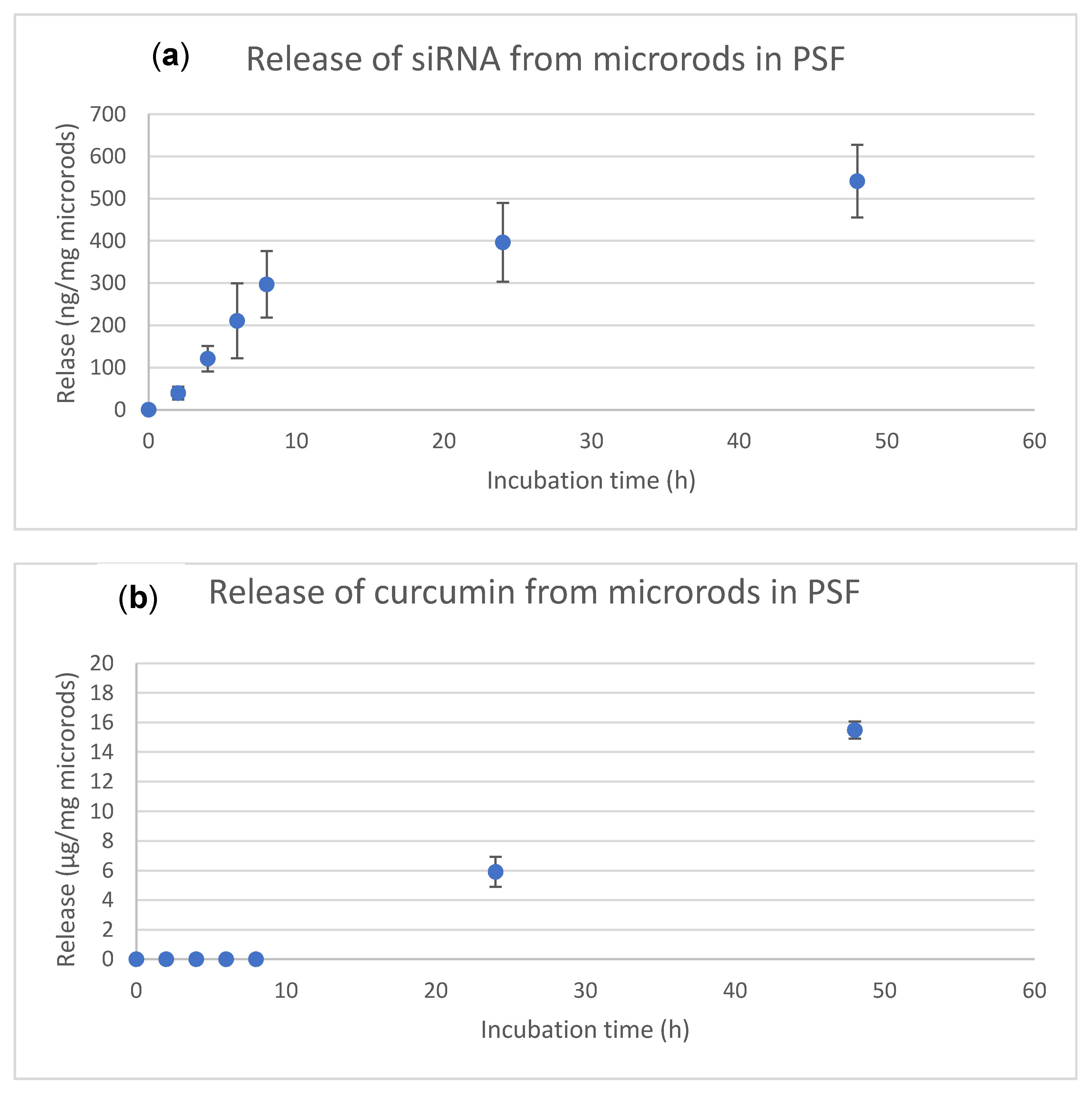
2.11. In Vitro Release of siRNA and Curcumin under Phagolysosomal Conditions
2.12. Quantification of TNF-α Release by Enzyme Linked Immunosorbent Assay (ELISA)
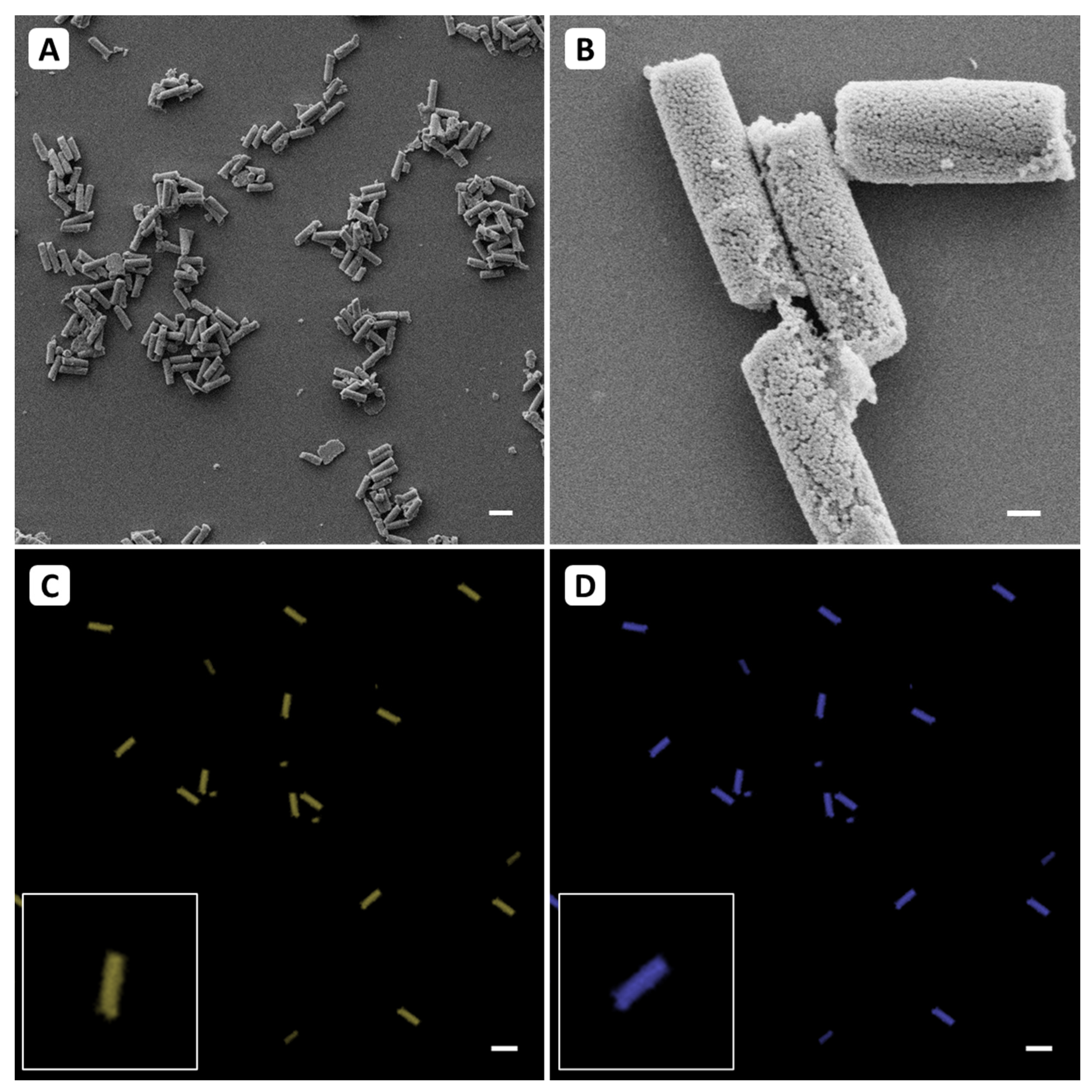
2.13. Scanning Electron Microscopy for Analysis of Rod Morphology
2.14. Confocal Laser Scanning Microscopy for Particle Visualization
2.15. Statistical Evaluation
3. Results
3.1. Particle Characterization
3.2. Evaluation of Aerodynamic Properties by Next Generation Impactor
- da = aerodynamic diameter;
- dve = volume equivalent diameter;
- χ = dynamic shape factor;
- = particle density;
- = standard density;
- Cc(dve) = Cunningham slip correction factor of the volume equivalent diameter;
- Cc(da) = Cunningham slip correction factor of the aerodynamic diameter.
3.3. Drug Loading and Release Kinetics
3.4. Cell Viability of Alveolar Basal Epithelial Cells and Macrophages
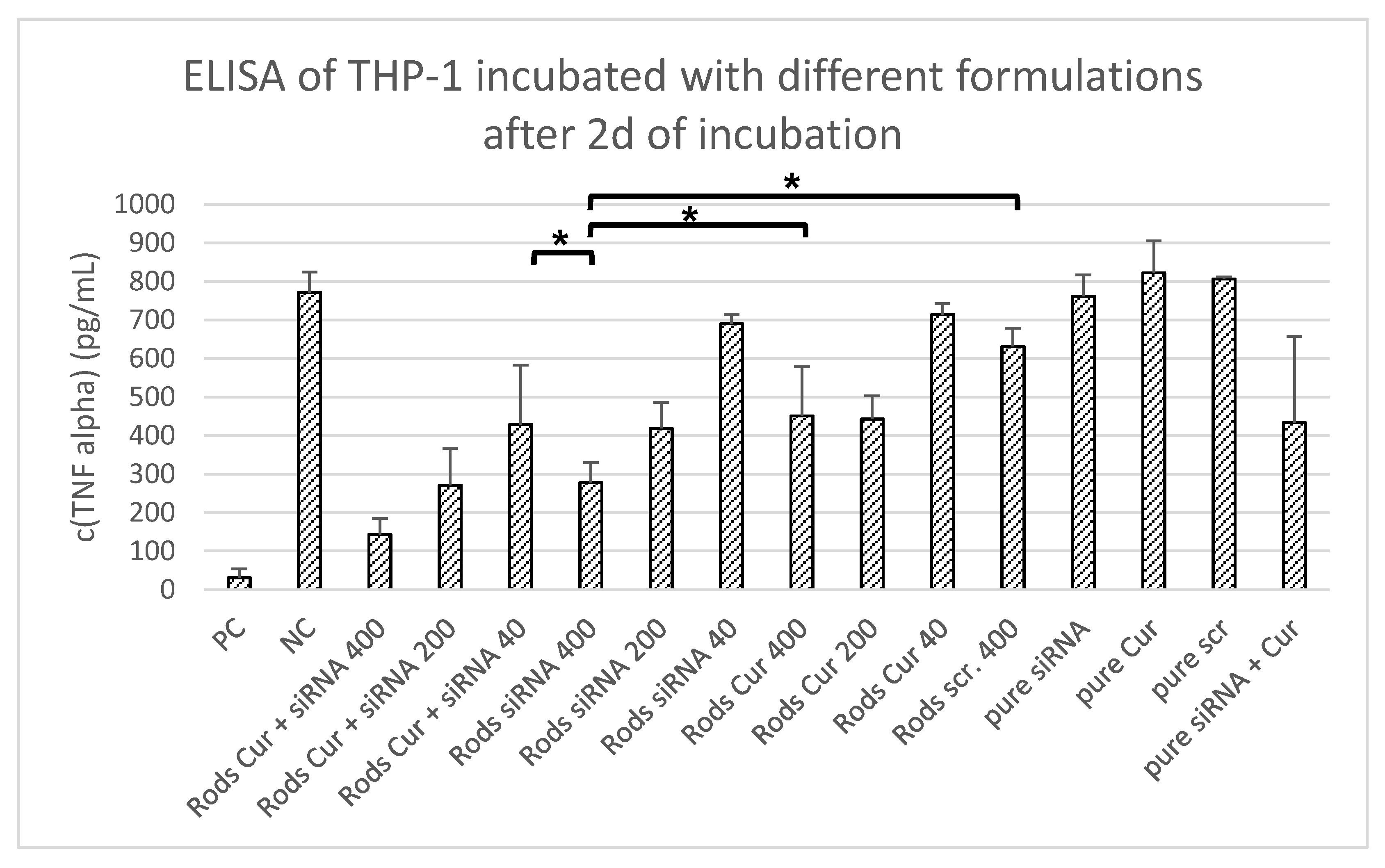
3.5. TNF-α Quantification via ELISA
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khaltaev, N.; Axelrod, S. Chronic respiratory diseases global mortality trends, treatment guidelines, life style modifications, and air pollution: Preliminary analysis. J. Thorac. Dis. 2019, 11, 2643. [Google Scholar] [CrossRef]
- Mehta, M.; Satija, S.; Paudel, K.R.; Malyla, V.; Kannaujiya, V.K.; Chellappan, D.K.; Bebawy, M.; Hansbro, P.M.; Wich, P.R.; Dua, K. Targeting respiratory diseases using miRNA inhibitor based nanotherapeutics: Current status and future perspectives. Nanomed. Nanotechnol. Biol. Med. 2020, 31, 102303. [Google Scholar] [CrossRef] [PubMed]
- Mirza, S.; Clay, R.D.; Koslow, M.A.; Scanlon, P.D. COPD Guidelines: A Review of the 2018 GOLD Report. Mayo Clin. Proc. 2018, 93, 1488–1502. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, G.J.; Rodrigo, C.; Hall, J.B. Acute asthma in adults: A review. Chest 2004, 125, 1081–1102. [Google Scholar] [CrossRef] [PubMed]
- Strausbaugh, S.D.; Davis, P.B. Cystic fibrosis: A review of epidemiology and pathobiology. Clin. Chest Med. 2007, 28, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Chin, L.H.; Hon, C.M.; Chellappan, D.K.; Chellian, J.; Madheswaran, T.; Zeeshan, F.; Awasthi, R.; Aljabali, A.A.; Tambuwala, M.M.; Dureja, H. Molecular mechanisms of action of naringenin in chronic airway diseases. Eur. J. Pharmacol. 2020, 879, 173139. [Google Scholar] [CrossRef] [PubMed]
- Mehta, M.; Dhanjal, D.S.; Paudel, K.R.; Singh, B.; Gupta, G.; Rajeshkumar, S.; Thangavelu, L.; Tambuwala, M.M.; Bakshi, H.A.; Chellappan, D.K. Cellular signalling pathways mediating the pathogenesis of chronic inflammatory respiratory diseases: An update. Inflammopharmacology 2020, 28, 795–817. [Google Scholar] [CrossRef]
- Lee, T.I.; Young, R.A. Transcriptional regulation and its misregulation in disease. Cell 2013, 152, 1237–1251. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, S.; Mahjoubin-Tehran, M.; Aghaee-Bakhtiari, S.H.; Jalili, A.; Movahedpour, A.; Khan, H.; Moghoofei, M.; Shojaei, Z.; Hamblin, M.R.; Mirzaei, H. Autophagy-related MicroRNAs in chronic lung diseases and lung cancer. Crit. Rev. Oncol. Hematol. 2020, 153, 103063. [Google Scholar] [CrossRef] [PubMed]
- Samuelson, D.R.; Welsh, D.A.; Shellito, J.E. Regulation of lung immunity and host defense by the intestinal microbiota. Front. Microbiol. 2015, 6, 1085. [Google Scholar] [CrossRef]
- Lubamba, B.A.; Jones, L.C.; O’Neal, W.K.; Boucher, R.C.; Ribeiro, C.M. X-box–binding protein 1 and innate immune responses of human cystic fibrosis alveolar macrophages. Am. J. Respir. Crit. Care Med. 2015, 192, 1449–1461. [Google Scholar] [CrossRef]
- Shetty, N.; Cipolla, D.; Park, H.; Zhou, Q.T. Physical stability of dry powder inhaler formulations. Expert Opin. Drug Deliv. 2020, 17, 77–96. [Google Scholar] [CrossRef]
- Singh, D.K.; Thakur, R.K. Dry Powder Inhalers. J. Crit. Rev. 2020, 7, 915–920. [Google Scholar]
- Lechanteur, A.; Evrard, B. Influence of Composition and Spray-Drying Process Parameters on Carrier-Free DPI Properties and Behaviors in the Lung: A review. Pharmaceutics 2020, 12, 55. [Google Scholar] [CrossRef]
- Nanologica. NLAB Spiro™; Nanologica: Södertälje, Sweden, 2020. [Google Scholar]
- Narayan, R.; Nayak, U.Y.; Raichur, A.M.; Garg, S. Mesoporous silica nanoparticles: A comprehensive review on synthesis and recent advances. Pharmaceutics 2018, 10, 118. [Google Scholar] [CrossRef] [PubMed]
- Manzano, M.; Vallet-Regí, M. Mesoporous silica nanoparticles for drug delivery. Adv. Funct. Mater. 2020, 30, 1902634. [Google Scholar] [CrossRef]
- Castillo, R.R.; Lozano, D.; Vallet-Regí, M. Mesoporous silica nanoparticles as carriers for therapeutic biomolecules. Pharmaceutics 2020, 12, 432. [Google Scholar] [CrossRef]
- Kong, Z.L.; Kuo, H.P.; Johnson, A.; Wu, L.C.; Chang, K.L.B. Curcumin-Loaded Mesoporous Silica Nanoparticles Markedly Enhanced Cytotoxicity in Hepatocellular Carcinoma Cells. Int. J. Mol. Sci. 2019, 20, 2918. [Google Scholar] [CrossRef] [PubMed]
- Kuang, G.; Zhang, Q.; He, S.; Liu, Y. Curcumin-loaded PEGylated mesoporous silica nanoparticles for effective photodynamic therapy. RSC Adv. 2020, 10, 24624–24630. [Google Scholar] [CrossRef]
- Yadav, Y.C.; Pattnaik, S.; Swain, K. Curcumin loaded mesoporous silica nanoparticles: Assessment of bioavailability and cardioprotective effect. Drug Dev. Ind. Pharm. 2019, 45, 1889–1895. [Google Scholar] [CrossRef] [PubMed]
- Mehmood, A.; Ghafar, H.; Yaqoob, S.; Gohar, U.F.; Ahmad, B. Mesoporous silica nanoparticles: A review. J. Dev. Drugs 2017, 6. [Google Scholar] [CrossRef]
- Crespilho, F.N.; Zucolotto, V.; Oliveira, O.N., Jr.; Nart, F.C. Electrochemistry of layer-by-layer films: A review. Int. J. Electrochem. Sci. 2006, 1, 194–214. [Google Scholar]
- Fischer, T.; Tschernig, T.; Drews, F.; Brix, K.; Meier, C.; Simon, M.; Kautenburger, R.; Schneider, M. siRNA delivery to macrophages using aspherical, nanostructured microparticles as delivery system for pulmonary administration. Eur. J. Pharm. Biopharm. 2021, 158, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Möhwald, M.; Pinnapireddy, S.R.; Wonnenberg, B.; Pourasghar, M.; Jurisic, M.; Jung, A.; Fink-Straube, C.; Tschernig, T.; Bakowsky, U.; Schneider, M. Aspherical, nanostructured microparticles for targeted gene delivery to alveolar macrophages. Adv. Healthc. Mater. 2017, 6, 1700478. [Google Scholar] [CrossRef]
- Carvalho, T.C.; Peters, J.I.; Williams, R.O., III. Influence of particle size on regional lung deposition—What evidence is there? Int. J. Pharm. 2011, 406, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sadozai, S.K.; Khan, S.A.; Karim, N.; Becker, D.; Steinbrück, N.; Gier, S.; Baseer, A.; Breinig, F.; Kickelbick, G.; Schneider, M. Ketoconazole-loaded PLGA nanoparticles and their synergism against Candida albicans when combined with silver nanoparticles. J. Drug Deliv. Sci. Technol. 2020, 56, 101574. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef]
- Kharat, M.; Du, Z.; Zhang, G.; McClements, D.J. Physical and chemical stability of curcumin in aqueous solutions and emulsions: Impact of pH, temperature, and molecular environment. J. Agric. Food Chem. 2017, 65, 1525–1532. [Google Scholar] [CrossRef]
- Hu, B.; Zhong, L.; Weng, Y.; Peng, L.; Huang, Y.; Zhao, Y.; Liang, X.-J. Therapeutic siRNA: State of the art. Signal Transduct. Target. Ther. 2020, 5, 1–25. [Google Scholar] [CrossRef]
- Durcan, N.; Murphy, C.; Cryan, S.-A. Inhalable siRNA: Potential as a therapeutic agent in the lungs. Mol. Pharm. 2008, 5, 559–566. [Google Scholar] [CrossRef]
- Mathaes, R.; Winter, G.; Besheer, A.; Engert, J. Influence of particle geometry and PEGylation on phagocytosis of particulate carriers. Int. J. Pharm. 2014, 465, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Kohler, D.A.L. Asymmetric Particles for Pulmonary Drug Delivery. Ph.D. Thesis, Naturwissenschaftlich-Technische Fakultät, Saarbrücken, Germany, 2010. [Google Scholar]
- Tracey, D.; Klareskog, L.; Sasso, E.H.; Salfeld, J.G.; Tak, P.P. Tumor necrosis factor antagonist mechanisms of action: A comprehensive review. Pharmacol. Ther. 2008, 117, 244–279. [Google Scholar] [CrossRef] [PubMed]
- Pham, K.N.; Fullston, D.; Sagoe-Crentsil, K. Surface charge modification of nano-sized silica colloid. Aust. J. Chem. 2007, 60, 662–666. [Google Scholar] [CrossRef]
- Xie, G.; Sun, J.; Zhong, G.; Shi, L.; Zhang, D. Biodistribution and toxicity of intravenously administered silica nanoparticles in mice. Arch. Toxicol. 2010, 84, 183–190. [Google Scholar] [CrossRef]
- Chenebault, P.; Schürenkämper, A. The Measurement of Small Surface Areas by the BET Adsorption Method1. J. Phys. Chem. 1965, 69, 2300–2305. [Google Scholar] [CrossRef]
- Kohler, D.; Schneider, M.; Krüger, M.; Lehr, C.M.; Möhwald, H.; Wang, D. Template-Assisted Polyelectrolyte Encapsulation of Nanoparticles into Dispersible, Hierarchically Nanostructured Microfibers. Adv. Mater. 2011, 23, 1376–1379. [Google Scholar] [CrossRef]
- Decher, G. Fuzzy nanoassemblies: Toward layered polymeric multicomposites. Science 1997, 277, 1232–1237. [Google Scholar] [CrossRef]
- Decher, G.; Hong, J.D. Buildup of Ultrathin Multilayer Films by a Self-Assembly Process, 1 Consecutive Adsorption of Anionic and Cationic Bipolar Amphiphiles on Charged Surfaces. Makromol. Chemie. Macromol. Symp. 1991, 46, 321–327. [Google Scholar] [CrossRef]
- Benhalima, T.; Ferfera-Harrar, H. Eco-friendly porous carboxymethyl cellulose/dextran sulfate composite beads as reusable and efficient adsorbents of cationic dye methylene blue. Int. J. Biol. Macromol. 2019, 132, 126–141. [Google Scholar] [CrossRef]
- Heo, R.; You, D.G.; Um, W.; Choi, K.Y.; Jeon, S.; Park, J.-S.; Choi, Y.; Kwon, S.; Kim, K.; Kwon, I.C. Dextran sulfate nanoparticles as a theranostic nanomedicine for rheumatoid arthritis. Biomaterials 2017, 131, 15–26. [Google Scholar] [CrossRef]
- Akinc, A.; Thomas, M.; Klibanov, A.M.; Langer, R. Exploring polyethylenimine-mediated DNA transfection and the proton sponge hypothesis. J. Gene Med. 2004, 7, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Marple, V.A.; Roberts, D.L.; Romay, F.J.; Miller, N.C.; Truman, K.G.; Van Oort, M.; Olsson, B.; Holroyd, M.J.; Mitchell, J.P.; Hochrainer, D. Next generation pharmaceutical impactor (a new impactor for pharmaceutical inhaler testing). Part I: Design. J. Aerosol Med. 2003, 16, 283–299. [Google Scholar] [CrossRef] [PubMed]
- Borgström, L. On the use of dry powder inhalers in situations perceived as constrained. J. Aerosol Med. 2001, 14, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Newman, S.P.; Morén, F.; Trofast, E.; Talaee, N.; Clarke, S.W. Terbutaline sulphate Turbuhaler: Effect of inhaled flow rate on drug deposition and efficacy. Int. J. Pharm. 1991, 74, 209–213. [Google Scholar] [CrossRef]
- Vehring, R. Pharmaceutical particle engineering via spray drying. Pharm. Res. 2008, 25, 999–1022. [Google Scholar] [CrossRef]
- Aquino, R.; Prota, L.; Auriemma, G.; Santoro, A.; Mencherini, T.; Colombo, G.; Russo, P. Dry powder inhalers of gentamicin and leucine: Formulation parameters, aerosol performance and in vitro toxicity on CuFi1 cells. Int. J. Pharm. 2012, 426, 100–107. [Google Scholar] [CrossRef]
- Seville, P.C.; Learoyd, T.; Li, H.-Y.; Williamson, I.; Birchall, J.C. Amino acid-modified spray-dried powders with enhanced aerosolisation properties for pulmonary drug delivery. Powder Technol. 2007, 178, 40–50. [Google Scholar] [CrossRef]
- Feng, A.; Boraey, M.; Gwin, M.; Finlay, P.; Kuehl, P.; Vehring, R. Mechanistic models facilitate efficient development of leucine containing microparticles for pulmonary drug delivery. Int. J. Pharm. 2011, 409, 156–163. [Google Scholar] [CrossRef]
- Abdelrahim, M.E.; Chrystyn, H. Aerodynamic characteristics of nebulized terbutaline sulphate using the next generation impactor (NGI) and CEN method. J. Aerosol Med. Pulm. Drug Deliv. 2009, 22, 19–28. [Google Scholar] [CrossRef]
- Lababidi, N.; Kissi, E.O.; Elgaher, W.A.; Sigal, V.; Haupenthal, J.; Schwarz, B.C.; Hirsch, A.K.; Rades, T.; Schneider, M. Spray-drying of inhalable, multifunctional formulations for the treatment of biofilms formed in cystic fibrosis. J. Control. Release 2019, 314, 62–71. [Google Scholar] [CrossRef]
- Torge, A.; Pavone, G.; Jurisic, M.; Lima-Engelmann, K.; Schneider, M. A comparison of spherical and cylindrical microparticles composed of nanoparticles for pulmonary application. Aerosol Sci. Technol. 2019, 53, 53–62. [Google Scholar] [CrossRef]
- Lada, A.T.; Rudel, L.L.; Clair, R.W.S. Effects of LDL enriched with different dietary fatty acids on cholesteryl ester accumulation and turnover in THP-1 macrophages. J. Lipid Res. 2003, 44, 770–779. [Google Scholar] [CrossRef]
- Foster, K.A.; Oster, C.G.; Mayer, M.M.; Avery, M.L.; Audus, K.L. Characterization of the A549 cell line as a type II pulmonary epithelial cell model for drug metabolism. Exp. Cell Res. 1998, 243, 359–366. [Google Scholar] [CrossRef]
- Stockert, J.C.; Horobin, R.W.; Colombo, L.L.; Blázquez-Castro, A. Tetrazolium salts and formazan products in Cell Biology: Viability assessment, fluorescence imaging, and labeling perspectives. Acta Histochem. 2018, 120, 159–167. [Google Scholar] [CrossRef]
- Fu, Y.; Kao, W.J. Drug release kinetics and transport mechanisms of non-degradable and degradable polymeric delivery systems. Expert Opin. Drug Deliv. 2010, 7, 429–444. [Google Scholar] [CrossRef] [PubMed]
- Stefaniak, A.; Guilmette, R.; Day, G.; Hoover, M.; Breysse, P.N.; Scripsick, R. Characterization of phagolysosomal simulant fluid for study of beryllium aerosol particle dissolution. Toxicol. Vitr. 2005, 19, 123–134. [Google Scholar] [CrossRef]
- Barbas, C.F.; Burton, D.R.; Scott, J.K.; Silverman, G.J. Quantitation of DNA and RNA. Cold Spring Harb. Protoc. 2007. [Google Scholar] [CrossRef] [PubMed]
- Lequin, R.M. Enzyme immunoassay (EIA)/enzyme-linked immunosorbent assay (ELISA). Clin. Chem. 2005, 51, 2415–2418. [Google Scholar] [CrossRef]
- Sun, J.; Shigemi, H.; Cao, M.; Qin, E.; Tang, J.; Shen, J.; Iwasaki, H. Minocycline induces autophagy and inhibits cell proliferation in LPS-stimulated THP-1 cells. BioMed Res. Int. 2020, 2020, 5459209. [Google Scholar] [CrossRef] [PubMed]
- Clay, C.S.; Peace, G.W. Ion beam sputtering: An improved method of metal coating SEM samples and shadowing CTEM samples. J. Microsc. 1981, 123, 25–34. [Google Scholar] [CrossRef]
- Jeelani, P.G.; Mulay, P.; Venkat, R.; Ramalingam, C. Multifaceted application of silica nanoparticles. A review. Silicon 2020, 12, 1337–1354. [Google Scholar] [CrossRef]
- Hurtado, Y.; Beltrán, C.; Zabala, R.D.; Lopera, S.H.; Franco, C.A.; Nassar, N.N.; Cortés, F.B. Effects of surface acidity and polarity of SiO2 nanoparticles on the foam stabilization applied to natural gas flooding in tight gas-condensate reservoirs. Energy Fuels 2018, 32, 5824–5833. [Google Scholar] [CrossRef]
- Möller, J.; Luehmann, T.; Hall, H.; Vogel, V. The race to the pole: How high-aspect ratio shape and heterogeneous environments limit phagocytosis of filamentous Escherichia coli bacteria by macrophages. Nano Lett. 2012, 12, 2901–2905. [Google Scholar] [CrossRef] [PubMed]
- Champion, J.A.; Mitragotri, S. Role of target geometry in phagocytosis. Proc. Natl. Acad. Sci. USA 2006, 103, 4930–4934. [Google Scholar] [CrossRef]
- Geiser, M.; Kreyling, W.G. Deposition and biokinetics of inhaled nanoparticles. Part. Fibre Toxicol. 2010, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Finlay, W.; Stapleton, K.; Zuberbuhler, P. Fine particle fraction as a measure of mass depositing in the lung during inhalation of nearly isotonic nebulized aerosols. J. Aerosol Sci. 1997, 28, 1301–1309. [Google Scholar] [CrossRef]
- Van Holsbeke, C.; Marshall, J.; De Backer, J.; Vos, W. Median mass aerodynamic diameter (MMAD) and fine particle fraction (FPF): Influence on lung deposition? Eur. Respir. J. 2014, 44, P912. [Google Scholar]
- De Boer, A.H.; Hagedoorn, P.; Hoppentocht, M.; Buttini, F.; Grasmeijer, F.; Frijlink, H.W. Dry powder inhalation: Past, present and future. Expert Opin. Drug Deliv. 2017, 14, 499–512. [Google Scholar] [CrossRef]
- DeCarlo, P.F.; Slowik, J.G.; Worsnop, D.R.; Davidovits, P.; Jimenez, J.L. Particle morphology and density characterization by combined mobility and aerodynamic diameter measurements. Part 1: Theory. Aerosol Sci. Technol. 2004, 38, 1185–1205. [Google Scholar] [CrossRef]
- Hinds, W.C. Aerosol Technology: Properties, Behavior, and Measurement of Airborne Particles; John Wiley & Sons: New York, NY, USA, 1999. [Google Scholar]
- Sturm, R. Nanotubes in the human respiratory tract–deposition modeling. Z. Für Med. Phys. 2015, 25, 135–145. [Google Scholar] [CrossRef]
- Darquenne, C.; Fleming, J.S.; Katz, I.; Martin, A.R.; Schroeter, J.; Usmani, O.S.; Venegas, J.; Schmid, O. Bridging the gap between science and clinical efficacy: Physiology, imaging, and modeling of aerosols in the lung. J. Aerosol Med. Pulm. Drug Deliv. 2016, 29, 107–126. [Google Scholar] [CrossRef]
- Heyder, J. Deposition of inhaled particles in the human respiratory tract and consequences for regional targeting in respiratory drug delivery. Proc. Am. Thorac. Soc. 2004, 1, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Slika, L.; Patra, D. A short review on chemical properties, stability and nano-technological advances for curcumin delivery. Expert Opin. Drug Deliv. 2020, 17, 61–75. [Google Scholar] [CrossRef]
- Aderem, A.; Underhill, D.M. Mechanisms of phagocytosis in macrophages. Annu. Rev. Immunol. 1999, 17, 593–623. [Google Scholar] [CrossRef] [PubMed]
- Alkekhia, D.; Hammond, P.T.; Shukla, A. Layer-by-layer biomaterials for drug delivery. Annu. Rev. Biomed. Eng. 2020, 22, 1–24. [Google Scholar] [CrossRef]
- Wood, K.C.; Boedicker, J.Q.; Lynn, D.M.; Hammond, P.T. Tunable drug release from hydrolytically degradable layer-by-layer thin films. Langmuir 2005, 21, 1603–1609. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, Q.; Han, N.; Bai, L.; Li, J.; Liu, J.; Che, E.; Hu, L.; Zhang, Q.; Jiang, T. Mesoporous silica nanoparticles in drug delivery and biomedical applications. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 313–327. [Google Scholar] [CrossRef] [PubMed]
- Hassanzadeh, S.; Read, M.I.; Bland, A.R.; Majeed, M.; Jamialahmadi, T.; Sahebkar, A. Curcumin: An inflammasome silencer. Pharmacol. Res. 2020, 159, 104921. [Google Scholar] [CrossRef]
- Tang, Z.; Wang, Y.; Podsiadlo, P.; Kotov, N.A. Biomedical applications of layer-by-layer assembly: From biomimetics to tissue engineering. Adv. Mater. 2006, 18, 3203–3224. [Google Scholar] [CrossRef]
- Sajid, M.I.; Moazzam, M.; Kato, S.; Yeseom Cho, K.; Tiwari, R.K. Overcoming Barriers for siRNA Therapeutics: From Bench to Bedside. Pharmaceuticals 2020, 13, 294. [Google Scholar] [CrossRef] [PubMed]
- Soni, D.M. Strategies and Approaches for siRNA Delivery. J. Drug Deliv. Ther. 2020, 10, 239–250. [Google Scholar] [CrossRef]
- Champion, J.A.; Katare, Y.K.; Mitragotri, S. Particle shape: A new design parameter for micro-and nanoscale drug delivery carriers. J. Control. Release 2007, 121, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Pittella, F.; Abcs, G.A.V.; Acm, G. Short Interfering RNA (siRNA) based Medicines and the Future of RNAi Therapy: A Mini Review. Curr. Trends Biomed. Eng. Biosci. 2020, 19, 116–121. [Google Scholar]
- Hibbitts, A.J.; Ramsey, J.M.; Barlow, J.; MacLoughlin, R.; Cryan, S.-A. In Vitro and In Vivo Assessment of PEGylated PEI for Anti-IL-8/CxCL-1 siRNA Delivery to the Lungs. Nanomaterials 2020, 10, 1248. [Google Scholar] [CrossRef] [PubMed]

| Unmodified mSNPs | APTS Modified mSNPs | Rhodamine B + APTS Modified mSNPs | |
|---|---|---|---|
| Hydr. Diameter (nm) | 262.13 ± 3.98 | 263.23 ± 1.33 | 245.8 ± 2.97 |
| PDI | 0.102 ± 0.013 | 0.045 ± 0.022 | 0.084 ± 0.068 |
| Zeta potential (mV) | −40.03 ± 0.80 | 36.07 ± 0.57 | 22.7 ± 1.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fischer, T.; Winter, I.; Drumm, R.; Schneider, M. Cylindrical Microparticles Composed of Mesoporous Silica Nanoparticles for the Targeted Delivery of a Small Molecule and a Macromolecular Drug to the Lungs: Exemplified with Curcumin and siRNA. Pharmaceutics 2021, 13, 844. https://doi.org/10.3390/pharmaceutics13060844
Fischer T, Winter I, Drumm R, Schneider M. Cylindrical Microparticles Composed of Mesoporous Silica Nanoparticles for the Targeted Delivery of a Small Molecule and a Macromolecular Drug to the Lungs: Exemplified with Curcumin and siRNA. Pharmaceutics. 2021; 13(6):844. https://doi.org/10.3390/pharmaceutics13060844
Chicago/Turabian StyleFischer, Thorben, Inga Winter, Robert Drumm, and Marc Schneider. 2021. "Cylindrical Microparticles Composed of Mesoporous Silica Nanoparticles for the Targeted Delivery of a Small Molecule and a Macromolecular Drug to the Lungs: Exemplified with Curcumin and siRNA" Pharmaceutics 13, no. 6: 844. https://doi.org/10.3390/pharmaceutics13060844
APA StyleFischer, T., Winter, I., Drumm, R., & Schneider, M. (2021). Cylindrical Microparticles Composed of Mesoporous Silica Nanoparticles for the Targeted Delivery of a Small Molecule and a Macromolecular Drug to the Lungs: Exemplified with Curcumin and siRNA. Pharmaceutics, 13(6), 844. https://doi.org/10.3390/pharmaceutics13060844