Bactericidal Properties of Rod-, Peanut-, and Star-Shaped Gold Nanoparticles Coated with Ceragenin CSA-131 against Multidrug-Resistant Bacterial Strains
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains, Media, and Growth Conditions
2.2. Antibacterial Compounds
2.2.1. Ceragenin (CSA-131)
2.2.2. Gold Nanoparticles (Au NPs) Functionalized by CSA-131
2.2.3. Physicochemical Properties of AuR NPs@CSA-131, AuP NPs@CSA-131 and AuS NPs@CSA-131 Nanoparticles
2.3. Estimation of Antibacterial Activity of the Nanosystems
2.3.1. Antimicrobial Susceptibility Testing
2.3.2. Killing Assay
2.4. Analysis of the Bacterial Response to Tested Nanosystems at Molecular Level
2.5. Haemolytic Activity of CSA-131 Nanosystems
2.6. Statistical Analysis
3. Results
3.1. Physicochemical Nature of Rod-, Peanut-, and Star-Shaped Au NPs
3.2. Antimicrobial Activity of the Rod-, Peanut-, and Star- Shaped Au NPs
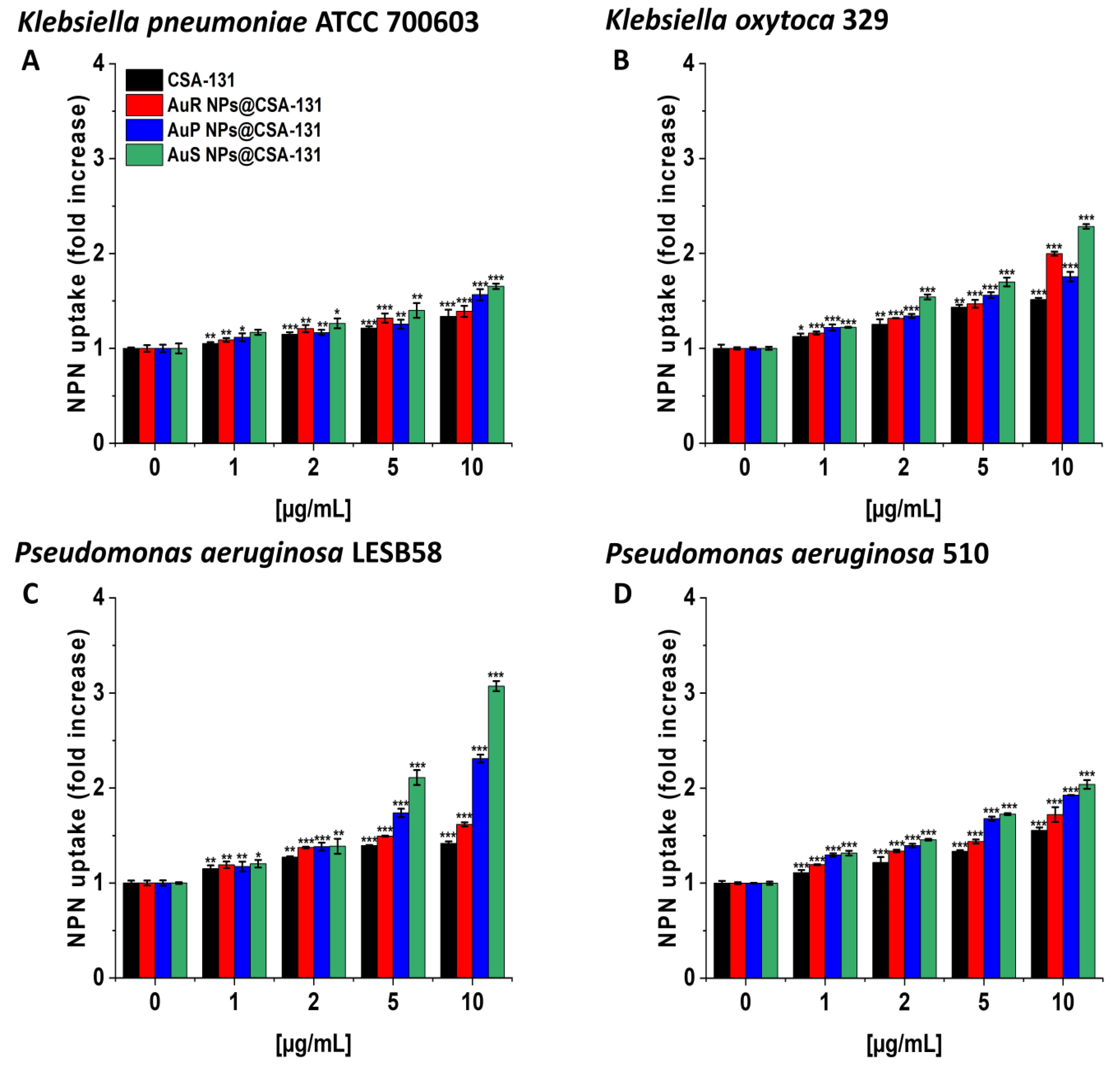
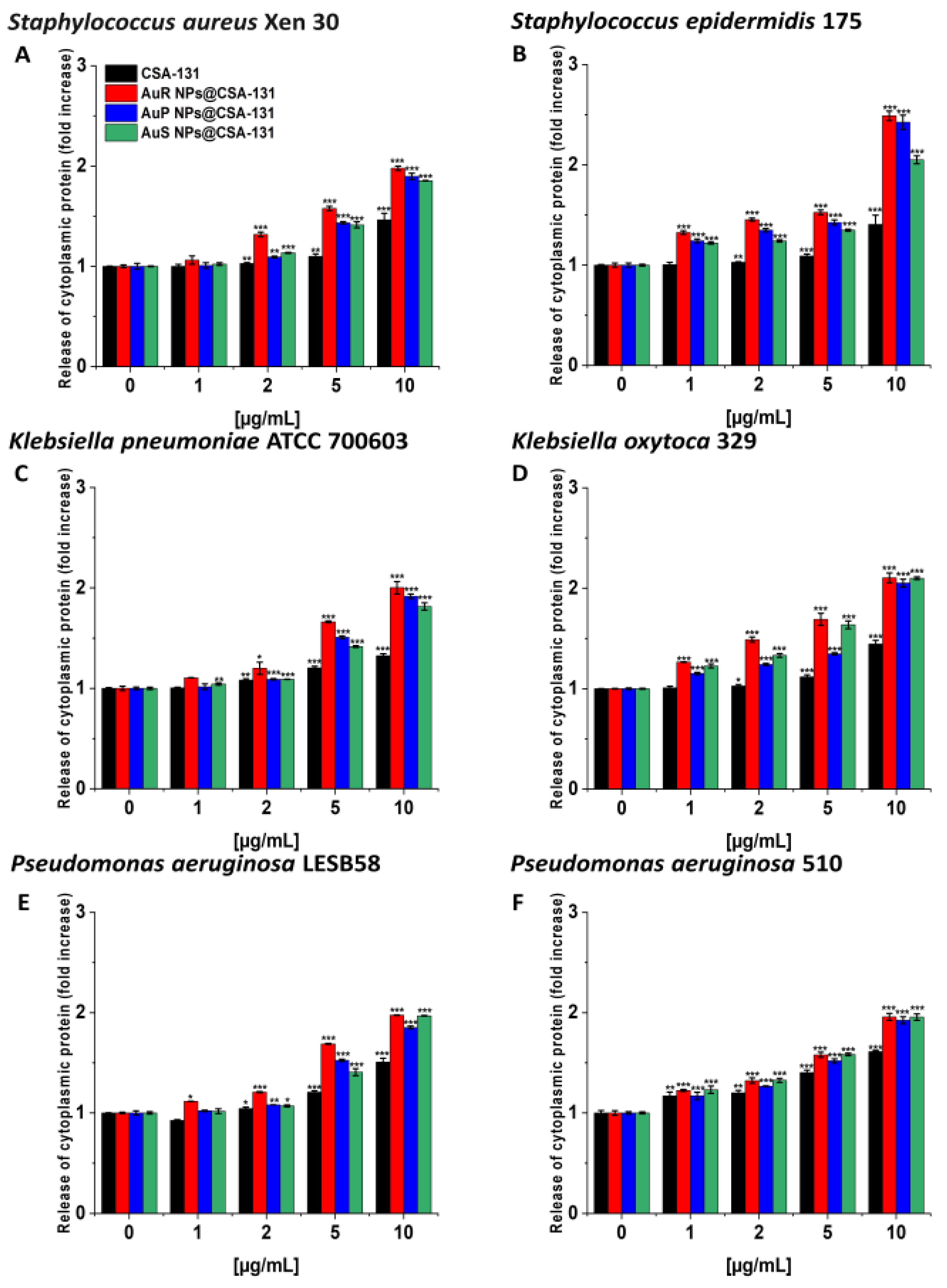
3.3. Bactericidal Mechanism of the Nanosystems Involves the Induction of Oxidative Stress as well as the Destruction and Depolarization of Bacterial Membranes and Protein Leakage
3.4. The Nanosystems Exert High Biocompatibility at Bactericidal Doses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- David, A.A.; Park, S.E.; Parang, K.; Tiwari, R.K. Antibiotics-Peptide Conjugates Against Multidrug-resistant Bacterial Pathogens. Curr. Top. Med. Chem. 2018, 18, 1926–1936. [Google Scholar] [CrossRef]
- Tao, C. Antimicrobial activity and toxicity of gold nanoparticles: Research progress, challenges and prospects. Lett. Appl. Microbiol. 2018, 67, 537–543. [Google Scholar] [CrossRef] [PubMed]
- New Report Calls for Urgent Action to Avert Antimicrobial Resistance Crisis. Available online: https://www.who.int/news-room/detail/29-04-2019-new-report-calls-for-urgent-action-to-avert-antimicrobial-resistance-crisis (accessed on 4 March 2021).
- Munir, M.U.; Ahmed, A.; Usman, M.; Salman, S. Recent Advances in Nanotechnology-Aided Materials in Combating Microbial Resistance and Functioning as Antibiotics Substitutes. Int. J. Nanomed. 2020, 15, 7329–7358. [Google Scholar] [CrossRef]
- Biggest Threats and Data, 2019 AR Threats Report. 2019. Available online: https://www.cdc.gov/drugresistance/biggest-threats.html (accessed on 27 July 2020).
- Shang, W.; Rao, Y.; Zheng, Y.; Yang, Y.; Hu, Q.; Hu, Z.; Yuan, J.; Peng, H.; Xiong, K.; Tan, L.; et al. β-Lactam Antibiotics Enhance the Pathogenicity of Methicillin-Resistant Staphylococcus aureus via SarA-Controlled Lipoprotein-Like Cluster Expression. mBio 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Hassoun, A.; Linden, P.K.; Friedman, B. Incidence, prevalence, and management of MRSA bacteremia across patient populations-a review of recent developments in MRSA management and treatment. Crit. Care 2017, 21, 211. [Google Scholar] [CrossRef]
- Antibiotics Resistance Threats in the United States 2019. Available online: https://www.cdc.gov/drugresistance/pdf/threats-report/mrsa-508.pdf (accessed on 4 March 2021).
- Pryor, R.; Viola-Luqa, C.; Hess, O.; Bearman, G. Barrier Precautions in the Era of Multidrug Pathogens. Curr. Treat. Options Infect. Dis. 2020, 12, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Surveillance Atlas of Infectious Disease Klebsiella pneumoniae. Available online: https://atlas.ecdc.europa.eu/public/index.aspx?Dataset=27&HealthTopic=4 (accessed on 4 March 2021).
- Özer, T.T. The Rate of Inducible MLSB Resistance in the Methicillin-Resistant Staphylococci Isolated from Clinical Samples. J. Clin. Lab. Anal. 2016, 30, 490–493. [Google Scholar] [CrossRef]
- Tamma, P.D.; Aitken, S.L.; Bonomo, R.A.; Mathers, A.J.; van Duin, D.; Clancy, C.J. Infectious Diseases Society of America Antimicrobial Resistant Treatment Guidance: Gram-Negative Bacterial Infections. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Rekomendacje Doboru Testów do Oznaczania Wrażliwości Bakterii na Antybiotyki i Chemioterapeutyki 2009. Oznaczanie Wrażliwości Pałeczek Gram-Ujemnych. Available online: https://korld.nil.gov.pl/pdf/02-Rek2009-Paleczki_z_rodziny_Enterobacteriaceae.pdf (accessed on 4 March 2021).
- Bitsori, M.; Galanakis, E. Treatment of Urinary Tract Infections Caused by ESBL-producing Escherichia coli or Klebsiella pneumoniae. Pediatr. Infect. Dis. J. 2019, 38, e332–e335. [Google Scholar] [CrossRef] [PubMed]
- Horcajada, J.P.; Montero, M.; Oliver, A.; Sorlí, L.; Luque, S.; Gómez-Zorrilla, S.; Benito, N.; Grau, S. Epidemiology and Treatment of Multidrug-Resistant and Extensively Drug-Resistant Pseudomonas aeruginosa Infections. Clin. Microbiol. Rev. 2019, 32. [Google Scholar] [CrossRef]
- Bucki, R.; Durnaś, B.; Wątek, M.; Piktel, E.; Cruz, K.; Wolak, P.; Savage, P.B.; Janmey, P.A. Targeting polyelectrolyte networks in purulent body fluids to modulate bactericidal properties of some antibiotics. Infect. Drug Resist. 2018, 11, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Surveillance Atlas of Infectious Disease Pseudomonas aeruginosa. Available online: https://atlas.ecdc.europa.eu/public/index.aspx?Dataset=27&HealthTopic=4 (accessed on 4 March 2021).
- Raman, G.; Avendano, E.E.; Chan, J.; Merchant, S.; Puzniak, L. Risk factors for hospitalized patients with resistant or multidrug-resistant. Antimicrob. Resist. Infect. Control 2018, 7, 79. [Google Scholar] [CrossRef]
- Zheng, Y.; Liu, W.; Qin, Z.; Chen, Y.; Jiang, H.; Wang, X. Mercaptopyrimidine-Conjugated Gold Nanoclusters as Nanoantibiotics for Combating Multidrug-Resistant Superbugs. Bioconjugate Chem. 2018, 29, 3094–3103. [Google Scholar] [CrossRef]
- Xie, Y.; Liu, Y.; Yang, J.; Hu, F.; Zhu, K.; Jiang, X. Gold Nanoclusters for Targeting Methicillin-Resistant Staphylococcus aureus In Vivo. Angew. Chem. Int. Ed. Engl. 2018, 57, 3958–3962. [Google Scholar] [CrossRef]
- Shaikh, S.; Nazam, N.; Rizvi, S.M.D.; Ahmad, K.; Baig, M.H.; Lee, E.J.; Choi, I. Mechanistic Insights into the Antimicrobial Actions of Metallic Nanoparticles and Their Implications for Multidrug Resistance. Int. J. Mol. Sci. 2019, 20, 2468. [Google Scholar] [CrossRef]
- Mohamed, M.M.; Fouad, S.A.; Elshoky, H.A.; Mohammed, G.M.; Salaheldin, T.A. Antibacterial effect of gold nanoparticles against. Int. J. Vet. Sci. Med. 2017, 5, 23–29. [Google Scholar] [CrossRef]
- Wang, L.; Hu, C.; Shao, L. The antimicrobial activity of nanoparticles: Present situation and prospects for the future. Int. J. Nanomed. 2017, 12, 1227–1249. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, T.; Chatterjee, B.K.; Chakrabarti, P. Modelling of growth kinetics of Vibrio cholerae in presence of gold nanoparticles: Effect of size and morphology. Sci. Rep. 2017, 7, 9671. [Google Scholar] [CrossRef]
- Hemeg, H.A. Nanomaterials for alternative antibacterial therapy. Int. J. Nanomed. 2017, 12, 8211–8225. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.; Misba, L.; Khan, A.U. Nano-therapeutics: A revolution in infection control in post antibiotic era. Nanomedicine 2017, 13, 2281–2301. [Google Scholar] [CrossRef] [PubMed]
- Penders, J.; Stolzoff, M.; Hickey, D.J.; Andersson, M.; Webster, T.J. Shape-dependent antibacterial effects of non-cytotoxic gold nanoparticles. Int. J. Nanomed. 2017, 12, 2457–2468. [Google Scholar] [CrossRef]
- Terracciano, R.; Sprouse, M.L.; Wang, D.; Ricchetti, S.; Hirsch, M.; Ferrante, N.; Butler, B.; Demarchi, D.; Grattoni, A.; Filgueira, C.S. Intratumoral Gold Nanoparticle-Enhanced CT Imaging: An in Vivo Investigation of Biodistribution and Retention. In Proceedings of the Virtual 2020 IEEE 20th International Conference on Nanotechnology (IEEE-NANO), Montreal, QC, Canada, 28–31 July 2020. [Google Scholar]
- Choi, M.R.; Stanton-Maxey, K.J.; Stanley, J.K.; Levin, C.S.; Bardhan, R.; Akin, D.; Badve, S.; Sturgis, J.; Robinson, J.P.; Bashir, R.; et al. A cellular Trojan Horse for delivery of therapeutic nanoparticles into tumors. Nano Lett. 2007, 7, 3759–3765. [Google Scholar] [CrossRef] [PubMed]
- León-Buitimea, A.; Garza-Cárdenas, C.R.; Garza-Cervantes, J.A.; Lerma-Escalera, J.A.; Morones-Ramírez, J.R. The Demand for New Antibiotics: Antimicrobial Peptides, Nanoparticles, and Combinatorial Therapies as Future Strategies in Antibacterial Agent Design. Front. Microbiol. 2020, 11, 1669. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.Y.; Ko, W.C.; Hsueh, P.R. Nanoparticles in the Treatment of Infections Caused by Multidrug-Resistant Organisms. Front. Pharmacol. 2019, 10, 1153. [Google Scholar] [CrossRef]
- Linklater, D.P.; Baulin, V.A.; Le Guével, X.; Fleury, J.B.; Hanssen, E.; Nguyen, T.H.P.; Juodkazis, S.; Bryant, G.; Crawford, R.J.; Stoodley, P.; et al. Antibacterial Action of Nanoparticles by Lethal Stretching of Bacterial Cell Membranes. Adv. Mater. 2020, 32, e2005679. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt-Guzel, C.; Hacioglu, M.; Savage, P.B. Investigation of the in vitro antifungal and antibiofilm activities of ceragenins CSA-8, CSA-13, CSA-44, CSA-131, and CSA-138 against Candida species. Diagn. Microbiol. Infect. Dis. 2018, 91, 324–330. [Google Scholar] [CrossRef]
- Hacioglu, M.; Haciosmanoglu, E.; Birteksoz-Tan, A.S.; Bozkurt-Guzel, C.; Savage, P.B. Effects of ceragenins and conventional antimicrobials on Candida albicans and Staphylococcus aureus mono and multispecies biofilms. Diagn. Microbiol. Infect. Dis. 2019, 95, 114863. [Google Scholar] [CrossRef] [PubMed]
- Chmielewska, S.J.; Skłodowski, K.; Piktel, E.; Suprewicz, Ł.; Fiedoruk, K.; Daniluk, T.; Wolak, P.; Savage, P.B.; Bucki, R. NDM-1 Carbapenemase-Producing Enterobacteriaceae are Highly Susceptible to Ceragenins CSA-13, CSA-44, and CSA-131. Infect. Drug Resist. 2020, 13, 3277–3294. [Google Scholar] [CrossRef]
- Güzel, Ç.B.; Avci, N.M.; Savage, P. Activities of the Cationic Steroid Antibiotics CSA-13, CSA-131, CSA-138, CSA-142, and CSA-192 Against Carbapenem-resistant. Turk. J. Pharm. Sci. 2020, 17, 63–67. [Google Scholar] [CrossRef]
- Vila-Farrés, X.; Callarisa, A.E.; Gu, X.; Savage, P.B.; Giralt, E.; Vila, J. CSA-131, a ceragenin active against colistin-resistant Acinetobacter baumannii and Pseudomonas aeruginosa clinical isolates. Int. J. Antimicrob. Agents 2015, 46, 568–571. [Google Scholar] [CrossRef]
- Hashemi, M.M.; Rovig, J.; Weber, S.; Hilton, B.; Forouzan, M.M.; Savage, P.B. Susceptibility of Colistin-Resistant, Gram-Negative Bacteria to Antimicrobial Peptides and Ceragenins. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Wnorowska, U.; Piktel, E.; Durnaś, B.; Fiedoruk, K.; Savage, P.B.; Bucki, R. Use of ceragenins as a potential treatment for urinary tract infections. BMC Infect. Dis. 2019, 19, 369. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, M.M.; Holden, B.S.; Taylor, M.F.; Wilson, J.; Coburn, J.; Hilton, B.; Nance, T.; Gubler, S.; Genberg, C.; Deng, S.; et al. Antibacterial and Antifungal Activities of Poloxamer Micelles Containing Ceragenin CSA-131 on Ciliated Tissues. Molecules 2018, 23, 596. [Google Scholar] [CrossRef] [PubMed]
- Oyardi, Ö.; Savage, P.B.; Erturan, Z.; Bozkurt-Guzel, C. In vitro assessment of CSA-131 and CSA-131 poloxamer form for the treatment of Stenotrophomonas maltophilia infections in cystic fibrosis. J. Antimicrob. Chemother. 2021, 76, 443–450. [Google Scholar] [CrossRef]
- Favi, P.M.; Valencia, M.M.; Elliott, P.R.; Restrepo, A.; Gao, M.; Huang, H.; Pavon, J.J.; Webster, T.J. Shape and surface chemistry effects on the cytotoxicity and cellular uptake of metallic nanorods and nanospheres. J. Biomed. Mater. Res. A 2015, 103, 3940–3955. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.N.; Smith, K.; Samuels, T.A.; Lu, J.; Obare, S.O.; Scott, M.E. Nanoparticles functionalized with ampicillin destroy multiple-antibiotic-resistant isolates of Pseudomonas aeruginosa and Enterobacter aerogenes and methicillin-resistant Staphylococcus aureus. Appl. Environ. Microbiol. 2012, 78, 2768–2774. [Google Scholar] [CrossRef]
- Depciuch, J.; Miszczyk, J.; Maximenko, A.; Zieliński, P.M.; Rawojć, K.; Panek, A.; Olko, P.; Parlińska-Wojtan, M. Gold Nanopeanuts as Prospective Support for Cisplatin in Glioblastoma Nano-Chemo-Radiotherapy. Int. J. Mol. Sci. 2020, 21, 9082. [Google Scholar] [CrossRef] [PubMed]
- Brady, P.N.; Macnaughtan, M.A. Evaluation of colorimetric assays for analyzing reductively methylated proteins: Biases and mechanistic insights. Anal. Biochem. 2015, 491, 43–51. [Google Scholar] [CrossRef]
- Goldring, J.P.D. Measuring Protein Concentration with Absorbance, Lowry, Bradford Coomassie Blue, or the Smith Bicinchoninic Acid Assay Before Electrophoresis. Methods Mol. Biol. 2019, 1855, 31–39. [Google Scholar] [CrossRef]
- Guo, G.Y.; Ebert, H. First-principles study of the magnetic hyperfine field in Fe and Co multilayers. Phys. Rev. B Condens. Matter. 1996, 53, 2492–2503. [Google Scholar] [CrossRef]
- Levin, C.S.; Janesko, B.G.; Bardhan, R.; Scuseria, G.E.; Hartgerink, J.D.; Halas, N.J. Chain-length-dependent vibrational resonances in alkanethiol self-assembled monolayers observed on plasmonic nanoparticle substrates. Nano Lett. 2006, 6, 2617–2621. [Google Scholar] [CrossRef]
- Bazylewski, P.; Dibigalpitiya, R.; Fanchini, G. In situ Raman spectroscopy distinguishes between reversible and inrreversible thiol modifications in L-cysteine. RSC Adv. 2017, 5, 2964–2970. [Google Scholar] [CrossRef]
- Deręgowska, A.; Depciuch, J.; Wojnaowska, R.; Polit, J.; Broda, D.; Nechai, H.G.M.; Sheregii, E. Study of optical properties of a glutathione capped gold nanoparticles using linker (MHDA) by Fourier transform infra red spectroscopy and surface enhanced Raman scattering. IJSRIT 2013, 7, 80–83. [Google Scholar]
- Niemirowicz-Laskowska, K.; Głuszek, K.; Piktel, E.; Pajuste, K.; Durnaś, B.; Król, G.; Wilczewska, A.Z.; Janmey, P.A.; Plotniece, A.; Bucki, R. Bactericidal and immunomodulatory properties of magnetic nanoparticles functionalized by 1,4-dihydropyridines. Int. J. Nanomed. 2018, 13, 3411–3424. [Google Scholar] [CrossRef]
- Wnorowska, U.; Fiedoruk, K.; Piktel, E.; Prasad, S.V.; Sulik, M.; Janion, M.; Daniluk, T.; Savage, P.B.; Bucki, R. Nanoantibiotics containing membrane-active human cathelicidin LL-37 or synthetic ceragenins attached to the surface of magnetic nanoparticles as novel and innovative therapeutic tools: Current status and potential future applications. J. Nanobiotechnology 2020, 18, 3. [Google Scholar] [CrossRef]
- Makhdoumi, P.; Karimi, H.; Khazaei, M. Review on Metal-Based Nanoparticles: Role of Reactive Oxygen Species in Renal Toxicity. Chem. Res. Toxicol. 2020, 33, 2503–2514. [Google Scholar] [CrossRef]
- Masri, A.; Anwar, A.; Khan, N.A.; Siddiqui, R. The Use of Nanomedicine for Targeted Therapy against Bacterial Infections. Antibiotics 2019, 8, 260. [Google Scholar] [CrossRef]
- Niemirowicz, K.; Piktel, E.; Wilczewska, A.Z.; Markiewicz, K.H.; Durnaś, B.; Wątek, M.; Puszkarz, I.; Wróblewska, M.; Niklińska, W.; Savage, P.B.; et al. Core-shell magnetic nanoparticles display synergistic antibacterial effects against. Int. J. Nanomed. 2016, 11, 5443–5455. [Google Scholar] [CrossRef]
- Pajerski, W.; Ochonska, D.; Brzychczy-Wloch, M.; Indyka, P.; Jarosz, M.; Golda-Cepa, M.; Sojka, Z.; Kotarba, A. Attachment efficiency of gold nanoparticles by Gram-positive and Gram-negative bacterial strains governed by surface charges. J. Nanoparticle Res. 2019, 21. [Google Scholar] [CrossRef]
- Bu, T.; Jia, P.; Liu, J.; Liu, Y.; Sun, X.; Zhang, M.; Tian, Y.; Zhang, D.; Wang, J.; Wang, L. Diversely positive-charged gold nanoparticles based biosensor: A label-free and sensitive tool for foodborne pathogen detection. Food Chem. X 2019, 3, 100052. [Google Scholar] [CrossRef] [PubMed]
- Shaker, M.A.; Shaaban, M.I. Formulation of carbapenems loaded gold nanoparticles to combat multi-antibiotic bacterial resistance: In vitro antibacterial study. Int. J. Pharm. 2017, 525, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Yetisgin, A.A.; Cetinel, S.; Zuvin, M.; Kosar, A.; Kutlu, O. Therapeutic Nanoparticles and Their Targeted Delivery Applications. Molecules 2020, 25, 2193. [Google Scholar] [CrossRef]
- Niemirowicz, K.; Surel, U.; Wilczewska, A.Z.; Mystkowska, J.; Piktel, E.; Gu, X.; Namiot, Z.; Kułakowska, A.; Savage, P.B.; Bucki, R. Bactericidal activity and biocompatibility of ceragenin-coated magnetic nanoparticles. J. Nanobiotechnology 2015, 13, 32. [Google Scholar] [CrossRef]
- Durnaś, B.; Piktel, E.; Wątek, M.; Wollny, T.; Góźdź, S.; Smok-Kalwat, J.; Niemirowicz, K.; Savage, P.B.; Bucki, R. Anaerobic bacteria growth in the presence of cathelicidin LL-37 and selected ceragenins delivered as magnetic nanoparticles cargo. BMC Microbiol. 2017, 17, 167. [Google Scholar] [CrossRef]
- Piktel, E.; Suprewicz, Ł.; Depciuch, J.; Cieśluk, M.; Chmielewska, S.; Durnaś, B.; Król, G.; Wollny, T.; Deptuła, P.; Kochanowicz, J.; et al. Rod-shaped gold nanoparticles exert potent candidacidal activity and decrease the adhesion of fungal cells. Nanomedicine 2020, 15, 2733–2752. [Google Scholar] [CrossRef] [PubMed]
- Piktel, E.; Ościłowska, I.; Suprewicz, Ł.; Depciuch, J.; Marcińczyk, N.; Chabielska, E.; Wolak, P.; Wollny, T.; Janion, M.; Parlinska-Wojtan, M.; et al. ROS-mediated apoptosis and autophagy in ovarian cancer cells treated with peanut-shaped gold nanoparticles. Int. J. Nanomed. 2021, 2021, 1993–2011. [Google Scholar] [CrossRef]
- Hameed, S.; Wang, Y.; Zhao, L.; Xie, L.; Ying, Y. Shape-dependent significant physical mutilation and antibacterial mechanisms of gold nanoparticles against foodborne bacterial pathogens (Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus) at lower concentrations. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 108, 110338. [Google Scholar] [CrossRef]
- Niemirowicz, K.; Durnaś, B.; Tokajuk, G.; Piktel, E.; Michalak, G.; Gu, X.; Kułakowska, A.; Savage, P.B.; Bucki, R. Formulation and candidacidal activity of magnetic nanoparticles coated with cathelicidin LL-37 and ceragenin CSA-13. Sci. Rep. 2017, 7, 4610. [Google Scholar] [CrossRef]
- Rajchakit, U.; Sarojini, V. Recent Developments in Antimicrobial-Peptide-Conjugated Gold Nanoparticles. Bioconjugate Chem. 2017, 28, 2673–2686. [Google Scholar] [CrossRef]
- Ruddaraju, L.K.; Pammi, S.V.N.; Guntuku, G.S.; Padavala, V.S.; Kolapalli, V.R.M. A review on anti-bacterials to combat resistance: From ancient era of plants and metals to present and future perspectives of green nano technological combinations. Asian J. Pharm. Sci. 2020, 15, 42–59. [Google Scholar] [CrossRef] [PubMed]
- Flores-Mireles, A.; Hreha, T.N.; Hunstad, D.A. Pathophysiology, Treatment, and Prevention of Catheter-Associated Urinary Tract Infection. Top. Spinal Cord Inj. Rehabil. 2019, 25, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Bandyk, D.F. The diabetic foot: Pathophysiology, evaluation, and treatment. Semin. Vasc. Surg. 2018, 31, 43–48. [Google Scholar] [CrossRef] [PubMed]







| Strain | Mechanism of Resistance | Type of Strain/Source |
|---|---|---|
| S. aureus Xen 30 | - MRSA, - constitutive MLSb | - Reference strain/purchased from Caliper Life Sciences (Hopkinton, MA) |
| S. epidermidis 175 | - MRSE, - constitutive MLSb | - Clinical strain/isolated from tracheobronchial secretions |
| K. pneumoniae ATCC 700603 | - ESBL caused by SHV-18 - impermeability to cephamycin as a mechanism associated with the alteration of porins | - Reference strain/purchased from American Type Culture Collection (ATCC, USA) |
| K. oxytoca 329 | - ESBL - acquired plasmid-mediated cephalosporinase AmpC | - Clinical strain/collected from the urine |
| P. aeruginosa LESB58 | - highly resistant to antibiotics with production of chromosomally encoded inducible AmpC-β-lactamase | - Hypervirulent and an epidemic cystic fibrosis strain isolated from sputum |
| P. aeruginosa 510 | - ESBL related to the active efflux pump - alteration of the outer membrane permeability resulting in OprD2 deficiency and finally resistance to carbapenems | - Clinical strain/collected from tracheobronchial secretions |
| Antibiotic/Chemotherapeutic | Staphylococcus aureus Xen 30 | Staphylococcus epidermidis 175 |
|---|---|---|
| Interpretation | Interpretation | |
| Screening test with cefoxitin | Positive | Positive |
| Oxacillin | R | R |
| Erythromycin | R | R |
| Clindamycin | R | R |
| Amikacin | R | R |
| Gentamicin | R | R |
| Ciprofloxacin | R | R |
| Levofloxacin | R | R |
| Linezolid | S | S |
| Daptomycin | S | S |
| Teicoplanin | S | S |
| Vancomycin | S | S |
| Antibiotic/Chemotherapeutic | Klebsiella pneumoniae ATCC 700603 | Klebsiella oxytoca 329 |
|---|---|---|
| Interpretation | Interpretation | |
| Ampicillin | R | R |
| Amoxicillin/Clavulanic acid | I * | R |
| Piperacillin/Tazobactam | I * | R |
| Cefuroxime | R | R |
| Cefuroxime axetil | R | R |
| Cefotaxime | R | R |
| Ceftazidime | R | R |
| Ertapenem | S | S |
| Meropenem | S | S |
| Amikacin | S | S |
| Gentamicin | R | R |
| Ciprofloxacin | S | R |
| Norfloxacin | S | R |
| Antibiotic/Chemotherapeutic | Pseudomonas aeruginosa LESB58 | Pseudomonas aeruginosa 510 |
|---|---|---|
| Interpretation | Interpretation | |
| Piperacillin | R | R |
| Piperacillin/Tazobactam | R | R |
| Ticarcillin/Clavulanic acid | R | R |
| Ceftazidime | R | R |
| Cefepime | R | R |
| Imipenem | S | R |
| Meropenem | S | R |
| Amikacin | R | R |
| Gentamicin | R | R |
| Tobramycin | R | R |
| Ciprofloxacin | R | R |
| Levofloxacin | R | R |
| Colistin | S | S |
| Assay | Indicator Reagent | Final Bacterial Inoculum | Final Concentration of the CSA-131 | Test Conditions | Results Recording |
|---|---|---|---|---|---|
| ROS generation | 2′,7′-dichlorofluorescin diacetate (DCFH-DA, Sigma-Aldrich, USA) | OD600 = 0.1 in PBS | 1–10 µg/mL | 60 min incubation at 37 °C with 20 µM DCFH-DA in PBS (in 96-well black plates) | fluorescence emission at wavelengths of 488/535 nm |
| Outer membrane permeabilization * | 1-N-phenylnapthylamine (NPN, Sigma-Aldrich, USA) | OD600 = 0.1 in PBS | 1–10 µg/mL | 5 min incubation at 37 °C with 0.5 mM NPN | fluorescence intensity λex = 348 nm/λem = 408 nm |
| diSC(3) | 3,3′-dipropylthiadicarbocyanine iodide (diSC(3), Sigma Aldrich, USA) | OD600–0.05 | 1–10 µg/mL | 60 min incubation at room temperature with 0.4 µM diSC(3), followed by 5 min incubation with 100 mM KCl | fluorescence emission at wavelengths of 622/670 nm |
| SYTO9/PI-dual staining ** | LIVE/DEAD BacLight Bacterial Viability Kit (CA, USA) | OD600–0.5 | 5 µg/mL and 10 µg/mL | 60 min incubation at 37 °C in PBS, followed by staining using SYTO9 dye and propidium iodide (PI) for 15 min | fluorescence microscopy (Zeiss AxioObserver.A1 Fluorescence Version Inverted Optical Microscope, JPK Instruments, German). |
| Protein leakage | Coomassie Brilliant Blue G-250 (Bradford reagent, Sigma-Aldrich, USA) | OD600–0.1 | 1–10 µg/mL | 60 min incubation at 37 °C, followed by 10 min centrifugation (5000 rpm at 4 °C) and incubation of supernatant with Bradford reagent (1:1 ratio) for 10 min in dark | absorbance level at 595 nm |
| Ceragenins/Nanosystems/ Microorganisms | S. aureus Xen 30 MIC/MBC (µg/mL) | S. epidermidis 175 MIC/MBC (µg/mL) | K. pneumoniae ATCC 700603 MIC/MBC (µg/mL) | K. oxytoca 329 MIC/MBC (µg/mL) | P. aeruginosa LESB58 MIC/MBC (µg/mL) | P. aeruginosa 510 MIC/MBC (µg/mL) |
|---|---|---|---|---|---|---|
| CSA-131 | 0.5/1 | 0.5/1 | 2/2 | 1/4 | 4/8 | 2/8 |
| AuR NPs@CSA-131 | 0.2/0.4 | 0.2/0.2 | 0.8/1.6 | 0.8/3.2 | 0.8/1.6 | 1.6/3.2 |
| AuP NPs@CSA-131 | 0.4/1.6 | 0.4/0.8 | 0.8/3.2 | 0.8/6.4 | 0.8/0.8 | 1.6/3.2 |
| AuS NPs@CSA-131 | 0.2/0.8 | 0.2/0.4 | 0.8/1.6 | 0.4/0.8 | 1.6/1.6 | 1.6/3.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chmielewska, S.J.; Skłodowski, K.; Depciuch, J.; Deptuła, P.; Piktel, E.; Fiedoruk, K.; Kot, P.; Paprocka, P.; Fortunka, K.; Wollny, T.; et al. Bactericidal Properties of Rod-, Peanut-, and Star-Shaped Gold Nanoparticles Coated with Ceragenin CSA-131 against Multidrug-Resistant Bacterial Strains. Pharmaceutics 2021, 13, 425. https://doi.org/10.3390/pharmaceutics13030425
Chmielewska SJ, Skłodowski K, Depciuch J, Deptuła P, Piktel E, Fiedoruk K, Kot P, Paprocka P, Fortunka K, Wollny T, et al. Bactericidal Properties of Rod-, Peanut-, and Star-Shaped Gold Nanoparticles Coated with Ceragenin CSA-131 against Multidrug-Resistant Bacterial Strains. Pharmaceutics. 2021; 13(3):425. https://doi.org/10.3390/pharmaceutics13030425
Chicago/Turabian StyleChmielewska, Sylwia Joanna, Karol Skłodowski, Joanna Depciuch, Piotr Deptuła, Ewelina Piktel, Krzysztof Fiedoruk, Patrycja Kot, Paulina Paprocka, Kamila Fortunka, Tomasz Wollny, and et al. 2021. "Bactericidal Properties of Rod-, Peanut-, and Star-Shaped Gold Nanoparticles Coated with Ceragenin CSA-131 against Multidrug-Resistant Bacterial Strains" Pharmaceutics 13, no. 3: 425. https://doi.org/10.3390/pharmaceutics13030425
APA StyleChmielewska, S. J., Skłodowski, K., Depciuch, J., Deptuła, P., Piktel, E., Fiedoruk, K., Kot, P., Paprocka, P., Fortunka, K., Wollny, T., Wolak, P., Parlinska-Wojtan, M., Savage, P. B., & Bucki, R. (2021). Bactericidal Properties of Rod-, Peanut-, and Star-Shaped Gold Nanoparticles Coated with Ceragenin CSA-131 against Multidrug-Resistant Bacterial Strains. Pharmaceutics, 13(3), 425. https://doi.org/10.3390/pharmaceutics13030425









